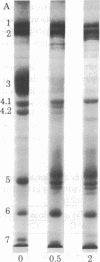
Abstract
Direct physical evidence for the linkage of a band 3 population to the cytoskeleton in the erythrocyte ghost membrane is presented. The rotational diffusion of band 3 proteins was mesured by observing flash-induced transient dichroism of a covalently bound eosin probe. After proteolytic release of a 40,000-dalton cytoplasmic segment of band 3 by trypsin, a considerable enhancement in the decay of the absorption anisotropy was observed. Analysis of the data indicates that proteolytic cleavage of band 3 produces a mobile band 3 population which has restricted mobility in the unperturbed membrane due to protein-protein interactions involving the cytoplasmic band 3 moiety. Band 2.1 (ankyrin) or 4.1 or both are likely to be involved in this interaction because a similar effect on band 3 mobility is observed after low-salt/high-salt extraction of these components. Quantitatively, it is estimated that up to 40% of band 3 may be linked to the cytoskeleton. Because the ankyrin-band 3 dimer stoichiometry in the membrane is approximately 1:5, only about 20% of band 3 dimers can be directly linked to ankyrin. The remainder could be explained by the existence of higher oligomers of band 3 linked to single ankyrin polypeptides or by linkages involving other components such as band 4.1 or 4.2.
Full text
PDF
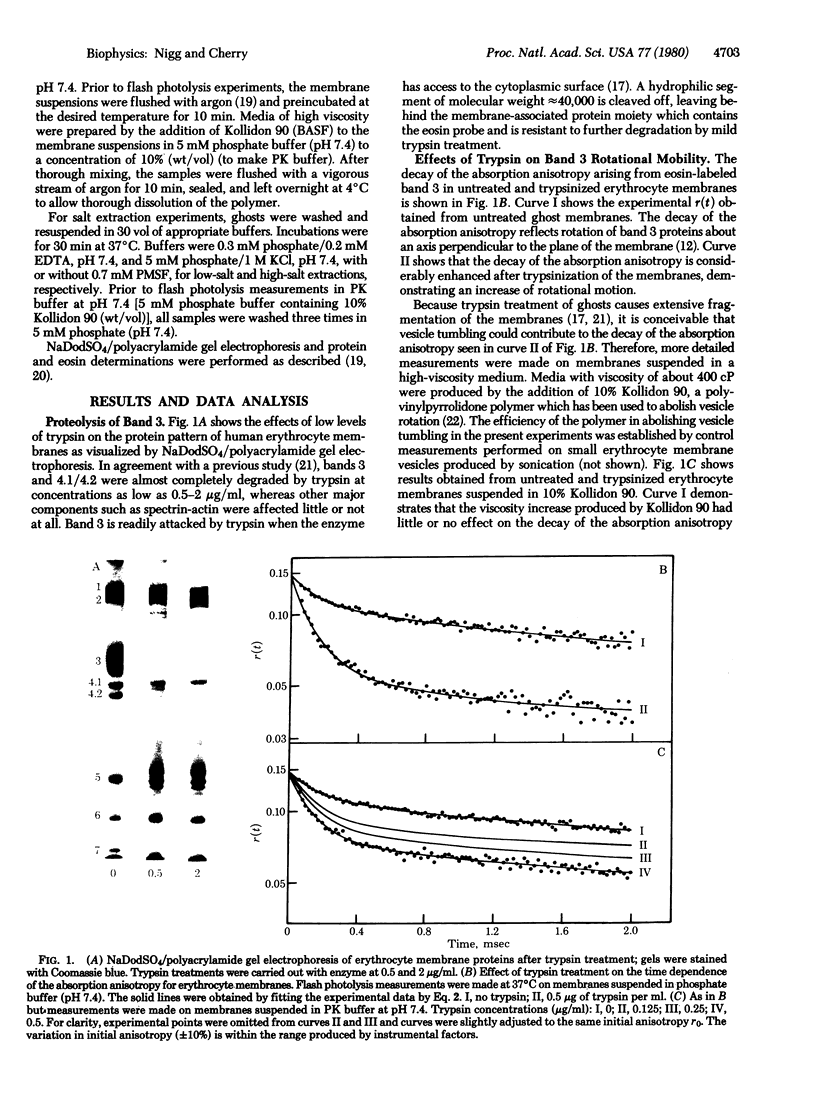
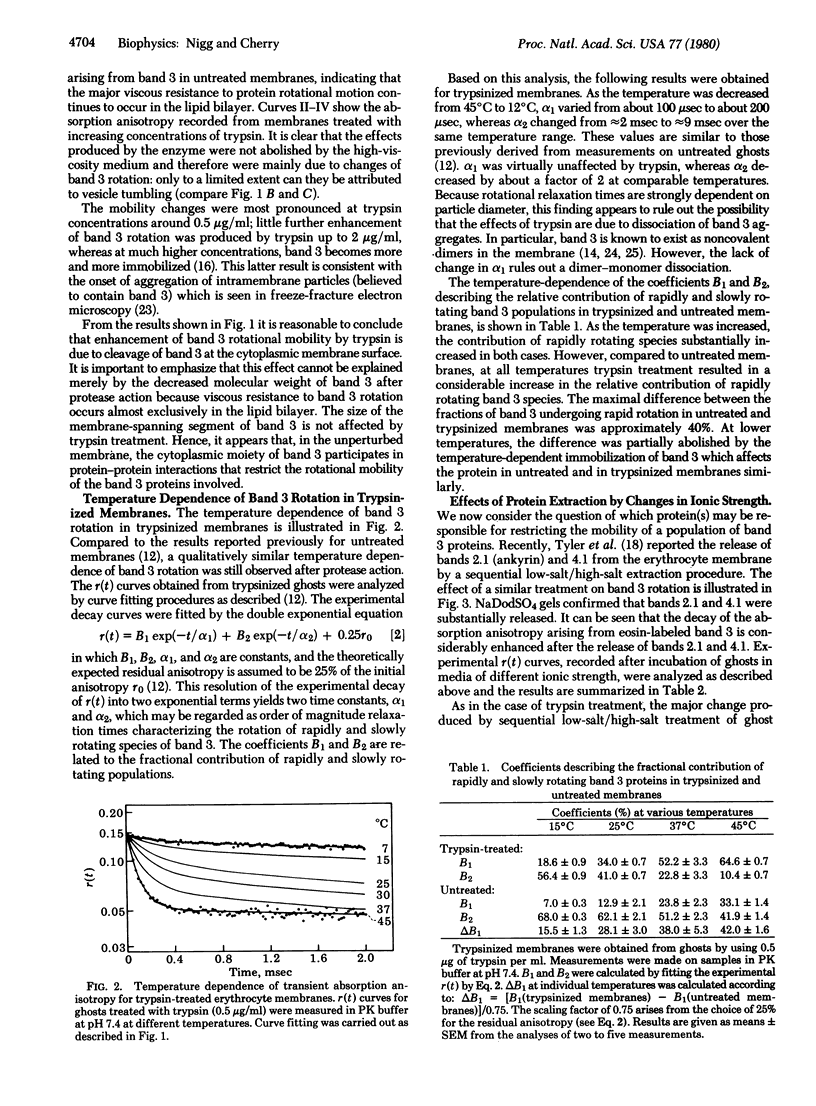
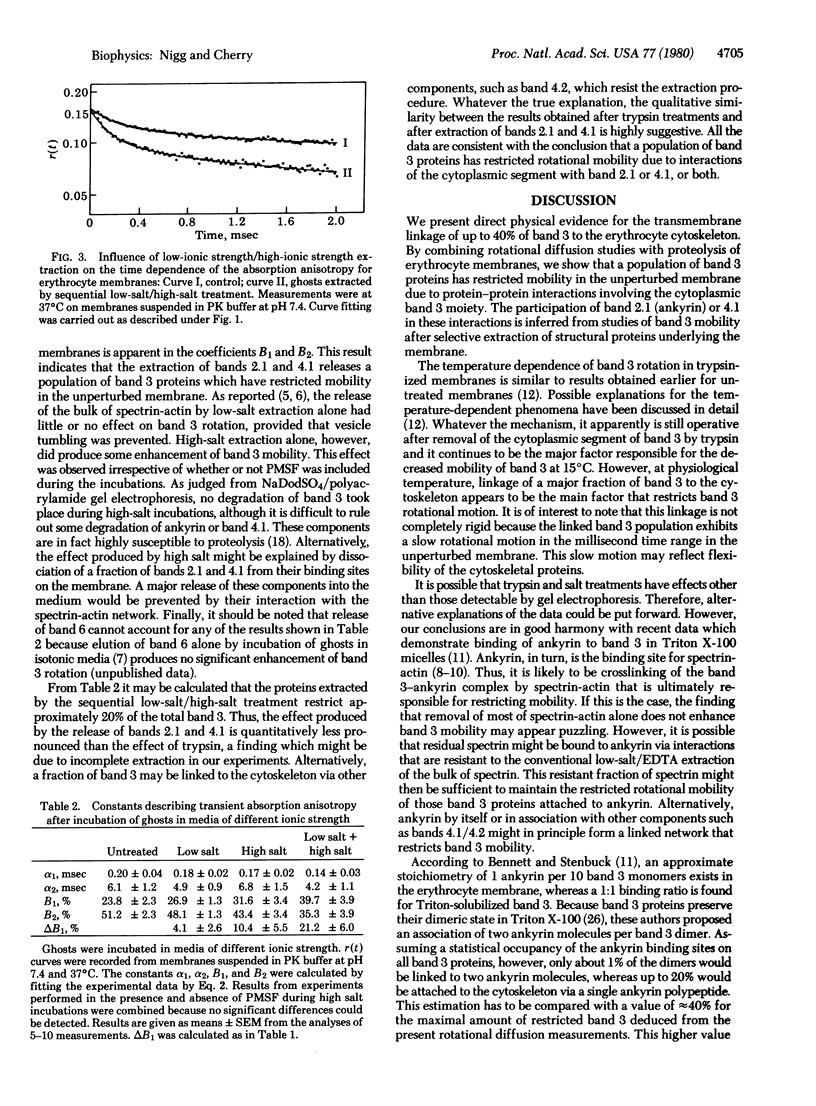

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Austin R. H., Chan S. S., Jovin T. M. Rotational diffusion of cell surface components by time-resolved phosphorescence anisotropy. Proc Natl Acad Sci U S A. 1979 Nov;76(11):5650–5654. doi: 10.1073/pnas.76.11.5650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avruch J., Price H. D., Martin D. B., Carter J. R. Effect of low levels of trypsin on erythrocyte membranes. Biochim Biophys Acta. 1973 Jan 26;291(2):494–505. doi: 10.1016/0005-2736(73)90501-4. [DOI] [PubMed] [Google Scholar]
- Bennett V., Stenbuck P. J. Identification and partial purification of ankyrin, the high affinity membrane attachment site for human erythrocyte spectrin. J Biol Chem. 1979 Apr 10;254(7):2533–2541. [PubMed] [Google Scholar]
- Bennett V., Stenbuck P. J. The membrane attachment protein for spectrin is associated with band 3 in human erythrocyte membranes. Nature. 1979 Aug 9;280(5722):468–473. doi: 10.1038/280468a0. [DOI] [PubMed] [Google Scholar]
- Cherry R. J., Bürkli A., Busslinger M., Schneider G., Parish G. R. Rotational diffusion of band 3 proteins in the human erythrocyte membrane. Nature. 1976 Sep 30;263(5576):389–393. doi: 10.1038/263389a0. [DOI] [PubMed] [Google Scholar]
- Cherry R. J. Measurement of protein rotational diffusion in membranes by flash photolysis. Methods Enzymol. 1978;54:47–61. doi: 10.1016/s0076-6879(78)54007-x. [DOI] [PubMed] [Google Scholar]
- Cherry R. J. Rotational and lateral diffusion of membrane proteins. Biochim Biophys Acta. 1979 Dec 20;559(4):289–327. doi: 10.1016/0304-4157(79)90009-1. [DOI] [PubMed] [Google Scholar]
- Elgsaeter A., Shotton D. M., Branton D. Intramembrane particle aggregation in erythrocyte ghosts. II. The influence of spectrin aggregation. Biochim Biophys Acta. 1976 Feb 19;426(1):101–122. doi: 10.1016/0005-2736(76)90433-8. [DOI] [PubMed] [Google Scholar]
- Greinert R., Staerk H., Stier A., Weller A. E-type delayed fluorescence depolarization, technique to probe rotational motion in the microsecond range. J Biochem Biophys Methods. 1979 May;1(2):77–83. doi: 10.1016/0165-022x(79)90014-9. [DOI] [PubMed] [Google Scholar]
- Heyn M. P., Cherry R. J., Müller U. Transient and linear dichroism studies on bacteriorhodopsin: determination of the orientation of the 568 nm all-trans retinal chromophore. J Mol Biol. 1977 Dec 15;117(3):607–620. doi: 10.1016/0022-2836(77)90060-2. [DOI] [PubMed] [Google Scholar]
- Kiehm D. J., Ji T. H. Photochemical cross-linking of cell membranes. A test for natural and random collisional cross-links by millisecond cross-linking. J Biol Chem. 1977 Dec 10;252(23):8524–8531. [PubMed] [Google Scholar]
- Luna E. J., Kidd G. H., Branton D. Identification by peptide analysis of the spectrin-binding protein in human erythrocytes. J Biol Chem. 1979 Apr 10;254(7):2526–2532. [PubMed] [Google Scholar]
- Lux S. E. Dissecting the red cell membrane skeleton. Nature. 1979 Oct 11;281(5731):426–429. doi: 10.1038/281426a0. [DOI] [PubMed] [Google Scholar]
- Marchesi V. T., Tillack T. W., Jackson R. L., Segrest J. P., Scott R. E. Chemical characterization and surface orientation of the major glycoprotein of the human erythrocyte membrane. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1445–1449. doi: 10.1073/pnas.69.6.1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nigg E. A., Bron C., Girardet M., Cherry R. J. Band 3-glycophorin A association in erythrocyte membrane demonstrated by combining protein diffusion measurements with antibody-induced cross-linking. Biochemistry. 1980 Apr 29;19(9):1887–1893. doi: 10.1021/bi00550a024. [DOI] [PubMed] [Google Scholar]
- Nigg E., Cherry R. J. Dimeric association of band 3 in the erythrocyte membrane demonstrated by protein diffusion measurements. Nature. 1979 Feb 8;277(5696):493–494. doi: 10.1038/277493a0. [DOI] [PubMed] [Google Scholar]
- Nigg E., Kessler M., Cherry R. J. Labeling of human erythrocyte membranes with eosin probes used for protein diffusion measurements: inhibition of anion transport and photo-oxidative inactivation of acetylcholinesterase. Biochim Biophys Acta. 1979 Jan 19;550(2):328–340. doi: 10.1016/0005-2736(79)90219-0. [DOI] [PubMed] [Google Scholar]
- Silva P. P., Nicolson G. L. Freeze-etch localization of concanavalin A receptors to the membrane intercalated particles of human erythrocyte ghost membranes. Biochim Biophys Acta. 1974 Sep 23;363(3):311–319. doi: 10.1016/0005-2736(74)90071-6. [DOI] [PubMed] [Google Scholar]
- Steck T. L. Cross-linking the major proteins of the isolated erythrocyte membrane. J Mol Biol. 1972 May 14;66(2):295–305. doi: 10.1016/0022-2836(72)90481-0. [DOI] [PubMed] [Google Scholar]
- Steck T. L., Ramos B., Strapazon E. Proteolytic dissection of band 3, the predominant transmembrane polypeptide of the human erythrocyte membrane. Biochemistry. 1976 Mar 9;15(5):1153–1161. doi: 10.1021/bi00650a030. [DOI] [PubMed] [Google Scholar]
- Steck T. L. The organization of proteins in the human red blood cell membrane. A review. J Cell Biol. 1974 Jul;62(1):1–19. doi: 10.1083/jcb.62.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stefanovic V., Mandel P., Rosenberg A. Effect of concanavalin A on the kinetics of ecto-5'-adenosine monophosphatase (5'-adenosine monophosphate phosphohydrolase) in the outer surface of intact neural cells in culture. Biochemistry. 1979 Jan 23;18(2):357–361. doi: 10.1021/bi00569a020. [DOI] [PubMed] [Google Scholar]
- Tyler J. M., Hargreaves W. R., Branton D. Purification of two spectrin-binding proteins: biochemical and electron microscopic evidence for site-specific reassociation between spectrin and bands 2.1 and 4.1. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5192–5196. doi: 10.1073/pnas.76.10.5192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J., Branton D. Reconstitution of intramembrane particles in recombinants of erythrocyte protein band 3 and lipid: effects of spectrin-actin association. Proc Natl Acad Sci U S A. 1976 Nov;73(11):3891–3895. doi: 10.1073/pnas.73.11.3891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J., Goodman S. R. Syndeins: the spectrin-binding protein(s) of the human erythrocyte membrane. Proc Natl Acad Sci U S A. 1979 May;76(5):2340–2344. doi: 10.1073/pnas.76.5.2340. [DOI] [PMC free article] [PubMed] [Google Scholar]