From 28–30 October 2011, more than 100 people assembled in Alexandria, Virginia, to participate in an American Diabetes Association–sponsored research symposium entitled “Diabetes and the Brain.” The objective of the symposium was to discuss the role of the brain in normal and abnormal metabolism and to consider the impact of diabetes on cerebral structure and function. Symposium participants were particularly interested in understanding how abnormalities in brain metabolism could affect the development of diabetes and obesity and how these diseases could, in turn, affect learning and memory. The growing epidemic of diabetes brought urgency to the meeting because of the ever-increasing number of people placed at risk for the cerebral complications of the disease. In this report, we present meeting highlights in five related areas as follows: 1) metabolism, blood flow, and epigenetics; 2) glucose sensing and hypoglycemia counterregulation; 3) insulin resistance and action in the brain; 4) neurocognition and imaging; and 5) energy homeostasis.
METABOLISM, BLOOD FLOW, AND EPIGENETICS
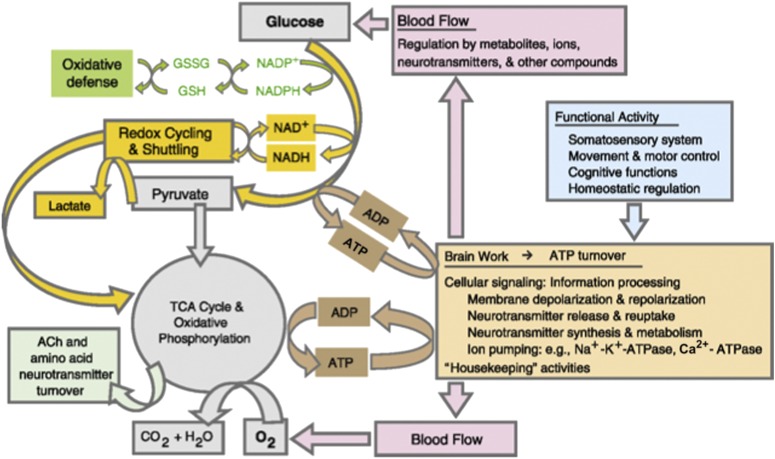
The symposium opened with an outstanding review of cerebral metabolism by Gerald Dienel, PhD, from the University of Arkansas. He emphasized that glucose is the major fuel for the adult brain and that it has multifunctional roles in brain function. Metabolism of glucose via the glycolytic, pentose phosphate shunt, and tricarboxylic acid cycle pathways provides ATP for energy, NADPH for defense against oxidative stress, and carbon for biosynthesis of amino acids and sugars used for synthesis of glycoproteins and glycolipids and for de novo synthesis of tricarboxylic acid cycle-derived neurotransmitters glutamate and γ-aminobutyric acid (GABA) (Fig. 1). Glycogen is stored predominately in astrocytes, and the CO2 fixation reaction required for glutamate biosynthesis occurs in astrocytes. Local rates of blood flow and glucose utilization are closely linked to local activities of brain cells, and metabolic rates increase with increased functional activity and decline when cellular signaling is reduced. Energy metabolism and neurotransmission are closely interrelated because glutamate, GABA, acetylcholine, and glycine are synthesized via glucose metabolic pathways, and catecholamine synthesis and degradation rely on oxygen. Metabolic brain imaging and magnetic resonance spectroscopic studies take advantage of the obligatory use of glucose as the predominant brain fuel, and labeled glucose or labeled glucose analogs are used to assay rates of specific reactions or pathways in living brain. Different labeled precursors are used to measure major pathway fluxes in neurons (glucose) and astrocytes (acetate), and they are used for trafficking of metabolites between these two major cell types. In experimental diabetes, changes in the metabolic rate of glucose in the brain are small, but increased brain glucose levels increase production of sorbitol, elevate oxidative stress, impair gap junctional communication among astrocytes, and cause other abnormalities. Although cognitive capability and other brain functions are affected by diabetes, its impact on the brain does not appear to be as severe as it is on peripheral organs.
FIG. 1.
Relationships among brain function, ATP use, glucose and oxygen metabolism, and blood flow. ACh, acetylcholine; GSH, glutathione; GSSG, oxidized glutathione; TCA, tricarboxylic acid.
Eric Newman, PhD, from the University of Minnesota, discussed the regulation of cerebral blood flow in the retina and used it as a model for regulation of brain blood flow. He emphasized that neuronal activity in the brain leads to localized increases in blood flow, a process termed functional hyperemia. Recent work suggests that activity-dependent increases in blood flow are mediated largely by a feedforward mechanism, whereby transmitter release from active neurons results in release of vasodilatory agents, such as arachidonic acid and its metabolites, either from activated glial cells or from other neurons. He went on to say that the regulation of central nervous system (CNS) blood flow is disrupted in diabetic patients. In diabetes, these changes have been best-characterized in retinas in which decreases in light-evoked vasodilation are seen, even before overt retinopathy is observed. Loss of the functional hyperemia response may result in hypoxia and may contribute to development of diabetic retinopathy.
Dr. Newman presented results from a recently completed study in his laboratory, where he found that light-evoked vasodilation was substantially reduced in rats with diabetes for a duration of 7 months. Loss of vasodilation reproduced the disruption of functional hyperemia observed in patients with diabetes. Inducible nitric oxide synthase (iNOS) was upregulated in these animals. He suggested that this response could be responsible for the reduction of functional hyperemia in diabetes, particularly because inhibiting iNOS with aminoguanidine reversed its loss. Dr. Newman concluded that targeting iNOS or its downstream signaling pathway with selective inhibitors to restore functional hyperemia ultimately might be an effective therapy for treating diabetic retinopathy.
Giulio Maria Pasinetti, MD, PhD, from Mount Sinai School of Medicine, focused on epigenetics (DNA methylation) in mouse models of diet-induced obesity. In the Tg2576 mouse, a high-fat diet for up to 5 months resulted in increased soluble β-amyloid in hippocampus and cortex (key CNS sites for learning and cognition) and impaired spatial memory behavioral performance. Using a model of the metabolic syndrome (increased body weight; glucose intolerance; and elevated blood pressure, plasma insulin, and cholesterol) developed in the wild-type C57BL/6J mouse, his group observed impairment in hippocampal electrophysiological measures that may be linked to impaired memory consolidation. Dr. Pasinetti summarized progress in identification of genes that are targets for DNA epigenetic modification and altered expression in the CNS of the mouse model of metabolic syndrome. These include genes for mitochondrial energy metabolism that are downregulated by diet-induced obesity secondary to hypermethylation of specific promoter sequences. A caloric restriction mimetic, Combi-phenol, protects against or normalizes several sequelae of the metabolic syndrome in this model, including brain mitochondrial dysfunction, cognitive behavioral performance, and CNS network connectivity evaluated with diffusion tensor imaging. The Combi-phenol is being studied in phase I and II clinical trials for safety, measurement of biomarkers, and memory assessments as a potential therapeutic or therapeutic adjunct for Alzheimer disease (AD).
GLUCOSE SENSING AND HYPOGLYCEMIA COUNTERREGULATION
Brain glucose-sensing mechanisms in the brain were the topic of the presentation by Vanessa Routh, PhD, from the New Jersey Medical School. She explained that the ventromedial hypothalamus (VMH) contains both glucose-excited and glucose-inhibited neurons that respond to physiologically relevant changes in glucose. Glucose-excited neurons increase firing as glucose is increased through a mechanism that involves closure of the KATP channel. The phenotype of these neurons remains uncertain, but some believe they may be producing pro-opiomelanocortin (POMC). Glucose-inhibited neurons reduce firing in response to an increase in glucose. This action appears to be mediated by AMP kinase and neuronal NOS and couples to a chloride channel. Glucose-inhibited neurons are distributed throughout the VMH, including approximately half of the neuropeptide Y (NPY)–producing neuronal population. The ability of the glucose-inhibited neurons to sense and respond to glucose parallels the ability of the brain to sense and respond to fasting and hypoglycemia in normal rodents. Moreover, the response of glucose-inhibited and glucose-excited neurons to decreased glucose can be blunted by satiety signals such as leptin or insulin and can be increased by the orexigenic peptide ghrelin. On the basis of these results, Dr. Routh proposed the hypothesis that glucose-sensing neurons are important for sensing decreased glucose and initiating compensatory responses under conditions of energy deficit, such as fasting or hypoglycemia. However, during normal energy balance, it is important that glucose-sensing neurons do not respond to small glucose decreases associated with meal-to-meal fluctuations in glucose because such a response could lead to inappropriate signals of overall energy deficit. As a result, satiety hormones mask and orexigenic agents enhance the responsiveness of glucose-sensing neurons to reductions in glucose.
Stephanie Amiel, MD, from King’s College London School of Medicine, presented a lecture entitled “Neural Circuits Involved in Hypoglycemia Counterregulation.” She reviewed the physiology underlying the counterregulatory response to hypoglycemia and then presented neuroimaging studies in humans that have helped visualize the global brain response to hypoglycemia. Many cerebral regions have been shown to participate in the detection of hypoglycemia and coordination of the counterregulatory response, including the hypothalamus, the thalamus, the hippocampus, and higher brain centers. With a new understanding of the parts of the brain involved in the counterregulatory response to hypoglycemia, she proposed that new therapeutic avenues to prevent and treat hypoglycemia may be possible.
How these different brain regions may detect and respond to hypoglycemia was discussed by E.R.S. MD, from the University of Minnesota, in her lecture entitled “Mechanisms Underlying the Development of Hypoglycemia-Associated Autonomic Failure.” She began her presentation by emphasizing that hypoglycemia-associated autonomic failure (HAAF) is a clinically significant problem that occurs in patients with type 1 and advanced type 2 diabetes. Recurrent hypoglycemia leads to HAAF because it shifts the glucose threshold that elicits adrenomedullary response (i.e., lower and lower blood sugars required to elicit response) and reduces the magnitude of hormonal counterregulatory response while having no effect on glucose level associated with neuroglycopenia symptoms. HAAF is reversible by avoiding hypoglycemia.
The mechanisms responsible for the development of HAAF remain uncertain, but at least three have been proposed. One potential mechanism is that hypoglycemia induces a change in brain glucose metabolism in such a way to prevent regulatory centers from sensing a change in blood glucose concentration during systemic hypoglycemia. A second potential mechanism is that the brain may increase the utilization of alternative fuels such as lactate and glycogen during hypoglycemia and therefore may not experience energy deficit in the setting of systemic hypoglycemia. A third mechanism suggests that changes in downstream signaling of GABA and opioids that link detection of hypoglycemia to counterregulatory response are altered in the setting of recurrent hypoglycemia.
INSULIN RESISTANCE AND INSULIN ACTION IN THE BRAIN
C. Ronald Kahn, MD, from the Joslin Clinic and the Harvard Medical School, discussed diabetes, insulin action, and the brain. One approach to identifying pathways through which insulin alters brain metabolism has been creation of mice in which the insulin receptor is knocked out only in neural tissue. These mice show increased food intake, increased adipose mass, hyperleptinemia, and insulin resistance; however, behaviorally, they are the same as wild-type mice at a young age. However, as they age, they begin to behave abnormally in open field tests (interpreted as a sign of anxiety), in tail suspension tests (interpreted as a sign of depression), and in forced swimming tests (interpreted as a loss of motivation). How the loss of insulin action causes these behavior changes remains unknown, but preliminary studies show that these mice have decreased phosphorylation of glycogen synthase kinase 3 and increased phosphorylation of τ, two proteins known to be important in the pathogenesis of AD. Dr. Kahn’s group recently performed a gene expression analysis on the hypothalamus from streptozotocin-treated C57BL/6 mice (a model for insulin-deficient diabetes) and found that genes involved in cholesterol biosynthesis are suppressed relative to controls. In addition, rates of cholesterol synthesis and the synaptosomal cholesterol content are decreased in the brains of these diabetic mice, as are levels of sterol regulatory element–binding protein 2 and srebp cleavage-activating protein (SCAP) mRNA and protein, two important regulators of cholesterol synthesis.
Mark W.J. Strachan, MD, from the Western General Hospital, Edinburgh, Scotland, also discussed insulin action in the brain in his lecture entitled “Emerging Data on the Relationship Between Insulin Resistance and Neurovascular Disorders.” Insulin receptors are found in high concentrations within the limbic system of the brain. In vitro, insulin affects neuronal excitability and synaptic plasticity. Direct relationships have been shown between insulin action and the pathogenesis of AD. The β-amyloid oligomers, which accumulate in the brain in AD, are intimately linked with neuronal dysfunction and cause a rapid and substantial loss of cell surface insulin receptors in hippocampal neuronal cultures. The development of insulin resistance in the brain then can set-up a vicious cycle because resultant activation of glucose synthase kinase 3 promotes hyperphosphorylation of τ protein and altered processing of amyloid precursor protein, which increases β-amyloid oligomer production. Moreover, insulin resistance (through attendant hyperinsulinemia) may reduce the ability of insulin-degrading enzyme in the brain to break-down β-amyloid oligomers. Proving the existence of these mechanisms and pathways in humans has been challenging, but insulin concentrations in the cerebrospinal fluid do appear lower in people with severe AD. In addition, administration of intravenous insulin (with concurrent glucose to maintain euglycemia) has been shown to enhance cognitive function. Direct administration of insulin via the intranasal route has a favorable impact on cognition and may provide new opportunities for treatment. Therapies designed to improve cerebral insulin action have produced mixed results. Administration of rosiglitazone was not of cognitive benefit to people with AD, perhaps because it does not cross the blood--brain barrier, whereas an exercise intervention did lead to improvement in insulin sensitivity and cognitive function in older adults.
Suzanne Craft, PhD, from the University of Washington, presented a programmatic analysis of a specific mechanism that may link type 2 diabetes with pathological brain changes, cognitive impairment, and even AD-related dementia. A key target (if not culprit) is insulin secretion and action. Dysregulation or other abnormalities in brain insulin play a role in normal aging-related cognitive decline, exacerbated cognitive decline with type 2 diabetes, and (when severe) mild cognitive impairment and AD. The two forms of insulin dysregulation that increase the risk of neurocognitive dysfunction, insulin resistance and peripheral hyperinsulinemia, also were discussed by other speakers in this symposium. Dr. Craft detailed several mechanisms through which insulin resistance and diabetes may increase the risk of cognitive impairment and AD. In addition to reduced brain insulin uptake, these specific mechanisms include: 1) impaired glucose and lipid metabolism; 2) disrupted regulation of β-amyloid clearance (i.e., a hallmark of AD neuropathology); 3) increased inflammation; and 4) compromised vascular function (e.g., microvascular lesions). Notably, insulin receptors are distributed in several critical brain regions (e.g., hippocampus, frontal lobe), and insulin performs several crucial brain cognition functions (e.g., increasing neurotransmitter levels, glucose utilization). The fact that numerous specific mechanisms can independently and perhaps interactively interrupt signaling in any of these regions creates a formidable challenge for clinical research.
If insulin resistance is a mechanism through which diabetes may compromise neurocognitive functioning, then there may be preventable or modifiable factors that lead to insulin dysregulation as well as therapeutics that may improve insulin resistance. Regarding the former, Dr. Craft reported that epidemiological studies suggest that the expanding prevalence of conditions related to insulin resistance in western societies may be attributable to decreased levels of physical activity and increased dietary intake of calories and saturated fats. Arguably, healthier lifestyles may be protective factors operating in several specific mechanisms through which insulin resistance can lead to neurocognitive dysfunction. Regarding the latter, Dr. Craft described her own research on the therapeutic strategy of administering intranasal insulin in an effort to improve neural insulin signaling. In her project known as the Study of Nasal Insulin to Fight Forgetfulness (SNIFF), she reported promising outcomes, including improved memory performance and improved performance in activities of daily living.
NEUROCOGNITION AND IMAGING
Christopher M. Ryan, PhD, from the University of Pittsburgh, presented a historically informative tour of the major issues and strategies for assessing cognitive function in diabetic patients. Although the field has benefited from a surge of interest in recent years, it has come to recognize that improvements can be implemented across research laboratories in crucial aspects such as: 1) establishing diagnostic standards and procedures for evaluating equivalences and systematic variations in clinical groups; 2) reducing inconsistencies in the cognitive testing batteries deployed (e.g., standardization, theoretical breadth, measurement purity, manifest vs. latent variable representations, and validity issues); 3) improving research sensitivity to special population characteristics that affect diabetes--cognition relationships (e.g., children, older adults, multiple impairments); and 4) addressing the importance of longitudinal follow-up for understanding the trajectories of cognitive change. Dr. Ryan, who was an early contributor and still is a continuing contributor to the field, focused on identifying a framework for representing some of the major approaches to selecting cognitive measures for research in diabetes and reviewing a large number of frequently used cognitive batteries.
Regarding the framework, Dr. Ryan described several related approaches. One approach emphasizes identifying relatively nonspecific cognitive markers of diabetes-related dysfunction. Neurocognitive speed has proven to be the most sensitive marker across multiple studies, even while using tasks that range widely in terms of their process purity. In a complementary approach, researchers use cognitive and neuropsychological test batteries to identify neurocognitive or clinical phenotypes (e.g., mild cognitive impairment) associated with diabetes. Results using this approach have improved as the batteries become more contemporary, comprehensive, and multidimensional. However, Dr. Ryan cautioned that it is crucial to map the expected cognitive effects to the brain changes (e.g., vascular) on the basis of theoretical and clinical data and to use batteries that are age-appropriate and change-sensitive to evaluate these relationships and performances at different phases of the life span.
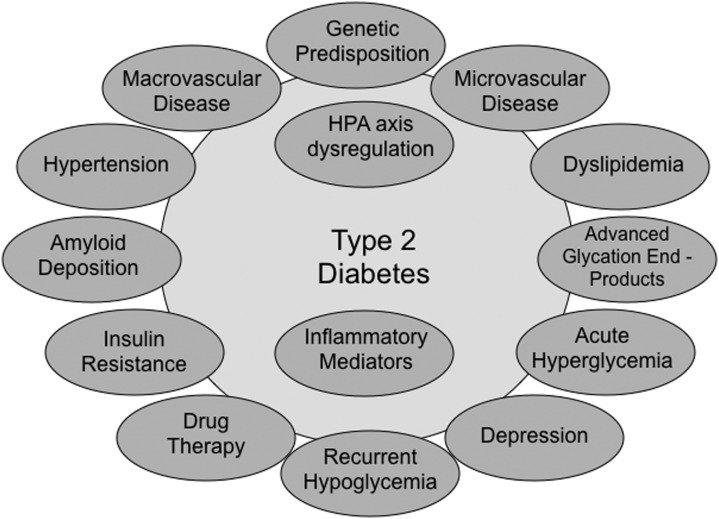
Brian M. Frier, MD, from the Royal Infirmary of Edinburgh, Scotland, shifted the focus from extant approaches and available test batteries to the correlative topic of observed cognitive effects of diabetes and their potential mechanisms. He began by noting that the “unsystematic” nature of early research in the area was likely attributable to cross-study variations and within-study limitations in sample sizes, cognitive batteries, and measurement of relevant covariates. Accordingly, Dr. Frier supplemented the typical organizing scheme of diabetes type. Rather than summarizing similar and dissimilar cognitive effects of type 1 and type 2 diabetes, he began by presenting the main observed (or to-be-tested) risk factors for cognitive decrements as associated with each. Both sets of risk factors include a genetic predisposition (although this remains an understudied aspect for both). For type 1 diabetes, the other risk factors include recurrent hypoglycemia, chronic hyperglycemia, long duration, and early age of onset, as well as a group of diabetes complications. Not surprisingly, for type 2 diabetes the list is much longer and more diverse (Fig. 2). It includes vascular issues (microvascular and macrovascular disease, hypertension), acute hyperglycemia, recurrent hypoglycemia, hyperinsulinemia, dyslipidemia, amyloid deposition in the brain, and inflammatory mediators, as well as drug therapy issues and psychosocial factors such as depression. The implicit message is that diabetes per se may affect neurocognitive performance in a variety of ways but does so not just through the separate prisms of diabetes type. It is also influenced within type by a diverse set of barely overlapping but likely interacting influences.
FIG. 2.
Potential risk factors for cognitive impairment in type 2 diabetes. HPA, hypothalamic-pituitary-adrenal.
For both disease types, there is consistent evidence that neurocognitive speed is affected, particularly straightforward indicators of mean performance rates that usually show slower average speeds for diabetic patients relative to controls. In addition, some downstream indicators of basic cognitive resources (e.g., executive function) and products (e.g., episodic memory) that may require speed for efficient performance are often implicated in research with both types of diabetes. According to Dr. Frier’s review, several of the promising risk factors for cognitive decrements in type 1 diabetes have not yet been confirmed empirically. One confirmed factor among children and adolescents is early disease onset (up to age 7), a factor that is also linked to progress and participation in schooling and social development. Regarding research on cognitive effects of type 2 diabetes, a key challenge is to differentiate neurocognitive changes that characterize normal aging from those exacerbated by diabetes or associated with progressive neurodegenerative decline. Researchers are increasingly considering the roles of modulating factors. In addition to age, sex, and education, these include the specific risk factors in Fig. 2, numerous comorbidities (e.g., obesity, vascular dysfunction), treatment and adherence (e.g., diet, oral agents), and exposure to risk-enhancing substances (alcohol, smoking, cholesterol). In summary, Dr. Frier’s recommendations for future research included examining multiple influences on longer-term progressive or accelerated declines in cognitive functioning. We await further results from prospective studies that feature larger sample sizes, broader coverage of key cognitive functions, and markers of important risk and mediator factors, all measured across extended bands of adulthood.
The presentation by Geert Jan Biessels, MD, PhD, from the University Medical Centre Utrecht, addressed etiology, risk factors, and biomarkers of diabetes-related cognitive dysfunction with an emphasis on novel opportunities provided by longitudinal research methods. A first important point is that cognitive dysfunction in diabetes should be characterized not just in terms of implicated aspects of cognition (e.g., episodic memory or neurocognitive speed) or even magnitude of the concurrent deficit Rather because most observed decrements are modest in magnitude, dysfunction also should be defined by characteristics of change, including direction, slope, trajectory, and outcome. Whereas for some diabetic patients neurocognitive deficits may decline only slowly over long periods of adulthood, others may experience trajectories that lead beyond normal decline to frank cognitive impairment and even dementia. Dr. Biessels reported that although diabetic patients who exhibit sustained modest decrement profiles of both types of diabetes (1 and 2) and of any age, those who experience more rapid neurocognitive decline are more likely to be patients with type 2 diabetes who eventually have development of overt dementia.
Dr. Biessels’ second major point was that the two types of cognitive dysfunction in diabetes may differ not only in terms of temporal and outcome characteristics but also in terms of associated biomarkers, risk factors, and etiological influences. In addition, Dr. Biessels noted that these dynamic and interactive processes can be directly examined with multiple assessments and appropriate statistical analyses. He described one promising longitudinal study that may yield data to answer many of the questions raised in his presentation. It features abundant risk factor data on large samples of type 2 diabetic patients from the Kaiser Permanente Northern California Diabetes Registry. Finally, a summary point from his biomarker and etiology discussion merits special attention. Dr. Biessels noted an emerging theory that may imply that the two types of diabetes--cognition trajectories may differ in part in the extent to which an additional (diabetes and immediate complications) “hit” from substantial brain pathology is absorbed by some patients. For example, a mild stroke or increased rates of amyloid deposition may accelerate the transition of some type 2 diabetic patients from normal cognition to impairment or dementia.
Gail Musen, PhD, from the Joslin Diabetes Center, highlighted the value of using neuroimaging techniques to shed light on neural correlates of cognitive dysfunction in diabetes. A principal rationale is that diabetes-related changes in the brain may be detectable with recent technology early in diabetes-related degenerative processes and that they may also precede and be differentially associated with specific diabetes-related cognitive manifestations and decremental patterns. Dr. Musen commented on the recent progress and future promise in this area, attributable in part to new and more widely available neuroimaging techniques. For example, early diabetes--brain research on white matter hyperintensities (often observed with magnetic resonance imaging) revealed mixed patterns across studies. Using the more sensitive, flexible, and objective diffusion tensor imaging, researchers can examine and compare a number of white matter abnormalities and connectivity patterns. These lines of investigation were not previously available because of limitations in imaging technology. In addition, voxel-based morphometry studies of cortical thickness (including both gray and white matter density) are being used to compare diabetic patients and healthy controls. Dr. Musen reported new research from her team on one promising avenue that may eventually link diabetes more closely with cognitive impairment and dementia risk. In normal adults, the default mode network is typically deactivated during goal-directed cognitive activity, with variations on activation and metabolism patterns for AD patients. She described one study in which similar disruptions occurred for type 1 diabetic patients and an effort to investigate similar issues with type 2 diabetes (a well-known risk factor for AD). Dr. Musen concluded her presentation with several forward-looking recommendations, including identification of neural mechanisms associated with potential cognitive dysfunctions in diabetes, pursuing this goal with larger-scale studies (in terms of both sample sizes and assessment waves), and collection of repeated assessments of target (neuroimaging, cognitive performance) and covariate (diabetes-related biomarkers) measures.
ENERGY HOMEOSTASIS
D.F.L., PhD, from the University of Washington, presented translational research demonstrating the effects of insulin and leptin on reward behavior and dopamine function in the rat. Her work has focused predominately on the rewarding value of sucrose, based on findings that sucrose drives motivation for intake of mixtures of sugar, fat, and flavor. She reviewed the CNS anatomy that mediates reward behavior and energy homeostasis and made three key points. First, these sets of circuitries are substantially interconnected. Second, specific sites within both sets of circuitries play a role in mediating different aspects of reward behavior. Third, insulin and leptin can decrease reward behavior through actions on both reward and energy homeostatic circuitries. The neurotransmitter dopamine plays a central role in reward and motivation. D.F.L. reviewed the effect of insulin to increase dopamine reuptake, which curtails dopamine signaling. There is evidence that this effect is attributable to increased cell-surface cycling of the dopamine reuptake transporter, and that this cycling is dependent on activation of phosphatidylinositol 3 kinase mechanisms. Additionally, it has been shown that leptin can decrease dopamine neuronal activity. Together, both hormones act to dampen dopamine signaling in normal animals. D.F.L. finished by reporting that exposure to a moderate-fat diet results in increased motivation for sucrose and resistance to insulin and leptin effects in rats that are metabolically normal and preobese. She concluded with examples from current human imaging studies suggesting that the effects of insulin and insulin resistance on reward circuitry in rodents are relevant for humans.
Tony Lam, PhD, from the University of Michigan, reviewed work from his laboratory in a lecture entitled “Gut--Brain Signaling.” He has shown that the influx of fatty acids into the duodenum leads to the release of cholecystokinin, subsequent activation of vagal afferents, and transmission of the signal to the nucleus of the solitary tract. As a result, the gut-brain-liver axis is activated to reduce hepatic glucose production. In addition, metabolism of lipids and glucose in the hypothalamus activates protein kinase C and KATP channels that regulate hepatic glucose production. Diabetes and obesity alter these nutrient-sensing pathways in both the gut and the brain, which leads to abnormalities in glucose homeostasis.
Joel Elmquist, DVM, PhD, from the University of Texas Southwestern Medical Center, reviewed the anatomical, synaptic, and cellular functions of the mediobasal hypothalamic circuitry in the coordinated regulation of multiple components of energy homeostasis. This circuitry has now been characterized for reciprocal regulatory interactions in locus, as well as for receiving and transmitting multiple signals from other CNS sites and from the periphery. Research has focused on the roles of (orexigenic) NPY/agouti-related protein (AGRP) and (anorexigenic) POMC neurons. Dr. Elmquist provided a historical perspective on the field that laid the groundwork for the utility of genetically modified mouse models to determine specific neuronal and functional aspects of leptin activity. Specific ablation of leptin receptors in POMC neurons, which produce the neuropeptide melanocortin, results in modest obesity with decreased energy expenditure and locomotor activity but no effect on food intake, along with mild glucose intolerance and insulin resistance. Specific reinstatement of functional leptin receptors in these POMC neurons normalizes peripheral glucose homeostasis and glucagon release. Interestingly, a model in which both insulin and leptin receptors were ablated in POMC neurons did not result in strong effects on body weight but showed profound glucose dyshomeostasis and reduced fertility. This suggested coordinated action of insulin and leptin signaling in POMC neurons that regulate reproductive and hepatic function. However, leptin receptor effects are distributed throughout a network; for example, the leptin receptors expressed on steroidogenic factor-1 neurons of the VMH appear to regulate energy expenditure. The melanocortin receptor subtype 4 (MCR4) mediates many of the energy homeostatic effects of melanocortin, and new evidence suggests that MCR4 activity in multiple central and peripheral nervous system sites is important for regulation of energy homeostasis. Thus, MCR4 receptors expressed in SIM1 neurons of the paraventricular nucleus of the hypothalamus and the amygdala correct hyperphagia in MCR4-null mice. MCR4 receptors expressed in cholinergic neurons of the sympathetic nervous system increase energy expenditure and regulate hepatic glucose production, and MCR4 receptors expressed in the cholinergic neurons of the parasympathetic nervous system may regulate endocrine pancreatic function.
Tamas Horvath, DVM, PhD, from Yale University, presented his work examining synaptic interactions directly between POMC and other local neurons. The AGRP/NPY neuron releases the inhibitory neurotransmitter GABA. In addition to a projection to the paraventricular nucleus of the hypothalamus, the AGRP/NPY neuron sends a GABAergic projection to local POMC neurons in the arcuate nucleus. This local circuit is a direct target for leptin action as well as for the orexigenic hormone/neuropeptide, ghrelin. Recent work from Horvath’s group has demonstrated ghrelin stimulation of UCP2 expression. Studies in a UCP2 knockout model have shown an impairment of ghrelin-stimulated electrical activity and mitochondriogenesis in NPY/AGRP neurons. Ghrelin effects have been observed in other CNS sites, i.e., the hippocampus and the dopaminergic neuron in the substantia nigra pars compacta. Taken together, the group’s recent findings clearly delineate a key role of the UCP2 protein in many aspects of CNS function and its modulation by nutritional and metabolic status.
The theme of hypothalamic/brain/periphery networking was further emphasized in the presentation by Joseph Bass, MD, PhD, from Northwestern University, that focused on circadian regulation of energy homeostasis. Dr. Bass reviewed the discovery of clock proteins in the 1990s, which set the stage for research on circadian function and its regulation in the subsequent years. In mammals, including humans, the CNS “master clock” has been identified as the suprachiasmatic nucleus (SCN), and circadian rhythms are encoded by the sequential and reciprocal expression of transcription activators, CLOCK/BMAL1, that induce expression of the transcription repressors, PER1–3/CRY1–3, that inhibit CLOCK/BMAL1 expression. The cycle of expression takes approximately 24 h and is entrained by light. The SCN has connections with key homeostatic nuclei in the hypothalamus, including the arcuate, paraventricular nucleus, ventromedial hypothalamus (VMH), lateral hypothalamus, and dorsal medial hypothalamus (DMH). Thus, there is direct input on behavior, activity, metabolism, and the hypothalamic-pituitary-adrenal axis, which in turn is a key diurnal regulator of activity and metabolism. Importantly, it is now appreciated that “clock proteins” are expressed in nearly all cells. Peripheral tissue clocks are entrained to food availability, adrenal glucocorticoids, and temperature, all of which can reset peripheral clocks. Although research is only beginning to uncover the complex crosstalk between stimuli and peripheral clocks, molecules that reflect metabolic status such as NAD and the nutrient sensor AMP kinase, relay information to the clocks and connectivity is bidirectional. A current line of investigation is examining the coupling of metabolic processes and CNS clock function. A high-fat diet in an animal model has been shown to increase period length of the daily activity rhythm and to phase-shift metabolic gene expression in the liver and adipose as well as in the hypothalamus. The CNS locus of this effect (SCN per se, or SCN projections) remains to be determined but clearly demonstrates metabolite/CNS clock interaction. Finally, sleep and activity abnormalities such as narcolepsy and genetic evidence in humans with altered sleep patterns suggest that pursuit of this research and links between clock activities and metabolic disease will reveal the importance of clock/environmental coordination to human metabolic health.
CONCLUSIONS
The workshop brought together scientists and clinicians working in disparate areas such as metabolism, psychology, imaging, and neuroscience who were all united by a desire to understand how diabetes affects cerebral structure and function. In the opening lecture, Dr. Gerald Dienel reported anecdotally that early in his career it was unusual for basic neuroscientists to consider diabetes as an influence on cognitive functioning, much less a risk factor for neurodegenerative disease. By the end of the workshop, new collaborations were formed that will support future multidisciplinary research in this area. Ideally, this work should be based on the following principles: both “neuro” and “cognitive” phenomena must be clinically relevant, theoretically selected, domain-specific, and carefully measured; research should emphasize, whenever possible, continuous (rather than categorical) assessments of phenomena related to diagnosis, performance, structure, modulating factors, and clinical outcomes; investigation should attend to a broad range of potential modulators, including proximal (e.g., insulin resistance), somatic or distal (e.g., functional biomarkers), comorbidities (e.g., vascular disruptions, inflammation), therapy--social (e.g., treatment availability and adherence, depression), and genetic susceptibilities (e.g., genetic polymorphisms related to diabetes, AD, obesity); work should continue to examine the complex systems through which endocrine and neural systems regulate energy metabolism; and, whenever possible, scientists should study these relationships dynamically using longitudinal follow-up assessments and appropriate change-sensitive statistical analyses.
ACKNOWLEDGMENTS
No potential conflicts of interest relevant to this article were reported.
E.R.S., D.F.L., and R.A.D. contributed to the drafting and revising of the manuscript.
The authors gratefully acknowledge the organizational support of Shirley Ash (American Diabetes Association).