Abstract
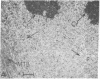
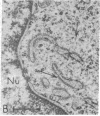
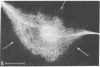
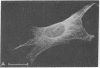
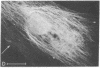
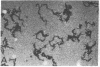
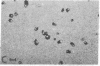
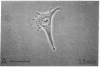
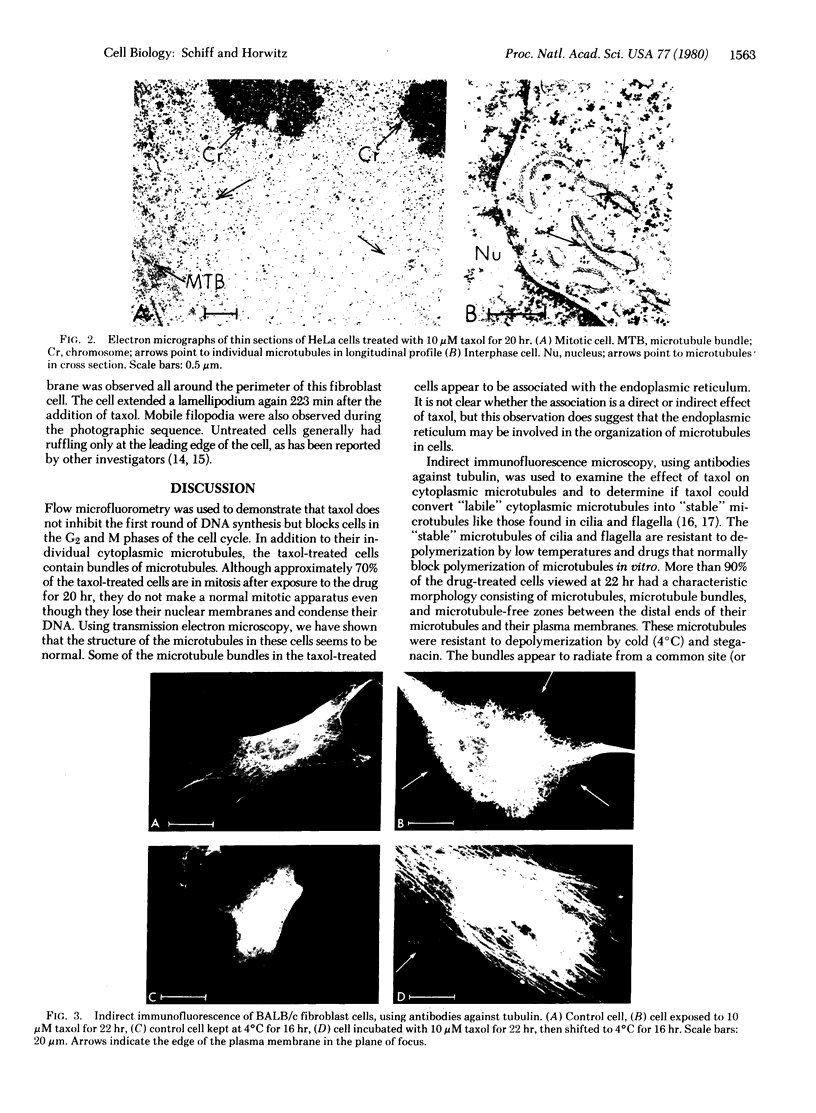
Taxol, a potent inhibitor of human HeLa and mouse fibroblast cell replication, blocked cells in the G2 and M phase of the cell cycle and stabilized cytoplasmic microtubules. The cytoplasmic microtubules of taxol-treated cells were visualized by transmission electron microscopy and indirect immunofluorescence microscopy. More than 90% of the cells treated with 10 micro M taxol for 22 hr at 37 degrees C displayed bundles of microtubules that appeared to radiate from a common site (or sites), in addition to their cytoplasmic microtubules. Untreated cells that were kept in the cold (4 degrees C) for 16 hr lost their microtubules, whereas cells that were pretreated with taxol for 22 hr at 37 degrees C continued to display their microtubules and bundles of microtubules in the cold. Taxol inhibited the migration behavior of fibroblast cells, but these cells did not lose their ability to produce mobile surface projections such as lamellipodia and filopodia.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abercrombie M., Heaysman J. E., Pegrum S. M. The locomotion of fibroblasts in culture. I. Movements of the leading edge. Exp Cell Res. 1970 Mar;59(3):393–398. doi: 10.1016/0014-4827(70)90646-4. [DOI] [PubMed] [Google Scholar]
- Abercrombie M., Heaysman J. E., Pegrum S. M. The locomotion of fibroblasts in culture. II. "RRuffling". Exp Cell Res. 1970 Jun;60(3):437–444. doi: 10.1016/0014-4827(70)90537-9. [DOI] [PubMed] [Google Scholar]
- Albrecht-Buehler G. The phagokinetic tracks of 3T3 cells. Cell. 1977 Jun;11(2):395–404. doi: 10.1016/0092-8674(77)90057-5. [DOI] [PubMed] [Google Scholar]
- Bhisey A. N., Freed J. J. Ameboid movement induced in cultured macrophages by colchicine or vinblastine. Exp Cell Res. 1971 Feb;64(2):419–429. doi: 10.1016/0014-4827(71)90096-6. [DOI] [PubMed] [Google Scholar]
- Crissman H. A., Steinkamp J. A. Rapid, simultaneous measurement of DNA, protein, and cell volume in single cells from large mammalian cell populations. J Cell Biol. 1973 Dec;59(3):766–771. doi: 10.1083/jcb.59.3.766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gail M. H., Boone C. W. Effect of colcemid on fibroblast motility. Exp Cell Res. 1971 Mar;65(1):221–227. doi: 10.1016/s0014-4827(71)80070-8. [DOI] [PubMed] [Google Scholar]
- Inoué S., Sato H. Cell motility by labile association of molecules. The nature of mitotic spindle fibers and their role in chromosome movement. J Gen Physiol. 1967 Jul;50(6 Suppl):259–292. [PMC free article] [PubMed] [Google Scholar]
- Margolis R. L., Wilson L., Keifer B. I. Mitotic mechanism based on intrinsic microtubule behaviour. Nature. 1978 Mar 30;272(5652):450–452. doi: 10.1038/272450a0. [DOI] [PubMed] [Google Scholar]
- Osborn M., Weber K. Cytoplasmic microtubules in tissue culture cells appear to grow from an organizing structure towards the plasma membrane. Proc Natl Acad Sci U S A. 1976 Mar;73(3):867–871. doi: 10.1073/pnas.73.3.867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiff P. B., Fant J., Horwitz S. B. Promotion of microtubule assembly in vitro by taxol. Nature. 1979 Feb 22;277(5698):665–667. doi: 10.1038/277665a0. [DOI] [PubMed] [Google Scholar]
- Schiff P. B., Kende A. S., Horwitz S. B. Steganacin: an inhibitor of HeLa cell growth and microtubule assembly in vitro. Biochem Biophys Res Commun. 1978 Nov 29;85(2):737–746. doi: 10.1016/0006-291x(78)91223-8. [DOI] [PubMed] [Google Scholar]
- Spiegelman B. M., Lopata M. A., Kirschner M. W. Multiple sites for the initiation of microtubule assembly in mammalian cells. Cell. 1979 Feb;16(2):239–252. doi: 10.1016/0092-8674(79)90002-3. [DOI] [PubMed] [Google Scholar]
- Vasiliev J. M., Gelfand I. M., Domnina L. V., Ivanova O. Y., Komm S. G., Olshevskaja L. V. Effect of colcemid on the locomotory behaviour of fibroblasts. J Embryol Exp Morphol. 1970 Nov;24(3):625–640. [PubMed] [Google Scholar]
- Vasiliev J. M., Gelfand I. M. Mechanisms of morphogenesis in cell cultures. Int Rev Cytol. 1977;50:159–274. doi: 10.1016/s0074-7696(08)60099-6. [DOI] [PubMed] [Google Scholar]
- Wang R. W., Rebhum L. I., Kupchan S. M. Antimitotic and antitubulin activity of the tumor inhibitor steganacin. Cancer Res. 1977 Sep;37(9):3071–3079. [PubMed] [Google Scholar]
- Wani M. C., Taylor H. L., Wall M. E., Coggon P., McPhail A. T. Plant antitumor agents. VI. The isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc. 1971 May 5;93(9):2325–2327. doi: 10.1021/ja00738a045. [DOI] [PubMed] [Google Scholar]
- Weber K., Bibring T., Osborn M. Specific visualization of tubulin-containing structures in tissue culture cells by immunofluorescence. Cytoplasmic microtubules, vinblastine-induced paracrystals, and mitotic figures. Exp Cell Res. 1975 Oct 1;95(1):111–120. doi: 10.1016/0014-4827(75)90615-1. [DOI] [PubMed] [Google Scholar]
- Weber K., Pollack R., Bibring T. Antibody against tuberlin: the specific visualization of cytoplasmic microtubules in tissue culture cells. Proc Natl Acad Sci U S A. 1975 Feb;72(2):459–463. doi: 10.1073/pnas.72.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheatley D. N. Cilia in cell-cultured fibroblasts. 3. Relationship between mitotic activity and cilium frequency in mouse 3T6 fibroblasts. J Anat. 1971 Dec;110(Pt 3):367–382. [PMC free article] [PubMed] [Google Scholar]