Abstract
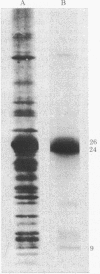
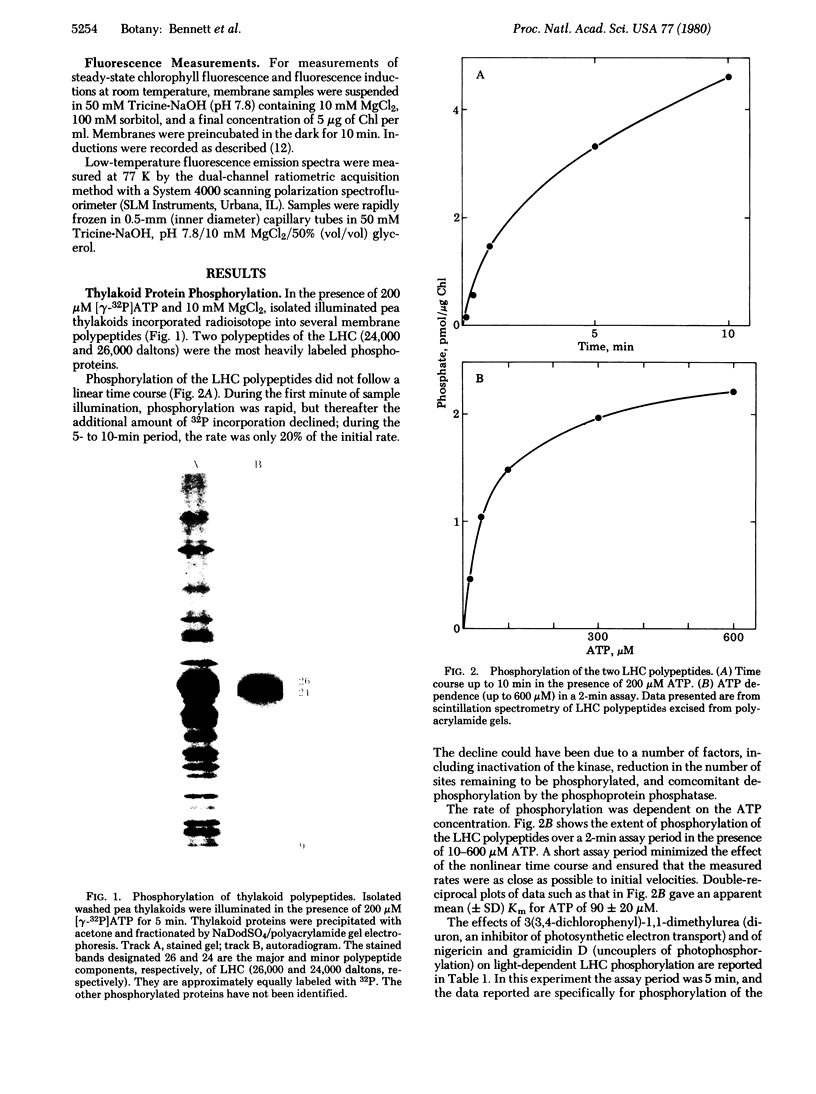
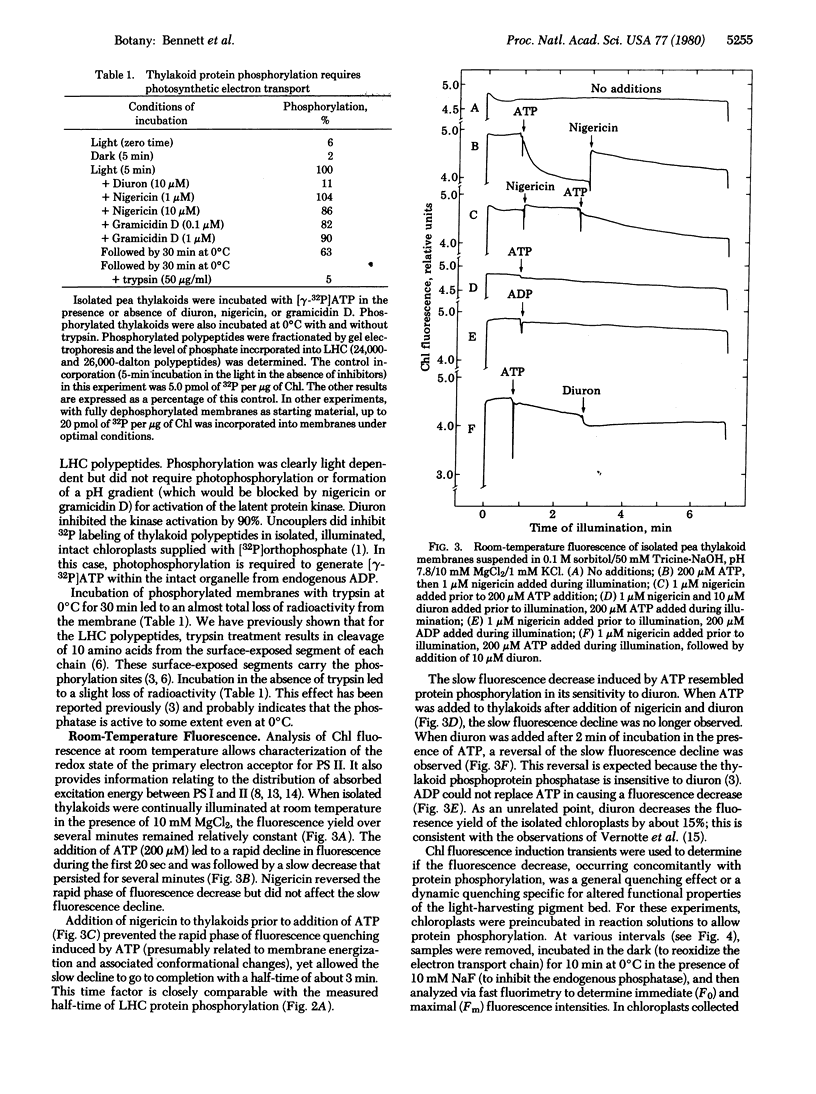
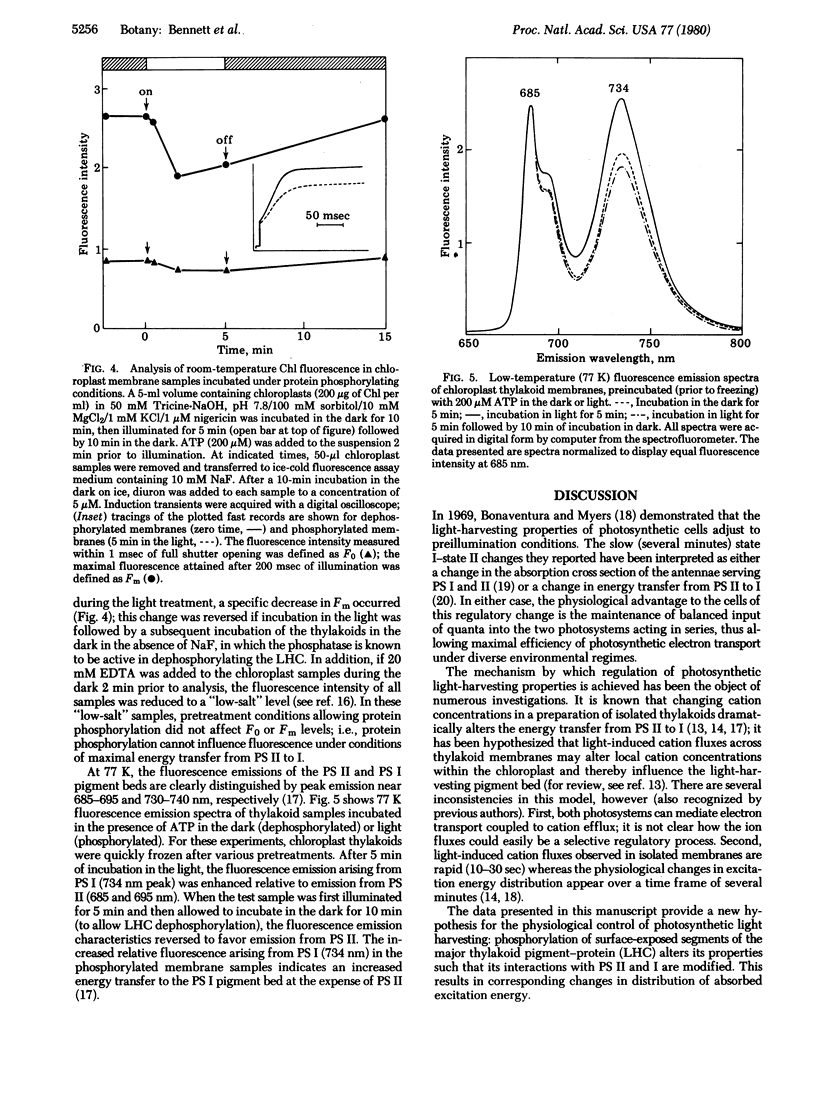
Incubation of isolated chloroplast thylakoid membranes with [gamma-32P]ATP results in phosphorylation of surface-exposed segments of several membrane proteins. The incorporation of 32P is light dependent, is blocked by 3(3,4-dichlorophenyl)-1,1-dimethylurea (diuron, an inhibitor of electron transport), but is insensitive to uncouplers of photophosphorylation. Polypeptides of the light-harvesting chlorophyll a/b-protein complex are the major phosphorylated membrane proteins. Addition of ATP to isolated chloroplast thylakoid membranes at 20 degrees C results in a time-dependent reduction of chlorophyll fluorescence emission; this is blocked by diuron but not by nigericin. ADP could not substitute for ATP. Chlorophyll fluorescence induction transients showed a decrease in the variable component after incubation of the membranes with ATP. Chlorophyll fluorescence at 77 K of phosphorylated thylakoid membranes showed an increase in long-wavelength emission compared with dephosphorylated controls. We conclude that a membrane-bound protein kinase can phosphorylate surface-exposed segments of the light-harvesting pigment-protein complex, altering the properties of its interaction with the two photosystems such that the distribution of absorbed excitation energy increasingly favors photosystem I.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bennett J. Chloroplast phosphoproteins. Evidence for a thylakoid-bound phosphoprotein phosphatase. Eur J Biochem. 1980 Feb;104(1):85–89. doi: 10.1111/j.1432-1033.1980.tb04403.x. [DOI] [PubMed] [Google Scholar]
- Bennett J. Chloroplast phosphoproteins. Phosphorylation of polypeptides of the light-harvesting chlorophyll protein complex. Eur J Biochem. 1979 Aug 15;99(1):133–137. doi: 10.1111/j.1432-1033.1979.tb13239.x. [DOI] [PubMed] [Google Scholar]
- Bennett J. Chloroplast phosphoproteins. The protein kinase of thylakoid membranes is light-dependent. FEBS Lett. 1979 Jul 15;103(2):342–344. doi: 10.1016/0014-5793(79)81358-7. [DOI] [PubMed] [Google Scholar]
- Bonaventura C., Myers J. Fluorescence and oxygen evolution from Chlorella pyrenoidosa. Biochim Biophys Acta. 1969;189(3):366–383. doi: 10.1016/0005-2728(69)90168-6. [DOI] [PubMed] [Google Scholar]
- Burke J. J., Ditto C. L., Arntzen C. J. Involvement of the light-harvesting complex in cation regulation of excitation energy distribution in chloroplasts. Arch Biochem Biophys. 1978 Apr 15;187(1):252–263. doi: 10.1016/0003-9861(78)90031-0. [DOI] [PubMed] [Google Scholar]
- Béliveau R., Bellemare G. Light-dependent phosphorylation of thylakoid membrane polypeptides. Biochem Biophys Res Commun. 1979 Jun 13;88(3):797–803. doi: 10.1016/0006-291x(79)91478-5. [DOI] [PubMed] [Google Scholar]
- Lieberman J. R., Bose S., Arntzen C. J. Requirement of the light-harvesting pigment.protein complex for magnesium ion regulation of excitation energy distribution in chloroplasts. Biochim Biophys Acta. 1978 Jun 8;502(3):417–429. doi: 10.1016/0005-2728(78)90075-0. [DOI] [PubMed] [Google Scholar]
- Murata N. Control of excitation transfer in photosynthesis. II. Magnesium ion-dependent distribution of excitation energy between two pigment systems in spinach chloroplasts. Biochim Biophys Acta. 1969 Oct 21;189(2):171–181. doi: 10.1016/0005-2728(69)90045-0. [DOI] [PubMed] [Google Scholar]
- Steinback K. E., Burke J. J., Arntzen C. J. Evidence for the role of surface-exposed segments of the light-harvesting complex in cation-mediated control of chloroplast structure and function. Arch Biochem Biophys. 1979 Jul;195(2):546–557. doi: 10.1016/0003-9861(79)90381-3. [DOI] [PubMed] [Google Scholar]
- Vernotte C., Etienne A. L., Briantais J. M. Quenching of the system II chlorophyll fluorescence by the plastoquinone pool. Biochim Biophys Acta. 1979 Mar 15;545(3):519–527. doi: 10.1016/0005-2728(79)90160-9. [DOI] [PubMed] [Google Scholar]
- Wang R. T., Myers J. On the state 1-state 2 phenomenon in photosynthesis. Biochim Biophys Acta. 1974 Apr 23;347(1):134–140. doi: 10.1016/0005-2728(74)90206-0. [DOI] [PubMed] [Google Scholar]