Abstract
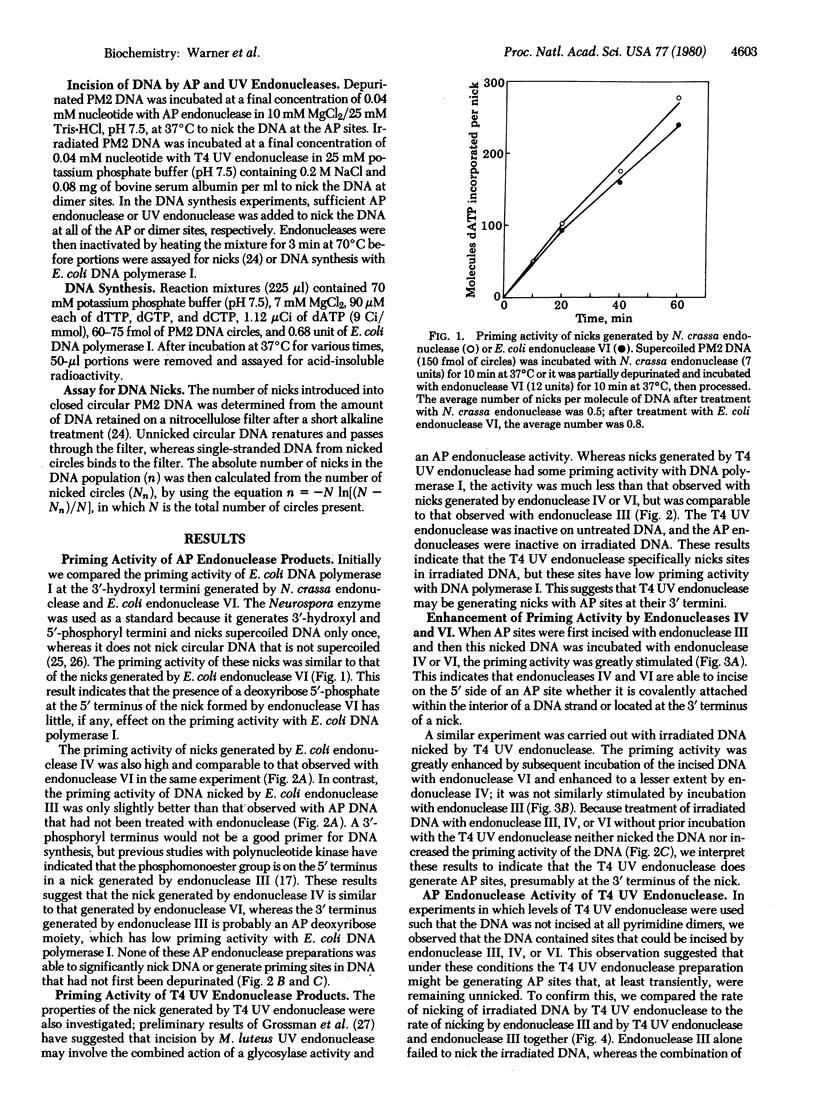
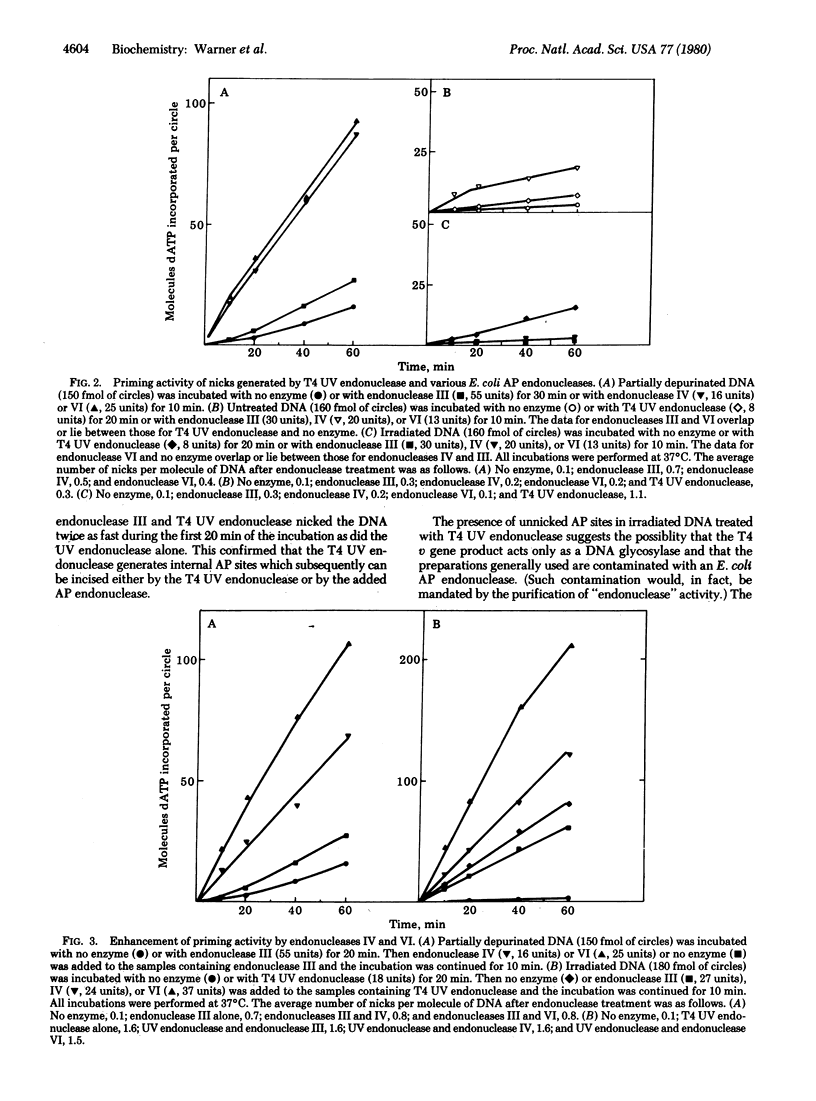
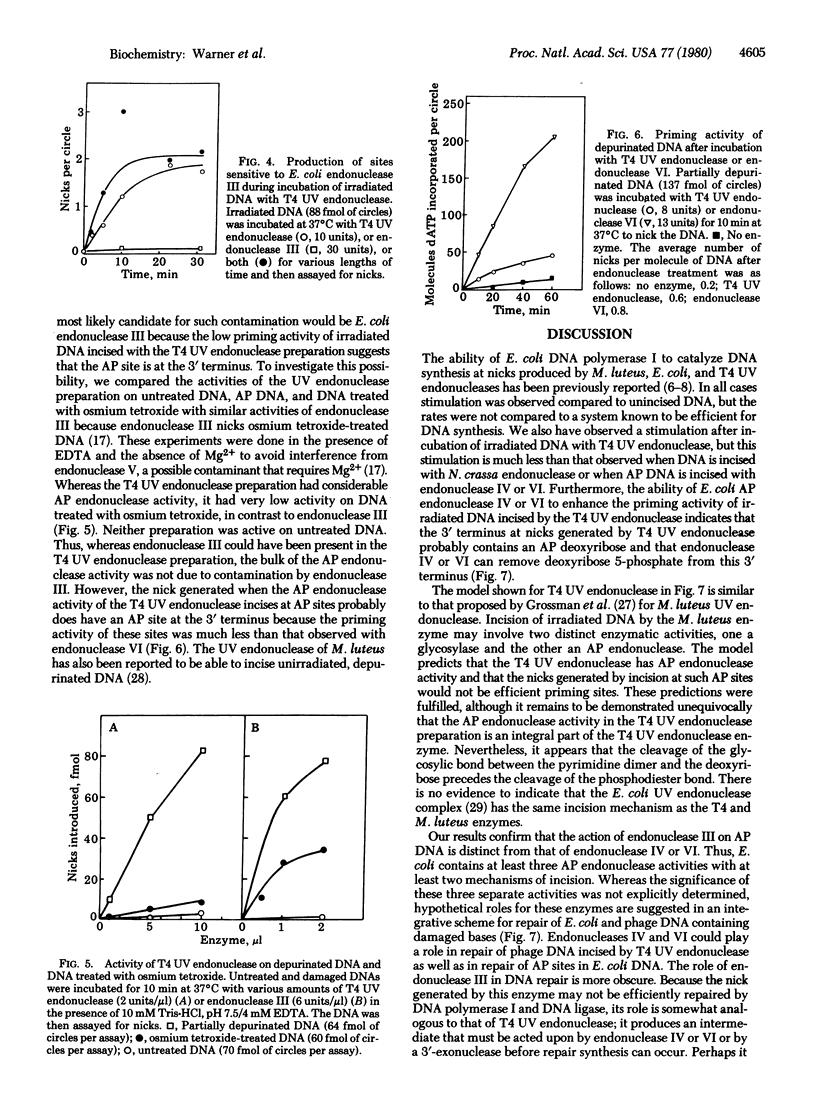
The characteristics of the nicks (single-strand breaks) introduced into damaged DNA by Escherichia coli endonucleases III, IV, and VI and by phage T4 UV endonuclease have been investigated with E. coli DNA polymerase I (DNA nucleotidyltransferase). Nicks introduced into depurinated DNA by endonuclease IV or VI provide good primer termini for the polymerase, whereas nicks introduced into depurinated DNA by endonuclease III or into irradiated DNA by T4 UV endonuclease do not. This result suggests that endonuclease IV nicks depurinated DNA on the 5' side of the apurinic site, as does endonuclease VI, whereas endonuclease III has a different incision mechanism. T4 UV endonuclease also possesses apurinic endonuclease activity that generates nicks in depurinated DNA with low priming activity for the polymerase. The priming activity of DNA nicked with endonuclease III or T4 UV endonuclease can be enhanced by an additional incubation with endonuclease VI and, to a lesser extent, by incubation with endonuclease IV. These results indicate that endonuclease III and T4 UV endonuclease (acting upon depurinated and irradiated DNA, respectively) generate nicks containing apurinic/apyrimidinic sites at their 3' termini and that such sites are not rapidly excised by the 3' leads to 5' activity of DNA polymerase I. However, endonuclease IV or VI apparently can remove such terminal apurinic/apyrimidinic sites as well as cleave on the 5' side of the unnicked sites. These results suggest roles for endonucleases III, IV, and VI in the repair of apurinic/apyrimidinic sites as well as pyrimidine dimer sites in DNA. Our results with T4 UV endonuclease suggest that the incision of irradiated DNA by T4 UV endonuclease involves both cleavage of the glycosylic bond at the 5' half of the pyrimidine dimer and cleavage of the phosphodiester bond originally linking the two nucleotides of the dimer. They also imply that the glycosylic bond is cleaved before the phosphodiester bond.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braun A. G., Radman M., Grossman L. Enzymatic repair of DNA: SITES OF HYDROLYSIS BY THE Escherichia coli endonuclease specific for pyrimidine dimers (correndonuclease II). Biochemistry. 1976 Sep 7;15(18):4116–4120. doi: 10.1021/bi00663a031. [DOI] [PubMed] [Google Scholar]
- Braun A., Grossman L. An endonuclease from Escherichia coli that acts preferentially on UV-irradiated DNA and is absent from the uvrA and uvrB mutants. Proc Natl Acad Sci U S A. 1974 May;71(5):1838–1842. doi: 10.1073/pnas.71.5.1838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedberg E. C., King J. J. Dark repair of ultraviolet-irradiated deoxyribonucleic acid by bacteriophage T4: purification and characterization of a dimer-specific phage-induced endonuclease. J Bacteriol. 1971 May;106(2):500–507. doi: 10.1128/jb.106.2.500-507.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gates F. T., Linn S. Endonuclease from Escherichia coli that acts specifically upon duplex DNA damaged by ultraviolet light, osmium tetroxide, acid, or x-rays. J Biol Chem. 1977 May 10;252(9):2802–2807. [PubMed] [Google Scholar]
- Gossard F., Verly W. G. Properties of the main endonuclease specific for apurinic sites of Escherichia coli (endonuclease VI). Mechanism of apurinic site excision from DNA. Eur J Biochem. 1978 Jan 16;82(2):321–332. doi: 10.1111/j.1432-1033.1978.tb12026.x. [DOI] [PubMed] [Google Scholar]
- Grossman L., Braun A., Feldberg R., Mahler I. Enzymatic repair of DNA. Annu Rev Biochem. 1975;44:19–43. doi: 10.1146/annurev.bi.44.070175.000315. [DOI] [PubMed] [Google Scholar]
- Grossman L., Riazuddin S., Haseltine W. A., Lindan C. Nucleotide excision repair of damaged DNA. Cold Spring Harb Symp Quant Biol. 1979;43(Pt 2):947–955. doi: 10.1101/sqb.1979.043.01.104. [DOI] [PubMed] [Google Scholar]
- Hadi S. M., Kirtikar D., Goldthwait D. A. Endonuclease II of Escherichia coli. Degradation of double- and single-stranded deoxyribonucleic acid. Biochemistry. 1973 Jul 3;12(14):2747–2754. doi: 10.1021/bi00738a030. [DOI] [PubMed] [Google Scholar]
- Kato A. C., Bartok K., Fraser M. J., Denhardt D. T. Sensitivity of superhelical DNA to a single-strand specific endonuclease. Biochim Biophys Acta. 1973 Apr 21;308(7):68–78. doi: 10.1016/0005-2787(73)90123-8. [DOI] [PubMed] [Google Scholar]
- Kuhnlein U., Penhoet E. E., Linn S. An altered apurinic DNA endonuclease activity in group A and group D xeroderma pigmentosum fibroblasts. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1169–1173. doi: 10.1073/pnas.73.4.1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LINN S., LEHMAN I. R. AN ENDONUCLEASE FROM NEUROSPORA CRASSA SPECIFIC FOR POLYNUCLEOTIDES LACKING AN ORDERED STRUCTURE. I. PURIFICATION AND PROPERTIES OF THE ENZYME. J Biol Chem. 1965 Mar;240:1287–1293. [PubMed] [Google Scholar]
- LINN S., LEHMAN I. R. AN ENDONUCLEASE FROM NEUROSPORA CRASSA SPECIFIC FOR POLYNUCLEOTIDES LACKING AN ORDERED STRUCTURE. II. STUDIES OF ENZYME SPECIFICITY. J Biol Chem. 1965 Mar;240:1294–1304. [PubMed] [Google Scholar]
- Lindahl T. DNA glycosylases, endonucleases for apurinic/apyrimidinic sites, and base excision-repair. Prog Nucleic Acid Res Mol Biol. 1979;22:135–192. doi: 10.1016/s0079-6603(08)60800-4. [DOI] [PubMed] [Google Scholar]
- Lindahl T. New class of enzymes acting on damaged DNA. Nature. 1976 Jan 1;259(5538):64–66. doi: 10.1038/259064a0. [DOI] [PubMed] [Google Scholar]
- Lindahl T., Nyberg B. Rate of depurination of native deoxyribonucleic acid. Biochemistry. 1972 Sep 12;11(19):3610–3618. doi: 10.1021/bi00769a018. [DOI] [PubMed] [Google Scholar]
- Ljungquist S. A new endonuclease from Escherichia coli acting at apurinic sites in DNA. J Biol Chem. 1977 May 10;252(9):2808–2814. [PubMed] [Google Scholar]
- Minton K., Durphy M., Taylor R., Friedberg E. C. The ultraviolet endonuclease of bacteriophage T4. Further characterization. J Biol Chem. 1975 Apr 25;250(8):2823–2829. [PubMed] [Google Scholar]
- RICHARDSON C. C., KORNBERG A. A DEOXYRIBONUCLEIC ACID PHOSPHATASE-EXONUCLEASE FROM ESCHERICHIA COLI. I. PURIFICATION OF THE ENZYME AND CHARACTERIZATION OF THE PHOSPHATASE ACTIVITY. J Biol Chem. 1964 Jan;239:242–250. [PubMed] [Google Scholar]
- Radman M. An endonuclease from Escherichia coli that introduces single polynucleotide chain scissions in ultraviolet-irradiated DNA. J Biol Chem. 1976 Mar 10;251(5):1438–1445. [PubMed] [Google Scholar]
- Riazuddin S., Grossman L. Micrococcus luteus correndonucleases. I. resolution and purification of two endonucleases specific for DNA containing pyrimidine dimers. J Biol Chem. 1977 Sep 25;252(18):6280–6286. [PubMed] [Google Scholar]
- Riazuddin S., Grossman L. Micrococcus luteus correndonucleases. II. Mechanism of action of two endonucleases specific for DNA containing pyrimidine dimers. J Biol Chem. 1977 Sep 25;252(18):6287–6293. [PubMed] [Google Scholar]
- Seeberg E. Reconstitution of an Escherichia coli repair endonuclease activity from the separated uvrA+ and uvrB+/uvrC+ gene products. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2569–2573. doi: 10.1073/pnas.75.6.2569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeberg E. Reconstitution of an Escherichia coli repair endonuclease activity from the separated uvrA+ and uvrB+/uvrC+ gene products. Proc Natl Acad Sci U S A. 1978 Jun;75(6):2569–2573. doi: 10.1073/pnas.75.6.2569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomilin N. V., Paveltchuk E. B., Mosevitskaya T. V. Substrate specificity of the ultraviolet-endonuclease from Micrococcus luteus. Endonucleolytic cleavage of depurinated DNA. Eur J Biochem. 1976 Oct 1;69(1):265–272. doi: 10.1111/j.1432-1033.1976.tb10882.x. [DOI] [PubMed] [Google Scholar]
- Yajko D. M., Weiss B. Mutations simultaneously affecting endonuclease II and exonuclease III in Escherichia coli. Proc Natl Acad Sci U S A. 1975 Feb;72(2):688–692. doi: 10.1073/pnas.72.2.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasuda S., Sekiguchi M. T4 endonuclease involved in repair of DNA. Proc Natl Acad Sci U S A. 1970 Dec;67(4):1839–1845. doi: 10.1073/pnas.67.4.1839. [DOI] [PMC free article] [PubMed] [Google Scholar]



