Abstract
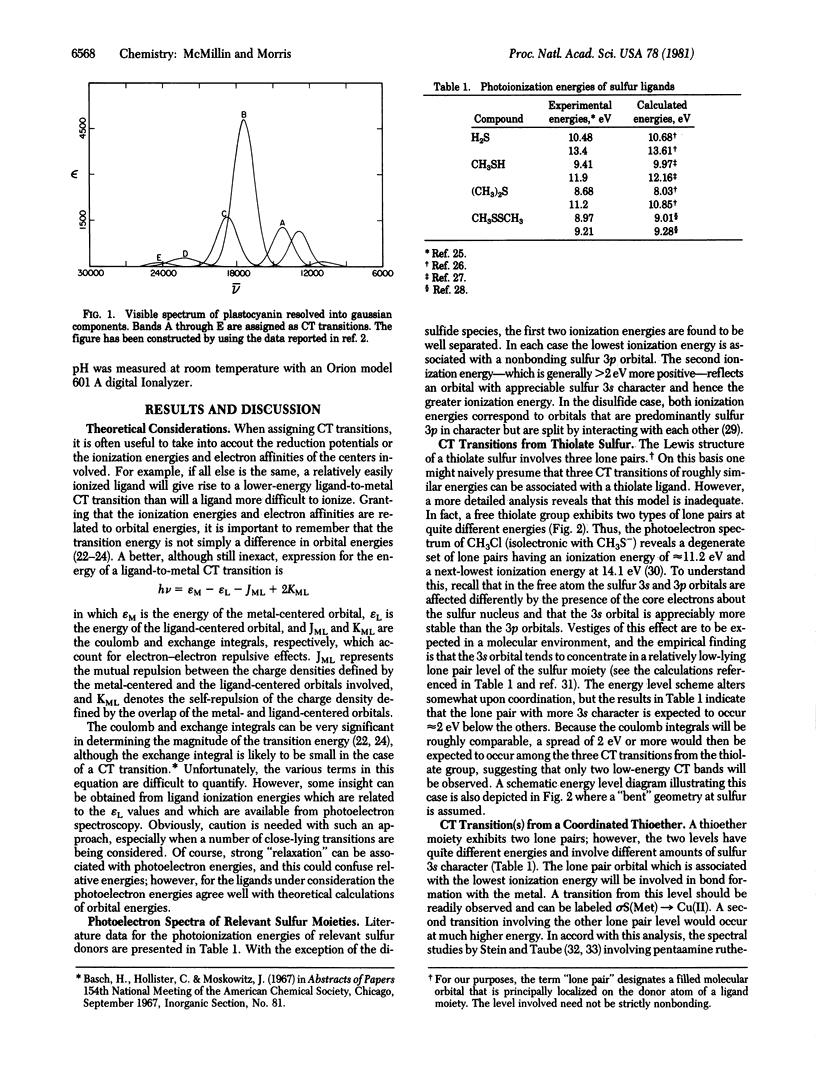
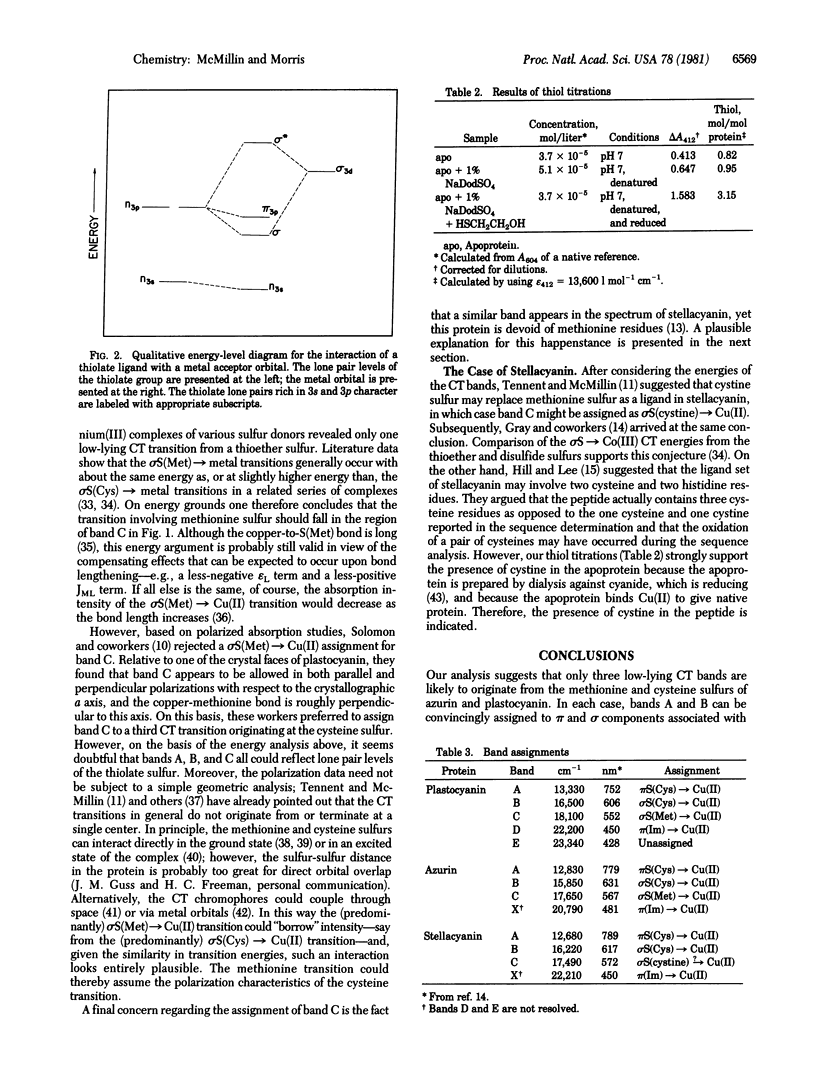
The charge transfer spectra of plastocyanin and azurin are analyzed in detail, taking into account the structural information that is available. The number and relative energies of the low-lying charge transfer bands attributable to cysteine and methionine sulfur are considered with reference to the energy levels associated with the donor centers and to experimental work on relevant complexes. Two cysteine → Cu(II) charge transfer transitions are assigned to bands that occur near 620 nm and near 770 nm. Energy considerations suggest that the methionine → Cu(II) transition occurs in the vicinity of 550 nm for both plastocyanin and azurin. The existence of a disulfide linkage in stellacyanin is established by thiol titrations, and the possibility exists that the fourth ligand in stellacyanin may involve disulfide sulfur. If so, the cystine → Cu(II) transition is also expected to appear around 550 nm.
Keywords: visible absorption, methionine, cysteine, cystine
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amundsen A. R., Whelan J., Bosnich B. Biological analogues. On the nature of the binding sites of copper-containing proteins. J Am Chem Soc. 1977 Sep 28;99(20):6730–6739. doi: 10.1021/ja00462a042. [DOI] [PubMed] [Google Scholar]
- Bergaman C., Gandvik E. K., Nyman P. O., Strid L. The amino acid sequence of Stellacyanin from the lacquer tree. Biochem Biophys Res Commun. 1977 Aug 8;77(3):1052–1059. doi: 10.1016/s0006-291x(77)80084-3. [DOI] [PubMed] [Google Scholar]
- Dooley D. M., Dawson J. H., Stephens P. J., Gray H. B. Spectroscopic studies of ascorbate oxidase. Electronic structure of the blue copper sites. Biochemistry. 1981 Mar 31;20(7):2024–2028. doi: 10.1021/bi00510a044. [DOI] [PubMed] [Google Scholar]
- ELLMAN G. L. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959 May;82(1):70–77. doi: 10.1016/0003-9861(59)90090-6. [DOI] [PubMed] [Google Scholar]
- Eklund H., Nordström B., Zeppezauer E., Söderlund G., Ohlsson I., Boiwe T., Brändén C. I. The structure of horse liver alcohol dehydrogenase. FEBS Lett. 1974 Aug 25;44(2):200–204. doi: 10.1016/0014-5793(74)80725-8. [DOI] [PubMed] [Google Scholar]
- Finazzi-Agrò A., Giovagnoli C., Avigliano L., Rotilio G., Mondovì B. Luminescence quenching in azurin. Eur J Biochem. 1973 Apr 2;34(1):20–24. doi: 10.1111/j.1432-1033.1973.tb02723.x. [DOI] [PubMed] [Google Scholar]
- Graziani M. T., Agrò A. F., Rotilio G., Barra D., Mondovi B. Parsley plastocyanin. The possible presence of sulfhydryl and tyrosine in the copper environment. Biochemistry. 1974 Feb 12;13(4):804–809. doi: 10.1021/bi00701a025. [DOI] [PubMed] [Google Scholar]
- Hill H. A., Lee W. K. Investigation of the structure of the blue copper protein from Rhus vernicifera stellacyanin by 1H nuclear magnetic resonance spectroscopy. J Inorg Biochem. 1979 Oct;11(2):101–113. doi: 10.1016/s0162-0134(00)80176-2. [DOI] [PubMed] [Google Scholar]
- Maret W., Dietrich H., Ruf H. H., Zeppezauer M. Active site-specific reconstituted copper(II) horse liver alcohol dehydrogenase: a biological model for type 1 Cu2+ and its changes upon ligand binding and conformational transitions. J Inorg Biochem. 1980 Jun;12(3):241–252. doi: 10.1016/s0162-0134(00)80205-6. [DOI] [PubMed] [Google Scholar]
- McMillin D. R., Holwerda R. A., Gray H. B. Preparation and spectroscopic studies of cobalt(II)-stellacyanin. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1339–1341. doi: 10.1073/pnas.71.4.1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMillin D. R., Rosenberg R. C., Gray H. B. Preparation and spectroscopic studies of cobalt(II) derivatives of blue copper proteins. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4760–4762. doi: 10.1073/pnas.71.12.4760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morpurgo L., Finazzi-Agrò A., Rotilio G., Mondovì B. Studies of the metal sites of copper proteins. IV. Stellacyanin: preparation of apoprotein and involvement of sulfhydryl and tryptophan in the copper chromophore. Biochim Biophys Acta. 1972 Jul 21;271(2):292–299. doi: 10.1016/0005-2795(72)90203-6. [DOI] [PubMed] [Google Scholar]
- Reinhammar B. Purification and properties of laccase and stellacyanin from Rhus vernicifera. Biochim Biophys Acta. 1970 Apr 7;205(1):35–47. doi: 10.1016/0005-2728(70)90059-9. [DOI] [PubMed] [Google Scholar]
- Solomon E. I., Hare J. W., Gray H. B. Spectroscopic studies and a structural model for blue copper centers in proteins. Proc Natl Acad Sci U S A. 1976 May;73(5):1389–1393. doi: 10.1073/pnas.73.5.1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tullius T. D., Frank P., Hodgson K. O. Characterization of the blue copper site in oxidized azurin by extended x-ray absorption fine structure: Determination of a short Cu-S distance. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4069–4073. doi: 10.1073/pnas.75.9.4069. [DOI] [PMC free article] [PubMed] [Google Scholar]



