Abstract
Background:
Mediator complex participates in transcriptional regulation by connecting regulatory DNA sequences to the RNA polymerase II initiation complex. Recently, we discovered through exome sequencing that as many as 70% of uterine leiomyomas harbour specific mutations in exon 2 of mediator complex subunit 12 (MED12). In this work, we examined the role of MED12 exon 2 mutations in other tumour types.
Methods:
The frequency of MED12 exon 2 mutations was analysed in altogether 1158 tumours by direct sequencing. The tumour spectrum included mesenchymal tumours (extrauterine leiomyomas, endometrial polyps, lipomas, uterine leiomyosarcomas, other sarcomas, gastro-intestinal stromal tumours), hormone-dependent tumours (breast and ovarian cancers), haematological malignancies (acute myeloid leukaemias, acute lymphoid leukaemias, myeloproliferative neoplasms), and tumours associated with abnormal Wnt-signalling (colorectal cancers (CRC)).
Results:
Five somatic alterations were observed: three in uterine leiomyosarcomas (3/41, 7% Gly44Ser, Ala38_Leu39ins7, Glu35_Leu36delinsVal), and two in CRC (2/392, 0.5% Gly44Cys, Ala67Val).
Conclusion:
Somatic MED12 exon 2 mutations were observed in uterine leiomyosarcomas, suggesting that a subgroup of these malignant tumours may develop from a leiomyoma precursor. Mutations in CRC samples indicate that MED12 may, albeit rarely, contribute to CRC tumorigenesis.
Keywords: MED12 , mutation screening, somatic mutation, benign tumours, malignant tumours
Mediator is a large multiprotein complex, which is involved in global as well as gene-specific transcriptional regulation of most protein coding genes. The complex can both activate and repress transcription by connecting transcription factors to the RNA polymerase II initiation complex (Taatjes, 2010). Mediator complex subunit 12 (MED12) gene on Xq13.1 encodes MED12 protein, which together with MED13, CDK8, and Cyclin C comprises a CDK8 submodule of the Mediator. This submodule participates in transcriptional regulation, as well as in scaffold formation and transcription elongation (Galbraith et al, 2010). Mediator complex subunit 12 is an essential regulator of the kinase activity of CDK8 submodule, and the protein directly interacts with various transcription factors (Knuesel et al, 2009; Taatjes, 2010). Mediator complex subunit 12 participates in various molecular pathways, for example, p53 and Wnt/β-catenin pathways, which have central roles in tumour development (Kim et al, 2006; Galbraith et al, 2010).
Uterine leiomyomas, also known as fibroids, are benign smooth muscle tumours that occur in approximately 70% of women by the age of 50 years (Day Baird et al, 2003). Despite their benign nature, these tumours cause various symptoms including abdominal pain, abnormal menstrual bleeding, pregnancy complications, and even infertility. Oestrogen and progesterone dependency is a characteristic feature for uterine leiomyomas, which usually occur in women of reproductive age and typically regress after menopause (Parker, 2007). Several recurrent, albeit infrequent, genetic aberrations have been observed in these tumours, including deletions in chromosome 7q, trisomy of chromosome 12, various rearrangements affecting the high mobility group AT-hook 2 (HMGA2) gene, and structural changes at 6p21 (Mitelman, 1998). Recently, we discovered by exome sequencing that as many as 70% of uterine leiomyomas harbour very specific somatic mutations in MED12 (Mäkinen et al, 2011a). Strikingly, all mutations resided in exon 2, and the vast majority of them affected a single codon glutamine 44. The finding has subsequently been validated in other populations (Mäkinen et al, 2011b; Je et al, 2012; Markowski et al, 2012; McGuire et al, 2012). Mediator complex subunit 12 had not been implicated in human tumorigenesis before identification of specific exon 2 mutations in uterine leiomyomas. High mutation frequency and the proteins’ key role in transcriptional regulation prompted us to study the genes role in other tumour types. We collected a comprehensive series of 1158 samples representing various tumour types that might harbour mutations in the gene. These include both benign and malignant mesenchymal tumours, oestrogen- and progesterone-dependent tumours, and haematological malignancies, as retroviral insertions in Med12 have been reported to participate in the development of leukaemias in murines (Dave et al, 2009). We also included tumours associated with abnormal Wnt-signalling, as preliminary data indicated this well-known cancer-related pathway may be dysregulated in MED12 mutation-positive uterine leiomyomas (Mäkinen et al, 2011a).
Materials and Methods
Subjects
Altogether 1158 tumours from as many patients were included in the study. The sample series consisted of 286 mesenchymal tumours (uterine leiomyosarcomas, other sarcomas, gastrointestinal stromal tumours, extrauterine leiomyomas, endometrial polyps, lipomas), 216 oestrogen- and progesterone-dependent tumours (ovarian and breast carcinomas), 264 haematological malignancies (acute myeloid leukaemias, acute lymphoid leukaemias, and myeloproliferative neoplasms), and 392 colorectal cancers (CRCs) as representatives of tumours commonly showing abnormal Wnt-signalling. See Table 1 for more details on the whole sample set utilised in the study, and Supplementary Information and Supplementary Tables S1,2 for additional information on uterine leiomyosarcomas and extrauterine leiomyomas.
Table 1. Tumours included in the MED12 exon 2 mutation screening.
| Tumour type | N | Sample type | Samples from |
|---|---|---|---|
| Mesenchymal | |||
| Uterine leiomyosarcoma | 39 | FFPE | PH/CFCH |
| Early onset (dg⩽45 years) | 27 | PH | |
| Unselected | 12 | CFCH | |
| Sarcoma | 104 | Fresh frozen | PH |
| Soft tissue sarcoma (including two ULMS) | 83 | ||
| Bone sarcoma | 21 | ||
| Gastrointestinal stromal tumour | 12 | FFPE | CFCH |
| Extrauterine leiomyoma | 42 | FFPE | CFCH/PH |
| Endometrial polyp | 54 | FFPE | PH |
| Lipoma | 35 | FFPE | CFCH |
| Oestrogen–progesterone dependent | |||
| Ovarian carcinoma | 122 | FFPE/fresh frozen | PH |
| Clear cell | 39 | ||
| Serous | 44 | ||
| Mucinous | 10 | ||
| Endometrioid | 10 | ||
| NOS | 19 | ||
| Breast cancer | 94 | Fresh frozen | OGH/PH |
| Ductal | 68 | ||
| Lobular | 14 | ||
| Medullary | 4 | ||
| Other | 8 | ||
| Haematological malignancies | |||
| Acute myeloid leukaemia | 131 | Fresh/fresh frozen | AAUH/MSKCC |
| Acute lymphoid leukaemiaa | 37 | Fresh frozen | AAUH |
| Myeloproliferative neoplasm | 96 | Fresh/fresh frozen | MSKCC |
| Polycythemia vera | 48 | ||
| Essential thrombocytosis | 48 | ||
| Abnormal Wnt-signalling associated | |||
| Colorectal cancer | 392 | Fresh frozen | FCH |
Abbreviations: AAUH=Department of Hematology at Aarhus University Hospital; CFCH=Central Finland Central Hospital; FCH=Finnish Central Hospitals; FFPE=formalin-fixed paraffin-embedded; MSKCC=Memorial Sloan-Kettering Cancer Center; NOS=not otherwise specified; OGH=Department of Obstetrics and Gynecology at Helsinki University Central Hospital; PH=Department of Pathology at Helsinki University Central Hospital; ULMS=uterine leiomyosarcoma.
T-cell origin.
The study was approved by the ethics review board of the Hospital District of Helsinki and Uusimaa (HUS), Helsinki, Finland, and the appropriate research permissions were obtained from local ethics committees.
DNA extraction and MED12 exon 2 mutation screening
Genomic DNA was extracted from archival formalin-fixed paraffin-embedded and fresh-frozen tissue samples with standard methods. Mediator complex subunit 12 exon 2 mutation status was determined by direct sequencing. Two different sets of primers were used in the study. See Supplementary Information for more details.
Results
Five MED12 exon 2 mutations were identified in 1158 tumour samples, three in uterine leiomyosarcomas (3/41; 7%) and two in colorectal tumours (2/392; 0.5%) (Supplementary Table S3). No mutations were observed in any other tumour type.
Two MED12 mutations were detected in histopathologically confirmed early-onset (dg⩽45 years) uterine leiomyosarcomas; one affecting the codon 44 that is frequently mutated in uterine leiomyomas (c.130G>A, p.Gly44Ser) and the other inserting 21 nucleotides and leading to an in-frame transcript (c.115_116ins21, p.Ala38_Leu39ins7). Corresponding normal DNA was available from these patients, and the somatic origin of the mutations was confirmed (Supplementary Figure S1). The third mutation was a three nucleotide deletion also resulting in an in-frame transcript (c.104_106delAAC, p.Glu35_Leu36delinsVal). This mutation was identified in a soft-tissue sarcoma sample, which was further diagnosed as a metastasis of uterine leiomyosarcoma. Unfortunately, normal DNA from this patient was not available for this study, and the somatic origin of this mutation could not be confirmed. It is likely, however, that this mutation is also somatic as it is located in a highly conserved area of the protein affecting the codon 36, which is the second most common mutational hotspot observed in uterine leiomyomas. Furthermore, no germline changes have been observed at this site in our own studies or reported in any published studies or in the databases.
The fourth mutation (c.130G>T, p.Gly44Cys), again hitting the hotspot codon 44, was identified in one CRC sample. The female patient had been diagnosed with a Dukes B/grade II/microsatellite stable tumour in the sigmoid colon at the age of 78 years. A variant with an unknown significance (c.200C>T, p.Ala67Val) was observed in one additional CRC sample. The codon is conserved among species (Figure 1), although according to in silico analyses with Polyphen2 (http://genetics.bwh.harvard.edu/pph2/) and SIFT (http://sift.bii.a-star.edu.sg/), this change is predicted to be tolerated. The female patient had been 78 years old when a Dukes A/grade II tumour in ascending colon was diagnosed. The tumour showed microsatellite instability. Both observed mutations in CRC samples were confirmed to be somatic (Supplementary Figure S2).
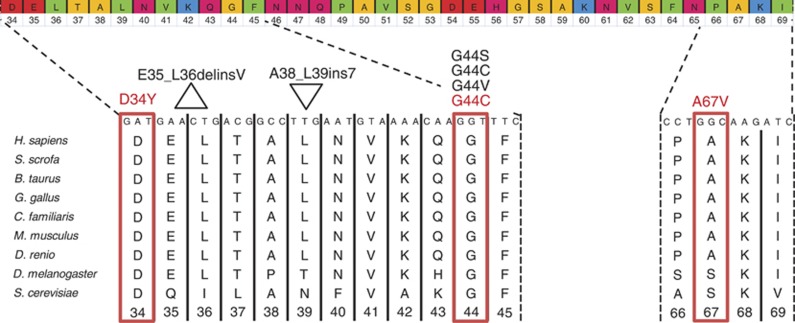
Figure 1.
Mutations in MED12 exon 2. The whole MED12 exon 2 with the amino acids and codon numbers is shown at the top, and multispecies alignment of the regions with the detected mutations is shown below. Mutations observed in uterine leiomyosarcoma and CRC samples are marked with black and red, respectively. Mutations G44C and G44V in ULMS were reported by Pérot et al (2012). Mutation G44C in CRC was observed in this study and also in the study by The Cancer Genome Atlas Network (2012). Mutation D34Y in CRC has been reported by Je et al (2012). Amino acids at the top are color-coded according to their side-chains' pKas (acid dissociation constant) and charge at physiological pH 7.4. Red = negatively charged, green = hydrophobic, yellow = small non-polar, magenta = polar, blue = positively charged.
In one endometrial polyp sample, MED12 exon 2 mutation (c.107T>G, p. Leu36Arg) was initially observed. Closer evaluation revealed that the sample also contained degenerated uterine leiomyoma tissue. Genomic DNA was extracted separately from both the polyp and leiomyoma, and the sequencing confirmed that the mutation originated from the leiomyoma.
Discussion
Our recent study revealed very specific mutations in MED12 exon 2 in as many as 70% of uterine leiomyomas (Mäkinen et al, 2011a). This study implicated, for the first time, a role for MED12 in human tumorigenesis. To analyse whether similar mutations can be found in other tumour types, we collected a broad spectrum of samples for the MED12 exon 2 mutation analyses.
Uterine leiomyomas originate from the smooth muscle cells and are thus of mesenchymal origin. Similar karyotypic changes that have been observed in uterine leiomyomas have also been seen in other benign mesenchymal tumours, such as extrauterine leiomyomas, endometrial polyps, and lipomas (Tallini et al, 2000). We therefore hypothesised that these tumours might also harbour mutations in MED12. We screened altogether 131 benign mesenchymal tumours, but no mutations were identified. Similar results were recently reported by Markowski et al (2012), who found only one mutation in a single endometrial polyp and no mutations in the lipomas. Of specific note is that none of the 42 extrauterine leiomyoma samples analysed in this study harboured MED12 mutations.
Altogether 155 malignant mesenchymal tumours were analysed for MED12 exon 2 mutations. Three mutations were observed, two in early-onset uterine leiomyosarcomas, and one in a metastasis of a uterine leiomyosarcoma (3/41; 7% of uterine leiomyosarcomas studied). This finding is in line with the results of a recent study by Pérot et al (2012), where MED12 mutations were reported in 2/10 (20%) uterine leiomyosarcomas and 1/9 (11%) smooth muscle tumour of uncertain malignant potential, respectively. Highly aggressive and malignant uterine leiomyosarcomas are not generally considered to develop from benign leiomyomas. The observed MED12 mutations are probably not the driving force behind the malignant transformation, but rather indicate that these tumours have developed from leiomyoma precursors. Indeed, it has also previously been suggested that a small subgroup of leiomyomas may actually develop into malignancy (Christacos et al, 2006). Would this be the case, identification of molecular markers that could be used in detecting such leiomyomas would be of high clinical significance as in most cases the leiomyosarcoma diagnosis is only made at surgery and many patients present with an advanced disease. In this study, no mutations were found in other sarcomas or in gastrointestinal stromal tumours. Overall, the low frequency of MED12 exon 2 mutations in various mesenchymal tumours suggests that the high mutation frequency observed in uterine leiomyomas is not a common feature for all mesenchymal tumours.
Development and growth of uterine leiomyomas are dependent on oestrogen and progesterone. Lesions show increased expression of oestrogen and progesterone receptors, and enhanced response to oestrogen stimulation compared with normal myometrial cells has been observed. Progesterone has also been reported to increase mitotic activity and regulate cell proliferation in uterine leiomyoma cells (Parker, 2007). To elucidate whether MED12 is involved in the tumorigenesis of other oestrogen- and progesterone-dependent tumours, we screened breast and ovarian cancer samples representing different histological subtypes for MED12 exon 2 mutations. None of the tumours harboured mutations, indicating that aberrant hormonal function is not the underlying cause, at least alone, for MED12 mutations in uterine leiomyomas.
Mediator complex subunit 12 interacts with β-catenin, and it has been identified as an important transducer of canonical Wnt/β-catenin signalling (Kim et al, 2006; Rocha et al, 2010). The preliminary results by us and Markowski et al implicate that this signalling pathway may be altered in MED12 mutation-positive uterine leiomyomas (Mäkinen et al, 2011a; Markowski et al, 2012). Dysregulation of Wnt/β-catenin pathway is involved in the development of many tumour types, including CRC (Polakis, 2000). Here, we identified two MED12 exon 2 mutations in altogether 392 CRC samples analysed. Similar results were recently reported by Je et al and The Cancer Genome Atlas Network, both of whom found one MED12 exon 2 mutation in 389 (0.3%) and 224 (0.4%) CRC samples, respectively (Je et al, 2012; The Cancer Genome Atlas Network, 2012). Although these mutations may have occurred just by chance, these findings suggest that specific MED12 exon 2 mutations may also be involved, albeit rarely, in the development of colorectal tumours.
Leukaemias have been shown to develop in murines with retroviral insertions in Med12 (Dave et al, 2009). A role for the CDK8 module/MED12 in hematopoiesis has also been suggested in Drosophila and zebrafish, respectively (Gobert et al, 2010; Keightley et al, 2011). High MED12 expression level has been observed in acute myeloid leukaemia and acute lymphoid leukaemia compared with other tumour types in the GeneSapiens database (http://www.genesapiens.org/) (Kilpinen et al, 2008). In line with a previous study by Je et al, no MED12 exon 2 mutations in any haematological malignancies were detected (Je et al, 2012).
Taken together, screening of 1158 samples representing various tumour types revealed five MED12 exon 2 mutations; three in uterine leiomyosarcomas and two in CRC samples. Three mutations in the confirmed uterine leiomyosarcomas indicate that these tumours may have developed through a leiomyoma precursor. Supported by the observations by Je et al and The Cancer Genome Atlas Network, we also suggest that MED12 exon 2 mutations may contribute, albeit rarely, to CRC tumorigenesis (Je et al, 2012; The Cancer Genome Atlas Network, 2012). Interestingly, Barbieri et al recently reported recurrent MED12 mutations affecting codon 1224 in 5 out of 111 (4.5%) prostate adenocarcinomas studied (Barbieri et al, 2012). It remains to be seen whether additional mutations in other parts of this large gene have a role in the development of various tumour types. The high MED12 exon 2 mutation frequency observed in uterine leiomyomas seem to associate with the location of the tumours in the uterus and also with their benign nature. The mechanistic details and the affected molecular pathways through which these extremely specific mutations promote tumorigenesis need to be unravelled at the molecular level utilising, for example, gene expression analyses, mouse models, and in vitro functional experiments. It is likely they alter a very specific function of the MED12 protein providing the cells with a growth advantage especially in the uterus.
Acknowledgments
We would like to thank Inga-Lill Svedberg, Mairi Kuris, Sini Nieminen, and Anni Tieva for their technical assistance, and Drs Soili Kytölä and Päivi Heikkilä for their help in sample collection. This study was supported by the Academy of Finland (Center of Excellence in Cancer Genetics Research), the Sigrid Jusélius Foundation, the Cancer Society of Finland, and the University of Helsinki Funds (personal grant to NM).
Footnotes
Supplementary Information accompanies the paper on British Journal of Cancer website (http://www.nature.com/bjc)
Supplementary Material
References
- Barbieri CE, Baca SC, Lawrence MS, Demichelis F, Blattner M, Theurillat JP, White TA, Stojanov P, Van Allen E, Stransky N, Nickerson E, Chae SS, Boysen G, Auclair D, Onofrio RC, Park K, Kitabayashi N, MacDonald TY, Sheikh K, Vuong T, Guiducci C, Cibulskis K, Sivachenko A, Carter SL, Saksena G, Voet D, Hussain WM, Ramos AH, Winckler W, Redman MC, Ardlie K, Tewari AK, Mosquera JM, Rupp N, Wild PJ, Moch H, Morrissey C, Nelson PS, Kantoff PW, Gabriel SB, Golub TR, Meyerson M, Lander ES, Getz G, Rubin MA, Garraway LA (2012) Exome sequencing identifies recurrent SPOP, FOXA1 and MED12 mutations in prostate cancer. Nat Genet 44: 685–689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- The Cancer Genome Atlas Network (2012) Comprehensive molecular characterization of human colon and rectal cancer. Nature 487: 330–337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christacos NC, Quade BJ, Dal Cin P, Morton CC (2006) Uterine leiomyomata with deletions of Ip represent a distinct cytogenetic subgroup associated with unusual histologic features. Genes Chromosomes Cancer 45: 304–312 [DOI] [PubMed] [Google Scholar]
- Dave UP, Akagi K, Tripathi R, Cleveland SM, Thompson MA, Yi M, Stephens R, Downing JR, Jenkins NA, Copeland NG (2009) Murine leukemias with retroviral insertions at Lmo2 are predictive of the leukemias induced in SCID-X1 patients following retroviral gene therapy. PLoS Genet 5: e1000491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day Baird D, Dunson DB, Hill MC, Cousins D, Schectman JM (2003) High cumulative incidence of uterine leiomyoma in black and white women: ultrasound evidence. Am J Obstet Gynecol 188: 100–107 [DOI] [PubMed] [Google Scholar]
- Galbraith MD, Donner AJ, Espinosa JM (2010) CDK8: a positive regulator of transcription. Transcription 1: 4–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gobert V, Osman D, Bras S, Auge B, Boube M, Bourbon HM, Horn T, Boutros M, Haenlin M, Waltzer L (2010) A genome-wide RNA interference screen identifies a differential role of the mediator CDK8 module subunits for GATA/RUNX-activated transcription in Drosophila. Mol Cell Biol 30: 2837–2848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Je EM, Kim MR, Min KO, Yoo NJ, Lee SH (2012) Mutational analysis of MED12 exon 2 in uterine leiomyoma and other common tumors. Int J Cancer 131(6): E1044–E1047 [DOI] [PubMed] [Google Scholar]
- Keightley MC, Layton JE, Hayman JW, Heath JK, Lieschke GJ (2011) Mediator subunit 12 is required for neutrophil development in zebrafish. PLoS One 6: e23845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilpinen S, Autio R, Ojala K, Iljin K, Bucher E, Sara H, Pisto T, Saarela M, Skotheim RI, Bjorkman M, Mpindi JP, Haapa-Paananen S, Vainio P, Edgren H, Wolf M, Astola J, Nees M, Hautaniemi S, Kallioniemi O (2008) Systematic bioinformatic analysis of expression levels of 17 330 human genes across 9783 samples from 175 types of healthy and pathological tissues. Genome Biol 9: R139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S, Xu X, Hecht A, Boyer TG (2006) Mediator is a transducer of Wnt/beta-catenin signaling. J Biol Chem 281: 14066–14075 [DOI] [PubMed] [Google Scholar]
- Knuesel MT, Meyer KD, Donner AJ, Espinosa JM, Taatjes DJ (2009) The human CDK8 subcomplex is a histone kinase that requires Med12 for activity and can function independently of mediator. Mol Cell Biol 29: 650–661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowski DN, Bartnitzke S, Loning T, Drieschner N, Helmke BM, Bullerdiek J (2012) MED12 mutations in uterine fibroids-their relationship to cytogenetic subgroups. Int J Cancer 131(7): 1528–1536 [DOI] [PubMed] [Google Scholar]
- McGuire MM, Yatsenko A, Hoffner L, Jones M, Surti U, Rajkovic A (2012) Whole exome sequencing in a random sample of North American women with leiomyomas identifies MED12 mutations in majority of uterine leiomyomas. PLoS One 7: e33251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitelman F (1998) Catalog of chromosomal aberration in cancer. Wiley Liss: New York [Google Scholar]
- Mäkinen N, Heinonen HR, Moore S, Tomlinson IP, van der Spuy ZM, Aaltonen LA (2011b) MED12 exon 2 mutations are common in uterine leiomyomas from South African patients. Oncotarget 2: 966–969 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mäkinen N, Mehine M, Tolvanen J, Kaasinen E, Li Y, Lehtonen HJ, Gentile M, Yan J, Enge M, Taipale M, Aavikko M, Katainen R, Virolainen E, Böhling T, Koski TA, Launonen V, Sjöberg J, Taipale J, Vahteristo P, Aaltonen LA (2011a) MED12, the mediator complex subunit 12 gene, is mutated at high frequency in uterine leiomyomas. Science 334: 252–255 [DOI] [PubMed] [Google Scholar]
- Parker WH (2007) Etiology, symptomatology, and diagnosis of uterine myomas. Fertil Steril 87: 725–736 [DOI] [PubMed] [Google Scholar]
- Pérot G, Croce S, Ribeiro A, Lagarde P, Velasco V, Neuville A, Coindre JM, Stoeckle E, Floquet A, Macgrogan G, Chibon F (2012) MED12 alterations in both human benign and malignant uterine soft tissue tumors. PLoS One 7: e40015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polakis P (2000) Wnt signaling and cancer. Genes Dev 14: 1837–1851 [PubMed] [Google Scholar]
- Rocha PP, Scholze M, Bleiss W, Schrewe H (2010) Med12 is essential for early mouse development and for canonical Wnt and Wnt/PCP signaling. Development 137: 2723–2731 [DOI] [PubMed] [Google Scholar]
- Taatjes DJ (2010) The human Mediator complex: a versatile, genome-wide regulator of transcription. Trends Biochem Sci 35: 315–322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tallini G, Vanni R, Manfioletti G, Kazmierczak B, Faa G, Pauwels P, Bullerdiek J, Giancotti V, Van Den Berghe H, Dal Cin P (2000) HMGI-C and HMGI(Y) immunoreactivity correlates with cytogenetic abnormalities in lipomas, pulmonary chondroid hamartomas, endometrial polyps, and uterine leiomyomas and is compatible with rearrangement of the HMGI-C and HMGI(Y) genes. Lab Invest 80: 359–369 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.