Abstract
Background:
Biomarkers predictive of pathological complete response (pCR) to neoadjuvant chemotherapy (NACT) of breast cancer are urgently needed.
Methods:
Using a training/validation approach for detection of predictive biomarkers in HER2-negative breast cancer, pre-therapeutic core biopsies from four independent cohorts were investigated: Gene array data were analysed in fresh frozen samples of two cohorts (n=86 and n=55). Quantitative reverse transcription polymerase chain reaction (qRT–PCR) was performed in formalin-fixed, paraffin-embedded (FFPE) samples from two neoadjuvant phase III trials (GeparTrio, n=212, and GeparQuattro, n=383).
Results:
A strong predictive capacity of thymosin beta 15 (TMSB15A) gene expression was evident in both fresh frozen cohorts (P<0.0001; P<0.0042). In the GeparTrio FFPE training cohort, a significant linear correlation between TMSB15A expression and pCR was apparent in triple-negative breast cancer (TNBC) (n=61, P=0.040). A cutoff point was then defined that divided TNBC into a low and a high expression group (pCR rate 16.0% vs 47.2%). Both linear correlation of TMSB15A mRNA levels (P=0.017) and the pre-defined cutoff point were validated in 134 TNBC from GeparQuattro (pCR rate 36.8% vs 17.0%, P=0.020). No significant predictive capacity was observed in luminal carcinomas from GeparTrio and GeparQuattro.
Conclusion:
In TNBC, TMSB15A gene expression analysis might help to select patients with a high chance for pCR after NACT.
Keywords: predictive factor, neoadjuvant chemotherapy, triple negative, breast cancer, thymosin beta 15
Neoadjuvant chemotherapy (NACT), initially an approach for locally advanced, inoperable tumours, has become an established treatment option for operable breast cancer (Goldhirsch et al, 2007; Kaufmann et al, 2012). Response to chemotherapy is evaluated histologically in the surgical specimen after NACT, and pathological complete response (pCR) is defined as the complete absence of invasive tumour cells in breast and axillary lymph nodes. Pathological complete response is also a surrogate marker for long-term survival for triple-negative breast cancer (TNBC), which lack the expression of hormone receptors (HR) and HER2 (Carey et al, 2007; Liedtke et al, 2008). Similarly, in HER2-positive carcinomas treated by trastuzumab-containing NACT, pCR indicates a favourable prognosis (Untch et al, 2011). The situation is less clear for HR-positive luminal cancers that have an intrinsic good prognosis (von Minckwitz et al, 2011a). With current chemotherapeutic approaches, approximately 25% of unselected patients achieve a pCR (von Minckwitz et al, 2011b). The strong dependence of pCR from molecular tumour type has been elucidated in the past years. Thus, pCR rates are only in the range of 9% in HR-positive (luminal) carcinomas, whereas TNBC achieve much higher rates (35–40%) (von Minckwitz et al, 2011a). Therefore, even in selected breast cancer patient subgroups, a considerable proportion of patients does not respond and is in danger of a dismal outcome. New biomarkers for response prediction are needed that help to stratify patients according to their risk profile for an optimal therapy.
In this project, we identified the predictive capacity of TMSB15A gene expression by DNA microarray analysis of fresh-frozen pre-therapeutic breast cancer biopsies of two independent patient cohorts that have been described in previous publications (Modlich et al, 2005; Rody et al, 2007). TMSB15A encodes thymosin beta 15 (TMSB15), a member of the beta thymosin family of regulatory proteins (Chen et al, 2005). It was the gene with the highest fold change in pCR cases as compared with non-pCR cases in both cohorts and lowest significance level after correction for multiple testing. As TMSB15A did not belong to a group of genes from biological motives already known to be associated with pCR (such as for example, proliferation or immune response, Denkert et al, 2010; Fasching et al, 2011), we decided to systematically validate this individual biomarker in FFPE core biopsies from two additional independent patient cohorts with HER2-negative carcinomas from the neoadjuvant phase III clinical trials GeparTrio (von Minckwitz et al, 2005, 2008a, 2008b) and GeparQuattro (von Minckwitz et al, 2010). In our methodology, we strictly adhered to the REporting recommendations for tumour MARKer prognostic studies (REMARK) (McShane et al, 2006), and used the recently described ‘prospective–retrospective’ study design (Simon et al, 2009).
Materials and methods
Gene expression data sets
From GeparTrio as well as from an independent population-based group of patients treated with neoadjuvant EC, 55 and 86 pre-therapeutic core biopsies were snap frozen and used for Affymetrix (U133A) gene array analysis (Affymetrix, Santa Clara, CA, USA) after isolation of RNA with Qiagen RNeasy (Qiagen, Hilden, Germany). Data were processed and normalised using Affymetrix Microarray Suite 5.0 software and expression values were log2 transformed. Receiver operating characteristic (ROC) analysis and logistic regression was performed for TMSB15A (probe set 205347_s_at). Details about the cohorts and generation of the Affymetrix data sets have been previously described (Modlich et al, 2005; Rody et al, 2007).
Study population
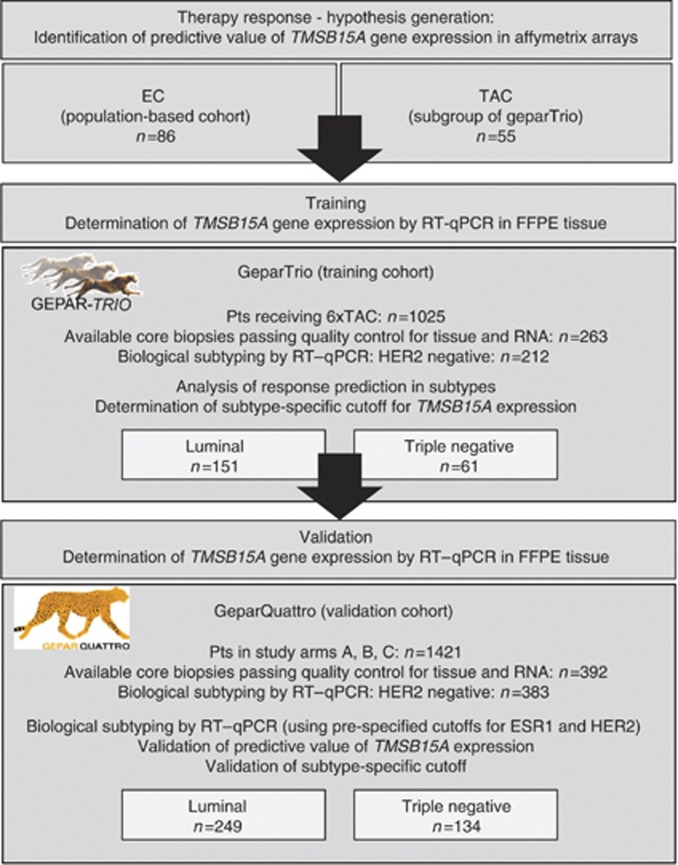
An overview on the complete study setup is given in the consort diagram (Figure 1). As an FFPE training cohort, samples from the prospective neoadjuvant phase III GeparTrio study (NCT00544765) or the GeparTrio pilot study performed by the German Breast Group, Neu-Isenburg, Germany were used. The details of study setup and treatments have been published before (von Minckwitz et al, 2005, 2008a, 2008b). For mRNA analysis, only patients who had received six cycles of TAC were included. HER2-positive patients had not received trastuzumab in GeparTrio, as this was not the standard of care during the study period; these patients were excluded from the present project. The final study population consisted of 212 patients with HER2-negative tumours, treated with 6 × TAC, and with available biomaterial. In all, 16 FFPE samples from GeparTrio were paired to fresh frozen samples used in the Affymetrix array analysis. As a validation cohort, pre-therapeutic core biopsies were used from 383 patients enroled in the prospective, neoadjuvant phase III GeparQuattro trial that compared EC-Doc vs EC-DocX vs EC-Doc-X (NCT00288002) (von Minckwitz et al, 2010). HER2-positive patients were excluded. Baseline clinico-pathological data as well as data on HER2 and HR status were extracted from the study databases. In GeparTrio, centrally evaluated data on HER2 status (based on immunohistochemistry (IHC) and silver-enhanced in situ hybridisation according to ASCO/CAP guidelines (Wolff et al, 2007)) were used, as HER2 determination was not yet fully established at the time the study was conducted. Grading and histology were also centrally determined; local data on HR expression were used (central evaluation: Institute of Pathology, Charité Berlin). Definition of pCR was complete absence of invasive tumour cells in the breast and lymph nodes as assessed at the time of surgery by the local pathologist (ypT0/Tis; ypN0). The baseline demographic and clinical characteristics of the patients with tissue available for this translational research project are shown in Table 1. The clinical trials were conducted in compliance with the Helsinki Declaration. The protocol was reviewed and approved by all responsible local ethics committees. The leading ethics committee was located at the Johann Wolfgang Goethe University, Frankfurt, Germany (Approval Number: 80/99). Consent of patient, pathologist and investigator to supply tumour material of biopsy and surgery for central pathological evaluation and examination of predictive factors was available.
Figure 1.
Consort statement: TMSB15A was a predictive factor in fresh frozen samples from a population-based cohort treated with EC (n=86), as well as in a subset of GeparTrio treated with TAC (fresh frozen tissue, n=55). Gene expression analysis was transferred to a qRT–PCR platform using 212 FFPE samples from GeparTrio. Biological tumour type was established by qRT–PCR of ESR1 and HER2 using pre-defined cutoff points, and logistic regression analysis for determination of the predictive value of TMSB15A gene expression was performed in luminal (n=151) and TNBC (n=61) separately. In the GeparTrio training cohort, a subtype-specific cutoff point for TMSB15A gene expression was determined additionally. The findings from GeparTrio were validated in 383 FFPE samples from GeparQuattro, comprising 249 luminal and 134 TNBC.
Table 1. Association of TMSB15A mRNA levels with baseline clinico-pathological parameters.
| n | Median TMSB15A gene expression, ΔCT (s.d.) | P (Mann–Whitney) | |
| GeparTrio | |||
| Biological tumour type | <0.0001 | ||
| Luminal | 151 | 9.14 (2.18) | |
| Triple-negative | 61 | 12.19 (3.18) | |
| Age | |||
| <50 years | 98 | 9.71 (2.82) | 0.006 |
| ⩾50 years | 114 | 9.21 (2.64) | |
| Tumour histology | |||
| Ductal/other | 189 | 9.47 (2.79) | 0.606 |
| Lobular | 17 | 9.66 (2.82) | |
| Missing | 6 | ||
| Tumour grade | |||
| G1–2 | 160 | 9.38 (2.62) | <0.0001 |
| G3 | 45 | 11.36 (2.96) | |
| Missing | 7 | ||
| cT | |||
| cT1–2 | 144 | 9.47 (2.89) | 0.791 |
| cT3–4 | 62 | 9.47 (2.51) | |
| Missing | 6 | ||
| cN | |||
| cN0 | 102 | 9.31 (2.84) | 0.31 |
| cN1–3 | 101 | 9.55 (2.78) | |
| Missing | 9 | ||
| GeparQuattro | |||
| Biological tumour type | <0.0001 | ||
| Luminal | 249 | 9.58 (2.21) | |
| Triple-negative | 134 | 12.59 (2.29) | |
| Age | |||
| <50 years | 176 | 10.35 (2.53) | 0.081 |
| ⩾50 years | 207 | 10.25 (2.78) | |
| Tumour histology | |||
| Ductal/other | 326 | 10.40 (2.66) | 0.009 |
| Lobular | 57 | 9.58 (2.59) | |
| Tumour grade | |||
| G1–2 | 248 | 9.84 (2.47) | <0.0001 |
| G3 | 125 | 11.99 (2.42) | |
| Missing | 10 | ||
| cT | |||
| cT1–2 | 264 | 11.44 (2.72) | 0.119 |
| cT3–4 | 119 | 10.09 (2.56) | |
| cN | |||
| cN0 | 187 | 10.41 (2.80) | 0.268 |
| cN1–3 | 196 | 10.21 (2.55) | |
Abbreviations: cN=clinical node stage; cT=clinical tumour stage.
Biomarker assays for determination of TMSB15 mRNA levels
RNA was isolated from FFPE tissue sections of core biopsies using a fully automated isolation method of total RNA based on silica-coated magnetic beads in combination with a liquid-handling robot, and RNA was then used in quantitative reverse transcription polymerase chain reaction (qRT–PCR) as described earlier (Bohmann et al, 2009; Müller et al, 2011). Expression of TMSB15A, ESR1, and HER2 as well as the normalisation genes RPL37A, CALM2, and OAZ1 was assessed in triplicate. Sequences of primers and probes are listed in Table 2. Biological tumour types were defined based on gene expression data of ESR1 and HER2 as follows: ESR1+/HER2−: luminal; ESR1+ or −/HER2+: HER2 positive; ESR1−/HER2−: TNBC. The ESR1 and HER2 mRNA cutoff values of 16 and 19.5 ΔCT were pre-defined based on two previous studies (Bohmann et al, 2009; Pentheroudakis et al, 2009). All biomarker analyses were performed blinded to patient outcome.
Table 2. Sequences of PCR primers and probes.
| Gene | FAM/TAMRA probe | Forward primer | Reverse primer | Amplicon length (bp) |
|---|---|---|---|---|
| TMSB15A | CAAGCTTTCAGGCTATCTTCTAGTCAA | CCGCGAACAGCCTTTCAC | CGACAAGTCTGGCTTATCACTCA | 77 |
| ESR1 | ATGCCCTTTTGCCGATGCA | GCCAAATTGTGTTTGATGGATTAA | GACAAAACCGAGTCACATCAGTAATAG | 73 |
| HER2 | ACCAGGACCCACCAGAGCGGG | CCAGCCTTCGACAACCTCTATT | TGCCGTAGGTGTCCCTTTG | 87 |
| OAZ1 | TGCTTCCACAAGAACCGCGAGGA | CGAGCCGACCATGTCTTCAT | AAGCCCAAAAAGCTGAAGGTT | 83 |
| CALM2 | TCGCGTCTCGGAAACCGGTAGC | GAGCGAGCTGAGTGGTTGTG | AGTCAGTTGGTCAGCCATGCT | 72 |
| RPL37A | TGGCTGGCGGTGCCTGGA | CCGCGAACAGCCTTTCAC | CGACAAGTCTGGCTTATCACTCA | 77 |
Sequences are given in 5′ to 3′ orientation.
Cell culture
Wild-type Cal-51 (DSMZ, Braunschweig, Germany) were cultured in RPMI supplemented with 10% fetal calf serum (FCS), and MCF-7 cells (DSMZ) were cultured in 5% FCS in the absence of phenol red. Paclitaxel (Sigma-Aldrich Chemie Gmbh, Munich, Germany)-resistant clones were generated by increasing drug concentration from 1.56 nℳ to 6.25 nℳ and repeated limiting dilution subcloning. Affymetrix arrays were performed as described above. Wild-type Cal-51 cells were transiently transfected with TMSB15A siRNA (100 ng, Sigma-Aldrich Chemie GmbH). Decrease of TMSB15A mRNA levels was determined by qRT–PCR. Relative gene expression was calculated from duplicate samples using the comparative CT method. Cytotoxicity assays for cell survival were performed as described previously (Stege et al, 2004). At 24 h after siRNA transfection, doxorubicin or paclitaxel were added in increasing concentrations. Cells were incubated with cytotoxic drugs for 72 h. Subsequently, cell viability was determined by sulforhodamine B assay. The absorbance difference of control cells without drug was set to be 100%. Each experiment was performed in triplicate at least.
Statistical evaluation
Statistical analysis was performed using SPSS Statistics 19 (IBM Corporation, Somers, NY, USA), GraphPad PRISM 5.01 (GraphPad software, La Jolla, CA, USA), JMP 7.0.2 (SAS, Cary, NC, USA), and the statistical language R (www.r-project.org). In logistic regression analyses, significance of the correlation with pCR was assessed by the Wald test. The cutoff point for TMSB15A gene expression was determined using an R-based software designed by our group, and which is available in the internet (http://molpath.charite.de/cutoff/): for cutoff determination, this software correlates the dichotomised biomarker with a binary outcome variable using logistic regression. The optimal cutoff is defined as the point with the most significant split (Fisher’s exact test). The details about this method will be the subject of a separate publication (manuscript submitted). All tests were two-sided, P-values <0.05 were considered as significant.
Results
Gene expression analysis
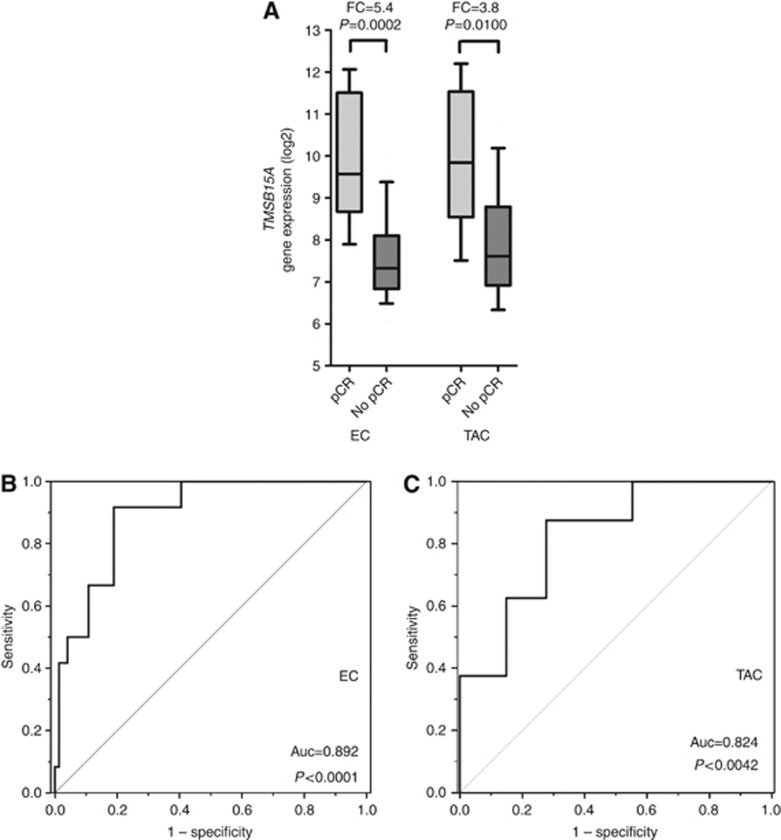
To identify biomarkers predictive of a pCR, gene expression array data from previously published studies were analysed and searched for genes with significant expression differences in pCR and non-pCR cases. TMSB15A was the gene with the highest fold change in pCR cases as compared with non-pCR cases in both cohorts. In comparison with patients who had not achieved a pCR, we found a 5.3-fold (P=0.0002) and a 3.8-fold (P=0.01) higher TMSB15A isoform expression in those patients who had achieved a pCR following EC (in the population-based cohort) and TAC NACT (in the GeparTrio cohort), respectively (Figure 2A). Receiver operating characterisitic (ROC) analysis revealed predictive power of TMSB15A gene isoform expression in both the EC as well as the TAC cohort (auc=0.89, P<0.0001, and auc=0.82, P<0.0042, respectively; Figure 2B and C). After combination of both cohorts, subgroup analysis was performed with molecular tumour types. A significant predictive value for TMSB15A gene expression in TNBC (auc=0.730, P=0.037), as well as in HER2-positive carcinomas (auc=0.821, P=0.012), and in the luminal subgroup (auc=0.918, P=0.046), although there were only two events (pCRs) within the luminal subgroup. As TMSB15A did not belong to a group of genes from biological motives already known to be associated with pCR (such as for example, proliferation or immune response), we decided to validate this individual biomarker in FFPE cohorts and to examine it functionally using siRNA technology.
Figure 2.
Distributions of TMSB15A gene expression in dependence of pCR in the EC cohort and the TAC cohort bold lines: medians; whiskers, 10–90th percentile; FC= fold change, P, Mann–Whitney test. (B, C) Reciever operator characteristic (ROC) curves for TMSB15A gene expression in the EC and TAC cohort. Abbreviation: Auc=area under the curve.
TMSB15A gene expression in FFPE tissue of GeparTrio
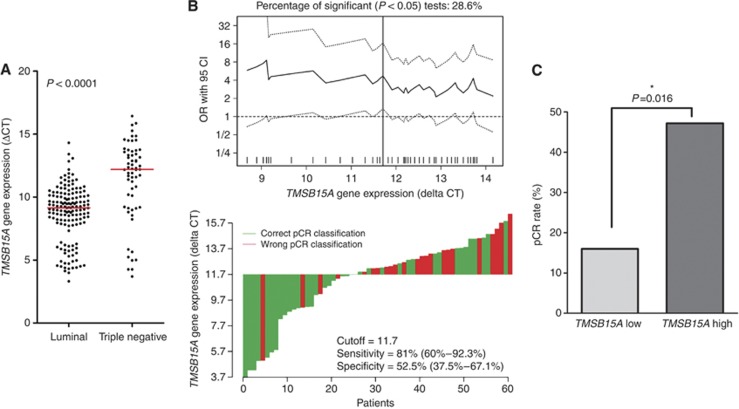
TMSB15A gene expression was subsequently investigated in routinely used FFPE samples from the GeparTrio trial using qRT–PCR. HER2-positive tumours had not been treated with neoadjuvant trastuzumab in GeparTrio, therefore only HER2-negative cases were included in the subsequent analyses. TMSB15A mRNA levels ranged from 3.32 to 16.44 ΔCT, with a median of 9.55 ΔCT. In TNBC, TMSB15A was strongly overexpressed as compared with luminal tumours (Figure 3A): median TMSB15A expression was 12.19 ΔCT in TNBC and 9.14 ΔCT in luminal carcinomas (P<0.0001, Table 1). A strong association was further seen with poor tumour differentiation (G3, P<0.0001); a weak association with younger age was evident, too (P=0.006, Table 1). Because of the different expression levels in TNBC and luminal carcinomas, further statistical investigations were performed separately in each biological subtype. An overview of clinico-pathological characteristics of the luminal and TNBC subgroup is given in Table 3; significant differences were seen for tumour grading, and pCR rate (P<0.0001 in each case). Logistic regression revealed that TMSB15A gene expression was a positive predictor of therapy response in TNBC. A doubling of the mRNA amount represented a 1.25-fold increase of the odds ratio (OR) for pCR (95% CI=1.01–1.55, P=0.040). A ΔCT of 11.7 was identified as the optimal cutoff point (Figure 3B) splitting the cohort into a low expression group (n=25) with a pCR rate of 16.0% and a high expression group (n=36) with an almost three-fold higher pCR rate of 47.2% (OR 4.70, 95% CI=1.34–16.45, P=0.016, Table 4, Figure 3C). This cutoff point had a sensitivity of 81.0% (95% CI=60–92.3%), and specificity of 52.5% (95% CI=37.5–67.1%) for response prediction (Figure 3B). Logistic regression with continuous TMSB15A mRNA data in luminal tumours did not show a significant predictive effect: OR=1.35 (95% CI=0.96–1.89, P=0.082). When molecular tumour types were defined by IHC, results were quite similar: In TNBC (ER−/PR−/HER2−, n=43) TMAB15A gene expression (continuous) was a significant predictor of pCR (OR=1.32 per ΔCT, 95% CI=1.00–1.74, P=0.049), however not in luminal carcinomas (ER+ or PR+/HER2−, n=141; OR=1.33 per ΔCT, 95% CI=0.96–1.83, P=0.083, data not shown).
Figure 3.
(A) Distribution of TMSB15A expression in luminal carcinomas and TNBC from GeparTrio. Dots indicate individual tumours. Red lines, medians. P, Mann–Whitney test. (B) Upper panel: determination of a cutoff point of TMSB15A gene expression for TNBC in GeparTrio. All possible cutoff points were considered and the corresponding odds ratios were calculated and plotted. Each data point in the line gives the corresponding OR and the 95% CI (dotted lines) on the y-axis. Vertical line: most significant split. Lower panel: waterfall plot for TNBC in GeparTrio charts each tumour as a vertical bar. Green bars represent cases with correct pCR classification, red bars represent cases with wrong pCR classification. Sensitivity and specificity of the cutoff point are indicated. (C) Pathological complete response (pCR) rates in dependence of TMSB15A status in TNBC from GeparTrio. For dichotomisation into a TMSB15A low and high expression group, the subtype-specific cutoff point was used (11.7 ΔCT). P-values were calculated by logistic regression. *Indicates significant values. The colour reproduction of this figure available at the British Journal of Cancer online.
Table 3. Clinico-pathological characteristics of luminal carcinomas and TNBC in GeparTrio and GeparQuattro.
| GeparTrio | GeparQuattro | |||||||
|---|---|---|---|---|---|---|---|---|
| Total n (%) | Luminal n (%) | Triple-negative n (%) | P | Total n (%) | Luminal n (%) | Triple-negative n (%) | P | |
| Total no. of samples | 212 | 151 | 61 | 383 | 249 | 134 | ||
| Age | ||||||||
| <50 years | 98 (46.2) | 65 (43.0) | 33 (54.1%) | 0.095a | 176 (46.0) | 108 (43.4) | 68 (50.7) | 0.123a |
| ⩽50 years | 114 (53.8) | 86 (57.0) | 28 (45.9%) | 207 (54.0) | 141 (56.6) | 66 (49.3) | ||
| Tumour histology | ||||||||
| Ductal | 184 (89.3) | 126 (86.3) | 58 (96.7) | 0.076b | 299 (78.1) | 180 (72.3) | 119 (88.8) | <0.0001b |
| Lobular | 17 (8.3) | 16 (11.0) | 1 (1.7) | 57 (14.9) | 53 (21.3) | 4 (3.0) | ||
| Other | 5 (2.4) | 4 (2.7) | 1 (1.7) | 27 (7.0) | 16 (6.4) | 11 (8.2) | ||
| Missing | 6 | 5 | 1 | |||||
| Tumour grade | ||||||||
| G1 | 21 (10.2) | 19 (13.1) | 2 (3.3) | <0.0001b | 9 (2.4) | 9 (3.8) | 0 (0) | <0.0001b |
| G2 | 139 (67.8) | 109 (75.2) | 30 (50.0) | 239 (64.1) | 188 (78.8) | 51 (38.1) | ||
| G3 | 45 (22.0) | 17 (11.7) | 28 (46.7) | 125 (33.5) | 42 (17.6) | 83 (61.9) | ||
| Missing | 7 | 6 | 1 | 10 | 10 | |||
| cT | ||||||||
| cT1 | 1 (0.5) | 0 (0) | 1 (1.7) | 0.246b | 5 (1.3) | 1 (0.4) | 4 (1.3) | 0.051b |
| cT2 | 138 (69.0) | 99 (70.2) | 39 (66.1) | 259 (67.6) | 163 (65.5) | 96 (71.6) | ||
| cT3 | 38 (19.0) | 24 (17.0) | 14 (23.7) | 59 (15.4) | 40 (16.1) | 19 (14.2) | ||
| cT4 | 23 (11.5) | 18 (12.8) | 5 (8.5) | 60 (15.7) | 45 (18.1) | 15 (11.2) | ||
| Missing | 12 | 10 | 2 | 10 | ||||
| cN | ||||||||
| cN0 | 102 (50.2) | 76 (53.5) | 26 (42.6) | 0.120a | 187 (48.8) | 115 (46.2) | 72 (53.7) | 0.096a |
| cN1–3 | 101 (49.8) | 66 (45.5) | 35 (57.4) | 196 (51.2) | 134 (53.8) | 62 (46.3) | ||
| Missing | 9 | 9 | ||||||
| pCR | ||||||||
| pCR | 32 (15.1) | 11 (7.3) | 21 (34.4) | <0.0001a | 60 (15.7) | 20 (8.0) | 40 (29.9) | <0.0001a |
| No pCR | 180 (84.9) | 140 (92.7) | 40 (65.6) | 323 (84.3) | 229 (92.0) | 94 (70.1) | ||
Abbreviations: cN=clinical node stage; cT=clinical tumour stage; pCR=pathological complete response; TNBC=triple-negative breast cancer.
Fisheŕs exact test.
Pearsońs chi-square test.
Table 4. Correlation with pCR: logistic regression analysis within TNBC.
| TMSB15A gene expression | n | Events | % pCR | OR | 95% CI | P |
|---|---|---|---|---|---|---|
| GeparTrio | ||||||
| Low (⩽11.7 ΔCT) | 25 | 4 | 16.0 | 1 | — | |
| High (>11.7 ΔCT) | 36 | 17 | 47.2 | 4.70 | 1.34–16.45 | 0.016 |
| GeparQuattro | ||||||
| Low (⩽11.7 ΔCT) | 47 | 8 | 17.0 | 1 | — | |
| High (>11.7 ΔCT) | 87 | 32 | 36.8 | 2.84 | 1.18–6.82 | 0.020 |
Abbreviations: CI=confidence interval; OR, odds ratio; pCR=pathological complete response; TNBC=triple-negative breast cancer.
Validation of the predictive impact of TMSB15A gene expression in GeparQuattro
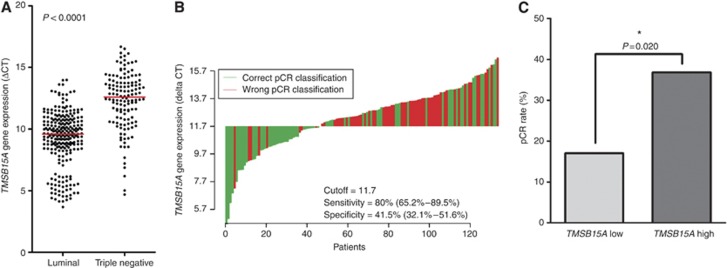
In the validation cohort GeparQuattro, distribution of TMSB15A mRNA levels was similar as in GeparTrio (range 3.67–16.67 ΔCT, median 10.29 ΔCT). A strong correlation with triple-negative subtype was seen as well: Median TMSB15A expression was 12.59 in TNBC and 9.58 in luminal tumours (P<0.0001, Table 1, Figure 4A). Association with non-lobular histology (P=0.009), and poor differentiation (P<0.0001) were further found (Table 1). As in GeparTrio, luminal and TNBC differed in tumour grading, and pCR rate but also in histology (P<0.0001 in each case, Table 3). The predictive value of TMSB15A gene expression found in GeparTrio was validated in TNBC from GeparQuattro: OR was 1.26 (95% CI=1.04–1.52) per doubling of the mRNA amount (ΔCT, P=0.017). The cutoff point of 11.7 ΔCT, which had been defined in GeparTrio, could be validated as well: pCR rate was 17.0% in TNBC with TMSB15A low expression and 36.8% in the high expression group (OR=2.84, 95% CI=1.18–6.82, P=0.020, Figure 4C, Table 4). Sensitivity was 80.0% and specificity was 41.5% (Figure 4B). Multivariate logistic regression analysis showed that in TNBC from GeparQuattro, TMSB15A gene expression was the only significant predictive factor, independently of patient age, as well as clinical tumour (cT) and nodal stage (cN, P=0.041). No significant predictive effect was seen in luminal carcinomas (OR=1.14 per ΔCT, 95% CI=0.91–1.43, P=0.249). Similarly to GeparTrio, the use of IHC-defined molecular tumour types yielded the same results. In TNBC (ER−/PR−/HER2−, n=126) TMSB15A gene expression was a significant predictive factor (OR=1.23 per ΔCT, 95% CI=1.01–1.49, P=0.041), and in luminal carcinomas (ER+ or PR+/HER2-, n=257) it was not (OR=1.05, 95% CI=0.85–1.31, P=0.652, data not shown).
Figure 4.
(A) Distribution of TMSB15A expression in luminal cancers and TNBC from GeparQuattro. Dots indicate individual tumours. Red lines, medians. P, Mann–Whitney test. (B) Waterfall plot for TNBC in GeparQuattro charts each tumour as a vertical bar. Green bars represent cases with correct pCR classification, red bars represent cases with wrong pCR classification. Sensitivity and specificity of the cutoff point are indicated. (C) Pathological complete response (pCR) rates in dependence of TMSB15A status in TNBC from GeparQuattro. For dichotomisation into a TMSB15A low and high expression group, the cutoff point of 11.7 ΔCT, which had been pre-defined in GeparTrio, was used. P-values were calculated by logistic regression. *Indicates significant values. The colour reproduction of this figure available at the British Journal of Cancer online.
Functional evaluation of the relationship between TMSB15A expression and chemosensitivity
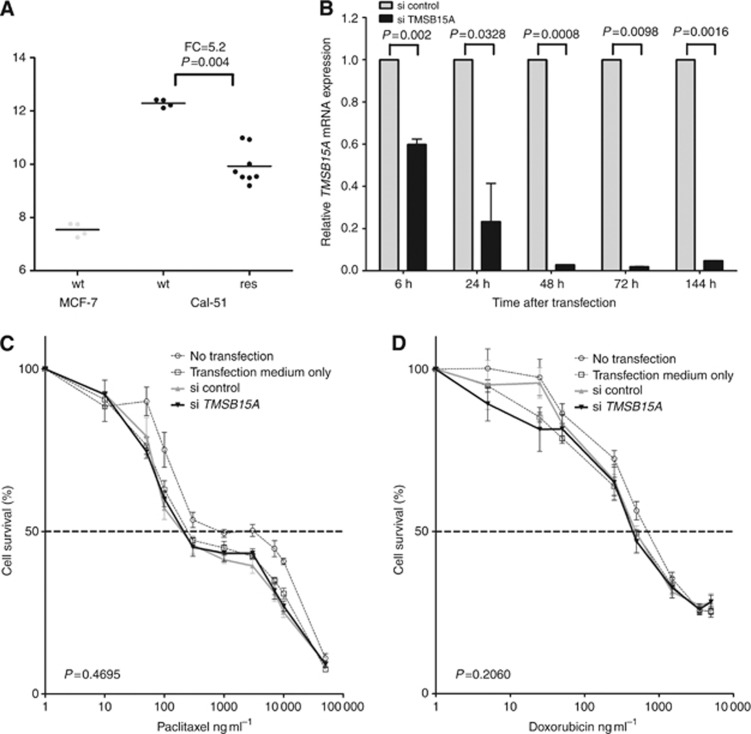
To investigate whether there is a functional link between TMSB15A mRNA levels and response to chemotherapy, exploratory cell culture experiments were conducted. Gene expression arrays that had been performed from the breast cancer cell lines MCF-7 and Cal-51 showed relatively low levels of TMSB15A mRNA detected in ER-positive (luminal) MCF-7 (Figure 5A). Triple-negative Cal-51 cells, however, showed high-level TMSB15A gene expression, and were therefore chosen for further experiments. Induction of secondary paclitaxel resistance in Cal-51 resulted in a significant decrease of TMSB15A gene expression as compared with parental cells (fold change 5.2, P=0.004; Mann–Whitney test, Figure 5A). Parental Cal-51 cells were then used for siRNA experiments. As shown in Figure 5B, transient transfection with TMSB15A siRNA successfully reduced gene expression by up to 98%, as determined by qRT–PCR. Transfected cells were subsequently tested for sensitivity to paclitaxel (Figure 5C) and doxorubicin (Figure 5D). However, no difference in sensitivity to both drugs was determined as compared with cells transfected with control siRNA (P=0.470 and P=0.206, respectively).
Figure 5.
(A) TMSB15A mRNA levels (Affymetrix HG U133A microarray; probe set 205347_s_at) in MCF-7 and Cal-51 breast cancer cell lines. For clones of wild type (wt), cells were measured (dots), four clones of paclitaxel-resistant (res) Cal-51 were measured twice (dots). Expression levels in wt and res Cal-51 were compared by Mann–Whitney test. Bold lines, mean; FC=fold change. (B) TMSB15A mRNA expression in parental Cal-51 cells transfected with siRNA against TMSB15A (si TMSB15A) relative to cells transfected with control siRNA (si control). P, columnar t-test. (C) Dose–response curves for wild-type Cal-51 treated with increasing concentrations of paclitaxel (left panel) and doxorubicin (right panel) 24 h after siRNA transfection. Comparison of si control (bold grey line) with si TMSB15A treated cells (bold black line) were made by paired t-test. Dashed line, IC50.
Discussion
In this project, we investigated TMSB15A gene expression as a novel biomarker for prediction of response to NACT in HER2-negative breast cancer. We identified the clinical validity of TMSB15A mRNA for prediction of response by gene expression analysis in two independent cohorts, and systematically validated it in the neoadjuvant clinical phase III GeparTrio and GeparQuattro trials. Taking into account the differing expression levels of TMSB15A in luminal tumours and TNBC, we investigated each biological subtype separately. In the GeparTrio training cohort and GeparQuattro validation cohort, TMSB15A significantly predicted pCR in TNBC, whereas in luminal carcinomas, no reliable predictive value could be observed. A cutoff point for TMSB15A mRNA expression levels that was defined in GeparTrio TNBC could be validated in GeparQuattro as well.
A prominent finding in our project is that TMSB15A expression is a marker for triple-negative tumour type as compared with luminal subtype. Based on the strongly differing expression levels in luminal cancers and TNBC evident in GeparTrio, we hypothesised that the utility as a predictive marker could be different within biological subtypes. Indeed, TMSB15A mRNA levels were reliably predictive only in TNBC, both on the continuous scale and after dichotomisation using a cutoff point. This cutoff point divided the triple-negative group in a low expression group with a pCR rates of 16.0%/17.0% in GeparTrio/GeparQuattro, which is lower than the pCR rate in unselected patients (von Minckwitz et al, 2011b), and a high expression group with a pCR rate of 47.2%/36.8% in GeparTrio/GeparQuattro. TNBC constitute a heterogeneous group of tumours that partially overlaps with the basal-like intrinsic subtype as defined by gene expression profiling. Several subtypes of TNBC have been described to date with the aid of molecular methods, for example the claudin-low or the HER2-enriched subtype (Perou, 2011) as described by gene expression profiling, or the 5 negative/core basal phenotype by IHC (Blows et al, 2010). The potential diagnostic or prognostic use is not clear up to now, therefore this subtyping has not yet found its way into the clinics. The current pragmatic definition of triple-negative disease used in the clinic is negativity for ER, PR, and HER2 by IHC/ISH (Badve et al, 2011). In our project, we molecularly classified luminal and TNBC similarly, however, determined ER (ESR1) and HER2 expression by qRT–PCR, which is not the clinical standard. We nevertheless showed that the predictive value of TMSB15A gene expression was also present when TNBC were defined by standard IHC/ISH, which indicates that it is not an artifact due to the use of a novel typing method. The systematic comparison between qRT–PCR and IHC/ISH for determination of tumour types is described in a recent publication of our group, which shows that molecular tumour types defined by qRT–PCR reveal a quite similar prognostic and predictive impact as those defined by IHC/ISH (Denkert et al, 2012).
The heterogeneity of TNBC is also reflected by the fact that, although in comparison to luminal carcinomas, response rates to CTX are significantly higher and are in the range of 40% for modern NACT (von Minckwitz et al, 2011b), a significant number of patients with TNBC do not achieve a pCR. Large retrospective studies have shown that pCR is a robust surrogate marker for long-term survival in patients with TNBC treated with NACT (Carey et al, 2007; Liedtke et al, 2008) Thus, patients with pCR can expect a quite favourable long-term outcome, but in patients without pCR, triple-negative disease follows an aggressive course with early metastasis and death from disease (Hudis and Gianni, 2011). Biomarkers that predict pCR in the triple-negative subgroup are therefore helpful to select those patients for whom NACT is the optimal treatment option. The cutoff point of 11.7 ΔCT detected those patients with a sensitivity of 81%/80% in GeparTrio/GeparQuattro and might therefore be an aid in the decision for NACT in combination with clinical parameters such as age (Huober et al, 2010), to which TMSB15A adds independent predictive information. On the other hand, patients with TNBC who are less likely to achieve a pCR and therefore are in danger of a dismal outcome, could be identified, too. For those patients unfortunately, no standard specific treatment exists today. However, much effort is made to develop and test experimental therapeutics for TNBC (Hudis and Gianni, 2011). In this regard, TMSB15A gene expression analysis might help to select patients with a lower chance for pCR for clinical trials investigating novel therapies in this poor prognosis subgroup.
The function of TMSB15, the protein encoded by TMSB15A, in TNBC or the mechanisms by which it is upregulated has not been investigated to date. TMSB15 belongs to the beta thymosin family of highly conserved 5-kDa proteins with three members in humans, TMSB4, TMSB10, and TMSB15 (Chen et al, 2005). All beta thymosins bind and sequester (monomeric) G-actin and remove it from the dynamic assembly/deassembly process, which constantly takes place during essential cellular functions, such as mitosis, migration, intracellular transport, and phagocytosis (Mannherz and Hannappel, 2009). Beta thymosins further have extracellular functions due to their ability to interact with various other proteins and are thereby involved in the regulation of immune response, wound healing, and angiogenesis (Chen et al, 2005). TMSB15 is the least well studied family member, however, there is increasing evidence for an essential role in tumour progression. It has been shown to be upregulated in cancer cell lines of various origin and be implicated in tumour cell migration and proliferation (Bao et al, 1996; Abdulrahman et al, 2007; Banyard et al, 2009). In breast cancer, TMSB15A might be upregulated to increase cell motility and transformation, which would explain that it is found in higher levels in aggressive, triple-negative and poorly differentiated carcinomas. Our cell culture data show an upregulation of TMSB15A in triple-negative Cal-51 cells as compared with luminal MCF-7 cell and therefore, reflect the situation found in vivo. These results further indicated that decreased TMSB15A expression seems to be a side-effect of chemoresistance, rather than a causal factor for chemoresistance, as no increase of sensitivity neither to anthracycline nor to taxane could be determined by TMSB15A knockdown with siRNA.
In luminal carcinomas, we did not observe a significant predictive effect of TMSB15A gene expression. Luminal tumour type by itself is a negative predictor for pCR, with a response rate of approximately 9% with current regimes (von Minckwitz et al, 2011). Although the identification of those few patients who benefit from NACT would be desirable, the detection of predictive biomarkers is hampered by the low number of events.
Some limitations of our study need to be mentioned. Sample size was limited by dealing with very small amounts of tissue (core biopsies) that have already been used for diagnostic purposes. Therefore, we did not reach two-thirds of the original study population as postulated by Simon for category IB evidence (Simon et al, 2009). Further, in our study cohorts, standard anthracyline/taxane-based NACT was applied, whereas the treatment protocols for triple-negative disease are changing. Especially platinum agents are increasingly applied (Hudis and Gianni, 2011). Therefore, we plan to investigate TMSB15A gene expression prospectively in patient cohorts treated with different agents to see whether it is a general maker for response to cytotoxic therapy.
In conclusion, we identified and validated TMSB15A mRNA levels as a predictive marker for response to NACT in TNBC. TMSB15A expression analysis might be helpful for the identification of patients who would benefit from anthracycline/taxane-based NACT. For patients with TMSB15A-negative TNBC, who have a smaller chance for therapy response, novel treatment options, and experimental therapeutics should be considered.
Acknowledgments
We thank Professor Hermann Lage (Experimental Pathology, Institute of Pathology, Charité Universitätsmedizin Berlin, Germany) for his kind support in the execution of cell cytotoxicity assays and Martina Eickmann for editorial assistance. This work was supported by a grant from the Bundesministerium für Bildung und Forschung (BMBF), Germany (grant number: 01ES1002, NEOpredict project).
Footnotes
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
References
- Abdulrahman M, Maina EN, Morris MR, Zatyka M, Raval RR, Banks RE, Wiesener MS, Richards FM, Johnson CM, Latif F, Maher ER (2007) Identification of novel VHL targets that are associated with the development of renal cell carcinoma. Oncogene 26: 1661–1672 [DOI] [PubMed] [Google Scholar]
- Badve S, Dabbs DJ, Schnitt SJ, Baehner FL, Decker T, Eusebi V, Fox SB, Ichihara S, Jacquemier J, Lakhani SR, Palacios J, Rakha EA, Richardson AL, Schmitt FC, Tan PH, Tse GM, Weigelt B, Ellis IO, Reis-Filho JS (2011) Basal-like and triple-negative breast cancers: a critical review with an emphasis on the implications for pathologists and oncologists. Mod Pathol 24: 157–167 [DOI] [PubMed] [Google Scholar]
- Banyard J, Barrows C, Zetter BR (2009) Differential regulation of human thymosin beta 15 isoforms by transforming growth factor beta 1. Genes Chromosomes Cancer 48: 502–509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bao L, Loda M, Janmey PA, Stewart R, Anand-Apte B, Zetter BR (1996) Thymosin beta 15: a novel regulator of tumour cell motility upregulated in metastatic prostate cancer. Nat Med 2: 1322–1328 [DOI] [PubMed] [Google Scholar]
- Blows FM, Driver KE, Schmidt MK, Broeks A, van Leeuwen FE, Wesseling J, Cheang MC, Gelmon K, Nielsen TO, Blomqvist C, Heikkilä P, Heikkinen T, Nevanlinna H, Akslen LA, Bégin LR, Foulkes WD, Couch FJ, Wang X, Cafourek V, Olson JE, Baglietto L, Giles GG, Severi G, McLean CA, Southey MC, Rakha E, Green AR, Ellis IO, Sherman ME, Lissowska J, Anderson WF, Cox A, Cross SS, Reed MW, Provenzano E, Dawson SJ, Dunning AM, Humphreys M, Easton DF, García-Closas M, Caldas C, Pharoah PD, Huntsman D (2010) Subtyping of breast cancer by immunohistochemistry to investigate a relationship between subtype and short and long term survival: a collaborative analysis of data for 10,159 cases from 12 studies. PLoS Med 7: e1000279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohmann K, Hennig G, Rogel U, Poremba C, Mueller BM, Fritz P, Stoerkel S, Schaefer KL (2009) RNA extraction from archival formalin-fixed paraffin-embedded tissue: a comparison of manual, semiautomated, and fully automated purification methods. Clin Chem 55: 1719–1727 [DOI] [PubMed] [Google Scholar]
- Carey LA, Dees EC, Sawyer L, Gatti L, Moore DT, Collichio F, Ollila DW, Sartor CI, Graham ML, Perou CM (2007) The triple negative paradox: primary tumor chemosensitivity of breast cancer subtypes. Clin Cancer Res 3: 2329–2334 [DOI] [PubMed] [Google Scholar]
- Chen C, Li M, Yang H, Chai H, Fisher W, Yao Q (2005) Roles of thymosins in cancers and other organ systems. World J Surg 29: 264–270 [DOI] [PubMed] [Google Scholar]
- Denkert C, Loibl S, Kronenwett R, Budczies J, von Törne C, Nekljudova V, Darb-Esfahani S, Solbach C, Sinn BV, Petry C, Müller BM, Hilfrich J, Altmann G, Staebler A, Roth C, Ataseven B, Kirchner T, Dietel M, Untch M, von Minckwitz G (2012) RNA-based determination of ESR1 and HER2 expression and response to neoadjuvant chemotherapy. Ann Oncol (in press) [DOI] [PubMed]
- Denkert C, Loibl S, Noske A, Roller M, Müller BM, Komor M, Budczies J, Darb-Esfahani S, Kronenwett R, Hanusch C, von Törne C, Weichert W, Engels K, Solbach C, Schrader I, Dietel M, von Minckwitz G (2010) Tumor-associated lymphocytes as an independent predictor of response to neoadjuvant chemotherapy in breast cancer. J Clin Oncol 28: 105–113 [DOI] [PubMed] [Google Scholar]
- Fasching PA, Heusinger K, Haeberle L, Niklos M, Hein A, Bayer CM, Rauh C, Schulz-Wendtland R, Bani MR, Schrauder M, Kahmann L, Lux MP, Strehl JD, Hartmann A, Dimmler A, Beckmann MW, Wachter DL (2011) Ki67, chemotherapy response, and prognosis in breast cancer patients receiving neoadjuvant treatment. BMC Cancer 11: 486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldhirsch A, Wood WC, Gelber RD, Coates AS, Thürlimann B, Senn HJ (2007) Progress and promise: highlights of the international expert consensus on the primary therapy of early breast cancer. Ann Oncol 18: 1133–1144 [DOI] [PubMed] [Google Scholar]
- Hudis CA, Gianni L (2011) Triple-negative breast cancer: an unmet medical need. Oncologist 16(Suppl 1): 1–11 [DOI] [PubMed] [Google Scholar]
- Huober J, von Minckwitz G, Denkert C, Tesch H, Weiss E, Zahm DM, Belau A, Khandan F, Hauschild M, Thomssen C, Högel B, Darb-Esfahani S, Mehta K, Loibl S (2010) Effect of neoadjuvant anthracycline-taxane-based chemotherapy in different biological breast cancer phenotypes: overall results from the GeparTrio study. Breast Cancer Res Treat 124: 133–140 [DOI] [PubMed] [Google Scholar]
- Kaufmann M, von Minckwitz G, Mamounas EP, Cameron D, Carey LA, Cristofanilli M, Denkert C, Eiermann W, Gnant M, Harris JR, Karn T, Liedtke C, Mauri D, Rouzier R, Ruckhaeberle E, Semiglazov V, Symmans WF, Tutt A, Pusztai L (2012) Recommendations from an International Consensus Conference on the Current Status and Future of Neoadjuvant Systemic Therapy in Primary Breast Cancer. Ann Surg 19: 1508–1516 [DOI] [PubMed] [Google Scholar]
- Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, Symmans WF, Gonzalez-Angulo AM, Hennessy B, Green M, Cristofanilli M, Hortobagyi GN, Pusztai L (2008) Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol 26: 1275–1281 [DOI] [PubMed] [Google Scholar]
- Mannherz HG, Hannappel E (2009) The beta-thymosins: intracellular and extracellular activities of a versatile actin binding protein family. Cell Motil Cytoskeleton 66: 839–851 [DOI] [PubMed] [Google Scholar]
- McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2006) Statistics Subcommittee of NCI-EORTC Working Group on Cancer Diagnostics. Reporting recommendations for tumor MARKer prognostic studies (REMARK). Breast Cancer Res Treat 2: 229–235 [DOI] [PubMed] [Google Scholar]
- Modlich O, Prisack HB, Munnes M, Audretsch W, Bojar H (2005) Predictors of primary breast cancers responsiveness to preoperative epirubicin/cyclophosphamide-based chemotherapy: translation of microarray data into clinically useful predictive signatures. J Transl Med 3: 32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller BM, Kronenwett R, Hennig G, Euting H, Weber K, Bohmann K, Weichert W, Altmann G, Roth C, Winzer KJ, Kristiansen G, Petry C, Dietel M, Denkert C (2011) Quantitative determination of estrogen receptor, progesterone receptor and HER2 mRNA in formalin-fixed paraffin-embedded tissue - a new option for predictive biomarker assessment in breast cancer. Diagn Mol Pathol 20: 1–10 [DOI] [PubMed] [Google Scholar]
- Pentheroudakis G, Kalogeras KT, Wirtz RM, Grimani I, Zografos G, Gogas H, Stropp U, Pectasides D, Skarlos D, Hennig G, Samantas E, Bafaloukos D, Papakostas P, Kalofonos HP, Pavlidis N, Fountzilas G (2009) Gene expression of estrogen receptor, progesterone receptor and microtubule-associated protein Tau in high-risk early breast cancer: a quest for molecular predictors of treatment benefit in the context of a Hellenic Cooperative Oncology Group trial. Breast Cancer Res Treat 116: 131–143 [DOI] [PubMed] [Google Scholar]
- Perou CM (2011) Molecular stratification of triple-negative breast cancers. Oncologist 16(Suppl 1): 61–70 [DOI] [PubMed] [Google Scholar]
- Rody A, Karn T, Gätje R, Kourtis K, Minckwitz G, Loibl S, Bauerfeind I, Hilfrich J, Eidtmann H, Gerber B, Hanusch C, Kühn T, du Bois A, Blohmer JU, Thomssen C, Dan Costa S, Jackisch C, Kaufmann M, Mehta K, Untch M (2007) Gene expression profiling of breast cancer patients treated with docetaxel, doxorubicin, and cyclophosphamide within the GEPARTRIO trial: HER-2, but not topoisomerase II alpha and microtubule-associated protein tau, is highly predictive of tumor response. Breast 16: 86–93 [DOI] [PubMed] [Google Scholar]
- Simon RM, Paik S, Hayes DF (2009) Use of archived specimens in evaluation of prognostic and predictive biomarkers. J Natl Cancer Inst 101: 1446–1452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stege A, Priebsch A, Nieth C, Lage H (2004) Stable and complete overcoming of MDR1/P-glycoprotein-mediated multidrug resistance in human gastric carcinoma cells by RNA interference. Cancer Gene Ther 11: 699–706 [DOI] [PubMed] [Google Scholar]
- Untch M, Fasching PA, Konecny GE, Hasmüller S, Lebeau A, Kreienberg R, Camara O, Müller V, du Bois A, Kühn T, Stickeler E, Harbeck N, Höss C, Kahlert S, Beck T, Fett W, Mehta KM, von Minckwitz G, Loibl S (2011) Pathologic complete response after neoadjuvant chemotherapy plus trastuzumab predicts favorable survival in human epidermal growth factor receptor 2-overexpressing breast cancer: results from the TECHNO trial of the AGO and GBG study groups. J Clin Oncol 25: 3351–3357 [DOI] [PubMed] [Google Scholar]
- von Minckwitz G, Blohmer JU, Raab G, Löhr A, Gerber B, Heinrich G, Eidtmann H, Kaufmann M, Hilfrich J, Jackisch C, Zuna I, Costa SD (2005) In vivo chemosensitivity-adapted preoperative chemotherapy in patients with early-stage breast cancer: the GEPARTRIO pilot study. Ann Oncol 16: 56–63 [DOI] [PubMed] [Google Scholar]
- von Minckwitz G, Kümmel S, Vogel P, Hanusch C, Eidtmann H, Hilfrich J, Gerber B, Huober J, Costa SD, Jackisch C, Loibl S, Mehta K, Kaufmann M (2008a) Intensified neoadjuvant chemotherapy in early-responding breast cancer: phase III randomized GeparTrio study. J Natl Cancer Inst 100: 552–562 [DOI] [PubMed] [Google Scholar]
- von Minckwitz G, Kümmel S, Vogel P, Hanusch C, Eidtmann H, Hilfrich J, Gerber B, Huober J, Costa SD, Jackisch C, Loibl S, Mehta K, Kaufmann M (2008b) Neoadjuvant vinorelbine-capecitabine versus docetaxel-doxorubicin-cyclophosphamide in early nonresponsive breast cancer: phase III randomized GeparTrio trial. J Natl Cancer Inst 100: 542–551 [DOI] [PubMed] [Google Scholar]
- von Minckwitz G, Loibl S, Maisch A, Untch M (2011a) Lessons from the neoadjuvant setting on how best to choose adjuvant therapies. Breast 20(Suppl 3): S142–S145 [DOI] [PubMed] [Google Scholar]
- von Minckwitz G, Rezai M, Loibl S, Fasching PA, Huober J, Tesch H, Bauerfeind I, Hilfrich J, Eidtmann H, Gerber B, Hanusch C, Kühn T, du Bois A, Blohmer JU, Thomssen C, Dan Costa S, Jackisch C, Kaufmann M, Mehta K, Untch M (2010) Capecitabine in addition to anthracycline- and taxane-based neoadjuvant treatment in patients with primary breast cancer: phase III GeparQuattro study. J Clin Oncol 28: 2015–2023 [DOI] [PubMed] [Google Scholar]
- von Minckwitz G, Untch M, Nüesch E, Loibl S, Kaufmann M, Kümmel S, Fasching PA, Eiermann W, Blohmer JU, Costa SD, Mehta K, Hilfrich J, Jackisch C, Gerber B, du Bois A, Huober J, Hanusch C, Konecny G, Fett W, Stickeler E, Harbeck N, Müller V, Jüni P (2011b) Impact of treatment characteristics on response of different breast cancer phenotypes: pooled analysis of the German neo-adjuvant chemotherapy trials. Breast Cancer Res Treat 125: 145–156 [DOI] [PubMed] [Google Scholar]
- Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, Allred DC, Cote RJ, Dowsett M, Fitzgibbons PL, Hanna WM, Langer A, McShane LM, Paik S, Pegram MD, Perez EA, Press MF, Rhodes A, Sturgeon C, Taube SE, Tubbs R, Vance GH, van de Vijver M, Wheeler TM, Hayes DF (2007) American Society of Clinical Oncology; College of American Pathologists. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol 25: 118–145 [DOI] [PubMed] [Google Scholar]