Abstract
Background
Li-Fraumeni (LFS) and Li-Fraumeni-like (LFL) syndromes are associated to germline TP53 mutations, and are characterized by the development of central nervous system tumors, sarcomas, adrenocortical carcinomas, and other early-onset tumors. Due to the high frequency of breast cancer in LFS/LFL families, these syndromes clinically overlap with hereditary breast cancer (HBC). Germline point mutations in BRCA1, BRCA2, and TP53 genes are associated with high risk of breast cancer. Large rearrangements involving these genes are also implicated in the HBC phenotype.
Methods
We have screened DNA copy number changes by MLPA on BRCA1, BRCA2, and TP53 genes in 23 breast cancer patients with a clinical diagnosis consistent with LFS/LFL; most of these families also met the clinical criteria for other HBC syndromes.
Results
We found no DNA copy number alterations in the BRCA2 and TP53 genes, but we detected in one patient a 36.4 Kb BRCA1 microdeletion, confirmed and further mapped by array-CGH, encompassing exons 9–19. Breakpoints sequencing analysis suggests that this rearrangement was mediated by flanking Alu sequences.
Conclusion
This is the first description of a germline intragenic BRCA1 deletion in a breast cancer patient with a family history consistent with both LFL and HBC syndromes. Our results show that large rearrangements in these known cancer predisposition genes occur, but are not a frequent cause of cancer susceptibility.
Keywords: Breast cancer, Copy number variation, MLPA, BRCA1 microdeletion, Li-Fraumeni syndrome
Background
Germline mutations of the tumor suppressor gene TP53 account for more than half of the families with classic Li-Fraumeni syndrome (LFS) [1], which is an inherited condition characterized by the development of sarcomas and other early-onset tumors, including breast cancer [2,3]. Families presenting incomplete features of LFS are referred as having Li-Fraumeni-like syndrome (LFL). Depending on the criteria adopted to classify the cancer phenotype in a given family, up to 22% of LFL pedigrees have detectable TP53 mutations [4-6]. Several cancer predisposition syndromes that involve breast cancer have been described to date, and include, in addition to LFS/LFL, the hereditary breast and ovarian cancer (HBOC), hereditary diffuse gastric cancer, and the Cowden and Peutz-Jeghers syndromes [7]. Due to the high frequency of breast and other cancers in LFS/LFL individuals, there may be an overlap of phenotypes, and often some families fulfill genetic testing criteria for more than one hereditary breast cancer syndrome [1,8,9].
Several studies have investigated the frequency of BRCA1/BRCA2 and TP53 germline mutations in families with multiple early-onset breast cancers [6,8,10,11]. Approximately 5-10% of breast cancer is estimated to result from dominant mutations in known single genes [12-14], particularly in the BRCA1 or BRCA2 genes. Germline TP53 mutations have been considered to be responsible for only a small fraction of the hereditary breast cancer cases overall [15], and have mostly been described in families with the other core-cancers of LFS/LFL [1,8,9]. Germline mutations of the BRCA2 gene have been described in families presenting both breast cancer and sarcomas, suggesting that BRCA2 mutations account for a proportion of LFS/LFL families negative for TP53 mutations [16,17]. As far as we are aware, germline BRCA1 mutations have not been detected in LFS/LFL kindreds, not even among families presenting a complex cancer history consistent both with LFL and other syndromes that constitute the HBC phenotype [6,8,11,18].
All known breast cancer susceptibility genes present germline point mutations in only approximately 20-25% of the cases fulfilling the criteria for genetic testing [12]. Gene rearrangements can contribute to disease through different mechanisms, resulting in either imbalance of gene dosage or gene disruption, and they are not usually detected by routine molecular diagnostic methods such as gene sequencing. In particular, large rearrangements, most often deletions, have been reported as a cause of cancer susceptibility, occurring in at least 30% of highly penetrant Mendelian cancer-predisposing genes [19].
BRCA1 germline rearrangements have been implicated in up to 30% of HBC families in certain populations [19-23]. The aim of the present study was to determine the frequency of germline copy number changes of TP53BRCA1, and BRCA2 genes in breast cancer patients with clinical diagnosis of Li-Fraumeni or Li-Fraumeni-like syndrome, and without detectable germline TP53 point mutations.
Results
All studied patients were females affected by breast cancer, two of them with bilateral disease, and 11 (45.8%) with more than one primary tumor. The average age at breast cancer diagnosis was 41 years (SD: 11.5; range: 26–61 years). Nineteen of the 23 families met genetic testing criteria for both LFL and another hereditary breast cancer syndrome (Table 1); two families met criteria for both classic LFS and another hereditary breast cancer syndrome, and two fulfill only the criteria for LFL.
Table 1.
Characteristics of the probands: clinical phenotype, type of tumor and age of diagnosis (years)
| Individual Code | Classification | Breast tumor (age at diagnosis) | Other tumors (age at diagnosis) |
|---|---|---|---|
|
Y6T000 |
Birch |
breast (79) |
Lymphoma (73), skin (81) |
|
Y29T000 |
Birch/HBC |
breast (26) |
Osteosarcoma (19), soft tissue sarcoma (23), head/neck (24) |
|
Y36T000 |
Birch/HBC |
breast (44) |
|
|
Y41T000 |
Chompret/HBC |
breast (28) |
Osteosarcoma (8) |
|
Y51T000 |
Eeles1 |
breast (53) |
|
|
Y54T000 |
Eeles1/HBC |
breast (41) |
Endometrium (44) |
|
Y83T000 |
Chompret/HBC |
breast (45) |
Soft tissue sarcoma (21) |
|
Y93T000 |
LFS/HBC |
breast (42) |
|
|
Y95T000 |
Eeles2/HBC |
breast (36) |
|
|
Y101T000 |
Eeles1/HBC |
breast (48) |
Thyroid (52) |
|
Y110T000 |
Eeles1/HBC |
breast (36) |
|
|
Y112T000 |
Chompret/HBOC |
breast (34) |
|
|
Y115T000 |
Chompret/HBC |
breast (36) |
|
|
Y116T000 |
Eeles2/HBC |
breast (48) |
|
|
Y117T000 |
Eeles1/HBC |
breast (44) |
|
|
Y122T000 |
Eeles1/HBC |
breast (61) |
Colorectal cancer(68) |
|
Y123T000 |
Eeles1/HBC |
breast bilateral (37) |
|
|
Y126T000 |
Chompret/HBC |
breast (39) |
Lymphoma (23), skin (40) |
|
Y135T000 |
Eeles1/HBC |
breast (30) |
|
|
Y143T000 |
Eeles1/HBC |
breast (42) |
|
|
Y145T000 |
Chompret/HBC |
breast, bilateral (36; 36) |
|
|
Y147T000 |
Chompret/HBC |
breast (35) |
Melanoma (36) |
| Y152T000 | LFS/HBC | breast (38) | Skin (36) |
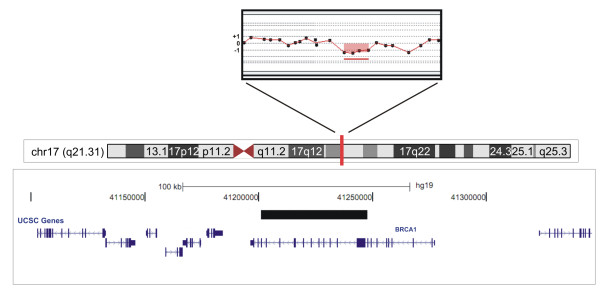
In the MLPA analysis none of the patients showed TP53, or BRCA2 deletions or duplications. We identified a single patient carrying a heterozygous intragenic BRCA1 microdeletion (Y54). Analysis using two different sets of MLPA probes (kits P087 and P002) and array-CGH allowed confirming a deletion that spanned from exon 9 to 19 (Figure 1 depicts the chromosome 17q21.31 array-CGH profile of the patient, indicating the position of the BRCA1 microdeletion). We tested two non-affected relatives of patient Y54 (III.13 and III.16) and found that one of them carries the BRCA1 deletion (III.16). Unfortunately, affected relatives of the patient Y54 could not be investigated for the presence of the BRCA1 deletion either because they were deceased or were not available.
Figure 1.
Mapping of the intragenicBRCA1deletion detected in a patient with multiple primary tumors and a cancer family history fulfilling criteria forTP53andBRCAtesting. In the upper panel, the array-CGH profile of a region at chromosome band 17q21.31, showing a heterozygous loss in copy number (red bar) of a genomic segment (image adapted from the Genomic Workbench software, Agilent Technologies). The lower panel displays the deleted segment (solid black bar) in the context of the genomic region, encompassing exons 9–19 of the BRCA1 gene according to the analysis of breakpoint sequencing data (image adapted from UCSC Genome Bioinformatics, http://genome.ucsc.edu, Build 37.1).
The DNA fragment containing the rearrangement breakpoints was sequenced and the results showed that the deletion starts at intron 8 and ends at intron 19 of the BRCA1 gene, resulting in a deletion-block identified as: g.29197_65577del36381 (Figure 2). Detailed in silico assessment of the genomic sequences surrounding the breakpoints showed that consensus Alu sequences flanked them.
Figure 2.
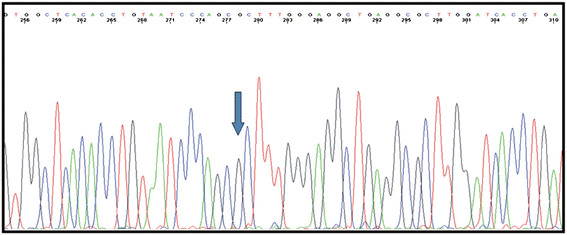
Breakpoint sequencing analysis. Eletropherogram showing the g.29197_65577del36381 mutation in the BRCA1 sequence; the intron 8 sequence is followed by intron 19 sequence. The blue arrow represents the inferred breakpoint.
Clinically, this family fulfilled genetic testing criteria for both hereditary breast and ovarian cancer (HBOC) and LFL (Eeles 1 criteria) syndromes; the cancer family history was significant for the presence of two individuals with multiple primary tumors, including the proband (Figure 3).
Figure 3.
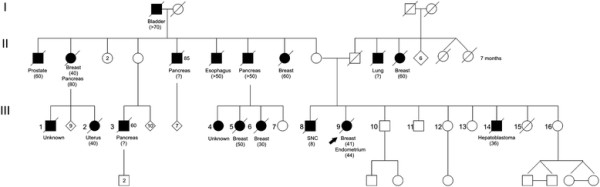
Pedigree of the family with a largeBRCA1rearrangement. Type of cancer is indicated under the subjects and the age of diagnosis is shown in brackets.
Discussion
In families with a breast cancer history that suggests the involvement of high risk genes such as TP53, BRCA1 and BRCA2, a more extensive analysis of these genes should be considered. In this study we have screened three major breast cancer predisposition genes for copy number changes in a group of 23 breast cancer patients with the clinical diagnosis of LFS/LFL who had no germline TP53 point mutations.
We did not identify large rearrangements encompassing TP53, which is in line with previous reports of low prevalence of such alterations, encountered in less than 5% of LFS/LFL families [24,25].
Similarly, large rearrangements in other breast cancer predisposition genes seem to be infrequent. A few BRCA2 deletions have been previously reported in families with male breast cancer [26], and contribute to inactivate this gene in breast cancer families [21,27]. Rearrangements affecting the BRCA2 gene have also been reported in breast/sarcoma families, causing a Li–Fraumeni type of cancer pattern [16]. Although none of the families included in this study had male breast cancer cases, nine of them had a breast cancer/sarcoma phenotype; however, no BRCA2 rearrangements were identified, which may be related to the relatively small sample size.
BRCA1 rearrangements, on the other hand, are more prevalent mostly due to the high density of Alu elements throughout the BRCA1 locus [28]. A large study by Walsh et al (2006) [11] suggested that the mutation spectra of BRCA1/BRCA2 includes several genomic rearrangements, and those alterations seem to be particularly frequent in certain populations (due to founder effect), and in families presenting individuals with multiple primary tumors [20,21,29,30]. Indeed, the “multiple primary tumors” phenotype was observed in the BRCA1 rearrangement-positive family identified in our series. Interestingly, the BRCA1 microdeletion identified here appears to be the same as the one identified in a breast cancer Italian patient [20]. Our patient is originally from southern Brazil, and since Italians have strongly contributed to the ethnic make-up of southern Brazilian population [31] it is possible that the Brazilian and the Italian patients have a common ancestry. Considering that we could not establish the parental origin of the rearrangement, this large genomic deletion may represent a breast cancer susceptibility allele rather than a more general cancer predisposition factor.
This study contributes to the understanding of the etiology of cancer susceptibility in Li-Fraumeni (LFS) and Li-Fraumeni-like (LFL) families, and their possible relation to large genomic rearrangements in high risk breast cancer susceptibility genes.
Conclusion
In patients with a cancer family history consistent with genetic testing criteria for multiple breast cancer syndromes, a comprehensive investigation, including full gene sequencing and rearrangement screening of multiple loci may be necessary to determine the precise molecular mechanisms underlying the disease. However, as illustrated with this study, in many families with cancer histories clearly indicative of hereditary cancer predisposition, the disease-causing molecular mechanisms remain elusive. Thus, despite the availability of extensive genotyping and sequencing approaches, determination of the precise pathogenic mechanisms of hereditary cancer in many cases is still a significant challenge.
Methods
Patients
The research protocol was approved by the institutional ethics committees of the participating Institutions (Protocol numbers 1175/08 and GPPG-HCPA 04–081), and recruitment of patients was done after signature of informed consent. DNA samples from 23 patients were obtained from peripheral blood; sample quality was assessed using Nanodrop and molecular weight was checked by electrophoresis in 0.8% agarose gels. TP53 mutation testing was previously performed by direct sequencing of exons 2–11, using the protocols published in http://www-p53.iarc.fr/p53sequencing.html[24].
Family history was recorded in detailed pedigrees with information traced as far backwards and laterally as possible, extending to paternal lines and including a minimum of three generations. Confirmation of the family history of cancer was attempted in all cases and pathology reports, medical records and/or death certificates were obtained whenever possible.
We selected 23 breast cancer patients with an indication for TP53 mutation testing due to a Li-Fraumeni or Li-Fraumeni-like phenotype according to the classical criteria [32] or at least one of the LFL definitions: Chompret, Birch or Eeles [4,33-35]. In all families, TP53 mutation testing was negative [36]. Additionally, some of these families also fulfilled mutation testing criteria for other hereditary breast cancer syndromes, as described in the NCCN Practice Guidelines in Oncology – v.1.2010 [37].
Clinical features of the 23 probands are summarized in Table 1.
Multiplex ligation probe amplification (MLPA)
Deletions and duplications affecting all coding exons of the TP53 gene (12 probes) were investigated by MLPA [38](MRC-Holland, Amsterdam, The Netherlands, kit P056). MLPA experiments were performed in duplicates for each patient sample, with simultaneous analysis of DNA samples from two healthy individuals from the general population (negative controls), and two patients carrying previously characterized germlineTP53 rearrangements (positive controls: a Li-Fraumeni patient with an intragenic TP53 deletion [39]; and a patient harboring a large 17p13 duplication from our in-house database). Deletions and duplications affecting BRCA1 and BRCA2 exons were also investigated by MLPA (MRC-Holland, Amsterdam, The Netherlands, kits P087 and P045, respectively; kit P002 was also used for confirmatory analysis of one detected BRCA1 microdeletion); duplicated experiments were performed simultaneously in samples from patients, two healthy individuals, and samples previously identified as carrying large duplications encompassing the BRCA1 and BRCA2 genes (positive controls; patients from our in-house database).
The PCR-amplified fragments were separated by capillary electrophoresis on an ABI 3130 XL genetic analyzer (Applied Biosystems, Foster City, California), and analyzed using the Coffalyser software (MRC Holland). We performed direct normalization with control probes as normalization factor, using the median of all imported samples, and two standard deviations. Values >1.3 were considered as possible duplications, and deletions were considered for probes exhibiting values < 0.7. Using this analysis, alterations present in all positive controls were detected.
Comparative genomic hybridization on microarrays (array-CGH)
Array-CGH analysis was performed as previously described [40] to confirm an intragenic BRCA1 deletion detected by MLPA in one patient (Y54). We used a whole-genome 180 K platform (Agilent Technologies), according to the manufacturer’s instructions; a gain or loss in copy number was considered when the log2 ratio of the Cy3/Cy5 intensities of a given genomic segment was > 0.6 or < −0.8, respectively. As reference DNA, we used commercially available human Promega female DNA (Promega, Madison, WI, USA).
Breakpoint Sequencing Analysis
To assess the microdeletion breakpoints, specific primers (forward: 5'- ACTCTGAGGACAAAGCAGCGGA -3'; reverse: 5'-GTGCCACCAAGCCCGGCTAA -3') were designed in order to amplify the breakpoint region of the BRCA1 rearrangement (microdeletion involving the same exons described by [20]. A 450 bp fragment was detected only in the sample with the microdeletion, and absent in the normal controls. The 450 bp fragment was purified from the gel using the Gel Band Purification Kit (Illustra, GE Healthcare UK limited, Buckinghamshire, United Kingdom) and sequenced (forward and reverse) using the Big Dye V3.1 Terminator Kit (Applied Biosystems, Forster City, CA, USA) on an automated sequencer ABI Prism 310 Genetic Analyser (Applied BioSystems,) according to the manufacturer’s instructions.
We performed an in silico analysis of the genomic sequences surrounding the breakpoints using the RepeatMasker program ( http://www.repeatmasker.org/) that screens DNA sequences for interspersed repeats and low complexity DNA sequences.
Abbreviations
LFS, Li-Fraumeni syndrome; LFL, Li-Fraumeni like syndrome; HBC, Hereditary breast cancer syndromes; HBOC, Hereditary breast and ovarian cancer syndrome; CNV, Copy number variation; Array-CGH, comparative genomic hybridization on microarrays; MLPA, Multiplex ligation probe amplification; NCCN, National Comprehensive Cancer Network; IARC, International Agency for Research on Cancer; UCSC, University of California, Santa Cruz.
Competing interests
The authors declare that they have no competing interests.
Authors’ contribution
AG carried out the molecular genetics studies and has drafted the manuscript. IPE carried out part of the MLPA assays and characterized the rearrangement breakpoints by sequencing. MS carried out part of the MLPA assays of the TP53 gene. MP characterized the rearrangement breakpoints by sequencing. AP characterized the rearrangement breakpoints by sequencing. AFN participated of the clinical trial and classification of the families. DMC participated in the design of the study and revised the manuscript. MIWA and PAP were physicians responsible for the clinical trial, selection and classification of the families, and critically revised the manuscript. PAP and MT supervised the BRCA1 MLPA and sequencing analyses and result interpretation. CR revised critically the manuscript. ACVK participated in the design of the study, performed part of the molecular genetics analysis, and helped to draft the manuscript. All authors read and approved the final manuscript.
Links
National Comprehensive Cancer Network [ http://www.nccn.org/]
Database of Genomic Variants [ http://projects.tcag.ca/variation/]
International Agency for Research on Cancer [ http://www.iarc.fr/]
UCSC Genome Bioinformatics [ http://genome.ucsc.edu/]
Pre-publication history
The pre-publication history for this paper can be accessed here:
Contributor Information
Amanda Gonçalves Silva, Email: amandagonc@yahoo.com.
Ingrid Petroni Ewald, Email: ipewald@gmail.com.
Marina Sapienza, Email: m_sapienza@hotmail.com.
Manuela Pinheiro, Email: manueladiaspinheiro@gmail.com.
Ana Peixoto, Email: anpeixoto@gmail.com.
Amanda França de Nóbrega, Email: amanda.nobrega@hcancer.org.br.
Dirce M Carraro, Email: dirce.carraro@hcancer.org.br.
Manuel R Teixeira, Email: manuel.teixeira@ipoporto.min-saude.pt.
Patricia Ashton-Prolla, Email: pprolla@hcpa.ufrgs.br.
Maria Isabel W Achatz, Email: miachatz@hcancer.org.br.
Carla Rosenberg, Email: carlarosenberg@uol.com.br.
Ana C V Krepischi, Email: ana.krepischi@gmail.com.
Acknowledgements
This work was supported by grants from the Brazilian National Institute of Science and Technology in Oncogenomics (FAPESP 2008/57887-9, and CNPq 573589/08-9), Fundo de Incentivo a Pesquisa (FIPE), Hospital de Clínicas de Porto Alegre (04–081 and 09–115), and FAPERGS (PRONEX "Programa de Apoio a Núcleos de Excelência”), Brazil. IPE received a grant for sandwich doctoral training from CAPES Process: 2317/10-9. We are indebted with the patients and their families.
References
- Malkin D, Li FP, Strong LC, Fraumeni JF, Nelson CE, Kim DH, Kassel J, Gryka MA, Bischoff FZ, Tainsky MA. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science. 1990;250(4985):1233–1238. doi: 10.1126/science.1978757. [DOI] [PubMed] [Google Scholar]
- Li FP, Fraumeni JF, Mulvihill JJ, Blattner WA, Dreyfus MG, Tucker MA, Miller RW. A cancer family syndrome in twenty-four kindreds. Cancer Res. 1988;48(18):5358–5362. [PubMed] [Google Scholar]
- Birch JM, Alston RD, McNally RJ, Evans DG, Kelsey AM, Harris M, Eden OB, Varley JM. Relative frequency and morphology of cancers in carriers of germline TP53 mutations. Oncogene. 2001;20(34):4621–4628. doi: 10.1038/sj.onc.1204621. [DOI] [PubMed] [Google Scholar]
- Birch JM, Heighway J, Teare MD, Kelsey AM, Hartley AL, Tricker KJ, Crowther D, Lane DP, Santibáñez-Koref MF. Linkage studies in a Li-Fraumeni family with increased expression of p53 protein but no germline mutation in p53. Br J Cancer. 1994;70(6):1176–1181. doi: 10.1038/bjc.1994.468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frebourg T, Barbier N, Yan YX, Garber JE, Dreyfus M, Fraumeni J, Li FP, Friend SH. Germ-line p53 mutations in 15 families with Li-Fraumeni syndrome. Am J Hum Genet. 1995;56(3):608–615. [PMC free article] [PubMed] [Google Scholar]
- Rapakko K, Allinen M, Syrjäkoski K, Vahteristo P, Huusko P, Vähäkangas K, Eerola H, Kainu T, Kallioniemi OP, Nevanlinna H. et al. Germline TP53 alterations in Finnish breast cancer families are rare and occur at conserved mutation-prone sites. Br J Cancer. 2001;84(1):116–119. doi: 10.1054/bjoc.2000.1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindor NM, McMaster ML, Lindor CJ, Greene MH. National Cancer Institute DoCP C.mmunity Oncology and Prevention Trials Research Group. Concise handbook of familial cancer susceptibility syndromes - second edition. J Natl Cancer Inst Monogr. 2008;38:1–93. doi: 10.1093/jncimonographs/lgn001. [DOI] [PubMed] [Google Scholar]
- Huusko P, Castrén K, Launonen V, Soini Y, Pääkkönen K, Leisti J, Vähäkangas K, Winqvist R. Germ-line TP53 mutations in Finnish cancer families exhibiting features of the Li-Fraumeni syndrome and negative for BRCA1 and BRCA2. Cancer Genet Cytogenet. 1999;112(1):9–14. doi: 10.1016/S0165-4608(98)00258-1. [DOI] [PubMed] [Google Scholar]
- Lehman TA, Haffty BG, Carbone CJ, Bishop LR, Gumbs AA, Krishnan S, Shields PG, Modali R, Turner BC. Elevated frequency and functional activity of a specific germ-line p53 intron mutation in familial breast cancer. Cancer Res. 2000;60(4):1062–1069. [PubMed] [Google Scholar]
- Lalloo F, Varley J, Ellis D, Moran A, O'Dair L, Pharoah P, Evans DG, Group EOBCS. Prediction of pathogenic mutations in patients with early-onset breast cancer by family history. Lancet. 2003;361(9363):1101–1102. doi: 10.1016/S0140-6736(03)12856-5. [DOI] [PubMed] [Google Scholar]
- Walsh T, Casadei S, Coats KH, Swisher E, Stray SM, Higgins J, Roach KC, Mandell J, Lee MK, Ciernikova S. et al. Spectrum of mutations in BRCA1, BRCA2, CHEK2, and TP53 in families at high risk of breast cancer. JAMA. 2006;295(12):1379–1388. doi: 10.1001/jama.295.12.1379. [DOI] [PubMed] [Google Scholar]
- Easton DF. How many more breast cancer predisposition genes are there? Breast Cancer Res. 1999;1(1):14–17. doi: 10.1186/bcr6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chompret A. Clinical and molecular diagnosis of inherited breast-ovarian cancer. J Gynecol Obstet Biol Reprod (Paris) 2003;32(2):101–119. [PubMed] [Google Scholar]
- Hirshfield KM, Rebbeck TR, Levine AJ. Germline mutations and polymorphisms in the origins of cancers in women. J Oncol. 2010;2010:297671. doi: 10.1155/2010/297671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Børresen AL, Andersen TI, Garber J, Barbier-Piraux N, Thorlacius S, Eyfjörd J, Ottestad L, Smith-Sørensen B, Hovig E, Malkin D. Screening for germ line TP53 mutations in breast cancer patients. Cancer Res. 1992;52(11):3234–3236. [PubMed] [Google Scholar]
- Evans DG, Wu CL, Birch JM. BRCA2: a cause of Li-Fraumeni-like syndrome. J Med Genet. 2008;45(1):62–63. doi: 10.1136/jmg.2007.054494. [DOI] [PubMed] [Google Scholar]
- Manoukian S, Peissel B, Pensotti V, Barile M, Cortesi L, Stacchiotti S, Terenziani M, Barbera F, Pasquini G, Frigerio S. et al. Germline mutations of TP53 and BRCA2 genes in breast cancer/sarcoma families. Eur J Cancer. 2007;43(3):601–606. doi: 10.1016/j.ejca.2006.09.024. [DOI] [PubMed] [Google Scholar]
- Lalloo F, Varley J, Moran A, Ellis D, O'dair L, Pharoah P, Antoniou A, Hartley R, Shenton A, Seal S. et al. BRCA1, BRCA2 and TP53 mutations in very early-onset breast cancer with associated risks to relatives. Eur J Cancer. 2006;42(8):1143–1150. doi: 10.1016/j.ejca.2005.11.032. [DOI] [PubMed] [Google Scholar]
- Kuiper RP, Ligtenberg MJ, Hoogerbrugge N, Geurts van Kessel A. Germline copy number variation and cancer risk. Curr Opin Genet Dev. 2010;20(3):282–289. doi: 10.1016/j.gde.2010.03.005. [DOI] [PubMed] [Google Scholar]
- Montagna M, Dalla Palma M, Menin C, Agata S, De Nicolo A, Chieco-Bianchi L, D'Andrea E. Genomic rearrangements account for more than one-third of the BRCA1 mutations in northern Italian breast/ovarian cancer families. Hum Mol Genet. 2003;12(9):1055–1061. doi: 10.1093/hmg/ddg120. [DOI] [PubMed] [Google Scholar]
- Woodward A, Davis T, Silva A, Kirk J, Leary J, Investigators K. Large genomic rearrangements of both BRCA2 and BRCA1 are a feature of the inherited breast/ovarian cancer phenotype in selected families. J Med Genet. 2005;42(5):e31. doi: 10.1136/jmg.2004.027961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen TO, Jønson L, Albrechtsen A, Andersen MK, Ejlertsen B, Nielsen FC. Large BRCA1 and BRCA2 genomic rearrangements in Danish high risk breast-ovarian cancer families. Breast Cancer Res Treat. 2009;115(2):315–323. doi: 10.1007/s10549-008-0088-0. [DOI] [PubMed] [Google Scholar]
- Hartmann C, John AL, Klaes R, Hofmann W, Bielen R, Koehler R, Janssen B, Bartram CR, Arnold N, Zschocke J. Large BRCA1 gene deletions are found in 3% of German high-risk breast cancer families. Hum Mutat. 2004;24(6):534. doi: 10.1002/humu.9291. [DOI] [PubMed] [Google Scholar]
- IARC - INTERNATIONAL AGENCY FOR RESEARCH ON CANCER. [http://www.iarc.fr] [DOI] [PubMed]
- Petitjean A, Mathe E, Kato S, Ishioka C, Tavtigian SV, Hainaut P, Olivier M. Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum Mutat. 2007;28(6):622–629. doi: 10.1002/humu.20495. [DOI] [PubMed] [Google Scholar]
- Tournier I, Paillerets BB, Sobol H, Stoppa-Lyonnet D, Lidereau R, Barrois M, Mazoyer S, Coulet F, Hardouin A, Chompret A. et al. Significant contribution of germline BRCA2 rearrangements in male breast cancer families. Cancer Res. 2004;64(22):8143–8147. doi: 10.1158/0008-5472.CAN-04-2467. [DOI] [PubMed] [Google Scholar]
- Agata S, Dalla Palma M, Callegaro M, Scaini MC, Menin C, Ghiotto C, Nicoletto O, Zavagno G, Chieco-Bianchi L, D'Andrea E. et al. Large genomic deletions inactivate the BRCA2 gene in breast cancer families. J Med Genet. 2005;42(10):e64. doi: 10.1136/jmg.2005.032789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pavlicek A, Noskov VN, Kouprina N, Barrett JC, Jurka J, Larionov V. Evolution of the tumor suppressor BRCA1 locus in primates: implications for cancer predisposition. Hum Mol Genet. 2004;13(22):2737–2751. doi: 10.1093/hmg/ddh301. [DOI] [PubMed] [Google Scholar]
- Gad S, Klinger M, Caux-Moncoutier V, Pages-Berhouet S, Gauthier-Villars M, Coupier I, Bensimon A, Aurias A, Stoppa-Lyonnet D. Bar code screening on combed DNA for large rearrangements of the BRCA1 and BRCA2 genes in French breast cancer families. J Med Genet. 2002;39(11):817–821. doi: 10.1136/jmg.39.11.817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazoyer S. Genomic rearrangements in the BRCA1 and BRCA2 genes. Hum Mutat. 2005;25(5):415–422. doi: 10.1002/humu.20169. [DOI] [PubMed] [Google Scholar]
- Pena SD, Di Pietro G, Fuchshuber-Moraes M, Genro JP, Hutz MH, Kehdy FeS, Kohlrausch F, Magno LA, Montenegro RC, Moraes MO. et al. The genomic ancestry of individuals from different geographical regions of Brazil is more uniform than expected. PLoS One. 2011;6(2):e17063. doi: 10.1371/journal.pone.0017063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li FP, Fraumeni JF. Soft-tissue sarcomas, breast cancer, and other neoplasms. A familial syndrome? Ann Intern Med. 1969;71(4):747–752. doi: 10.7326/0003-4819-71-4-747. [DOI] [PubMed] [Google Scholar]
- Eeles RA. Germline mutations in the TP53 gene. Cancer Surv. 1995;25:101–124. [PubMed] [Google Scholar]
- Chompret A, Brugières L, Ronsin M, Gardes M, Dessarps-Freichey F, Abel A, Hua D, Ligot L, Dondon MG, Bressac-de Paillerets B. et al. P53 germline mutations in childhood cancers and cancer risk for carrier individuals. Br J Cancer. 2000;82(12):1932–1937. doi: 10.1054/bjoc.2000.1167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chompret A, Abel A, Stoppa-Lyonnet D, Brugiéres L, Pagés S, Feunteun J, Bonaïti-Pellié C. Sensitivity and predictive value of criteria for p53 germline mutation screening. J Med Genet. 2001;38(1):43–47. doi: 10.1136/jmg.38.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Achatz MI, Olivier M, Le Calvez F, Martel-Planche G, Lopes A, Rossi BM, Ashton-Prolla P, Giugliani R, Palmero EI, Vargas FR. et al. The TP53 mutation, R337H, is associated with Li-Fraumeni and Li-Fraumeni-like syndromes in Brazilian families. Cancer Lett. 2007;245(1–2):96–102. doi: 10.1016/j.canlet.2005.12.039. [DOI] [PubMed] [Google Scholar]
- NCCN - National Comprehensive Cancer Network. [http://www.nccn.org]
- Schouten JP, McElgunn CJ, Waaijer R, Zwijnenburg D, Diepvens F, Pals G. Relative quantification of 40 nucleic acid sequences by multiplex ligation-dependent probe amplification. Nucleic Acids Res. 2002;30(12):e57. doi: 10.1093/nar/gnf056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shlien A, Baskin B, Achatz MI, Stavropoulos DJ, Nichols KE, Hudgins L, Morel CF, Adam MP, Zhukova N, Rotin L. et al. A common molecular mechanism underlies two phenotypically distinct 17p13.1 microdeletion syndromes. Am J Hum Genet. 2010;87(5):631–642. doi: 10.1016/j.ajhg.2010.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krepischi A, Achatz M, Santos E, Costa S, Lisboa B, Brentani H, Santos T, Gonçalves A, Nóbrega A, Pearson P, Germline DNA copy number variation in familial and early-onset breast cancer. Breast Cancer Res. 2012. in press. [DOI] [PMC free article] [PubMed]