Abstract
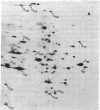
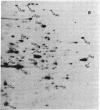
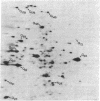
The synthesis of cytoplasmic proteins from quiescent and serum-stimulated Swiss 3T3 cells was compared by two-dimensional polyacrylamide gel electrophoresis. Four new proteins of Mrs 26,000, 28,000, 45,000, and 47,000 designated N26, N28, N45, and N47, which were not detectable in quiescent cells, appeared 60 min after addition of serum. During the same period, the amount of [35S]methionine incorporated into 10 proteins present in quiescent cells, ranging in Mr from 23,000 to 98,000 and designated Q23-98, increased up to 6-fold, whereas the amount incorporated into three other proteins decreased by a factor of approximately 2. Of the new proteins, N26 was no longer detectable, and the amount of [35S]methionine incorporated into N47 was significantly reduced by 150 min. During this same time, a fifth new protein, N56, appeared, and there was a large increase in the amount of radioactivity incorporated into another protein, Q121. The increases in nine of the proteins were either strongly or completely inhibited by actinomycin D, arguing that the expression of these proteins was under transcriptional control. In contrast, the increases in seven other proteins were unaffected by actinomycin D, suggesting that their expression was under translational control. These proteins will serve as useful markers for determining how cells progress through early lag phase.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alton T. H., Lodish H. F. Translational control of protein synthesis during the early stages of differentiation of the slime mold Dictyostelium discoideum. Cell. 1977 Sep;12(1):301–310. doi: 10.1016/0092-8674(77)90208-2. [DOI] [PubMed] [Google Scholar]
- Bandman E., Gurney T., Jr Differences in the cytoplasmic distribution of newly synthesized poly (A) in serum-stimulated and resting cultures of BALB/c 3T3 cells. Exp Cell Res. 1975 Jan;90(1):159–168. doi: 10.1016/0014-4827(75)90369-9. [DOI] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Bravo R., Bellatin J., Celis J. E. [35S]-methionine labelled polypeptides from HELA cells. Coordinates and percentage of some major polypeptides. Cell Biol Int Rep. 1981 Jan;5(1):93–96. doi: 10.1016/0309-1651(81)90162-4. [DOI] [PubMed] [Google Scholar]
- Brooks R. F. Continuous protein synthesis is required to maintain the probability of entry into S phase. Cell. 1977 Sep;12(1):311–317. doi: 10.1016/0092-8674(77)90209-4. [DOI] [PubMed] [Google Scholar]
- Croall D. E., Morrison M. R. Polysomal and non-polysomal messenger RNA in neuroblastoma cells. Lack of correlation between polyadenylation or initiation efficiency and messenger RNA location. J Mol Biol. 1980 Jul 15;140(4):549–564. doi: 10.1016/0022-2836(80)90270-3. [DOI] [PubMed] [Google Scholar]
- Fox T. O., Pardee A. B. Proteins made in the mammalian cell cycle. J Biol Chem. 1971 Oct 25;246(20):6159–6165. [PubMed] [Google Scholar]
- Garrels J. I. Two dimensional gel electrophoresis and computer analysis of proteins synthesized by clonal cell lines. J Biol Chem. 1979 Aug 25;254(16):7961–7977. [PubMed] [Google Scholar]
- Gates B. J., Friedkin M. Mid-G1 marker protein(s) in 3T3 mouse fibroblast cells. Proc Natl Acad Sci U S A. 1978 Oct;75(10):4959–4961. doi: 10.1073/pnas.75.10.4959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershko A., Mamont P., Shields R., Tomkins G. M. "Pleiotypic response". Nat New Biol. 1971 Aug;232(33):206–211. [PubMed] [Google Scholar]
- Holley R. W. Control of growth of mammalian cells in cell culture. Nature. 1975 Dec 11;258(5535):487–490. doi: 10.1038/258487a0. [DOI] [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- Lee G. T., Engelhardt D. L. Growth-related fluctuation in messenger RNA utilization in animal cells. J Cell Biol. 1978 Oct;79(1):85–86. doi: 10.1083/jcb.79.1.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee G. T., Engelhardt D. L. Peptide coding capacity of polysomal and non-polysomal messenger RNA during growth of animal cells. J Mol Biol. 1979 Apr 5;129(2):221–233. doi: 10.1016/0022-2836(79)90278-x. [DOI] [PubMed] [Google Scholar]
- Lodish H. F. Translational control of protein synthesis. Annu Rev Biochem. 1976;45:39–72. doi: 10.1146/annurev.bi.45.070176.000351. [DOI] [PubMed] [Google Scholar]
- O'Farrell P. Z., Goodman H. M., O'Farrell P. H. High resolution two-dimensional electrophoresis of basic as well as acidic proteins. Cell. 1977 Dec;12(4):1133–1141. doi: 10.1016/0092-8674(77)90176-3. [DOI] [PubMed] [Google Scholar]
- Pelham H. R., Jackson R. J. An efficient mRNA-dependent translation system from reticulocyte lysates. Eur J Biochem. 1976 Aug 1;67(1):247–256. doi: 10.1111/j.1432-1033.1976.tb10656.x. [DOI] [PubMed] [Google Scholar]
- Peterson J. L., McConkey E. H. Non-histone chromosomal proteins from HeLa cells. A survey by high resolution, two-dimensional electrophoresis. J Biol Chem. 1976 Jan 25;251(2):548–554. [PubMed] [Google Scholar]
- Riddle V. G., Dubrow R., Pardee A. B. Changes in the synthesis of actin and other cell proteins after stimulation of serum-arrested cells. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1298–1302. doi: 10.1073/pnas.76.3.1298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudland P. S. Control of translation in cultured cells: continued synthesis and accumulation of messenger RNA in nondividing cultures. Proc Natl Acad Sci U S A. 1974 Mar;71(3):750–754. doi: 10.1073/pnas.71.3.750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudland P. S., Jimenez de Asua L. Action of growth factors in the cell cycle. Biochim Biophys Acta. 1979 Feb 4;560(1):91–133. doi: 10.1016/0304-419x(79)90004-0. [DOI] [PubMed] [Google Scholar]
- Rudland P. S., Weil S., Hunter A. R. Changes in RNA metabolism and accumulation of presumptive messenger RNA during transition from the growing to the quiescent state of cultured mouse fibroblasts. J Mol Biol. 1975 Aug 25;96(4):745–766. doi: 10.1016/0022-2836(75)90150-3. [DOI] [PubMed] [Google Scholar]
- Salas J., Green H. Proteins binding to DNA and their relation to growth in cultured mammalian cells. Nat New Biol. 1971 Feb 10;229(6):165–169. doi: 10.1038/newbio229165a0. [DOI] [PubMed] [Google Scholar]
- Thomas G., Gordon J. Regulation of protein synthesis during the shift of quiescent animal cells into the proliferative state. Cell Biol Int Rep. 1979 Jul;3(4):307–320. doi: 10.1016/s0309-1651(79)80001-6. [DOI] [PubMed] [Google Scholar]
- Thomas G., Siegmann M., Bowman P. D., Gordon J. The isolation and analysis of polysomes and ribosomal RNA from cells growing in monolayer culture. Exp Cell Res. 1977 Sep;108(2):253–258. doi: 10.1016/s0014-4827(77)80032-3. [DOI] [PubMed] [Google Scholar]
- Thomas G., Siegmann M., Gordon J. Multiple phosphorylation of ribosomal protein S6 during transition of quiescent 3T3 cells into early G1, and cellular compartmentalization of the phosphate donor. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3952–3956. doi: 10.1073/pnas.76.8.3952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas G., Siegmann M., Kubler A. M., Gordon J., Jimenez de Asua L. Regulation of 40S ribosomal protein S6 phosphorylation in Swiss mouse 3T3 cells. Cell. 1980 Apr;19(4):1015–1023. doi: 10.1016/0092-8674(80)90092-6. [DOI] [PubMed] [Google Scholar]
- Williams J. G., Penman S. The messenger RNA sequences in growing and resting mouse fibroblasts. Cell. 1975 Oct;6(2):197–206. doi: 10.1016/0092-8674(75)90010-0. [DOI] [PubMed] [Google Scholar]
- de Asua L. J., O'Farrell M. K., Clingan D., Rudland P. S. Temporal sequence of hormonal interactions during the prereplicative phase of quiescent cultured 3T3 fibroblasts. Proc Natl Acad Sci U S A. 1977 Sep;74(9):3845–3849. doi: 10.1073/pnas.74.9.3845. [DOI] [PMC free article] [PubMed] [Google Scholar]