Abstract
Background
This study evaluated potential correlations between the allele burden of the Janus kinase 2 (JAK2) V617F mutation and clinicohematologic characteristics in patients with myeloproliferative neoplasms (MPN).
Methods
Clinical and hematologic features were reviewed for 103 MPN patients, including patients with polycythemia vera (PV, 22 patients), essential thrombocythemia (ET, 64 patients), and primary myelofibrosis (PMF, 17 patients). JAK2 V617F allele status and allele burdens were measured by allele-specific PCR and pyrosequencing, respectively.
Results
The JAK2 V617F mutation was detected in 95.5%, 68.8%, and 52.9% of PV, ET, and PMF patients, respectively. JAK2 V617F-positive ET patients were significantly older and exhibited higher neutrophil fractions, a higher frequency of thrombotic events, and a higher myelofibrosis rate than JAK2 V617F-negative patients (P <0.05). PV patients carried the highest mean T allele burden (66.0%±24.9%) compared with ET (40.5%±25.2%) and PMF patients (31.5%±37.0%) (P =0.00). No significant correlations were detected between V617F allele burden and patient age, white blood cell count, Hb, Hct, or the platelet count for PV, ET, or PMF patients. ET patients with organomegaly had a higher JAK2 V617F allele burden (53.4%±23.7%) than patients without organomegaly (35.6%±24.3%) (P =0.03).
Conclusions
The JAK2 V617F mutational status and its allele burden correlate with the clinicohematologic phenotypes of ET patients, including older age, higher neutrophil count, and greater rates of organomegaly, thrombotic events, and myelofibrosis. For PV and PMF patients, larger-scale studies involving more MPN patients are needed.
Keywords: Janus kinase 2, Allele, Myeloproliferative neoplasm
INTRODUCTION
The somatic mutation V617F in Janus kinase 2 (JAK2) is an established diagnostic marker in BCR-ABL1-negative myeloproliferative neoplasms (MPN), e.g., polycythemia vera (PV), essential thrombocythemia (ET), and primary myelofibrosis (PMF) [1-3]. However, correlations among the mutational status or allele burden of JAK2 V617F and clinicohematologic characteristics of these diseases remain undetermined. Several retrospective studies have evaluated correlations between the JAK2 V617F mutation and parameters such as age, white blood cell (WBC) count, Hb concentration, Hct, spleen size, disease duration, pruritus, and fibrosis in subjects with MPN; however, the results of these studies have been inconsistent [4-18]. The present study was undertaken to: (1) determine whether MPN patients harboring the JAK2 V617F mutation exhibit different clinicohematologic characteristics than patients lacking it; and (2) identify whether the allele burden of the JAK2 V617F mutation is associated with the severity of clinicohematologic features of MPN.
METHODS
1. Study population and sample collection
In this study, we used stored bone marrow (BM) cells of 103 patients who were diagnosed with MPN during 2005-2010. Clinicohematologic data obtained from patient records and BM histology were reviewed. Hematological diagnoses and subtyping results were reconfirmed according to the 2008 WHO criteria [1]. Patient data included the following major thrombotic events: ischemic stroke, transient ischemic attack, myocardial infarction, angina pectoris, and deep vein thrombosis. Karyotyping and reverse transcription-PCR (RT-PCR) for the BCR-ABL1 rearrangement confirmed that no patients harbored the BCR-ABL1 fusion gene. This study was approved by the Institutional Review Board of the Dongsan Medical Center.
2. Analysis of JAK2 V617F mutation and allele burden
Each patient's JAK2 V617F mutation status (n=103) was determined by allele-specific PCR, according to the manufacturer's instructions (Seegene, Seoul, Korea). For patients with sufficient stored BM samples (n=85), JAK2 V617F allele burdens were determined by pyrosequencing. Briefly, DNA was isolated using a QIAamp DNA mini kit (Qiagen, Hilden, Germany) and amplified using the following biotin-labeled primers: forward 5'-GAAGCAGCAAGTATGATGAGCA-3'; reverse 5'-TGCTCTGAGAAAGGCATTAGAA-3'. Single-stranded biotinylated templates were then isolated and sequenced using the sequencing primer 5'-TTACTTACTCTCGTCTCCAC-3'. All primers were designed using the Pyromark Assay Design 2.0 software (Qiagen). Percentages of JAK2 V617F mutant alleles were determined using the Allele Frequency Quantification function in PyroMark Q24 Software 2.0, according to the manufacturer's specifications (Qiagen).
3. Statistical analysis
Correlations between the mutational status of JAK2 V617F and WBC count, neutrophil fraction, Hb, Hct, or platelet count were analyzed using independent T-tests. Frequency differences of sex, organomegaly, thrombotic event, or myelofibrosis according to the JAK2 V617F mutational status were assessed using a Chi-square or Fisher's exact test. Correlations between clinicohematologic parameters and JAK2 V617F allele burden were examined using ANOVA or independent-T-tests. All analyses were conducted using SPSS v19.0 (SPSS Inc., Chicago, IL, USA). Statistical significance was assigned at P <0.05.
RESULTS
1. Clinicohematologic findings by MPN subtype
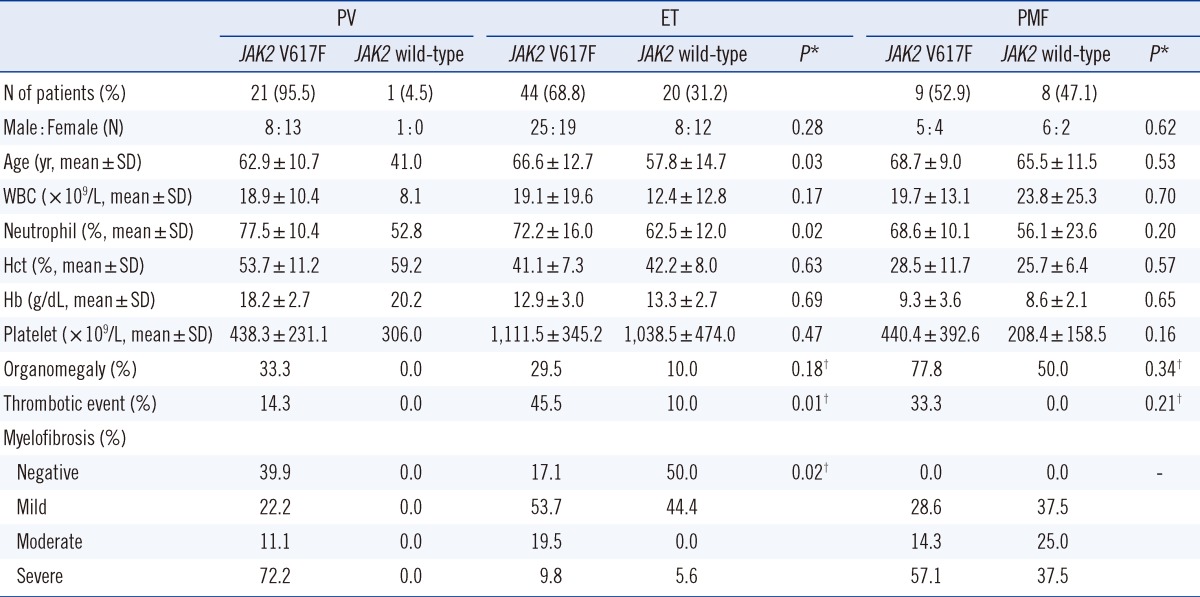
All 103 MPN patients (53 men, 50 women; mean age 64.0±12.8) were diagnosed with chronic-phase MPN with the following subtypes: 22 PV (mean age 62.2±11.6), 64 ET (mean age 63.8±13.8), and 17 PMF (mean age 63.8±13.8) (Table 1). The mean age of patients across MPN subgroups was not statistically different. Mean WBC counts and neutrophil fractions were statistically similar across MPN subgroups, except for a significantly higher neutrophil fraction in PV patients compared with PMF patients (P =0.01). Mean Hct and Hb concentrations significantly differed by disease subgroup, with PV patients exhibiting the highest values, followed by ET and PMF patients, in that order (P =0.00). Mean platelet counts were highest in ET patients (P =0.00) and did not differ significantly between PV and PMF patients. Organomegaly, including splenomegaly and hepatomegaly, was most frequent in PMF patients (64.7%) compared with ET (23.4%) or PV patients (31.8%). Thrombotic events occurred most frequently in ET patients (34.4%) and least frequently in PV patients (13.6%). Myelofibrosis was detected in all PMF patients, in 73.7% of PV patients, and in 72.9% of ET patients. Severe-grade myelofibrosis occurred most frequently among PMF patients (46.7%).
Table 1.
Clinicohematologic characteristics according to myeloproliferative neoplasm (MPN) subtypes
Abbreviations: PV, polycythemia vera; ET, essential thrombocythemia; PMF, primary myelofibrosis; WBC, white blood cell.
2. JAK2 V617F status and clinicohematologic findings
The JAK2 V617F mutation was detected in 74 of 103 patients (71.8%). Subtype-specific JAK2 V617F detection rates were 95.5% for PV patients, 68.8% for ET patients, and 52.9% for PMF patients (Table 2). When clinicohematologic data were compared between JAK2 V617F-positive and V617F-negative patients in the PV and PMF subgroups, no significant differences were detected. In contrast, JAK2 V617F-positive ET patients were significantly older and displayed higher neutrophil fractions, higher frequencies of thrombotic events, and higher myelofibrosis rates than JAK2 V617F-negative ET patients.
Table 2.
Clinicohematologic characteristics according to Janus kinase 2 (JAK2) mutation status
*P values describe differences between JAK2 V617F and JAK2 wild-type by independent T-test or †Chi-square test.
Abbreviations: PV, polycythemia vera; ET, essential thrombocythemia; PMF, primary myelofibrosis; WBC, white blood cell.
3. JAK2 V617F allele burden and clinicohematologic findings
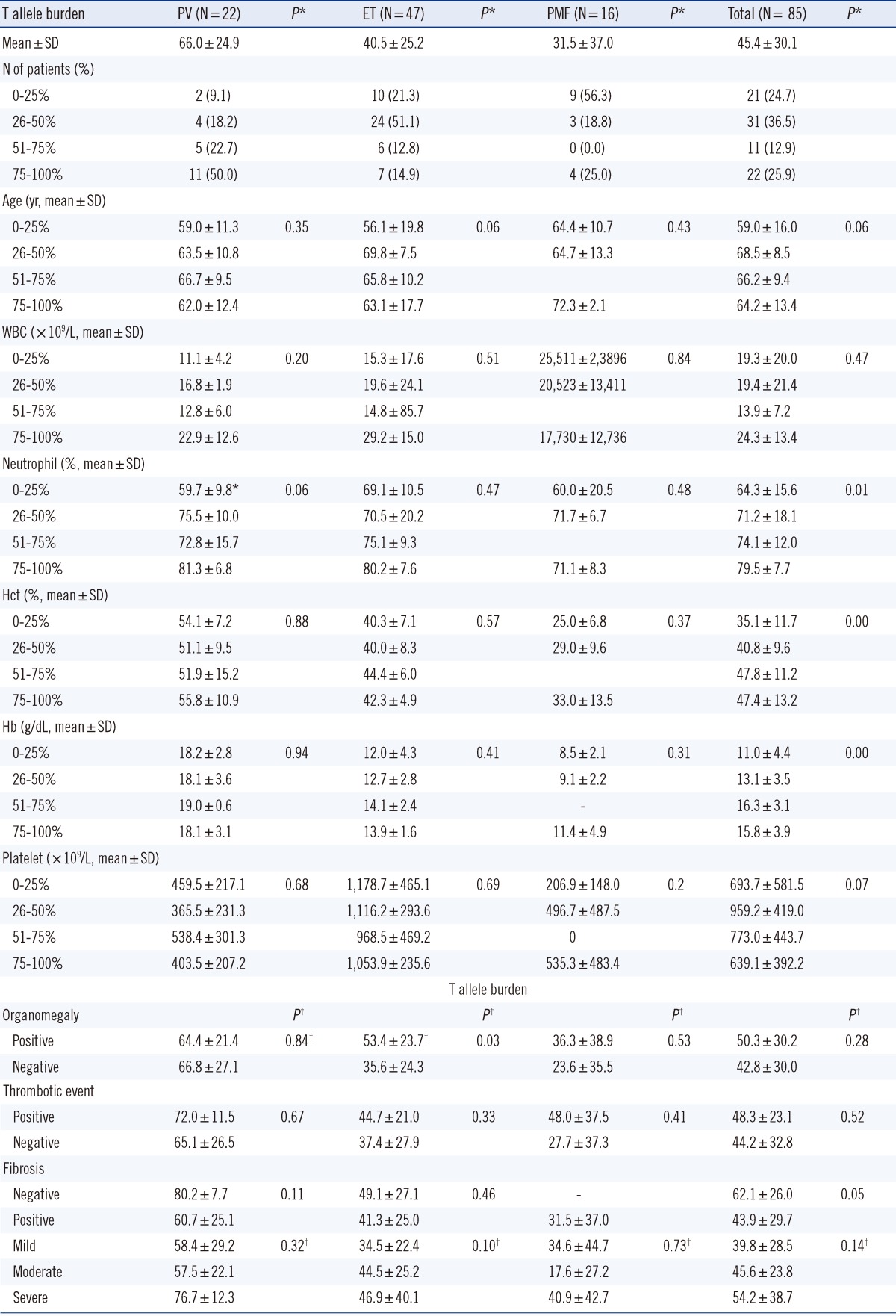
JAK2 V617F allele burdens could be measured for 85 of the 103 patients in the study population. This corresponded to 22 PV patients, 47 ET patients, and 16 PMF patients (Table 3). The PV subgroup carried the highest mean JAK2 V617F allele burden (66.0%±24.9%) compared with ET (40.5%±25.2%) and PMF patients (31.5%±37.0%) (P =0.00). To examine the relationship between clinicohematologic parameters and V617F allele burdens, PV, ET, and PMF patients were stratified by V617F allele burden as follows: 0-25%, 26-50%, 51-75%, or 76-100%. PV patients were most frequently classified into the 76-100% allele group (50.0%), whereas ET and PMF patients more often populated the 0-25% or 26-50% groups. No significant differences were detected when comparing mean age, WBC count, neutrophil fraction, Hb concentration, Hct, or platelet count among the 4 allele groups for each disease subgroup. However, many of these characteristics tended to increase in severity with incremental increases in the V617F allele burden in each disease subgroup. In contrast, platelet counts tended to be decreased with high V617F allele burdens. ET patients with organomegaly were significantly more likely to harbor high V617F allele burdens (53.4%±23.7%) than patients carrying wild-type JAK2 (35.6%±24.3%) (P =0.03). Organomegaly and V617F allele burden were uncorrelated among PV and PMF patient subgroups. Patients with a history of thrombotic events displayed a slight tendency toward carrying a higher T allele burden compared with patients who did not experience thrombotic events in each disease group. The V617F allele burdens of PV and ET patients with fibrosis tended to be lower than those of patients without fibrosis, but this difference was not significant. When V617F allele burdens were stratified by the extent of fibrosis (i.e., mild, moderate, or severe) in each disease subgroup, no significant associations were observed.
Table 3.
Associations of clinicohematologic parameters with Janus kinase 2 (JAK2) V617F allele burden
*P values describe differences of parameters among four groups according to V617F allele burden measured (by ANOVA); †P values describe differences of mean V617F allele burden between negative and positive groups for organomegaly, thrombotic event, and fibrosis (by independent T-test); ‡P values describe differences of V617F allele burdens among mild, moderate, and severe fibrosis groups (by ANOVA).
Abbreviations: PV, polycythemia vera; ET, essential thrombocythemia; PMF, primary myelofibrosis; WBC, white blood cell.
When patients were stratified into 2 groups on the basis of V617F allele burden, that is, <50% and ≥50%, no significant differences were detected with regard to most of the clinicohistological parameters, except older age of PMF patients in the ≥50% V617F allele burden group (P =0.03).
DISCUSSION
The JAK2 V617F acquired somatic mutation is the most commonly described mutation associated with MPN. Current estimates suggest that the JAK2 V617F mutation is detected in >95% of patients diagnosed with PV, approximately 60% of ET patients, and 50% of PMF patients [19-21]. In agreement with these reports, we detected the JAK2 V617F mutation in 95.5% of PV patients and 52.9% of PMF patients. However, ET patients in our study carried slightly higher V617F allele frequencies (68.8%) than previously reported. This might be explained by the smaller population examined in our study compared with previous study populations.
In numerous studies, the JAK2 V617F allele has been variably associated with higher indices of erythropoiesis, unchanged or decreased platelet counts, greater occurrence of thrombosis, increased BM fibrosis or cytoreductive treatments, older age, longer disease duration, or poorer survival in MPN [2-8]. Similarly, it was reported that the JAK2 V617F mutation in ET or PMF patients may skew their presenting phenotype toward a more erythremic and less thrombocythemic phenotype related to high erythropoietic and granulopoietic parameters and low platelet counts [4, 7]. Among ET patients in our study, we observed correlations between JAK2 V617F positivity and older age, higher neutrophil fraction, higher frequency of thrombotic events, and higher myelofibrosis rates; these results are consistent with previous reports. However, we did not observe low platelet counts in JAK2 V617F-positive patients. Instead, we observed a slight tendency in groups of high JAK2 V617F allele burden to exhibit lower platelet counts among ET patients.
Similar to the JAK2 V617F mutational status, previous studies have frequently correlated JAK2 V617F allele burdens with features of stimulated erythropoiesis (e.g., elevated Hct and decreased mean cell volume, serum ferritin level, and erythropoietin level), myelopoiesis (e.g., increased WBC count, neutrophil count, and serum lactate dehydrogenase level), neutrophil activation (e.g., elevated leukocyte count and alkaline phosphatase level), and high complication rate in MPN [9-18]. Compared to JAK2 V617F heterozygous patients, JAK2 V617F homozygous patients (>50% JAK2 V617F allele burden) with PV or ET exhibited significantly higher Hb levels, WBC counts, and Hct; older age; larger spleen volumes; higher incidence of pruritus; and/or more frequent progression to secondary myelofibrosis [11, 12]. In another study, patients with >75% JAK2 V617F allele burdens were at a higher relative risk of presenting with large spleens, pruritus, major cardiovascular events, or requiring chemotherapy compared with patients with a mutant allele burden <25%; thus, >75% JAK2 V617F allele burden at diagnosis was suggested as one of the high risk factors [10]. A separate study also correlated the JAK2 V617F allele burden with advanced myelofibrosis and greater splenomegaly [13].
The JAK2 V617F allele burden has been associated with disease duration in PV patients, such that the allele burden was significantly elevated in patients examined more than 5 years after their initial diagnosis [14]. To minimize this potential bias, we limited our analyses to samples collected at each patient's initial diagnosis. However, we detected an association only between high V617F allele burden and a higher frequency of organomegaly in ET patients, but not for other clinicohematologic parameters among PV or PMF patients. PV, ET, and PMF patients with thrombotic events tended to have higher allele burdens, but this finding was not statistically significant.
Interestingly, a low JAK2 V617F allele burden at diagnosis has been proposed as a surrogate marker for shortened survival in PMF patients [17]. In that study, the group with low V617F allele burden showed a significantly lower survival rate than the group with high V617F allele burden, but the JAK2 V617F mutational status did not affect the survival rate [17]. In contrast, another study demonstrated that the JAK2 V617F mutation was associated with poorer survival in PMF patients [18]. Although our short observation period precluded survival studies, previous findings support the likelihood of the JAK2 V617F allele burden as a prognostic marker in PMF patients.
Our study was limited by the small patient population, particularly for the PV and PMF patient subgroups, and by our method of JAK2 V617F allele detection. We identified JAK2 V617F from whole BM cells, whereas most previous studies have used peripheral blood granulocytes or CD34+ cells. Since JAK2 V617F is a myeloid lineage-specific mutation that is not found in lymphocytes, our analysis may have been influenced by a low detection rate [22, 23].
Although we could observe only a few associations of the JAK2 V617F allele with MPN phenotype parameters, our results support the hypothesis that the mutational status or allele burden of JAK2 V617F affects the severity of MPN-associated clinical and hematologic phenotypes with a larger allele burden, tending to produce a more severe phenotype (e.g., high myelopoiesis or high frequency of complications). Considering the limited patient number in the present study, a larger data set might improve the analysis of significance of JAK2 V617F alleles across clinical or phenotypic subgroups of MPN patients.
Acknowledgement
The research was supported by the Bisa Research Grant of Keimyung University in 2012. We thank KS Park, Ph.D. and WA Bae, Ph.D. for their assistance in sample collection, DNA extraction, and pyrosequencing analysis.
Footnotes
No potential conflicts of interest relevant to this article were reported.
References
- 1.Tefferi A, Vardiman JW. Classification and diagnosis of myeloproliferative neoplasms: the 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia. 2008;22:14–22. doi: 10.1038/sj.leu.2404955. [DOI] [PubMed] [Google Scholar]
- 2.Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR, et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med. 2005;352:1779–1790. doi: 10.1056/NEJMoa051113. [DOI] [PubMed] [Google Scholar]
- 3.Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S, et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet. 2005;365:1054–1061. doi: 10.1016/S0140-6736(05)71142-9. [DOI] [PubMed] [Google Scholar]
- 4.Campbell PJ, Scott LM, Buck G, Wheatley K, East CL, Marsden JT, et al. Definition of subtypes of essential thrombocythaemia and relation to polycythaemia vera based on JAK2 V617F mutation status: a prospective study. Lancet. 2005;366:1945–1953. doi: 10.1016/S0140-6736(05)67785-9. [DOI] [PubMed] [Google Scholar]
- 5.Wolanskyj AP, Lasho TL, Schwager SM, McClure RF, Wadleigh M, Lee SJ, et al. JAK2 mutation in essential thrombocythaemia: clinical associations and long-term prognostic relevance. Br J Haematol. 2005;131:208–213. doi: 10.1111/j.1365-2141.2005.05764.x. [DOI] [PubMed] [Google Scholar]
- 6.Barosi G, Bergamaschi G, Marchetti M, Vannucchi AM, Guglielmelli P, Antonioli E, et al. JAK2 V617F mutational status predicts progression to large splenomegaly and leukemic transformation in primary myelofibrosis. Blood. 2007;110:4030–4036. doi: 10.1182/blood-2007-07-099184. [DOI] [PubMed] [Google Scholar]
- 7.Rudzki Z, Sacha T, Stój A, Czekalska S, Wójcik M, Skotnicki AB, et al. The gain-of-function JAK2 V617F mutation shifts the phenotype of essential thrombocythemia and chronic idiopathic myelofibrosis to more "erythremic" and less "thrombocythemic": a molecular, histologic, and clinical study. Int J Hematol. 2007;86:130–136. doi: 10.1532/IJH97.E0607. [DOI] [PubMed] [Google Scholar]
- 8.De Stefano V, Za T, Rossi E, Vannucchi AM, Ruggeri M, Elli E, et al. Increased risk of recurrent thrombosis in patients with essential thrombocythemia carrying the homozygous JAK2 V617F mutation. Ann Hematol. 2010;89:141–146. doi: 10.1007/s00277-009-0788-5. [DOI] [PubMed] [Google Scholar]
- 9.Silver RT, Vandris K, Wang YL, Adriano F, Jones AV, Christos PJ, et al. JAK2(V617F) allele burden in polycythemia vera correlates with grade of myelofibrosis, but is not substantially affected by therapy. Leuk Res. 2011;35:177–182. doi: 10.1016/j.leukres.2010.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vannucchi AM, Antonioli E, Guglielmelli P, Longo G, Pancrazzi A, Ponziani V, et al. Prospective identification of high-risk polycythemia vera patients based on JAK2(V617F) allele burden. Leukemia. 2007;21:1952–1959. doi: 10.1038/sj.leu.2404854. [DOI] [PubMed] [Google Scholar]
- 11.Tefferi A, Lasho TL, Schwager SM, Strand JS, Elliott M, Mesa R, et al. The clinical phenotype of wild-type, heterozygous, and homozygous JAK2V617F in polycythemia vera. Cancer. 2006;106:631–635. doi: 10.1002/cncr.21645. [DOI] [PubMed] [Google Scholar]
- 12.Vannucchi AM, Antonioli E, Guglielmelli P, Rambaldi A, Barosi G, Marchioli R, et al. Clinical profile of homozygous JAK2 617V>F mutation in patients with polycythemia vera or essential thrombocythemia. Blood. 2007;110:840–846. doi: 10.1182/blood-2006-12-064287. [DOI] [PubMed] [Google Scholar]
- 13.Passamonti F, Rumi E, Pietra D, Elena C, Boveri E, Arcaini L, et al. A prospective study of 338 patients with polycythemia vera: the impact of JAK2 (V617F) allele burden and leukocytosis on fibrotic or leukemic disease transformation and vascular complications. Leukemia. 2010;24:1574–1579. doi: 10.1038/leu.2010.148. [DOI] [PubMed] [Google Scholar]
- 14.Tefferi A, Strand JJ, Lasho TL, Knudson RA, Finke CM, Gangat N, et al. Bone marrow JAK2V617F allele burden and clinical correlates in polycythemia vera. Leukemia. 2007;21:2074–2075. doi: 10.1038/sj.leu.2404724. [DOI] [PubMed] [Google Scholar]
- 15.Antonioli E, Guglielmelli P, Pancrazzi A, Bogani C, Verrucci M, Ponziani V, et al. Clinical implications of the JAK2 V617F mutation in essential thrombocythemia. Leukemia. 2005;19:1847–1849. doi: 10.1038/sj.leu.2403902. [DOI] [PubMed] [Google Scholar]
- 16.Antonioli E, Guglielmelli P, Poli G, Bogani C, Pancrazzi A, Longo G, et al. Influence of JAK2V617F allele burden on phenotype in essential thrombocythemia. Haematologica. 2008;93:41–48. doi: 10.3324/haematol.11653. [DOI] [PubMed] [Google Scholar]
- 17.Guglielmelli P, Barosi G, Specchia G, Rambaldi A, Lo Coco F, Antonioli E, et al. Identification of patients with poorer survival in primary myelofibrosis based on the burden of JAK2V617F mutated allele. Blood. 2009;114:1477–1483. doi: 10.1182/blood-2009-04-216044. [DOI] [PubMed] [Google Scholar]
- 18.Campbell PJ, Griesshammer M, Döhner K, Döhner H, Kusec R, Hasselbalch HC, et al. V617F mutation in JAK2 is associated with poorer survival in idiopathic myelofibrosis. Blood. 2006;107:2098–2100. doi: 10.1182/blood-2005-08-3395. [DOI] [PubMed] [Google Scholar]
- 19.Delhommeau F, Jeziorowska D, Marzac C, Casadevall N. Molecular aspects of myeloproliferative neoplasms. Int J Hematol. 2010;91:165–173. doi: 10.1007/s12185-010-0530-z. [DOI] [PubMed] [Google Scholar]
- 20.Passamonti F, Maffioli M, Caramazza D. New generation small-molecule inhibitors in myeloproliferative neoplasms. Curr Opin Hematol. 2012;19:117–123. doi: 10.1097/MOH.0b013e32834ff575. [DOI] [PubMed] [Google Scholar]
- 21.De Stefano V, Rossi E, Za T, Ciminello A, Betti S, Luzzi C, et al. JAK2 V617F mutational frequency in essential thrombocythemia associated with splanchnic or cerebral vein thrombosis. Am J Hematol. 2011;86:526–528. doi: 10.1002/ajh.22024. [DOI] [PubMed] [Google Scholar]
- 22.Tefferi A, Lasho TL, Schwager SM, Steensma DP, Mesa RA, Li CY, et al. The JAK2(V617F) tyrosine kinase mutation in myelofibrosis with myeloid metaplasia: lineage specificity and clinical correlates. Br J Haematol. 2005;131:320–328. doi: 10.1111/j.1365-2141.2005.05776.x. [DOI] [PubMed] [Google Scholar]
- 23.Lasho TL, Mesa R, Gilliland DG, Tefferi A. Mutation studies in CD3+, CD19+ and CD34+ cell fractions in myeloproliferative disorders with homozygous JAK2(V617F) in granulocytes. Br J Haematol. 2005;130:797–799. doi: 10.1111/j.1365-2141.2005.05682.x. [DOI] [PubMed] [Google Scholar]