Abstract
Although WNTs have been long thought of as regulators of cell fate, recent studies highlight their involvement in crucial aspects of synaptic development in the nervous system. Particularly compelling are recent studies of the neuromuscular junction in nematodes, insects, fish and mammals. These studies place WNTs as major determinants of synapse differentiation and neurotransmitter receptor clustering.
Morphogens of the WNT family have crucial functions during the development of all metazoan organisms by providing positional information to cells in the embryo or during metamorphosis in insects1. However, mis-regulation of the WNT pathway in humans is linked to conditions such as Alzheimer’s disease, Huntington’s disease, schizophrenia and bipolar disorder2–6, which suggests that members of this family have a role in postmitotic neurons. Indeed, studies of the nervous system have uncovered roles for WNTs in axon pathfinding, dendritic development, synaptogenesis, synapse maturation and plasticity7,8. Particularly intriguing are a series of recent studies that implicate WNTs in the development of neuromuscular junctions (NMJs) across species ranging from worms to mammals7,9. These studies are unravelling a role for WNTs in neurotransmitter receptor clustering and the organization of presynaptic and postsynaptic specializations. Here we review recent progress in understanding the mechanisms by which WNTs regulate NMJ development and function.
The WNT family is composed of multiple family members, including 5 in worms, 7 in flies, 15 in zebrafish, and 19 in mice and humans. Adding to this diversity is the presence of a myriad of typical WNT receptors, known as Frizzled receptors, which include 3 in worms, 5 in flies, 12 in fish and 11 in mammals, as well as non-conventional receptors such as Derailed (DRL) — a member of the RYK (related to receptor tyrosine kinase) subfamily of receptor tyrosine kinases — and ROR2 (receptor tyrosine kinase-like orphan receptor 2). This complexity of WNT signalling is further heightened by the activation of at least five different WNT transduction pathways, which trigger various cellular processes7,10 (BOX 1).
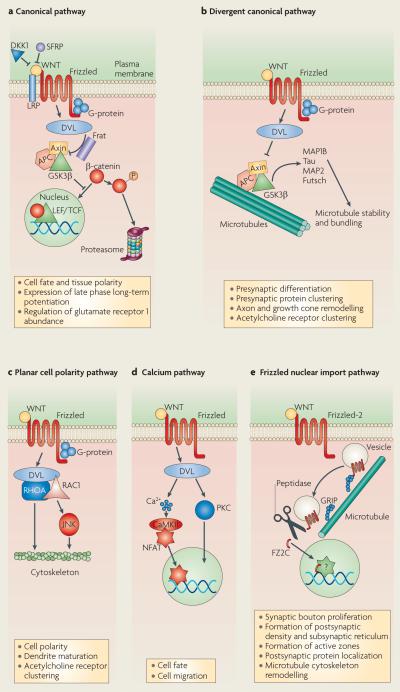
Box 1. WNT signalling pathways.
Canonical pathway
This is the best-characterized WNT signalling pathway (see part a of the figure), in which WNT binding to Frizzled receptors activates the scaffolding protein Dishevelled (DVL), which disassembles a so-called ‘destruction complex’ formed by glycogen synthase kinase 3β (GSK3β), Axin and adenomatous polyposis coli (APC) — a complex that normally leads to the degradation of β-catenin. WNT binding to Frizzled disrupts the destruction complex, and this results in cytoplasmic stabilization of β-catenin and its import into the nucleus, where it regulates gene expression through association with lymphoid enhancer factor/T cell factor (LEF/TCF) transcription factors. In this pathway, Frizzled collaborates with a co-receptor, LRP5/6 of the low-density lipoprotein receptor related protein (LRP) family. This pathway is antagonized by the secreted protein Dickkopf1 (DKK1) and secreted Frizzled related proteins (SFRPs). Also depicted is an inhibitor of GSK3b, Frat.
Divergent canonical pathway
DVL binds to microtubules and regulates GSK3β-dependent phosphorylation of microtubule-associated proteins (MAPs), such as MAP1B, Tau, MAP275,76, and the related Drosophila melanogaster protein Futsch59. Inhibition of GSK3β upon activation of the WNT divergent canonical pathway (see part b of the figure), thus enhances microtubule stability.
Planar cell polarity pathway
In this pathway (see part c of the figure), DVL activation turns on the small GTPases RHOA or RAC1 and the JUN N-terminal kinase (JNK) to regulate actin and microtubule cytoskeletons.
WNT calcium pathway
This is a fourth signalling pathway (see part d of the figure) in which DVL activation induces an elevation in the levels of intracellular Ca2+ and activation of protein kinase C (PKC) and calcium/ calmodulin-dependent protein kinase II (CaMKII). This results in the nuclear import of the transcription factor nuclear-factor of activated T cells (NFAT), which regulates gene expression.
Frizzled nuclear import pathway
An alternative transduction pathway (see part e of the figure) in which WNT receptors themselves are internalized, cleaved and imported into the nucleus7,77. Trafficking of the Frizzled-2 receptor towards the nucleus depends on its binding partner GRIP (7-PDZ-domain glutamate-receptor binding protein). This mechanism has been substantiated at the Drosophila melanogaster neuromuscular junction60, as well as during the development of cortical neurons in mammals78.
The first hint that WNTs function in synapse development emerged from studies of the developing cerebellum, which suggested that WNT7a operated in a retrograde manner to enhance presynaptic differentiation8,11. A similar retrograde role for WNT3, involving the divergent canonical pathway was also demonstrated in sensory neurons and motor neurons12. WNT7b has also been involved in dendrite development in the hippocampus, probably through the activation of canonical and non-canonical WNT pathways8,13. Recent studies have extended WNT function to the development of the NMJ in both vertebrate and invertebrate organisms.
WNT signalling at the vertebrate NMJ
Organization of postsynaptic receptor clusters
The vertebrate NMJ is composed of the motor axon terminal, which sits in a shallow trough at the muscle surface and is capped by terminal Schwann cells that cover the entire NMJ. The motor axon releases acetylcholine (ACh), the primary excitatory transmitter at these junctions. In the mature NMJ, the postsynaptic membrane forms junctional folds, which organize the postsynaptic apparatus. ACh receptors (AChRs) aggregate at the top of these folds in direct apposition to presynaptic active zones, which are the sites of neurotransmitter release14. The organization of AChR at the junctional folds involves positive and negative signals that lead to the clustering of AChRs at the endplate, and the dispersal of aneural AChR clusters, as well as an increased expression of AChRs by synaptic myonuclei and the suppression of AChR expression by extra-synaptic myonuclei15. AChR clustering requires the transmembrane MUSK (muscle, skeletal, receptor tyrosine kinase), and the secreted heparan-sulphate proteoglycan Agrin, which activates tyrosine phosphorylation of MUSK. MUSK controls AChR clustering through Rapsyn, a protein that binds, clusters and anchors AChRs16. ACh also functions to disperse AChR clusters, a role counteracted by the release of nerve-derived Agrin14. This mechanism seems to ensure proper apposition between the presynaptic and postsynaptic apparatus, as well as mono-synaptic innervation of each muscle. Indeed, in mutant mice that lack the ACh synthetic enzyme choline acetyltransferase, NMJs had abnormal branching patterns and excessively broad endplate bands, and muscles were often innervated by more than one NMJ17.
The small GTPases RAC and RHO are also sequentially activated by Agrin, and this induces the formation of AChR micro-clusters and their coalescence into full-sized clusters18,19. Initial hypotheses posed that nerve-derived Agrin, through MUSK, triggered AChR clustering. However, it was subsequently found that before innervation, AChR clusters were already present at presumptive endplates in a MUSK-dependent manner20,21. Therefore the formation of these aneural or pre-patterned AChR clusters is independent of Agrin22,23. However the maintenance and further growth of the clusters require Agrin, which suggests that Agrin serves a stabilizing function rather than an AChR cluster-inducing function14,24.
Role of WNTs in AChR clustering
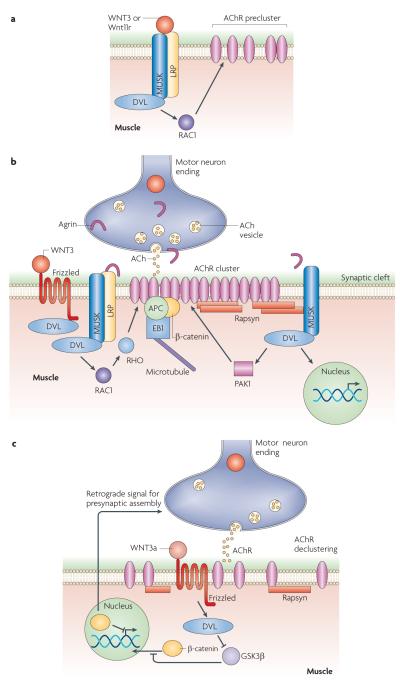
A link between AChR clustering and WNTs emerged from observations that DVL (mammalian homologue of Drosophila Dishevelled) interacted directly with MUSK, coupling it to the actin regulator PAK1 (p21 protein (Cdc42/Rac)-activated kinase 1) — an interaction that is crucial for AChR clustering by Agrin in cultured mouse muscle cells25. Further, it was found that WNT1 regulates MUSK expression26 and that neural Agrin activates PAK1 in a DVL-dependent manner25. Although there is some disparity in the published literature regarding specific roles of WNTs in the regulation of AChR clustering, WNT3a has been reported to inhibit Agrin-induced AChR clustering in mice27. Conversely, mouse WNT3 and zebrafish Wnt11r are positive regulators of AChR clustering28,29. It is important to note that although several studies suggest that WNTs are secreted by the presynaptic or the postsynaptic cell, WNTs that influence synapse development might also be released by other cell types (for example, by glial cells30 or, in the case of Wnt11r, probably by somites29). Furthermore, autocrine regulation of WNT signalling by WNT-expressing cells has also been reported31 and suggested at the Drosophila melanogaster NMJ7. Support for a positive role for WNT3 during AChR clustering emerged from the finding that exposing embryonic chick wings or cultured mouse myotubes to WNT3, resulted in an increase in the number and size of Agrin-dependent AChR clusters28. Moreover, in Dvl1 mutant mice AChR clusters had a more disperse distribution at the endplate28. Whereas SFRP1 (secreted Frizzled-related protein 1) blocked the effects of WNT3, DKK1 (dickkopf homolog 1) did not, suggesting the involvement of non-canonical signalling (see BOX1). WNT3 induced a rapid activation of RAC1 and the accumulation of transient AChR microclusters (FIG. 1a), which were transformed into full-sized clusters when Agrin was present28 (FIG. 1b). Thus, WNT3-dependent microclusters might be stabilized by Agrin and serve as nucleating centres for the formation of full-size AChR clusters28. Interestingly, recent studies of zebrafish show that Dvl is required for aneural AChR cluster formation (FIG. 1a), and that in the absence of aneural AChR clusters motor axon pathfinding is disrupted29. Analysis of zebrafish injected with Wnt11r morpholinos showed that Wnt11r was required for AChR pre-patterning and for normal navigation of presynaptic terminals, involving a pathway similar to the planar cell polarity pathway.
Figure 1. Role of WNTs in AChR clustering at the vertebrate neuromuscular junction.
a ∣ WNT3 (in mice) and Wnt11r (in zebrafish) induce AChr (acetylcholine receptor) preclustering before innervation. b ∣ WNT3 has been implicated in AChr clustering after innervation. c ∣ ACh release and WNT3a mediate AChr declustering. A retrograde signal induced by muscle β-catenin, to regulate the development of the presynaptic motor endings, is also shown. APC, adenomatous polyposis coli; DVL, mammalian homologue of Drosophila Dishevelled; GSK3β, glycogen synthase kinase 3β; LRP, low density lipoprotein receptor-related protein; MUsK, muscle, skeletal, receptor tyrosine kinase; PAK1, protein (Cdc42/rac)-activated kinase 1.
Inhibitory roles of WNTs at the NMJ were supported by the finding that WNT3a inhibited Agrin-dependent AChR cluster formation and induced the dispersal of already formed clusters in cultured myotubes and in vivo, through Rapsyn gene repression27 (FIG. 1c). Furthermore, Rapsyn overexpression prevented WNT3a-dependent cluster dispersal in cultured myoblasts27. This inhibitory effect of WNT3a is consistent with studies showing that inhibition of GSK3β (glycogen synthase kinase 3β) in muscle reduces AChR clustering28. Moreover, expressing β-catenin (also known as cadherin-associated protein-β) in limb muscles of mice in vivo inhibited Agrin-dependent AChR cluster formation27, and, conversely, mutant mice lacking β-catenin in muscle showed an increase in the size of AChR clusters32,33. However, there is some disparity on the evidence implicating β-catenin in AChR cluster formation, as it has also been reported that downregulating β-catenin in cultured myotubes inhibits Agrin-dependent AChR clusters34. Rapsyn gene expression was shown to be reduced upon β-catenin upregulation27. The inhibitory function of WNT3a is likely to be mediated through canonical WNT signalling, as β-catenin was involved, and as DKK1 opposed the effects of WNT3a27. However, T cell factor (TCF)-mediated transcription does not seem to be required, as mutating TCF motifs in the Rapsyn promoter region had no effect on Rapsyn levels, and expressing a TCF dominant-negative in myotubes did not alter Agrin-dependent AChR cluster formation27. Notably, there are also NF-κB (nuclear factor-κB) binding sites and an e-box in the Rapsyn promoter35,36, implying the possibility of Rapsyn gene regulation by β-catenin through these sites. Thus, WNTs can serve both synaptogenic and anti-synaptogenic functions. This antagonistic role for WNTs might serve to refine synaptic architecture, and might also have a role during synapse elimination37.
Intriguingly, Zhang et al.34 found that β-catenin interacted directly with Rapsyn and surface AChRs, and that Agrin enhanced the association between β-catenin and surface AChRs34 (FIG. 1b). α-Catenin was also present in the complex, probably through association with β-catenin, which suggests that β-catenin could serve as a link between AChRs and the α-catenin-associated cytoskeleton.
A retrograde signalling pathway down-stream of β-catenin at the NMJ was suggested by the finding that mutants lacking β-catenin in muscle had abnormal presynaptic differentiation32 (FIG. 1c). These mutants also had a reduction in evoked release, defects in short-term plasticity, as well as calcium sensitivity32. Interestingly, SFRPs are present in muscles, localize to the NMJ and are upregulated upon denervation38, which raises the possibility that WNT signalling might mediate these β-catenin-dependent processes32.
Important additional evidence for the interaction between WNT signalling and AChR clustering was provided by the finding that LRP4 (low density lipoprotein receptor-related protein 4) interacts with MUSK and binds Agrin (FIG. 1a,b). Although a wealth of evidence had indicated that MUSK was an Agrin receptor, no evidence for a direct interaction had been forth-coming, which suggests the presence of a co-receptor39. Initial clues as to the identity of the Agrin co-receptor were provided by the finding that Lrp4 mutant mice lacked AChR clusters, had aberrant presynaptic branching and a reduction in presynaptic sites40 — a phenotype remarkably similar to that of Musk mutants41. Two recent studies42,43 have supported the view that LRP4 is the long-sought Agrin co-receptor. The extracellular domain of LRP4 binds to neural Agrin and forms a complex with MUSK, which was shown to be required for MUSK activation by Agrin and for AChR clustering in myotubes42. LRP4 co-localizes with MUSK at the NMJ and in cultured muscle cells40,42,43. It was found that LRP4 interacted directly with both Agrin and MUSK, and that the interaction between LRP4 and MUSK was enhanced by Agrin42,43. Although LRP4 alone could bind Agrin, the MUSK–LRP4 complex had higher binding affinity at high Agrin concentrations, such as those predicted in the synaptic cleft42,43.Whereas Zhang et al.43 showed tyrosine phosphorylation of the intracellular domain of LRP4 in Agrin stimulated muscle cells, Kim et al.42 could not detect tyrosine phosphorylation of LRP4. Notably, Lrp4 mutant43 mice, similar to MUSK mutants but unlike Agrin mutants, also lacked aneural AChR clusters, suggesting an Agrin-independent, and perhaps WNT-dependent pathway for aneural AChR cluster formation22 (FIG. 1a). This idea has been supported by recent studies in zebrafish, demonstrating that Wnt11r and Dvl are required for AChR pre-patterning29 (see ReF. 44 for a summary of the role of WNTs in AChR pre-patterning at the NMJ).
The earlier studies of WNT signalling at the NMJ also demonstrated that APC (adenomatous polyposis coli) is localized at the NMJ and binds directly to the β-subunit of AChRs45 (FIG. 1b). APC, beyond its role in antagonizing the canonical WNT pathway, organizes and stabilizes the microtubule cytoskeleton in epithelial cells by binding the microtubule plus-end binding protein EB1 (REFS 46,47). At the NMJ APC is required for Agrin-dependent nicotinic AChR clustering45. APC also bound to postsynaptic density 93 (PSD93), β-catenin and EB1 to regulate neuronal AChR surface levels and clusters48,49 (FIG. 1b). Thus, at the vertebrate NMJ, WNTs are intimately involved in the signalling mechanisms that specify the localization of innervation and the cellular machinery that induces AChR clustering.
WNT signalling at the invertebrate NMJ
Development of Drosophila melanogaster larval NMJs
The body wall muscles of the D. melanogaster larva are stereotypically innervated by glutamatergic as well as by peptidergic and octopamine-containing motor neurons. Presynaptic endings are organized into synaptic boutons, containing synaptic vesicles and the release apparatus. At the postsynaptic membrane, glutamate receptor (GluR) clusters are exactly apposed to active zones, the sites of neurotransmitter release. GluR clustering (as well as their enhanced synthesis) is induced by innervation50 and this is modulated by non-vesicular51,52, but not by vesicular53, glutamate release. A molecular mechanism that directs GluR clustering in a similar manner to Agrin- and WNT-dependent clustering of AChRs at the vertebrate NMJ has not been identified at the D. melanogaster NMJ (but see next section for a description of the role of WNTs in D. melanogaster NMJ development)54,55.
A hallmark of this system is the continuous formation of new synaptic boutons to compensate for the striking increase in muscle size during larval development and to maintain excitation–contraction efficacy. The bone morphogenic protein (BMP) pathway, which initiates a retrograde signal by which muscles influence presynaptic growth, has emerged to be key for presynaptic and postsynaptic communication56. In addition, WNTs function both in an anterograde and probably an autocrine fashion in motor neurons to regulate the presynaptic and postsynaptic apparatus. Although initial establishment of connectivity at the larval NMJ is mainly independent of electrical activity, the expansion of the NMJ during muscle growth is strongly influenced by synaptic activity, which has made this preparation an excellent model system to study synaptic plasticity.
Regulation of Drosophila melanogaster larval NMJ development by WNTs
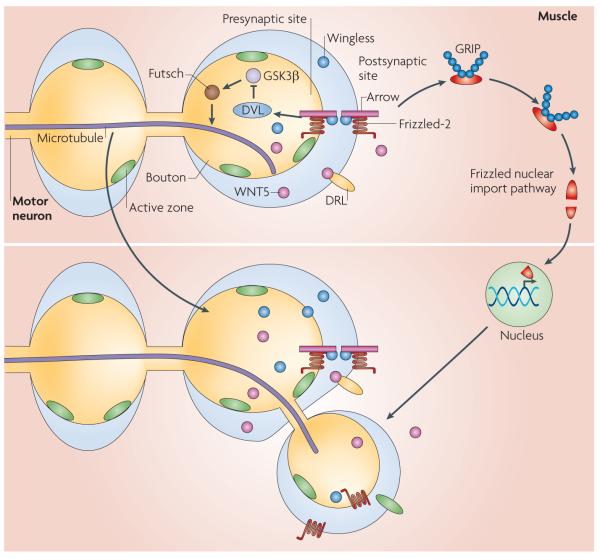
The role of WNTs in invertebrate synapses was recognized by the finding that Wingless (also known as WNT1) and its receptor Frizzled-2 were present at the D. melanogaster larval NMJ57. Through the use of a temperature sensitive wingless mutant (wgts), which allowed for a temporal block of Wingless secretion, thus bypassing early roles of Wingless in embryogenesis, as well as various molecular manipulations, it was shown that Wingless was released by presynaptic boutons, but probably not by muscles. blocking Wingless release during larval growth, led to a decrease in synaptic bouton number and to changes in bouton morphology, that were rescued by restoring Wingless in the motor neurons57. by contrast, increasing the levels of Wingless in motor neurons led to synaptic bouton overgrowth. In wgts mutants, presynaptic boutons had abnormal postsynaptic Discs large (DLG), a PSD95 family member, and GluR localization. Most strikingly, a subset of boutons (‘ghost boutons’) were filled with synaptic vesicles, but were devoid of active zones, postsynaptic specializations and mitochondria, which suggests that Wingless has central roles during synapse differentiation57,58. The mutants also had disruptions in the presynaptic microtubule cytoskeleton, as demonstrated by examining the microtubule-associated protein 1b (MAP1b)-related protein Futsch, which has been shown to be phosphorylated by GSK3β59. Interfering with Frizzled-2 function in the muscle alone resulted in similar synaptic growth and morphology defects57, suggesting that Wingless activates both anterograde and retrograde signalling (FIG. 2).
Figure 2. Role of WNTs during Drosophila melanogaster larval neuromuscular junction development.
Wingless secreted from presynaptic motor neuron endings, binds to Frizzled-2 and co-receptor Arrow, which are localized presynaptically and postsynaptically. In the presynaptic cell, Wingless activates a divergent canonical pathway, involving DVL (Dishevelled) activation, inhibition of GSK3β (glycogen synthase kinase 3β) activity and the regulation of the microtubule cytoskeleton through Futsch. In the postsynaptic cell, Wingless activates the Frizzled nuclear import pathway, which involves the cleavage and nuclear import of Frizzled-2. grIP (7-PDZ-domain glutamate-receptor binding protein) is required for the trafficking of receptors from the postsynaptic membrane towards the nucleus. WNT5 is also released from the presynaptic boutons and binds to its receptor Derailed (DrL) on the postsynaptic membrane to regulate synaptic bouton growth.
The search for the transduction cascade activated by Wingless at the D. melanogaster NMJ led to the finding of a previously unrecognized alternative WNT pathway in larval muscles, the Frizzled nuclear import pathway60 (BOX 1; FIG. 2), in which a fragment of the Frizzled-2 receptor itself is cleaved and imported into the nucleus. The importance of Frizzled-2 cleavage was demonstrated by Frizzled-2 mutant rescue experiments, which showed that although expressing a full-length Frizzled-2 transgene in muscles rescued the defects in bouton number, expressing a transgene lacking the cleavage site did not. Notably, expressing the Frizzled-2C fragment did not bypass the requirement for Wingless signalling, raising the possibility that Frizzled-2C is modified in a Wingless-dependent fashion before nuclear import60. The Frizzled nuclear import pathway was also shown to depend on the D. melanogaster homologue of GRIP (7-PDZ-domain glutamate-receptor binding protein), which interacts directly with the carboxy-terminal PDZ binding sequence of Frizzled-2, and which is required to traffic the receptor from the synapse to the nucleus58 (FIG. 2). Although in mammals GRIP also seems to be crucial for post-synaptic development of neurons in culture61, an association between GRIP and WNT pathways has not been as yet established in mammals. A similar mechanism involving cleavage and import has been implicated in establishing communication between the cell surface and the nucleus by several other receptors, including Notch, EGFR (epidermal growth-factor receptor) and the voltage-gated calcium channel (Cav1.2)62–64.
Recently synaptic Wingless signalling was also shown to underlie activity-dependent remodelling of the NMJ65. Wingless secretion was enhanced by activity and this was correlated to rapid activity-dependent NMJ growth. Spaced stimulation, by potassium-induced depolarization, motor nerve stimulation or light activation of neuronally expressed channelrhodopsin-2 (ChR2) induced the formation of dynamic filopodia- like extensions (synaptopods) and ghost boutons, as well as a potentiation of spontaneous neurotransmitter release 2 hours after the stimulation began. This was blocked by low extracellular calcium and by genetic manipulations that blocked action potentials or neurotransmitter release. Live imaging of ghost boutons from live non-dissected preparations demonstrated that they could acquire GluRs and active zones, and thus represent synaptic bouton intermediates. Although ghost boutons were also observed in non-stimulated larvae, albeit at very low frequency, the activity-induced formation of ghost boutons required four to five cycles of spaced simulation and was blocked by transcriptional and translational inhibitors. This is akin to long-term behavioural and physiological plasticity, which also requires spaced training and/or stimulation and new protein synthesis66.
Given that disrupting the Frizzled nuclear import pathway leads to poor bouton proliferation and the formation of ghost boutons, the authors speculated that this transduction pathway might be involved in the acute activity-dependent synaptic growth. Indeed, heterozygous wingless mutants suppressed the activity-dependent synaptic growth, which was rescued by restoring Wingless in motor neurons. Importantly, over-expressing Wingless in motor neurons bypassed some of the requirements for spaced stimulation in the formation of ghost boutons — whereas wild-type larvae required four to five cycles of spaced stimulations, Wingless over-expressing larvae required only three. As expected, activity also regulated the Frizzled nuclear import pathway in the muscle cell. Spaced stimulation or chronic increase in activity through the use of mutations in potassium-channel subunits, eag Sh increased Frizzled-2C in the nucleus. This increase could be prevented by decreasing wingless gene dosage in the eag Sh mutant background. Conversely, manipulations that blocked motor neuron action potentials or neurotransmitter release decreased levels of Frizzled-2C in the nucleus.
In the presynaptic compartment, WNT signalling was found to involve the regulation of GSK3β activity, as GSK3β inhibition was required in motor neurons for activity-dependent synaptic growth (FIG. 2). Whereas over-expressing GSK3β in motor neurons prevented bouton growth, expressing a GSK3β dominant-negative form bypassed activity requirements, as was observed by Wingless over-expression in motor neurons. Thus, Wingless release in an activity-dependent manner activates bidirectional pathways in the presynaptic and postsynaptic cell, with a divergent canonical pathway being activated in motor neurons and presumably regulating the presynaptic cytoskeleton, and the Frizzled nuclear import pathway activated in muscles presumably to regulate the development of the postsynaptic apparatus. The bidirectional activation of alternative pathways represents a mechanism to precisely match the development of presynaptic and postsynaptic structures, a crucial process during synapse development. Whether such a bidirectional signalling mechanism could also operate at the vertebrate NMJ is still unclear.
Further evidence that Wingless activated a divergent canonical pathway in motor neurons was provided by the finding that GSK3β over-expression, like mutations in wingless, also disrupted the presynaptic microtubule cytoskeleton67. However, it has also been suggested that GSK3β functions through AP1 by regulating the JUN N-terminal kinase (JNK) pathway68. Miech et al.67 further found that Arrow (also known as LRP5/6) and DVL but not the β-catenin-homologue Armadillo were present at the NMJ67. Mutations in arrow mimicked the wingless mutant phenotypes at the presynaptic terminal. However, Arrow seemed to have both presynaptic and postsynaptic functions as some phenotypes were rescued by expressing an arrow transgene in either presynaptic or postsynaptic cell. Disruption of DVL in neurons, by expressing a dominant-negative transgene mimicked the phenotypes resulting from disrupting Wingless and Arrow. However, no such effect was found on disrupting the function of the TCF homologue Pangolin or Armadillo, suggesting that presynaptic development is not regulated by the canonical pathway, but rather by the divergent canonical pathway (FIG. 2). However, the involvement of JNK68, an enzyme of the planar cell polarity pathway, suggests additional complexity on the pathways involved.
Besides Wingless (or WNT1), WNT5 and its atypical receptor DRL also function as positive regulators of NMJ development69 (FIG. 2). DRL is present at the NMJ and drl mutants have a significant reduction in synaptic bouton number. In addition, in wnt5 mutants, the density of active zones was decreased, although they remained unaffected in drl mutants, suggesting DRL-independent functions of WNT5. Functional defects in wnt5 mutants included a reduction in the amplitude of evoked excitatory junctional currents (EJCs), as well as the frequency of spontaneous miniature EJCs (mEJCs) similar to the defects in gsk3β68. However, both inhibition and overexpression of GSK3β led to a reduction in the amplitude of EJCs. WNT5 seemed to function in part in an anterograde manner, as over-expressing WNT5 in motor neurons suppressed the drl phenotype and DRL was required in muscle for normal NMJ growth. Further, expressing WNT5 in neurons but not in muscles, rescued the reduced synaptic bouton number of the wnt5 mutant, and over-expressing WNT5 in motor neurons led to synaptic overgrowth. However, the active zone phenotype was restored either by neuronal or muscle WNT5 expression, suggesting a potential retrograde function. Thus, more than one WNT pathway can function in parallel to positively regulate synapse development.
An inhibitory role for WNT signalling in Caenorhabditis elegans NMJ development
In C. elegans, muscles are innervated by cholinergic (excitatory) or GAbAergic (inhibitory) inputs. The contact between the axon and the muscle is through specialized muscle projections, called muscle arms that reach out to the nerve cord and form en-passant synapses70. A crucial factor for proper development of the C. elegans NMJ is the transmembrane protein LEV-10, which functions in AChR clustering71. In addition, AChR clustering is mediated by the MUSK-related orphan receptor tyrosine kinase CAM-1 (also known as KIN-8)72.
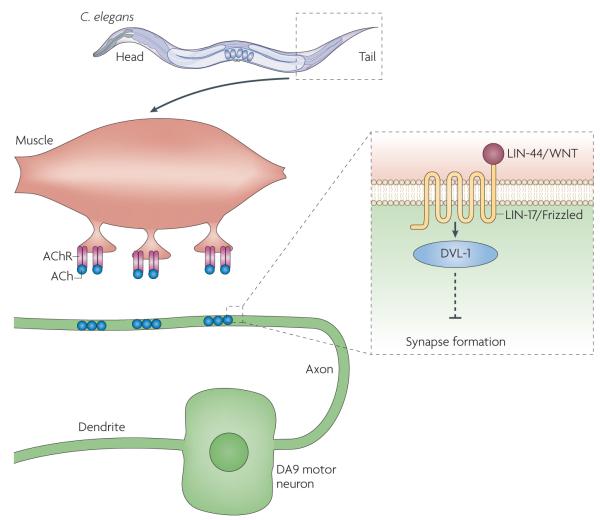
Two synaptic extracellular matrix (ECM) proteins have also been shown to regulate NMJ formation, collagen XVIII (CLE-1) and nidogen (NID-1)73. In C. elegans the WNTs, LIN-44 and EGL-20, and the Frizzled receptor LIN-17 inhibit synapse formation as shown by the observations that in lin-44 and lin-17 mutants the DA9 motor neuron forms ectopic NMJs and that these phenotypes are enhanced in egl-20 lin-44 double mutants74 (FIG. 3). Rescue experiments suggested that LIN-17, as well as DVL, but neither the β-catenins (BAR-1 and WRM-1) nor POP-1 (a lymphoid enhancer factor/TCF transcription factor), were required in the DA9 motor neuron to regulate the location of neuromuscular endings. This suggests the involvement of non-canonical WNT signalling. However, some components of non-canonical WNT signalling, such as FMI-1 (the homologue of Flamingo) or UNC-43 (the homologue of CaMKII) did not seem to be required.
Figure 3. inhibitory role of WNT signalling at the neuromuscular junction of Caenorhabditis elegans.
At the tail region of C. elegans, the axon from the DA9 motor neuron forms neuromuscular junctions (NMJs) with the body wall muscles. synaptic terminals do not form posteriorly as the axon receives the anti-synaptogenic WNT/LIN-44 signal. Inset: WNT/LIN-44 binds its receptor Frizzled/ LIN-17, which leads to activation of DVL-1 (Dishevelled 1). This signalling pathway prevents neuromuscular synapse formation. ACh, acetylcholine; AChr, ACh receptor.
WNT signalling during NMJ assembly seems to be local rather than global. This was demonstrated by shifting the distribution of Lin-44 by ectopic expression, which led to a shift of the asynaptic region within the DA9 motor neuron. The anti-synaptogenic influence of Lin-44 during C. elegans NMJ development is consistent with the finding in mammalian synapses that WNT3a inhibits NMJ formation. Future studies will be required to demonstrate if, as in other systems, C. elegans WNTs also have synaptogenic functions.
Perspectives
WNTs have emerged as crucial regulators of synaptic development throughout evolution. The studies discussed in this Progress article highlight the roles of WNT signalling in various aspects of synaptic development, including the organization of presynaptic and postsynaptic components, cytoskeletal structure and gene regulation at the NMJ.
These pathways can operate in parallel in the same cell to positively regulate different aspects of synapse formation or have an antagonist effect. The versatility of WNT signalling at synapses is further supported by evidence suggesting that WNTs can serve as anterograde, retrograde and autocrine signals. Moreover, WNT secretion by nonneuronal cells may also regulate synapse development. An important future question will be to determine whether WNTs have a role during synapse elimination. Given the diversity of WNTs and their receptors, the studies described in this Progress article are likely to represent just the tip of the iceberg, and therefore additional studies will be required to gain a better understanding of the breadth of WNT function in synapse development and function.
Acknowledgements
We thank Drs K. Koles, S. Speese, J. Ashley and P. Ramachandran for comments on the manuscript. V.B. is supported by grant MH70000 from the National Institutes of Health (NIH), USA.
Footnotes
DATABASES Flybase: http://flybase.org/
Arrow ∣ Dishevelled ∣ DrL ∣ Frizzled-2 ∣ Wingless ∣ Wnt5
OMIM: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=OMIM
Alzheimer’s disease ∣ Huntington’s disease ∣ schizophrenia
UniProtKB: http://www.uniprot.org
AChr ∣ Agrin ∣ EB1 ∣ MUsK ∣ PAK1 ∣ rapsyn ∣ WNT1 ∣ WNT3 ∣WNT7a ∣ WNT7b ∣ Wnt11r
FURTHER INFORMATION Vivian Budnik’s homepage: http://www.umassmed.edu/neuroscience/faculty/budnik.cfm
References
- 1.Siegfried E, Perrimon N. Drosophila wingless: a paradigm for the function and mechanism of Wnt signaling. Bioessays. 1994;16:395–404. doi: 10.1002/bies.950160607. [DOI] [PubMed] [Google Scholar]
- 2.Gould TD, Manji HK. The Wnt signaling pathway in bipolar disorder. Neuroscientist. 2002;8:497–511. doi: 10.1177/107385802237176. [DOI] [PubMed] [Google Scholar]
- 3.De Ferrari GV, Inestrosa NC. Wnt signaling function in Alzheimer’s disease. Brain Res. Brain Res. Rev. 2000;33:1–12. doi: 10.1016/s0165-0173(00)00021-7. [DOI] [PubMed] [Google Scholar]
- 4.Caricasole A, et al. Two sides of the same coin: Wnt signaling in neurodegeneration and neuro-oncology. Biosci. Rep. 2005;25:309–327. doi: 10.1007/s10540-005-2893-6. [DOI] [PubMed] [Google Scholar]
- 5.Inestrosa N, et al. Wnt signaling involvement in β-amyloid-dependent neurodegeneration. Neurochem. Int. 2002;41:341–344. doi: 10.1016/s0197-0186(02)00056-6. [DOI] [PubMed] [Google Scholar]
- 6.Johnson ML, Rajamannan N. Diseases of Wnt signaling. Rev. Endocr. Metab. Disord. 2006;7:41–49. doi: 10.1007/s11154-006-9003-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Speese SD, Budnik V. Wnts: up-and-coming at the synapse. Trends Neurosci. 2007;30:268–275. doi: 10.1016/j.tins.2007.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Salinas PC, Zou Y. Wnt signaling in neural circuit assembly. Annu. Rev. Neurosci. 2008;31:339–358. doi: 10.1146/annurev.neuro.31.060407.125649. [DOI] [PubMed] [Google Scholar]
- 9.Song Y, Balice-Gordon R. New dogs in the dogma: Lrp4 and Tid1 in neuromuscular synapse formation. Neuron. 2008;60:526–528. doi: 10.1016/j.neuron.2008.11.003. [DOI] [PubMed] [Google Scholar]
- 10.Widelitz R. Wnt signaling through canonical and noncanonical pathways: recent progress. Growth Factors. 2005;23:111–116. doi: 10.1080/08977190500125746. [DOI] [PubMed] [Google Scholar]
- 11.Lucas FR, Salinas PC. WNT-7a induces axonal remodeling and increases synapsin I levels in cerebellar neurons. Dev. Biol. 1997;192:31–44. doi: 10.1006/dbio.1997.8734. [DOI] [PubMed] [Google Scholar]
- 12.Krylova O, et al. WNT-3, expressed by motoneurons, regulates terminal arborization of neurotrophin-3-responsive spinal sensory neurons. Neuron. 2002;35:1043–1056. doi: 10.1016/s0896-6273(02)00860-7. [DOI] [PubMed] [Google Scholar]
- 13.Rosso SB, Sussman D, Wynshaw-Boris A, Salinas PC. Wnt signaling through Dishevelled, Rac and JNK regulates dendritic development. Nature Neurosci. 2005;8:34–42. doi: 10.1038/nn1374. [DOI] [PubMed] [Google Scholar]
- 14.Kummer TT, Misgeld T, Sanes JR. Assembly of the postsynaptic membrane at the neuromuscular junction: paradigm lost. Curr. Opin. Neurobiol. 2006;16:74–82. doi: 10.1016/j.conb.2005.12.003. [DOI] [PubMed] [Google Scholar]
- 15.Schaeffer L, de Kerchove d’Exaerde A, Changeux JP. Targeting transcription to the neuromuscular synapse. Neuron. 2001;31:15–22. doi: 10.1016/s0896-6273(01)00353-1. [DOI] [PubMed] [Google Scholar]
- 16.Sanes JR, Lichtman JW. Induction, assembly, maturation and maintenance of a postsynaptic apparatus. Nature Rev. Neurosci. 2001;2:791–805. doi: 10.1038/35097557. [DOI] [PubMed] [Google Scholar]
- 17.Misgeld T, et al. Roles of neurotransmitter in synapse formation: development of neuromuscular junctions lacking choline acetyltransferase. Neuron. 2002;36:635–648. doi: 10.1016/s0896-6273(02)01020-6. [DOI] [PubMed] [Google Scholar]
- 18.Weston C, Yee B, Hod E, Prives J. Agrin-induced acetylcholine receptor clustering is mediated by the small guanosine triphosphatases Rac and Cdc42. J. Cell Biol. 2000;150:205–212. doi: 10.1083/jcb.150.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Weston C, et al. Cooperative regulation by Rac and Rho of agrin-induced acetylcholine receptor clustering in muscle cells. J. Biol. Chem. 2003;278:6450–6455. doi: 10.1074/jbc.M210249200. [DOI] [PubMed] [Google Scholar]
- 20.Kim N, Burden SJ. MuSK controls where motor axons grow and form synapses. Nature Neurosci. 2008;11:19–27. doi: 10.1038/nn2026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lefebvre JL, Jing L, Becaficco S, Franzini-Armstrong C, Granato M. Differential requirement for MuSK and dystroglycan in generating patterns of neuromuscular innervation. Proc. Natl Acad. Sci. USA. 2007;104:2483–2488. doi: 10.1073/pnas.0610822104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lin W, et al. Distinct roles of nerve and muscle in postsynaptic differentiation of the neuromuscular synapse. Nature. 2001;410:1057–1064. doi: 10.1038/35074025. [DOI] [PubMed] [Google Scholar]
- 23.Yang X, et al. Patterning of muscle acetylcholine receptor gene expression in of motor innervation. Neuron. 2001;30:399–410. doi: 10.1016/s0896-6273(01)00287-2. [DOI] [PubMed] [Google Scholar]
- 24.Bezakova G, Rabben I, Sefland I, Fumagalli G, Lomo T. Neural agrin controls acetylcholine receptor stability in skeletal muscle fibers. Proc. Natl Acad. Sci. USA. 2001;98:9924–9929. doi: 10.1073/pnas.171539698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Luo ZG, et al. Regulation of AChR clustering by Dishevelled interacting with MuSK and PAK1. Neuron. 2002;35:489–505. doi: 10.1016/s0896-6273(02)00783-3. [DOI] [PubMed] [Google Scholar]
- 26.Kim CH, Xiong WC, Mei L. Regulation of MuSK expression by a novel signaling pathway. J. Biol. Chem. 2003;278:38522–38527. doi: 10.1074/jbc.M305058200. [DOI] [PubMed] [Google Scholar]
- 27.Wang J, et al. Wnt/β-catenin signaling suppresses Rapsyn expression and inhibits acetylcholine receptor clustering at the neuromuscular junction. J. Biol. Chem. 2008;283:21668–21675. doi: 10.1074/jbc.M709939200. [DOI] [PubMed] [Google Scholar]
- 28.Henriquez JP, et al. Wnt signaling promotes AChR aggregation at the neuromuscular synapse in collaboration with agrin. Proc. Natl Acad. Sci. USA. 2008;105:18812–18817. doi: 10.1073/pnas.0806300105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jing L, Lefebvre JL, Gordon LR, Granato M. Wnt signals organize synaptic prepattern and axon guidance through the zebrafish unplugged/MuSK receptor. Neuron. 2009;61:721–733. doi: 10.1016/j.neuron.2008.12.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Castelo-Branco G, et al. Ventral midbrain glia express region-specific transcription factors and regulate dopaminergic neurogenesis through Wnt-5a secretion. Mol. Cell Neurosci. 2006;31:251–262. doi: 10.1016/j.mcn.2005.09.014. [DOI] [PubMed] [Google Scholar]
- 31.Hooper JE. Distinct pathways for autocrine and paracrine Wingless signalling in Drosophila embryos. Nature. 1994;372:461–464. doi: 10.1038/372461a0. [DOI] [PubMed] [Google Scholar]
- 32.Li XM, et al. Retrograde regulation of motoneuron differentiation by muscle beta-catenin. Nature Neurosci. 2008;11:262–268. doi: 10.1038/nn2053. [DOI] [PubMed] [Google Scholar]
- 33.Wang J, LUO Z-G. The role of Wnt/ß-catenin signaling in postsynaptic differentiation. Commun. Integr. Biol. 2008;1:1–3. doi: 10.4161/cib.1.2.7099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang B, et al. Beta-catenin regulates acetylcholine receptor clustering in muscle cells through interaction with rapsyn. J. Neurosci. 2007;27:3968–3973. doi: 10.1523/JNEUROSCI.4691-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ohno K, Sadeh M, Blatt I, Brengman JM, Engel AG. E-box mutations in the RAPSN promoter region in eight cases with congenital myasthenic syndrome. Hum. Mol. Genet. 2003;12:739–748. doi: 10.1093/hmg/ddg089. [DOI] [PubMed] [Google Scholar]
- 36.Deng J, et al. Beta-catenin interacts with and inhibits NF-κB in human colon and breast cancer. Cancer Cell. 2002;2:323–334. doi: 10.1016/s1535-6108(02)00154-x. [DOI] [PubMed] [Google Scholar]
- 37.Sanes JR, Lichtman JW. Development of the vertebrate neuromuscular junction. Annu. Rev. Neurosci. 1999;22:389–442. doi: 10.1146/annurev.neuro.22.1.389. [DOI] [PubMed] [Google Scholar]
- 38.Svensson A, Norrby M, Libelius R, Tagerud S. Secreted frizzled related protein 1 (Sfrp1) and Wnt signaling in innervated and denervated skeletal muscle. J. Mol. Histol. 2008;39:329–337. doi: 10.1007/s10735-008-9169-y. [DOI] [PubMed] [Google Scholar]
- 39.Glass DJ, et al. Agrin acts via a MuSK receptor complex. Cell. 1996;85:513–523. doi: 10.1016/s0092-8674(00)81252-0. [DOI] [PubMed] [Google Scholar]
- 40.Weatherbee SD, Anderson KV, Niswander LA. LDL-receptor-related protein 4 is crucial for formation of the neuromuscular junction. Development. 2006;133:4993–5000. doi: 10.1242/dev.02696. [DOI] [PubMed] [Google Scholar]
- 41.DeChiara TM, et al. The receptor tyrosine kinase MuSK is required for neuromuscular junction formation in vivo. Cell. 1996;85:501–512. doi: 10.1016/s0092-8674(00)81251-9. [DOI] [PubMed] [Google Scholar]
- 42.Kim N, et al. Lrp4 is a receptor for Agrin and forms a complex with MuSK. Cell. 2008;135:334–342. doi: 10.1016/j.cell.2008.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhang B, et al. LRP4 serves as a coreceptor of agrin. Neuron. 2008;60:285–297. doi: 10.1016/j.neuron.2008.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang B, Xiong WC, Mei L. Get ready to Wnt: prepatterning in neuromuscular junction formation. Dev. Cell. 2009;16:325–327. doi: 10.1016/j.devcel.2009.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang J, et al. Regulation of acetylcholine receptor clustering by the tumor suppressor APC. Nature Neurosci. 2003;6:1017–1018. doi: 10.1038/nn1128. [DOI] [PubMed] [Google Scholar]
- 46.Akhmanova A, Hoogenraad CC. Microtubule plus-end-tracking proteins: mechanisms and functions. Curr. Opin. Cell Biol. 2005;17:47–54. doi: 10.1016/j.ceb.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 47.Reilein A, Nelson WJ. APC is a component of an organizing template for cortical microtubule networks. Nature Cell Biol. 2005;7:463–473. doi: 10.1038/ncb1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rosenberg MM, et al. Adenomatous polyposis coli plays a key role, in vivo, in coordinating assembly of the neuronal nicotinic postsynaptic complex. Mol. Cell Neurosci. 2008;38:138–152. doi: 10.1016/j.mcn.2008.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Temburni MK, Rosenberg MM, Pathak N, McConnell R, Jacob MH. Neuronal nicotinic synapse assembly requires the adenomatous polyposis coli tumor suppressor protein. J. Neurosci. 2004;24:6776–6784. doi: 10.1523/JNEUROSCI.1826-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Broadie K, Bate M. Innervation directs receptor synthesis and localization in Drosophila embryo synaptogenesis. Nature. 1993;361:350–353. doi: 10.1038/361350a0. [DOI] [PubMed] [Google Scholar]
- 51.Featherstone DE, Rushton E, Broadie K. Developmental regulation of glutamate receptor field size by nonvesicular glutamate release. Nature Neurosci. 2002;5:141–146. doi: 10.1038/nn789. [DOI] [PubMed] [Google Scholar]
- 52.Augustin H, Grosjean Y, Chen K, Sheng Q, Featherstone DE. Nonvesicular release of glutamate by glial xCT transporters suppresses glutamate receptor clustering in vivo. J. Neurosci. 2007;27:111–123. doi: 10.1523/JNEUROSCI.4770-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Daniels RW, et al. A single vesicular glutamate transporter is sufficient to fill a synaptic vesicle. Neuron. 2006;49:11–16. doi: 10.1016/j.neuron.2005.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liebl FL, Featherstone DE. Identification and investigation of Drosophila postsynaptic density homologs. Bioinform Biol. Insights. 2008;2:375–387. doi: 10.4137/bbi.s2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liebl FL, Featherstone DE. Genes involved in Drosophila glutamate receptor expression and localization. BMC Neurosci. 2005;6:44. doi: 10.1186/1471-2202-6-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Marques G, Zhang B. Retrograde signaling that regulates synaptic development and function at the Drosophila neuromuscular junction. Int. Rev. Neurobiol. 2006;75:267–285. doi: 10.1016/S0074-7742(06)75012-7. [DOI] [PubMed] [Google Scholar]
- 57.Packard M, et al. The Drosophila wnt, wingless, provides an essential signal for pre- and postsynaptic differentiation. Cell. 2002;111:319–330. doi: 10.1016/s0092-8674(02)01047-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ataman B, et al. Nuclear trafficking of Drosophila Frizzled-2 during synapse development requires the PDZ protein dGRIP. Proc. Natl Acad. Sci. USA. 2006;103:7841–7846. doi: 10.1073/pnas.0600387103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gogel S, Wakefield S, Tear G, Klambt C, Gordon-Weeks PR. The Drosophila microtubule associated protein Futsch is phosphorylated by Shaggy/Zestewhite 3 at an homologous GSK3β phosphorylation site in MAP1B. Mol. Cell Neurosci. 2006;33:188–199. doi: 10.1016/j.mcn.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 60.Mathew D, et al. Wingless signaling at synapses is through cleavage and nuclear import of receptor DFrizzled2. Science. 2005;310:1344–1347. doi: 10.1126/science.1117051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hoogenraad CC, Milstein AD, Ethell IM, Henkemeyer M, Sheng M. GRIP1 controls dendrite morphogenesis by regulating EphB receptor trafficking. Nature Neurosci. 2005;8:906–915. doi: 10.1038/nn1487. [DOI] [PubMed] [Google Scholar]
- 62.Gomez-Ospina N, Tsuruta F, Barreto-Chang O, Hu L, Dolmetsch R. The C terminus of the l-type voltage-gated calcium channel CaV1.2 encodes a transcription factor. Cell. 2006;127:591–606. doi: 10.1016/j.cell.2006.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lin SY, et al. Nuclear localization of EGF receptor and its potential new role as a transcription factor. Nature Cell Biol. 2001;3:802–808. doi: 10.1038/ncb0901-802. [DOI] [PubMed] [Google Scholar]
- 64.Baron M, et al. Multiple levels of Notch signal regulation. Mol. Membr. Biol. 2002;19:27–38. doi: 10.1080/09687680110112929. [DOI] [PubMed] [Google Scholar]
- 65.Ataman B, et al. Rapid activity-dependent modifications in synaptic structure and function require bidirectional wnt signaling. Neuron. 2008;57:705–718. doi: 10.1016/j.neuron.2008.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Barco A, Bailey CH, Kandel ER. Common molecular mechanisms in explicit and implicit memory. J. Neurochem. 2006;97:1520–1533. doi: 10.1111/j.1471-4159.2006.03870.x. [DOI] [PubMed] [Google Scholar]
- 67.Miech C, Pauer HU, He X, Schwarz TL. Presynaptic local signaling by a canonical wingless pathway regulates development of the Drosophila neuromuscular junction. J. Neurosci. 2008;28:10875–10884. doi: 10.1523/JNEUROSCI.0164-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Franciscovich AL, Mortimer AD, Freeman AA, Gu J, Sanyal S. Overexpression screen in Drosophila identifies neuronal roles of GSK-3β/shaggy as a regulator of AP-1-dependent developmental plasticity. Genetics. 2008;180:2057–2071. doi: 10.1534/genetics.107.085555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liebl FL, et al. Derailed regulates development of the Drosophila neuromuscular junction. Dev. Neurobiol. 2008;68:152–165. doi: 10.1002/dneu.20562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dixon SJ, Roy PJ. Muscle arm development in Caenorhabditis elegans. Development. 2005;132:3079–3092. doi: 10.1242/dev.01883. [DOI] [PubMed] [Google Scholar]
- 71.Gally C, Eimer S, Richmond JE, Bessereau JL. A transmembrane protein required for acetylcholine receptor clustering in Caenorhabditis elegans. Nature. 2004;431:578–582. doi: 10.1038/nature02893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Francis MM, et al. The Ror receptor tyrosine kinase CAM-1 is required for ACR-16-mediated synaptic transmission at the C. elegans neuromuscular junction. Neuron. 2005;46:581–594. doi: 10.1016/j.neuron.2005.04.010. [DOI] [PubMed] [Google Scholar]
- 73.Ackley BD, et al. The basement membrane components nidogen and type XVIII collagen regulate organization of neuromuscular junctions in Caenorhabditis elegans. J. Neurosci. 2003;23:3577–3587. doi: 10.1523/JNEUROSCI.23-09-03577.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Klassen MP, Shen K. Wnt signaling positions neuromuscular connectivity by inhibiting synapse formation in C. elegans. Cell. 2007;130:704–716. doi: 10.1016/j.cell.2007.06.046. [DOI] [PubMed] [Google Scholar]
- 75.Ciani L, Krylova O, Smalley MJ, Dale TC, Salinas PC. A divergent canonical WNT-signaling pathway regulates microtubule dynamics: dishevelled signals locally to stabilize microtubules. J. Cell Biol. 2004;164:243–253. doi: 10.1083/jcb.200309096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lucas FR, Goold RG, Gordon-Weeks PR, Salinas PC. Inhibition of GSK-3β leading to the loss of phosphorylated MAP-1B is an early event in axonal remodelling induced by WNT-7a or lithium. J. Cell Sci. 1998;111:1351–1361. doi: 10.1242/jcs.111.10.1351. [DOI] [PubMed] [Google Scholar]
- 77.Zhong W. Going nuclear is again a winning (Wnt) strategy. Dev. Cell. 2008;15:635–636. doi: 10.1016/j.devcel.2008.10.012. [DOI] [PubMed] [Google Scholar]
- 78.Lyu J, Yamamoto V, Lu W. Cleavage of the Wnt receptor Ryk regulates neuronal differentiation during cortical neurogenesis. Dev. Cell. 2008;15:773–780. doi: 10.1016/j.devcel.2008.10.004. [DOI] [PubMed] [Google Scholar]