Abstract
Objective
The aim of this study was to compare the efficacy of indirect and combined bypass surgery for treatment of adult moyamoya disease (MMD). The definition of combined bypass surgery is a combination of superficial temporal artery-middle cerebral artery (STA-MCA) anastomosis and indirect anastomosis. Development of collateral circulation after surgery was investigated.
Methods
Forty three patients (58 hemispheres) with MMD were followed by cerebral angiography for at least six months after surgery, between May 2002 and July 2011. Indirect and combined revascularization surgeries were performed in 33 and 25 cases, respectively. Good outcome was defined as more than group B, in accordance with the method suggested by Matsushima.
Results
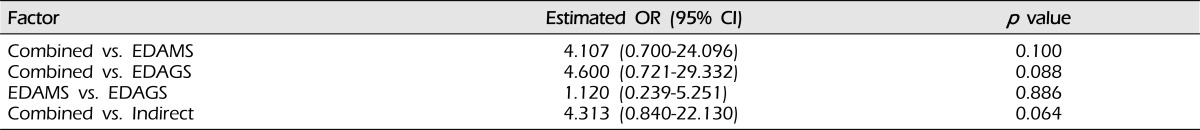
Development of collateral circulation was not affected by sex (p = 0.493), clinical features (p = 0.206), or Suzuki stage (p = 0.428). Based on postoperative cerebral angiography, the combined bypass surgery group showed a better angiographic outcome, than the encephaloduroarteriomyosynangiosis (EDAMS) group (p = 0.100, odds ratio [OR] 4.107, 95% confidence interval [CI] 0.700 - 24.096). The combined bypass group showed a better response than the encephaloduroarteriogaleosynangiosis (EDAGS) group (p = 0.088, OR 4.600, 95% CI 0.721 - 29.332). Similar responses were observed for EDAGS and EDAMS (p = 0.886, OR 1.120, 95% CI 0.239 - 5.251). The combined bypass group showed a better response than the indirect group (p = 0.064, OR 4.313, 95% CI 0.840 - 22.130).
Conclusion
Results of this study demonstrate that combined bypass results in better revascularization on angiographic evaluation in adult MMD. Therefore, among surgical procedures, combined bypass is a choice that can be recommended.
Keywords: Moyamoya disease, Cerebral revascularization, Indirect bypass surgery, Combined bypass surgery
INTRODUCTION
Moyamoya disease (MMD) is characterized by gradual stenosis or occlusion of the bilateral internal carotid arteries; this occlusion induces development of collateral circulation, called moyamoya vessels in the basal ganglia.19),24) Occurrence of this disease is more frequent among young teenagers and adults in their 30s to 40s, and represents the most common cause of ischemic stroke in children.5) MMD is generally characterized by cerebral ischemia and cerebral infarction in children and by hemorrhagic lesions in adults. Various symptoms, including aphasia and hemiparesis, may appear, depending on the sites of cerebral ischemia or hemorrhage.7),19)
Medical treatment for MMD is ineffective,19) however, there are three representative techniques used in surgical treatment.15) The first is a direct anastomosis, such as a superficial temporal artery-middle cerebral artery (STA-MCA) procedure; the second is an indirect anastomosis such as encephaloduroarteriogaleosynangiosis (EDAGS), encephalomyosynangiosis (EMS), encephaloduroarteriomyosynangiosis (EDAMS), encephalogaleosynangiosis (EGS) and multiple burr-hole surgery; and the third is a combined bypass surgery.1),4),6),10),12),14),18),20),22) The definition of combined bypass surgery is a combination of STA-MCA anastomosis and indirect anastomosis. The goal of these surgical treatments is to minimize and prevent symptoms of cerebral ischemia by effectively forming collateral circulation from the outside to the inside of the cranial cavity.15)
Currently, among these methods, there are no clear data indicating definite superiority. The indirect revascularization method is aimed at stimulating development of new vascular networks and is thought to lead to delayed collateralization, however, the extent of revascularization is considered unpredictable, whereas, with direct revascularization, selective perfusion of ischemic areas can be performed immediately, however, use of this procedure could result in hyperperfusion syndrome and hemorrhage as a complication.
In the current study, we evaluated the effectiveness of various surgical methods for treatment of MMD by analysis of cerebral angiographic results. In addition, factors affecting efficient collateral circulation were also evaluated, in terms of sex, clinical presentation, and Suzuki stage.
MATERIALS AND METHODS
Fifty eight hemispheres of 43 patients over the age of 15 years (mean 42.3 ± 15.8 years, 16-62 years) with confirmed or probable MMD underwent indirect or combined bypass surgery between May 2002 and July 2011. Each patient was followed by cerebral angiography for at least six months. There were 21 male hemispheres (36.2%) and 37 female hemispheres (63.8%). The average follow-up period was 23 ± 20.0 months (range, 6-67 months). Indirect revascularization surgery was performed as EDAMS in 20 cases, EDAGS in 13 cases, and combined revascularization surgery was performed in 25 cases. The combined group included 20 cases of STA-MCA plus EDAGS, and five cases of STA-MCA plus EDAMS.
Surgery was performed under general anesthesia. During surgery, patients were monitored continuously in order to maintain normal carbon dioxide tension (PCO2), blood pressure, and body temperature. Urine output was measured, and appropriate fluids were supplied in order to prevent dehydration.20)
For EDAGS, the scalp incision was made directly above the parietal branch of the STA and extended anteriorly in a blunt angle to the distal frontal branch of the STA. Both branches of the STA were exposed for as long as possible, and the galea and skin were widely divided in order to obtain sufficient galea attached and surround the STA. Then, two lines of incisions were placed in the galea two to three cm apart from each side of both branches of the STA to the main trunk of the STA in the shape of the letter Y, such that a pedicle of the galea was attached to the STA over its entire exposed length and separated them from the underlying temporal fascia or the periosteum in a bridge form. After the Y-shaped STA-galeal flap (STAGF) was retracted to either side, the exposed periosteum and the fascia were incised by means of an electric cautery in a straight line in order to expose the cranium underneath. One burr hole was then made for performance of craniotomy: one was placed just proximal to the main STA, then, the temporal base was removed piece by piece in order to preserve the middle meningeal artery (MMA) after craniotomy. The dura was incised into the pedicle-based bone window and rolled back on the brain surface. The larger of two branches of the STA was cut at its distal side with the attached galea together in a flap. Then, the STAGF was inverted so as, through its richly vascularized outer surface, to contact the brain cortex exposed at the site of the dural defect. The arachnoid membrane was opened at many sites. The galeal edges of the STAGF were connected to the margins of the opening of the dura mater by suturing (using No. 3 silk thread) for completion of EDAGS. Consequently, the exposed brain surface was fully covered with STAGF as widely and closely as possible.
For EDAMS, the skin was incised beginning 1 cm above the root of zygoma of the temporal bone and continuing along the STA, while sufficiently preserving the surrounding soft tissues, and curving toward the back from the intersection with the linea temporalis. After detaching the temporal muscle, an incision 5-6 cm in length and 6-7 cm in width was made in the skull. The dura was incised, while preserving the main branch of the MMA, and the dura was pushed inside between the dura and the arachnoid membrane so that the periosteal layer of the incised dura mater would contact the surface of the arachnoid membrane. The arachnoid membrane was opened at many sites; and the STA was sutured to the pia mater using 10-0 nylon sutures. Then, the temporalis muscle was sutured to a dural edge, and the inner table of the bone flap was removed in order to reduce the mass effect. If the mass effect was a concern due to the thickness of the temporalis muscle, the muscle layer was divided into two halves, and only the inner half was transplanted to the pia mater, with its edge sutured to the dura.20)
For the direct technique, the parietal or, less often, the frontal branch of the STA (donor vessels; over 1 mm in diameter) was first checked by doppler sonography and then dissected along its course via a linear incision (8 to 10 cm of dissection). This was followed by performance of a small craniotomy (5 to 6 cm in diameter) on Chater's point, which corresponds to the end of the sylvian fissure. After finding a suitable branch of the MCA, direct anastomosis between the STA branch and the cortical MCA branch was achieved with 8 to 10 interrupted stitches of 10-0 or 11-0 suture. We used the thickest branch among the angular, posterior temporal, and posterior parietal arteries.20)
Cerebral angiography was performed for a minimum of six months after surgery, in order to evaluate development of collateral circulation. In accordance with the method suggested by Matsushima et al.,21) a good score (group A) indicated revascularization of more than two-thirds of the MCA distribution through the external carotid artery (ECA), a fair score (group B) indicated revascularization of one- to two-thirds of the MCA distribution, and a poor score (group C) indicated slight or no revascularization. Subjects in group A and B were considered as having a good angiographic outcome.
SPSS version 12.0 (SPSS Inc., Chicago, IL) was used in performance of statistical analysis. Pearson's Chi-square test was used for analysis of development of collateral circulation according to gender, surgical technique, clinical presentation and Suzuki classification.
RESULTS
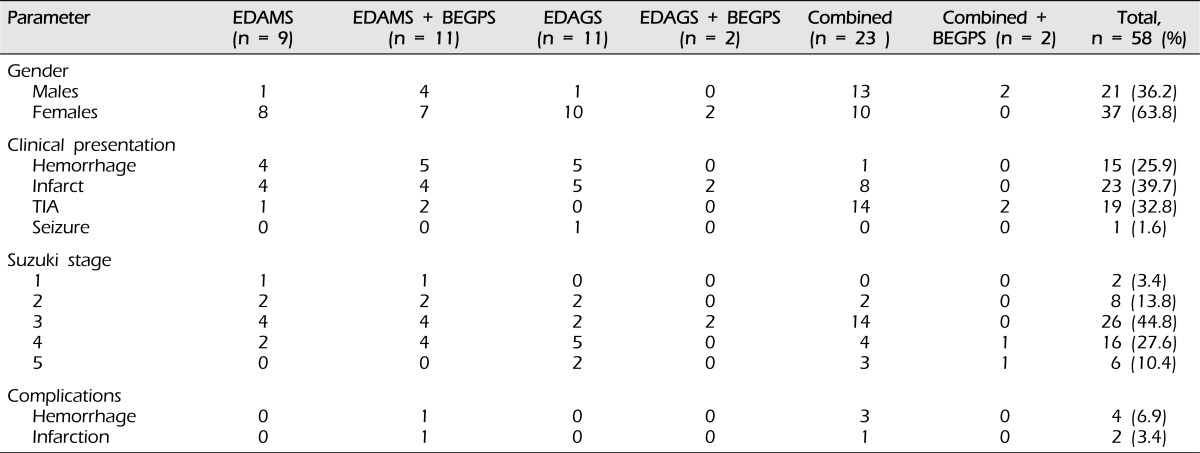
There were 21 male hemispheres (36.2%) and 37 female hemispheres (63.8%). In the indirect group, cerebral infarction was the most frequent clinical feature at the time of incidence (15 cases; 45.5%). In the combined group, transient ischemic attack (TIA) was the most frequent clinical feature at the time of incidence (16 cases; 64.0%). Six complications occurred. In the combined group, there were three hemorrhages and one infarction. In the indirect group, there was one hemorrhage and one infarction (Table 1).
Table 1.
Clinical summary of the 58 hemispheres with moyamoya.
EDAMS = encephaloduroarteriomyosynangiosis; EDAGS = encephaloduroarteriogaleosynangiosis; BEGPS = bifrontal encephalogaleoperiosteal synangiosis; Combined = direct + indirect; TIA = transient ischemic attack
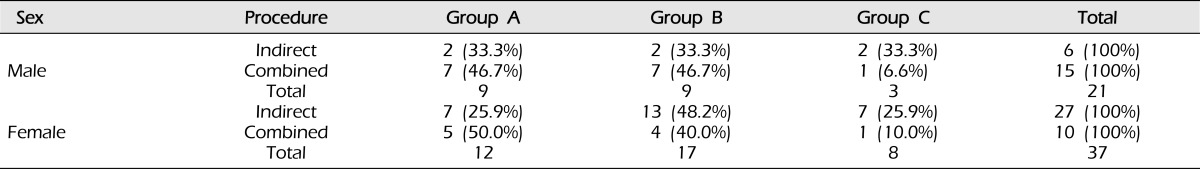
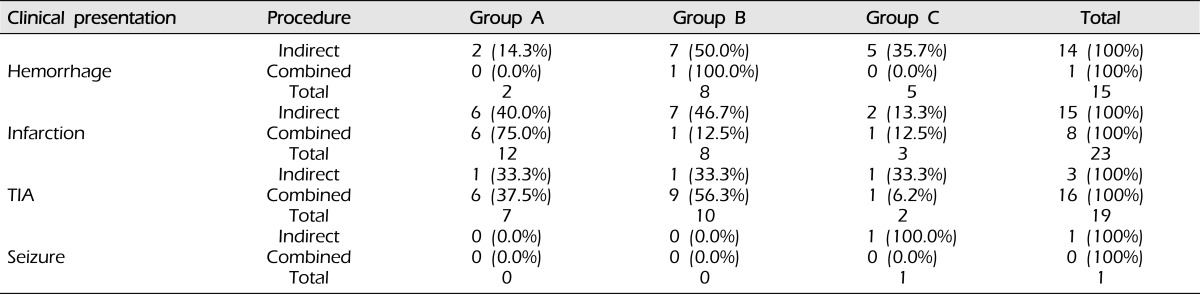
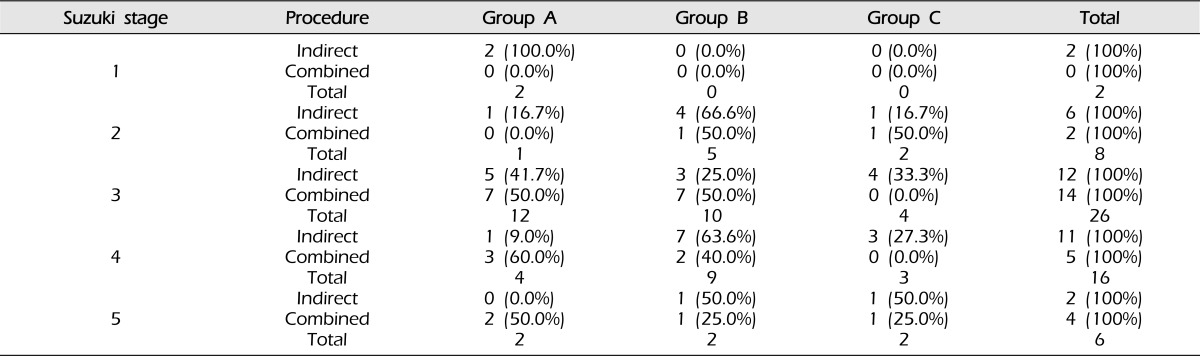
Angiographic outcome after surgery did not differ according to sex (p = 0.493, Table 2), clinical presentation (p = 0.206, Table 3), or Suzuki stage (p = 0.428, Table 4).
Table 2.
Surgical outcomes of bypass according to gender (p = 0.493).
Analyzed using the chi-square test.
Group A: Revascularization area was more than two-thirds of the middle cerebral artery (MCA) distribution.
Group B: Revascularization area was between one-third and two-thirds of the MCA distribution.
Group C: Revascularization area was less than one-third of the MCA distribution.
Table 3.
Surgical outcomes of bypass according to clinical presentation (p = 0.206).
Analyzed using the chi-square test.
Group A: Revascularization area was more than two-thirds of the middle cerebral artery (MCA) distribution.
Group B: Revascularization area was between one-third and two-thirds of the MCA distribution.
Group C: Revascularization area was less than one-third of the MCA distribution.
Table 4.
Surgical outcomes for bypass according to Suzuki stage (p = 0.478).
Analyzed using the chi-square test.
Group A: Revascularization area was more than two-thirds of the middle cerebral artery (MCA) distribution.
Group B: Revascularization area was between one-third and two-thirds of the MCA distribution.
Group C: Revascularization area was less than one-third of the MCA distribution.
As shown in Table 5, in the combined group, 23 cases (92.0%) showed a good outcome. In the EDAMS and EDAGS groups, positive outcomes were observed 15 (75.0%) and nine cases (69.2%), respectively. A better response was observed in the Combined group, compared with the EDAMS group (p = 0.100, odds ratio [OR] 4.107, 95% confidence interval [CI] 0.700 - 24.096). Patients in the Combined group showed a better response than those in the EDAGS group (p = 0.088, OR 4.600, 95% CI 0.721 - 29.332). EDAGS and EDAMS showed a similar response (p = 0.886, OR 1.120, 95% CI 0.239 - 5.251). The Combined group showed a better response than the indirect group (p = 0.064, OR 4.313, 95% CI 0.840 - 22.130, Table 6).
Table 5.
Surgical outcomes of bypass according to procedure.
Group A: Revascularization area was more than two-thirds of the middle cerebral artery (MCA) distribution.
Group B: Revascularization area was between one-third and two-thirds of the MCA distribution.
Group C: Revascularization area was less than one-third of the MCA distribution.
EDAMS = encephaloduroarteriomyosynangiosis; EDAGS = encephaloduroarteriogaleosynangiosis; Combined = direct surgery + indirect surgery
Table 6.
Multiple logistic regression analysis in surgical outcomes.
Analyzed using the chi-square test.
Group A: Revascularization area was more than two-thirds of the middle cerebral artery (MCA) distribution.
Group B: Revascularization area was between one-third and two-thirds of the MCA distribution.
Group C: Revascularization area was less than one-third of the MCA distribution.
DISCUSSION
The aim of surgical treatment for patients with MMD has been creation of new collateral channels between the external carotid system and cerebral cortical arteries.11) All surgical techniques are essentially based on the same surgical principle in which arteries or vascularized tissues (eg, the temporal muscle, the dura mater, the galea, and the omentum) are placed indirectly on the brain surface or direct anastomosis. In this study, we found that combined bypass tended to result in better development of collateral circulation. This result suggests that an indirect operation is sometimes insufficient, and combined surgery induces not only an immediate but also a slow yet long-standing revascularization by use of all vessels capable of providing the collateral circulation from the external carotid artery to the brain cortex in MMD.
Therefore, we confirmed that the combination of STA-MCA anastomosis and other indirect surgery is an attempt to achieve a maximal increase in collateral circulation from the STA with surrounding wide galeal tissue by contact through its highly vascularized external surface with the ischemic brain cortex as widely and closely as possible.16) Many recent studies have reported good results using combined revascularization, rather than a single direct or indirect bypass operation.2),10),17),19),23)
However, our study has some limitations. First, we analyzed revascularization extent using a lateral angiographic view, which excluded the anteroposterior view of the angiographic image. Thus, a true, accurate three-dimensional volume-concept analysis of revascularization extent was not performed. Second, the number of cases was relatively small (43 patients, 58 hemispheres). To solve these problems, we will conduct a comparison of the degree of collateral circulation on accurate three-dimensional volume-concept analysis and study more patients with long-term follow-up. Finally, in this study, we used only angiographic results. Angiographic results do not always show correlation with clinical ones; therefore, in order to perform a more exact evaluation of the effect of various surgical methods, both results should be considered in a future study.
CONCLUSION
Although the difference was not statistically significant, compared with indirect, combined bypass surgery resulted in better development of collateral circulation. A similar response was observed for EDAMS and EDAGS. Therefore, among surgical procedures, combined bypass is a choice that can be recommended.
References
- 1.Amine AR, Moody RA, Meeks W. Bilateral temporal-middle cerebral artery anastomosis for Moyamoya syndrome. Surg Neurol. 1977 Jul;8(1):3–6. [PubMed] [Google Scholar]
- 2.Czabanka M, Vajkoczy P, Schmiedek P, Horn P. Age-dependent revascularization patterns in the treatment of moyamoya disease in a European patient population. Neurosurg Focus. 2009 Apr;26(4):E9. doi: 10.3171/2009.1.FOCUS08298. [DOI] [PubMed] [Google Scholar]
- 3.Dauser RC, Tuite GF, McCluggage CW. Dural inversion procedure for moyamoya disease. Technical note. J Neurosurg. 1997 Apr;86(4):719–723. doi: 10.3171/jns.1997.86.4.0719. [DOI] [PubMed] [Google Scholar]
- 4.Erickson DL, Koivukangas J. The treatment of Moyamoya disease by superficial temporal-middle cerebral artery (STA-MCA) anastomosis. Ann Clin Res. 1986;18(Suppl 47):21–24. [PubMed] [Google Scholar]
- 5.Goda M, Isono M, Ishii K, Kamida T, Abe T, Kobayashi H. Long-term effects of indirect bypass surgery on collateral vessel formation in pediatric moyamoya disease. J Neurosurg. 2004 Feb;100(2 Suppl Pediatrics):156–162. doi: 10.3171/ped.2004.100.2.0156. [DOI] [PubMed] [Google Scholar]
- 6.Golby AJ, Marks MP, Thompson RC, Steinberg GK. Direct and combined revascularization in pediatric moyamoya disease. Neurosurgery. 1999 Jul;45(1):50–58. doi: 10.1097/00006123-199907000-00013. discussion 58-60. [DOI] [PubMed] [Google Scholar]
- 7.Han DH, Nam DH, Oh CW. Moyamoya disease in adults: characteristics of clinical presentation and outcome after encephalo-duro-arterio-synangiosis. Clin Neurol Neurosurg. 1997 Oct;99(Suppl 2):S151–S155. doi: 10.1016/s0303-8467(97)00058-9. [DOI] [PubMed] [Google Scholar]
- 8.Houkin K, Kamiyama H, Takahashi A, Kuroda S, Abe H. Combined revascularization surgery for childhood moyamoya disease: STA-MCA and encephalo-duro-arterio-myo-synangiosis. Childs Nerv Syst. 1997 Jan;13(1):24–29. doi: 10.1007/s003810050034. [DOI] [PubMed] [Google Scholar]
- 9.Ishii R. [Surgical treatment of moyamoya disease] No Shinkei Geka. 1986 Aug;14(9):1059–1068. Japanese. [PubMed] [Google Scholar]
- 10.Ishikawa T, Kamiyama H, Kuroda S, Yasuda H, Nakayama N, Takizawa K. Simultaneous superficial temporal artery to middle cerebral or anterior cerebral artery bypass with pan-synangiosis for Moyamoya disease covering both anterior and middle cerebral artery territories. Neurol Med Chir (Tokyo) 2006 Sep;46(9):462–468. doi: 10.2176/nmc.46.462. [DOI] [PubMed] [Google Scholar]
- 11.Karasawa J, Kikuchi H, Furuse S, Kawamura J, Sakaki T. Treatment of moyamoya disease with STA-MCA anastomosis. J Neurosurg. 1978 Nov;49(5):679–688. doi: 10.3171/jns.1978.49.5.0679. [DOI] [PubMed] [Google Scholar]
- 12.Karasawa J, Kikuchi H, Furuse S, Sakaki T, Yoshida Y. A surgical treatment of moyamoya disease encephalo-myo synangiosis. Neurol Med Chir (Tokyo) 1977;17(1 Pt 1):29–37. doi: 10.2176/nmc.17pt1.29. [DOI] [PubMed] [Google Scholar]
- 13.Kashiwagi S, Kato S, Yamashita K, Takasago T, Akimura T, Okamura S, et al. Revascularization with split duro-encephalo-synangiosis in the pediatric moyamoya disease-surgical result and clinical outcome. Clin Neurol Neurosurg. 1997 Oct;99(Suppl 2):S115–S117. doi: 10.1016/s0303-8467(97)00069-3. [DOI] [PubMed] [Google Scholar]
- 14.Kawaguchi T, Fujita S, Hosoda K, Shose Y, Hamano S, Iwakura M, et al. Multiple burr-hole operation for adult moyamoya disease. J Neurosurg. 1996 Mar;84(3):468–476. doi: 10.3171/jns.1996.84.3.0468. [DOI] [PubMed] [Google Scholar]
- 15.Kim DS, Kang SG, Yoo DS, Huh PW, Cho KS, Park CK. Surgical results in pediatric moyamoya disease: angiographic revascularization and the clinical results. Clin Neurol Neurosurg. 2007 Feb;109(2):125–131. doi: 10.1016/j.clineuro.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 16.Kim DS, Yoo DS, Huh PW, Kang SG, Cho KS, Kim MC. Combined direct anastomosis and encephaloduroarteriogaleosynangiosis using inverted superficial temporal artery-galeal flap and superficial temporal artery-galeal pedicle in adult moyamoya disease. Surg Neurol. 2006 Oct;66(4):389–394. doi: 10.1016/j.surneu.2006.02.047. discussion 395. [DOI] [PubMed] [Google Scholar]
- 17.Kim SH, Choi JU, Yang KH, Kim TG, Kim DS. Risk factors for postoperative ischemic complications in patients with moyamoya disease. J Neurosurg. 2005 Nov;103(5 Suppl):433–438. doi: 10.3171/ped.2005.103.5.0433. [DOI] [PubMed] [Google Scholar]
- 18.Kinugasa K, Mandai S, Kamata I, Sugiu K, Ohmoto T. Surgical treatment of moyamoya disease: operative technique for encephalo-duro-arterio-myo-synangiosis, its follow-up, clinical results, and angiograms. Neurosurgery. 1993 Apr;32(4):527–531. doi: 10.1227/00006123-199304000-00006. [DOI] [PubMed] [Google Scholar]
- 19.Kuroda S, Houkin K. Moyamoya disease: current concepts and future perspectives. Lancet Neurol. 2008 Nov;7(11):1056–1066. doi: 10.1016/S1474-4422(08)70240-0. [DOI] [PubMed] [Google Scholar]
- 20.Lee JP, Cho SJ, Park HK, Park SQ, Chang JC, Choi SK. Angiographic and Clinical Results of Indirect Bypass Surgery for Moyamoya Disease. Korean J Cerebrovasc Surg. 2010 Dec;12(4):250–258. [Google Scholar]
- 21.Matsushima T, Fukui M, Kitamura K, Hasuo K, Kuwabara Y, Kurokawa T. Encephalo-duro-arterio-synangiosis in children with moyamoya disease. Acta Neurochir (Wien) 1990;104(3-4):96–102. doi: 10.1007/BF01842826. [DOI] [PubMed] [Google Scholar]
- 22.Park JH, Yang SY, Chung YN, Kim JE, Kim SK, Han DH, et al. Modified encephaloduroarteriosynangiosis with bifrontal encephalogaleoperiosteal synangiosis for the treatment of pediatric moyamoya disease. Technical note. J Neurosurg. 2007 Mar;106(3 Suppl):237–242. doi: 10.3171/ped.2007.106.3.237. [DOI] [PubMed] [Google Scholar]
- 23.Starke RM, Komotar RJ, Connolly ES. Optimal surgical treatment for moyamoya disease in adults: direct versus indirect bypass. Neurosurg Focus. 2009 Apr;26(4):E8. doi: 10.3171/2009.01.FOCUS08309. [DOI] [PubMed] [Google Scholar]
- 24.Suzuki J, Takaku A. Cerebrovascular moyamoya disease. Disease showing abnormal net-like vessels in base of brain. Arch Neurol. 1969 Mar;20(3):288–299. doi: 10.1001/archneur.1969.00480090076012. [DOI] [PubMed] [Google Scholar]