Abstract
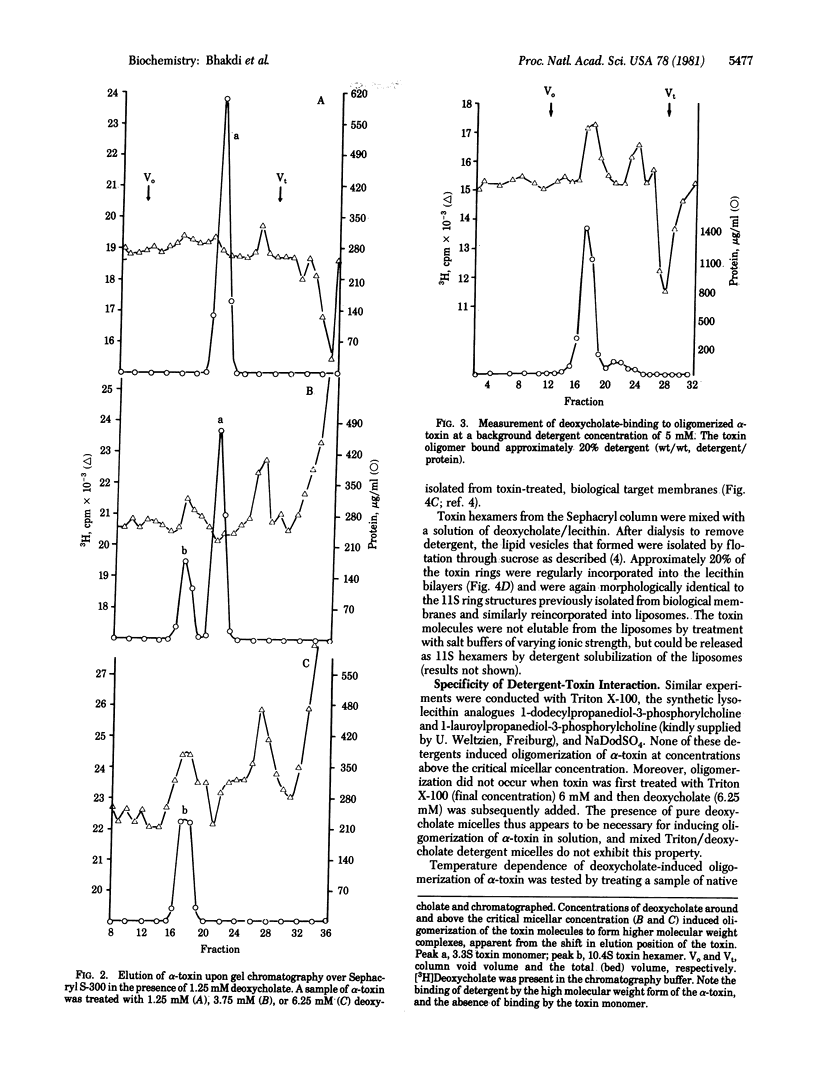
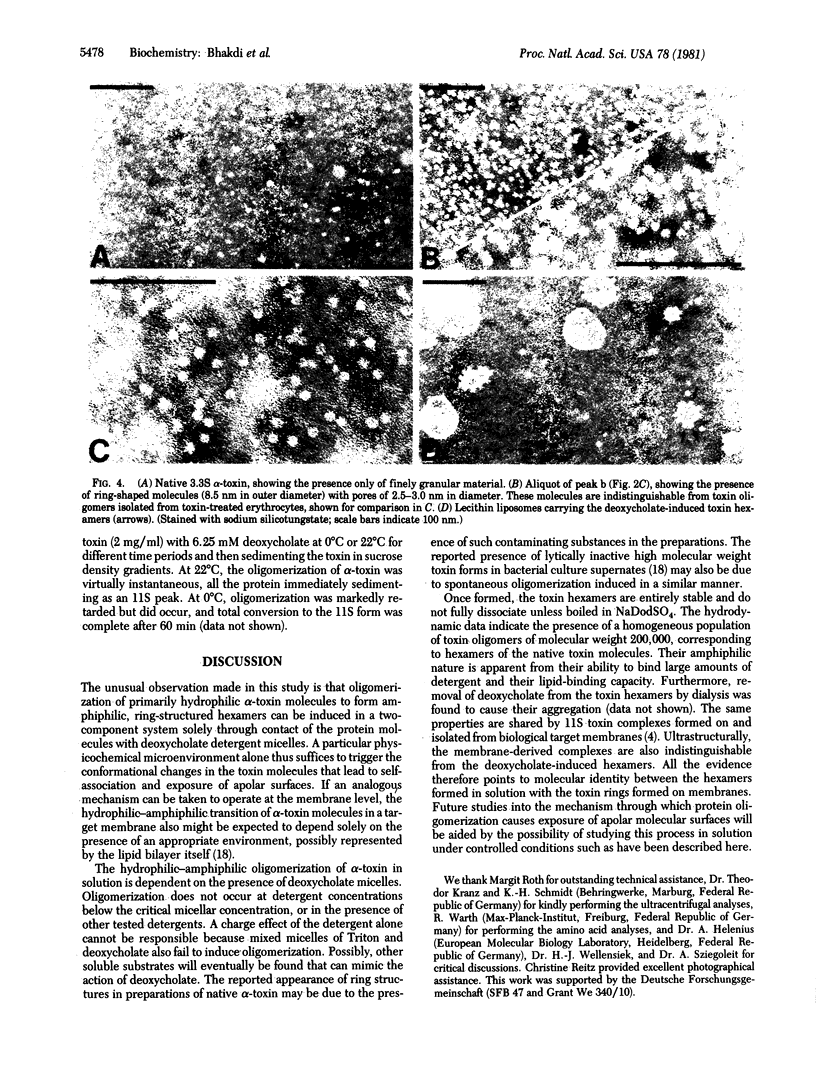
Native staphylococcus aureus alpha-toxin is secreted as a hydrophilic polypeptide chain of Mr 34,000. The presence of deoxycholate above the critical micellar concentration induced the toxin monomers to self-associate, forming ring or cylindrical oligomers. The oligomers were amphiphilic and bound detergent. In deoxycholate solution, the protein-detergent complexes exhibited a sedimentation coefficient of 10.4 S. A Mr of 238,700 was determined by ultracentrifugation analyses at sedimentation equilibrium. Because quantitative detergent-binding studies indicated a protein/detergent ratio of approximately 5:1 (wt/wt), the protein moiety in each protein-detergent complex was determined to be approximately Mr 200000, corresponding to a hexamer of the native molecule. The amphiphilic toxin hexamers were ultrastructurally indistinguishable from the cytolytic, annular toxin complexes that form on and in biological target membranes. They bound lipid and could be incorporated into artificial lecithin lipid vesicles. The transition of toxin protein molecules from a hydrophilic monomer to an amphiphilic oligomer through self-association has thus been shown to be inducible solely through contact of the native protein molecules with an appropriate amphiphilic substrate.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arbuthnott J. P., Freer J. H., Bernheimer A. W. Physical states of staphylococcal alpha-toxin. J Bacteriol. 1967 Oct;94(4):1170–1177. doi: 10.1128/jb.94.4.1170-1177.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbuthnott J. P., Freer J. H., Billcliffe B. Lipid-induced polymerization of staphylococcal -toxin. J Gen Microbiol. 1973 Apr;75(2):309–319. doi: 10.1099/00221287-75-2-309. [DOI] [PubMed] [Google Scholar]
- Bhakdi S., Tranum-Jensen J. Molecular nature of the complement lesion. Proc Natl Acad Sci U S A. 1978 Nov;75(11):5655–5659. doi: 10.1073/pnas.75.11.5655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassidy P., Harshman S. Characterization of detergent-solubilized iodine-125-labeled alpha-toxin bound to rabbit erythrocytes and mouse diaphragm muscle. Biochemistry. 1979 Jan 9;18(1):232–236. doi: 10.1021/bi00568a036. [DOI] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Freer J. H., Arbuthnott J. P., Bernheimer A. W. Interaction of staphylococcal alpha-toxin with artificial and natural membranes. J Bacteriol. 1968 Mar;95(3):1153–1168. doi: 10.1128/jb.95.3.1153-1168.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freer J. H., Arbuthnott J. P., Billcliffe B. Effects of staphylococcal -toxin on the structure of erythrocyte membranes: a biochemical and freeze-etching study. J Gen Microbiol. 1973 Apr;75(2):321–332. doi: 10.1099/00221287-75-2-321. [DOI] [PubMed] [Google Scholar]
- Helenius A., Simons K. Solubilization of membranes by detergents. Biochim Biophys Acta. 1975 Mar 25;415(1):29–79. doi: 10.1016/0304-4157(75)90016-7. [DOI] [PubMed] [Google Scholar]
- Helenius A., Simons K. The binding of detergents to lipophilic and hydrophilic proteins. J Biol Chem. 1972 Jun 10;247(11):3656–3661. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Mayer M. M. Mechanism of cytolysis by complement. Proc Natl Acad Sci U S A. 1972 Oct;69(10):2954–2958. doi: 10.1073/pnas.69.10.2954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogolsky M. Nonenteric toxins of Staphylococcus aureus. Microbiol Rev. 1979 Sep;43(3):320–360. doi: 10.1128/mr.43.3.320-360.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Six H. R., Harshman S. Physical and chemical studies on staphylococcal -toxins A and B . Biochemistry. 1973 Jul 3;12(14):2677–2683. doi: 10.1021/bi00738a020. [DOI] [PubMed] [Google Scholar]
- Six H. R., Harshman S. Purification and properties of two forms of staphylococcal toxin. Biochemistry. 1973 Jul 3;12(14):2672–2677. doi: 10.1021/bi00738a019. [DOI] [PubMed] [Google Scholar]
- Tanford C., Reynolds J. A. Characterization of membrane proteins in detergent solutions. Biochim Biophys Acta. 1976 Oct 26;457(2):133–170. doi: 10.1016/0304-4157(76)90009-5. [DOI] [PubMed] [Google Scholar]
- Tranum-Jensen J., Bhakdi S., Bhakdi-Lehnen B., Bjerrum O. J., Speth V. Complement lysis: the ultrastructure and orientation of the C5b-9 complex on target sheep erythrocyte membranes. Scand J Immunol. 1978;7(1):45–46. doi: 10.1111/j.1365-3083.1978.tb00425.x. [DOI] [PubMed] [Google Scholar]