Abstract
Regulation of Armadillo (Arm) protein levels through ubiquitin-mediated degradation plays a central role in the Wingless (Wg) signaling. Although zeste-white3 (Zw3)-mediated Arm phosphorylation has been implicated in its degradation, we have recently shown that casein kinase Iα (CKIα) also phosphorylates Arm and induces its degradation. However, it remains unclear how CKIα and Zw3, as well as other components of the Arm degradation complex, regulate Arm phosphorylation in response to Wg. In particular, whether Wg signaling suppresses CKIα- or Zw3-mediated Arm phosphorylaytion in vivo is unknown. To clarify these issues, we performed a series of RNA interference (RNAi)-based analyses in Drosophila S2R+ cells by using antibodies that specifically recognize Arm phosphorylated at different serine residues. These analyses revealed that Arm phosphorylation at serine-56 and at threonine-52, serine-48, and serine-44, is mediated by CKIα and Zw3, respectively, and that Zw3-directed Arm phosphorylation requires CKIα-mediated priming phosphorylation. Daxin stimulates Zw3- but not CKIα-mediated Arm phosphorylation. Wg suppresses Zw3- but not CKIα-mediated Arm phosphorylation, indicating that a vital regulatory step in Wg signaling is Zw3-mediated Arm phosphorylation. In addition, further RNAi-based analyses of the other aspects of the Wg pathway clarified that Wg-induced Dishevelled phosphoylation is due to CKIα and that presenilin and protein kinase A play little part in the regulation of Arm protein levels in Drosophila tissue culture cells.
The Wnt/Wingless (Wg) signaling pathway is essential for many aspects of animal development, and mutations in components of the Wnt pathway are oncogenic (reviewed in references 3, 30, and 45). Wnts are secreted glycoproteins that exert their effects on neighboring cells by binding to a receptor protein complex consisting of the Frizzled (Fz) transmembrane receptor family and the single-pass transmembrane proteins of the low density lipoprotein receptor-related protein family. A variety of studies have set a general framework for the Wnt/Wg pathway and revealed that components of this pathway are structurally and functionally conserved in various species. However, it remains unclear how the Wnt signal is transduced from receptors to downstream components such as Dvl/Dishevelled (Dsh). In this pathway, the stabilization of cytoplasmic pools of β-catenin/Armadillo (Drosophila homolog of β-catenin, Arm) is a key regulatory step.
Several components of this pathway, including Dvl/Dsh, glycogen synthase kinase-3β (GSK-3β)/zeste-white3 (Zw3), β-catenin/Arm, adenomatous polyposis coli (APC) protein/Dapc, and protein phosphatase 2A (PP2A), have been shown to form a large multimeric protein complex on the scaffold protein Axin/Daxin, and Wnt/Wg-regulated phosphorylation of β-catenin/Arm known to occur in this complex (6, 11, 20, 31, 32, 33, 47). In the absence of Wnt/Wg signaling, β-catenin/Arm is phosphorylayted at four conserved serine (Ser) and threonine (Thr) residues at the N terminus of the protein (1, 2, 22, 50), and phosphorylated β-catenin/Arm is targeted to the ubiquitin-proteasome pathway for degradation via β-Trcp/Slimb, a subunit of the E3 ubiquitin ligase (1). Moreover, the importance of β-catenin phosphorylation in controlling degradation has been inferred from mutations at four conserved Ser and Thr, residues of β-catenin in tumor cells (reviewed in reference 30). Upon Wnt/Wg stimulation, Dvl/Dsh, by an unknown mechanism, inhibits β-catenin/Arm phosphorylation, thereby allowing it to accumulate in the cytoplasm (41). β-Catenin/Arm then forms a complex with the Tcf-Lef/D-Tcf family of transcription factors and activates the transcription of specific target genes.
A group of GSK-3β substrates are formed by prior phosphorylation from other kinases, an event known as “priming,” to generate the sequence S/T-X-X-X-S/T-PO4 where S/T corresponds to Ser or Thr and X refers to any other residues (reviewed in reference 10). Indeed, recent crystallographic studies of GSK-3β have revealed the existence of a phosphate-binding site, which explains the unique specificity for primed substrates and inactivation by phosphorylation (7, 13). Because suppression of GSK-3β/Zw3 led to an elevation in β-catenin/Arm levels, and the four conserved Ser and Thr residues at the N terminus of β-catenin/Arm match the consensus target sequences for GSK-3β/Zw3 phosphorylation, GSK-3β/Zw3 has been assumed to phosphorylate these sites (28, 36, 50). On the other hand, by using double-stranded RNA-mediated interference (RNAi), we have demonstrated that casein kinase Iα (CKIα) stimulates Arm degradation, thus functioning as a negative regulator of Wg signaling, and that CKIα phosphorylates Arm at Ser56, one of the four conserved Ser and Thr residues in vitro (48).
It has long been believed that β-catenin/Arm did not require a priming phosphate and may rely on high-affinity interactions in a multiprotein complex with GSK-3β/Zw3 (13). Indeed, GSK-3β-mediated phosphorylation of β-catenin is stimulated 20,000-fold in the presence of Axin (6). Nonetheless, it has recently been shown in Xenopus embryos and mammalian cells that the GSK-3β-mediated sequential phosphorylation of β-catenin at Thr41, Ser37, and Ser33 requires CKI-mediated priming phosphorylation at Ser45 (2, 22; reviewed in reference 9). Our finding that CKIα-mediated phosphorylation of Arm at Ser56 is essential for its degradation is consistent with the notion that this phosphorylated Ser56 could function as a priming phosphate for the Zw3-dependent phosphorylation of Arm at Thr52, Ser48, and Ser44 (48).
The presenilins (PS) are structurally and functionally well-conserved polytopic proteins with six to eight transmembrane domains that are required for the regulated intramembrane proteolysis of the amyloid precursor protein and the Notch receptors and thereby associated with familial Alzheimer's disease and Notch signaling (16, 27, 38). On the other hand, recent reports implicate PS as a negative regulator of β-catenin/Arm independent of Wnt/Wg signaling. A PS1 (one of the two PS genes in mice) deficiency in primary fibroblasts stabilizes levels of free β-catenin (16). In a genetic screen, Drosophila presenilin (DPS) was identified as a negative modifier of Wg signaling and a DPS deficiency resulted in an increase in cytoplasmic Arm at the expense of its adherens junction-associated pools (27). Moreover, using mammalian tissue culture cells, Kang et al. have found that PS1 functions as a scaffold that couples β-catenin phosphorylation through two sequential kinase activities, a protein kinase A (PKA)-mediated priming phosphorylation at Ser45 and subsequent GSK-3β-dependent phosphorylation at Thr41, Ser37, and Ser33 and that this PS1-dependent control of β-catenin phosphorylation and degradation occurs outside of the Wnt-regulated Axin complex (16).
Although the Wnt/Wg pathway has been intensively investigated, it is not yet verified in Drosophila whether CKIα and Zw3 phosphorylate Arm cooperatively in the suspected manner. In addition, it remains unclear and controversial whether CKI-mediated priming phosphorylaytion of β-catenin/Arm is affected by Wnt/Wg signaling (2, 16, 22), how exactly components of the Arm destruction complex (Daxin, Dsh, and PP2A) regulate distinct Arm phosphorylations in response to Wg (20, 21, 31, 32, 41, 44), and whether two isoforms of CKI (CKIα and CKIɛ) exert similar or different functions in Wnt/Wg signaling (2, 15, 17, 18, 23, 26, 29, 33, 34, 35, 48).
In the present study, we performed a series of RNAi-based loss-of-function analyses in Drosophila tissue culture cells to address these issues. These analyses revealed the following: (i) Zw3-mediated progressive phosphorylation of Arm at Thr52, Ser48, and Ser44 absolutely requires CKIα-mediated priming phosphorylation at Ser56; (ii) Daxin markedly stimulates Zw3-mediated but not CKIα-mediated Arm phosphorylation; (iii) Wg signaling suppresses Zw3-mediated Arm phosphorylation, whereas CKIα-mediated priming phosphorylation of Arm is not affected, indicating that CKIα-mediated Arm phosphorylation is constitutive and not modulated by Wg signaling; (iv) Wg-induced phosphorylation of Dsh is abolished by CKIα-RNAi, suggesting that CKIα is responsible for the Dsh phosphorylation evoked by Wg treatment; (v) and finally, in contrast to earlier reports, DPS- and PKA-RNAi have little effect on Arm protein levels in Drosophila cultured cells, suggesting that the proposed role of DPS and PKA in Arm degradation must be revaluated.
MATERIALS AND METHODS
Cell cultures and transfections.
Drosophila S2R+ cells, a subline of Schneider S2 cells which respond to Wingless signaling, and mouse L cells were cultured as described previously (19, 40, 46). The Kc cell line and ML-DmBG2cl6 (abbreviated as BG2), a cell line established from the central nervous system of Drosophila third-instar larvae (39) were maintained in Schneider insect medium supplemented with 10% heat-inactivated fetal bovine serum. Expression plasmids were introduced into S2R+ cells by using Effectine reagent (Qiagen). Stable S2R+ transfectants that expressed the Zw3, CKIα, CKIɛ, or PKA catalytic subunit, tagged with the hemagglutinin (HA) sequences, Flag-tagged-Daxin, and myc-tagged-Dsh were generated with pMK33-based-vectors as described previously (19, 47, 48, 49). Expression of the transfected genes was induced by adding 0.5 mM CuSO4.
Immunoblot analyses and antibodies.
The cell lysate was subjected to Western blot analysis as described previously (49). For phosphorylation analysis of β-catenin and Arm, two anti-β-catenin phosphopeptide antibodies (Cell Signaling Technology) were used; anti-phospho-The41/Ser45- and anti-phospho-Ser33/37/Thr41-β-catenin. To analyze the phosphorylation status of Zw3 at Ser9, anti-phospho-Ser21-GSK-3α and anti-phospho-Ser9-GSK-3β (both from Cell Signaling Technology) were used. The rabbit polyclonal anti-Par-1 antibody is a gift from D. St Johnston (Wellcome/CRC Institute and University of Cambridge). The other antibodies used were as follows, and their origins were described previously (19, 38, 46, 47, 48, 49): the mouse monoclonal anti-Arm antibody N2-7A1; the rat polyclonal anti-Dsh region I antibody; the rat monoclonal anti-Drosophila-α-catenin antibody DCAT-1; the rat antibody against the C-terminal region of Daxin; the mouse monoclonal anti-Zw3 antibody; the rabbit polyclonal anti-Drosophila protein phosphatase 2A catalytic subunit antibody; the rabbit polyclonal anti-human CKIɛ antibody; the rabbit polyclonal anti-DPS antibody; the mouse monoclonal anti-β-catenin antibody; the mouse monoclonal anti-GSK-3β antibody; the rabbit polyclonal anti-glutathione S-transferase (GST) antibody Z-5 (Santa Cruz Biotechnology); the rat monoclonal anti-α-catenin antibody α18; the rabbit polyclonal anti-Axin antibody H-98 (Santa Cruz Biotechnology); the mouse monoclonal anti-Flag antibody M2 (Sigma); the mouse monoclonal anti-HA antibody 12CA5 (Roche); the mouse monoclonal anti-human c-myc antibody 9E10 (Calbiochem); and peroxidase-conjugated secondary antibodies against mouse immunoglobulin G (IgG; Bio-Rad), rat IgG (Santa Cruz Biotechnology), and rabbit IgG (New England Biolabs). The blots were visualized with enhanced chemiluminescence reagent (Amersham Pharmacia Biotech).
Wg and Wnt3A treatment of cells.
Details of Wg treatment of S2R+ cells were described previously (40, 47). Wnt3A treatment of mouse L cells and the method used for the preparation of conditioned medium (CM) from Wnt-3A-producing L cells or control L cells were described previously (19). Briefly, confluent S2R+ cell cultures and subconfluent cultures of L cells, both preincubated or not with lactacystin (20 μM) for 4 h, were cocultivated with S2-HS-wg (S2 cells expressing Wg under the control of a heat shock promoter) or control S2 cells and incubated in Wnt-3A-CM or Control-CM, respectively, for a further 1 or 3 h in the presence or absence of lactacystin. Then the cell extracts for sodium dodecyl sulfate-polyacrylamide gel electrophoresis were prepared. Since Kc cells are not adherent, Wg treatment of Kc cells was performed as follows. Kc cell suspensions preincubated or not with lactacystin were cocultivated with confluent cultures of S2-HS-Wg or S2 cells in each well of a six-well plate. After Wg treatment, Kc cells were recovered and subjected to Western blot analysis.
His6-tagged Arm proteins and GST-Arm fusion proteins.
GST-Arm fusion proteins containing the N-terminal 39 amino acids (from codons 37 to 75) of the wild-type and mutant forms of Arm and various myc-tagged Arm cDNA in pBluescript II were described previously (48). Using the pQE 30 (Qiagen) vector, His6 tag was added to the N terminus of wild-type and mutant forms of the myc-tagged Arm, and the Arm proteins doubly tagged with His6 and myc epitopes thus generated were purified as described previously (48).
dsRNA production and RNAi procedures.
The RNAi experiments in Drosophila S2R+, BG2, and Kc cells were performed as described previously (106 cells were incubated with 15 μg of double-stranded RNA [dsRNA] in each well of a six-well plate [4]). The S2R+ cell cultures preincubated with various dsRNAs for 30 h were further incubated for 6 h in the presence or absence of 20 μM lactacystin and then harvested. Primer sequences used to generate dsRNAs were as follows: Daxin, DDBJ/EMBL/GenBank accession no. AF086811, sense-primer (S-P) 221-241, antisense primer (AS-P) 882-901; Par-1, accession no. AF258462, S-P 974-992, AS-P 1619-1637; Drosophila PKA catalytic subunit, accession no. M18655, S-P 931-948, AS-P 1613-1630; Drosophila PP2A-C, accession no. X55199, S-P 189-209, AS-P 882-902; Drosophila presenilin (DPS), accession no. U78084, S-P 83-97, AS-P 769-783; and CKIɛ (new version), accession no. AF055583, S-P 950-975, AS-P 1361-1387. Primers to generate LacZ-, CKIα-, Dα-catenin-, Zw3-, and casein kinase II-dsRNA, as well as the old version of CKIɛ-dsRNA, were described previously (48).
Expression constructs.
pMK33, pMK-CKIαHA, pMK-CKIɛHA, pMK-Zw3HA, pMK-DaxinFlag, and pMK-DshMyc plasmids were described previously (47, 48, 49). To express wild-type and mutant forms of myc-tagged Arm under the control of the Drosophila actin 5C gene promoter, various myc-tagged Arm cDNAs in pBluescript II were digested with BamHI and blunted, and the resulting 2.7-kb fragments were inserted into the EcoRV site of pAC5.1/V5-His-C vector (Invitrogen). To add the HA epitope to the carboxyl terminus of full-length PKA-C, the entire coding sequence of PKA-C was amplified by reverse transcription-PCR with the single-stranded cDNA synthesized from Drosophila embryonic poly(A)+ RNA and the following set of primers: S-P with a XhoI site (5-TAGCTCGAGGAGCAGCTAGCCAGGATGGACAAG-3′) and AS-P with a SpeI site (5′-CAGACTAGTGTCCGCGATCAGGGGCTTGCCGTT-3′). The PKA-C-reverse transcription-PCR product was double-digested with XhoI and SpeI before being cloned into the XhoI-SpeI-cleaved pMK33-HA. The resulting plasmid was named pMK-PKA-HA.
In vitro kinase assay.
The stable pMK-CKIαHA or pMK-Zw3HA transfectants were treated with CuSO4 for 14 h. From the lysates of these cells, HA-tagged CΚIα or HA-tagged Zw3 was immunoprecipitated with the rabbit polyclonal anti-HA antibody (Y-11; Santa Cruz Biotechnology) and protein A-Sepharose (Amersham Pharmacia Biotech). The HA-immunoprecipitate from naive S2R+ cells was used as a negative control. The immune complexes were washed with lysis buffer and with kinase buffer (10 mM HEPES [pH 7.5], 75 mM KCl, 5 mM MgCl2, 20 μM ATP, and 1 mM dithiothreitol) before being suspended in 60 μl of kinase buffer containing either 10 μg of various GST-Arm proteins or 10 μg of various His6-tagged Arm proteins. Ten minutes after incubation at 30°C, 5 μl of the reaction mixture was taken and subjected to Western blot analysis.
The sequential phosphorylation of Arm with CKI and GSK-3β was performed as follows: 3 μg of the wild-type or S56A-mutant form of Arm, which was doubly tagged with His6 and myc epitopes, was incubated in 40 μl of kinase buffer containing 125 U of CKI for 10 min at 30°C. Then, 360 μl of immunoprecipitation buffer (10 mM Tris-HCl [pH 7.5], 1% NP-40, 150 mM NaCl, 20 mM β-glycerophosphate, 0.5 mM Na3VO4), 5 μg of goat anti-myc antibody (A-14; Santa Cruz Biotechnology), and 50 μl of protein G-Sepharose (Amersham Pharmacia Biotech) were added, and the mixture was incubated for 1 h at 4°C. Next, the immune complexes were washed twice with immunoprecipitation buffer and once with kinase buffer before being suspended in 40 μl of kinase buffer containing 150 U of GSK-3β. Ten minutes after incubation at 30°C, 5 μl of the reaction mixture was taken and subjected to Western blot analysis.
RESULTS
Characterization of anti-β-catenin phosphopeptide antibodies in Drosophila system.
Recent studies showed that antibodies specifically recognizing either Ser45-phosphorylated β-catenin or β-catenin phosphorylated at Ser33, Ser37, and Thr41 are quite useful for analyzing how Wnt signaling regulates β-catenin phosphorylation in vivo (2, 16, 22). On the other hand, except our previous study suggesting cooperation of CKIα with Zw3 in Arm phosphorylation (48), there has been no study on how Arm phosphorylation is regulated in vivo. Alignment of the N-terminal amino acid sequences of Arm and β-catenin (Fig. 1A) suggests that antibodies for phosphorylated β-catenin could cross-react with corresponding phosphorylated Arm and would become valuable tools for the analysis of Arm phosphorylation in Drosophila. Thus, we first clarified the specificity of the commercial phospho-41/45-β-catenin and phospho-33/37/41-β-catenin antibodies (data not shown) and confirmed the results described by Kang et al. (16): the phospho-41/45 antibody only recognized phospho-45; thus, it should be designated phospho-45-β-catenin antibody. Moreover, neither phospho-41/45 nor phospho-33/37/41 antibody reacted with the β-catenin peptide singly phosphorylated at Thr41 or the unphosphorylated recombinant GST-β-catenin fusion protein, indicating that there is no overlap in antigen recognition between the two phospho-specific antibodies.
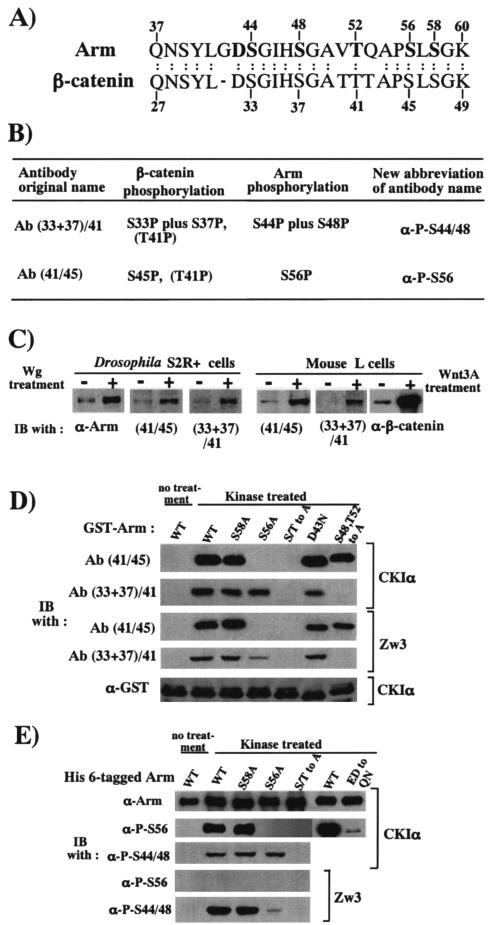
FIG. 1.
Anti-phospho-Thr41/Ser45- and anti-phospho-Ser33/37/Thr41-β-catenin antibody recognizes phospho-Ser56 Arm and phospho-Ser44/48 Arm, respectively. (A) Alignment of conserved N-terminal amino acid sequences of Arm and β-catenin. The Arm sequence from codons 37 to 60 and the β-catenin sequence from codons 27 to 49 are shown. Ser, Thr, and Asp residues mutated in some constructs are shown with boldface. (B) Antibodies for phosphorylated β-catenin cross-react with corresponding phosphorylated Arm counterparts. T41P in parentheses indicates that, in contrast to the manufacturer's descriptions, the batches of the antibodies used in the present study did not recognize T41P. (C) Phosphorylated β-catenin in L cells and putative phosphorylated Arm in S2R+ cells accumulates in response to Wnt3A and Wg treatment, respectively. (D and E) Immunoblot analyses to confirm cross-reactivities of antibodies for phosphorylated β-catenin with phosphorylated Arm proteins and to determine phosphorylated amino acid sequences in the N-terminal region of Arm which were recognized by anti-phospho-β-catenin antibodies. (D) Various GST-Arm fusion proteins treated with CKIα or ZW3 were subjected to Western blot analysis with the antibodies indicated. In the S/T-to-A mutant, the Sers at codons 44, 48, and 56 and the Thr at codon 52 were changed to Ala. A GST fusion protein with the wild-type Arm sequence not treated with kinase was used as a negative control. (E) The wild-type and mutated Arm proteins doubly taggedwith the His6 and myc epitopes were phosphorylated by CKIα or Zw3 and subjected to Western blot analysis with the antibodies indicated. In the ED-to-QN mutant, a stretch of acidic amino acids (E and D) was replaced with Q and N (E at 61, 63, 64, and 66 to Q and D at 62 to N). Wild-type Arm not treated with kinase was used as a negative control.
Wnt3A treatment for 3 h elevated steady-state levels of total, as well as the Ser45- and Ser33/37/Thr41-phosphorylated β-catenin in mouse L cells. Similarly, Wg treatment for 3 h elevated the amount of total Arm, as well as putative phosphorylated Arm species, which were recognized by the phospho-41/45- and phospho-33/37/41-β-catenin antibodies in S2R+ cells (Fig. 1C). Thus, the phospho-specific β-catenin antibodies appeared to cross-react with the corresponding phosphorylated Arm.
To confirm the phosphorylated amino acid sequences in Arm, which are recognized by the phospho-41/45- and phospho-33/37/41-β-catenin antibodies, various GST-Arm fusion proteins (Fig. 1D) or His6-tagged full-length Arm proteins (Fig. 1E) with wild-type or mutant forms of Arm sequences were phosphorylated by Zw3 or CKIα, and their reactivities with the phospho-specific β-catenin antibodies were examined by Western blotting. As expected, GST-Arm fusion protein with the wild-type Arm sequence was recognized by both phospho-specific β-catenin antibodies upon phosphorylation with CKIα or Zw3. The reactivity of these antibodies with GST-Arm fusion protein was not affected by alanine (Ala) and asparagine substitutions at Ser58 and Asp43, respectively, but was completely abolished by Ala substitutions at Ser44, Ser48, Thr52, and Ser56. Moreover, the reactivity of the phospho-41/45- but not phospho-33/37/41-β-catenin-antibody was abolished by an Ala substitution at Ser56. Conversely, the reactivity of the phospho-33/37/41- but not phospho-41/45-β-catenin antibody was abolished by Ala substitutions at Ser48 and Thr52. These results are consistent with the notion that phosphorylated Ser44 and phosphorylated Ser48 of Arm, together with neighboring residues, constitute the recognition motif for the phospho-33/37/41-β-catenin antibody, while phosphorylated Ser56 of Arm, together with neighboring residues, constitutes that of the phospho-41/45-β-catenin-antibody (phosphorylated Ser residues recognized by these two antibodies are summarized in Fig. 1B). Thus, based on the Arm codon, the phospho-33/37/41- and phospho-41/45-β-catenin antibodies were renamed the anti (α)-phospho-Ser44/48- and α-phospho-Ser56-Arm antibodies, respectively.
Analyses with His6-tagged Arm proteins confirmed the results with the GST-Arm fusion proteins described above, but the following points should be noted. (i) In contrast to GST-Arm fusion protein, Ser56 in His6-tagged Arm was efficiently phosphorylated by CKIα but not by Zw3. (ii) Although both CKIα and Zw3 phosphorylated Ser44 and Ser48 in His6-tagged wild-type-Arm, the Ala substitution at Ser56 severely suppressed Zw3-mediated but not CKIα-mediated phosphorylation at Ser44 and Ser48. (iii) Consistent with our previous report (48), replacement of a stretch of acidic amino acids (E and D, which are conserved in β-catenin and plakoglobin) with Q and N (E at 61, 63, 64, and 66 to Q and D at 62 to N) suppressed CKIα-mediated phosphorylation at Ser56 (Fig. 1E), suggesting that CKIα-mediated phosphorylation of Ser56 requires a neighboring cluster of acidic amino acids (12). In this regard, Marin et al. (24) have recently reported that the sequence downstream from Ser45 of β-catenin (Ser56 of Arm as well) is very similar to a sequence recognized by CKI in nuclear factor for activated T cells 4; the common features include an SLS (Ser-Leu-Ser) motif, followed two to five residues downstream by a cluster of acidic residues. These authors also reported that the substitution of all five acidic residues in β-catenin (E53A, D54A, E55A, D56A, and D58A) eliminated Ser45 phosphorylation by CKI. Interestingly, a search for proteins with similar motifs yielded many important signaling proteins, including APC, Ras GTPase-activating protein 2, tumor suppressor P53 binding protein, and tyrosine-protein kinase receptor Ror2.
CKI functions as a priming kinase for Arm phosphorylation by Zw3.
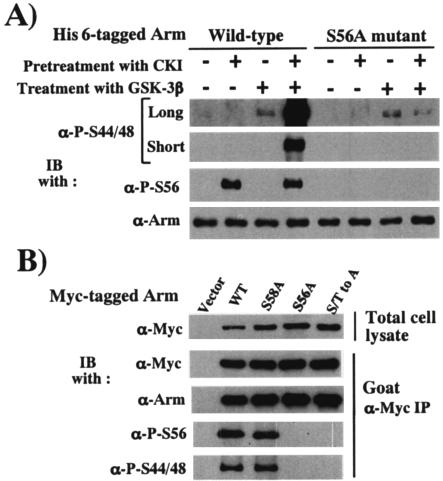
Using the wild-type and Ser56-mutated forms of full-length Arm doubly tagged with His6 and myc epitope at the N terminus and in the C-terminal region, respectively, we sought to determine which of the two kinases, CKIα and Zw3, preferentially phosphorylates Ser56 and two neighboring Ser residues, Ser44 and Ser48, and evaluated whether CKIα really functions as a priming kinase for Zw3 in vitro, where a scaffold protein, Daxin, is not present (Fig. 2A). In this experiment, substrates had to be quantitatively and sequentially phosphorylated by two kinases. For this reason, purified recombinant CKI (rat CKI-δ with a C-terminal truncation and thus structurally similar to CKIα; New England Biolabs) and purified recombinant rabbit GSK-3β (New England Biolabs) were used instead of Drosophila CKIαΗΑ and Zw3HA immunoprecipitates. Western blotting with anti-phospho-Ser44/48 and anti-phospho-Ser56-Arm antibodies demonstrated the following. (i) In these experimental conditions, Ser56 was phosphorylated solely by CKI but not GSK-3β and, conversely, Ser44 and Ser48 were phosphorylated by GSK-3β but not CKI. (ii) Although GSK-3β by itself weakly phosphorylated Ser44 and Ser48 in both wild-type and Ser56-mutated forms of Arm, prior phosphorylation of Ser56 by CKI in wild-type Arm markedly enhanced GSK-3β-mediated phosphorylation of Ser44 and Ser48, whereas in the Ser56-mutated form prior treatment with CKI failed to stimulate GSK-3β-mediated phosphorylation (Fig. 2A). These in vitro experiments with recombinant CKI and GSK-3β strongly suggested that CKIα-mediated priming phosphorylation of Arm at Ser56 enhances Zw3-mediated phosphorylation of Arm at Ser44 and Ser48 in Drosophila even in the absence of Daxin.
FIG. 2.
CKI functions as a priming kinase for Zw3 in Arm phosphorylation. (A) in vitro kinase experiment to demonstrate that priming phosphorylation by CKI facilitates subsequent GSK-3β-mediated Arm phosphorylation. The wild-type or S56A-mutant form of Arm proteins doubly tagged with the His6 and myc epitopes was pretreated or not with CKI, immunoprecipitated with anti-myc antibody, again treated or not with GSK-3β, and then subjected to Western blot analysis. The top and second panels show long and short exposures, respectively of the same immunoblot. (B) Arm phosphorylation at Ser44 and Ser48 requires phosphorylation at Ser56 in vivo. First, 1 μg of pAc5.1/V5-His C vector or various pAcArm-Myc constructs were transfected into S2R+ cells and, after 48 h, the myc-tagged Arm was immunoprecipitated with the goat polyclonal anti-myc antibody in combination with protein G-Sepharose. The cell lysates and the immunoprecipitates were subjected to Western blot analysis with the antibodies indicated.
Next, to clarify whether the priming phosphorylation at Ser56 is required for subsequent phosphorylation of Arm in vivo, we analyzed the effect of the Ala substitution at Ser56 on the phosphorylation at Ser44 and Ser48 in S2R+ cells (Fig. 2B). Because an actin 5C promoter with very strong transcriptional activity was used for the protein expression, all myc-tagged Arm species showed similar expression levels, although the stability of each protein appeared to differ significantly. Thus, among various Arm proteins, almost the same amounts were immunoprecipitated with anti-myc antibody. Although the Ala substitution at Ser58 affected neither Ser56 phosphorylation nor Ser44 plus Ser48 phosphorylation, the Ala substitution occurring at Ser56 not only abolished Ser56 phosphorylation but also Ser44 plus Ser48 phosphorylation in S2R+ cells, indicating that prior phosphorylation at Ser56 is essential for the subsequent phosphorylation of Arm at Ser44 plus Ser48 in vivo.
Priming phosphorylation of Arm at Ser56 and phosphorylation at Ser44 plus Ser48 is mediated by CKIα and Zw3, respectively, in vivo.
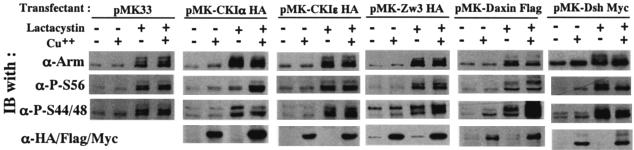
The notion that the priming phosphorylation of Arm at Ser56 and subsequent phosphorylations at Ser44 plus Ser48 are due to CKIα and ZW3, respectively, in vivo remains unproven in Drosophila. To address this issue, we upregulated and downregulated (by overexpression and RNAi, respectively) CKIα, Zw3, and Daxin expression in S2R+ cells and examined its effect on the amount of Arm phosphorylated at Ser56 (P-S56-Arm) and at Ser44 plus Ser48 (P-S44/48-Arm). Since Arm is a protein with a rapid turnover, these analyses were performed in S2R+ cells treated or not with lactacystin, a specific inhibitor of proteasome. Actually, only in the presence of lactacystin were the effects of overexpression of CKIα, ZW3, and Daxin prominent (Fig. 3). Overexpression of CKIα elevated P-S56-Arm levels but not P-S44/48-Arm protein levels. Conversely, overexpression of Zw3 elevated P-S44/48-Arm but not P-S56-Arm levels. At least in this system, overexpression of CKIɛ affected neither Ser56 nor Ser44 plus Ser48 phosphorylation. These results suggested that phosphorylation of Arm at Ser56 and at Ser44 plus Ser48 is mediated by CKIα and Zw3, respectively.
FIG. 3.
Effect of overexpression of CKIα, CKIɛ, Zw3, Daxin, and Dsh on Arm phosphorylation status. Stable S2R+ transfectants which could overexpress CKIα, CKIɛ, or Zw3 (all of which are HA tagged), as well as Daxin (Flag tagged) and Dsh (myc tagged), were cultured in the presence or absence of CuSO4 for 8 h. The cultures were then incubated for a further 6 h in the presence or absence of lactacystin (20 μM). Cell lysates were subjected to Western blot analysis with the antibodies indicated. Total cell lysates were used for this analysis because the amount of membrane-bound Arm is very limited (<5%) in lactacystin-treated S2R+ cells.
On the other hand, Daxin overexpression resulted in a marked and slight elevation of P-S44/48-Arm and P-S56-Arm, respectively, in the presence of lactacystin, indicating that Daxin mainly facilitates Zw3-mediated Arm phosphorylation in Drosophila. This result is in striking contrast to earlier reports for Xenopus and the mammalian system that expression of Axin dramatically stimulated β-catenin priming-phosphorylation at Ser45 (2, 22). In the presence of lactacystin, overexpression of Dsh diminished P-S56-Arm, as well as P-S44/48-Arm levels, whereas in the absence of lactacystin, total Arm levels, as well as P-S56- and P-S44/48-Arm levels, increased upon overexpression. These findings are in line with the notion that Dvl/Dsh is a positive regulator of the Wnt/Wg pathway and consistent with the earlier report that in the presence of MG132, a proteasome inhibitor, overexpression of Dvl1 suppressed both priming phosphorylation of β-catenin at Ser45 and GSK-3β-mediated β-catenin phosphorylation at Ser33 plus Ser37 (2).
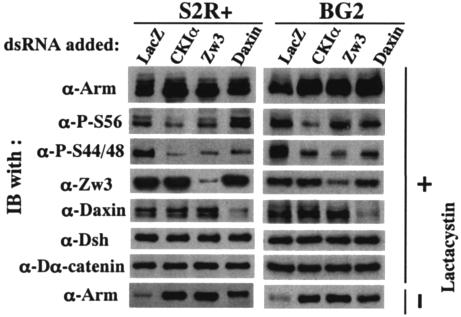
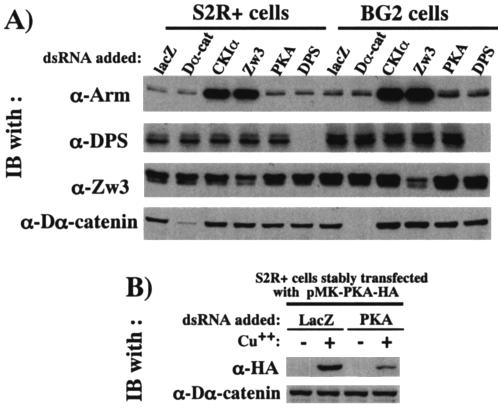
By using RNAi, we further characterized the roles of CKIα, Zw3, and Daxin in the phosphorylation of Arm in a reciprocal way. The S2R+ cells and BG2 cells were used for this analysis and both of these cell lines gave similar results (Fig. 4). Since CKIα, Zw3, and Daxin are all negative regulators of Wg signaling, RNAi of these proteins all resulted in a marked elevation of Arm in the absence of lactacystin, confirming RNAi is working. In the presence of lactacystin, however, CKIα- but not Zw3- or Daxin-RNAi decreased P-S56-Arm, which is consistent with the observation that overexpression of CKIα but not Zw3 or Daxin induced a large increase in P-S56-Arm levels (Fig. 3). On the other hand, CKIα-, Zw3- or Daxin-RNAi all decreased P-S44/48-Arm. We interpreted this as follows. Zw3 appears to be directly responsible for the phosphorylation of Arm at Ser44 plus Ser48, and Daxin brings Arm and Zw3 into close proximity, thereby enhancing the Zw3-mediated phosphorylation of Arm. Thus, it is convincing that Zw3- and Daxin-RNAi suppressed the phosphorylation at Ser44 plus Ser48. Moreover, the decrease in the P-S44/48-Arm caused by CKIα-RNAi appears to indicate that CKIα-RNAi diminished the phosphorylation at Ser56 that was required for subsequent Zw3-mediated phosphorylation at Ser44 plus Ser48. Taken together, a series of gain-of-function analyses in S2R+ cells and loss-of-function analyses in S2R+ and BG2 cells showed that the phosphorylation of Arm at Ser56 and at Ser44 plus Ser48 is mediated by CKIα and Zw3, respectively, in vivo. In addition, Daxin markedly enhances the Zw3- but not CKIα-mediated phosphorylation, indicating that, in Drosophila scaffolding protein, Daxin is not essential for the initial priming phosphorylation but is necessary for bridging the two phosphorylation events.
FIG. 4.
Effect of RNAi-mediated downregulation of CKIα, Zw3, and Daxin expression on Arm phosphorylation. To the S2R+ or BG2 cell cultures preincubated with dsRNA for 30 h, 20 μM lactacystin was added or not, and then the cultures were further incubated for 6 h. Western blots of the cell lysates are shown. The bottom panel shows the effect of various RNAi on steady-state levels of Arm protein in the absence of lactacystin.
CKIα but not CKIɛ destabilizes Arm.
Using S2R+ transfectants overexpressing HA-tagged CKIɛ or CKIα (because good antibody against Drosophila CKIα is not available), we have previously shown that dsRNA corresponding to the catalytic region of CKIɛ selectively suppressed expression of CKIɛ but not of CKIα (48). In addition, although the induction level was much lower than that of CKIα-dsRNA, CKIɛ-dsRNA evoked a significant accumulation of Arm protein (Fig. 5B). Thus, in a previous study, we concluded that CKIɛ by itself destabilizes Arm (48). However, in view of the nucleotide sequence similarity between CKIɛ and CKIα in their kinase-encoding domains, we could not rule out the possibility that in naive S2R+ cells, CKIɛ-dsRNA slightly diminished endogenous CKIα protein levels, thereby moderately inducing the accumulation of Arm. In addition, using a Drosophila cultured cell-based Tcf-dependent reporter assay, Lum et al. recently found that CKIɛ-RNAi has no effect on the Wg pathway, whereas overexpression of CKIɛ elevates the basal activity of the pathway (23).
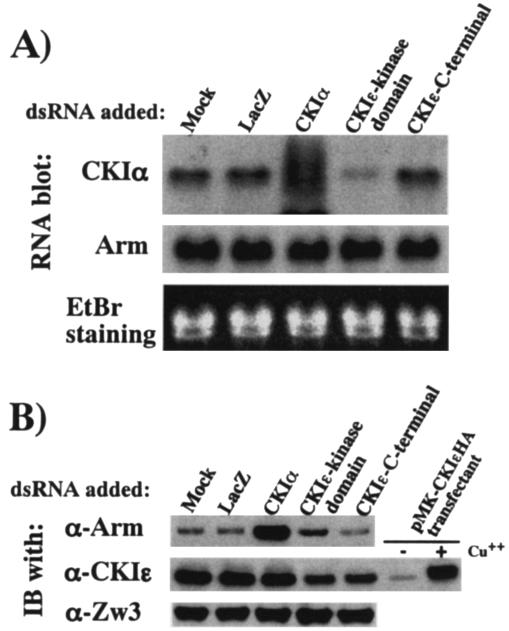
FIG. 5.
Arm protein levels are not affected by CKIɛ-RNAi. (A) Northern blot analysis showing the effect of various dsRNAs on CKIα and Arm mRNA levels. Northern blots were produced as described previously (48). The bottom panel is an ethidium bromide staining of the gels showing that the same amount (10 μg) of total RNA was loaded in each lane. (B) Effect of various dsRNAs on Arm, CKIɛ, and ZW3 protein levels. In the middle blot, lysates from the pMK-CKIɛHA transfectant treated with or without CuSO4 were used to demonstrate that the antibody against human CKIɛ recognizes Drosophila CKIe.
To determine the effect of CKIɛ specific-RNAi on Wg signaling, we generated a new CKIɛ-dsRNA corresponding to the C-terminal extracatalytic region (which is unique for this isoform) of CKIɛ and evaluated its influence on Arm protein levels. Northern analysis revealed that whereas the old version of CKIɛ-dsRNA reduced steady-state levels of CKIα mRNA by 60%, the new one had no effect on CKIα mRNA levels (Fig. 5A). Moreover, in contrast to the old version, this new CKIɛ-dsRNA did not elevate Arm, although both of the CKIɛ-dsRNA reduced CKIɛ protein levels to the same extent (Fig. 5B). Therefore, we corrected the previous report (48) and concluded that only CKIα and not CKIɛ destabilizes Arm. These results are in line with the observation that overexpression of CKIɛ did not stimulate Arm phosphorylation (Fig. 3). The finding that overexpression of CKIɛ had no effect on Arm protein levels (Fig. 3) appears to be inconsistent with the result from the reporter assay that overexpression of CKIɛ elevates basal activity of the Wg pathway (23). However, the report that phosphorylation of Tcf3 by CKIɛ stimulates its binding to β-catenin might explain this discrepancy (18).
Effect of Wg/Wnt3A signaling on CKIα- and Zw3/GSK-3β-mediated phosphorylation of Arm/β-catenin in S2R+, Kc, and L cells.
There is disagreement as to whether Wnt signaling inhibits GSK-3β- or CKI-mediated β-catenin phosphorylation in mammalian cells (2, 16, 22). Moreover, the effect of Wg signaling on Zw3- and CKIα-mediated Arm phosphorylation has not been documented. Therefore, S2R+, Kc, and L cells were each treated with Wg and Wnt3A, and their effects on CKIα- and Zw3/GSK-3β-mediated-phosphorylation of Arm/β-catenin were analyzed in the presence or absence of lactacystin (Fig. 6). In addition, to compare the effects of short and long-term Wg/Wnt treatment, cells treated with Wg/Wnt3A for 1 and 3 h were examined.
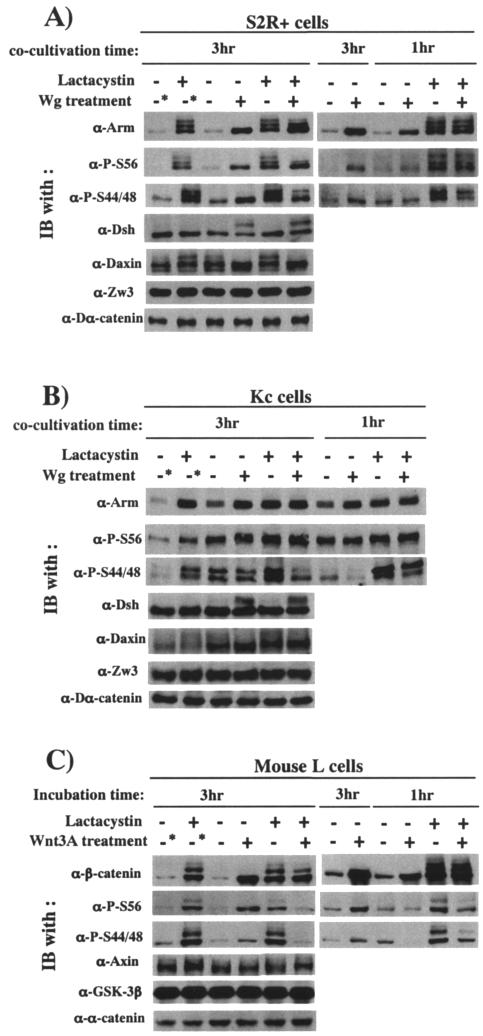
FIG. 6.
Wg/Wnt3A signaling-induced changes in Arm/β-catenin phosphorylation in the presence or absence of lactacystin. Effects of Wg/Wnt3A treatment for 1 and 3 h were compared. Effects of Wg signaling on phosphorylation of Arm, Dsh, and Daxin were analyzed in S2R+ (A) and Kc (B) cells by Western blotting. Asterisks indicate that these cultures were treated in the same way as the others, except that the cocultivation with plain S2 cells for 3 h was replaced with incubation in plain medium (in the presence or absence of lactacystin) for 3 h. Zw3- and Dα-catenin blots were used as loading controls. (C) Effect of Wnt3A signaling on phosphorylation status of β-catenin in L cells. Asterisks indicate that these cultures were treated in the same way as the others, except that they were incubated for a further 3 h in plain medium (in the presence or absence of lactacystin) but not in the control conditioned medium. Total cell lysates were used for this analysis, because L cells do not express cadherin; thus, the amount of membrane-bound β-catenin is very limited. Amounts of P-Ser45- and P-Ser44/48-β-catenin were monitored by using α-P-S56 and α-P-S44/48 antibodies, respectively.
Consistent with the results shown in Fig. 1C, treatment of S2R+, Kc, and L cells with Wg or Wnt3A for 3 h in the absence of lactacystin elevated steady-state levels of total Arm/β-catenin as well as Arm/β-catenin species undergoing CKIα- or Zw3/GSK-3β-mediated phosphorylation. Thus, in cells treated with Wg/Wnt3A for a long time, the steady-state levels of Arm/β-catenin phosphorylated by either CKIα or Zw3/GSK-3β appear to increase dependent on total Arm/β-catenin levels. Consistent with previous reports, Wg signaling induced and suppressed phosphorylation of Dsh (19, 49) and Daxin (32, 44), respectively. Further analysis with cells treated with Wg/Wnt3A for 1 h in the absence of lactacystin, however, revealed the following. Total Arm or β-catenin levels in cells subjected to a 1-h treatment were lower than those in cells subjected to a 3-h treatment; Wnt3A treatment rapidly decreased steady-state levels of Ser33/37-phosphorylated β-catenin (detected by α-P-S44/48 antibody), but those of Ser45-phosphorylated β-catenin were not affected. Similarly, a 1-h Wg treatment markedly decreased P-S44/48-Arm levels in Kc cells, but this was not obvious in S2R+ cells. In S2R+ cells, a 1-h Wg treatment slightly elevated P-S56-Arm protein levels.
Avoiding the effect of Arm/β-catenin turnover, we also performed similar analyses in the presence of lactacystin. As expected, either in S2R+, Kc, and L cells, the total amount of Arm or β-catenin was little affected by Wg or Wnt3A treatment under these conditions. Irrespective of the length of treatment, Wg markedly decreased P-S44/48-Arm protein levels, whereas P-S56-Arm protein levels remained constant in both S2R+ and Kc cells (Fig. 6A and B). In S2R+ cells treated with Wg for 3 h, the Arm with the highest electrophoretic mobility increased at the expense of Arm proteins showing less electrophoretic mobility, which may correspond to Arm species fully phosphorylated by both CKIα and Zw3 and ubiquitinated to various extents, but we noted that the total amount of P-S56-Arm protein was not affected by Wg treatment (Fig. 6A). In accordance with the Wg result, 1 or 3 h of Wnt3A treatment in the presence of lactacystin markedly decreased Ser33/37-phosphorylated β-catenin levels. However, in contrast to the Wg result, but in line with a previous report (2), Wnt3A diminished Ser45-phosphorylated β-catenin levels, and this effect was prominent in the 3-h Wnt3A treatment. This appeared to be related to the earlier reports in mammalian cells that CKIα-mediated β-catenin phosphorylation is highly dependent on Axin (2, 22) and that prolonged Wnt3A treatment decreased total Axin protein levels (44). Against expectation, however, either in the presence or in the absence of lactacystin, 3-h Wnt3A treatment little affected total Axin protein levels (Fig. 6C).
Taken together, Wg signaling suppresses ZW3-mediated Arm phosphorylation at Ser44 and Ser48 but not CKIα-mediated phosphorylation at Ser56. Wnt3A signaling mainly inhibits GSK-3β-mediated β-catenin phosphorylation at Ser33/37, but prolonged Wnt3A treatment also decreases CKIα-mediated β-catenin phosphorylation at Ser45.
Nevertheless, the primary target of Wnt3A signaling is clearly GSK-3β-mediated phosphorylation of β-catenin, because short Wnt3A treatment rapidly decreased Ser33/37- but not Ser45-phosphorylated β-catenin levels in the absence of lactacystin. Kang et al. (16) and Liu et al. (22) have reported basically the same results with Wnt3A-treated L cells and Wnt1-treated Rat2 cells, respectively. On the other hand, Amit et al. have concluded that Wnt regulates the pathway primarily by inhibiting CKI-mediated β-catenin phosphorlation at Ser45, because in the presence of MG132, Wnt3A treatment for 5 h (in L, HeLa, and Jurkat cells) and Dvl overexpression (in 293T cells) both resulted in inhibition of Ser45 and Ser33+Ser37 phosphorylation (2).
Our finding that Wg/Wnt3A signaling suppresses ZW3/GSK-3β-mediated Arm/β-catenin phosphorylation appears to be inconsistent with the observation that treatment of S2R+, Kc, and L cells with Wg or Wnt3A for 3 h (but not 1 h), in the absence of lactacystin, elevated steady-state-levels of P-S44/48-Arm and Ser33/37-phosphorylated β-catenin (Fig. 1C and 6). To address these questions, we analyzed in detail (0, 30, 60, 90, 120, 150, and 180 min after Wg/Wnt3A treatment) the kinetics of Wg/Wnt3A-induced changes in levels of total Arm/β-catenin, as well as Arm/β-catenin species undergoing CKIα- or Zw3/GSK-3β-mediated phosphorylation in S2R+ and L cells (data not shown). The results can be summarized as follows. (i) Consistent with the notion that CKIα-mediated phosphorylation of Arm/β-catenin is constitutive, the amounts of both P-S56-Arm and Ser45-phosphorylated β-catenin were elevated progressively in proportion to the total amount of Arm/β-catenin, which was markedly elevated depending on the length of Wg/Wnt3A treatment. (ii) In contrast, in the lag phase (up to 30 and 60 min after the Wg and Wnt3A treatments, respectively), where increase in total Arm/β-catenin protein levels was little or very limited, the amounts of P-S44/48-Arm and Ser33/37-phosphorylated β-catenin decreased slightly and sharply, respectively (as shown in Fig. 6C). (iii) However, at 60 min after Wg treatment, the amount of P-S44/48-Arm recovered to the same level as that at 0 min (as shown in Fig. 6A). After 90 min, amount of P-S44/48-Arm exceeded that at 0 min, and it elevated gradually as the total amount of Arm protein elevated progressively. (iv) Similarly, at 90 min after Wnt3A treatment, the amount of Ser33/37-phosphorylated β-catenin recovered to the same level as that at 0 min in L cells. After 120 min, the amount of Ser33/37-phosphorylated β-catenin became greater than that at 0 min and elevated gradually in a time-dependent fashion.
Based on these observations, the following explanation is possible for the elevation of Zw3/GSK-3β-phosphorylated Arm/β-catenin in cells treated with Wg/Wnt3A for 3 h: even though Wg/Wnt3A suppresses Zw3/GSK-3β-mediated phosphorylation of Arm/β-catenin, it also induces a marked increase in total amount of Arm/β-catenin protein, a substrate for Zw3/GSK-3β at the later phase of treatment. Because of this secondary effect, long-term Wg/Wnt3A treatment causes a significant increase in the amount of Arm/β-catenin phosphorylated by Zw3/GSK-3β. However, we could not rule out the possibility that Wg/Wnt3A signaling not only regulates Arm/β-catenin phosphorylation but also blocks Arm/β-catenin degradation downstream of the phosphorylation events (at the step of ubiquitination of the phosphorylated Arm/β-catenin, for example), thus causing the elevation of Zw3/GSK-3β-phosphorylated Arm/β-catenin.
Wg signaling does not affect Zw3 phosphorylation at Ser9.
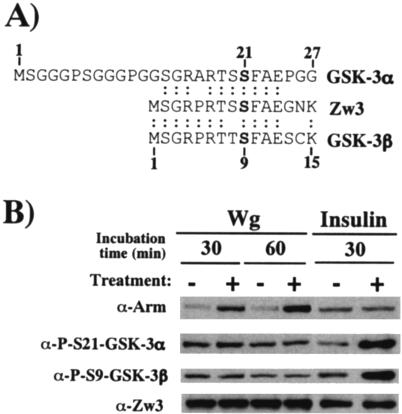
Thus far, we have shown that Wg/Wnt diminishes the amount of Arm/β-catenin undergoing Zw3/GSK-3β-mediated phosphorylation. However, the molecular mechanisms underlining this process are poorly understood. Using GSK-3 substrate peptide, it was shown that the kinase activity of GSK-3/Zw3 was partially inhibited by Wnt-1 in 293, CHO, and C57MG cells (8) and by Wg in mouse 10T1/2 fibroblasts (5) and Drosophila clone-8 cells (32). In spite of these reports, the effect of Wg/Wnt signaling on the kinase activity of Zw3/GSK-3β still remains controversial. In insulin signaling, on the other hand, protein kinase B/Akt-mediated inhibitory phosphorylation at either Ser9 (GSK-3β) or Ser21 (GSK-3α) is well established as a molecular mechanism by which insulin inhibits GSK-3 activity. Surprisingly, the fundamental question to date of whether or not Wg increases the inhibitory N-terminal phosphorylation of Zw3 remains unanswered. Therefore, we analyzed this by using anti-phospho-GSK-3β (Ser9) and anti-phospho-GSK-3α (Ser21) antibodies, both of which are found to cross-react with Zw3 only when its Ser9 is phosphorylated (Fig. 7A). Insulin but not Wg treatment promoted the inhibitory phosphorylation of Zw3 at Ser9 in S2R+ cells, indicating that stimulation of inhibitory phosphorylation of Zw3 is not the mechanism by which Wg suppresses Zw3-mediated-phosphorylation of Arm (Fig. 7B). Similarly, insulin but not Wnt3A induced inhibitory phosphorylation of GSK-3 in L, Rat2, and NIH 3T3 cells (data not shown). These results appear to substantiate reports excluding the involvement of the components of the Akt pathway in Wg signaling: (i) Wg signaling was not affected by inhibitors of phosphatidylinositol 3-kinase (5); (ii) the kinase activity of Drosophila and mammalian Akt was not stimulated by Wg and Wnt-1, respectively (8, 32); and (iii) Wg signaling was not affected by RNAi of Drosophila Akt (23).
FIG. 7.
Zw3 phosphorylation at Ser9 is affected by insulin but not Wg in S2R+ cells. (A) Alignment of conserved N-terminal amino acid sequences of GSK-3α, GSK-3β, and Zw3. The GSK-3α (human) sequence from codons 1 to 27 and Zw3 and GSK-3β (human) sequences from codons 1 to 15 are shown. Ser residues phosphorylated upon insulin treatment are shown in boldface. Anti-phospho-GSK-3α and anti-phospho-GSK-3β antibody detects endogenous levels of GSK-3α and GSK-3β, respectively, only when Ser21 of GSK-3α and Ser9 of GSK-3β are phosphorylated. Both of these antibodies recognize the Ser9-phosphorylated form of Zw3. (B) Wg signaling does not induce Zw3 phosphorylation at Ser9. Confluent S2R+ cell cultures were either cocultivated with S2-HS-wg or plain S2 cells for the time indicated or treated with 200 nM insulin for 30 min, and the cell lysates were subjected to Western blot analysis with the antibodies indicated.
PP2A catalytic subunit counteracts Arm and Daxin phosphorylation but does not appears to play an indispensable role in Wg signaling.
PP2A is a multisubunit Ser/Thr phosphatase that has a wide range of substrates and is involved in many cellular processes. It comprises a regulatory A subunit, a catalytic C subunit, and variable regulatory B subunits, which may target the location and/or action of the holoenzyme (reviewed in reference 42). The catalytic subunit of PP2A (PP2A-C) is known to bind to Axin (31, 47), whereas regulatory subunits of the PP2A-B′ family can interact with APC (21). Thus, PP2A could serve to modulate the effect of CKIα and Zw3/GSK-3β on Arm/β-catenin or other proteins in the Arm/β-catenin degradation complex. An earlier report with Xenopus and mammalian cells has indicated a positive regulatory role for PP2A-C at multiple levels in the Wnt pathway (31). However, the specific role of PP2A-C in the Wg pathway remains unclear. Therefore, using RNAi, we determined whether PP2A-C antagonizes CKIα- and Zw3-mediated-Arm phosphorylation, as well as Daxin phosphorylation (Fig. 8A), and whether PP2A-C promotes Arm dephosphorylation in a Wg-dependent manner, thereby playing a pivotal role in Wg-induced Arm accumulation (Fig. 8B).
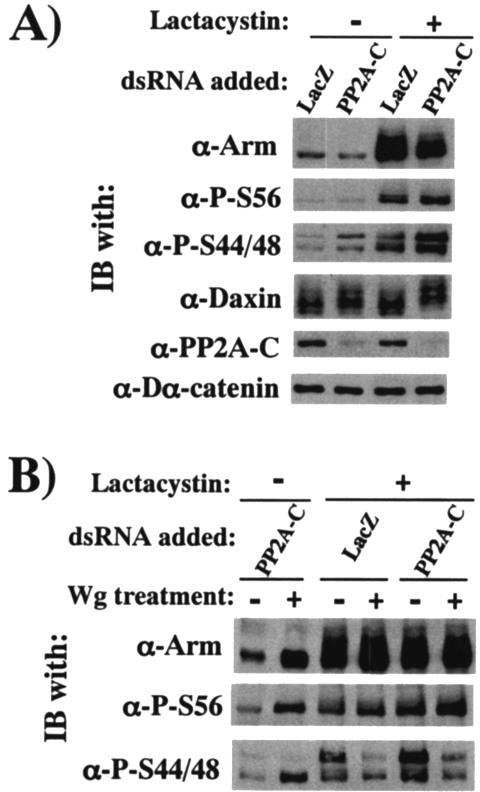
FIG. 8.
RNAi experiments to evaluate the function of PP2A-C in Wg signaling. (A) PP2A-C counteracts Arm phosphorylation at Ser56 and at Ser44 plus Ser48, as well as Daxin phosphorylation. To the S2R+ cell cultures preincubated with dsRNA for 30 h, 20 μM lactacystin was added or not, and then the cultures were further incubated for 6 h. The cell lysates were subjected to Western blot analysis with the antibody indicated. (B) Wg-induced decrease in Arm phosphorylation at Ser44 plus Ser48 is not caused by phosphatase activity of PP2A-C. The S2R+ cell cultures were preincubated with dsRNA for 30 h, further treated or not with lactacystin for 6 h, cocultivated with S2 or S2-HS-wg cells for a further 3 h, and then harvested for Western blot analysis.
In the absence of lactacystin, PP2A-C-RNAi slightly decreased the total Arm protein level, which is consistent with the report that PP2A-C functions as a positive regulator of Wnt signaling (31). Either in the presence or in the absence of lactacystin, PP2A-C-RNAi slightly elevated both P-S56-Arm and P-S44/48-Arm, as well as hyperphosphorylated Daxin, suggesting that, in vivo, PP2A-C antagonizes CKI- and Zw3-mediated Arm phosphorylation and Daxin phosphorylation, which appears to be mediated by Zw3 (32, 44).
In Fig. 6A, we have shown that Wg reduces Arm phosphorylation at Ser44 and Ser48. Although the N-terminal inhibitory phosphorylation of Zw3 is not associated with this process (Fig. 7B), it might also involve alterations in phosphatase activity: Wg/Wnt signaling in some way enhances the enzymatic activity of PP2A, thereby abolishing Zw3/GSK-3β-mediated Arm/β-catenin phosphorylation that eventually leads to its accumulation. If so, PP2A-C-RNAi may severely block the Wg-induced increase in Arm in the absence of lactacystin and strongly inhibit the Wg-induced decrease in P-S44/48-Arm in the presence of lactacystin. However, PP2A-C RNAi induced neither of these molecular events. Upon stimulation with Wg, S2R+ cells treated with PP2A-C-dsRNA showed levels of Arm similar to those of S2R+ cells treated with Laz-dsRNA. In the presence of lactacystin, PP2A-C-RNAi had no effect on the Wg-induced decrease in P-S44/48-Arm (Fig. 8B). Hence, as far as this RNAi-based analysis is concerned, PP2A-C counteracts Arm and Daxin phosphorylation, but this PP2A-C function does not appear to play a crucial role in normal Wg signaling.
Characterization of kinases involved in Wg-induced Dsh phosphorylation.
We have previously shown that Dsh/Dvl protein becomes phosphorylated in response to Wg/Wnt3A treatment in S2R+ and mouse L cells (19, 49), indicating that Dsh/Dvl phosphorylation occurs concomitantly with Wg/Wnt-induced Arm/β-catenin elevation. However, whether this Dsh/Dvl phosphorylation is an essential integral process in Wg/Wnt signal transduction or just a Wg/Wnt-induced molecular event which by itself has little function in the Wg/Wnt pathway remains unclear. In addition, it is still controversial which of the two Dsh-associated kinases, CKI or Par-1, is responsible for Wg/Wnt-induced Dsh/Dvl phosphorylation (15, 18, 26, 29, 37). In some reports, CKI-7, a CKI inhibitor was used at higher concentrations (100 to 600 μM) to show that Wnt-3A-induced Dvl phosphorylation in L cells was due to CKI (15, 26). However, we have noticed that at higher concentrations, CKI-7 does not necessarily work as a specific inhibitor of CKI in tissue culture systems (see Discussion).
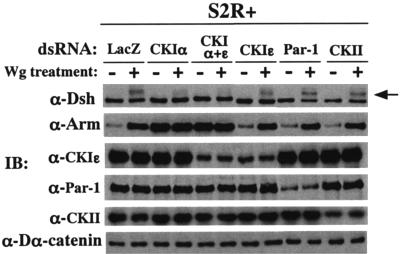
To identify the kinase responsible for the Wg-induced Dsh phosphorylation in a more convincing way, the expression of four Dsh-associated kinases—CKIα, CKIɛ, casein kinase II (CKII [43]), and Par-1—was downregulated by RNAi, and their effects on Dsh phosphorylation were examined. The Wg-induced Dsh phosphorylation was almost completely abolished by CKIα-RNAi but not by RNAi of the other Dsh-associated kinases (Fig. 9). Strictly speaking, this result only indicates CKIα is required for the Wg-induced Dsh phosphorylation, but we conclude that CKIα is the kinase directly responsible for the Wg-induced Dsh phosphorylation because CKIα is a Dsh-associated kinase and its overexpression in vivo induces hyperphosphorylation of Dsh (48). On the other hand, Par-1-, CKIɛ-, or CKII-RNAi had little effect on the Wg-induced accumulation of Arm, suggesting that these three kinases do not play an essential role in Wg signaling. The present results are inconsistent with the report that Par-1 potentiates Wg/Wnt signaling and is responsible for the Wg/Wnt-induced phosphorylation of Dsh/Dvl in Drosophila and mammalian cells (37). The reason for the discrepancy is not clear.
FIG. 9.
Wg-induced phosphorylation of Dsh is due to CKIα. Expression of each of four Dsh-associated kinases was suppressed by preincubating S2R+ cells with dsRNA for 30 h, and these S2R+ cell cultures were treated or not with Wg for 3 h and then harvested. The amounts of phosphorylated Dsh (shown with an arrow), Arm, CKIɛ, Par-1, CKII α subunit, and Dα-catenin among these cells were compared by Western blottings. LacZ dsRNA was used as a negative control.
PKA and PS play little part in Arm degradation in Drosophila tissue culture cells.
A recent report documented that PS functions as a scaffold linking PKA-mediated priming phosphorylation of β-catenin at Ser45 to subsequent GSK-3β-mediated phosphorylation at Ser33, Ser37, and Thr41, thereby downmodulating β-catenin expression in mammalian cells (16). To elucidate whether this PS-controlled regulation of Arm family protein is conserved in Drosophila and to biochemically evaluate the report in Drosophila embryos that a DPS deficiency led to a cytoplasmic Arm elevation (27), expression of the PKA catalytic subunit and DPS was disrupted by RNAi, and their effects on Arm levels were examined in S2R+ and BG2 cells. PKA-RNAi suppressed the expression of HA-tagged PKA in pMK-PKA-ΗΑ transfectant, indicating that PKA-RNAi is working (Fig. 10B). Furthermore, DPS protein levels expressed in both S2R+ and BG2 cells were diminished by DPS-RNAi. Although Zw3- and CKIα-RNAi markedly elevated Arm protein levels in both S2R+ and BG2 cells, DPS- and PKA-RNAi had little effect, suggesting that the assumed Arm degradation complex consisting of PKA, DPS, and Zw3 is not present or plays a marginal role in Arm protein metabolism at least in these Drosophila tissue culture cells (Fig. 10A). Although Drosophila genetic studies support the negative regulation of Arm by DPS, our RNAi-based findings in tissue culture cells demonstrated that the model recently proposed in mammalian cells is not applicable to Drosophila. Further study is required to clarify the biochemical relationship between DPS and Arm.
FIG. 10.
Arm protein levels are little affected by DPS and PKA in Drosophila tissue culture cells. (A) Expression of CKIα, ZW3, PKA, and DPS was downregulated by RNAi, and their effects on Arm protein levels were analyzed in S2R+ and BG2 cells. S2R+ or BG2 cells were incubated with dsRNA for 60 h. The cell lysates were then subjected to Western blot analysis with the antibody indicated. LacZ- and Dα-catenin-RNAi were used as negative controls. Total cell lysates were used for this analysis, because the amount of membrane-bound Arm is very limited in S2R+ and BG2 cells. (B) The PKA-RNAi experiment works. The stable pMK-PKA-HA transfectant was incubated with dsRNA for 60 h, treated or not with CuSO4 for a further 12 h, and subjected to Western blotting.
DISCUSSION
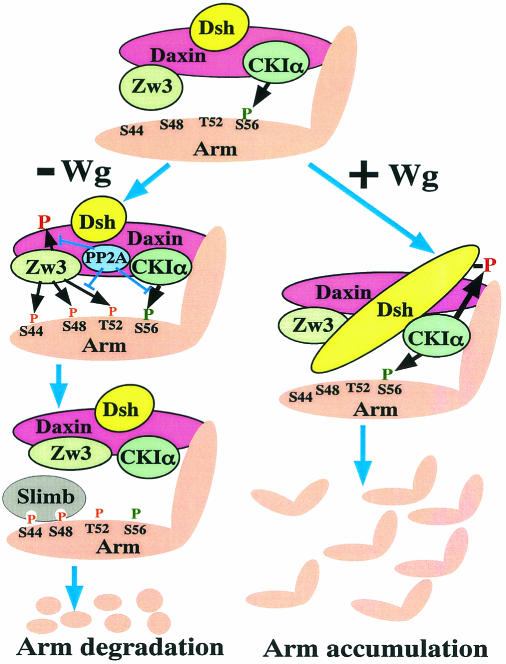
In this RNAi-based study, we have provided compelling evidence that phosphorylation of Arm at Ser56 by CKIα primes it for subsequent phosphorylation at Thr52, Ser48, and Ser44 by Zw3 and that Daxin enhances the Zw3- but not CKIα-mediated Arm phosphorylation. Moreover, we have clarified that Wg signaling inhibits the Zw3- but not CKIα-mediated phosphorylation of Arm and that PP2A-C does not play a primary role in this Wg-induced decrease in Arm phosphorylation. In addition, we have confirmed that Wg-induced Dsh phosphoylation is due to CKIα. Taking these findings into account, we present a model of how Wg signaling modulates the phosphorylation of Arm, as well as other components of the destruction complex, thereby regulating the stability of Arm (Fig. 11).
FIG. 11.
Model depicting how the Wg signal controls phosphorylation-dependent degradation of Arm. Daxin assembles Zw3, CKIα, Dsh, PP2A-C, and Arm in close proximity. Drosophila APC is not shown in this figure because the present study does not deal with this protein. CKIα constitutively primes Arm phosphorylation at Ser56. In the absence of Wg, Zw3 recognizes this priming phosphate then sequentially phosphorylates Thr52, Ser48, and Ser44. The latter two phosphorylations are required for the binding of an F-box protein, Slimb, that targets Arm for ubiquitination and degradation. PP2A-C appears to counteract the CKIα-mediated-phosphorylation of Arm, as well as the Zw3-mediated phosphorylation of Arm and Daxin. Although its molecular mechanism is unknown, the binding of Wg to the Dfrizzled2-Arrow receptor complex is speculated to induce conformational changes in Dsh that prevent Zw3 from phosphorylating Arm at Thr52, Ser48, and Ser44 on the one hand and elicit CKIα-mediated phosphorylation of Dsh itself on the other. However, CKIα-mediated Arm phosphorylation at Ser56 is not affected by Wg signaling. The mechanism by which Wg signaling counteracts Zw3-mediated phosphorylation of Arm is poorly understood, but the present study ruled out both Ser9 phosphorylation-induced inactivation of Zw3 kinase (Fig. 7B) and the involvement of PP2A (Fig. 8B). In this process, Frat/GBP has been proposed to evoke the dissociation of GSK-3β from Axin in vertebrates (6, 11, 20). However, Drosophila has no apparent Frat/GBP counterpart. Wnt/Wg signaling also diminishes GSK-3β/Zw3-mediated phosphorylation of Axin/Daxin (Fig. 6A), which appears to lower Axin/Daxin's affinity to β-catenin (44) and Zw3 (32), which were shown to stimulate the release of β-catenin from the degradation complex and speculated to reduce Arm phosphorylation, respectively. In Drosophila, it still remains unclear whether Wg-induced phosphorylation of Dsh by CKIα is essential for normal Wg signal transduction. However, in vertebrates Wnt-induced Dvl/Xdsh phosphorylation by CKIɛ has been shown to enhance the binding of Frat/GBP to Dvl/Xdsh that promotes the disintegration of the β-catenin destruction complex, which eventually leads to activation of the Wnt pathway (15, 18).
As shown in Fig. 9, CKIα plus CKIɛ-RNA elevates Arm levels, suggesting that the overall effect of inhibition of the CKI family is to induce the accumulation of Arm in S2R+ cells. In addition, the Wg-induced accumulation of Arm was not blocked by any of the CKI-RNAi (data not shown). Given that the 50% inhibitory concentration of CKI-7 for CKI is 9.5 μM, we treated S2R+ cells with various concentrations (10 to 1,000 μM) of CKI-7 for 5 h and analyzed their effect on Arm levels. CKI-7 did not induce Arm accumulation, whereas at higher concentrations (≥300 μM), it inhibited Wg-induced Arm elevation and Dsh phosphorylation. Similarly, CKI-7 did not induce β-catenin elevation but inhibited both Wnt3A-induced β-catenin accumulation and Dvl phosphorylation in L cells (data not shown). These results indicate that CKI-7-induced changes in Wnt/Wg signaling components are not necessarily due to its function as a CKI inhibitor.
In contrast to the finding in mammalian fibroblasts that PS1 and PKA functions as a scaffold and a priming kinase, respectively, for β-catenin degradation (16), RNAi in S2R+ and BG2 cells showed that DPS and PKA play little part in Arm degradation (Fig. 10). The reason for this discrepancy is not clear, but it should be noted that even in a mammalian system, Marambaud et al. have reported PS1 as a positive regulator of β-catenin: PS1 cleaves E-cadherin at the membrane-cytoplasmic interface, thereby releasing its intracellular domain, which eventually leads to disassembly of the E-cadherin-catenin complex and an increase in the cytoplasmic pools of β-catenin (25). However, this mechanism of Arm regulation may not exist in S2R+ and BG2 cells because these cells express little Drosophila E-cadherin (46).
Using RNAi in combination with a Drosophila Kc cell-based Tcf-dependent reporter assay, Lum et al. have systematically screened the functional roles of all kinases and phosphatases encoded by the Drosophila genome in Wg signaling (23). This screen identified Zw3 and CKIα but not CKIɛ, PKA, and PP2A as components of the Wg/Arm pathway, a finding which is in line with our results that CKIɛ-, PKA-, and PP2A-C-RNAi have little effect on Arm protein levels.
There is disagreement as to whether different CKI isoforms play the same or different roles (both positive or negative) in Wnt/Wg signaling. The different experimental systems and approaches used in different studies make the situation more complicated. Mainly based on observations that overexpression of CKI induced dorsal axis duplication in Xenopus embryos and stimulated the Tcf/lef reporter in mammalian cells, Graff's group have reported that all CKI isoforms except CKIγ positively regulate Wnt signaling in vertebrates (26, 29), whereas Sakanaka et al. have identified CKIɛ but not CKIα as a positive regulator of the Wnt pathway (34). Lee et al. have reported that CKIɛ strengthened Tcf3-β-catenin and GBP/Frat-Xdsh interactions, which led to β-catenin stabilization (18), whereas Rubinfeld et al. have demonstrated that CKIɛ mediates Axin-dependent phosphorylation of APC, thereby stimulating β-catenin degradation (33). However, these proposals for mechanisms of CKI action have not been evaluated by others.
In contrast, in three recent studies (2, 22, 48) and in the present study, the function of CKIα as a priming kinase for GSK-3β/Zw3 in β-catenin/Arm phosphorylation, and thus its negative role in Wnt/Wg signaling, has been established in both vertebrates and Drosophila. Liu et al. (22) and the present study have ruled out a role for CKIɛ as a priming kinase, whereas Ben-Neriah's group have demonstrated that in some cell types CKIɛ also functions as a priming kinase (2, 14). In addition, Schwarz-Romond et al. have reported that the ankyrin repeat protein Diversin recruits CKIɛ but not CKIα to the β-catenin degradation complex and allows efficient phosphorylation of β-catenin (35). These reports support negative roles for CKIɛ (as a priming kinase) in Wnt signaling. However, in two consecutive papers (15, 17), a positive role for CKIɛ but not CKIα in Wnt signaling has been reported: CKIɛ phosphorylates Dvl and thereby enhances the binding of Frat to Dvl, which leads to inhibition of Dvl-Axin interaction and a malfunction of the β-catenin degradation complex. Clearly, further studies are necessary to reconcile discrepancies about CKI's role in Wnt/Wg signaling and to figure out the real functions of CKI in the Wnt/Wg pathway.
Acknowledgments
We are grateful to D. St Johnston (University of Cambridge) and K. Tei (University of Tokyo) for providing anti-PAR-1 antibody and BG2 cells, respectively.
This study was supported by a grant-in-aid from the Ministry of Education, Science, Sports, and Culture of Japan to S.Y.
REFERENCES
- 1.Aberle, H., A. Bauer, J. Stappert, A. Kispert, and R. Kemler. 1997. β-Catenin is a target for the ubiquitin-proteasome pathway. EMBO J. 16:3797-3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Amit, S., A. Hatzuba, Y. Birman, J. S. Andersen, E. Ben-Shushan, M. Mann, Y. Ben-Neriah, and I. Alkalay. 2002. Axin-mediated CKI phosphorylation of β-catenin at Ser 45: a molecular switch for the Wnt pathway. Genes Dev. 16:1066-1076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cadigan, K., and R. Nusse. 1997. Wnt signaling: a common theme in animal development. Genes Dev. 11:3286-3305. [DOI] [PubMed] [Google Scholar]
- 4.Clements, J. C., C. A. Worby, N. Simonson-Leff, M. Muda, T. Maehara, B. A. Hemmings, and J. E. Dixon. 2000. Use of double-stranded RNA interference in Drosophila cell lines to dissect signal transduction pathways. Proc. Natl. Acad. Sci. USA 97:6499-6503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cook, D., M. J. Fry, K. Hughes, R. Sumathipala, J. R. Woodgett, and T. C. Dale. 1996. Wingless inactivates glycogen synthase kinase-3 via an intracellular signaling pathway which involves a protein kinase C. EMBO J. 15:4526-4536. [PMC free article] [PubMed] [Google Scholar]
- 6.Dajani, R., E. Fraser, S. M. Roe, M. Yeo, V. M. Good, V. Thompson, T. C. Dale, and L. H. Pearl. 2003. Structural basis for recuruitment of glycogen synthase kinase 3β to the axin-APC scaffold complex. EMBO J. 22:494-501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dajani, R., E. Fraser, S. M. Roe, N. Young, V. Good, T. C. Dale, and L. H. Pearl. 2001. Crystal structure of glycogen synthase kinase 3β: structural basis for phosphate-primed substrate specificity and auto-inhibition. Cell 105:721-732. [DOI] [PubMed] [Google Scholar]
- 8.Ding, V. W., R.-H. Chen, and F. McCormick. 2000. Differential regulation of glycogen synthase kinase 3β by insulin and Wnt signaling. J. Biol. Chem. 275:32475-32481. [DOI] [PubMed] [Google Scholar]
- 9.Ding, Y., and T. Dale. 2002. Wnt signal ttransduction: kinase cogs in a nano- machine? Trends Biochem. Sci. 7:327-329. [DOI] [PubMed] [Google Scholar]
- 10.Doble, B. W., and J. R. Woodgett. 2003. GSK-3: tricks of the trade for a multi-tasking kinase. J. Cell Sci. 116:1175-1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ferkey, D. M., and D. Kimelman. 2002. Glycogen synthase kinase3β mutagenesis identifies a common binding site for GBP and Axin. J. Biol. Chem. 277:16147-16152. [DOI] [PubMed] [Google Scholar]
- 12.Flotow, H., and P. J. Roach. 1991. Role of acidic residues as substrate determinants for casein kinase I. J. Biol. Chem. 266:3724-3727. [PubMed] [Google Scholar]
- 13.Frame, S., P. Cohen, and M. Biondi. 2001. A common phosphate binding site explains the unique substrate specificity of GSK3 and its inactivation by phosphorylation. Mol. Cell 7:1321-1327. [DOI] [PubMed] [Google Scholar]
- 14.He, X. 2003. A Wnt-Wnt situation. Dev. Cell 4:791-797. [DOI] [PubMed] [Google Scholar]
- 15.Hino, S., T. Michiue, M. Asashima, and A. Kikuchi. 2003. Casein kinase Iɛ enhances the binding of Dvl-1 to Frat-1 and is essential for Wnt-3a-induced accumulation of β-catenin. J. Biol. Chem. 278:14066-14073. [DOI] [PubMed] [Google Scholar]
- 16.Kang, D. E., S. Soriano, X. Xia, C. G. Eberhart, B. De Strooper, H. Zheng, and E. H. Koo. 2002. Presenilin couples the paird phosphorylation of β-catenin independent of axin: implications for β-catenin activation in tumorigenesis. Cell 110:751-762. [DOI] [PubMed] [Google Scholar]
- 17.Kishida, M., S.-i. Hino, T. Michiue, H. Yamamoto, S. Kishida, A. Fukui, M. Asashima, and A. Kikuchi. 2001. Synergistic activation of the Wnt signaling pathway by DVl and casein kinase 1ɛ. J. Biol. Chem. 276:33147-33155. [DOI] [PubMed] [Google Scholar]
- 18.Lee, E., A. Salic, and M. W. Kirschner. 2001. Physiological regulation of β-catenin stability by Tcf3 and CK1ɛ. J. Cell Biol. 154:983-994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee, J.-S., A. Ishimoto, and S. Yanagawa. 1999. Characterization of mouse Dishevelled (Dvl) proteins in Wnt/Wingless signaling pathway. J. Biol. Chem. 274:21464-21470. [DOI] [PubMed] [Google Scholar]
- 20.Li, L., H. Yuan, C. D. Weaver, J. Mao, G. H. Farr III, D. J. Sussman, J. Jonker, D. Kimelman, and D. Wu. 1999. Axin and Frat1 interact with Dvl and GSK, bridging Dvl to GSK in Wnt-mediated regulation of LEF-1. EMBO J. 18:4233-4240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li, X., H. J. Yost, D. M. Virshup, and J. M. Seeling. 2001. Protein phosphatase 2A and its B56 regulatory subunit inhibit Wnt signaling in Xenopus. EMBO J. 20:4122-4131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu, C., Y. Li, M. Semenov, C. Han, G.-H. Bang, Y. Tan, Z. Zhang, X. Lin, and X. He. 2002. Control of β-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell 108:837-847. [DOI] [PubMed] [Google Scholar]
- 23.Lum, L., S. Yao, B. Mozer, A. Rovescalli, D. Von Kessler, M. Nirenberg, and P. A. Beachy. 2003. Identification of hedgehog pathway components by RNAi in Drosophila cultured cells. Science 299:2039-2045. [DOI] [PubMed] [Google Scholar]
- 24.Marin, O., V. H. Bustos, L. Cesaro, F. Meggio, M. A. Pagano, M. Antonelli, C. C. Allende, L. A. Pinna, and J. E. Allende. 2003. A noncanonical sequence phosphorylated by casein kinase 1 in β-catenin may play a role in casein kinase 1 targeting of important signaling proteins. Proc. Natl. Acad. Sci. USA 100:10193-10200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marmbaud, P., J. Shioi, G. Serban, A. Georgakopoulos, S. Sarner, V. Nagy, L. Baki, P. Wen, S. Efthimiopoulos, Z. Shao, T. Wisniewski, and N. K. Robakis. 2002. A presenilin-1/γ-secretase claevage release the E-cadherin intracellular domain and regulates disassembly of adherens junctions. EMBO J. 21:11948-11956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mckay, R. M., J. M. Peters, and J. M. Graff. 2001. The casein kinase I family in Wnt signaling. Dev. Biol. 235:388-396. [DOI] [PubMed] [Google Scholar]
- 27.Noll, E., M. Medina, J. D. Hartley, Zhou, N. Perrimon, and K. S. Kosik. 2000. Presenilin affects Arm/β-catenin localization and function in Drosophila. Dev. Biol. 227:450-464. [DOI] [PubMed] [Google Scholar]
- 28.Pai, L.-M. S., S. Orsulic, A. Bejsovec, and M. Peifer. 1997. Negative regulation of armadillo, a wingless effector in Drosophila. Development 124:2255-2266. [DOI] [PubMed] [Google Scholar]
- 29.Peters, J. M., R. M. Mckay, J. P. McKay, and J. M. Graff. 1999. Casein kinase I transduces Wnt signals. Nature 401:345-350. [DOI] [PubMed] [Google Scholar]
- 30.Polakis, P. 2000. Wnt signaling and cancer. Genes Dev. 14:1837-1851. [PubMed] [Google Scholar]
- 31.Ratcliffe, M. J., K. Itoh, and S. Sokol. 2000. A positive role for the PP2A catalytic subunit in the Wnt signal transduction. J. Biol. Chem. 275:35680-35683. [DOI] [PubMed] [Google Scholar]
- 32.Ruel, L., V. Stambolic, A. Ali, A. S. Manoukian, and J. R. Woodgett. 1999. Regulation of the protein kinase activity of Shaggy by components of the Wingless pathway in Drosophila cells. J. Biol. Chem. 274:21790-21796. [DOI] [PubMed] [Google Scholar]
- 33.Rubinfeld, B., D. A. Tice, and P. Polakis. 2001. Axin-dependent phosphorylation of the adenomatous polyposis coli protein mediated by casein kinase 1 epsilon. J. Biol. Chem. 276:39037-39045. [DOI] [PubMed] [Google Scholar]
- 34.Sakanaka, C., P. Leong, L. Xu, S. D. Harrison, and L. T. Williams. 1999. Casein kinase Iɛ in the Wnt pathway: regulation of β-catenin function. Proc. Natl. Acad. Sci. USA 96:12548-12552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schwarz-Romond, T., C. Asbrand, J. Bakkers, M. Kuhl, H.-J. Schaeffer, J. Huelsken, J. Berhrens, M. Hammerscmit, and W. Birchmeier. 2002. The ankyrin repeat protein Diversin recuruit casein kinase Iɛ to the β-catenin degradation complex and acts in both canonical Wnt and Wnt/JNK signaling. Genes Dev. 16:2073-2084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Siegfried, E., T.-B. Chou, and N. Perrimon. 1992. wingless signaling acts through zeste-white 3, the Drosophila homolog of glycogen synthase kinase-3 to regulate engrailed and establish cell fate. Cell 71:1167-1179. [DOI] [PubMed] [Google Scholar]
- 37.Sun, T.-Q., B. Lu, J.-J. Feng, C. Reinhard, Y. N. Jan, W. J. Fanti, and L. Williams. 2001. PAR-1 is a dishevelled-associated kinase and a positive regulator of Wnt signalling. Nat. Cell Biol. 3:628-636. [DOI] [PubMed] [Google Scholar]
- 38.Takasugi, N., Y. Takahashi, Y. Morohashi, T. Tomita, and T. Iwatsubo. 2002. The mechanism of γ-secretase activities through high molecular weight complex formation of presenilin is conserved in Drosophila melanogaster and mammals. J. Biol. Chem. 277:50198-50205. [DOI] [PubMed] [Google Scholar]
- 39.Ui, K., S. Nishihara, M. Sakuma, S. Togashi, R. Ueda, Y. Miyata, and T. Miyake. 1994. Newly established cell lines from Drosophila larval CNS express neural specific characteristics in vitro. Cell. Biol. 30A:209-216. [DOI] [PubMed] [Google Scholar]
- 40.van Leeuwen, F., C. Harryman-Samos, and R. Nusse. 1994. Biological activity of soluble wingless protein in cultured Drosophila imaginal disc cells. Nature 368:342-344. [DOI] [PubMed] [Google Scholar]
- 41.van Noort, M., J. Meeldijk, R. van der Zee, O. Destree, and H. Clevers. 2002. Wnt signaling controls the phosphorylation status of β-catenin. J. Biol. Chem. 277:17901-17905. [DOI] [PubMed] [Google Scholar]
- 42.Virshup, D. 2000. Protein phosphatase 2A: a panoply enzymes. Curr. Opin. Cell Biol. 12:180-185. [DOI] [PubMed] [Google Scholar]
- 43.Willert, K., M. Brink, A. Wodarz, H. Varmus, and R. Nusse. 1997. Casein kinase 2 associates with and phosphorylates Dishevelled. EMBO J. 16:3089-3096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Willert, K., S. Shibamoto, and R. Nusse. 1999. Wnt-induced dephosphorylation of Axin release β-catenin from the Axin complex. Genes Dev. 13:1768-1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wodarz, A., and R. Nusse. 1998. Mechanisms of Wnt signaling in development. Annu. Rev. Cell Dev. Biol. 14:59-88. [DOI] [PubMed] [Google Scholar]
- 46.Yanagawa, S., J.-S. Lee, and A. Ishimoto. 1998. Identification and characterization of a novel line of Drosophila Schneider S2 cells that respond to wingless signaling. J. Biol. Chem. 273:32353-32359. [DOI] [PubMed] [Google Scholar]
- 47.Yanagawa, S., J.-S. Lee, Y. Matsuda, and A. Ishimoto. 2000. Biochemical characterization of the Drosophila Axin protein. FEBS Lett. 474:189-194. [DOI] [PubMed] [Google Scholar]
- 48.Yanagawa, S., Y. Matsuda, J.-S. Lee, S. H. Matsubayashi, Sese, T. Kadowaki, and A. Ishimoto. 2002. Casein kinase I phosphorylates the Armadillo protein and induces its degradation in Drosophila. EMBO J. 21:1733-1742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yanagawa, F. S., van Leeuwen, A. Wodarz, J. Klingensmith, and R. Nusse. 1995. The dishevelled protein is modified by wingless signaling in Drosophila. Genes Dev. 9:1087-1097. [DOI] [PubMed] [Google Scholar]
- 50.Yost, C., M. Torres, J. R. Miller, E. Huang, D. Kimelman, and R. Moon. 1996. The axis-inducing activity, and subcellular distribution of β-catenin is regulated in Xenopus embryos by glycogen synthase kinase 3. Genes Dev. 10:1443-1454. [DOI] [PubMed] [Google Scholar]