Abstract
Visceral leishmaniasis (kala-azar) is a disease caused by protozoa of genus Leishmania. It is currently regarded as the second most dreaded parasitic disease, next to malaria. There have been very few case reports of visceral leishmaniasis among the renal transplant recipients. We present a renal allograft recipient with symptoms of fever, sore throat, hoarseness of voice, lymphadenopathy, splenomegaly, and pancytopenia after 7 years post-transplant period. On investigating, he was diagnosed to have extensive visceral leishmaniasis with laryngeal involvement. Despite extensive PubMed literature search, we could not find any case report of postrenal transplant visceral and laryngeal leishmaniasis and to the best of our knowledge this is the first case report of this kind.
Keywords: Postrenal transplant, visceral leishmaniasis, vocal cord
Introduction
Leishmaniasis, also known as kala-azar (literally meaning black fever or black disease) is a zoonosis caused by protozoan of the genus Leishmania, which is transmitted by bite of insect vector phlebotomus sandfly. Visceral leishmaniasis is an endemic infectious disease in India and Nepal. Still it is a rare infection in postrenal transplant setting with atypical presentation leading to delayed diagnosis and considerable morbidity and mortality. High index of suspicion, particularly in patients from endemic areas, is required in early diagnosis and improve the disease outcome. We report a case of 50-year-old male renal allograft recipient who presented to us with fever, sore throat, hoarseness of voice, lymphadenopathy, and splenomegaly and was diagnosed to have visceral and laryngeal leishmaniasis.
Case Report
A 50-year-old male, resident of Nepal underwent live-related renal transplant in 2003 for a basic renal disease – membranous glomerulopathy. Donor was his wife. No induction was used and he was on maintenance immunosuppression with cyclosporine, azathioprine, and prednisolone. Post-transplant period was uneventful and he was on regular follow up. He presented after 7 years with the history of sore throat of 3 months duration and fever of 1-month duration. He consulted a local ENT surgeon 3 months before presenting to us and was told to have vocal cord polyp. For this conservative management was advised. He was also seen by a local practitioner for fever and treated empirically with ceftriaxone 2 g daily for 7 days, however fever persisted.
On examination he was febrile, had bilateral cervical lymphadenopathy measuring up to 3 cm. He also had nontender, firm splenomegaly. Renal allograft was normal on examination. Blood investigations revealed serum creatinine 1.1 mg/dL, total bilirubin 1.5 mg/dL, direct bilirubin 0.5 mg/dL, AST 108 U/L, ALT 91 U/L, total protein 5.0 g/dL, serum albumin 2.3 g/dL, and serum globulin 2.7 g/dL. HIV, HBsAg and anti-HCV tests were negative by ELISA. Urinalysis was normal. Blood and urine cultures and Widal test were negative. He had pancytopenia (Hb, 9.3 g/dL; total leukocyte count, 1,560 cells/mm3, platelet count, 85 × 103/μL). Reticulocyte count was 0.5%. Peripheral smear showed a normocytic normochromic picture with leukopenia and thrombocytopenia. Bone marrow biopsy showed hypocellular marrow spaces with proportionate reduction of normal hemopoetic elements. In addition, there were intra- and extracellular Leishman–Donovan bodies (amastigote form) [Figure 1]. X-ray chest was normal. Ultrasound abdomen showed splenomegaly (spleen size, 14 cm) and multiple enlarged retroperitoneal lymph nodes. Renal allograft was normal.
Figure 1.
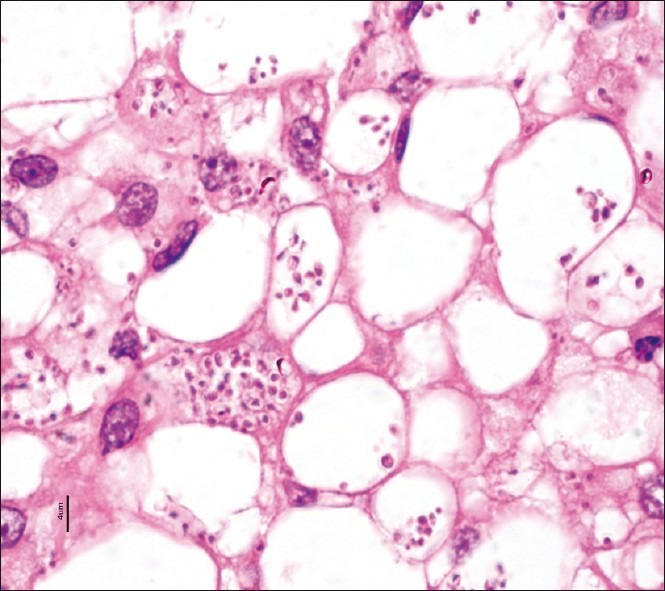
Bone marrow trephine biopsy show intra- and extracellular amastigote forms of leishmaniasis (H and E stain, ×100)
Excisional biopsy of cervical lymph node revealed complete replacement of nodal architecture by infectious pathology and was studded with intra-and extracellular amastigote form of protozoa (Leishman–Donovan bodies) accompanied with plasma cell response. He was also evaluated for hoarseness of voice and sore throat by ENT surgeon. Microlaryngoscopy showed irregular lesion on the left vocal cord, that was diffuse and involving full length. Biopsy of the lesion showed dense ulceration with exudate. There were numerous intra- and extracellular Leishman–Donovan bodies (amastigote form) and accompanying acute inflammatory cells [Figure 2]. Giemsa, PAS, and silver stain in both lymph node and vocal cord biopsy confirmed the organisms as Leishmaniasis.
Figure 2.
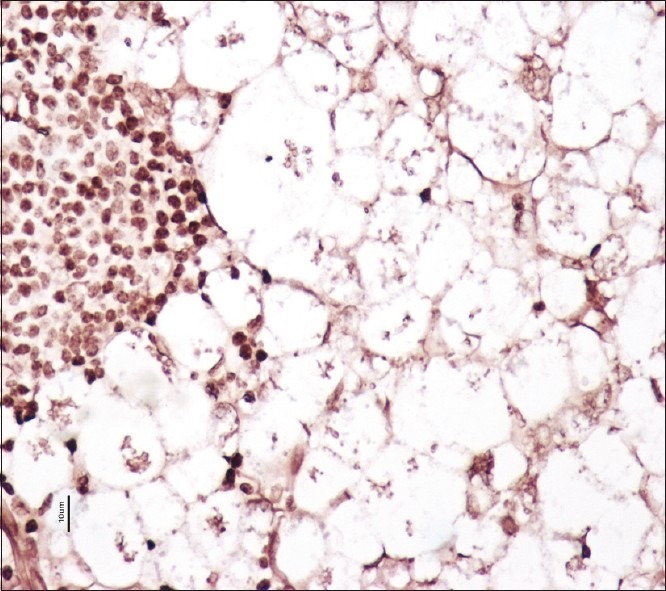
Vocal cord biopsy shows numerous extracellular amastigote forms of leishmaniasis (PASM stain, ×40)
He was started on treatment with liposomal amphotericin B. He completed total cumulative dose of 2 g IV Amphotericin B. The fever subsided and pancytopenia improved. After 8 months of follow-up, patient has remained asymptomatic without any further recurrence of the disease.
Discussion
Leishmaniasis is endemic in India, Bangladesh, Nepal, parts of Africa, southwest Asia, Spain, as well as other Mediterranean coast. In Indian subcontinent, transmission is anthroponotic and humans are the reservoir. Cell-mediated immunity determines the outcome of infection, and hence only minority of infected individuals develop clinically apparent disease. The immunological response, particularly interleukin-12 driven Th1 response has been widely accepted as promoting protective immunity against all species of leishmania. Immunosuppressive drugs used in transplant patients hamper T-cell activation and proliferation, thereby altering defense mechanisms against intracellular microorganisms. With increasing frequency of renal transplantation as the preferred renal replacement treatment modality, several cases of visceral leishmaniasis are being detected in renal transplant recipients. Among the organ transplant leishmaniasis disease, renal transplant patients are more commonly affected than others.[1] The immunosuppressive regimen used that is most commonly seen with disease is: Cyclosporine plus azathioprine and steroids.[1] The total number of cases reported in renal transplant is less than 100.[2]
Hernandez-Perez et al. reported five cases of leishmaniasis in solid organ transplant recipient out of which three were renal transplant recipients.[3] Oliveira et al. reported eight cases (1%) of leishmaniasis in renal transplant recipients.[4] Veroux et al. reported five renal transplant recipients presenting in early post-transplant period and eventually leading to graft loss in two.[2] The median time between transplantation and the onset of disease is 18 months.[1] It presents with fever (94%), hepatomegaly (42%), splenomegaly (75%), and pancytopenia (85%).[1] Lymphadenopathy in not commonly seen in transplant patients.[1] Our patient had classical presentation of fever, splenomegaly, pancytopenia suggesting bone marrow suppression and in addition, lymphadenopathy. He had the deranged liver function test in form of mild hyperbilirubinemia, hypoalbuminemia, and elevated AST and ALT.
Definitive diagnosis of visceral leishmaniasis requires demonstration of parasite. Histopathological diagnosis rests on visualization of amastigotes, which are spherical, or ovoid bodies measuring 1 to 5 μm. Culture requires inoculation in Novy-McNeal-Nicolle or other parasitic growth media.[5] Other diagnostic methods include serum antibody tests using IFA, ELISA, or DAT. The diagnosis of VL and ML in our case was done on the basis of biopsy of the lymph node, bone marrow, and vocal cord lesion, which provided unequivocal evidence of Leishman–Donovan bodies.
Laryngeal involvement in leishmaniasis is a rare presentation even in general population.[6–11] It can either present with other systemic manifestations or isolated laryngeal involvement.[7] Mucosal involvement of the nose, oral cavity, pharynx, or larynx occurs in 2–5% of persons infected with Leishmania braziliensis.[11] Nandy et al. described three cases of post kala-azar dermal leishmaniasis presenting with hoarseness of voice and biopsy of nodular lesions on vocal cord confirmed leishmaniasis in nontransplant patients.[10] In separate reports, authors have described isolated laryngeal involvement in immunocompetent patients.[6,11] Some of the patients have presented like mass lesions simulating neoplasia. Despite extensive pubmed literature search, we could not find a report of vocal cord leishmaniasis in a renal allograft recipient.
Agents used for treatment of visceral leishmaniasis include amphotericin B, pentavalent antimonial drugs, paromomycin, and miltefosine. Liposomal amphotericin B has highest therapeutic efficacy and it is safest among all these,[12,13] although conventional amphotericin B has considerable renal toxicity. In India and Nepal, high level of resistance to antimonial drugs has been observed and hence amphotericin B is being used as first line agents.[14–16]
Although leishmaniasis is a rare disease in a post-transplant setting even in endemic areas, it has got high morbidity and mortality in untreated cases. Clinical presentation may be atypical and high index of suspicion is a prerequisite for timely evaluation and appropriate treatment which can be life saving.
Acknowledgments
We sincerely thank Mrs. Tulasi Kumari, Mrs. Hema Nagaraj, Mr. Nagaraj, and rest for their outstanding technical support in Histopathology section.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
References
- 1.Antinori S, Cascio A, Parravicini C, Bianchi R, Corbellino M. Leishmaniasis among organ transplant recipients. Lancet Infect Dis. 2008;8:191–9. doi: 10.1016/S1473-3099(08)70043-4. [DOI] [PubMed] [Google Scholar]
- 2.Veroux M, Corona D, Giuffrida G, Cacopardo B, Sinagra N, Tallarita T, et al. Visceral leishmaniasis in the early post-transplant period after kidney transplantation: Clinical features and therapeutic management. Transpl Infect Dis. 2010;12:387–91. doi: 10.1111/j.1399-3062.2010.00520.x. [DOI] [PubMed] [Google Scholar]
- 3.Hernández-Pérez J, Yebra-Bango M, Jiménez-Martínez E, Sanz-Moreno C, Cuervas-Mons V, Alonso Pulpón L, et al. Visceral leishmaniasis (kala-azar) in solid organ transplantation: Report of five cases and review. Clin Infect Dis. 1999;29:918–21. doi: 10.1086/520457. [DOI] [PubMed] [Google Scholar]
- 4.Oliveira CM, Oliveira ML, Andrade SC, Girão ES, Ponte CN, Mota MU, et al. Visceral leishmaniasis in renal transplant recipients: Clinical aspects, diagnostic problems, and response to treatment. Transplant Proc. 2008;40:755–60. doi: 10.1016/j.transproceed.2008.02.039. [DOI] [PubMed] [Google Scholar]
- 5.Sundar S, Rai M. Laboratory diagnosis of visceral leishmaniasis. Clin Diagn Lab Immunol. 2002;9:951–8. doi: 10.1128/CDLI.9.5.951-958.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abrams J, Bocker W. A case of leishmaniasis with isolated disease of the larynx. Laryngorhinootologie. 1992;71:142–4. doi: 10.1055/s-2007-997265. [DOI] [PubMed] [Google Scholar]
- 7.Granel B, Serratrice J, Christides C, Richard-Vitton T, Minodier P, Horshowski N, et al. Epiglottis involvement in a visceral leishmaniasis. J Infect. 2002;45:196–7. [PubMed] [Google Scholar]
- 8.Grant A, Spraggs PD, Grant HR, Bryceson AD. Laryngeal leismaniasis. J Laryngol Otol. 1994;108:1086–8. doi: 10.1017/s0022215100128968. [DOI] [PubMed] [Google Scholar]
- 9.Kumar B, Ghimire A, Karki S, Upadhyaya P. Primary laryngeal leishmaniasis: A rare case report. Indian J Pathol Microbiol. 2009;52:62–4. doi: 10.4103/0377-4929.44967. [DOI] [PubMed] [Google Scholar]
- 10.Nandy A, Addy M, Banerjee D, Guha SK, Maji AK, Saha AM. Laryngeal involvement during post kala-azar dermal leishmaniasis in India. Trop Med Int Health. 1997;2:371–3. doi: 10.1111/j.1365-3156.1997.tb00153.x. [DOI] [PubMed] [Google Scholar]
- 11.Schroeder D, Foguena AK, Cometta A, Greub G, Cavassini M. A 48-year-old man with laryngeal mass and vocal cord palsy. Clin Infect Dis. 2010;50:1635. doi: 10.1086/652858. 80-1. [DOI] [PubMed] [Google Scholar]
- 12.Bern C, Adler-Moore J, Berenguer J, Boelaert M, den Boer M, Davidson RN, et al. Liposomal amphotericin B for the treatment of visceral leishmaniasis. Clin Infect Dis. 2006;43:917–24. doi: 10.1086/507530. [DOI] [PubMed] [Google Scholar]
- 13.Sundar S, Mehta H, Suresh AV, Singh SP, Rai M, Murray HW. Amphotericin B treatment for Indian visceral leishmaniasis: Conventional versus lipid formulations. Clin Infect Dis. 2004;38:377–83. doi: 10.1086/380971. [DOI] [PubMed] [Google Scholar]
- 14.Murray HW. Treatment of visceral leishmaniasis in 2004. Am J Trop Med Hyg. 2004;71:787–94. [PubMed] [Google Scholar]
- 15.Rijal S, Chappuis F, Singh R, Bovier PA, Acharya P, Karki BM, et al. Treatment of visceral leishmaniasis in south-eastern Nepal: Decreasing efficacy of sodium stibogluconate and need for a policy to limit further decline. Trans R Soc Trop Med Hyg. 2003;97:350–4. doi: 10.1016/s0035-9203(03)90167-2. [DOI] [PubMed] [Google Scholar]
- 16.Sundar S, Jha TK, Thakur CP, Mishra M, Singh VP, Buffels R. Single-dose liposomal amphotericin B in the treatment of visceral leishmaniasis in India: A multicenter study. Clin Infect Dis. 2003;37:800–4. doi: 10.1086/377542. [DOI] [PubMed] [Google Scholar]


