Abstract
We investigated the correlation between in vitro susceptibility of CD3+ T lymphocytes to equine arteritis virus (EAV) infection and establishment of persistent infection among 14 stallions following natural infections. The data showed that carrier stallions with a CD3+ T lymphocyte susceptibility phenotype to in vitro EAV infection may be at higher risk of becoming carriers than those that lack this phenotype (P = 0.0002).
TEXT
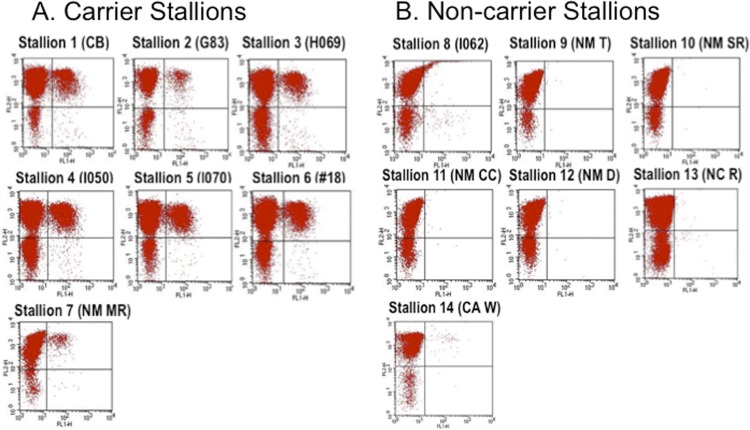
Equine arteritis virus (EAV) is a small, enveloped virus with a positive-sense, single-stranded RNA genome that belongs to the family Arteriviridae (genus Arterivirus, order Nidovirales) (4, 11). It is the causal agent of equine viral arteritis (EVA), which is a respiratory and reproductive disease of horses and other equids (3, 14). While the vast majority of natural infections with EAV are unapparent or subclinical (3, 2, 14), occasional outbreaks of EVA occur that are characterized by influenza-like signs in adult horses, abortion in mares, and the possibility of pneumonia in foals (7, 14). Following EAV infection, a variable proportion of stallions (30 to 70%) can become persistently infected and continuously shed the virus in their semen (14; S. M. Neu, P. J. Timoney, and W. H. McCollum, presented at the 5th International Conference Equine Infectious Diseases, Lexington, Kentucky, 1987). Carrier stallions are the natural reservoir of EAV; they ensure the virus is maintained in equine populations between breeding seasons (14, 16). The continued growth in the international trade of horses and semen has served as a significant means of dissemination of EAV strains around the world (1, 8, 13–16). Therefore, identification of carrier stallions is of critical epidemiological importance in the prevention and control of EAV infection in countries worldwide (2, 3, 12–14). While the mechanism of persistence of EAV in the male reproductive tract is not clear, it has been established that viral persistence in the stallion is testosterone dependent (9; T. V. Little, G. R. Holyoak, W. H. McCollum, and P. J. Timoney, presented at the 6th International Conference on Equine Infectious Diseases, Cambridge, England, 1992). Although multiple factors may contribute to development of the carrier state, host genetic variation is assumed to explain individual differences in occurrence of the carrier state as well as virus clearance, because such host variations exist even after exposure to the same virus. Moreover, the prevalence of EAV infection varies markedly among different breeds of horses, thus strengthening the assumption of genetic influence (14). Recently, we demonstrated differences in in vitro susceptibility of CD3+ T lymphocytes to EAV infection that have a strong association of genetic markers with de facto proof that the trait is under genetic control by conducting a genome-wide association study (GWAS) (5). The objective of the present study was to investigate whether there is a possible correlation between susceptibility of CD3+ T lymphocytes to EAV infection and establishment of long-term (>1 year) persistent infection in stallions. We were fortunate in identifying stallions that became EAV carriers after natural exposure as well as stallions that did not become carriers after exposure to EAV. To confirm long-term carrier status, isolation of EAV from equine semen samples was attempted in a high-passage-number (399 to 409 passages) rabbit kidney 13 cell line according to a previously described protocol (10). Subsequently, peripheral blood mononuclear cells (PBMCs) were collected from long-term carrier (n = 7) and noncarrier (n = 7) stallions (Table 1). The susceptible or resistant phenotype of each animal was defined by dual-color flow cytometric analysis of in vitro EAV-infected CD3+ T lymphocytes as described previously (6). Interestingly, CD3+ T lymphocytes from all EAV carrier stallions were susceptible to in vitro EAV infection (Fig. 1A), while those from stallions that did not become carriers were resistant to infection (Fig. 1B). While the number of stallions used in this study seems small, the results are highly significant based on a two-by-two chi-square test (P = 0.0002). The percentage of the CD3+ T lymphocyte population that was susceptible to EAV infection ranged from 4% to 17% among carrier stallions. As previously described, a majority of the CD3+ T lymphocytes infected with EAV are CD4+ rather than CD8+ T cells (6; data not shown). The data suggested that carrier stallions that have the susceptible CD3+ T lymphocyte phenotype in response to EAV infection could be at higher risk of becoming persistently infected than stallions that lack this phenotype. However, the exact role of the CD3+ T cell subpopulation in establishment of the long-term carrier state needs to be further investigated. We hypothesize that this CD3+ T cell subpopulation may represent regulatory T lymphocytes that may play a role in modulating the immune response, or they may be involved in transporting the virus to the site of persistent infection in the male reproductive tract. Alternatively, the persistent carrier status may simply be a consequence of expression of a molecule which acts as a viral receptor on cells of the reproductive tract of the stallion and which can allow the establishment of persistent infection; the expression of that same gene on CD3+ T cells may simply be a coincidence unrelated to the persistent carrier state. Studies are under way to identify the CD3+ T cell subpopulation and the EAV cellular receptor in our laboratory.
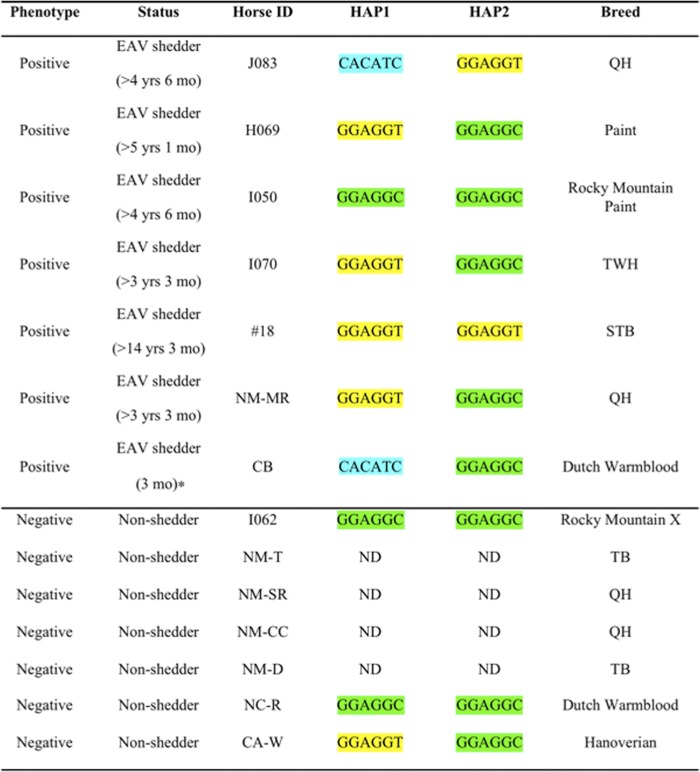
Table 1.
Haplotype of carrier and noncarrier stallions for selected SNPs in ECA11 49572804–49643932a
ND, not determined; QH, Quarter horse; STB, Standardbred; TB, Thoroughbred; Rocky Mountain X, Rocky Mountain cross. *, Not monitored regularly beyond 3 months for shedding of the virus. The positive and negative phenotypes refer to in vitro CD3+ T lymphocyte susceptibility and resistance, respectively. The haplotype GGAGGT, which is associated with CD3+ T cell susceptibility, is highlighted in yellow, as described elsewhere (6). Each haplotype is highlighted with a different color.
Fig 1.
Dual-color immunofluorescence flow cytometry analysis of carrier and seropositive noncarrier stallions. (A) Carrier status was confirmed by isolating EAV from semen. (B) Noncarrier stallions are seropositive horses with antibodies specific to EAV after natural exposure to EAV and not due to vaccination. PBMCs that were double stained with anti-CD3 and anti-EAV Nsp1 (12A4) MAbs are shown in the upper right quadrant of each dot plot (5).
Previously, we identified a dominant haplotype associated with the in vitro susceptible CD3+ T cell phenotype in a region of equine chromosome 11 (ECA11; 49572804 to 49643932) by testing four horse breeds (Thoroughbred, American Saddlebred, Standardbred, and Quarter Horses) (5). The presence of the common haplotype suggested that the trait is descended from a common ancestor for all four breeds. To evaluate whether the same haplotype is associated with carrier and noncarrier status, the ECA11 49572804-49643932 region of stallions included in the study was genotyped using a Sequenom Massarray system (Sequenom Inc., San Diego, CA). Briefly, genomic DNA was isolated from PBMCs of each animal using a Puregene whole-blood extraction kit (Qiagen, Valencia, CA) following the manufacturer's instructions. The distribution of single nucleotide polymorphisms (SNPs) was compared between carrier and noncarrier stallions as previously described (5). The SNP data (Table 1) showed that five of seven carrier stallions with the CD3+ T lymphocyte susceptible phenotype had the GGAGGT haplotype, which was previously reported to be closely associated with the trait in other breeds (5). While the number of animals was small, clearly the persistent carrier phenotype was not completely associated with genetic markers previously associated with the in vitro CD3+ T cell phenotype. The horses in this study were of diverse breeds, and the most likely explanation for these results is that the haplotype associated with the trait among Thoroughbred, Standardbred, Saddlebred, and Quarter Horses is not the same as that found in these other breeds (Table 1). This observation highlights the need to identify the actual gene(s) and mutation(s) responsible for the CD3+ T cell phenotype.
Based on previous studies, the CD3+ T cell phenotype was shown to have a genetic basis (5). The data presented in this study demonstrate that there is also a genetic difference between stallions that become long-term carriers after natural exposure to EAV and those that are able to clear the virus (stallions that do not become carriers) (Fig. 2). However, it is a challenge to find a large number of natural carrier and noncarrier stallions for these studies, especially the noncarrier stallions that have seroconverted following natural exposure to the virus. As a result, the horses in this study belonged to different breeds with different genetic backgrounds. Regardless of their diverse origins, all carriers possessed the in vitro susceptible CD3+ T cell phenotype while the noncarriers did not. Ultimately, results from this study need to be confirmed in an in vivo study with experimental EAV infection of stallions with susceptible and resistant CD3+ T lymphocyte EAV phenotypes to see whether stallions with the susceptible CD3+ T lymphocyte phenotype are at higher risk of establishing long-term carrier status than those with the resistant phenotype.
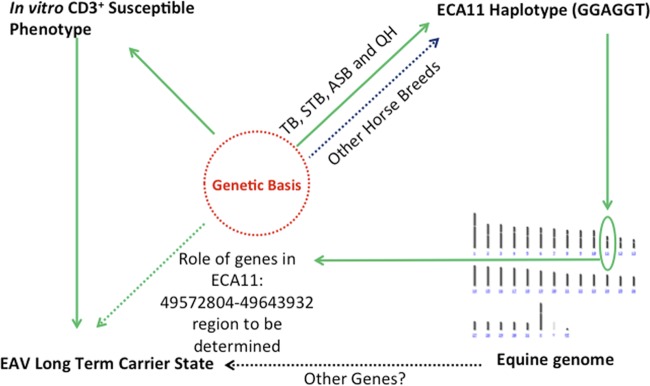
Fig 2.
Model for a hypothesis regarding the genetic cause of the persistent carrier state in stallions. This study shows an association of the CD3+ T cell susceptibility phenotype with the occurrence of the carrier state. The CD3+ T cell susceptibility phenotype was previously shown to be genetically determined (5). This study shows that the genetic definition of the susceptibility phenotype is imperfect based on the haplotype association. The genetic basis for this phenotype should be identified through additional genomic studies of this region.
In summary, predisposition for establishment of the carrier state following natural infections could be assessed by determining the susceptibility of their CD3+ T lymphocytes to in vitro infection with the virulent Bucyrus strain of EAV. However, this assay is expensive, time-consuming, and cumbersome to run on a routine or large-scale basis. This study also demonstrated that the ECA11 haplotype described in the previous work (5) did not show a complete association for these horses, possibly because of the diverse breeds and genetic distances between them. Therefore, the precise genetic basis for this virus infectivity trait should be determined to more effectively pursue investigations of the naturally occurring and experimentally induced carrier state in stallions (Fig. 2). Eventually, findings from this and subsequent studies will allow us to develop diagnostic tools to predict possible carrier stallions of EAV and limit dissemination of the virus.
ACKNOWLEDGMENTS
We thank Robert B. Perce, Bosque, New Mexico who kindly provided samples for the study.
This work was supported by the Morris Animal Foundation, Denver, CO, the Maxwell H. Gluck Equine Research Center intramural research grant program, and the Frederick Van Lennep Chair endowment fund at the Maxwell H. Gluck Equine Research Center, University of Kentucky. Yun Young Go was supported by a Geoffrey C. Hughes Foundation graduate fellowship.
Footnotes
Published ahead of print 29 August 2012
REFERENCES
- 1. Balasuriya UB, et al. 1998. Serologic and molecular characterization of an abortigenic strain of equine arteritis virus isolated from infective frozen semen and an aborted equine fetus. J. Am. Vet. Med. Assoc. 213:1586–1589 [PubMed] [Google Scholar]
- 2. Balasuriya UB, MacLachlan NJ. 2007. Equine viral arteritis, p 153–164 In Sellon DC, Long MT. (ed), Equine infectious diseases. Saunders Elsevier, St. Louis, MO [Google Scholar]
- 3. Balasuriya UB, MacLachlan NJ. 2004. The immune response to equine arteritis virus: potential lessons for other arteriviruses. Vet. Immunol. Immunopathol. 102:107–129 [DOI] [PubMed] [Google Scholar]
- 4. Cavanagh D. 1997. Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch. Virol. 142:629–633 [PubMed] [Google Scholar]
- 5. Go YY, et al. 2011. Genome-wide association study among four horse breeds identifies a common haplotype associated with the in vitro CD3+ T cell susceptibility/resistance to equine arteritis virus infection. J. Virol. 85:13174–13184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Go YY, et al. 2010. Complex interactions between the major and minor envelope proteins of equine arteritis virus determine its tropism for equine CD3+ T lymphocytes and CD14+ monocytes. J. Virol. 84:4898–4911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Holyoak GR, Balasuriya UB, Broaddus CC, Timoney PJ. 2008. Equine viral arteritis: current status and prevention. Theriogenology 70:403–414 [DOI] [PubMed] [Google Scholar]
- 8. Hullinger PJ, Gardner IA, Hietala SK, Ferraro GL, MacLachlan NJ. 2001. Seroprevalence of antibodies against equine arteritis virus in horses residing in the United States and imported horses. J. Am. Vet. Med. Assoc. 219:946–949 [DOI] [PubMed] [Google Scholar]
- 9. McCollum WH, Little TV, Timoney PJ, Swerczek TW. 1994. Resistance of castrated male horses to attempted establishment of the carrier state with equine arteritis virus. J. Comp. Pathol. 111:383–388 [DOI] [PubMed] [Google Scholar]
- 10. OIE 2008. OIE manual of diagnostic tests and vaccines for terrestrial animals, 6th ed., vol 2 Office Internationale des Epizooties, Paris, France [Google Scholar]
- 11. Snijder EJ, Meulenberg JJ. 1998. The molecular biology of arteriviruses. J. Gen. Virol. 79:961–979 [DOI] [PubMed] [Google Scholar]
- 12. Timoney PJ. 2000. Factors influencing the international spread of equine diseases. Vet. Clin. North Am. Equine Pract. 16:537–551 [DOI] [PubMed] [Google Scholar]
- 13. Timoney PJ. 2000. The increasing significance of international trade in equids and its influence on the spread of infectious diseases. Ann. N. Y. Acad. Sci. 916:55–60 [DOI] [PubMed] [Google Scholar]
- 14. Timoney PJ, McCollum WH. 1993. Equine viral arteritis. Vet. Clin. North Am. 9:295–309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Timoney PJ, et al. 1987. The carrier state in equine arteritis virus infection in the stallion with specific emphasis on the venereal mode of virus transmission. J. Reprod. Fertil Suppl. 35:95–102 [PubMed] [Google Scholar]
- 16. Timoney PJ, McCollum WH, Roberts AW, Murphy TW. 1986. Demonstration of the carrier state in naturally acquired equine arteritis virus infection in the stallion. Res. Vet. Sci. 41:279–280 [PubMed] [Google Scholar]