Abstract
The immune system has evolved multipronged responses that are critical to effectively defend the body from invading pathogens and to clear infection. However, the same weapons employed to eradicate infection can have caustic effects on normal bystander cells. Therefore, tight regulation is vital and the host must balance engendering correct and sufficient immune responses to pathogens while limiting errant and excessive immunopathology. To accomplish this task a complex network of positive and negative immune signals are delivered that in most instances successfully eliminate pathogen. However, in response to some viral infections, immune function is rapidly suppressed leading to viral persistence. Immune suppression is a critical obstacle to the control of many persistent virus infections such as HIV, hepatitis C and hepatitis B virus, which together affect more than 500 million individuals worldwide. Thus, the ability to therapeutically enhance immunity is a potentially powerful approach to resolve persistent infections. The host derived cytokine IL-10 is a key player in the establishment and perpetuation of viral persistence. This chapter discusses the role of IL-10 in viral persistence and explores the exciting prospect of therapeutically blocking IL-10 to increase antiviral immunity and vaccine efficacy.
1. INTRODUCTION
Immune dynamics during acute and persistent virus infection: the failure of antiviral immunity
Viral invasion is rapidly detected through the triggering of pattern recognition receptors like toll like receptors (TLRs) and the retinoic acid-inducible gene 1-like helicases (RIG-I). The presence of viral genome triggers TLRs and RIG-I to stimulate production of type I interferon (i.e., IFNα and β) as well as other immunomodulatory proteins. (Koyama et al., 2008; Schlee and Hartmann, 2010). Type I interferons control early viral replication by directly fostering apoptosis and hindering proliferation of virally infected cells, as well as strongly stimulating cytotoxic natural killer (NK) cells. (Biron et al., 1999; Gartel et al., 1996; Tanaka et al., 1998) In addition to orchestrating the innate inflammatory environment, interferons play important roles in directing adaptive immune responses to effectively combat viral assault (Goodbourn et al., 2000).
Successful resolution of viral infection and the subsequent establishment of lasting immunological memory hinge upon the effecter cells of the adaptive immune system. The interplay of CD4 and CD8 T cell, and B cell responses ultimately dictates the outcome of infection. Naïve CD4 and CD8 T cell responses are primed by dendritic cells (DC). DC direct the type of T cell responses generated based on the cytokine environment and general millieau in which antigen is encountered (Banchereau and Steinman, 1998). Effective antiviral CD8+ T cytolytic lymphocytes (CTL) responses are primed by DC via cognate interactions of the T cell receptor (TCR) with peptide/major histocompatibility class I (MHC I) complexes resulting in cellular activation and a clonal expansion of antigen specific cells. CTL produce multiple inflammatory cytokines, such as IFNγ and TNFα and have the ability to lyse infected cells. Concurrently, CD4+ T cells are activated by DC and differentiate into distinct T helper (Th) subsets that shape ensuing CD8 T cell and B cell responses (Fahey and Brooks, 2010). Classically speaking, CD4+ Th1 responses are tailored to combat intracellular infections via production of IFNγ, TNFα, IL-2 and the perpetuation of the inflammatory environment. Levels of co-stimulatory molecules (i.e. CD80/86 and CD40) and the cytokine environment (notably IL12 and interferons) promote Th1 differentiation (Constant and Bottomly, 1997). At the same time, and dependent on CD4 T cell help, B cells are activated and produce antibody to further neutralize virus. After the peak of this acute response, virus-specific T cells undergo significant contraction and further differentiation into stable memory populations (Kaech et al., 2002). Ultimately, it is the summation of all these events that lead to successful viral clearance and memory differentiation to prevent re-infection.
In most situations this concerted effort of innate and adaptive responses is effective in eliminating the pathogen. However, in some cases the acute resolution of infection is incomplete and viral persistence results. Herpes simplex virus, human cytomegalovirus (HCMV) and Epstein-Barr virus (EBV), along with γ2-herpes virus in mice, are hallmark examples of infections that develop lifelong viral persistence by ‘hiding’ from the immune response. This presence of latent/reactivating infection is associated with functional T cell responses that control viral replication upon re-emergence. In contrast, human immunodeficiency virus (HIV), hepatitis C virus (HCV), and hepatitis B virus (HBV) infections in humans, and lymphocytic choriomeningitis virus (LCMV) infection in rodents establish persistent infections characterized by sustained high levels of viral replication and immunosuppression (Klenerman and Hill, 2005). In response to these persistent infections, virus-specific CD4 and CD8 T cells are physically deleted or persist in an attenuated (termed exhausted) developmental program unable to proliferate to viral antigens or produce important antiviral and immunostimulatory cytokines (e.g., IFNγ, TNFα, IL-2) (Brooks et al., 2006a; Gallimore et al., 1998; Wherry et al., 2003; Wherry et al., 2007; Zajac et al., 1998). The physical deletion of high affinity CTL and the low amount of remaining virus-specific CD8 T cells, in conjunction with the loss of cytokine production, the inability to proliferate to viral antigen and attenuated CD4 Th cell and B cell responses all culminate in the failure to purge infection. The exhausted state is characterized by a unique transcriptional profile featuring up-regulation of the transcription factor Blimp1 and inhibitory cell surface molecules, such as programmed death receptor 1 (PD1), along with down modulation of cytokine and TCR signaling molecules (Agnellini et al., 2007; Shin et al., 2009; Wherry et al., 2007). Thus, T cell exhaustion is a unique T cell developmental program, still active and exerting some control over virus replication (Battegay et al., 1994; Elsaesser et al., 2009; Frohlich et al., 2009; Matloubian et al., 1994; Yi et al., 2009), but distinct from the productive T cell responses during acute infection or the anergic/tolerant responses to self-proteins (Wherry et al., 2007).
Although potentially counter-intuitive, induction of this exhausted state is an important mechanism by which excessive immunopathology and death are avoided in the face of persistent antigen load. Our lab and others have demonstrated that host immunosuppressive factors potentiate the exhausted phenotype, even at the expense of facilitating viral persistence (Barber et al., 2006; Brooks et al., 2006b; Ejrnaes et al., 2006; Thimme et al., 2001) The benefits of this immune strategy are apparent in persistent LCMV infection where rapid mortality occurs when T cell responses are therapeutically augmented to prevent exhaustion during the initial response (Barber et al., 2006; Yi et al., 2009). Excitingly, however there are conditions in which early augmentation to prevent T cell exhaustion can be productive in clearing persistent infections without producing fatal immunopathology (Brooks et al., 2006b; Ejrnaes et al., 2006; Tinoco et al., 2009).
The immunoregulatory cytokine IL-10 has been shown to be a key host factor in inducing and maintaining T cell exhaustion and facilitating viral persistence (Bachmann et al., 2007; Brooks et al., 2006b; Ejrnaes et al., 2006; Humphreys et al., 2007; Moore et al., 2001). This review focuses on the potential of therapeutically manipulating IL-10 to safely promote clearance of viral infection without disturbing normal immune homeostasis and inadvertently inducing immunopathology.
2. THE IMPACT OF HOST-BASED REGULATORY MECHANISMS IN T CELL EXHAUSTION
2.1 Discovery that host-based suppressive factors inhibit clearance of persistent viral infection
Both general and virus-specific immune suppression has been well established during persistent viral infection, however, the mechanisms that govern this phenomenon have only recently been brought to light. Many persisting viruses (e.g., HCMV, HIV, HCV) encode proteins that actively suppress immunity either by direct inhibition of T cell responses and/or by down-regulating antigen recognition molecules (Klenerman and Hill, 2005; Slobedman et al., 2009). Other persistent viruses (e.g., LCMV) do not encode suppressive factors yet their infection still rapidly leads to a suppressive state (Ahmed et al., 1984). This suggests that the inability to rapidly control infection triggers a suppressive program within the host.
The seminal discovery by Rafi Ahmed and colleagues that blockade of the host-encoded protein programmed death (PD) ligand 1 (PD-L1) restored function to exhausted CD8 T cells and enhanced control of persistent LCMV infection, led to the realization that the host itself potentiates immune suppression during viral persistence (Barber et al., 2005). The suppressive role of PD-1/PD-L1 interaction was promptly demonstrated to suppress CD8 T cell responses to a variety of diverse persistent infections in vitro including the RNA viruses HIV (a retrovirus) and HCV (a flavivirus) and the DNA virus, HBV (a hepadnavirus) as well as in vivo against SIV (a non-human primate retrovirus) (Boni et al., 2007; Day et al., 2006; Petrovas et al., 2006; Trautmann et al., 2006; Urbani et al., 2006b; Velu et al., 2008). The diversity of these viruses with respect to replication strategies, target organs and infected cell types highlights the conserved role of PD-1/PD-L1 mediated immunosuppression and establish that the host is a powerful inhibitor of T cell immunity during persistent infection.
2.2 Multiple regulatory factors suppress T cell responses during viral persistence
Shortly following the identification of PD-1 mediated immunosuppresion during viral persistence, the dominant role of IL-10 in attenuating effector T cell responses to initiate persistent infection was established (Brooks et al., 2006b; Ejrnaes et al., 2006). Infection of mice with a persistent, but not an acutely cleared variant of LCMV leads to sustained expression of IL-10 by multiple immune cell subsets and functional exhaustion of CD4 and CD8 T cells (Brooks et al., 2006b; Ejrnaes et al., 2006). However, when IL-10 activity was neutralized, either using mice genetically deficient in IL-10 expression or antibodies that block the IL-10 receptor (IL-10R), immune function was sustained and the otherwise persistent virus was rapidly cleared. Persistent LCMV replicated to high titers in wild-type and IL-10 deficient mice 5 days post infection and prior to the onset of T cell responses. By day 9, T cells did not lose function in IL-10 deficient mice and they were able to clear persistent LCMV whereas viral titers remained high in wild type mice (Brooks et al., 2006b). CD8 T cells were required for this clearance since depletion of CD8 T cells in IL-10 deficient mice prior to infection led to LCMV persistence (E. Wilson and D. Brooks, unpublished observation). This was the first identification that a single factor was responsible for derailing the immune response to permit viral persistence and importantly, that sustaining T cell immunity could facilitate the clearance of an otherwise persistent infection.
Subsequently, multiple immunoregulatory factors were identified to limit T cell responses during viral persistence, including TGFβ, Tim3, CTLA4, CD27/CD70 (Jones et al., 2008; Kaufmann et al., 2007; Matter et al., 2006; Tinoco et al., 2009). Exhausted CD8 T cells simultaneously express multiple negative regulatory factors during persistent infection (Blackburn et al., 2009) and these factors can simultaneously, but via different pathways, limit T cell activity (Blackburn et al., 2009; Brooks et al., 2008a). Further, a single suppressive factor can differentially affect distinct T cell populations. For example, blockade of CTLA4 during persistent LCMV infection did not impact CD8 T cell responses in vivo (Barber et al., 2005), whereas it did enhance HIV-specific CD4 T cell responses in vitro (Kaufmann et al., 2007). Similarly, IL-10 directly limits CD4 T cell responses, but not CD8 T cell responses, to an acute LCMV infection (Brooks et al., 2010). In addition to T cells, IL-10 and other suppressive factors modulate multiple immune subsets including B cells, DC, macrophages and NK cells to contribute to enhanced virus control. The diversity in suppressive mechanisms provides the potential opportunity to individually manipulate T cell responses (particularly in combination with therapeutic vaccines) to produce the optimal effector response required to control a specific viral infection (see discussion below). Antibody therapies that block multiple suppressive pathways additively increase antiviral T cell activity (Blackburn et al., 2009; Brooks et al., 2008a). Thus, while increased production of suppressive factors by the immune system itself ultimately leads to the demise and failure of antiviral immunity, excitingly these factors can be inhibited for therapeutic benefit.
2.3 Differential impact of reversing T cell exhaustion during viral persistence
Understanding the mechanisms and coordination of the multitude of suppressive factors involved in immunoregulation is crucial to the design of effective antiviral therapies. Therapeutic strategies that target host-based factors to restore immune function are less susceptible to resistance via viral mutation as they do not target a specific viral protein. Thus, blockade of host-based negative regulatory factors could be effective against diverse persistent viruses that induce T cell exhaustion without engendering viral resistance.
Although extremely promising, the efficacy of blocking suppressive factors to enhance antiviral immunity in humans remains unclear. However, recent evidence blocking PD-1 in SIV infected rhesus macaques suggests that these blockade strategies may be effective (Velu et al., 2008). Short-term PD-1 blockade (4 treatments over 10 days) provided long-term restoration of T cell responses that correlated with enhanced SIV control. Interestingly, memory B cell responses and SIV-specific antibody production were also increased following PD-1 blockade. The reason for the increased B cell responses was not elucidated and could be due to either direct or indirect mechanisms (e.g., enhanced CD4 T cell help to B cells), but does indicate the exciting prospect that targeting a single molecule may simultaneously enhance multiple arms of the immune response culminating in virus control.
In total these studies indicate the incredible effect of blocking regulatory factors to enhance T cell function during viral persistence; however, this is not without potential negative impact. These dominant regulatory factors and pathways are instilled to prevent errant or unrestricted immune responses. Even in the absence of overt infection, deficiencies in these factors can lead to the massive expansion of effector-like T cells and a variety of autoimmune disorders (Hedrich and Bream, 2010; Moore et al., 2001). In response to an infection, the inability to attenuate T cell responses can lead to severe immunopathology and death. For example, persistent LCMV infection is fatal in PD-L1 knockout mice (Barber et al., 2005) and while IL-10 deficient mice survive and clear persistent LCMV infection (Brooks et al., 2006b; Ejrnaes et al., 2006) they are more susceptible to death in response to higher doses of persistent LCMV as compared to IL-10 sufficient hosts (D.G. Brooks, unpublished observations). Further, treatment with the immunostimulatory cytokine IL-21 during the early phase of persistent LCMV infection dramatically elevated virus-specific CD8 T cell responses and mortality (Yi et al., 2009). Thus, although detrimental to virus clearance, it is likely that the increased expression of negative regulatory factors and T cell exhaustion is a conserved and rapid mechanism to prevent lethal immunopathology when the host ‘senses’ that virus replication has out-competed the immune response to it. In some instances enhanced immunopathology is observed in the absence of IL-10 regulation without an effect on viral replication, as is the case during a neurotropic model of mouse hepatitis virus infection (Lin et al., 1998). On the other hand, once chronic infection has been established and T cell numbers have contracted, reversing exhaustion appears to be well handled in animal models of LCMV and SIV infection (Barber et al., 2005; Brooks et al., 2008a; Brooks et al., 2008b; Velu et al., 2008). The relationship between T cell exhaustion (i.e., attenuating T cell responses), excessive immunopathology and host survival must be carefully considered when optimizing therapies targeting host suppressive factors, particularly if instituted early during persistent virus infection.
Despite freeing virus-specific T cells to fight infection, blockade of regulatory factors may simultaneously unleash the regulation of self-specific immune cells or ‘tolerant’ immune cells in multiple organs and in the case of IL-10 blockade, particularly in the gut (Kuhn et al., 1993). Such a result could have the unintended consequence of triggering autoimmunity or immune responses to ingested food or endogenous enteric bacterial microbiota. It should be noted that overt autoimmunity was not observed in our studies when IL-10R blockade was implemented during the chronic phase of LCMV infection (D.G. Brooks, unpublished observation) nor following PD-1 blockade in SIV-infected macaques (Velu et al., 2008). However, these studies utilized short-term treatment regimens and longer term therapy or different individuals dependence on a particular pathway to maintain immune homeostasis could affect negative responses. Thus, while therapies that block negative regulators of immune function clearly hold tremendous antiviral potential the possible consequences should be carefully evaluated.
3. THE IMPACT OF IL-10 TOWARD VIRAL PERSISTENCE
3.1 The diverse roles of IL-10 during viral infection
IL-10 was initially known as cytokine synthesis inhibitory factor (CSIF) and was first identified as a CD4 produced Th2 cytokine with the ability to indirectly repress Th1 responses (Fiorentino et al., 1989; Moore et al., 2001). It is now evident that multiple cell types including DC, B cells, macrophages, CD4 T cells, CD8 T cells, NK cells as well as innate and adaptive regulatory T cells can produce IL-10 (Mege et al., 2006; Moore et al., 2001).
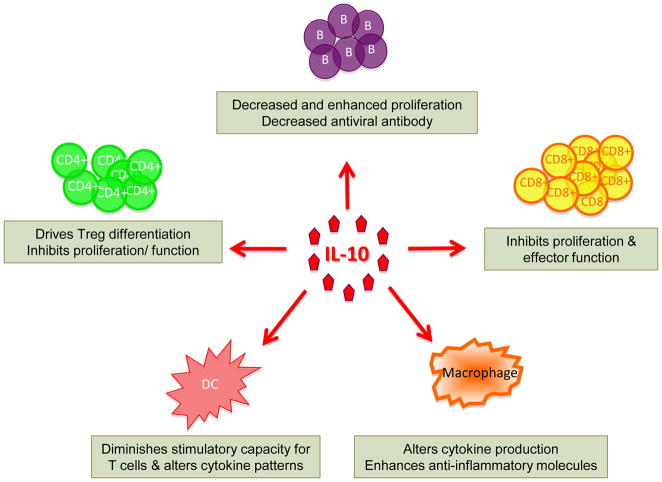
The IL-10 receptor (IL-10R) is a class II cytokine family member composed of two subunits, IL-10R1 is the unique ligand binding subunit and IL-10R2 is the signaling subunit that is shared with other family member cytokines (IL-22, IL-26, IL-28 and IL-29) (Donnelly et al., 2004). Dimerization of the receptor by IL-10 results in signaling through STAT3 and activation of gene expression (Kotenko et al., 1997; Liu et al., 1994; Moore et al., 2001; Spencer et al., 1998). Specificity of IL-10 responsiveness is dictated both by the expression of IL-10R1 and availability of the cytokine (Brooks et al., 2006b; Brooks et al., 2010). While the signaling subunit (IL-10R2) is constitutively expressed by most cells, IL-10R1 is differentially regulated by activation in a cell type specific manner in hematopoietic cells and is inducible on non-hematopoietic cells (Donnelly et al., 2004; Moore et al., 2001). Figure 1 illustrates some of important targets of IL-10 signaling in persistent viral infections.
Figure 1. Effects of IL-10 on distinct cellular targets during persistent viral infections.
IL-10 mediated immunosuppression in response to viral infection occurs via both direct and indirect modulation of APC function, T and B cell activity. This figure denotes some of the important functional consequences of IL-10 targeting of distinct immune subsets. Note that IL-10 likely acts on multiple other immune and non-immune cells during viral persistence and the overall influence of IL-10 is likely a summation of all these effects. Additional mechanisms of IL-10 mediated immune modulation have been described in bacterial and autoimmune models and investigating these pathways in viral persistence will be informative.
IL-10 aborts T cell responses when present during priming and can inhibit ongoing T cell activity to viral infections (Brooks et. al., 2006c; Ejrnaes et al., 2006; Groux et al., 1996; Steinbrink et al., 1997). It acts directly on antigen presenting cells to decrease stimulatory molecule expression (i.e., MHC class I and class II, B7-1, B7-2), alter cytokine production and prevent maturation ultimately dampening T cell activation (Carbonneil et al., 2004; Fiorentino et al., 1991; Moore et al., 2001; Steinbrink et al., 1997). In addition to these indirect effects, IL-10 can also act directly on T cells to limit proliferation, functional differentiation and effector activity (Brooks et al., 2010; Maynard and Weaver, 2008). Although controversial, emerging data also indicates that genetic polymorphisms in the IL-10 promoter that result in lower IL-10 production are associated with clearance of HCV infection and enhanced virus control during chronic HCV, HBV, HIV and EBV infections further supporting the important role of this cytokine in host immunosuppression (Cheong et al., 2006; Helminen et al., 1999; Paladino et al., 2006; Shin et al., 2003; Shin et al., 2000).
Counter to its negative regulatory functions, IL-10 can positively stimulate NK cells, in some instances CD8 T cells and induce B cell proliferation and antibody production (Foulds et al., 2006; Kang and Allen, 2005; Moore et al., 2001). Thus, although generally immunosuppressive, IL-10 can function through a variety of mechanisms (likely simultaneously) to fine-tune the pathogen-specific immune response. In total, these data emphasize the diverse functions (many of which may be pathogen specific) that IL-10 plays to regulate multiple immune parameters.
IL-10 plays a major role in limiting autoimmune disease under steady-state conditions by controlling circulating, self- or gut microbiota-reactive T cells. Consistent with this role, IL-10 knockout (KO) mice develop inflammatory bowel disease (IBD)/colitis approximately 8 weeks after birth (Davidson et al., 1996; Kuhn et al., 1993). Both CD4 T cells as well as resident enteric bacteria were required for the emergence of colitis indicating that under steady-state conditions IL-10 serves to suppress immune activation against the endogenous gut microbiota (Kuhn et al., 1993; Sellon et al., 1998). Although autoimmunity was not generally observed in other organs of IL-10 deficient mice it is likely that the cytokine also negatively regulates immunity in other tissue compartments. Specific incidence of IBD and colitis demonstrate the key importance of IL-10 in immune homeostasis to gut antigens.
3.2 Sources, mechanisms and targets of IL-10 during persistent virus infection
Sources
As discussed above, IL-10 can be produced by a variety of cell types to regulate their own and other cells functions. During persistent virus infection multiple cell types have been identified to produce IL-10, generally with immunosuppressive effects (Belkaid and Tarbell, 2009; Rouse et al., 2006). The dominant IL-10 producing cell type varies with different virus infections likely reflecting inherent differences in the pathogen-specific response as well as tissue-specific immune regulation. Although individual IL-10 expressing cells may exert some level of suppression, it is likely that during persistent virus infections the suppressive state mediated by IL-10 is maintained by multiple cells types that individually limit a variety of immune parameters, with the final effect being the inability to clear infection.
CD4 T cells were the first identified and are probably the most recognized IL-10 producing cell type. Upon activation and in response to the antigenic environment CD4 T cells differentiate into multiple Th subsets. These different subsets have both unique and overlapping qualities with multiple Th subsets capable of producing IL-10 (Maynard and Weaver, 2008). Initially identified as a Th2 cytokine (Fiorentino et al., 1989), IL-10 can also be produced by Th1, Th17, T follicular helper and both natural (Foxp3+) and induced (Foxp3−) T regulatory (Treg) cells (Maynard and Weaver, 2008). In general, IL-10 production by CD4 T cells during viral persistence is associated with the inducible Tr1 Treg population consisting of both virus-specific and non-specific cells (Rouse et al., 2006). The mechanism of Tr1 cell emergence in persistent infection is not known. They may arise non-specifically as a bystander effect of general T cell activation, they may be preferentially induced in an effort to limit excessive immunopathology or they could have deliberately evolved to arise in instances of prolonged immune activation. Finally, a variety of IL-10 expressing CD4 Th cells may be lumped into the Tr1 category with ‘regulation’ as only one of their potentially diverse functions.
The actual impact of Treg produced IL-10 in limiting virus-specific responses during persistent viral infection remains largely unclear. IL-10-mediated Treg activity during viral persistence is observed following infection of mice with Friend virus (FV). IL-10 expressing CD4 Treg cells are activated following FV infection and limit antiviral CD8 T cell responses in vivo facilitating increased persistent virus replication (Dittmer et al., 2004). However inhibition of Treg activity alone did not enhance control of virus replication, which required the adoptive transfer of large amounts of FV-specific CD8 T cells that could now function in the absence of IL-10 signals. Interestingly, FV-induced immunosuppression was IL-10 dependent, however IL-10 was not the Treg effector mechanism required to suppress CD8 T cells (Dittmer et al., 2004; Dittmer et al., 2002). IL-10 present during CD4 T cell priming can induce anergy, but also programs the development of additional IL-10 producing CD4 T cells (Groux et al., 1996; Groux et al., 1997). In such a manner, Treg produced IL-10 could promote the differentiation of more Treg cells as opposed to directly suppressing antiviral function, the latter being performed by other Treg produced inhibitory mechanims. Thus, IL-10 would be important for suppressing T cell responses, but would not itself be the direct effector mechanism.
Early following persistent LCMV infection, virus-specific IL-10 producing CD4 T cells are observed, but IL-10 protein expression rapidly decreases in conjunction with other Th1 cytokines (Brooks et al., 2005). IL-10 producing CD4 T cells are observed throughout persistent LCMV infection; however, they are relegated to the non-virus-specific CD4 T cell subset (Brooks et al., 2006b) and (D. Brooks, unpublished observation). Further, the amount of non-LCMV-specific IL-10 producing CD4 T cells in the spleen is similar during acute and persistent LCMV infection (E. Wilson, D. Brooks, unpublished observation). It will ultimately be interesting and important to determine whether these non-LCMV-specific CD4 T cells regulate the virus-specific immune response and the outcome of deleting these cells toward clearance of persistent LCMV infection.
In addition to CD4 T cells, IL-10 producing CD8 T cells, monocytes/macrophages, dendritic cells, B cells and NK cells are observed during persistent viral infections (Couper et al., 2008). During persistent LCMV infection IL-10 is produced by multiple cell types including NK cells, DC and B cells (Brooks et al., 2006b) all of which likely contribute to immune regulation. MCMV persistence in the salivary gland of infected mice is dependent on IL-10 producing CD4 T cells (Humphreys et al., 2007), whereas B cell produced IL-10 suppresses CD8 T cells in the spleen during MCMV infection (Madan et al., 2009). Thus, different cell types utilize IL-10 in a compartmentalized fashion to suppress distinct facets of immunity in the same host during persistent infection. A multi-cell mediated IL-10 response is also observed during HIV infection (Brockman et al., 2009; Yang et al., 2009), with monocyte/macrophages often comprising the largest IL-10 producing subset in the peripheral blood (Hagiwara et al., 1995; Kumar et al., 1998; Said et al., 2010). However, whether IL-10 producing cells differ in PBMC and tissue during HIV infection, and if so, what cells produce IL-10 in various tissue compartments, remains to be determined.
It will ultimately be critical to establish whether persistent viral infections are the result of IL-10 production by a single cell type with other IL-10 producing cells playing an auxiliary role or whether IL-10 production by multiple cell types is necessary. In the case of the former it will be important to define what cell type produces the ‘relevant’ IL-10 and how it aborts immunity. In the latter case, how each cell type suppresses individual immune components will need to be determined. Answers to these questions are critical from both a biologic standpoint to define the pathogenesis of persistent infection as well as from a therapeutic standpoint to modulate IL-10 expression by cells inhibiting antiviral activity while leaving other IL-10 producing cells intact to prevent autoimmunity and immunopathology.
Perhaps most strongly corroborating the importance of IL-10 toward viral persistence, several persistent viruses encode their own IL-10 homologs, including EBV, HCMV and some poxviruses to modulate the immune response and facilitate replication, spread and/or persistence (Slobedman et al., 2009). The first viral IL-10 (vIL-10) homolog to be identified was encoded by EBV with ebvIL-10 exhibiting ~70% amino acid sequence identity with human IL-10 (hIL-10) (Moore et al., 1990). EBV infects B cells leading to latent infection and in some cases B cell transformation. Both hIL-10 and ebvIL-10 have similar immunosuppressive activity and stimulated B cell proliferation, differentiation and antibody production (Slobedman et al., 2009). However, ebvIL-10 had ~1000-fold lower affinity for the cellular IL-10R and failed to promote MHC class II upregulation by B cells or to inhibit IL-2 production by CD4 T cells (Liu et al., 1997; Slobedman et al., 2009). Thus, in addition to its suppressive role permitting immune escape, an important function of ebvIL-10 may be to target B cell proliferation and differentiation thereby increasing the amount, permissiveness and/or transformation of infected cells without affecting the immune-stimulatory capacity. Similarly, HCMV encodes an IL-10 homolg with 27% identity to hIL-10 (Kotenko et al., 2000; Lockridge et al., 2000). hIL-10 and cmvIL-10 exhibit similar immunosuppressive and stimulatory characteristics: inhibiting LPS-induced DC maturation, cytokine production and upregulation of multiple T cell co-stimulatory molecules (Chang et al., 2004; Raftery et al., 2004). cmvIL-10 also inhibited type I interferon production by pDC, a major source of type-I interferon during viral infection (Chang et al., 2009). As discussed, type I interferons stimulate the virus-specific immune response and trigger a general antiviral state, but they also potently block HCMV infection. As a result, cmvIL-10 may enhance the spread of HCMV while simultaneously suppressing the early immune response. Interestingly, in vivo infection of mouse DC by murine CMV (MCMV) induced many of these same immunosuppressive effects despite not encoding an IL-10 homolog (Andrews et al., 2001). Thus, in addition to the direct affect of HCMV encoded IL-10, HCMV replication in DC in vivo may itself trigger an immunosuppressive program. During viral latency HCMV produces a shorter differentially spliced IL-10 variant sharing some of the immunosuppressive qualities of the cmvIL-10 produced during productive infection (e.g., down-regulation of MHC class II on monocytes) however, this homolog did not suppress DC maturation, co-stimulatory molecule induction or induce proliferation of a B cell line (Jenkins et al., 2004; Jenkins et al., 2008; Spencer et al., 2008). The decreased expression of MHC II inhibited CD4 T cell identification of latently infected cells allowing HCMV to evade immune recognition without affecting other immune functions that may compromise infection (Cheung et al., 2009). Thus, in its lifecycle, HCMV utilizes different IL-10 mediated suppressive mechanisms at different stages to persist.
Induction
One constant among persistent viruses is elevated expression of IL-10 and its direct correlation with virus replication (Brockman et al., 2009; Brooks et al., 2010; Cacciarelli et al., 1996; Yang et al., 2009). In addition to stimulatory factors, virus replication inherently triggers counter-regulatory measures to ultimately contain the immune response. Many signals inherent to immune activation induce IL-10 expression, but the precise ‘sensors’ of prolonged/heightened virus replication during persistent infection have yet to be determined. Pathogen specific IL-10 induction is likely achieved through the integration of multiple virus- and host-derived mechanisms and therefore will likely be dictated in a conserved and in a pathogen specific. It is possible that the same mechanisms responsible for the initial recognition of viral infection and induction of IL-10 continue to function throughout persistent infection. On the other hand (but certainly excluding the latter), prolonged/elevated viral levels may trigger subsequent factors that serve to continually stimulate IL-10 production.
As discussed in the introduction, the innate immune system initially senses viral infection via pattern recognition receptors (including multiple TLRs) leading to type-I interferon production and activation of the immune response (Koyama et al., 2008; Schlee and Hartmann, 2010) However, TLR signaling also induces counter-regulatory molecules, including IL-10 (Saraiva and O’Garra, 2010). Components of HCV, CMV, EBV and LCMV all bind to TLR2 and TLR2 in turn can induce IL-10 expression via recruitment of the signaling adaptor MyD88 and activation of ERK pathways. (Ariza et al., 2009; Compton et al., 2003; Dolganiuc et al., 2006; Zhou et al., 2008). In humans, HIV glycoprotein binding to a mannose C-type lectin receptor (likely DC-SIGN) on the surface of monocyte derived-DC led to IL-10 expression (Shan et al., 2007). In addition to stimulating IL-10, HIV and LCMV infections also lead to dysregulated type-I interferon production promoting a suppressive environment and further dampening the antiviral response (Taylor et al., 1999; Zuniga et al., 2008). This is also true for HCV, where interaction of the core protein with TLR2 results not only in up-regulation of IL-10 expression but also decreased expression of type-I interferon by Kupffer cells and pDC (Dolganiuc et al., 2006; Tu et al., 2010). While a second HCV protein, NS3, concurrently up-regulated IL-10 and down-regulated IL-12 expression by macrophages and DC leading to diminished T cell stimulatory capacity in vitro (Dolganiuc et al., 2003; Eisen-Vandervelde et al., 2004). Therefore increased/prolonged levels of antigen may continue to trigger these same innate receptors throughout infection leading to sustained IL-10 expression while simultaneously down-modulating stimulatory factors and potentiating the immunosuppressive environment.
Continued viral infection also stimulates the de novo expression of factors that potentially modulate IL-10 expression. PD-1/PD-L1 interaction suppresses antiviral T cell activity during persistent virus infection, and has also been shown to increase IL-10 expression (Dong et al., 1999). A recent study (Said et al., 2010) demonstrated that peripheral blood monocytes from HIV viremic individuals express high levels of PD-1. Stimulation of PD-1 with antibody or PD-L1 transfected cells induced IL-10 expression capable of limiting CD4 T cell proliferation in vitro. These data demonstrate that PD-1 stimulation can activate IL-10 expression and suppression of antiviral immunity. Interestingly, during persistent LCMV infection we observed similar levels of IL-10 RNA expression in wild-type and PD-L1 KO mice (Brooks et al., 2008a), indicating that PD-L1 and IL-10 largely interact via different pathways. Functioning through different suppressive pathways was also consistent with the ability of dual IL-10 and PD-L1 blockade to additively increase exhausted T cell function compared to either IL-10R or PD-L1 blockade alone (Brooks et al., 2008a). The difference between these studies may relate to the fact that although most of the monocytes express PD-1, very few produced IL-10 upon PD-1 triggering. As a result, in PD-L1 KO mice the amount of IL-10 triggered by PD-1 on monocytes may not substantially impact the overall level of IL-10 expression. On the other hand, although only a small fraction of monocytes were stimulated to produce IL-10 by PD-1stimulation, these cells may be functionally distinct from other monocyte subsets and therefore, may have an enhanced ability to affect T cell immunity while not contributing significantly to global IL-10 production. Thus, it remains to be determined whether the population of IL-10 producing monocytes in vivo impact CD4 T cell responses similarly to that observed in vitro.
We and others also recently identified the important and progressive role of IL-21 in sustaining CD8 T cell responses during prolonged periods of virus replication (Elsaesser et al., 2009; Frohlich et al., 2009; Yi et al., 2009). For us, these experiments were initiated in our effort to define the mechanism(s) that induce IL-10 during LCMV persistence and based on the known role of IL-21 in stimulating IL-10 expression (Spolski et al., 2009). However, no change in IL-10 expression was observed in mice lacking IL-21R expression [(Frohlich et al., 2009) and D.G. Brooks, unpublished observation]. Further, we have not observed changes in IL-10 RNA or serum protein expression during persistent LCMV infection in mice deficient in factors that stimulate IL-10 in other models of disease (E. Wilson and D. Brooks, unpublished observations), including IL-27 and Galectin-1 (Ilarregui et al., 2009), TLR2 (Sing et al., 2002) and MyD88 (Boonstra et al., 2006) In total the discrepancy between IL-10 inducing factors in other disease models compared to persistent virus infection again indicates that multiple regulatory mechanisms can be instituted to suppress immunity in a pathogen/disease specific manner.
Another mechanism of IL-10 induction may not result specifically from alterations in factors produced, but instead changes in antigen presenting cell subsets. One of the defining characteristics of persistent LCMV variants is their ability to bind with high affinity to their cellular receptor α-dystroglycan enabling efficient infection of dendritic cells (Cao et al., 1998; Smelt et al., 2001). Due to infection, DC become targets for CTL lysis and the loss of DC was associated with the ensuing immunosuppression (Borrow et al., 1995; Sevilla et al., 2004). In particular, the CD8α+ DC subset is depleted during persistent LCMV infection and the remaining CD8α− DC were shown to increase IL-10 production by virus-specific CD4 T cells, which might in turn suppress antiviral CD8 T cell responses (Ejrnaes et al., 2006). The preferential killing of mature DC during HIV infection by NK cells in an IL-10 dependent fashion would similarly increase the frequency of immature DC and potentially augment T cell responses (Alter et al., 2010). Further, MCMV disruption of APC function leads to insufficient T cell activation, but the decreased levels of MHC may also prevent DC interaction with T cells, thereby effectively shifting the APC subsets that prime/sustain T cells. In reality, it is likely a culmination of all these events (and more yet to be discovered) that account for IL-10 mediated immune suppression to persistent virus infection. The elucidation of how IL-10 suppresses the immune response to facilitate persistence is actively being investigated in our laboratory and we anticipate the results of these studies will lead to important insight into how the host ‘senses’ the level of virus replication then progressively translates those signals into immune suppression.
Targets
Since IL-10 has been shown to attenuate numerous important biological functions in persistent virus infections it is likely that many relevant cellular targets of IL-10 exist. The sum of all the IL-10 induced events (in conjunction with those induced by other suppressive factors) act in concert to orchestrate immune suppression and facilitate viral persistence. Therapeutically, the targeting of multiple cell types by IL-10 means that alleviating IL-10 mediated immunosuppression would enhance several immune parameters compromised by persistent infection.
In HIV infected individuals, IL-10 produced by PBMC inhibits CD4 and CD8 T cell proliferation and cytokine production and blockade of IL-10 efficiently restores these functions in vitro (Brockman et al., 2009; Clerici et al., 1994; Landay et al., 1996; Said et al., 2010). Interaction of HIV with DC stimulates IL-10 production resulting in multiple functional defects (Alter et al., 2010; Shan et al., 2007). Interestingly, immature and mature DC respond differently to HIV-induced IL-10 upregulation. Immature DC exhibit an aberrant resistance to NK cell mediated cytolysis, whereas mature DC are targeted and destroyed by DC (Alter et al., 2010). This APC switch results in an over represented presence of ‘toleregenic’ DC during HIV infection that may fail to sustain T cell responses and/or ineffectively prime de novo T cell responses against evolving HIV mutants. There is also evidence that IL-10 augments B cell responses during HIV infection, inducing B cell exhaustion that may hinder antibody production (Moir et al., 2008). In some circumstances IL-10 is a positive regulator of CD8 T cells (Foulds et al., 2006; Groux et al., 1998; Kang and Allen, 2005; Santin et al., 2000). In these situations IL-2 is required for the stimulatory effect of IL-10 on CD8 T cells (Groux et al., 1998; Santin et al., 2000). However, IL-2 production by CD4 and CD8 T cells is rapidly lost during persistent infections (Brooks et al., 2005; Clerici et al., 1996; Klenerman and Hill, 2005; Petrovas et al., 2006; Semmo et al., 2005; Wherry et al., 2003; Younes et al., 2003). Thus, the presence of IL-10 without IL-2 may lead to suppressive instead of stimulatory CD8 T cell programming and highlights the important interplay between stimulatory and suppressive factors that fine tune the immune response to affect the outcome of infection.
We and others have clearly established the dominant role of IL-10 in facilitating LCMV persistence and based on its translatability to human persistent viral infections; it is likely that LCMV will be an important system to address IL-10 induced immunosuppression. IL-10 is produced by multiple APC subsets during persistent LCMV infection (Brooks et al., 2006b). Although priming of virus-specific CD4 and CD8 T cells is initially effective during persistent infection (Brooks et al., 2006a), the subsequent interactions with APC (i.e., occurring after the initial priming events) may attenuate ongoing T cell responses. CD4 T cell help is critical during persistent LCMV infection to sustain antiviral immune responses (Battegay et al., 1994; Matloubian et al., 1994). IL-10 directly targets CD4 T cells during an acute LCMV infection (Brooks et al., 2010) and similar diminution/alteration of the CD4 response by IL-10 during viral persistence may attenuate help, further exasperating the debilitated immune response. IL-10 may also be acting directly on virus-specific CD8 T cells, B cells and/or NK cells to attenuate their function and facilitate viral persistence. Identification of the relevant targets of IL-10 in vivo are currently underway and should yield important insight into the mechanisms that abort immune responses to facilitate viral persistence.
4. TARGETING IMMUNOSUPPRESSION: POTENTIAL THERAPEUTIC APPLICATIONS FOR BLOCKING IL-10
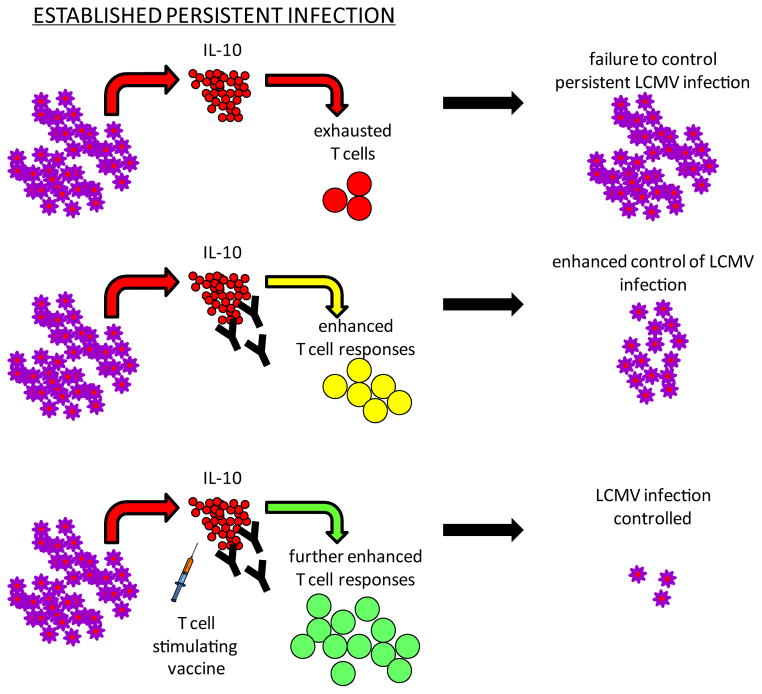
The initial finding that IL-10R blockade prevented T cell exhaustion and facilitated immune-mediated eradication of an otherwise persistent LCMV infection (Brooks et al., 2006b; Ejrnaes et al., 2006) was the first example that single factor could alone suppress antiviral immunity to prevent virus clearance. In addition to early blockade of IL-10 to prevent T cell exhaustion and LCMV persistence, late blockade of IL-10 activity also enhanced T cell responses leading to control of an established persistent infection (Brooks et al., 2008a; Brooks et al., 2008b). These findings indicate that IL-10 suppresses and can be targeted to restore antiviral immunity at multiple stages throughout persistent infection (Figure 2). Similarly, IL-10R blockade prevents MCMV persistence, although in the latter case the enhanced antiviral effects were accompanied by increased immunopathology (Brooks et al., 2006b; Campbell et al., 2008; Ejrnaes et al., 2006; Humphreys et al., 2007; Oakley et al., 2008).
Figure 2. Blocking IL-10 to enhance antiviral immunity and vaccine efficacy.
Overcoming IL-10-mediated immunosuppression represents an exciting strategy to enhance antiviral T cell responses both alone and in combination with other immunotherapies. Antibody blockade of IL-10 activity alone boosts T cell function and enhances control of an established persistent LCMV infection. Further, by alleviating IL-10 mediated suppression, virus-specific T cells become reactive to otherwise ineffective therapeutic vaccines facilitating markedly enhanced T cell responses and control of persistent infection. The upregulation of IL-10 during HIV and HCV infections suggests that similar strategies may also effectively enhance antiviral immunity and control infection.
Elevated serum levels of IL-10 are observed during many persistent virus infections in humans and similar to LCMV infection, correlates with diminished T cell activity and increased virus replication (Brady et al., 2003; Cacciarelli et al., 1996; Clerici et al., 1996; Dolganiuc et al., 2003; Marin-Serrano et al., 2006; Orsilles et al., 2006; Rico et al., 2001). Blockade of IL-10 in vitro restores function to HIV-specific and HCV-specific T cells (Cacciarelli et al., 1996; Clerici et al., 1994; Landay et al., 1996; Rigopoulou et al., 2005). Consistent with the correlation between HIV replication and IL-10 expression, antibody blockade of IL-10 only increased T cell function when cells were isolated from productively infected individuals and had minimal impact when cells originating from patients with effectively suppressed levels of HIV replication were analyzed (Brockman et al., 2009). However, in a separate study IL-10 blockade was shown to efficiently boost T cell responses even during effective anti-HIV therapy (in which HIV replication and IL-10 expression were low) (Yang et al., 2009). In total, these multiple studies demonstrate that blocking IL-10 during HIV infection can enhance antiviral T cell responses, but importantly, they indicate that IL-10 differentially affects different individuals and does so at distinct phases of infection.
The tight relationship between IL-10 and virus titers suggests that IL-10 may serve as a rheostat to constantly modulate immunity in relation to changing levels of virus replication. In agreement with this function, the experiments in which IL-10 was blocked to enhance HIV and HCV specific responses were performed after cell isolation techniques that eliminated in vivo produced IL-10 (Brockman et al., 2009; Rigopoulou et al., 2005). These findings indicate that de novo IL-10 production continually suppress T cell responses- T cells that are teetering on a fine line between exhaustion and productive immunity. Once function is diminished, blockade of no single factor restores T cell activity to that observed during an acute infection. However, therapies that alleviate some level of suppression appear to propel T cells across that fine line to better fight infection.
Recent research has clearly established that multiple negative immune-regulatory mechanisms are invoked to suppress T cell responses during viral persistence (Blackburn et al., 2009). We recently demonstrated that at least two of these factors (i.e., IL-10 and PD-L1) operate through distinct pathways to suppress immunity (Brooks et al., 2008a). As a result, dual antibody blockade of IL-10 and PD-L1 during LCMV persistence significantly enhanced antiviral T cell responses compared to blockade of either factor alone and rapidly controlled systemic viral replication. Similarly, simultaneous blockade of PD-L1 and another inhibitory receptor Lag-3, further enhanced T cell responses despite Lag-3 blockade alone having only minimal effect (Blackburn et al., 2009; Richter et al., 2010). This is particularly important because it indicates that there are layers of immunosuppression and that some negative regulatory pathways are dominant over others during persistent infection. However, once the dominant suppression is relieved other factors that appear to have limited function may become relevant and serve as targets to additionally enhance antiviral immunity.
Due to the varied lifecycles of persistent viruses, it will ultimately be important to determine how IL-10 blockade (as well as blockade of other negative regulatory factors) impacts control of different viral infections. HIV rapidly establishes a long-lived latent reservoir that is able to rekindle infection after prolonged periods of virus control (Chun et al., 1997; Finzi et al., 1997; Wong et al., 1997). During latent infection HIV remains hidden from immune recognition and as a result would not be targeted by therapies that amplify immune function (Brooks et al., 2003). However in cases like HCV infection where a latent viral reservoir is not established and therapeutic elimination of infection can be achieved, overcoming exhaustion and boosting immunity by blocking IL-10 activity could further control HCV replication. Thus, blockade of IL-10 (or other regulatory factors) may facilitate control (perhaps even long-term control) over HIV infection, but may not be able to completely eradicate infection because of immune-resistant latent reservoirs. On the other hand, a similar blockade of IL-10 during HCV infection may ultimately facilitate long term clearance of infection due to the absence of a long-lived latent reservoir.
IL-10 also regulates immunity to acute viral infections, limiting the magnitude of the ensuing response, the production of effector cytokines and consequently immunopathology, but generally without substantially impacting viral titers or clearance kinetics. Infection of mice with Influenza often leads to severe immunopathology, morbidity and mortality and IL-10 limits these negative effects (Sun et al., 2009). Following influenza infection of mice, a large population of lung infiltrating CD4 and particularly CD8 T cells produce IL-10 (Sun et al., 2009). Blockade of IL-10 enhanced IFNγ production leading to increased immunopathology and mortality without impacting virus clearance. These data suggest that in response to unknown cues, virus-specific CD8 T cells are capable of producing IL-10 to curb their own responses. IL-10 expression also restricted CD4 T cell responses during influenza resulting in decreased antibody titers; whereas, lack of IL-10 expression led to increased influenza specific antibody production and enhanced survival (Sun and Metzger, 2008; Sun et al., 2010). Interestingly, IL-10 mediated immunosuppression was linked to heightened susceptibility to secondary bacterial infection following influenza infection (van der Sluijs et al., 2004) although many factors likely contribute (Sun and Metzger, 2008). IL-10 is rapidly upregulated following acute LCMV-Armstrong infection and although not to the extent observed following persistent LCMV infection, it negatively regulated what is generally considered the ‘optimal’ antiviral immune response (Brooks et al., 2010). Interestingly, blockade of IL-10 activity directly enhanced both the quality and the quantity of virus-specific CD4 T cell responses without affecting virus-specific CD8 T cells, illustrating the differential regulation of T cell subsets following infection. Minor decreases in virus titers were observed in acute LCMV infected, IL-10 deficient mice, although both wild-type and IL-10 deficient mice cleared virus with a comparable kinetic. Excitingly, this promiscuity of IL-10 further substantiates the ability to restore many diverse effector mechanisms by blocking a single molecule. The increased expression and inhibitory activity of IL-10 following acute viral infection suggest that IL-10 blockade may be an effective adjuvant to prophylactic (i.e., preventative) vaccines to further enhance immunity (Brooks et al., 2010; Darrah et al., 2010). This would be particularly true for vaccines in which heightened CD4 T cell responses would be beneficial, such as HCV wherein the strength of the CD4 T cell response is an important determinant of clearance (Gerlach et al., 1999; Grakoui et al., 2003; Thimme et al., 2001; Urbani et al., 2006a).
Unlike prophylactic vaccines that aim to engender immune memory de novo, therapeutic vaccines (i.e., vaccines delivered during an established viral infection) must rebuild a debilitated immune response to now overcome the infection that it could not initially control. Along this line, vaccine agents that are immunogenic when administered to antigen naïve individuals often fail to efficiently stimulate immunity when provided prophylactically (Autran et al., 2004). Additionally, many prophylactic vaccines rely on stimulating antibody production, whereas therapeutic vaccines will likely have to restore/stimulate antiviral T cell responses as well as other immune parameters that are often refractory to further stimulation (Brooks et al., 2008b; Ha et al., 2008; Wherry et al., 2005; Zuniga et al., 2008). The finding that IL-10 actively inhibited T cell responses during persistent infection led us to hypothesize that one reason therapeutic vaccination strategies have thus far failed to resurrect/sustain T cell responses and control persistent infection is because they do not alleviate the immunosuppressive environment. Consequently, even if T cell responses could be restored they would rapidly again succumb to the same constraints that had previously limited their responsiveness. Consistent with this mechanism we demonstrated that antibody blockade of IL-10 during an established persistent viral infection permitted an otherwise ineffective DNA vaccine now highly efficient at stimulating CD4 and CD8 T cell responses leading to accelerated clearance of the persistent infection (Figure 2) (Brooks et al., 2008b). In conjunction, Rafi Ahmed’s group demonstrated that during persistent LCMV infection PD-L1 blockade similarly enhanced therapeutic vaccination with a live-replicating vaccine vector (Ha et al., 2008). Together, our findings established the immunosuppressive environment as an important factor inhibiting vaccination attempts to restore antiviral T cell function during persistent viral infection and suggested that blockade of negative immune regulatory molecules may ultimately prove a powerful strategy to aid therapeutic vaccination and purge an established persistent viral infection.
5. Past, Present and Future
The initial discovery of IL-10 as an inhibitor of Th1 differentiation has rapidly diversified such that now IL-10 is widely considered a ‘master-regulator’ of host immunity. The conserved nature of IL-10 mediated suppression among evolutionarily distinct species and the ability to boost immune function by blocking a single factor, despite the presence of other very powerful negative immune regulators, is quite astounding and speaks clearly to the significance of this pathway. However, many important discoveries remain to be made concerning IL-10 mediated suppression and how best to manipulate it for therapeutic benefit. The promiscuity of IL-10 production and function suggests that its blockade could amplify multiple antiviral mechanisms to control persistent virus replication. By understanding how IL-10 regulates distinct components of the immune response it may be possible to block IL-10 production by or function on certain cells and unleash antiviral T cells while maintaining regulation of those cells prevent immunopathology. Ultimately, blockade of immune-regulatory factors holds great promise as an approach to restore immunity and purge established persistent viral infections. The ability of IL-10 and other inhibitory factors to operate at distinct levels of immune function and on different cell subsets indicates the possibility of combinatorial blockade cocktails to specifically enhance desired immune cell subsets and evoke different immune responses; thus, paving the way into an age of rationale vaccine design.
Acknowledgments
Our work was supported by the UCLA Center for AIDS Research, the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA, the Johanna and Joseph Shaper Family Chair and grants from the National Institutes of Health (AI082975, AI085043 to D.G.B.).
References
- Agnellini P, Wolint P, Rehr M, Cahenzli J, Karrer U, Oxenius A. Impaired NFAT nuclear translocation results in split exhaustion of virus-specific CD8+ T cell functions during chronic viral infection. Proc Natl Acad Sci U S A. 2007;104:4565–4570. doi: 10.1073/pnas.0610335104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed R, Salmi A, Butler LD, Chiller JM, Oldstone MB. Selection of genetic variants of lymphocytic choriomeningitis virus in spleens of persistently infected mice. Role in suppression of cytotoxic T lymphocyte response and viral persistence. J Exp Med. 1984;160:521–540. doi: 10.1084/jem.160.2.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alter G, Kavanagh D, Rihn S, Luteijn R, Brooks D, Oldstone M, van Lunzen J, Altfeld M. IL-10 induces aberrant deletion of dendritic cells by natural killer cells in the context of HIV infection. J Clin Invest. 2010 doi: 10.1172/JCI40913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews DM, Andoniou CE, Granucci F, Ricciardi-Castagnoli P, Degli-Esposti MA. Infection of dendritic cells by murine cytomegalovirus induces functional paralysis. Nat Immunol. 2001;2:1077–1084. doi: 10.1038/ni724. [DOI] [PubMed] [Google Scholar]
- Ariza ME, Glaser R, Kaumaya PT, Jones C, Williams MV. The EBV-encoded dUTPase activates NF-kappa B through the TLR2 and MyD88-dependent signaling pathway. J Immunol. 2009;182:851–859. doi: 10.4049/jimmunol.182.2.851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Autran B, Carcelain G, Combadiere B, Debre P. Therapeutic vaccines for chronic infections. Science. 2004;305:205–208. doi: 10.1126/science.1100600. [DOI] [PubMed] [Google Scholar]
- Bachmann MF, Wolint P, Walton S, Schwarz K, Oxenius A. Differential role of IL-2R signaling for CD8+ T cell responses in acute and chronic viral infections. Eur J Immunol. 2007;37:1502–1512. doi: 10.1002/eji.200637023. [DOI] [PubMed] [Google Scholar]
- Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, Ahmed R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2005;439:682–687. doi: 10.1038/nature04444. [DOI] [PubMed] [Google Scholar]
- Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, Ahmed R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006;439:682–687. doi: 10.1038/nature04444. [DOI] [PubMed] [Google Scholar]
- Battegay M, Moskophidis D, Rahemtulla A, Hengartner H, Mak TW, Zinkernagel RM. Enhanced establishment of a virus carrier state in adult CD4+ T-cell-deficient mice. J Virol. 1994;68:4700–4704. doi: 10.1128/jvi.68.7.4700-4704.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belkaid Y, Tarbell K. Regulatory T cells in the control of host-microorganism interactions (*) Annu Rev Immunol. 2009;27:551–589. doi: 10.1146/annurev.immunol.021908.132723. [DOI] [PubMed] [Google Scholar]
- Biron CA, Nguyen KB, Pien GC, Cousens LP, Salazar-Mather TP. Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu Rev Immunol. 1999;17:189–220. doi: 10.1146/annurev.immunol.17.1.189. [DOI] [PubMed] [Google Scholar]
- Blackburn SD, Shin H, Haining WN, Zou T, Workman CJ, Polley A, Betts MR, Freeman GJ, Vignali DA, Wherry EJ. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat Immunol. 2009;10:29–37. doi: 10.1038/ni.1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boni C, Fisicaro P, Valdatta C, Amadei B, Di Vincenzo P, Giuberti T, Laccabue D, Zerbini A, Cavalli A, Missale G, et al. Characterization of hepatitis B virus (HBV)-specific T-cell dysfunction in chronic HBV infection. J Virol. 2007;81:4215–4225. doi: 10.1128/JVI.02844-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boonstra A, Rajsbaum R, Holman M, Marques R, Asselin-Paturel C, Pereira JP, Bates EE, Akira S, Vieira P, Liu YJ, et al. Macrophages and myeloid dendritic cells, but not plasmacytoid dendritic cells, produce IL-10 in response to MyD88- and TRIF-dependent TLR signals, and TLR-independent signals. J Immunol. 2006;177:7551–7558. doi: 10.4049/jimmunol.177.11.7551. [DOI] [PubMed] [Google Scholar]
- Borrow P, Evans CF, Oldstone MB. Virus-induced immunosuppression: immune system-mediated destruction of virus-infected dendritic cells results in generalized immune suppression. J Virol. 1995;69:1059–1070. doi: 10.1128/jvi.69.2.1059-1070.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brady MT, MacDonald AJ, Rowan AG, Mills KH. Hepatitis C virus non-structural protein 4 suppresses Th1 responses by stimulating IL-10 production from monocytes. Eur J Immunol. 2003;33:3448–3457. doi: 10.1002/eji.200324251. [DOI] [PubMed] [Google Scholar]
- Brockman MA, Kwon DS, Tighe DP, Pavlik DF, Rosato PC, Sela J, Porichis F, Le Gall S, Waring MT, Moss K, et al. IL-10 is upregulated in multiple cell types during viremic HIV infection and reversibly inhibits virus-specific T cells. Blood. 2009 doi: 10.1182/blood-2008-12-191296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks DG, Ha SJ, Elsaesser H, Sharpe AH, Freeman GJ, Oldstone MB. IL-10 and PD-L1 operate through distinct pathways to suppress T-cell activity during persistent viral infection. Proc Natl Acad Sci U S A. 2008a;105:20428–20433. doi: 10.1073/pnas.0811139106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks DG, Hamer DH, Arlen PA, Gao L, Bristol G, Kitchen CM, Berger EA, Zack JA. Molecular characterization, reactivation, and depletion of latent HIV. Immunity. 2003;19:413–423. doi: 10.1016/s1074-7613(03)00236-x. [DOI] [PubMed] [Google Scholar]
- Brooks DG, Lee AM, Elsaesser H, McGavern DB, Oldstone MB. IL-10 blockade facilitates DNA vaccine-induced T cell responses and enhances clearance of persistent virus infection. J Exp Med. 2008b;205:533–541. doi: 10.1084/jem.20071948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks DG, McGavern DB, Oldstone MB. Reprogramming of antiviral T cells prevents inactivation and restores T cell activity during persistent viral infection. J Clin Invest. 2006a;116:1675–1685. doi: 10.1172/JCI26856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks DG, Teyton L, Oldstone MB, McGavern DB. Intrinsic functional dysregulation of CD4 T cells occurs rapidly following persistent viral infection. Journal of Virology. 2005;79:10514–10527. doi: 10.1128/JVI.79.16.10514-10527.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks DG, Trifilo MJ, Edelmann KH, Teyton L, McGavern DB, Oldstone MB. Interleukin-10 determines viral clearance or persistence in vivo. Nat Med. 2006b;12:1301–1309. doi: 10.1038/nm1492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooks DG, Walsh KB, Elsaesser H, Oldstone MB. IL-10 directly suppresses CD4 but not CD8 T cell effector and memory responses following acute viral infection. Proc Natl Acad Sci U S A. 2010;107:3018–3023. doi: 10.1073/pnas.0914500107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cacciarelli TV, Martinez OM, Gish RG, Villanueva JC, Krams SM. Immunoregulatory cytokines in chronic hepatitis C virus infection: pre- and posttreatment with interferon alfa. Hepatology. 1996;24:6–9. doi: 10.1002/hep.510240102. [DOI] [PubMed] [Google Scholar]
- Campbell AE, Cavanaugh VJ, Slater JS. The salivary glands as a privileged site of cytomegalovirus immune evasion and persistence. Med Microbiol Immunol. 2008;197:205–213. doi: 10.1007/s00430-008-0077-2. [DOI] [PubMed] [Google Scholar]
- Cao W, Henry MD, Borrow P, Yamada H, Elder JH, Ravkov EV, Nichol ST, Compans RW, Campbell KP, Oldstone MB. Identification of alpha-dystroglycan as a receptor for lymphocytic choriomeningitis virus and Lassa fever virus. Science. 1998;282:2079–2081. doi: 10.1126/science.282.5396.2079. [DOI] [PubMed] [Google Scholar]
- Carbonneil C, Donkova-Petrini V, Aouba A, Weiss L. Defective dendritic cell function in HIV-infected patients receiving effective highly active antiretroviral therapy: neutralization of IL-10 production and depletion of CD4+CD25+ T cells restore high levels of HIV-specific CD4+ T cell responses induced by dendritic cells generated in the presence of IFN-alpha. J Immunol. 2004;172:7832–7840. doi: 10.4049/jimmunol.172.12.7832. [DOI] [PubMed] [Google Scholar]
- Chang WL, Barry PA, Szubin R, Wang D, Baumgarth N. Human cytomegalovirus suppresses type I interferon secretion by plasmacytoid dendritic cells through its interleukin 10 homolog. Virology. 2009;390:330–337. doi: 10.1016/j.virol.2009.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang WL, Baumgarth N, Yu D, Barry PA. Human cytomegalovirus-encoded interleukin-10 homolog inhibits maturation of dendritic cells and alters their functionality. J Virol. 2004;78:8720–8731. doi: 10.1128/JVI.78.16.8720-8731.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheong JY, Cho SW, Hwang IL, Yoon SK, Lee JH, Park CS, Lee JE, Hahm KB, Kim JH. Association between chronic hepatitis B virus infection and interleukin-10, tumor necrosis factor-alpha gene promoter polymorphisms. J Gastroenterol Hepatol. 2006;21:1163–1169. doi: 10.1111/j.1440-1746.2006.04304.x. [DOI] [PubMed] [Google Scholar]
- Cheung AK, Gottlieb DJ, Plachter B, Pepperl-Klindworth S, Avdic S, Cunningham AL, Abendroth A, Slobedman B. The role of the human cytomegalovirus UL111A gene in down-regulating CD4+ T-cell recognition of latently infected cells: implications for virus elimination during latency. Blood. 2009;114:4128–4137. doi: 10.1182/blood-2008-12-197111. [DOI] [PubMed] [Google Scholar]
- Chun TW, Stuyver L, Mizell SB, Ehler LA, Mican JA, Baseler M, Lloyd AL, Nowak MA, Fauci AS. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc Natl Acad Sci U S A. 1997;94:13193–13197. doi: 10.1073/pnas.94.24.13193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clerici M, Balotta C, Salvaggio A, Riva C, Trabattoni D, Papagno L, Berlusconi A, Rusconi S, Villa ML, Moroni M, Galli M. Human immunodeficiency virus (HIV) phenotype and interleukin-2/interleukin-10 ratio are associated markers of protection and progression in HIV infection. Blood. 1996;88:574–579. [PubMed] [Google Scholar]
- Clerici M, Wynn TA, Berzofsky JA, Blatt SP, Hendrix CW, Sher A, Coffman RL, Shearer GM. Role of interleukin-10 in T helper cell dysfunction in asymptomatic individuals infected with the human immunodeficiency virus. J Clin Invest. 1994;93:768–775. doi: 10.1172/JCI117031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Compton T, Kurt-Jones EA, Boehme KW, Belko J, Latz E, Golenbock DT, Finberg RW. Human cytomegalovirus activates inflammatory cytokine responses via CD14 and Toll-like receptor 2. J Virol. 2003;77:4588–4596. doi: 10.1128/JVI.77.8.4588-4596.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Constant SL, Bottomly K. Induction of Th1 and Th2 CD4+ T cell responses: the alternative approaches. Annu Rev Immunol. 1997;15:297–322. doi: 10.1146/annurev.immunol.15.1.297. [DOI] [PubMed] [Google Scholar]
- Couper KN, Blount DG, Riley EM. IL-10: the master regulator of immunity to infection. J Immunol. 2008;180:5771–5777. doi: 10.4049/jimmunol.180.9.5771. [DOI] [PubMed] [Google Scholar]
- Darrah PA, Hegde ST, Patel DT, Lindsay RW, Chen L, Roederer M, Seder RA. IL-10 production differentially influences the magnitude, quality, and protective capacity of Th1 responses depending on the vaccine platform. J Exp Med. 2010 doi: 10.1084/jem.20092532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson NJ, Leach MW, Fort MM, Thompson-Snipes L, Kuhn R, Muller W, Berg DJ, Rennick DM. T helper cell 1-type CD4+ T cells, but not B cells, mediate colitis in interleukin 10-deficient mice. J Exp Med. 1996;184:241–251. doi: 10.1084/jem.184.1.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day CL, Kaufmann DE, Kiepiela P, Brown JA, Moodley ES, Reddy S, Mackey EW, Miller JD, Leslie AJ, DePierres C, et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature. 2006;443:350–354. doi: 10.1038/nature05115. [DOI] [PubMed] [Google Scholar]
- Dittmer U, He H, Messer RJ, Schimmer S, Olbrich AR, Ohlen C, Greenberg PD, Stromnes IM, Iwashiro M, Sakaguchi S, et al. Functional impairment of CD8(+) T cells by regulatory T cells during persistent retroviral infection. Immunity. 2004;20:293–303. doi: 10.1016/s1074-7613(04)00054-8. [DOI] [PubMed] [Google Scholar]
- Dittmer U, Race B, Peterson KE, Stromnes IM, Messer RJ, Hasenkrug KJ. Essential roles for CD8+ T cells and gamma interferon in protection of mice against retrovirus-induced immunosuppression. J Virol. 2002;76:450–454. doi: 10.1128/JVI.76.1.450-454.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolganiuc A, Chang S, Kodys K, Mandrekar P, Bakis G, Cormier M, Szabo G. Hepatitis C virus (HCV) core protein-induced, monocyte-mediated mechanisms of reduced IFN-alpha and plasmacytoid dendritic cell loss in chronic HCV infection. J Immunol. 2006;177:6758–6768. doi: 10.4049/jimmunol.177.10.6758. [DOI] [PubMed] [Google Scholar]
- Dolganiuc A, Kodys K, Kopasz A, Marshall C, Do T, Romics L, Jr, Mandrekar P, Zapp M, Szabo G. Hepatitis C virus core and nonstructural protein 3 proteins induce pro- and anti-inflammatory cytokines and inhibit dendritic cell differentiation. J Immunol. 2003;170:5615–5624. doi: 10.4049/jimmunol.170.11.5615. [DOI] [PubMed] [Google Scholar]
- Dong H, Zhu G, Tamada K, Chen L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med. 1999;5:1365–1369. doi: 10.1038/70932. [DOI] [PubMed] [Google Scholar]
- Donnelly RP, Sheikh F, Kotenko SV, Dickensheets H. The expanded family of class II cytokines that share the IL-10 receptor-2 (IL-10R2) chain. J Leukoc Biol. 2004;76:314–321. doi: 10.1189/jlb.0204117. [DOI] [PubMed] [Google Scholar]
- Eisen-Vandervelde AL, Waggoner SN, Yao ZQ, Cale EM, Hahn CS, Hahn YS. Hepatitis C virus core selectively suppresses interleukin-12 synthesis in human macrophages by interfering with AP-1 activation. J Biol Chem. 2004;279:43479–43486. doi: 10.1074/jbc.M407640200. [DOI] [PubMed] [Google Scholar]
- Ejrnaes M, Filippi CM, Martinic MM, Ling EM, Togher LM, Crotty S, von Herrath MG. Resolution of a chronic viral infection after interleukin-10 receptor blockade. J Exp Med. 2006;203:2461–2472. doi: 10.1084/jem.20061462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elsaesser H, Sauer K, Brooks DG. IL-21 is required to control chronic viral infection. Science. 2009;324:1569–1572. doi: 10.1126/science.1174182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fahey LM, Brooks DG. Opposing positive and negative regulation of T cell activity during viral persistence. Curr Opin Immunol. 2010;22:348–354. doi: 10.1016/j.coi.2010.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finzi D, Hermankova M, Pierson T, Carruth LM, Buck C, Chaisson RE, Quinn TC, Chadwick K, Margolick J, Brookmeyer R, et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science. 1997;278:1295–1300. doi: 10.1126/science.278.5341.1295. [DOI] [PubMed] [Google Scholar]
- Fiorentino DF, Bond MW, Mosmann TR. Two types of mouse T helper cell. IV. Th2 clones secrete a factor that inhibits cytokine production by Th1 clones. J Exp Med. 1989;170:2081–2095. doi: 10.1084/jem.170.6.2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiorentino DF, Zlotnik A, Vieira P, Mosmann TR, Howard M, Moore KW, O’Garra A. IL-10 acts on the antigen-presenting cell to inhibit cytokine production by Th1 cells. J Immunol. 1991;146:3444–3451. [PubMed] [Google Scholar]
- Foulds KE, Rotte MJ, Seder RA. IL-10 is required for optimal CD8 T cell memory following Listeria monocytogenes infection. J Immunol. 2006;177:2565–2574. doi: 10.4049/jimmunol.177.4.2565. [DOI] [PubMed] [Google Scholar]
- Frohlich A, Kisielow J, Schmitz I, Freigang S, Shamshiev AT, Weber J, Marsland BJ, Oxenius A, Kopf M. IL-21R on T cells is critical for sustained functionality and control of chronic viral infection. Science. 2009;324:1576–1580. doi: 10.1126/science.1172815. [DOI] [PubMed] [Google Scholar]
- Gallimore A, Glithero A, Godkin A, Tissot AC, Pluckthun A, Elliott T, Hengartner H, Zinkernagel R. Induction and exhaustion of lymphocytic choriomeningitis virus-specific cytotoxic T lymphocytes visualized using soluble tetrameric major histocompatibility complex class I-peptide complexes. J Exp Med. 1998;187:1383–1393. doi: 10.1084/jem.187.9.1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gartel AL, Serfas MS, Tyner AL. p21--negative regulator of the cell cycle. Proc Soc Exp Biol Med. 1996;213:138–149. doi: 10.3181/00379727-213-44046. [DOI] [PubMed] [Google Scholar]
- Gerlach JT, Diepolder HM, Jung MC, Gruener NH, Schraut WW, Zachoval R, Hoffmann R, Schirren CA, Santantonio T, Pape GR. Recurrence of hepatitis C virus after loss of virus-specific CD4(+) T-cell response in acute hepatitis C. Gastroenterology. 1999;117:933–941. doi: 10.1016/s0016-5085(99)70353-7. [DOI] [PubMed] [Google Scholar]
- Goodbourn S, Didcock L, Randall RE. Interferons: cell signalling, immune modulation, antiviral response and virus countermeasures. J Gen Virol. 2000;81:2341–2364. doi: 10.1099/0022-1317-81-10-2341. [DOI] [PubMed] [Google Scholar]
- Grakoui A, Shoukry NH, Woollard DJ, Han JH, Hanson HL, Ghrayeb J, Murthy KK, Rice CM, Walker CM. HCV persistence and immune evasion in the absence of memory T cell help. Science. 2003;302:659–662. doi: 10.1126/science.1088774. [DOI] [PubMed] [Google Scholar]
- Groux H, Bigler M, de Vries JE, Roncarolo MG. Interleukin-10 induces a long-term antigen-specific anergic state in human CD4+ T cells. J Exp Med. 1996;184:19–29. doi: 10.1084/jem.184.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groux H, Bigler M, de Vries JE, Roncarolo MG. Inhibitory and stimulatory effects of IL-10 on human CD8+ T cells. J Immunol. 1998;160:3188–3193. [PubMed] [Google Scholar]
- Groux H, O’Garra A, Bigler M, Rouleau M, Antonenko S, de Vries JE, Roncarolo MG. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature. 1997;389:737–742. doi: 10.1038/39614. [DOI] [PubMed] [Google Scholar]
- Ha SJ, Mueller SN, Wherry EJ, Barber DL, Aubert RD, Sharpe AH, Freeman GJ, Ahmed R. Enhancing therapeutic vaccination by blocking PD-1-mediated inhibitory signals during chronic infection. J Exp Med. 2008;205:543–555. doi: 10.1084/jem.20071949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagiwara E, Abbasi F, Mor G, Ishigatsubo Y, Klinman DM. Phenotype and frequency of cells secreting IL-2, IL-4, IL-6, IL-10, IFN and TNF-alpha in human peripheral blood. Cytokine. 1995;7:815–822. doi: 10.1006/cyto.1995.0098. [DOI] [PubMed] [Google Scholar]
- Hedrich CM, Bream JH. Cell type-specific regulation of IL-10 expression in inflammation and disease. Immunol Res. 2010 doi: 10.1007/s12026-009-8150-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helminen M, Lahdenpohja N, Hurme M. Polymorphism of the interleukin-10 gene is associated with susceptibility to Epstein-Barr virus infection. J Infect Dis. 1999;180:496–499. doi: 10.1086/314883. [DOI] [PubMed] [Google Scholar]
- Humphreys IR, de Trez C, Kinkade A, Benedict CA, Croft M, Ware CF. Cytomegalovirus exploits IL-10-mediated immune regulation in the salivary glands. J Exp Med. 2007;204:1217–1225. doi: 10.1084/jem.20062424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ilarregui JM, Croci DO, Bianco GA, Toscano MA, Salatino M, Vermeulen ME, Geffner JR, Rabinovich GA. Tolerogenic signals delivered by dendritic cells to T cells through a galectin-1-driven immunoregulatory circuit involving interleukin 27 and interleukin 10. Nat Immunol. 2009;10:981–991. doi: 10.1038/ni.1772. [DOI] [PubMed] [Google Scholar]
- Jenkins C, Abendroth A, Slobedman B. A novel viral transcript with homology to human interleukin-10 is expressed during latent human cytomegalovirus infection. J Virol. 2004;78:1440–1447. doi: 10.1128/JVI.78.3.1440-1447.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkins C, Garcia W, Godwin MJ, Spencer JV, Stern JL, Abendroth A, Slobedman B. Immunomodulatory properties of a viral homolog of human interleukin-10 expressed by human cytomegalovirus during the latent phase of infection. J Virol. 2008;82:3736–3750. doi: 10.1128/JVI.02173-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones RB, Ndhlovu LC, Barbour JD, Sheth PM, Jha AR, Long BR, Wong JC, Satkunarajah M, Schweneker M, Chapman JM, et al. Tim-3 expression defines a novel population of dysfunctional T cells with highly elevated frequencies in progressive HIV-1 infection. J Exp Med. 2008;205:2763–2779. doi: 10.1084/jem.20081398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaech SM, Hemby S, Kersh E, Ahmed R. Molecular and functional profiling of memory CD8 T cell differentiation. Cell. 2002;111:837–851. doi: 10.1016/s0092-8674(02)01139-x. [DOI] [PubMed] [Google Scholar]
- Kang SS, Allen PM. Priming in the presence of IL-10 results in direct enhancement of CD8+ T cell primary responses and inhibition of secondary responses. J Immunol. 2005;174:5382–5389. doi: 10.4049/jimmunol.174.9.5382. [DOI] [PubMed] [Google Scholar]
- Kaufmann DE, Kavanagh DG, Pereyra F, Zaunders JJ, Mackey EW, Miura T, Palmer S, Brockman M, Rathod A, Piechocka-Trocha A, et al. Upregulation of CTLA-4 by HIV-specific CD4+ T cells correlates with disease progression and defines a reversible immune dysfunction. Nat Immunol. 2007;8:1246–1254. doi: 10.1038/ni1515. [DOI] [PubMed] [Google Scholar]
- Klenerman P, Hill A. T cells and viral persistence: lessons from diverse infections. Nat Immunol. 2005;6:873–879. doi: 10.1038/ni1241. [DOI] [PubMed] [Google Scholar]
- Kotenko SV, Krause CD, Izotova LS, Pollack BP, Wu W, Pestka S. Identification and functional characterization of a second chain of the interleukin-10 receptor complex. Embo J. 1997;16:5894–5903. doi: 10.1093/emboj/16.19.5894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kotenko SV, Saccani S, Izotova LS, Mirochnitchenko OV, Pestka S. Human cytomegalovirus harbors its own unique IL-10 homolog (cmvIL-10) Proc Natl Acad Sci U S A. 2000;97:1695–1700. doi: 10.1073/pnas.97.4.1695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koyama S, Ishii KJ, Coban C, Akira S. Innate immune response to viral infection. Cytokine. 2008;43:336–341. doi: 10.1016/j.cyto.2008.07.009. [DOI] [PubMed] [Google Scholar]
- Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–274. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- Kumar A, Angel JB, Daftarian MP, Parato K, Cameron WD, Filion L, Diaz-Mitoma F. Differential production of IL-10 by T cells and monocytes of HIV-infected individuals: association of IL-10 production with CD28-mediated immune responsiveness. Clin Exp Immunol. 1998;114:78–86. doi: 10.1046/j.1365-2249.1998.00689.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landay AL, Clerici M, Hashemi F, Kessler H, Berzofsky JA, Shearer GM. In vitro restoration of T cell immune function in human immunodeficiency virus-positive persons: effects of interleukin (IL)-12 and anti-IL-10. J Infect Dis. 1996;173:1085–1091. doi: 10.1093/infdis/173.5.1085. [DOI] [PubMed] [Google Scholar]
- Lin MT, Hinton DR, Parra B, Stohlman SA, van der Veen RC. The role of IL-10 in mouse hepatitis virus-induced demyelinating encephalomyelitis. Virology. 1998;245:270–280. doi: 10.1006/viro.1998.9170. [DOI] [PubMed] [Google Scholar]
- Liu Y, de Waal Malefyt R, Briere F, Parham C, Bridon JM, Banchereau J, Moore KW, Xu J. The EBV IL-10 homologue is a selective agonist with impaired binding to the IL-10 receptor. J Immunol. 1997;158:604–613. [PubMed] [Google Scholar]
- Liu Y, Wei SH, Ho AS, de Waal Malefyt R, Moore KW. Expression cloning and characterization of a human IL-10 receptor. J Immunol. 1994;152:1821–1829. [PubMed] [Google Scholar]
- Lockridge KM, Zhou SS, Kravitz RH, Johnson JL, Sawai ET, Blewett EL, Barry PA. Primate cytomegaloviruses encode and express an IL-10-like protein. Virology. 2000;268:272–280. doi: 10.1006/viro.2000.0195. [DOI] [PubMed] [Google Scholar]
- Madan R, Demircik F, Surianarayanan S, Allen JL, Divanovic S, Trompette A, Yogev N, Gu Y, Khodoun M, Hildeman D, et al. Nonredundant roles for B cell-derived IL-10 in immune counter-regulation. J Immunol. 2009;183:2312–2320. doi: 10.4049/jimmunol.0900185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marin-Serrano E, Rodriguez-Ramos C, Diaz F, Martin-Herrera L, Giron-Gonzalez JA. Modulation of the anti-inflammatory interleukin 10 and of proapoptotic IL-18 in patients with chronic hepatitis C treated with interferon alpha and ribavirin. J Viral Hepat. 2006;13:230–234. doi: 10.1111/j.1365-2893.2005.00679.x. [DOI] [PubMed] [Google Scholar]
- Matloubian M, Concepcion RJ, Ahmed R. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J Virol. 1994;68:8056–8063. doi: 10.1128/jvi.68.12.8056-8063.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matter M, Odermatt B, Yagita H, Nuoffer JM, Ochsenbein AF. Elimination of chronic viral infection by blocking CD27 signaling. J Exp Med. 2006;203:2145–2155. doi: 10.1084/jem.20060651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maynard CL, Weaver CT. Diversity in the contribution of interleukin-10 to T-cell-mediated immune regulation. Immunol Rev. 2008;226:219–233. doi: 10.1111/j.1600-065X.2008.00711.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mege JL, Meghari S, Honstettre A, Capo C, Raoult D. The two faces of interleukin 10 in human infectious diseases. Lancet Infect Dis. 2006;6:557–569. doi: 10.1016/S1473-3099(06)70577-1. [DOI] [PubMed] [Google Scholar]
- Moir S, Ho J, Malaspina A, Wang W, DiPoto AC, O’Shea MA, Roby G, Kottilil S, Arthos J, Proschan MA, et al. Evidence for HIV-associated B cell exhaustion in a dysfunctional memory B cell compartment in HIV-infected viremic individuals. J Exp Med. 2008;205:1797–1805. doi: 10.1084/jem.20072683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore KW, de Waal Malefyt R, Coffman RL, O’Garra A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765. doi: 10.1146/annurev.immunol.19.1.683. [DOI] [PubMed] [Google Scholar]
- Moore KW, Vieira P, Fiorentino DF, Trounstine ML, Khan TA, Mosmann TR. Homology of cytokine synthesis inhibitory factor (IL-10) to the Epstein-Barr virus gene BCRFI. Science. 1990;248:1230–1234. doi: 10.1126/science.2161559. [DOI] [PubMed] [Google Scholar]
- Oakley OR, Garvy BA, Humphreys S, Qureshi MH, Pomeroy C. Increased weight loss with reduced viral replication in interleukin-10 knock-out mice infected with murine cytomegalovirus. Clin Exp Immunol. 2008;151:155–164. doi: 10.1111/j.1365-2249.2007.03533.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orsilles MA, Pieri E, Cooke P, Caula C. IL-2 and IL-10 serum levels in HIV-1-infected patients with or without active antiretroviral therapy. Apmis. 2006;114:55–60. doi: 10.1111/j.1600-0463.2006.apm_108.x. [DOI] [PubMed] [Google Scholar]
- Paladino N, Fainboim H, Theiler G, Schroder T, Munoz AE, Flores AC, Galdame O, Fainboim L. Gender susceptibility to chronic hepatitis C virus infection associated with interleukin 10 promoter polymorphism. J Virol. 2006;80:9144–9150. doi: 10.1128/JVI.00339-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrovas C, Casazza JP, Brenchley JM, Price DA, Gostick E, Adams WC, Precopio ML, Schacker T, Roederer M, Douek DC, Koup RA. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J Exp Med. 2006;203:2281–2292. doi: 10.1084/jem.20061496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raftery MJ, Wieland D, Gronewald S, Kraus AA, Giese T, Schonrich G. Shaping phenotype, function, and survival of dendritic cells by cytomegalovirus-encoded IL-10. J Immunol. 2004;173:3383–3391. doi: 10.4049/jimmunol.173.5.3383. [DOI] [PubMed] [Google Scholar]
- Richter K, Agnellini P, Oxenius A. On the role of the inhibitory receptor LAG-3 in acute and chronic LCMV infection. Int Immunol. 2010;22:13–23. doi: 10.1093/intimm/dxp107. [DOI] [PubMed] [Google Scholar]
- Rico MA, Quiroga JA, Subira D, Castanon S, Esteban JM, Pardo M, Carreno V. Hepatitis B virus-specific T-cell proliferation and cytokine secretion in chronic hepatitis B e antibody-positive patients treated with ribavirin and interferon alpha. Hepatology. 2001;33:295–300. doi: 10.1053/jhep.2001.21147. [DOI] [PubMed] [Google Scholar]
- Rigopoulou EI, Abbott WG, Haigh P, Naoumov NV. Blocking of interleukin-10 receptor--a novel approach to stimulate T-helper cell type 1 responses to hepatitis C virus. Clin Immunol. 2005;117:57–64. doi: 10.1016/j.clim.2005.06.003. [DOI] [PubMed] [Google Scholar]
- Rouse BT, Sarangi PP, Suvas S. Regulatory T cells in virus infections. Immunol Rev. 2006;212:272–286. doi: 10.1111/j.0105-2896.2006.00412.x. [DOI] [PubMed] [Google Scholar]
- Said EA, Dupuy FP, Trautmann L, Zhang Y, Shi Y, El-Far M, Hill BJ, Noto A, Ancuta P, Peretz Y, et al. Programmed death-1-induced interleukin-10 production by monocytes impairs CD4+ T cell activation during HIV infection. Nat Med. 2010;16:452–459. doi: 10.1038/nm.2106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santin AD, Hermonat PL, Ravaggi A, Bellone S, Pecorelli S, Roman JJ, Parham GP, Cannon MJ. Interleukin-10 increases Th1 cytokine production and cytotoxic potential in human papillomavirus-specific CD8(+) cytotoxic T lymphocytes. J Virol. 2000;74:4729–4737. doi: 10.1128/jvi.74.10.4729-4737.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraiva M, O’Garra A. The regulation of IL-10 production by immune cells. Nat Rev Immunol. 2010;10:170–181. doi: 10.1038/nri2711. [DOI] [PubMed] [Google Scholar]
- Schlee M, Hartmann G. The Chase for the RIG-I Ligand-Recent Advances. Mol Ther. 2010 doi: 10.1038/mt.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sellon RK, Tonkonogy S, Schultz M, Dieleman LA, Grenther W, Balish E, Rennick DM, Sartor RB. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect Immun. 1998;66:5224–5231. doi: 10.1128/iai.66.11.5224-5231.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semmo N, Day CL, Ward SM, Lucas M, Harcourt G, Loughry A, Klenerman P. Preferential loss of IL-2-secreting CD4+ T helper cells in chronic HCV infection. Hepatology. 2005;41:1019–1028. doi: 10.1002/hep.20669. [DOI] [PubMed] [Google Scholar]
- Sevilla N, McGavern DB, Teng C, Kunz S, Oldstone MB. Viral targeting of hematopoietic progenitors and inhibition of DC maturation as a dual strategy for immune subversion. J Clin Invest. 2004;113:737–745. doi: 10.1172/JCI20243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shan M, Klasse PJ, Banerjee K, Dey AK, Iyer SP, Dionisio R, Charles D, Campbell-Gardener L, Olson WC, Sanders RW, Moore JP. HIV-1 gp120 mannoses induce immunosuppressive responses from dendritic cells. PLoS Pathog. 2007;3:e169. doi: 10.1371/journal.ppat.0030169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin H, Blackburn SD, Intlekofer AM, Kao C, Angelosanto JM, Reiner SL, Wherry EJ. A role for the transcriptional repressor Blimp-1 in CD8(+) T cell exhaustion during chronic viral infection. Immunity. 2009;31:309–320. doi: 10.1016/j.immuni.2009.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin HD, Park BL, Kim LH, Jung JH, Kim JY, Yoon JH, Kim YJ, Lee HS. Interleukin 10 haplotype associated with increased risk of hepatocellular carcinoma. Hum Mol Genet. 2003;12:901–906. doi: 10.1093/hmg/ddg104. [DOI] [PubMed] [Google Scholar]
- Shin HD, Winkler C, Stephens JC, Bream J, Young H, Goedert JJ, O’Brien TR, Vlahov D, Buchbinder S, Giorgi J, et al. Genetic restriction of HIV-1 pathogenesis to AIDS by promoter alleles of IL10. Proc Natl Acad Sci U S A. 2000;97:14467–14472. doi: 10.1073/pnas.97.26.14467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sing A, Roggenkamp A, Geiger AM, Heesemann J. Yersinia enterocolitica evasion of the host innate immune response by V antigen-induced IL-10 production of macrophages is abrogated in IL-10-deficient mice. J Immunol. 2002;168:1315–1321. doi: 10.4049/jimmunol.168.3.1315. [DOI] [PubMed] [Google Scholar]
- Slobedman B, Barry PA, Spencer JV, Avdic S, Abendroth A. Virus-encoded homologs of cellular interleukin-10 and their control of host immune function. J Virol. 2009;83:9618–9629. doi: 10.1128/JVI.01098-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smelt SC, Borrow P, Kunz S, Cao W, Tishon A, Lewicki H, Campbell KP, Oldstone MB. Differences in affinity of binding of lymphocytic choriomeningitis virus strains to the cellular receptor alpha-dystroglycan correlate with viral tropism and disease kinetics. J Virol. 2001;75:448–457. doi: 10.1128/JVI.75.1.448-457.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer JV, Cadaoas J, Castillo PR, Saini V, Slobedman B. Stimulation of B lymphocytes by cmvIL-10 but not LAcmvIL-10. Virology. 2008;374:164–169. doi: 10.1016/j.virol.2007.11.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer SD, Di Marco F, Hooley J, Pitts-Meek S, Bauer M, Ryan AM, Sordat B, Gibbs VC, Aguet M. The orphan receptor CRF2-4 is an essential subunit of the interleukin 10 receptor. J Exp Med. 1998;187:571–578. doi: 10.1084/jem.187.4.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spolski R, Kim HP, Zhu W, Levy DE, Leonard WJ. IL-21 mediates suppressive effects via its induction of IL-10. J Immunol. 2009;182:2859–2867. doi: 10.4049/jimmunol.0802978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinbrink K, Wolfl M, Jonuleit H, Knop J, Enk AH. Induction of tolerance by IL-10-treated dendritic cells. J Immunol. 1997;159:4772–4780. [PubMed] [Google Scholar]
- Sun J, Madan R, Karp CL, Braciale TJ. Effector T cells control lung inflammation during acute influenza virus infection by producing IL-10. Nat Med. 2009;15:277–284. doi: 10.1038/nm.1929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun K, Metzger DW. Inhibition of pulmonary antibacterial defense by interferon-gamma during recovery from influenza infection. Nat Med. 2008;14:558–564. doi: 10.1038/nm1765. [DOI] [PubMed] [Google Scholar]
- Sun K, Torres L, Metzger DW. A detrimental effect of interleukin-10 on protective pulmonary humoral immunity during primary influenza A virus infection. J Virol. 2010;84:5007–5014. doi: 10.1128/JVI.02408-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka N, Sato M, Lamphier MS, Nozawa H, Oda E, Noguchi S, Schreiber RD, Tsujimoto Y, Taniguchi T. Type I interferons are essential mediators of apoptotic death in virally infected cells. Genes Cells. 1998;3:29–37. doi: 10.1046/j.1365-2443.1998.00164.x. [DOI] [PubMed] [Google Scholar]
- Taylor DR, Shi ST, Romano PR, Barber GN, Lai MM. Inhibition of the interferon-inducible protein kinase PKR by HCV E2 protein. Science. 1999;285:107–110. doi: 10.1126/science.285.5424.107. [DOI] [PubMed] [Google Scholar]
- Thimme R, Oldach D, Chang KM, Steiger C, Ray SC, Chisari FV. Determinants of viral clearance and persistence during acute hepatitis C virus infection. J Exp Med. 2001;194:1395–1406. doi: 10.1084/jem.194.10.1395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tinoco R, Alcalde V, Yang Y, Sauer K, Zuniga EI. Cell-intrinsic transforming growth factor-beta signaling mediates virus-specific CD8+ T cell deletion and viral persistence in vivo. Immunity. 2009;31:145–157. doi: 10.1016/j.immuni.2009.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trautmann L, Janbazian L, Chomont N, Said EA, Gimmig S, Bessette B, Boulassel MR, Delwart E, Sepulveda H, Balderas RS, et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat Med. 2006;12:1198–1202. doi: 10.1038/nm1482. [DOI] [PubMed] [Google Scholar]
- Tu Z, Pierce RH, Kurtis J, Kuroki Y, Crispe IN, Orloff MS. Hepatitis C virus core protein subverts the antiviral activities of human Kupffer cells. Gastroenterology. 2010;138:305–314. doi: 10.1053/j.gastro.2009.09.009. [DOI] [PubMed] [Google Scholar]
- Urbani S, Amadei B, Fisicaro P, Tola D, Orlandini A, Sacchelli L, Mori C, Missale G, Ferrari C. Outcome of acute hepatitis C is related to virus-specific CD4 function and maturation of antiviral memory CD8 responses. Hepatology. 2006a;44:126–139. doi: 10.1002/hep.21242. [DOI] [PubMed] [Google Scholar]
- Urbani S, Amadei B, Tola D, Massari M, Schivazappa S, Missale G, Ferrari C. PD-1 expression in acute hepatitis C virus (HCV) infection is associated with HCV-specific CD8 exhaustion. J Virol. 2006b;80:11398–11403. doi: 10.1128/JVI.01177-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Sluijs KF, van Elden LJ, Nijhuis M, Schuurman R, Pater JM, Florquin S, Goldman M, Jansen HM, Lutter R, van der Poll T. IL-10 is an important mediator of the enhanced susceptibility to pneumococcal pneumonia after influenza infection. J Immunol. 2004;172:7603–7609. doi: 10.4049/jimmunol.172.12.7603. [DOI] [PubMed] [Google Scholar]
- Velu V, Titanji K, Zhu B, Husain S, Pladevega A, Lai L, Vanderford TH, Chennareddi L, Silvestri G, Freeman GJ, et al. Enhancing SIV-specific immunity in vivo by PD-1 blockade. Nature. 2008 doi: 10.1038/nature07662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry EJ, Blattman JN, Ahmed R. Low CD8 T-cell proliferative potential and high viral load limit the effectiveness of therapeutic vaccination. J Virol. 2005;79:8960–8968. doi: 10.1128/JVI.79.14.8960-8968.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry EJ, Blattman JN, Murali-Krishna K, van der Most R, Ahmed R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J Virol. 2003;77:4911–4927. doi: 10.1128/JVI.77.8.4911-4927.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry EJ, Ha SJ, Kaech SM, Haining WN, Sarkar S, Kalia V, Subramaniam S, Blattman JN, Barber DL, Ahmed R. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity. 2007;27:670–684. doi: 10.1016/j.immuni.2007.09.006. [DOI] [PubMed] [Google Scholar]
- Wong JK, Hezareh M, Gunthard HF, Havlir DV, Ignacio CC, Spina CA, Richman DD. Recovery of replication-competent HIV despite prolonged suppression of plasma viremia. Science. 1997;278:1291–1295. doi: 10.1126/science.278.5341.1291. [DOI] [PubMed] [Google Scholar]
- Yang H, Guimaraes-Walker A, Hibbs S, Dong T, Stacey A, Borrow P, Hanke T, Davenport MP, McMichael A, Dorrell L. Interleukin-10 responses to therapeutic vaccination during highly active antiretroviral therapy and after analytical therapy interruption. AIDS. 2009;23:2226–2230. doi: 10.1097/QAD.0b013e328331a424. [DOI] [PubMed] [Google Scholar]
- Yi JS, Du M, Zajac AJ. A vital role for interleukin-21 in the control of a chronic viral infection. Science. 2009;324:1572–1576. doi: 10.1126/science.1175194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Younes SA, Yassine-Diab B, Dumont AR, Boulassel MR, Grossman Z, Routy JP, Sekaly RP. HIV-1 viremia prevents the establishment of interleukin 2-producing HIV-specific memory CD4+ T cells endowed with proliferative capacity. J Exp Med. 2003;198:1909–1922. doi: 10.1084/jem.20031598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zajac AJ, Blattman JN, Murali-Krishna K, Sourdive DJ, Suresh M, Altman JD, Ahmed R. Viral immune evasion due to persistence of activated T cells without effector function. J Exp Med. 1998;188:2205–2213. doi: 10.1084/jem.188.12.2205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou S, Halle A, Kurt-Jones EA, Cerny AM, Porpiglia E, Rogers M, Golenbock DT, Finberg RW. Lymphocytic choriomeningitis virus (LCMV) infection of CNS glial cells results in TLR2-MyD88/Mal-dependent inflammatory responses. J Neuroimmunol. 2008;194:70–82. doi: 10.1016/j.jneuroim.2007.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuniga EI, Liou LY, Mack L, Mendoza M, Oldstone MB. Persistent virus infection inhibits type I interferon production by plasmacytoid dendritic cells to facilitate opportunistic infections. Cell Host Microbe. 2008;4:374–386. doi: 10.1016/j.chom.2008.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]