Abstract
Neural cancers display cellular hierarchies with self-renewing tumorigenic cancer stem cells (CSCs) at the apex. Instructive cues to maintain CSCs are generated by both intrinsic networks and the niche microenvironment. The CSC-microenvironment relationship is complex as CSCs can modify their environment and extrinsic forces induce plasticity in the cellular hierarchy.
Neural cancers represent a diverse set of over 100 diagnoses that collectively cause significant morbidity and mortality. The most common primary brain tumor, glioblastoma multiforme (GBM), is nearly universally lethal with a five-year survival rate of 2%, and current therapies only offer palliative relief. As the name implies, GBMs display striking cellular heterogeneity, which may be responsible for the frequent progression or recurrence in nodular or regional patterns observed in patients. Models to explain the origin of tumor cellular heterogeneity include a stochastic model in which tumor cells have a random probability of developing mutations to permit tumor maintenance, and a hierarchical model in which sustained tumor growth is restricted to selected subpopulations, called cancer stem cells (CSCs). Recently, the Dick and Greaves laboratories presented leukemia studies that suggest these two models are not mutually exclusive, but rather that tumor cell evolution may involve simultaneous changes governed by both models (commented on in Burgess, 2011). Based on the concept that cancers are organ systems governed by aberrant development and homeostasis, the CSC hypothesis is consistent with the existence of cellular populations that maintain tumor growth through self-renewal and generation of the larger tumor bulk in cooperation with a supportive microenvironment. Numerous laboratories have demonstrated the existence of CSCs in several high-grade primary brain tumors (gliomas, medulloblastomas, and ependymomas) and neuroblastomas, but CSCs from lower grade tumors are less clearly defined. CSCs in neural cancers display similarities with neural stem cells (NSCs) in terms of their capacity for self-renewal and sphere-forming growth in serum-free conditions, in their stem cell marker expression, and multi-lineage differentiation potential. However, CSCs and NSCs can also be distinguished based on aberrant expression of differentiation markers, chromosomal abnormalities, and tumor formation. It is important to note that the CSC hypothesis does not require a stem cell cell-of-origin and different cancers appear to originate from stem cells, lineage restricted precursors, or more than one cell type.
Sizing up the roster
Several sources of confusion have challenged the concepts inherent to the CSC model, including discrepancies in the nomenclature utilized by members of the field. Some reports have employed the term ‘tumor initiating cells’, but this designation may inadvertently confuse the idea of which cell is responsible for originating the disease, as opposed to reflecting the capacity of isolated cancer cells to replicate a tumor in vivo. In contrast, CSCs are better described as tumor propagating cells that also generate the non-tumorigenic tumor cells that do not contribute to secondary transplantation assays. For the scope of this review, the fraction of tumor forming cells in a transplantation assay will be referred to as CSCs, but it should be acknowledged that the premise that the bulk tumor arises exclusively downstream of CSCs has not been fully validated in most cancers. Finally, it is also important to consider that CSCs need not be rare and the ability to prospectively identify and enrich for CSCs using a single set of markers (i.e. an immunophenotype) is not universal for any cancer, perhaps reflecting the intertumoral diversity within cancer subtypes. Several genetic and expression studies, including those of Philips, Parsons and the Cancer Genome Atlas, have distinguished at least two tumor GBM subtypes (proneural and mesenchymal) that are enriched for different genetic lesions, but the relationship between these tumor subgroups and CSCs remains to be defined (reviewed in Huse et al., 2011). Despite these difficulties, neural CSCs have been heavily studied due to their implied role in promoting resistance to conventional therapies and their potential contribution to tumor recurrence.
As CSCs may contribute to treatment failure, elucidating the molecular pathways that regulate CSC maintenance and survival may inform the development of novel therapies. Intrinsic regulation of CSCs occurs through key proliferative and survival pathways including c-Myc, Oct4 (POU5F1), Olig2, and Bmi1 (reviewed in Li et al., 2009b). Extrinsically, CSCs are regulated by growth factors as well as cell-to-cell and cell-to-extracellular matrix (ECM) interactions. Extrinsic interactions occur in distinct anatomical locations, termed microenvironments or niches. These niches are responsible for CSC maintenance and may have a role in promoting therapeutic resistance (Rosen and Jordan, 2009). In the context of neural CSCs, the concept of a niche is likely to be mitogenic rather than quiescent, as it remains unclear if a quiescent neural CSC exists. Understanding CSCs and their niches in one cancer may have broad implications for other tumors as the CSC niche for one tumor type is generally not unique and regulatory components present in one cancer microenvironment can often be identified in other CSC niches. Moving beyond the idea that niches regulate CSC maintenance, recent work has demonstrated that the bidirectional communication between CSCs and their microenvironment has the ability to influence cell fate in GBMs (Bautch, 2010; Heddleston et al., 2009; Hjelmeland et al., 2010). Better defining how CSCs interact with their microenvironment has direct implications on the basic biology of the tumor and is likely to provide insight into the design of more effective therapies.
Several models have been employed to interrogate the nature of communication between CSCs and the niche. Currently, however, no system exists to fully recapitulate the genetic and epigenetic diversity representative of the human condition. Immunodeficient animal models are often used for in vivo assays, but this approach fails to replicate the immune system component that is present in human patients, and that may serve as a powerful driver of cellular hierarchy. Highly immunocompromised systems, such as the NSG-SCID model, have been proposed as an improvement over other systems, largely due to the higher frequency of tumor growth in these more permissive systems. However, it has yet to be proven that such models better represent tumor growth patterns that occur in humans, and it is possible that their immunocompromised status may permit growth of tumor cells that would not occur in patients. In contrast, genetically engineered models may yield syngeneic hosts that eliminate the presence of artificial immune modulation and species differences in cytokine signaling, but still pose challenges with respect to their relative lack of cellular heterogeneity. While tumorigenesis can be classified into spontaneous models (e.g. the Ptch medulloblastoma model), and models that require manipulation may induce inflammation prior to tumor initiation (e.g. medulloblastomas and GBMs from the Nestin tv-a model). Furthermore, manipulated tumor models may contain cells that express stem cell markers but do not fully recapitulate tumor formation, and thus lack a predicted CSC hierarchy. Whether these differences reflect different tumor subtypes or suggest loss of hierarchy with different models has yet to be defined. Thus, no single model can provide for complete characterization of CSC-microenvironmental interplay, and thus achieving an appropriately bi-directional model will likely require the combination of complementary approaches.
Support from the sidelines
NSCs commonly reside in perivascular regions, which exhibit restricted oxygen availability and distinct ECM profiles. In parallel, neural CSCs are enriched in perivascular niches, and possibly also within regions of hypoxia and at the invasive edge of the tumor (Fig. 1). The perivascular niche is the best characterized CSC niche due to the established relationship between tumor growth and angiogenesis. In a variety of brain cancers, CSCs are located within close proximity to blood vessels, which facilitates the transfer of signals necessary for their maintenance (as demonstrated in a 2007 Cancer Cell report from Gilbertson and colleagues, who examined GBM, ependymoma, and medulloblastoma samples, reviewed in Gilbertson and Rich, 2007). In the same vein, Bao et al. provided evidence that GBM CSCs secrete vascular endothelial growth factor (VEGF) that stimulates endothelial cell growth to support a local vascular environment (reviewed in Gilbertson and Rich, 2007). In turn, endothelial cells express Notch ligands that may stimulate Notch receptors essential for CSC maintenance (Fan et al., 2010). Endothelial cells may also secrete nitric oxide to activate the Notch pathway (Charles et al., 2010). CSCs are also regulated through ECM receptors, such as integrin α6, that are enriched on GBM CSCs and promote their maintenance (Lathia et al., 2010). Continuing studies elucidating the potential signaling crosstalk at the cell surface between key proteins such as integrins, Notch, and growth factor receptors may clarify our understanding of CSC maintenance. The significance of the CSC niche is supported by studies from Bao and coauthors, which demonstrate that GBM CSCs display resistance to conventional therapies (reviewed in Gilbertson and Rich, 2007). This work was extended in a mouse model of medulloblastoma whereby tumor cells within the perivascular space were resistant to radiation via Akt signaling which, when targeted, resulted in greater radiation efficiency (Hambardzumyan et al., 2008). Thus, the perivascular niche may offer an indirect CSC therapeutic target, e.g. anti-VEGF interventions such as bevacizumab (reviewed in Gilbertson and Rich, 2007). The limited clinical success of bevacizumab, complicated by post-treatment invasion, warrants further understanding of perivascular regulation in GBM development and therapies.
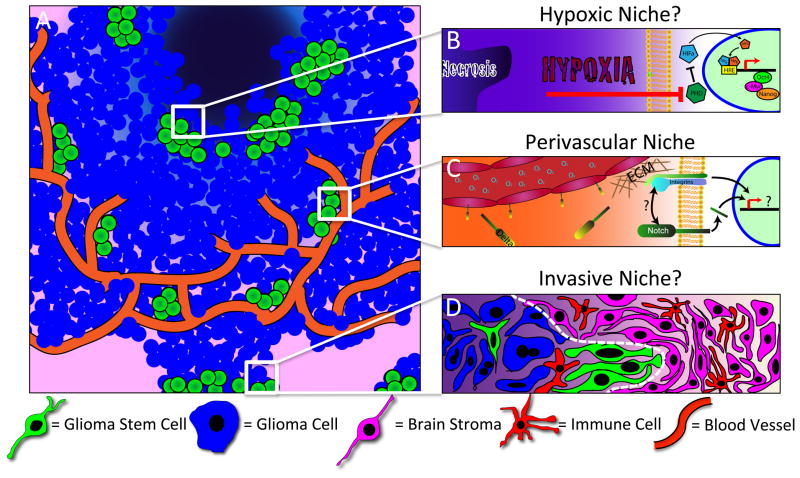
Figure 1. Microenvironmental factors modulate intrinsic cancer stem cell pathways.
Cancer stem cells (CSC) in neural tumors (A) are enriched in niches including the perivascular compartment (C), hypoxic regions (B) and possibly the invasive edge (D). (B) Regions of hypoxia in neural tumors have been well-characterized near areas of necrosis. Recent studies have suggested that low oxygen helps to maintain the CSC population, therefore supporting hypoxic regions as a niche for CSCs. Hypoxia modulates cell phenotypes via HIF signaling, which drives expression of stem cell related genes. (C) The perivascular niche is the most well-defined CSC niche in neural tumors. Experimental evidence has demonstrated that cells expressing integrin alpha 6 are localized in the perivascular region denoted by CD31-positive endothelial cells of the blood vessel. Interaction of the integrin cell surface proteins with the extracellular matrix (ECM) within the perivascular space supports CSC maintenance in gliomas. Notch-ligand interaction between endothelial cells of the blood vessels and the cancer cell is also crucial for CSC maintenance but the cross-talk between key signaling receptors such as integrins and Notch are yet to be fully elucidated. Inhibition of the Notch pathway has been shown to have detrimental effects on the CSC phenotype as well as survival following irradiation in gliomas. (D) The invasive front of neural tumors is thought to contain CSCs and provide an interface of interactions with immune cells and normal brain stroma. It is hypothesized that the microenvironment of these invasive regions supports CSC maintenance. However, the invasive niche has not been well characterized.
The metabolic demands of rapidly proliferating tumors often exceed local supply, and necessitating neovasculature, but newly formed vessels are often inefficient. Given the plasticity of cancer, which extends to the CSC compartment, recent work by Ricci-Vitiani et al. and Wang et al. suggests that GBM CSCs can directly participate in vasculogenic processes (reviewed by Bautch, 2010). These groups both identified endothelial cells in GBM microvessels that contained tumor-specific chromosomal aberrations, suggesting these endothelial cells were of tumor origin. Furthermore, CSCs isolated from GBMs could differentiate into endothelial-like cells both in vitro and in vivo, which suggested that the tumor cells were able to cross lineage boundaries via some degree of transdifferentiation. These findings may have profound implications on our understanding and therapeutic approaches to cancer. CSCs may be opportunistic, in that when key microenvironment components are not readily available, they can exhibit lineage plasticity and give rise to progeny that supply the deficient cell type. Theoretically, this process may also occur in other tumor components such as the stroma and tumor-related immune cells. Elucidation of phenotypic variation of CSCs with regards to other tumor components will provide much needed insight into this hypothesis and may help explain differences in normal and tumor-related stroma and immune compartments. However, these ideas remain attractive hypotheses that will require experimental confirmation.
Thriving under pressure
Hypoxia has been demonstrated to be a key factor in CSC maintenance in GBMs and neuroblastoma (Li et al., 2009a; Pietras et al., 2009; Seidel et al., 2010). As hypoxia promotes radioresistance and angiogenesis – both of which are considered CSC traits -- the negative impact of hypoxia on patient survival may be explained in part by increased maintenance of CSCs. Regions of hypoxia can arise within the tumor due to necrosis or the failure of tumor vasculature to efficiently exchange oxygen, even in the perivascular niche. Hypoxia may not only facilitate GBM CSC maintenance but also promote acquisition of stem-like characteristics within the tumor (Heddleston et al., 2009). Molecular responses to hypoxia are commonly governed by the hypoxia inducible factors (HIFs), which were linked to angiogenesis in a rat GBM model and shown to be important in regulating GBM cell survival (Acker et al., 2005). HIFs are also critical for GBM and neuroblastoma CSC maintenance and intimately linked to angiogenesis. Specifically, it has been shown that HIF1α regulates medulloblastoma and GBM CSCs (Soeda et al., 2009; Pistollato et al., 2010), while HIF2α is specifically expressed in neural CSCs (Li et al., 2009a; Pietras et al., 2009; Seidel et al., 2010) and promotes stem cell maintenance and CSC phenotypes (Heddleston et al., 2009). Stabilization of HIF2α protein under physiologic hypoxia or overexpression of nondegradable HIF2α increases self-renewal in vitro and tumor formation in vivo. This effect was observed not only in the initial CSC subpopulation but also in more differentiated non-stem tumor cells. Neural stem cells reside within low oxygen regions (Mohyeldin et al., 2010) and exhibit a growth dependence on blood vessels as shown by Temple and colleagues in a 2004 Science paper (reviewed by Gilbertson and Rich, 2007), suggesting that these key niches are shared by both normal and neoplastic neural stem cells and that extrinsic regulation from the microenvironment promotes plasticity within the neoplastic compartment. However, challenges still remain in the definitive anatomical identification of hypoxic niches due to the rapidly evolving composition of the tumor and lack of experimental reagents; but the molecular mechanism contributing to the CSC phenotype via hypoxia are being defined.
Regional and temporal perturbations of the microenvironment can profoundly modulate resident cellular physiology relative to steady state conditions. Attention has been paid to hypoxia within this context but other extracellular states may also be important. Acidic stress, which is not uniform across a tumor, induces plasticity in non-stem glioma cells making them more GBM CSC-like (Hjelmeland et al., 2010). Following exposure to physiologic tumor pH (6.5), nonstem glioma cells upregulate stem cell associated genes and display increased growth and tumorigenic potential. Similar to the hypoxic situation, these studies suggest that culturing cells under physiological tumor conditions rather than artificial conditions that promote unrestricted proliferation may maintain cellular heterogeneity better and perhaps improve the ability to model tumors. Proliferation in cell culture is limited by glucose availability, leading to routine use of extremely supraphysiologic glucose concentrations. The availability of such metabolic factors, such as lactate in addition to glucose, could significantly affect the intrinsic phenotype in a manner similar to what is seen in response to hypoxia and acidic stress. The elucidation of how the microenvironment regulates CSC maintenance is not only critical to our understanding of tumor biology but also provides an opportunity to unravel mechanisms of therapeutic resistance. Signals provided by both the perivascular and hypoxic niches are likely contributing factors, highlighting the potential of targeting the CSCs through therapeutic modalities designed to disrupt the niche.
Keeping an eye on the blindside
A hallmark of GBMs and other primary brain tumors is their propensity to invade into normal brain tissue, which prevents curative surgical resection. As CSC models may display in vivo invasion similar to human tumors (Wakimoto et al., 2009), it is tempting to speculate that the invasive edge may function as another CSC niche. Detailed examination of this hypothesis has thus far been limited by a variety of factors, not the least of which is the challenge of detecting and visualizing the invasive edge in vivo over time. Other key questions also remain open matters, such as whether CSCs migrate faster than non-stem tumor cells. Technical advances such as slice culture and multiphoton imaging may provide the tools necessary to better elucidate this putative niche. Dissecting this niche has implications beyond CSC maintenance, as anti-invasive therapies are likely to be useful in combination with traditional GBM therapeutic paradigms. There is a risk that CSCs located at the invasive edge will remain following surgical resection, given that tumor cells likely exist beyond the margin detectible by current imaging techniques, and potential cost to normal brain tissue must weighed against removal of the tumor margin. Additionally, better understanding of this niche may clarify the ability for one niche to repopulate or recreate another niche after therapy. This analysis of the invasive edge may also represent an opportunity to understand how immune cells that reside within the tumor interact with the tumor components (Markovic et al., 2009), and possibly with the CSCs, specifically. Constructing models to address this relationship at the tumor interface will address another major limitation of our current knowledge.
The recent work discussed in this review underscores our evolving understanding of the tumor microenvironment, in that beyond functioning as a regulator of CSC maintenance, components of the niche may also serve as a putative catalyst of the CSC phenotype. The relationship of tumor cells to the niche is far from being fully defined. In rapidly evolving neural tumors, it remains unresolved if CSCs develop a niche or if a niche exists that recruits CSCs. Additional in vivo studies utilizing high resolution imaging modalities and reporter systems may help resolve this cause and effect relationship. Another relationship, which is yet to be fully defined, is the interaction of tumor cells with the stroma and it is likely that there exists a bidirectional relationship. The contribution of CSCs to the invading tumor margin and possible interactions with the immune system are additional relationships that are only beginning to be investigated. The plasticity present within tumors has long been noted but recent work suggests that even CSCs may be plastic and can be viewed as opportunistic entities, readily able to contribute to their surrounding microenvironment. Gaining insight into communication mechanisms between CSCs and the microenvironment will be informative with regards to the contribution of CSCs to tumor initiation, growth, maintenance, invasion, and therapeutic resistance. Additionally, these communication mechanisms are likely to drive the development of more effective therapies for malignant tumors such as GBMs.
Acknowledgments
We regret that space limitations prevent a full representation of the many important studies by investigators of the microenvironment and CSCs. We thank members of the Rich lab for their constructive criticism. Work in the Rich lab is supported by the National Brain Tumor Society, Goldhirsh Foundation, and NIH grants NS054276 (JNR), CA129958 (JNR), CA116659 (JNR), CA154130 (JNR), and National Research Service Awards CA142159 (JDL) and NS058042 (MV). JNR is a Damon Runyon-Lilly Clinical Investigator supported by the Damon Runyon Cancer Research Foundation. JDL and MV are supported by an American Brain Tumor Association Basic Research Fellowship (sponsored by the Joelle Syverson Fund, JDL).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Acker T, Diez-Juan A, Aragones J, Tjwa M, Brusselmans K, Moons L, Fukumura D, Moreno-Murciano MP, Herbert JM, Burger A, et al. Cancer Cell. 2005;8:131–141. doi: 10.1016/j.ccr.2005.07.003. [DOI] [PubMed] [Google Scholar]
- Bautch VL. Nature. 2010;468:770–771. doi: 10.1038/468770a. [DOI] [PubMed] [Google Scholar]
- Burgess DJ. Nature Reviews Cancer. 2011;11:153. doi: 10.1038/nrc3019. [DOI] [PubMed] [Google Scholar]
- Charles N, Ozawa T, Squatrito M, Bleau AM, Brennan CW, Hambardzumyan D, Holland EC. Cell Stem Cell. 2010;6:141–152. doi: 10.1016/j.stem.2010.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan X, Khaki L, Zhu TS, Soules ME, Talsma CE, Gul N, Koh C, Zhang J, Li YM, Maciaczyk J, et al. Stem Cells. 2010;28:5–16. doi: 10.1002/stem.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilbertson RJ, Rich JN. Nature Reviews. 2007;7:733–736. doi: 10.1038/nrc2246. [DOI] [PubMed] [Google Scholar]
- Hambardzumyan D, Becher OJ, Rosenblum MK, Pandolfi PP, Manova-Todorova K, Holland EC. Genes Dev. 2008;22:436–48. doi: 10.1101/gad.1627008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heddleston JM, Li Z, McLendon RE, Hjelmeland AB, Rich JN. Cell Cycle. 2009;8:3274–3284. doi: 10.4161/cc.8.20.9701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hjelmeland AB, Wu Q, Heddleston JM, Choudhary GS, Macswords J, Lathia JD, McLendon R, Lindner D, Sloan A, Rich JN. Cell Death Differ. 2011;18:829–840. doi: 10.1038/cdd.2010.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huse JT, Phillips HS, Brennan CW. Glia. 2011 doi: 10.1002/glia.21165. [DOI] [PubMed] [Google Scholar]
- Lathia JD, Gallagher J, Heddleston JM, Wang J, Eyler CE, Macswords J, Wu Q, Vasanji A, McLendon RE, Hjelmeland AB, Rich JN. Cell Stem Cell. 2010;6:421–432. doi: 10.1016/j.stem.2010.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Bao S, Wu Q, Wang H, Eyler C, Sathornsumetee S, Shi Q, Cao Y, Lathia J, McLendon RE, et al. Cancer Cell. 2009a;15:501–513. doi: 10.1016/j.ccr.2009.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Wang H, Eyler CE, Hjelmeland AB, Rich JN. J Biol Chem. 2009b;284:16705–16709. doi: 10.1074/jbc.R900013200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markovic DS, Vinnakota K, Chirasani S, Synowitz M, Raguet H, Stock K, Sliwa M, Lehmann S, Kälin R, van Rooijen N, et al. Proc Natl Acad Sci U S A. 2009;106:12530–12535. doi: 10.1073/pnas.0804273106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohyeldin A, Garzon-Muvdi T, Quinones-Hinojosa A. Cell Stem Cell. 2010;7:150–161. doi: 10.1016/j.stem.2010.07.007. [DOI] [PubMed] [Google Scholar]
- Pietras A, Hansford LM, Johnsson AS, Bridges E, Sjolund J, Gisselsson D, Rehn M, Beckman S, Noguera R, Navarro S, et al. Proc Natl Acad Sci U S A. 2009;106:16805–16810. doi: 10.1073/pnas.0904606106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pistollato F, Rampazzo E, Persano L, Abbadi S, Frasson C, Denaro L, D’Avella D, Panchision DM, Della Puppa A, Scienza R, et al. Stem Cells. 2010;28:1918–1929. doi: 10.1002/stem.518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen JM, Jordan CT. Science. 2009;324:1670–1673. doi: 10.1126/science.1171837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidel S, Garvalov BK, Wirta V, von Stechow L, Schanzer A, Meletis K, Wolter M, Sommerlad D, Henze AT, Nister M, et al. Brain. 2010;133:983–995. doi: 10.1093/brain/awq042. [DOI] [PubMed] [Google Scholar]
- Soeda A, Park M, Lee D, Mintz A, Androutsellis-Theotokis A, McKay RD, Engh J, Iwama T, Kunisada T, Kassam AB, et al. Oncogene. 2009;28:3949–3959. doi: 10.1038/onc.2009.252. [DOI] [PubMed] [Google Scholar]
- Wakimoto H, Kesari S, Farrell CJ, Curry WT, Jr, Zaupa C, Aghi M, Kuroda T, Stemmer-Rachamimov A, Shah K, Liu TC, et al. Cancer Res. 2009;69:3472–3481. doi: 10.1158/0008-5472.CAN-08-3886. [DOI] [PMC free article] [PubMed] [Google Scholar]