Abstract
CONTEXT:
Pathogenesis of uterine synechia remains unsolved, the causal relationship between synechia and infertility is not clearly established.
AIMS:
To evaluate the rabbit as an experimental model for Asherman's syndrome using the endometrial curettage as trigger mechanism then to evaluate its impact on fertility.
SETTINGS AND DESIGN:
Experimental study
MATERIALS AND METHODS:
13 female rabbits. All submitted traumatic endometrial curettage. Animals of Group 1 (n = 7) were sacrificed at various times following surgery (day 7, 15 and 30), animals of Group 2 (n = 6) were bred and sacrificed during pregnancy. Main outcome were synechia occurring, number of implanted fetus, lumen surface/ global horn perimeter ratio (LS-GHP ratio) and epithelium thickness.
STATISTICAL ANALYSIS:
Means were compared using Student ‘t’ test (P < 0.05 was considered significant). Number of implantation sites of two horns were compared with the Wilcoxon test.
RESULTS:
No synechia have been observed. Examinations at Day 7, 15 and 30 demonstrate a complete regeneration of endometrium. We observed a significant diminished LS-GHP ratio at day 7 (0.042 ± 0.004 vs 0.074 ± 0.002 mm; P = 0.013) with a higher simple columnar epithelium compared to control (16.6 ± 3.39 vs 10.98 ± 1.7; P = 0.001). We observed a diminished ovum implantation in traumatized horns, even if it was not statistically significant.
CONCLUSION:
Even if no intrauterine adhesion were observed, this model represents a pathogenesis condition in the rabbit similar to intrauterine adhesions observed in the human with negative impact on implantation.
KEY WORDS: Asherman's syndrome, experimental study, rabbit, synechia
INTRODUCTION
Asherman's syndrome, or uterine synechia, is defined as a partial or complete obliteration of the uterine cavity and/or the cervical canal. This condition can cause menstrual abnormalities, infertility and recurrent pregnancy losses.[1]
Most often, synechia are a direct consequence of surgical endometrial lesions of a gravid uterus. It essentially occur after a uterine curettage, performed in cases of miscarriage, pregnancy termination[2] or evacuation of placental products in the postpartum period.[3] Synechia may also appear after a Cesarean section[4] or a uterine devascularization in cases of severe postpartum hemorrhage.[5]
Trauma to a nongravid uterine cavity may also result in Asherman's syndrome. Diagnostic curettage or hysteroscopic surgery, including mymectomy, polypectomy and endometrial ablation, can induce Asherman's syndrome. Myomectomy is associated with the highest risk for inducing uterine adhesions. Synechias can occur in up to 31.7% following surgical interventions using unipolar energy.[6] The risk appears reduced with the use of bipolar energy.[7]
Infections are also recognized as a cause of intrauterine adhesions particularly tuberculosis which has been shown to induce severe damage and was found in 4% of the 1856 synechia cases reported by Schenker and Al.[8]
The menstrual abnormalities associated with Asherman's syndrome are due to cervical adhesions blocking menstrual flow or severe endometrial fibrosis.[9]
Infertility may results from severe endometrial destruction, affecting implantation, or be caused by the occlusion of ostia.[9]
The causal relationship between synechia and infertility is not clearly established. In Schenker's study, infertility was only present in 43% of 2151 cases of Asherman's syndrome but these findings were based on a retrospective study with obvious bias: asymptomatic, fertile patients were not diagnosed. Evidence of this relationship can only be established based on a prospective study comparing the fertility of patients with known Asherman's syndrome, either treated or not. This kind of study is, of course, impossible to realize in human subjects because of ethical considerations. In the same study, however, among 292 patients with untreated Asherman's syndrome, a rate of 45.5% of spontaneous pregnancies was reported, but these were associated with pregnancy loss in 40% of the cases.[8]
Several questions remain unsolved. It is still very difficult to explain the high interindividual variability for the onset of intrauterine adhesions. So far, the exact mechanism of synechia formation is not known, nor is the influence of gravidity. For all these reasons and also because there is a paucity of effective preventive treatments, there is a real need for experimental animal model to explain synechia pathogenesis, prevention and treatment.
In this study, we evaluated the rabbit as an experimental model of synechia. The rabbit is classified as bicornuate, i.e., it is characterized by the presence of two completely separate uterine horns with two cervices, allowing one horn to be used as control for the other [Figure 1.] We used traumatic curettage as a trigger mechanism.
Figure 1.
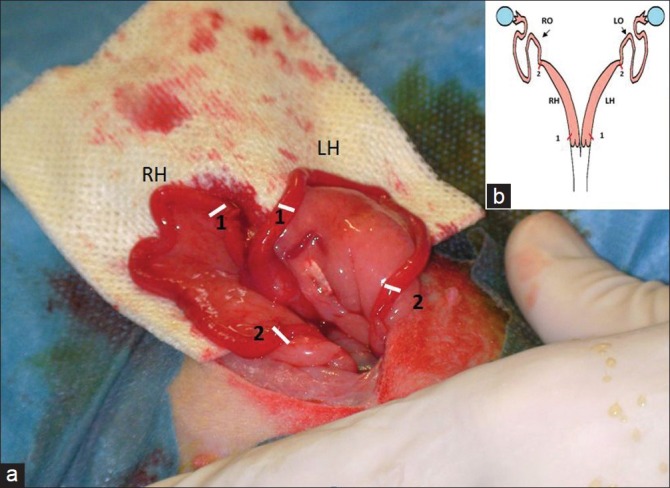
(a) Schematic illustration of the rabbit uterus. Red marks: incisions sites (b) peroperative photograph of the two uterine horns. White marks: incisions sites (1) Proximal incisions (2) distal incisions. (RH) Right horn; (LH) Left horn; (RO) right oviduct t; (LO) Left oviduct
The first objective of this study was to create a rabbit model of intrauterine adhesions in order to provide a new tool for further investigations in this field such as experimentation of new preventive treatments. The second objective is to evaluate the impact of curettage on fertility in this model.
MATERIALS AND METHODS
This is an experimental, prospective study
The experiment was performed in accordance with the International Guiding Principles for Biomedical Research involving Animals as promulgated by the Society for the Study of Reproduction and in accordance with the European Convention on Animal experimentation. Researchers involved in the work with the animals possessed an animal experimentation license delivered by the veterinary services. The euthanasia of the animals was performed at the local experimental slaughterhouse according to the protocol approved by the local ethics committee and the veterinary services. The whole experiment was approved by the local scientific committee for animal use.
A total of 13 mature, previously fertile female New Zealand White rabbits (INRA 1007 line) weighing 3600-4300 g were placed in a controlled environment with free access to food and water. They were housed individually in the same building with a temperature and light controlled environment.
All animals were submitted to experimental curettage as described below. They were then divided in two groups: Group 1 (non pregnant) where animals were sacrificed at various times following surgery, in order to evaluate the histological consequences of the curettage, and Group 2 (pregnant) where they were bred and sacrificed during pregnancy, in order to evaluate the consequence of the intrauterine surgery in terms of future spontaneous fertility [Figure 3].
Figure 3.
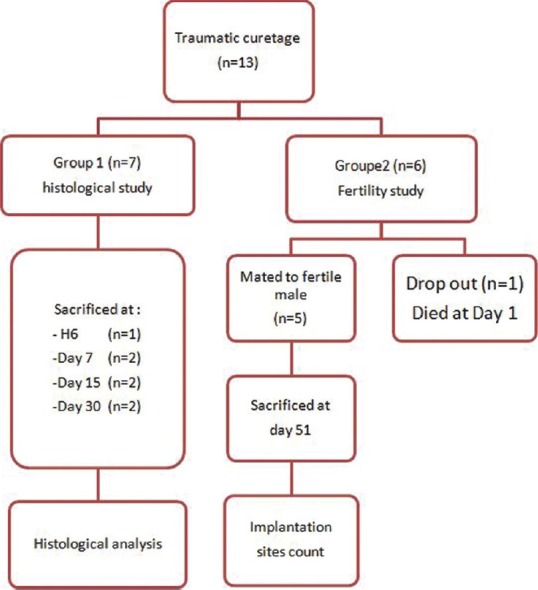
Experimental protocol
Anesthesia was induced by Flutothane inhalation after overnight fasting. Following laparotomy, the pelvic region was examined in order to exclude any animal with macroscopic abnormality which may interfere with fertility.
Five-mm long incisions were performed on the left horn: the first one was located right after the cervix (proximal incision) and the second one at the uterotubal junction (distal incision) [Figure 1]. A complete curettage of the endometrium was performed using a Basnier curette (2-mm diameter) through these incisions [Figure 2]. The right horn was used as control, the same incisions being performed without any curettage (sham operation).
Figure 2.
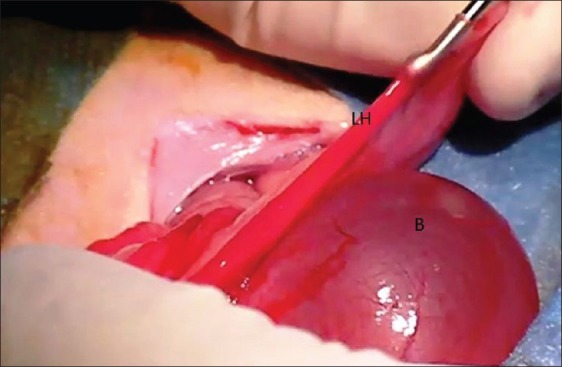
Surgical procedure: Intrauterine curettage of the left horn using a Curette de Basnier. (B): bladder, (LH): Left Horn
One female was sacrificed 6 hours after surgery to make sure that the surgical technique was effective to destroy the endometrial epithelium.
Thereafter, two rabbits were euthanized respectively on Day 7, 15 and 30 after surgery to analyze histological damages following curettage and the evolution of the regeneration process with time (Group 1). At postmortem, the uterus was removed and uterine horns fixed in 10% formalin.
The six remaining does were used for a fertility study (Group 2). They were mated to a fertile male on day 30 and subsequently sacrificed on day 51 [Figure 3] and implantation sites were counted.
In the rabbit scarified at the sixth hour, four sections were performed in each horn and rate of endometrial destruction was measured.
In group 1, three sections of each uterine horn were performed, paraffin-embedded and stained for histological examination. Slides were scanned using Nanozoomer (HamamatsuÒ, Japan) and images were analyzed with Image J (http://rsbweb.nih.gov/ij). In each section, the surface of the lumen (LS), the global horn perimeter (GHP) and the proportion of damaged endometrial luminal epithelium were measured. In each section, six measurements of endometrial thickness (ET) were performed. Inflammatory elements were also observed and histological changes in time were analyzed.
In Group 2, the number of implantations sites was counted macroscopically for each horn.
Primary outcomes are intrauterine adhesions formation defined as a complete or partial obstruction of horn lumen and difference in the number of gestations between the operated and nonoperated horns.
Secondary Outcomes are represented by the time-related histological modifications after intrauterine curettage: difference in term of luminal surface -global perimeter ratio (LS-GHP ratio) and ET, presence of inflammatory elements and regeneration process.
In group1: For each date (Day 7, Day 15 and 30), the mean LS-GHP ratio and the mean endometrial thickness were calculated. Means were compared using Student ‘t’ test. A value of P < 0.05 was considered significant.
In group 2: For fertility data, numbers of implantation sites of tow horns were compared with the Wilcoxon test for paired samples. All statistical analysis was performed using SPSS 13 for Windows.
RESULTS
Outcomes of the experimental animals are summarized in Figure 3. No gross pathologies were observed in all subjects. One Group 2 rabbit died on Day 1. All others subjects had no postoperative complications.
Histological findings in the immediate postoperative period (6 hours) showed significant epithelial damage, 64% of the total endometrial surface, in the left horn, together with significant inflammatory reaction. No endometrial destruction was observed in the right horn [Figure 4].
Figure 4.
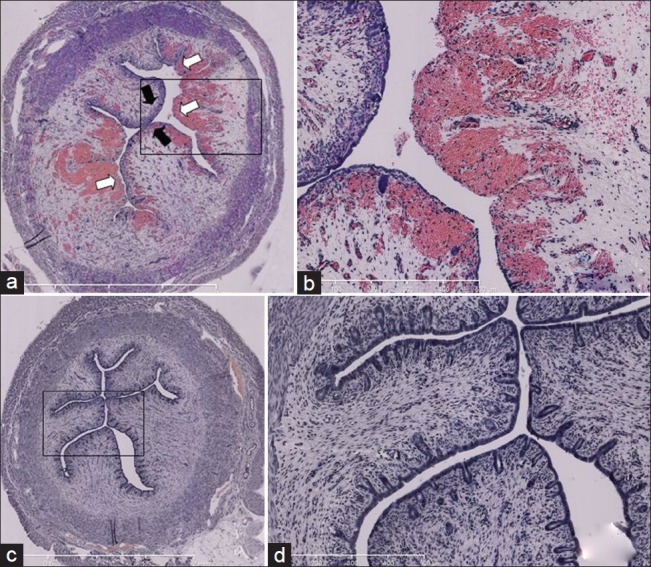
Histological findings 6 hours after traumatic curettage of endometrium in Rabbit (a) right horn (control): normal epithelium (a) Left horn (curettage): Disrupted epithelium covering 64 % of total endometrium; hyervascularized submucosa (b) Focused image of left horn (c) Right horn (control): No endometrial disruption (d) Focused image of right horn. Black narrow: Normal epithelium; White narrow: disrupted endometrium
In group 1, no intrauterine synechia was observed in the different sections observed.
Complete regeneration of endometrium was observed in all animals from day 7, without any inflammatory reactions [Figure 5].
Figure 5.
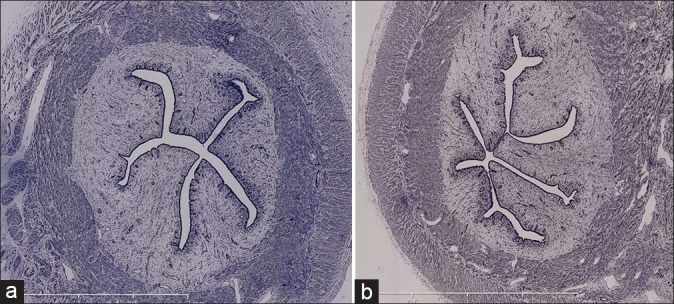
Histological findings at day 15: no endometrial disruption observed in left horn (a) and right horn (b) (control). Same aspect has been observed at day 7 and 30
At Day 7, the mean LS-GHP ration of the operated horn was statistically lower compared to the control. For Day 15 and 30, there was no difference, between two horns, for this parameter.
At day 7 and 30, ET was significantly higher in the traumatized horn.
The mean LS-GHP ratios and mean ET for each date are summarized in Table 1.
Table 1.
Histological findings in group student test was used for comparing means

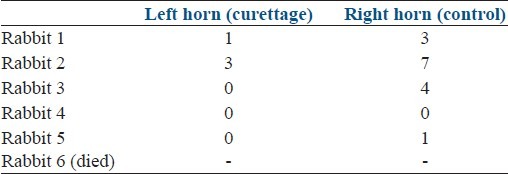
There was a decrease in the number of implantation sites on the traumatized left horn. Gestation was systematically lower in four cases. In one case, no gestations occurred on both sides [Table 2]. However, the difference between the two horns in term of gestations was not significant according to the Wilcoxon test.
Table 2.
Implantation count at day 51 in group 2 Wilcoxon test was used for comparing number of implantation sites
DISCUSSION
In this study, we attempted to create an animal model of postoperative synechia using the rabbit, based on a true clinical situation: the endometrial curettage. The examination of uterine section, taken at the sixth hour, shows that curettage was effective for endometium destruction. Histological examination at different times (Day 7, 15 and 30), demonstrate a complete regeneration of endometrium. We observed a significant diminished lumen surface at day 7 with a higher simple columnar epithelium compared to control. We observed a diminished ovum implantation in traumatized horns, even if it was not statistically significant. We did not objectify intrauterine synechia as observed in humans. The human uterus displays some idiosyncrasies compared to other mammals: a uterine muscle with an important thickness can only be observed in some of higher order primates. The high-regeneration frequency of the endometrium, due to monthly menstrual menses, is also quite specific of the Human species. In theory, the macaque would have been an ideal model, but it would have raised ethical, concerns and also would have pulled a considerable additional cost. In the present study, the choice of the rabbit as an animal model was justified by the short gestation period (31-32 days), the high fertility, the bicornual uterine structure, with two separate cervices (that offer an ideal control). Finally both woman and female rabbit possess a hemochorial placentation, what contributes to move closer to the clinical situations observed in human, especially for fertility study, in contrast to the horse where spontaneous synechia were reported.[10]
Preserving fertility after endouterine surgery is a great concern for clinicians and a synechia occurring is considered as a postoperative complication which, in some cases, may be hard to treat. In contrast with this great interest, little is currently known about the process of synechia formation and its physiopathology, while this could help in developing effective preventive treatments. For these considerations, creating an animal model is essential because it represents a tool for time-related histological changes description after endouterine trauma. Moreover, since causal relationship between endometrial surgery and infertility has been established based on retrospective studies.[8] synechia induction in an animal model may allow further investigations of this relationship. In contrast with the high number of publications using animal models for intraperitoneal adhesions,[11] intrauterine adhesions are rarely investigated or described in animals. To our knowledge, there is only one experimental model of intrauterine adhesions, as described by Schenker et al. in the rabbit.[12,13] Spontaneous occurrence of intrauterine synechia was also reported, in a unique retrospective study, in 10% of 87 hysteroscopic examinations realized in hypofertiles mares.[10] Schenker and al conducted their studies in order to investigate a treatment for abnormal genital bleeding or for contraception. They performed a traumatic curettage of the endometrium in rabbits pretreated with estrogens,[14] progesterone[15] or without pretreatment.[16] None of these experimentations succeeded in creating intrauterine adhesions and the authors report a complete regeneration of endometrium at day 7 of surgery.[16] Despite the fact that Schenker failed to create synechia using curettage, we choose to reuse the endometrial curettage as it is a frequent procedure in routine gynecological activity and because we aimed to evaluate its consequence on spontaneous fertility even in case of no adhesions formation. In accordance with Schenker findings, we failed to reveal intrauterine adhesions using curettage but we demonstrate a diminished lumen surface probably due to a high endometrial thickness. We cannot exclude an unrevealed minimal or filmy synechia as it is hard to explore all the horn length microscopally.
Schenker and al also used other surgical approaches such as endometrial destruction by cryosurgery or chemical aggression, all without success.[17,18] Finally, they performed a complex 2 step-procedure that eventually led to the establishment of synechia of the rabbit horn. First, a sponge was grafted subcutaneously and colonized by fibroblasts. After three weeks, the subcutaneous sponge was retrieved and inserted into the uterine horn. With this model they obtained total intrauterine adhesions. In this paper, they conclude that fibroblast have a major role in Asherman's syndrome pathology.[12,13] This procedure is not comparable to clinical situations leading to Asherman's syndrome and unhelpful for clinical purpose. Furthermore, these “synechia” are probably a simple immunological reaction against a foreign body. Our results are in concordance with Schenker experimentation using curettage.[16] Despite of a significant endometrial destruction, a complete regeneration is observed at day 7 contrasting with the histological aspect at the sixth hour. This apparent histological restitution of the endometrium contrasts with a decrease in level of implantation in the traumatized horn. We show in this study, that histological morphology of endomterium was not correlated with its functional aspect. These findings suggest limits of histological examination to predict implantation. In our model, implantation impairment may be explained by the existence of an inflammatory environment unexplored by classical histology. However, inflammation impact on implantation remains unclear: while some publications reports negative impact of inflammation,[19] other authors have shown that a local injury of the endometrium induced an inflammatory response that promotes successful implantation.[20] This model allowed an evaluation of histology limits for appreciation of endometrial function but also demonstrates the potential impact of curettage on fertility. In spite of we could not observe Ashermans/synechia, we accepted the demonstration they were deemed to have occurred and the option of inflammatory reaction is the consequence on subsequent fertility. This model may be used to investigate safety and effectiveness of preventive therapeutics of Asherman's syndrome.
In the present study, we propose a model of pathogenesis condition in the rabbit similar to intrauterine adhesions observed in the human. Even if no synechia have been observed, we concluded to a negative impact of endometrium curettage on spontaneous fertility similar to synechia, which will help in our understanding of the physiopathology of Asherman's syndrome, and also to evaluate several therapeutic options to prevent its occurrence.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Asherman JG. Traumatic intrauterine adhesions. J Obstet Gynaecol Br Emp. 1950;57:892–6. doi: 10.1111/j.1471-0528.1950.tb06053.x. [DOI] [PubMed] [Google Scholar]
- 2.Tam WH, Lau WC, Cheung LP, Yuen PM, Chung TK. Intrauterine adhesions after conservative and surgical management of spontaneous abortion. J Am Assoc Gynecol Laparosc. 2002;9:182–5. doi: 10.1016/s1074-3804(05)60129-6. [DOI] [PubMed] [Google Scholar]
- 3.Westendorp IC, Ankum WM, Mol BW, Vonk J. Prevalence of Ashermans syndrome after secondary removal of placental remnants or a repeat curettage for incomplete abortion. Hum Reprod. 1998;13:3347–50. doi: 10.1093/humrep/13.12.3347. [DOI] [PubMed] [Google Scholar]
- 4.Badawy SZ, Orbuch L, Khurana KK. Secondary amenorrhea with severe intrauterine adhesions and chronic uterine torsion after cesarean section in a teenage girl. J Pediatr Adolesc Gynecol. 1998;11:93–6. doi: 10.1016/s1083-3188(98)70119-6. [DOI] [PubMed] [Google Scholar]
- 5.Roman H, Sentilhes L, Cingotti M, Verspyck E, Marpeau L. Uterine devascularization and subsequent major intrauterine synechiae and ovarian failure. Fertil Steril. 2005;83:755–7. doi: 10.1016/j.fertnstert.2004.07.975. [DOI] [PubMed] [Google Scholar]
- 6.Taskin O, Sadik S, Onoglu A, Gokdeniz R, Erturan E, Burak F, et al. Role of endometrial suppression on the frequency of intrauterine adhesions after resectoscopic surgery. J Am Assoc Gynecol Laparosc. 2000;7:351–4. doi: 10.1016/s1074-3804(05)60478-1. [DOI] [PubMed] [Google Scholar]
- 7.Touboul C, Fernandez H, Deffieux X, Berry R, Frydman R, Gervaise A. Uterine synechiae after bipolar hysteroscopic resection of submucosal myomas in patients with infertility. Fertil Steril. 2009;9:1690–3. doi: 10.1016/j.fertnstert.2008.08.108. [DOI] [PubMed] [Google Scholar]
- 8.Schenker JG, Margalioth EJ. Intrauterine adhesions: An updated appraisal. Fertil Steril. 1982;37:593–610. doi: 10.1016/s0015-0282(16)46268-0. [DOI] [PubMed] [Google Scholar]
- 9.Yu D, Wong YM, Cheong Y, Xia E, Li TC. Ashermans syndrome-one century later. Fertil Steril. 2008;89:759–79. doi: 10.1016/j.fertnstert.2008.02.096. [DOI] [PubMed] [Google Scholar]
- 10.Bracher V, Mathias S, Allen WR. Videoendoscopic evaluation of the mare's uterus: Findings in subfertile mares. Equine Vet J. 1992;24:279–84. doi: 10.1111/j.2042-3306.1992.tb02835.x. [DOI] [PubMed] [Google Scholar]
- 11.Ward BC, Panitch A. Abdominal adhesions: Current and novel therapies. J Surg Res. 2011;165:91–111. doi: 10.1016/j.jss.2009.09.015. [DOI] [PubMed] [Google Scholar]
- 12.Polishuk WZ, Schenker JG. Induction of intrauterine adhesions in the rabbit with autogenous fibroblast implants. Am J Obstet Gynecol. 1973;115:789–94. doi: 10.1016/0002-9378(73)90522-x. [DOI] [PubMed] [Google Scholar]
- 13.Schenker JG, Yaffe H. Induction of intrauterine adhesions in experimental animals and in women. Isr J Med Sci. 1978;14:261–6. [PubMed] [Google Scholar]
- 14.Schenker JG, Polishuk WZ, Sacks MI. Regeneration of the endometrium in rabbits after curettage. Pretreatment with estrogens. J Reprod Med. 1973;11:43–8. [PubMed] [Google Scholar]
- 15.Schenker JG, Polishuk WZ, Sacks MI. Regeneration of the endometrium in rabbits after curettage. Pretreatment with progesterone. J Reprod Med. 1973;11:49–57. [PubMed] [Google Scholar]
- 16.Schenker JG, Sacks MI, Polischuk WZ. Regeneration of rabbit endometrium following curettage. Am J Obstet Gynecol. 1971;111:970–8. doi: 10.1016/0002-9378(71)90955-0. [DOI] [PubMed] [Google Scholar]
- 17.Schenker JG, Polishuk WZ. Regeneration of rabbit endometrium after cryosurgery. Obstet Gynecol. 1972;40:638–45. [PubMed] [Google Scholar]
- 18.Schenker JG, Polishuk WZ. Regeneration of rabbit endometrium following intrauterine instillation of chemical agents. Gynecol Invest. 1973;4:1–13. doi: 10.1159/000301703. [DOI] [PubMed] [Google Scholar]
- 19.Weiss G, Goldsmith LT, Taylor RN, Bellet D, Taylor HS. Inflammation in reproductive disorders. Reprod Sci. 2009;16:216–29. doi: 10.1177/1933719108330087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gnainsky Y, Granot I, Aldo PB, Barash A, Or Y, Schechtman E, et al. Local injury of the endometrium induces an inflammatory response that promotes successful implantation. Fertil Steril. 2010;94:2030–6. doi: 10.1016/j.fertnstert.2010.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]


