Abstract
Background/Aim:
Chronic diarrhea is defined as a decrease in fecal consistency lasting for four or more weeks. Prevalence of this complication in the general population is 5%. Mast cells that play an important role in the regulation of gastrointestinal visceral sensitivity and vascular permeability may be involved in functional chronic diarrhea. In this study we tried to evaluate mast cells density in colonic mucosa of patients with chronic diarrhea.
Patients and Methods:
50 patients with chronic diarrhea and 50 persons as control group were investigated. All specimens were immunohistochemically stained for mast cell tryptase (MCT) with monoclonal mouse anti-human MCT as well as toluidine blue. Mean number of mucosal mast cells were counted in 10 high power microscopic fields of patients and control groups.
Results:
In patients group (age range, 15-78 years; 26 females), the number of mast cells per high power field in the immunohistochemistry staining was 21.3 ± 4.8 compared to 14.2 ± 3.4 in the control group (age range, 18–78 years; 24 females) [P < 0.001]. Also number of mast cells in toluidine blue staining was 10.3 ± 3.6 per high power field in cases and 7.1 ± 2.4 in the control group (P < 0.001).
Conclusion:
Elevated number of colonic mast cells exist in patients with chronic diarrhea. Further research should be considered on application of these findings for new therapeutic opportunities.
Keywords: Chronic diarrhea, colonic mucosa, mast cell, toluidine blue
Chronic diarrhea is defined as loose or watery stool lasting for four or more weeks. The prevalence of this complication is about 5% in the general population of the United States.[1,2] Unlike acute diarrhea that has often infectious etiology, most causes of chronic diarrhea are non-infectious and include secretory diarrhea, osmotic diarrhea, fatty diarrhea, and inflammatory diarrhea.[2] Most of these conditions can be diagnosed by a detailed history, physical examination, routine laboratory tests, therapeutic trials, stool analysis, and further evaluations such as culture studies, radiography, endoscopy, and biopsies.[1–3] Despite these efforts and diagnostic procedures, the diagnosis remains unknown and treatment is disappointing in some patients.[1,2] This form of disease is known as functional chronic diarrhea.[1]
Basic understanding of colonic physiology has shown that mast cells are the main regulators of motor and sensory function of the intestine, and reactive to unknown stimuli involved in functional chronic diarrhea.[1] Mast cells are certain cells with hematopoietic origin, containing abundant specific basophilic metachromatic granules in their cytoplasm that have a tissue distribution and are abundant in the gastrointestinal mucosa.[4] Based on protease content, two subpopulation of mast cells are differentiated: reactive mucosal mast cells containing tryptase that increases in response to stimuli, and sub mucosal mast cells containing chymase and tryptase that are associated with angiogenesis and tissue repair and have no interaction with the immune system.[5]
Because the colon biopsy specimens in some patients with chronic diarrhea do not show pathological findings in routine haematoxylin – eosin stained sections, we decided to study the number of mucosal mast cells in these patients with toluidine blue staining and immunohistochemical analysis for mast cells tryptase.
PATIENTS AND METHODS
Patients group
The study group composed of 50 adult patients having chronic diarrhea who were referred to our centre between March 2007 and December 2010 (age range 15-78 years, mean age 39.5 years, 26 females and 24 males) with macroscopically and histologically normal colonic mucosa. Organic and metabolic causes of diarrhea had also been excluded by complete blood count, stool examination, previous endoscopy and/or barium enema. The patients underwent a full colonoscopy and random biopsy samples (4-8 specimens) were taken from different parts of the colon.
Control group
The control group composed of 50 persons (age range 18–78 years, mean age 40.2 years, 24 females and 26 males) with colonic biopsies obtained during investigations for anemia, family history of colonic cancer, mucosa adjacent to adenomatous polyps or those with upper gastrointestinal complaints (Helicobacter gastritis and gastroesophageal reflux disease).
In both groups, persons with a history of allergy, recent traveling and drugs consumption (such as anti-cholinergic, antihistamines and non-steroidal anti-inflammatory drugs [NSAIDs]) were excluded.
Histopathologic evaluation
After reviewing of slides all sections were immunohistochemically stained for mast cell tryptase (MCT) as follows: 4-mm-thin sections were cut, dried, and deparaffinized before they were treated with protease 1 for 4 minutes and then incubated with monoclonal mouse anti-human MCT (clone AA1, code M 7052; DAKO, Carpentaria, California; dilution 1:200) for 30 minutes.
In addition, all sections were stained with toluidine blue stock solution as follows:
1 gr toluidine blue O, 100 ml 70% alcohol, were mixed to dissolve and then sodium chloride (1%) was added, pH is adjusted to 2.0~2.5 using glacial acetic acid.
4-mm-thin sections were cut, dried and deparaffinized to distilled water, embedded in toluidine blue working solution for 3 minutes, washed in distilled water (3 times), dehydrated quickly through 95% and 2 times of 100% alcohol and finally 2 times were cleared in xylene.
A neurofibroma tissue was also used as positive control. Primary antibody in immunohistochemistry (IHC) procedure was omitted as a negative control. Also we used internal negative control in biopsied specimens.
Immunohistochemically positive-stained mast cells were counted in 10 consecutive non-overlapping microscopic fields at a magnification of 400 and the average number of mast cells per high power field was calculated. In biopsies with a low number of pieces mast cells counted in 5 high power microscopic fields. Muscularis mucosa and submucosa were excluded from mast cell counts. Only the intact mast cells with visible nuclei and darkly staining cytoplasm's were counted. Wisps of extracellular, apparently degranulated tryptase were excluded from counts. In toluidine blue staining, metachromatic cells with red to purple granules were counted.
The number of mast cells were counted and compared between the cases and control groups.
RESULTS
The study consisted of 100 samples, including 50 patients suffering from chronic diarrhea (cases) and 50 persons without chronic diarrhea (control group) with normal appearing colonic mucosa on routine haematoxylin-eosin staining. Comparison of gender showed no significant difference (P = 0.689). The mean age was 39.5 ± 14.5 in cases and 40.2 ± 16.7 in the control group with no significant difference (P = 0.658).
The density of mucosal mast cells was 14.2 ± 3.4 cells per high-power field in the control group compared to 21.3 ± 4.8 cells per high power field in the study group (P < 0.001) [Figures 1–4].
Figure 1.
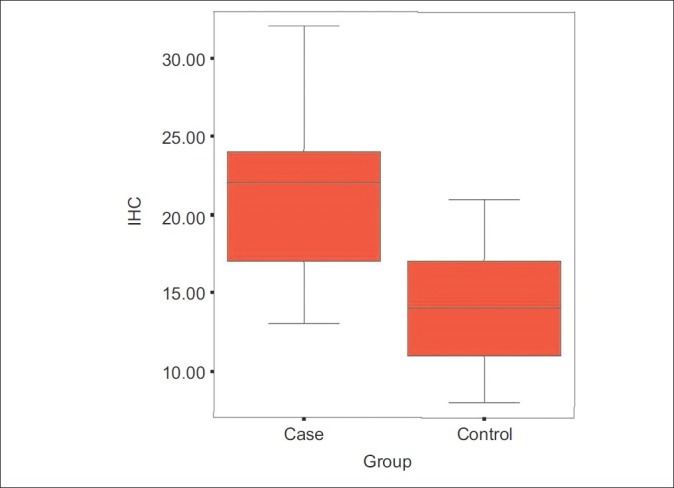
Distribution of mast cells in IHC staining
Figure 4.
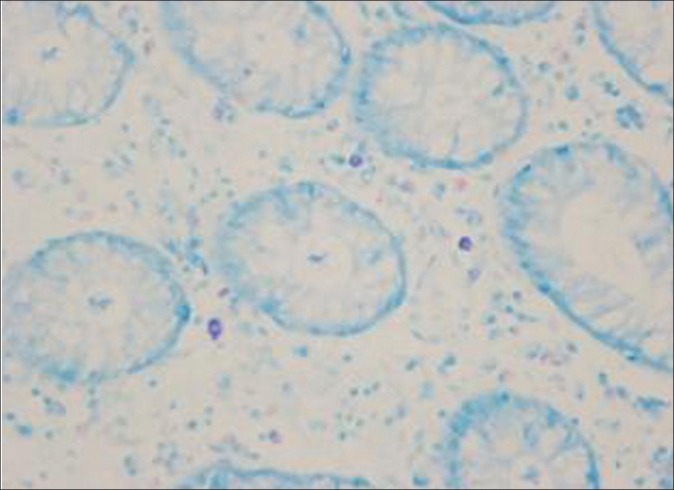
Distribution of mast cells in patients with chronic diarrhea visualized by toluidine blue staining (original magnification × 400)
Figure 2.
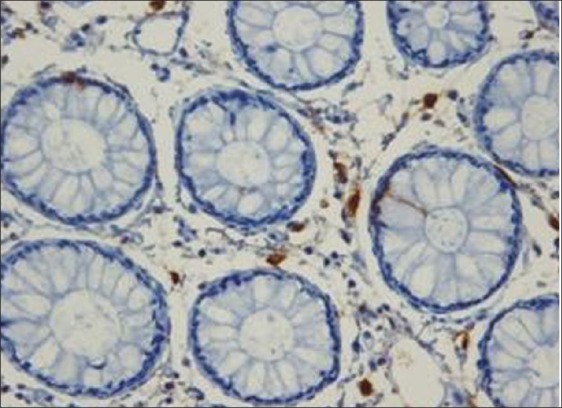
Distribution of mast cells in patients with chronic diarrhea visualized by immunohistochemical staining for mast cell tryptase (original magnification × 400)
Figure 3.
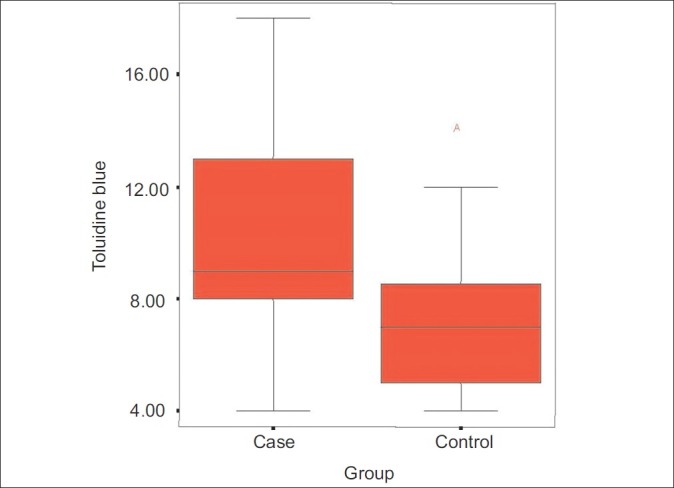
Distribution of mast cells in toluidine blue staining
There was a strong relationship between the number of mast cells in the IHC staining and toluidine blue method (correlation coefficient 0.730; P < 0.001).
No relationship between the age and number of mast cells was detected in cases group (correlation coefficient in the IHC staining was 0.09, P = 0.897; correlation coefficient in toluidine blue staining was - 0.127, P = 0.507).
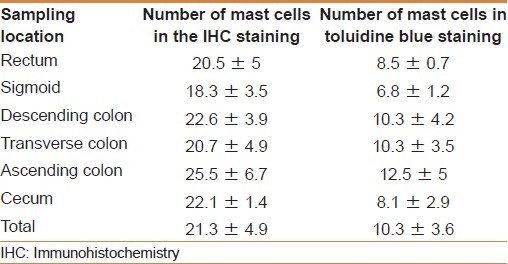
Mean number of mast cells in cases group in terms of sampling location was not statistically significant (P = 0.461 for IHC staining and P = 0.281 for toluidine blue staining) [Table 1].
Table 1.
Mean number of mast cells in the cases group in terms of sampling location
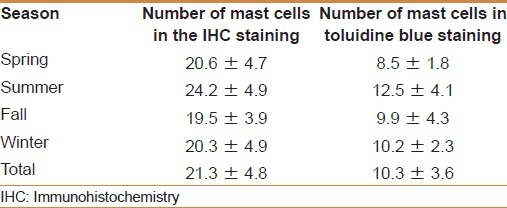
Also the mean number of mast cells in cases based on sampling in different seasons of the year was analyzed. There is no significant difference for different seasons of the year (P = 0.387 for IHC method and P = 0.192 for toluidine blue staining) [Table 2].
Table 2.
Mean number of mast cells in cases based on sampling in different seasons of year
DISCUSSION
Mast cells are functionally and morphologically distinct cells that develop from CD-34 positive or c-kit positive progenitor cells of bone marrow.[6] Mast cells contain abundant specific basophilic metachromatic granules in their cytoplasm, have a tissue distribution and are abundant in the gastrointestinal mucosa.[4] Up to 40% of the volume of mast cell is occupied by secretory granules which originate from the Golgi apparatus, and is responsible for the synthesis and organization of the preformed mediators contained therein.[7,8] Upon activation mast cell can release its mediators to fulfill its biological functions. Physiology of the colon recognized that mast cells that are the main regulator of motor and sensory function of the intestine, react in response to unknown stimuli involved in functional chronic diarrhea.[1] These cells that constitute 2-5% of mononuclear cells in the gastrointestinal lamina propria, have the average level of 13 cells per high power microscopic field in colon and duodenal mucosa.[1,9]
The role of mast cells and their mediators have been studied in a number of gastrointestinal diseases that cause chronic diarrhea. Increased, normal or decreased numbers of mast cells in patients with inflammatory bowel disease (IBD),[4,10,11] ulcerative colitis,[1,12,13] Crohn's disease,[14] collagenous colitis[15,16] and gluten- sensitive enteropathy[17] have been reported. Considering the role of mast cells in the pathogenesis of these diseases according to the diffuse tissue distribution, its close relationship with the intestinal nervous system is inevitable.
Among the diseases causing chronic diarrhea, diagnosis of irritable bowel syndrome (IBS) is controversial and is based on the patient's symptoms and exclusion of other diseases. The role of mast cells in the pathogenesis of this disease is still being investigated and there are no specific criteria for diagnosis. Although one study suggested a cutoff of 20 mast cells per high power field (as 2 SD higher than normal number of mast cells in the general population),[1] but other studies did not support these findings. Colonic biopsy of patients that were enrolled in this study showed the average of 21.3 ± 4.8 mast cells per high power field that was significantly greater than control group.[18]
Results of our study are consistent with those of previous studies. Jakate and colleagues in their study in 2006 investigated the number of intestinal mucosal mast cells in patients with chronic diarrhea and revealed more than twenty cells in each high magnification field of microscope.[1] Also diarrhea-predominant IBS patients showed marked increase in mast cell numbers and higher tryptase concentration in jejunal fluid, according to Guilarte et al. study.[5] Barbara and colleagues in 2004 studied IBS patients with gastrointestinal symptoms and demonstrated higher mast cells density than controls.[19] The numbers of mast cells in patients with constipation-predominant IBS and diarrhea-predominant IBS at the ileum were significantly more than that in the normal group in a study by Wang and coworkers.[20] Also Park et al, in 2006 demonstrated that numbers of mast cells were significantly greater in the terminal ileum, ascending colon and rectum of diarrhea-predominant IBS patients compared with controls.[21] Walker and colleagues in 2009 studied duodenal mastocytosis, eosinophilia and intraepithelial lymphocytosis as possible disease markers in IBS and functional dyspepsia. They showed significant increase in number of duodenal mast cells in IBS patients.[22] Similarly, an increased number of mast cells has been found in the cecum, terminal ileum, and colon of patients with IBS in other studies.[23–26]
Findings of our study also indicated that mean number of mast cells per high power microscopic field with toluidine blue staining was significantly higher in cases group compared to control group but in comparison with the results of IHC staining, fewer number of mast cell was determined (because only heavily granulated mast cells showed metachromasia with toluidine blue staining). This finding is similar to study of Jakate and colleagues. They eventually eliminated results of toluidine blue staining from their analysis.[1]
This study did not show any correlation between age, gender, site of colonic biopsy and sampling in different seasons of year with mast cell number, whereas some studies have shown a potential influence of clinical history or age on mucosal mast cell numbers.[19,26–27]
CONCLUSION
In chronic intractable diarrhea, colonic biopsy specimens may appear unremarkable on routine haematoxylin-eosin staining, but increased mast cells may be demonstrated by immunohistochemistry for mast cell tryptase. Similar increases in mast cells were not apparent in control population. Our study also revealed that age, gender, site of colonic biopsy and different seasons of year had no effect on number of mast cells. Further research should be considered on application of these data in treatment of patients with drugs affecting mast cell function.
Footnotes
Source of Support: This research was supported by Tehran university of medical sciences
Conflict of Interest: None declared.
REFERENCES
- 1.Jakate S, Demeo M, John R, Tobin M, Keshavarzian A. Mastocytic enterocolitis: Increased mucosal mast cells in chronic intractable diarrhea. Arch Pathol Lab Med. 2006;130:362–7. doi: 10.5858/2006-130-362-MEIMMC. [DOI] [PubMed] [Google Scholar]
- 2.Fine KD, Schiller LR. AGA technical review on the evaluation and management of chronic diarrhea. Gastroenterology. 1999;116:1464–86. doi: 10.1016/s0016-5085(99)70513-5. [DOI] [PubMed] [Google Scholar]
- 3.Schiller LR. Chronic diarrhea. Gasroenterology. 2004;127:287–93. doi: 10.1053/j.gastro.2004.05.028. [DOI] [PubMed] [Google Scholar]
- 4.He SH. Key role of mast cells and their major secretory products in inflammatory bowel disease. World J Gastroenterol. 2004;10:309–18. doi: 10.3748/wjg.v10.i3.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guilarte M, Santos J, de Torres I, Alonso C, Vicario M, Ramos L, et al. Diarrhea-predominant IBS patients show mast cell activation and hyperplasia in the jejunum. Gut. 2007;56:203–9. doi: 10.1136/gut.2006.100594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kirshenbaum AS, Goff JP, Semere T, Foster B, Scott LM, Metcalfe DD. Demonstration that human mast cells arise from a progenitor cell population that is CD34 (+), c-kit (+), and expresses aminopeptidase N (CD13) Blood. 1999;94:2333–42. [PubMed] [Google Scholar]
- 7.Schwartz LB. Tryptase from human mast cells: Biochemistry, biology and clinical utility. Monogr Allergy. 1990;27:90–113. [PubMed] [Google Scholar]
- 8.Caulfield JP, Lewis RA, Hein A, Austen KF. Secretion in dissociated human pulmonary mast cells. Evidnece of solubilization of granule contents before discharge. J Cell Biol. 1980;85:299–312. doi: 10.1083/jcb.85.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Feldman M, Friedman L, Brandt L. Gastrointestinal and liver disease. 8th ed. Philadelphia, PA: Saunders; 2006. p. 31. [Google Scholar]
- 10.Stoyanova II, Gulubova MV. Mast cells and inflammatory mediators in chronic ulcerative colitis. Acta Histochem. 2002;104:185–92. doi: 10.1078/0065-1281-00641. [DOI] [PubMed] [Google Scholar]
- 11.Bischoff SC, Wedemeyer J, Herrmann A, Meier PN, Trautwein C, Cetin Y, et al. Quantitative assessment of intestinal eosinophils and mast cells in inflammatory bowel disease. Histopathology. 1996;28:1–13. doi: 10.1046/j.1365-2559.1996.262309.x. [DOI] [PubMed] [Google Scholar]
- 12.Raithel M, Winterkamp S, Pacurar A, Ulrich P, Hochberger J, Hahn EG. Release of mast cell tryptase from human colorectal mucosa in inflammatory bowel disease. Scand J Gastroenterol. 2001;36:174–9. doi: 10.1080/003655201750065933. [DOI] [PubMed] [Google Scholar]
- 13.Raithel M, Matek M, Baenkler HW, Jorde W, Hahn EG. Mucosal histamine content and histamine secretion in Crohn's disease, ulcerative colitis and allergic enteropathy. Int Arch Allergy Immunol. 1995;108(2):127–33. doi: 10.1159/000237129. [DOI] [PubMed] [Google Scholar]
- 14.Dvorak AM, Monahan RA, Osage JE, Dickersin GR. Crohn's disease: Transmission electron microscopic studies. II. Immunologic inflammatory response. Alterations of mast cells, basophils, eosinophils, and the microvasculature. Hum Pathol. 1980;11:606–19. doi: 10.1016/s0046-8177(80)80072-4. [DOI] [PubMed] [Google Scholar]
- 15.Schwab D, Raithel M, Hahn EG. Evidence for mast cell activation in collagenous colitis. Inflamm Res. 1998;47(suppl 1):S64–5. doi: 10.1007/s000110050276. [DOI] [PubMed] [Google Scholar]
- 16.Nishida Y, Murase K, Isomoto H, Furusu H, Mizuta Y, Riddell RH, et al. Different distribution of mast cells and macrophages in colonic mucosa of patients with collagenous colitis and inflammatory bowel disease. Hepatogastroenterology. 2002;49:678–82. [PubMed] [Google Scholar]
- 17.Suranyi Y, Freier S, Faber J, Dollberg L. Intestinal mast cells in different stages of celiac disease. Isr J Med Sci. 1986;22:370–5. [PubMed] [Google Scholar]
- 18.Hahn HP, Hornick JL. Immunoreactivity for CD25 in gastrointestinal mucosal mast cells is specific for systemic mastocytosis. Am J Surg Pathol. 2007;31:1669–76. doi: 10.1097/PAS.0b013e318078ce7a. [DOI] [PubMed] [Google Scholar]
- 19.Barbara G, Stanghellini V, De Giorgio R, Cremon C, Cottrell GS, Santini D, et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology. 2004;126:693–702. doi: 10.1053/j.gastro.2003.11.055. [DOI] [PubMed] [Google Scholar]
- 20.Wang SH, Dong L, Luo JY, Gong J, Li L, Lu XL, et al. Decreased expression of serotonin in the jejunum and increased numbers of mast cells in the terminal ileum in patients with irritable bowel syndrome. World J Gastroenterol. 2007;13:6041–7. doi: 10.3748/wjg.v13.45.6041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park JH, Rhee PL, Kim HS, Lee JH, Kim YH, Kim JJ, et al. Mucosal mast cell counts correlate with visceral hypersensitivity in patients with diarrhea predominant irritable bowel syndrome. J Gastroenterol Hepatol. 2006;21(1 Pt 1):71–8. doi: 10.1111/j.1440-1746.2005.04143.x. [DOI] [PubMed] [Google Scholar]
- 22.Walker MM, Talley NJ, Prabhakar M, Pennaneac’h CJ, Aro P, Ronkainen J, et al. Duodenal mastocytosis, eosinophilia and intra-epithelial lymphocytosis as possible disease markers in the irritable bowel syndrome and functional dyspepsia. Aliment Pharmacol Ther. 2009;29:765–73. doi: 10.1111/j.1365-2036.2009.03937.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chadwick VS, Chen W, Shu D, Paulus B, Bethwaite P, Tie A, et al. Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology. 2002;122:1778–83. doi: 10.1053/gast.2002.33579. [DOI] [PubMed] [Google Scholar]
- 24.Spiller RC. Post infectious irritable bowel syndrome. Gastroenterology. 2003;124:1662–71. doi: 10.1016/s0016-5085(03)00324-x. [DOI] [PubMed] [Google Scholar]
- 25.Barbara G, Stanghellini V, De Giorgio R, Corinaldesi R. Functional gastrointestinal disorders and mast cells: Implication for therapy. Neurogastroenterol Motil. 2006;18:6–17. doi: 10.1111/j.1365-2982.2005.00685.x. [DOI] [PubMed] [Google Scholar]
- 26.Cremon C, Gargano L, Morselli-Labate AM, Santini D, Cogliandro RF, De Giorgio R, et al. Mucosal immune activation in irritable bowel syndrome: Gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392–400. doi: 10.1038/ajg.2008.94. [DOI] [PubMed] [Google Scholar]
- 27.Dunlop SP, Jenkins D, Spiller RC. Age-related decline in rectal mucosal lymphocytes and mast cells. Eur J Gastroenterol Hepatol. 2004;16:1011–5. doi: 10.1097/00042737-200410000-00010. [DOI] [PubMed] [Google Scholar]


