Abstract
Background
BRAF inhibitors such as vemurafenib are a new family of biological drugs, recently available to treat metastatic malignant melanoma.
Methods
We present the case of a 38-year-old man affected by metastatic melanoma who had been under treatment with vemurafenib for a few days. The patient suffered from sudden onset of abdominal pain due to intra-abdominal hemorrhage with profuse hemoperitoneum. An emergency abdominal sonography confirmed the clinical suspicion of a splenic rupture.
Results
The intraoperative finding was hemoperitoneum due to splenic two-step rupture and splenectomy was therefore performed. Histopathology confirmed splenic hematoma and capsule laceration, in the absence of metastasis.
Conclusions
This report describes the occurrence of a previously unreported adverse event in a patient with stage IV melanoma receiving vemurafenib.
Keywords: Spontaneous splenic rupture, Melanoma, BRAF, Vemurafenib
Background
Atraumatic splenic rupture (ASR) is a rare pathology that can be idiopathic (7%) or pathological (93%) in nature [1]. While in the first group there are no abnormal histopathological findings, in the second one several etiological factors can be identified, including neoplastic (30%), infectious (27%), inflammatory non-infectious (20%), drug- or treatment-related (9%), and mechanical disorders (6%) [1].
ASR is an often unrecognized and potentially fatal cause of acute abdomen development; it should be routinely considered in the differential diagnosis of such pathology and when present promptly managed, at best with laparotomic approach [2]. The commonest causes of this rare pathology are: malignant hematological disorders (16%) such as acute leukemia and non-Hodgkin’s lymphoma; viral infections (15%) as infectious mononucleosis and cytomegalovirus infection; and local inflammatory and neoplastic disorders (11%) such as acute and chronic pancreatitis. Splenomegaly is a common feature, affecting 55% of patients. There is a male gender prevalence of 2:1, with a mean age of 45 years, (median 45, range 18–86 years). The overall ASR-related mortality rate is 12% [1].
Although, nowadays, the laparoscopic approach is the routine procedure for various diseases requiring elective splenectomy, in emergency cases laparotomy still retains its effectiveness, first and foremost with regard to patients with unstable hemodynamic conditions.
Case presentation
In September 2011, a 38-year-old man was admitted in urgency regimen to our department because of a moderate left flank pain starting 6 h before hospital admission. The patient also presented vomiting and diffuse abdominal tenderness. Minor discomfort had been present in the left flank for a few days. The patient had a 3-year history of melanoma, starting in November 2008 with the removal of a forehead superficial spreading melanoma, 7 mm in diameter, Breslow thickness being 0.44 mm, with discrete lymphocytic infiltrate mainly in the surrounding tissue, without ulceration or regression. Only radicalization of local excision and follow-up were required according to the initial staging; sentinel node biopsy was therefore not performed.
In June 2011, 31 months after primary tumor excision, and 3 months before the episode of acute abdominal pain, the patient underwent an emergency orchiectomy because of the onset of acute scrotal swelling, with a pathologic diagnosis of metastatic melanoma to the testis (with an immunohistochemical profile positive for HMB-45, S-100, MART-1, and negative for pancytokeratin and inhibin). At that time, PET and CT scans showed several possible metastases, in soft tissues, gastro-intestinal tract, lungs, and brain; biopsies of masseter muscle, stomach, and duodenum confirmed the diagnosis.
The patient, after testing positive on screening for MO 255515 protein, was recruited to an experimental protocol using BRAF inhibitors and thus received a first cycle of therapy with vemurafenib at an oral dose of 960 mg twice daily. He did not undergo combined therapy with cytotoxic drugs, but received vemurafenib alone. Such experimental protocol was part of a compassionate care strategy and foresaw specific exclusion criteria, such as the presence of second malignancies, renal failure and diabetes mellitus, co-morbidities that our patient did not have. The acute abdominal event occurred 12 days after starting treatment, when the patient had assumed a total cumulative dose of about 23 g.
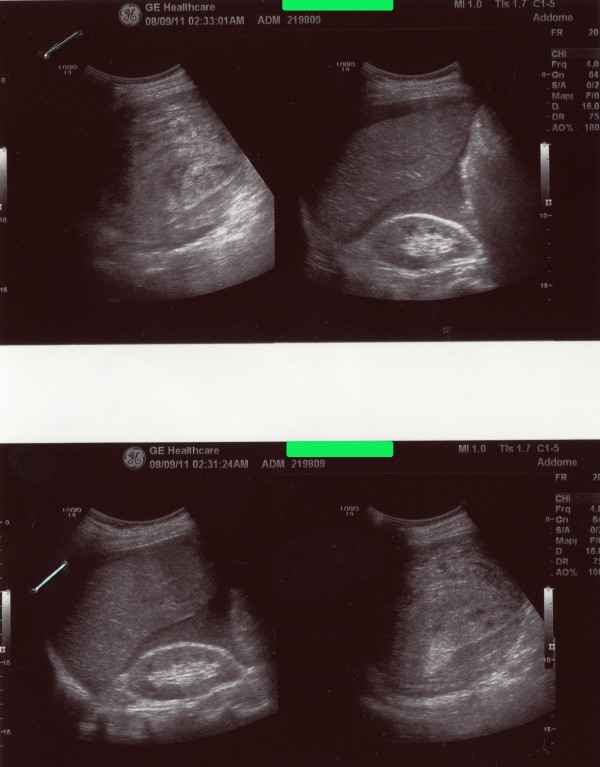
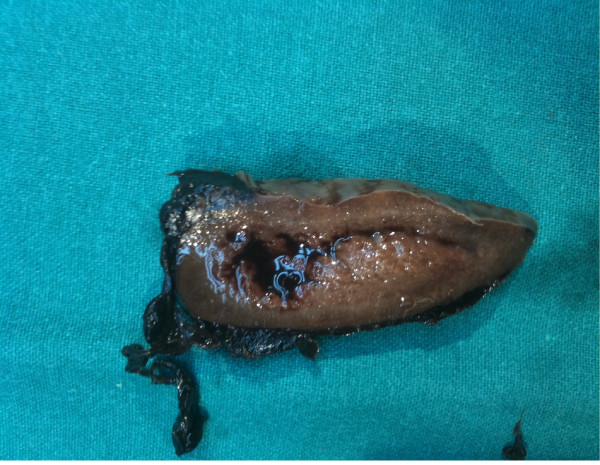
On admission the patient was pale and suffering, and his vital parameters were as follows: oxygen saturation 100%; heart rate 90 bpm, temperature 36.5°C, blood pressure 90/45 mmHg. His medical history was otherwise negative, and no trauma or accident had been previously reported. An abdominal examination showed diffuse pain and peritoneal fluid was noticed at percussion; a nasogastric tube was placed without detecting gastric retention and a urinary catheterization showed oliguria. A complete blood count showed moderate leukocytosis (13.67 WBC x103) and anemia (RBC 3.42 x106, Hb 10.4 g/dL, Ht 30.8%). An immediate abdominal sonography confirmed the clinical suspicion of spontaneous rupture of the spleen with massive hemoperitoneum (Figure 1). The patient immediately underwent an emergency laparotomy. Intraoperative findings have been massive hemoperitoneum (approximately 3 L of blood and coagula) due to a two-step rupture of the spleen, with subsequent subcapsular hematoma and complete decortication, that required a splenectomy; splenic size was within the normal range (Figure 2). Some suspected metastatic nodes were removed from the great omentum; histopathology confirmed the metastatic nature of the omental nodes while the spleen was decorticated in the absence of metastases.
Figure 1.
Abdominal ultrasonography showing the spleen rupture with hemoperitoneum.
Figure 2.
Section of spleen in which can be seen laceration of the capsule due to rupture in two times.
The postoperative course was complicated by the onset, on the fourth day, of partial thrombosis of the great saphenous vein; the diagnosis was confirmed by sonography and a therapy of 40 mg low molecular weight heparin (LMWH) (Clexane®, Aventis Pharma, Bad Soden, Germany; 0.4 mL prefilled syringes) subcutaneously administered twice daily was undertaken. The abdominal drainage was removed 5 days after the operation, and the patient was discharged on the following day.
Discussions
Melanoma is the deadliest form of skin cancer; its incidence has been increasing continuously and over 40,000 people die of this disease each year worldwide [3]. The estimated number of new cases of melanoma in 2010 in the United States was 68,130, of which 57% affected males and 43% females. In the same period, the estimated deaths caused by this pathology were 8,700 (65% males and 35% females) [4].
The disease presents genetically and clinically distinct subgroups that could indicate the need of patient-specific management strategies [5]. For example, head and neck mucosal malignant melanomas behave much more aggressively than limb or trunk tumors and their prognostic markers have not yet been fully elucidated [6]. The majority of metastases appears within 3 years from the diagnosis of primary melanoma [7]. Primary melanoma metastasizes most frequently to lymph nodes and lungs (about 70%). Less common sites of metastasis are liver (58%), brain (54%), bones (48%), adrenal glands (46%), and gastrointestinal tract (43%). Among this group, small intestine and spleen are affected in 36.5% and 30.6% of cases, respectively [7]. The 1-year survival rate for patients with systemic metastasis from cutaneous melanoma ranged from 40% to 60% in the American Joint Committee on Cancer (AJCC) Melanoma Staging Database [8]. Our patient had a stage IV melanoma, and, therefore, an extremely poor prognosis with median survival ranging from 6 to 18 months after diagnosis [9] and a 5-year survival rate of less than 5% [10-12].
Melanoma treatment has experienced a rapid change in recent years through recent discoveries of target therapies and immunotherapeutic antibodies. With the continuous progress of research, prolonged therapeutic success may be achieved through the tailored use of molecular markers and immunotherapies in sequential or combinatorial methods [13,14]. Advanced melanomas often have multiple genetic defects affecting several biochemical pathways. Somatic point mutations in RAF occur in approximately 8% of human tumors, most frequently in melanoma, colorectal, and thyroid cancers [4,14,15]. The targeting of this single oncogenic alteration with specific inhibitory nucleic acids or chemical RAF inhibitors in melanoma cell lines showed growth arrest and induction of apoptosis [16,17].
Although melanoma metastases have been found to contain thousands of mutations, the V600E BRAF mutation is clearly a driver of the neoplastic phenotype and is present in approximately 50% of melanomas [5,18]. The discovery of activating mutations (V600E) in the BRAF kinase encouraged the development of compounds to inhibit aberrant BRAF activity [5]. Vemurafenib (also known as PLX4032, RG7204, or RO5185426) marketed as Zelboraf is a BRAF enzyme inhibitor developed by Plexxikon (Berkeley, CA, USA, now part of the Daiichi Sankyo Group) and Roche [15]. PLX4032 was recently observed to increase median overall survival in metastatic melanoma. However, resistance through up-regulation of receptors or by activating mutations in oncogenic signaling and alternative enzymes is proving to be an emerging problem [3]. The comparison between vemurafenib and conventional treatment with dacarbazine led to encouraging results regarding 6-month survival, which was 84% in the vemurafenib group and 64% in the dacarbazine group. The risk of either death or disease progression was also reduced in the vemurafenib group [9]. In summary, these studies reveal that, for the first time, several immunotherapeutic and targeted agents are yielding some clinical responses and improvements in overall survival in patients with unresectable stage III and IV melanoma [3]. In patients with metastatic melanoma featuring BRAF V600E mutation, phase 1 and 2 clinical trials of the BRAF kinase inhibitor vemurafenib (PLX4032) have shown response rates of more than 50% [9].
Selective BRAF inhibitors show good tolerability with infrequent severe toxicities. Among the common adverse events associated with vemurafenib are skin changes 50% to 70% (rash, alopecia, keratoacanthoma or squamous-cell carcinoma, photosensitivity), fatigue, and arthralgia 30% to 50%, diarrhea 10% to 30%, and nausea 10% to 20% [16]. About 15% to 30% of patients treated with type I BRAF inhibitors, such as vemurafenib, develop squamous-cell carcinomas and keratoacanthomas. These non-melanoma skin cancers are not deadly, but they can become dangerous, when they rarely become metastatic [19]. Squamous cell tumors from patients treated with BRAF inhibitors have a distinct mutational profile. A higher frequency of activating RAS mutations was found in tumors from patients treated with vemurafenib (about 60%) vs. sporadic cases (range between 3% and 30%) [20]. Vemurafenib alone does not increase the number of tumors; it just decreases their latency by promoting pre-existent mutations. This is evidenced by the early appearance of lesions (within the first few weeks) after assumption of vemurafenib, and only in a subset of patients [21]. Therefore, testing the RAS status should be useful in patients who undergo treatment with BRAF inhibitors. Assuming that concomitant administration of MEK inhibitors can stop vemurafenib-induced acceleration of tumor growth in patients with RAS mutation, it may be possible to develop a new generation of BRAF inhibitors [22-24].
Another emerging problem is the resistance to BRAF inhibitors that develops within months [25].
Recent studies suggest that it could depend on tyrosine-kinase receptors (like PDGFR and IGFR-1) [26,27]. Inhibitors of c-Kit and mitogen-activated protein kinase (MEK) have also been found to act against melanomas, and MEK inhibitors are now being examined as a strategy to overcome BRAF inhibitor resistance [27].
ASR is uncommon but fatal if untreated. Its diagnosis should not be missed or delayed because of low clinical suspicion; in contrast it should be routinely considered in the differential diagnosis of acute surgical abdomen and of a wide range of medical conditions [28]. Various hypotheses could be advanced concerning the possible correlations between underlying disease, drug therapy, and acute complications. The possibility that a link between cancer and spontaneous splenic rupture exists in the absence of splenic metastasis or chemotherapy has been suggested for a long time, and the reasons are to be found in a hypercoagulable state secondary to the underlying malignancy [29-32]. Furthermore, the possibility of splenic rupture without a previous trauma in patients under treatment for abdominal manifestations of metastatic cancer has already been described, but metastases were assumed to be the cause [33,34]. Another possible cause might be found in alterations of angiogenesis pathways; BRAFV600E-dependent VEGF production has been suggested as angiogenetic promoter mechanism [35]. Oncogenic BRAF V600E mutation seems to enhance the expression of several proangiogenic and proinflammatory molecules, including VEGF-A [36]. BRAF binds to and is downstream from the main effectors of KRAS, whose activating mutations are believed to support the chaotic tumor vascularity, by up-regulating the transcription of several angiogenic inducers, including VEGF-A [37]. This might have caused splenic parenchyma fragility, resulting in a greater tendency to a spontaneous or minor trauma-related rupture; in fact, whether and to what measure BRAF regulates and alters angiogenesis is still unclear.
Conclusions
As shown by clinical data, we can assume a relationship, which is currently not verifiable, between the intake of BRAF inhibitors and spontaneous rupture of the spleen; also superficial venous thrombosis in the postoperative course has been reported. With this report we intend to comment on an unusual event, namely the spontaneous rupture of the spleen occurred in a patient with stage IV melanoma under treatment with vemurafenib, in the absence of neoplastic involvement of the spleen, splenomegaly, or major alterations of coagulation.
Consent
Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Abbreviations
AJCC: American Joint Committee on Cancer; ASR: Atraumatic Splenic Rupture; BRAF: V-raf murine sarcoma viral oncogene homolog B1; CT: Computer Tomography; Hb: Hemoglobin; HMB-45: Human Melanoma Black; Ht: Hematocrit; IGFR-1: Insulin-like Growth Factor 1 (IGF-1) Receptor; IU: International Units; LMWH: Low Molecular Weight Heparin; MART-1: Melanoma Antigen Recognized by T-cells; MEK: Mitogen-Activated protein Kinase; PDGFR: Platelet-Derived Growth Factor (PDGF) Receptors; PET: Positron Emission Tomography; RBC: Red Blood Cells; S-100: 100% Soluble protein (in ammonium); WBC: White Blood Cells.
Competing interests
The authors state that none of the authors involved in the manuscript preparation has any conflicts of interest regarding the manuscript itself, neither financial nor moral conflicts. Furthermore, none of the authors received support in the form of grants, equipment, and/or pharmaceutical items.
Authors’ contributions
All authors contributed equally to this work, read, and approved the final manuscript.
Contributor Information
Elisa Castellani, Email: elisa.ecv@gmail.com.
Piero Covarelli, Email: pcovarelli@med.unipg.it.
Carlo Boselli, Email: carloboselli@yahoo.it.
Roberto Cirocchi, Email: cirocchiroberto@yahoo.it.
Antonio Rulli, Email: rulli@unipg.it.
Francesco Barberini, Email: francescobarberini@hotmail.it.
Daniela Caracappa, Email: caracappadaniela@hotmail.it.
Carla Cini, Email: carlaci75@libero.it.
Jacopo Desiderio, Email: djdesi85@hotmail.it.
Gloria Burini, Email: gipsy@technet.it.
Giuseppe Noya, Email: gnoya@unipg.it.
References
- Renzulli P, Hostettler A, Schoepfer AM, Gloor B, Candinas D. Systematic review of atraumatic splenic rupture. Br J Surg. 2009;96:1114–1121. doi: 10.1002/bjs.6737. [DOI] [PubMed] [Google Scholar]
- Rhee SJ, Sheena Y, Imber C. Spontaneous rupture of the spleen: a rare but important differential of an acute abdomen. Am J Emerg Med. 2008;26:733. doi: 10.1016/j.ajem.2007.11.003. [DOI] [PubMed] [Google Scholar]
- Natarajan N, Telang S, Miller D, Chesney J. Novel immunotherapeutic agents and small molecule antagonists of signalling kinases for the treatment of metastatic melanoma. Drugs. 2011;71:1233–1250. doi: 10.2165/11591380-000000000-00000. [DOI] [PubMed] [Google Scholar]
- Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- Vultur A, Villanueva J, Herlyn M. Targeting BRAF in advanced melanoma: a first step toward manageable disease. Clin Cancer Res. 2011;17:1658–1663. doi: 10.1158/1078-0432.CCR-10-0174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerr EH, Hameed O, Lewis JS Jr, Bartolucci AA, Wang D, Said-Al-Naief N. Head and neck mucosal malignant melanoma: clinicopathologic correlation with contemporary review of prognostic indicators. Int J Surg Pathol. 2012;20(1):37–46. doi: 10.1177/1066896911417970. [DOI] [PubMed] [Google Scholar]
- Di Libero L, Sciascia V, Esposito D, Varriale R, Tartaglia E, Santini L. Surgical treatment of metastases from cutaneous melanoma to the small intestine and the spleen. Case reports and review of the literature. Ann Ital Chir. 2011;82:233–238. [PubMed] [Google Scholar]
- Balch CM, Gerschenwald JE, Soong S, Thompson JF, Atkins MB, Byrd DR, Buzaid AC, Cochran AJ, Coit DG, Ding S, Eggermont AM, Flaherty KT, Gimotty PA, Kirkwood JM, McMasters KM, Mihm MC Jr, Morton DL, Ross MI, Sober AJ, Sondak VK. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199–6206. doi: 10.1200/JCO.2009.23.4799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J, Dummer R, Garbe C, Testori A, Maio M, Hogg D, Lorigan P, Lebbe C, Jouary T, Schadendorf D, Ribas A, O’Day SJ, Sosman JA, Kirkwood JM, Eggermont AM, Dreno B, Nolop K, Li J, Nelson B, Hou J, Lee RJ, Flaherty KT, McArthur GA. BRIM-3 Study Group: Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507–2516. doi: 10.1056/NEJMoa1103782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollack LA, Li J, Berkowitz Z, Weir HK, Wu XC, Ajani UA, Ekwueme DU, Li C, Pollack BP. Melanoma survival in the United States, 1992 to 2005. J Am Acad Dermatol. 2011;Suppl 1:s78–86. doi: 10.1016/j.jaad.2011.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hafner C. Therapie des metastasierten Melanoms mit BRAF-Inhibitoren. Hautarzt. 2011;62:696–698. doi: 10.1007/s00105-011-2232-8. [DOI] [PubMed] [Google Scholar]
- Rondelli F, Vedovati MC, Becattini C, Tomassini GM, Messina S, Noya G, Simonetti S, Covarelli P. Prognostic role of sentinel node biopsy in patients with thick melanoma: a meta-analysis. J Eur Acad Dermatol Venereol. 2012;26(5):560–565. doi: 10.1111/j.1468-3083.2011.04109.x. [DOI] [PubMed] [Google Scholar]
- Hamid O, Boasberg PD, Rosenthal K, O’Day SJ. Systemic treatment of metastatic melanoma: New approaches. J Surg Oncology. 2011;104:425–429. doi: 10.1002/jso.22034. [DOI] [PubMed] [Google Scholar]
- Pratilas CA, Solit DB. Targeting the mitogen-activated protein kinase pathway: physiological feedback and drug response. Clin Cancer Res. 2010;16:3329–3334. doi: 10.1158/1078-0432.CCR-09-3064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bollag G, Hirth P, Tsai J, Zhang J, Ibrahim PN, Cho H, Spevak W, Zhang Y, Habets G, Burton EA, Wong B, Tsang G, West BL, Powell B, Shellooe R, Marimuthu A, Nguyen H, Zhan KY, Artis DR, Schlessinger J, Su F, Higgins B, Iyer R, D’Andrea K, Koehler A, Stumm M, Lin PS, Lee RJ, Grippo J, Puzanov I. et al. Clinical efficacy of a RAF inhibitor needs broad target blockade in BRAF- mutant melanoma. Nature. 2010;467:596–599. doi: 10.1038/nature09454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arkenau HT, Kefford R, Long GV. Targeting BRAF for patients with melanoma. Br J Cancer. 2011;104:392–398. doi: 10.1038/sj.bjc.6606030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calipel A, Lefevre G, Pouponnot C, Mouriaux F, Eychene A, Mascarelli F. Mutation of B-Raf in Human choroidal melanoma cells mediates cell proliferation and transformation through the MEK/ERK pathway. J Biol Chem. 2003;278:42409–42418. doi: 10.1074/jbc.M308709200. [DOI] [PubMed] [Google Scholar]
- Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S, Teague J, Woffendin H, Garnett MJ, Bottomley W, Davis N, Dicks E, Ewing R, Floyd J, Gray K, Hall S, Hawes R, Hughes J, Kosmidou V, Menzies A, Mould C, Parker A, Stevens C, Watt S, Hooper S, Wilson R, Jayatilake H, Gusterson BA, Cooper C, Shipley J. et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–954. doi: 10.1038/nature00766. [DOI] [PubMed] [Google Scholar]
- Flaherty KT, Puzanov I, Kim KB, Ribas A, McArthur GA, Sosman JA, O’Dwyer PJ, Lee RJ, Grippo JF, Nolop K, Chapman PB. Inhibition of mutated, activated BRAF in metastatic melanoma. N Engl J Med. 2010;363:809–819. doi: 10.1056/NEJMoa1002011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberholzer PA, Kee D, Dziunycz P, Sucker A, Kamsukom N, Jones R, Roden C, Chalk CJ, Ardlie K, Palescandolo E, Piris A, Macconaill LE, Robert C, Hofbauer GF, McArthur GA, Schadendorf D, Garraway LA. RAS mutations are associated with the development of cutaneous squamous cell tumors in patients treated with RAF inhibitors. J Clin Oncol. 2012;30:316–321. doi: 10.1200/JCO.2011.36.7680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su F, Viros A, Milagre C, Trunzer K, Bollag G, Spleiss O, Reis-Filho JS, Kong X, Koya RC, Flaherty KT, Chapman PB, Kim MJ, Hayward R, Martin M, Yang H, Wang Q, Hilton H, Hang JS, Noe J, Lambros M, Geyer F, Dhomen N, Niculescu-Duvaz I, Zambon A, Niculescu-Duvaz D, Preece N, Robert L, Otte NJ, Mok S, Kee D. et al. RAS mutations in cutaneous squamous-cell carcinomas in patients treated with BRAF inhibitors. N Engl J Med. 2012;366:207–15. doi: 10.1056/NEJMoa1105358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poulikakos PI, Zhang C, Bollag G, Shokat KM, Rosen N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature. 2010;464:427–30. doi: 10.1038/nature08902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heidorn SJ, Milagre C, Whittaker S, Nourry A, Niculescu-Duvas I, Dhomen N, Hussain J, Reis-Filho JS, Springer CJ, Pritchard C, Marais R. Kinase-dead BRAF and oncogenic RAS cooperate to drive tumor progression through CRAF. Cell. 2010;140:209–21. doi: 10.1016/j.cell.2009.12.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Infante JR, Fecher LA, Nallapareddy S, Gordon MS, Flaherty KT, Cox DS, DeMarini DJ, Morris SR, Burris HA, Messersmith WA. Safety and efficacy results from the first-in-human study of the oral MEK 1/2 inhibitor GSK1120212. J Clin Oncol. 2010;28(15):2503. [Google Scholar]
- Weeraratna AT. RAF around the Edges - The Paradox of BRAF Inhibitors. N Engl J Med. 2012;366:271–273. doi: 10.1056/NEJMe1111636. [DOI] [PubMed] [Google Scholar]
- Nazarian R, Shi H, Wang Q, Kong X, Koya RC, Lee H, Chen Z, Lee MK, Attar N, Sazegar H, Chodon T, Nelson SF, McArthur G, Sosman JA, Ribas A, Lo RS. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature. 2010;468:973–977. doi: 10.1038/nature09626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villanueva J, Vultur A, Lee JT, Somasundaram R, Fukunaga-Kalabis M, Cipolla AK, Wubbenhorst B, Xu X, Gimotty PA, Kee D, Santiago-Walker AE, Letrero R, D’Andrea K, Pushparajan A, Hayden JE, Brown KD, Laquerre S, McArthur GA, Sosman JA, Nathanson KL, Herlyn M. Acquired resistance to BRAF inhibitors mediated by a RAF kinase switch in melanoma can be overcome by cotargeting MEK and IGF-1R/PI3K. Cancer Cell. 2010;18:683–695. doi: 10.1016/j.ccr.2010.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orloff MJ, Peskin GW. Spontaneous rupture of the normal spleen: a surgical enigma. Int Abstr Surg. 1958;106:1–11. [PubMed] [Google Scholar]
- Sugahara K, Togashi H, Aoki M, Mitsuhashi H, Matsuo T, Watanabe H, Abe T, Ohno S, Saito K, Saito T, Shinzawa H, Tanida H, Ito M, Takahashi T. Spontaneous splenic rupture in a patient with large hepatocellular carcinoma. Am J Gastroenterol. 1999;94:276–278. doi: 10.1111/j.1572-0241.1999.00820.x. [DOI] [PubMed] [Google Scholar]
- Smith WM, Lucas JG, Frankel WL. Splenic rupture: a rare presentation of pancreatic carcinoma. Arch Pathol Lab Med. 2004;128:1146–1150. doi: 10.5858/2004-128-1146-SRARPO. [DOI] [PubMed] [Google Scholar]
- Kyriacou A, Arulai N, Varia H. Acute abdomen due to spontaneous splenic rupture as the first presentation of lung malignancy: a case report. J Med Case Reports. 2011;5:444. doi: 10.1186/1752-1947-5-444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karakousis CP, Elias EG. Spontaneous (pathologic) rupture of spleen in malignancies. Surgery. 1974;76:674–677. [PubMed] [Google Scholar]
- Richter RH, Reingruber B, Grüneis C, Altendorf-Hofmann A, Rupprecht H. Spontaneous splenic rupture in metastatic malignant melanoma. Chir. 2001;126:630–631. doi: 10.1055/s-2001-16561. [DOI] [PubMed] [Google Scholar]
- Buzbee TM, Legha SS. Spontaneous rupture of spleen in a patient with splenic metastasis of melanoma. A case report. Tumori. 1992;78:47–48. doi: 10.1177/030089169207800111. [DOI] [PubMed] [Google Scholar]
- Sharma A, Trivedi NR, Zimmerman MA, Tuveson DA, Smith CD, Robertson GB. Mutant V599EB-Raf regulates growth and vascular development of malignant melanoma tumors. Cancer Res. 2005;65:2412. doi: 10.1158/0008-5472.CAN-04-2423. [DOI] [PubMed] [Google Scholar]
- Bottos A, Martini M, Di Nicolantonio F, Comunanza V, Maione F, Minassi A, Appendino G, Bussolino F, Bardelli A. Targeting oncogenic serine/threonine-protein kinase BRAF in cancer cells inhibits angiogenesis and abrogates hypoxia. PNAS. 2012;109:353–359. doi: 10.1073/pnas.1105026109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rak J, Mitsuhashi Y, Bayko L, Filmus J, Shirasawa S, Sasazuki T, Kerbel RS. Mutant ras oncogenes upregulate VEGF/VPF expression: Implications for induction and inhibition of tumor angiogenesis. Cancer Res. 1995;55:4575–4580. [PubMed] [Google Scholar]