Abstract
Myasthenia gravis is an autoimmune disorder of the neuromuscular junction. A number of molecules, including ion channels and other proteins at the neuromuscular junction, may be targeted by autoantibodies leading to abnormal neuromuscular transmission. In approximately 85% of patients, autoantibodies, directed against the postsynaptic nicotinic acetylcholine receptor can be detected in the serum and confirm the diagnosis, but in general, do not precisely predict the degree of weakness or response to therapy. Antibodies to the muscle-specific tyrosine kinase are detected in approximately 50% of generalized myasthenia gravis patients who are seronegative for anti-acetylcholine receptor antibodies, and levels of anti-muscle-specific tyrosine kinase antibodies do appear to correlate with disease severity and treatment response. Antibodies to other muscle antigens may be found in the subsets of myasthenia gravis patients, potentially providing clinically useful diagnostic information, but their utility as relevant biomarkers (measures of disease state or response to treatment) is currently unclear.
Keywords: AChR, biomarkers, diagnosis, MuSK, myasthenia gravis
Autoimmune myasthenia gravis (MG) is a relatively rare disease affecting approximately 20 per 100,000 people [1]. Patients with MG exhibit characteristic fatigable weakness of voluntary muscles including ocular, facial, oropharyngeal, limb and respiratory muscles [2]. MG is a well-established organ-specific, autoantibody- mediated disease caused by circulating antibodies directed against skeletal muscle receptors and proteins at the neuromuscular junction (NMJ) [2–4]. These antibodies bind to components of the NMJ, disturbing their normal function and impairing neuromuscular transmission.
In approximately 85% of MG patients, circulating antibodies against the acetylcholine receptor (AChR) bungarotoxin-binding site are not only the pathogenic effector immune molecules but also provide a sensitive and specific diagnostic test [2]. Up to 50% of the remaining 15% of MG patients harbor antibodies against muscle-specific tyrosine kinase (MuSK), an enzyme critical for NMJ formation and agrin-induced AChR clustering [5,6]. In the remaining cases, antibodies against AChR and MuSK are not detectable by conventional assays, perhaps indicating a distinct autoantigenic target or possibly a lack of assay sensitivity. Some of these patients have been found to have circulating antibodies that can only be detected by binding of clustered AChRs in a cell-based assay [7]. In general, detecting circulating anti-AChR and anti-MuSK antibodies in MG provides an important means to confirm the clinical diagnosis in patients with suspected disease, allowing specific treatment.
MG is also associated with other antibodies that recognize skeletal muscle proteins (other than AChR or MuSK), which may be useful in identifying the subtypes of MG [8], although their role in disease pathogenesis is unclear. The molecules that have been identified as immune targets on the postsynaptic membrane and muscle cell are depicted in Figure 1. The identification of the antibodies that bind to these proteins aids in the classification of MG clinical subtypes, as shown in Table 1. However, there is a variable relationship between the levels of serum anti-AChR, anti-MuSK or other MG-related muscle autoantibodies to disease severity and response to treatment in MG. In this paper, the authors will review the muscle autoantibodies that have been associated with MG and discuss their role in MG pathology, diagnosis and their potential utility as therapeutic biomarkers.
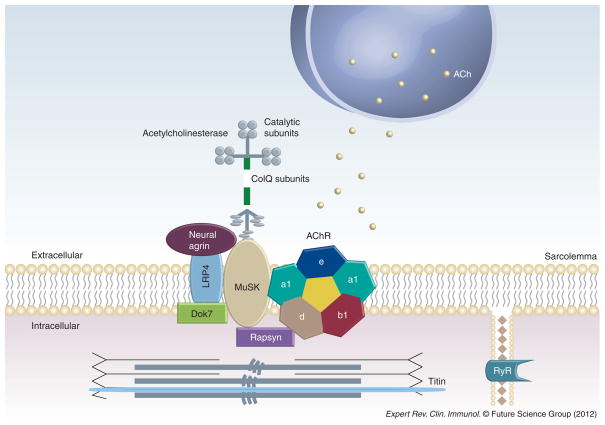
Figure 1. Muscle autoantigens in myasthenia gravis.
The major pathologic antibodies in myasthenia gravis target AChR and MuSK on the postsynaptic membrane. Autoantibodies may also target other end-plate proteins (LRP4, rapsyn) and proteins involved in muscle contraction and excitation–contraction coupling (titin, myofibrillar proteins and RyR).
ACh: Acetylcholine; AChR: Acetylcholine receptor; ColQ: Collagen Q; LRP4: Lipoprotein-related protein receptor 4; MuSK: Muscle-Specific tyrosine kinase; RyR: Ryanodine receptor.
Table 1.
Clinical myasthenia gravis subtypes and associated autoantibodies.
| MG subtype | Age at onset | Thymic histology | Muscle autoantibodies |
|---|---|---|---|
| Early onset | <50 years | Hyperplasia |
|
| Late onset | >50 years | Normal |
|
| Thymomatous | Any age; peak at 40–60 years | Neoplasia |
|
| MuSK | – | Minimal |
|
| Ocular | Variable | Unknown |
|
| Double-seronegative | Variable | Unknown |
|
AChR: Acetylcholine receptor; LRP4: Lipoprotein-related protein receptor 4; MG: Myasthenia gravis; MuSK: Muscle-specific tyrosine kinase; VGKC: Voltage-gated potassium channel.
Anti-AChR antibodies
In most patients with MG, the disease arises from a humoral auto-immune response directed against the muscle nicotinic AChR [2–4]. Evidence from classical experiments indicates that anti-AChR antibodies are pathogenic (the main cause of weakness in MG), leading to end-plate AChR loss, simplification of the postsynaptic membrane and derangement of neuromuscular transmission [9]. AChR antibodies from MG patients bind to the NMJ and cause weakness when injected into experimental animals [10]. In addition, the removal of circulating antibodies by thoracic duct drainage [11] or plasma exchange [12] results in improvement in MG symptoms. Anti-AChR antibodies in human MG are comprised of the IgG subclass 1 or 3 and mainly target the main immunogenic region of the AChR, which is located at the extracellular region of the AChR-α subunit [13,14]. Antibody binding reduces the number and/or function of muscle AChRs by three main mechanisms [9,15]: complement activation resulting in destruction and focal lysis of the postsynaptic folds at the NMJ leading to the destruction of AChR and AChR-related proteins at the end-plate (i.e., rapsyn and voltage-gated sodium channels); cross-linking of adjacent AChRs resulting in their accelerated internalization and degradation; and blocking of the acetylcholine (ACh)-binding site. Anti-AChR antibodies are highly specific for MG because they are not detected in healthy individuals and are only rarely present in patients with other autoimmune or neuromuscular disorders (see below).
Role of the thymus gland
The thymus gland plays an incompletely understood but critical role in the pathogenesis of MG with AChR autoantibodies. Most MG patients have thymic abnormalities, with more than 50% having thymic hyperplasia, and 10–15% having a thymic tumor [16]. The hyperplastic thymus glands of MG patients contain all the functional components (T cells, B cells and plasma cells, as well as muscle-like myoid cells that express AChR) necessary for the development of an immune response to AChR, and thymocytes in culture spontaneously produce anti-AChR antibodies [17]. These findings support the concept of an intrathymic source for the initiation of the anti-AChR immune response and the production of AChR antibodies in MG patients with thymic hyperplasia.
Thymoma is a relatively rare neoplasm of thymic epithelial cells that is often associated with autoimmunity, probably due to the presentation of altered self-antigens expressed by neoplastic cells. Thymomas may express numerous self-like antigens, including AChR-, titin- and ryanodine receptor-like epitopes [18]. Unlike the case in thymic hyperplasia, there is no significant autoantibody production within thymomas. However, sensitized autoreactive T lymphocytes may proliferate, leave the tumor and stimulate B cells to produce autoantibodies. Virtually 100% of patients with thymomatous MG have detectable serum AChR antibodies [19]. The antibodies and their epitopes differ in their fine specificities from those in MG patients without thymoma [20,21]. A number of other muscle autoantibodies are also associated with thymoma and will be discussed below.
While the precise mechanism of auto-sensitization to the AChR is not clear, abnormalities of the thymus gland (hyperplasia and neoplasia) almost certainly play a role in the majority of MG patients. As a primary site for the establishment of immune regulation, derangements in the thymus gland may lead to a defect in the immune system’s suppression of autoreactive lymphocytes, allowing for the development of anti-AChR immune responses. AChR-specific CD4+ T cells are present in the blood of MG patients with these antibodies [22] and are particularly abundant in the thymus, supporting the idea that the thymus is the site where T-cell autosensitization takes place in MG [23,24]. Interestingly, healthy subjects also have circulating T cells that can be activated in the presence of various autoantigens, including AChR, indicating that autoreactive T cells may normally gain access to the peripheral immune system but are held in check by peripheral tolerance mechanisms [25].
In thymoma, frequent concurrent autoimmunity against seemingly unrelated autoantigens suggests that potentially cross-reacting proteins expressed by the tumor play a role in disease production [26]. Thymomas from MG patients are rich in auto-reactive T cells, consistent with this postulate [27]. It may also be hypothesized that these autoreactive T cells are positively selected (selected for survival) and exported to the periphery, where they are activated and provide help for autoantibody-producing B cells. Negative selection and regulation of potentially self-reactive T cells may also play a role in abnormal thymus tissue due to a deficiency in the expression of the autoimmune regulator gene and possibly the selective loss of regulatory T cells [28,29].
Diagnostic testing
AChR-related antibodies in MG can be classified into three types (based on the effects of the antibodies on AChRs and AChR turnover): binding, blocking and modulating [30]. The most commonly utilized and clinically useful AChR-antibody assay (anti-AChR binding assay) measures IgG binding to 125I-α-bungarotoxin-labeled AChR by a radioimmunoprecipitation assay [31]. As noted, autoantibodies against AChRs can be detected in approximately 80–85% of patients with generalized MG and 50–75% of patients with ocular MG [31–34]. In general, an elevated level of anti-AChR-binding antibodies in a patient with compatible clinical features confirms the diagnosis of MG. Patients with early-onset MG (prior to age 40 years) and generalized weakness and patients with thymoma tend to have the highest concentrations of serum AChR-binding antibody levels compared with other subgroups, and anti-AChR antibodies are less frequently detected in MG patients with mild disease or restricted muscle weakness [35,36]. Anti-AChR-binding antibodies are relatively specific for MG, although they may also rarely be found in patients with autoimmune liver disease, systemic lupus, rheumatoid arthritis patients receiving penicillamine, in allogeneic bone marrow transplantation patients who develop graft-versus-host disease [37], and in patients with thymoma without MG [38], as well as in neuromyelitis optica [39]. Patients may be ‘falsely seronegative’ due to immunosuppression or if the test is performed early in the disease course [40].
Blocking AChR antibodies interfere with the interaction of ACh with AChR by occupying the ACh-binding site and are measured by determining the inhibition of 125I-α-bungarotoxin labeling of AChR by patient serum [30]. AChR-blocking antibodies are reported as a percentage of inhibition of α-bungarotoxin-binding sites on solubilized AChR and have been suggested to be important pathologically in acute exacerbations of MG, although they appear to represent a minority of AChR antibodies. These antibodies usually occur in association with AChR-binding antibodies and have a higher prevalence in generalized MG compared with disease restricted to the ocular muscles (ocular MG). A significant correlation between the degree of AChR blockade and the severity of generalized muscle weakness has been observed [30]. As a diagnostic test, however, AChR blocking antibodies are of limited value since they are very rarely present in the absence of AChR-binding antibodies.
Modulating AChR antibodies accelerate the rate of AChR internalization by cross-linking adjacent receptors and are detected by measuring the amount of internalized, processed 125I-α-bungarotoxin-labeled AChR released from cultured cells [30]. A positive result is most useful when the AChR-binding assay is negative, which occurs in approximately 3–4% of patients [36]. High levels of AChR-modulating antibodies have been reported in association with thymoma [41]. It should be noted that this assay cannot separate blocking antibody-released radioactivity from modulating antibody-released radioactivity and, therefore, cannot distinguish these two types of AChR antibodies [30].
Binding, blocking and modulating AChR antibody assays were designed to quantitate the three main pathologic mechanisms for loss of functional AChR mediated by circulating AChR antibodies (see above). Various AChR-testing algorithms have been proposed in an effort to optimize the relative sensitivity of each AChR antibody subtype with respect to diagnosis, but a comparison of the various studies is complicated by the use of alternative methods in different patient populations. In addition, it is likely that different patients have different AChR antibody subtypes with varying degrees of pathogenicity. In general, however, binding AChR antibodies are the most useful diagnostic test, with modulating, and particularly blocking AChR assays, adding relatively little to the diagnostic sensitivity.
Anti-AChR antibodies as measures of disease severity or treatment response
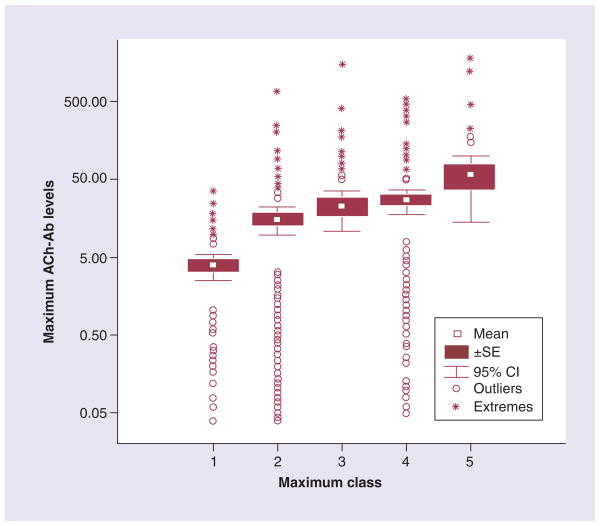
It is generally believed that no consistent correlation exists between anti-AChR antibody serum titers and MG disease severity. However, patients with ocular MG tend to have lower antibody titers compared with patients with generalized MG [34–36], and in individual patients, serial antibody titers tend to correlate with disease status. Tindall reported serum AChR antibody titers in MG patients according to disease severity as measured by the Osserman MG classification and found a correlation between antibody titers and disease severity [42]. In a population of 865 MG patients from a single academic center, there was a correlation between anti-AChR antibody levels and maximum disease severity per the Myasthenia Gravis Foundation of America (MGFA) disease classification (Figure 2) [43], but with many outliers and exceptions [Sanders DB et al., Unpublished Data]. This lack of a precise correlation is likely explained by multiple factors, including differences in the specificities of AChR antibodies, the immunoglobulin subclass of the antibodies and their ability to activate complement, as well as differences in serum and tissue antibody concentrations.
Figure 2. Maximum acetylcholine receptor antibody levels in 865 patients with seropositive myasthenia gravis versus maximum class.
ACh-Ab: Acetylcholine antibody; SE: Standard error.
Reproduced with permission from [Sanders DB et al., Unpublished Data] © DB Sanders (2012).
The usefulness of serial measurements of anti-AChR antibody levels to monitor treatment response in individual MG patients is also unclear. A number of studies have reported reductions in AChR antibody titers correlating with clinical improvement in response to immunomodulation and thymectomy [42,44–48]. In one of the larger cohorts, there was a strong correlation between change in AChR antibody titer and clinical status after treatment with prednisone or immunotherapy, as well as post-thymectomy in 75 patients [45,46]. In a prospective study of 60 MG patients, changes in AChR-binding antibody levels correlated with changes in the quantitative clinical score in most patients, and correlations with clinical state were stronger for binding compared with blocking AChR antibodies [44].
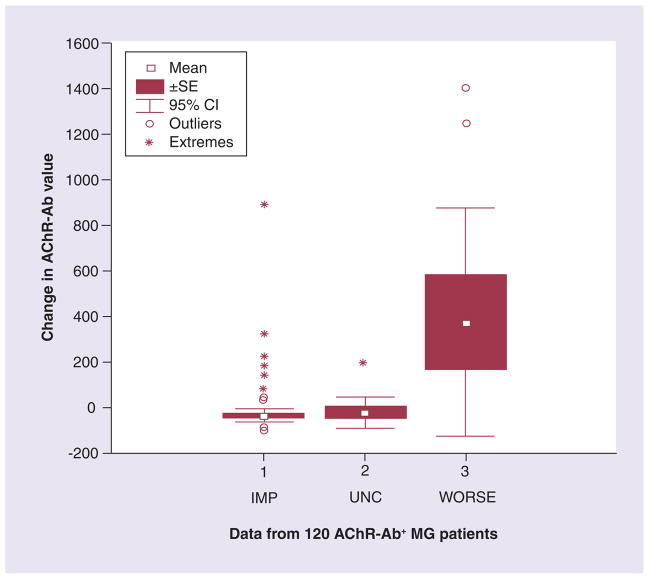
In a population of 120 patients in whom response to treatment was graded by the MGFA postintervention status score, a reduction in antibodies generally accompanied clinical improvement, but again, there were numerous exceptions and outliers (Figure 3) [Sanders DB et al., Unpublished Data]. A comparison of AChR-binding antibody level with change in disease severity determined by MGFA postintervention status or masked physician assessment in 153 patients showed that antibody levels decreased in almost all patients who improved after treatment and also in many who did not [Sanders DB et al., Unpublished Data].. In that study, a fall in binding antibody level had a positive predictive value of 78% for clinical improvement and a negative predictive value of only 60%.
Figure 3. Anti-acetylcholine receptor antibody levels in 120 myasthenia gravis patients grouped according to treatment response as assessed by the Myasthenia Gravis Foundation of America postintervention status scale.
All AChR-Ab values; n = 120.
AChR-Ab: Acetylcholine receptor antibody; IMP: Improved; MG: Myasthenia gravis; SE: Standard error; UNC: Unchanged; WORSE: Patient worsened.
Reproduced with permission from [Sanders DB et al., Unpublished Data]. © DB Sanders (2012).
The published studies and the authors’ observations in a large patient cohort suggest that clinical manifestations may, at least in part, be a function of the serum anti-AChR antibody concentrations. However, neither the presence nor the absolute concentration of antibodies precisely predicts disease class in all MG patients, nor does it accurately predict clinical disease course or therapeutic response in individual patients.
Anti-MuSK antibodies
MuSK is an NMJ protein that is specifically expressed at the postsynaptic membrane, where it colocalizes with AChR, and plays a critical role in the maintenance of the normal functional integrity of the NMJ by mediating clustering of AChRs [49]. The role of MuSK in mature adult muscle is less clear, but inhibition of MuSK synthesis has been found to cause AChR dispersion and end-plate disruption [50]. MuSK and the NMJ protein and low-density lipoprotein receptor-related protein 4 (LRP4; see below) together function as a receptor for neural agrin (Figure 1), a nerve-derived extracellular protein [49]. As noted, approximately half of generalized MG patients without anti-AChR antibodies have anti-MuSK antibodies (MuSK MG) [5,6]. MuSK antibodies bind to the N-terminal half of the extra-cellular domain of MuSK, functionally inhibiting agrin-induced AChR clustering in vitro [5].
The pathophysiology underlying anti-MuSK-positive MG has not been entirely defined. Studies comparing AChR loss and complement deposition in muscle from MuSK MG patients have shown relatively little change in AChR density and less frequent complement deposition compared with patients with anti-AChR-positive MG [51]. However, IgG from anti-MuSK-positive patients has been shown to cause myasthenic weakness in mice associated with a progressive reduction in the density of postsynaptic AChR combined with changes in the nerve terminal and its relationship to the postsynaptic membrane [52]. Furthermore, myasthenic weakness has been produced in experimental animals by immunization with recombinant MuSK protein, accompanied by reduced AChR clustering at the postsynaptic membrane [53,54]. Passive transfer of human anti-MuSK antibodies also influences the activity of MuSK in regenerating end-plates, diminishing their size without reducing MuSK levels [52,55].
The anti-MuSK antibodies in human MG belong predominantly to the IgG4 subclass and thus do not activate complement [56]. Active immunization of animals with MuSK protein, conversely, results in the production of antibodies that do activate complement [53,54]. Recent studies, however, demonstrate that complement activation may not be necessary for the onset of MuSK MG in mice and that both divalent and monovalent antibodies may induce MuSK dysfunction without the activation of complement [57,58]. It has also recently been shown that purified human anti-MuSK IgG4, passively transferred into experimental mice, binds to mouse NMJs and causes severe reduction of postsynaptic ACh sensitivity and depression of presynaptic ACh release during high-rate activity, culminating in fatigable muscle weakness [59]. This suggests that MuSK antibodies may have a presynaptic as well as postsynaptic effect on neuromuscular transmission and that activation of complement is not necessary for these effects [57]. Finally, it has been shown that anti-MuSK IgG interferes with the binding of MuSK to the collagen tail (collagen Q or [ColQ]) of end-plate acetylcholinesterase [60], suggesting that at least in some patients, the main target of anti-MuSK antibodies is the MuSK–ColQ interaction, and perhaps providing an explanation for the experimental observation of hypersensitivity to acetylcholinesterase inhibitors in the murine model [57], and the clinical observation that acetylcholinesterase inhibitors are ineffective in many MuSK MG patients [61]. Since dimerization, endocytosis and autophosphorylation of MuSK is required for its function [62], it is conceivable that antibodies binding to MuSK may also block these processes.
Diagnosis
Anti-MuSK antibodies have been reported in up to 50% of patients with generalized MG who lack anti-AChR antibodies [6,63,64]. The identification of anti-MuSK antibodies effectively confirms the diagnosis of MuSK MG, as false-positive results have not been reported and anti-MuSK antibodies are very seldom found in patients with anti-AChR antibodies [65]. The incidence of MuSK MG varies among geographic regions, the highest being closer to the equator and the lowest closer to the poles [66]. The frequency of anti-MuSK MG is also much lower in Asian populations [67], and it is likely that genetic or environmental factors or both play a role in these differences.
While MG patients with anti-MuSK antibodies may have presentations similar to anti-AChR-positive MG, they frequently have atypical clinical features, such as selective facial, bulbar, neck and respiratory muscle weakness and marked muscle atrophy, occasionally with relative sparing of ocular muscles [64,66]. Weakness may involve muscles that are not usually symptomatic in MG, such as paraspinal and upper esophageal muscles [68]. The preferential involvement of certain muscles in MuSK MG may reflect a different composition of the end-plates in these muscles. As noted, enhanced sensitivity, nonresponsiveness or even clinical worsening in response to anticholinesterase agents have also been reported [61]. Disease onset in MuSK MG patients tends to be earlier, and patients are predominantly female [66]. Thymus histology in these patients typically shows age-related atrophy without hyperplasia or neoplasia [69].
Anti-MuSK antibodies as measures of disease severity or treatment response
Anti-MuSK antibody serum levels have been shown to correlate with clinical symptoms and response to immunotherapy. In one report, the distribution of antibody levels in 83 serum samples from 40 patients correlated with both disease classification and clinical score, and in individual patients, immunosuppressive therapy led to a marked decrease in MuSK IgG [70]. Interestingly, no change in anti-MuSK antibody levels was observed after thymectomy, consistent with the reported lack of thymic pathology and response to thymectomy in these patients [64,66,69]. Niks et al. reported that levels of IgG4 (and not IgG1) correlated significantly with disease severity in a linear effect model [71]. While these studies strongly suggest that anti-MuSK IgG4 levels may serve as valuable biologic markers of disease, long-term longitudinal studies in larger patient populations are needed for confirmation.
Double-seronegative MG
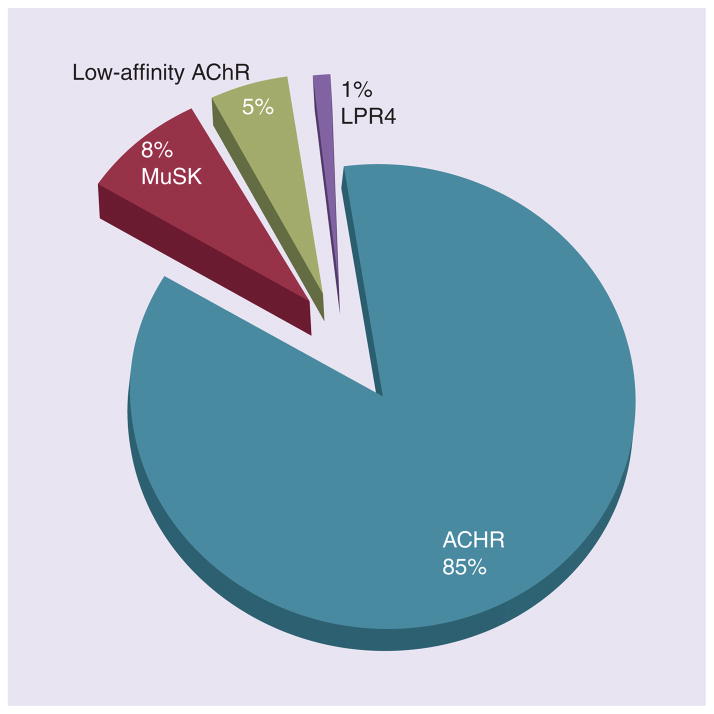
According to available published studies, approximately 7–8% of patients with MG have neither anti-AChR or anti-MuSK antibodies (Figure 4). Autoantibodies to AChR and MuSK detected in the serum of MG patients as described above are ‘high-affinity’ antibodies, meaning that they bind avidly to extracted AChR. ‘Low-affinity’ anti-AChR antibodies binding to AChRs clustered on the surface of a nonmuscle cell line (presenting antigenic epitopes in a more native conformation) have been found in 66% of generalized MG patients who were antibody negative on conventional AChR and MuSK assays [7]. Like anti-AChR antibodies detected by the conventional assay, these antibodies have also been shown to activate complement. It remains to be determined whether low-affinity anti-AChR antibodies are present in ocular MG, but this cell-based assay may eventually provide a more sensitive diagnostic test in this subgroup as well. A similar cell-based assay may enhance the detection of anti-MuSK antibodies.
Figure 4. Distribution of diagnostic autoantibody results in myasthenia gravis.
AChR: Acetylcholine receptor; LRP4: Lipoprotein-related protein receptor 4; MuSK: Muscle-specific tyrosine kinase.
The percentage of double seronegative MG patients in a given population also very likely varies based on genetics, geography and other characteristics of the cohort of interest (academic practice vs community based). For example, in a large tertiary referral academic practice, only 69% of 734 MG patients without thymoma were seropositive for AChR or MuSK antibodies (Table 2) [Sanders DB et al., Unpublished Data]. This relatively low seropositive rate may be explained by the exclusion of thymoma patients (who are essentially 100% positive for AChR antibodies), by the fact that seronegative patients are more likely to be referred to a tertiary center and by the availability of sensitive electrophysiologic tests for confirmation of the diagnosis of MG in seronegative patients. Finally, other antigenic targets at the NMJ may be responsible for disease in the remaining percentage of MG patients who are anti-AChR and anti-MuSK negative and do not have low-affinity anti-AChR antibodies. These are discussed below.
Table 2.
Anti-acetylcholine receptor antibody measurements in 734 patients with autoimmune myasthenia gravis without thymoma.
| Maximum severity† | I (Ocular) | II | IIa | IIb | III | IIIa | IIIb | IV | IVa | IVb | V | All patients |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N (MuSK antibody positive) | 116 (0) | 225 (4) | 138 (2) | 86 (2) | 194 (13) | 41 (0) | 153 (13) | 105 (5) | 66 (1) | 39 (4) | 67 (9) | 707 (31)‡ |
| AChR antibody positive (%) | 47 (40.5) | 138 (61) | 83 (60) | 55 (64) | 146 (75) | 32 (78) | 114 (75) | 76 (72) | 47 (71) | 29 (74) | 49 (73) | 456 (64) |
| AChR antibody | ||||||||||||
| Mean§ | 4.21 | 12.46 | 9.19 | 17.41 | 16.89 | 25.94 | 14.36 | 37.13 | 47.76 | 19.89 | 83.23 | 24.38 |
| SD | 7.3 | 30.55 | 24.88 | 36.98 | 37.75 | 64.31 | 25.18 | 77.19 | 94.51 | 25.67 | 301.0 | 107.39 |
| Maximum | 36 | 244 | 205 | 244 | 356 | 356 | 200 | 380 | 380 | 90 | 1795 | 1795 |
Disease severity by Myasthenia Gravis Foundation of America Clinical Classification [43].
Maximum severity data were missing in 27 patients.
Maximum values, seropositive patients only, measured at Mayo Medical Laboratories.
AChR: Acetylcholine receptor; MuSK: Muscle-specific tyrosine kinase; N: Total number of patients in each disease class; SD: Standard deviation.
Autoantibodies directed against non-AChR skeletal muscle proteins
Antibodies against striated muscle (striational antibodies)
While the evidence for the pathogenicity of anti-AChR antibodies in MG is quite strong [2], non-AChR autoantibodies that react with striated muscle antigens may also be found in up to 95% of MG patients with a thymoma and in 50% of late-onset MG patients without thymoma [8]. Striated muscle (striational) antibodies recognize muscle intracellular proteins (titin, myosin, actin and ryanodine receptors) and are therefore not directly accessible to autoantibodies. Presumably, this would suggest that these antibodies are not pathogenic, but, as will be discussed below, some of them are correlated with MG disease severity and therefore may contribute to weakness by unknown mechanisms. Striational antibodies are not specific for MG and may occur in patients with other autoimmune diseases and in patients with thymoma without MG [72]. In the setting of MG, these antibodies only rarely occur in the absence of anti-AChR antibodies and thus are not generally useful (in isolation) for the diagnosis of MG. However, striational antibodies may be helpful in the diagnosis of thymomatous MG and can reflect thymic pathology [73]. As a marker of thymoma, they are most useful in patients with anti-AChR-positive MG with onset before the age of 40 years. They are more frequent in older MG patients and in those with more severe disease, with or without thymoma, suggesting that disease severity may be related to a more heterogeneous immune attack against multiple muscle antigens [74].
One of the major antigenic targets of striational antibodies is titin, an intracellular protein with a molecular mass of 3000 kDa stretching throughout the sarcomere, with a length of more than 1 μm, making it the largest known protein [75]. Anti-titin antibodies are present in 70–90% of thymoma MG patients, and in approximately 50% of late-onset AChR-MG patients without thymoma [76–78]. In general, anti-titin antibodies correlate with disease severity [77,78] and may identify patients more likely to be refractory to therapy, including thymectomy. The coexistence of myopathy (see below) and disturbed neuromuscular transmission in titin antibody-positive MG patients may explain the more severe muscle weakness frequently observed in these patients. Anti-titin antibodies are rarely present in early-onset, anti-AChR-positive MG patients and have not been reported in MuSK MG or in double-seronegative MG.
The mechanisms underlying the production of anti-titin antibodies and mediating potential pathogenicity in MG patients are poorly characterized, but titin is known to be important for myofibrillogenesis and sarcomere structure and elasticity [75]. Anti-titin antibodies belong mainly to the IgG1 subclass and can activate complement in vitro [79]. Peripheral blood mononuclear cells from patients with anti-titin antibody-positive MG proliferate when cultured with the main immunogenic region of titin, indicative of a titin-specific T-cell response [80]. Since titin antibodies are not usually found in early-onset MG patients, it is unlikely that their production is triggered by focal end-plate lysis caused by anti-AChR antibodies. Titin is expressed in both hyperplastic thymus and thymoma tissue, potentially providing a thymic site for primary autosensitization against titin antigens [81,82]. However, this does not explain the presence of anti-titin antibodies in late-onset MG patients without thymoma or their absence in patients with early-onset MG and thymic hyperplasia.
The ryanodine receptor (RyR) is the calcium channel of the sarcoplasmic reticulum. Upon opening, the RyR releases Ca2+ into the sarcoplasm resulting in muscle contraction, and therefore plays a crucial role in excitation–contraction coupling [83]. RyR antibodies are closely associated with thymomatous MG, and patients typically have prominent bulbar and respiratory muscle weakness [84,85]. RyR antibodies are mainly of the IgG1 and IgG3 subclass [86], with the ability to activate complement, and in vitro can inhibit Ca2+ release from the sarcoplasmic reticulum [87]. Interestingly, RyR antibodies from MG patients bind to both the skeletal and cardiac muscle forms of the RyR [88], perhaps underlying the association with cardiac disease and sudden death. Some reports also suggest excitation–contraction coupling defects in thymomatous MG with RyR antibodies [89].
Striational antibodies may also target several other myofibrillar proteins that play a role in muscle contraction. Antibodies against myosin, actin, tropomyosin and troponin have been demonstrated in the serum of MG patients [90,91], but their clinical significance is unclear.
Striational antibodies: diagnosis & disease monitoring
The titin antibody assay is clinically useful since 95% of thymomatous MG patients have anti-titin antibodies, a sensitivity that is essentially equivalent to CT scanning of the chest [77,78,92]. Unfortunately, the specificity of titin antibodies for thymoma is low as approximately 50% of all late-onset MG patients may harbor these antibodies, with or without thymoma [8,77,78]. RyR antibodies are more specific for thymoma but are found in only approximately 75% of thymomatous MG patients [8]. The combination of titin and RyR antibody positivity is approximately 95% sensitive and 70% specific for thymoma in MG [8,92]. RyR antibodies are positively associated with invasive/malignant thymoma [77], so that their presence in an MG patient undergoing thymectomy should alert the surgeon to choose a technique that assures complete exploration and removal. Titin and RyR antibodies may also be associated with the presence of myositis and myocarditis, suggesting that these antibodies may target skeletal and cardiac muscle antigens [93]. As the presence of titin and RyR antibodies correlates with MG severity, the antibody status has been proposed to be potentially useful when assessing disease prognosis, treatment and follow-up [77]. However, there are no published studies examining the use of these antibodies as biomarkers for therapeutic response.
Antibodies to non-AChR ion channels
Anti-Kv1.4 antibodies that target α-subunits (Kv1.4) of the voltage-gated potassium channel (VGKC) have been reported in 12–28% of Japanese MG patients [94,95]. The presence of these antibodies is correlated with bulbar symptoms, myasthenic crisis, thymoma, myocarditis and prolonged QT time on electrocardiography and may identify a subgroup of patients with a favorable response to calcineurin inhibitors [94]. Interestingly, while anti-Kv1.4 antibodies were present in severe disease with bulbar weakness in Japanese patients, they have recently been found in mild or predominantly ocular MG in a Caucasian cohort [96]. These antibodies are present in MG without clinical or electrical neuromyotonia, suggesting that the targeted antigen may not be the neuronal VGKC, but perhaps the VGKC on muscle fibers.
Anti-muscle antibodies: summary
A number of skeletal muscle proteins with functional relevance to the AChR may be targeted by autoantibodies in MG patients. Their intracellular location suggests that they are not pathogenic, but their presence is associated with distinctive clinical features (thymoma, myositis and myocarditis) and more severe disease with prominent oropharyngeal and respiratory involvement. While the presence of titin and ryanodine antibodies correlates with MG severity, the practical usefulness of striational muscle antibody levels in prognosis and predicting treatment effect remains to be investigated.
Antibodies to non-AChR NMJ proteins
Rapsyn is an intracellular end-plate protein that is necessary for the clustering of AChRs at the postsynaptic folds of the NMJ [97]. Anti-rapsyn antibodies have been found in almost 15% of MG patients, most commonly in thymomatous MG [92], but have also been found in patients with other autoimmune diseases [98]. Antibodies to end-plate acetylcholinesterase have also been reported in MG patients [99], but the pathogenic role of these antibodies is questionable since they are present in other autoimmune diseases and healthy controls [100].
As noted above, low-density LRP4 is an end-plate protein that, along with MuSK, serves as an agrin receptor and is required for AChR clustering and normal NMJ formation [49]. Antibodies to LRP4 have been detected in the serum of double-seronegative MG patients by three groups to date [101–103]. Most recently, these antibodies were found in 9.2% of patients with double-seronegative MG, but not in anti-AChR or anti-MuSK positive patients [103]. These antibodies appeared to be specific for MG, as they were not found in patients with other neurologic or psychiatric diseases. Most LRP4 antibodies appeared to be IgG1, which would indicate that they would be able to activate complement. LRP4 autoanti-bodies may alter the agrin-signaling pathway and have an adverse effect on AChR clustering and the agrin–LRP4 interaction [49,103], but the mechanism(s) responsible for the potential pathogenicity of LRP4 antibodies remains to be studied.
Expert commentary & five-year view
Currently, two well-characterized autoantibodies are known to be specific for, and to play a causative role in MG; namely, the anti-AChR and the anti-MuSK autoantibodies. While very useful for diagnostic purposes, these autoantibodies have questionable utility for disease monitoring, with the possible exception of anti-MuSK antibodies. It is important to realize that the status of current knowledge of the autoantibody repertoire of MG patients is limited to studies that utilized varied techniques to detect autoantibodies, and in many cases were performed before the identification of anti-MuSK antibodies. Furthermore, it is becoming apparent that MG is not a single disease, but is probably comprised of a number of clinical subtypes that may be distinguished not only by their auto-antibody profile, but also by thymic pathology and clinical disease presentation [2]. Future studies examining autoantibody profiles in MG patients will need to take this into consideration by subdividing patients into relevant clinical MG subtypes to specifically determine the usefulness of various muscle autoantibody measurements in their diagnosis and monitoring. Further investigations will also need to determine IgG subclass, as a switch in subclass may be an important sign of disease state and/or response to treatment.
AChR and MuSK autoantibodies in MG are currently detected using radioimmunopreciptation assays, with the associated concerns related to the use of radioactive materials. The development of cell-based and fluorescence immunoprecipitation assays will likely provide nonradioactive alternatives in the future, with equal or enhanced sensitivity [104]. In addition, techniques for measuring muscle autoantibodies have been limited by the small number of purified proteins (i.e., extracted AChRs) available as sources of self-antigens. The development of new assays that allow for large-scale and high-throughput autoantibody analysis could enhance our understanding of the autoantibody repertoire in MG, and how it may change during the course of disease and in response to therapy. Antigen arrays allow the study of antibody reactivity against a large number of antigens using small volumes of fluid with greater sensitivity than ELISA [105]. The parallel detection of antibodies with different specificities in human serum, a procedure also called antibody profiling, would greatly enhance our ability to characterize MG disease subtypes on the basis of autoantigen reactivity and could potentially provide a much-needed immunologic biomarker for MG.
Key issues.
Two well-characterized autoantibodies playing a role in disease pathogenesis are found in the serum of most patients with myasthenia gravis (MG) – anti-acetylcholine receptor (AChR) and anti-muscle-specific tyrosine kinase (MuSK) antibodies.
While anti-AChR antibody levels generally correlate with disease severity and response to treatment, the absolute concentration of antibodies does not precisely predict disease severity in all MG patients or therapeutic response in individual patients.
Antibodies to striated muscle proteins generally occur in anti-AChR positive MG and aid in identifying patients with thymoma, myositis or cardiomyopathy.
Anti-MuSK and anti-striational antibodies (titin and ryanodine receptor) are correlated with disease severity and may predict treatment response but have not been studied in large enough patient numbers to recommend their use as disease biomarkers.
Autoantibodies to other end-plate proteins may play a role in patients with anti-AChR- and anti-MuSK negative MG. Recent studies have identified anti-lipoprotein receptor-related protein 4 antibodies in approximately 9% of double-seronegative generalized MG patients.
Footnotes
For reprint orders, please contact reprints@expert-reviews.com
Financial & competing interests disclosure
MN Meriggioli is a consultant and serves on the speakers bureau for Athena Diagnostics. He receives funding from the NIH (grant no. K08NS058800; National Institute of Neurologic Disorders and Stroke), Gliknik Inc. and the Muscular Dystrophy Association (MDA 34545 and MDA 185924). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
Papers of special note have been highlighted as:
• of interest
•• of considerable interest
- 1.Phillips LH., 2nd The epidemiology of myasthenia gravis. Ann NY Acad Sci. 2003;998:407–412. doi: 10.1196/annals.1254.053. [DOI] [PubMed] [Google Scholar]
- 2.Meriggioli MN, Sanders DB. Autoimmune myasthenia gravis: emerging clinical and biological heterogeneity. Lancet Neurol. 2009;8(5):475–490. doi: 10.1016/S1474-4422(09)70063-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Drachman DB. Myasthenia gravis. N Engl J Med. 1994;330(25):1797–1810. doi: 10.1056/NEJM199406233302507. [DOI] [PubMed] [Google Scholar]
- 4.Patrick J, Lindstrom J. Autoimmune response to acetylcholine receptor. Science. 1973;180(4088):871–872. doi: 10.1126/science.180.4088.871. [DOI] [PubMed] [Google Scholar]
- 5•.Hoch W, McConville J, Helms S, Newsom-Davis J, Melms A, Vincent A. Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med. 2001;7(3):365–368. doi: 10.1038/85520. Original report of anti-muscle-specific tyrosine kinase (MuSK) antibodies in anti-acetylcholine receptor (AChR) seronegative generalized myasthenia gravis (MG) patients. [DOI] [PubMed] [Google Scholar]
- 6.McConville J, Farrugia ME, Beeson D, et al. Detection and characterization of MuSK antibodies in seronegative myasthenia gravis. Ann Neurol. 2004;55(4):580–584. doi: 10.1002/ana.20061. [DOI] [PubMed] [Google Scholar]
- 7•.Leite MI, Jacob S, Viegas S, et al. IgG1 antibodies to acetylcholine receptors in ‘seronegative’ myasthenia gravis. Brain. 2008;131(Pt 7):1940–1952. doi: 10.1093/brain/awn092. Reports the presence of ‘low-affinity’ antibodies to the AChR in approximately 66% of patients in a cohort of double-seronegatiove MG patients using a cell-based immunoassay. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Romi F, Skeie GO, Aarli JA, Gilhus NE. Muscle autoantibodies in subgroups of myasthenia gravis patients. J Neurol. 2000;247(5):369–375. doi: 10.1007/s004150050604. [DOI] [PubMed] [Google Scholar]
- 9.Drachman DB, Adams RN, Stanley EF, Pestronk A. Mechanisms of acetylcholine receptor loss in myasthenia gravis. J Neurol Neurosurg Psychiatr. 1980;43(7):601–610. doi: 10.1136/jnnp.43.7.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Toyka KV, Drachman DB, Griffin DE, et al. Myasthenia gravis. Study of humoral immune mechanisms by passive transfer to mice. N Engl J Med. 1977;296(3):125–131. doi: 10.1056/NEJM197701202960301. [DOI] [PubMed] [Google Scholar]
- 11.Lefvert AK. Immunoglobulins in myasthenia gravis. Kinetic properties of the acetylcholine-receptor antibody studied during lymph drainage. Clin Exp Immunol. 1978;34(1):111–117. [PMC free article] [PubMed] [Google Scholar]
- 12.Newsom-Davis J, Pinching AJ, Vincent A, Wilson SG. Function of circulating antibody to acetylcholine receptor in myasthenia gravis: investigation by plasma exchange. Neurology. 1978;28(3):266–272. doi: 10.1212/wnl.28.3.266. [DOI] [PubMed] [Google Scholar]
- 13.Lindstrom J. An assay for antibodies to human acetylcholine receptor in serum from patients with myasthenia gravis. Clin Immunol Immunopathol. 1977;7(1):36–43. doi: 10.1016/0090-1229(77)90027-7. [DOI] [PubMed] [Google Scholar]
- 14.Tzartos SJ, Lindstrom JM. Monoclonal antibodies used to probe acetylcholine receptor structure: localization of the main immunogenic region and detection of similarities between subunits. Proc Natl Acad Sci USA. 1980;77(2):755–759. doi: 10.1073/pnas.77.2.755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gomez AM, Van Den Broeck J, Vrolix K, et al. Antibody effector mechanisms in myasthenia gravis-pathogenesis at the neuromuscular junction. Autoimmunity. 2010;43(5–6):353–370. doi: 10.3109/08916930903555943. [DOI] [PubMed] [Google Scholar]
- 16.Willcox N, Leite MI, Kadota Y, et al. Autoimmunizing mechanisms in thymoma and thymus. Ann NY Acad Sci. 2008;1132:163–173. doi: 10.1196/annals.1405.021. [DOI] [PubMed] [Google Scholar]
- 17.Schluep M, Willcox N, Vincent A, Dhoot GK, Newsom-Davis J. Acetylcholine receptors in human thymic myoid cells in situ: an immunohistological study. Ann Neurol. 1987;22(2):212–222. doi: 10.1002/ana.410220205. [DOI] [PubMed] [Google Scholar]
- 18.Morgenthaler TI, Brown LR, Colby TV, Harper CM, Jr, Coles DT. Thymoma. Mayo Clin Proc. 1993;68(11):1110–1123. doi: 10.1016/s0025-6196(12)60907-0. [DOI] [PubMed] [Google Scholar]
- 19.Aarli J. Myasthenia gravis and thymoma. In: Lisak RP, editor. Handbook of Myasthenia Gravis and Myasthenic Syndromes. Marcel Dekker, Inc; NY, USA: pp. 207–224. [Google Scholar]
- 20.Heidenreich F, Vincent A, Willcox N, Newsom-Davis J. Anti-acetylcholine receptor antibody specificities in serum and in thymic cell culture supernatants from myasthenia gravis patients. Neurology. 1988;38(11):1784–1788. doi: 10.1212/wnl.38.11.1784. [DOI] [PubMed] [Google Scholar]
- 21.Gilhus NE, Aarli JA, Matre R. Myasthenia gravis: the specificities of skeletal muscle and thymus antibodies. Acta Neurol Scand. 1983;68(5):328–336. doi: 10.1111/j.1600-0404.1983.tb04840.x. [DOI] [PubMed] [Google Scholar]
- 22.Hohlfeld R, Kalies I, Kohleisen B, Heininger K, Conti-Tronconi B, Toyka KV. Myasthenia gravis: stimulation of antireceptor autoantibodies by autoreactive T cell lines. Neurology. 1986;36(5):618–621. doi: 10.1212/wnl.36.5.618. [DOI] [PubMed] [Google Scholar]
- 23.Melms A, Schalke BC, Kirchner T, Müller-Hermelink HK, Albert E, Wekerle H. Thymus in myasthenia gravis. Isolation of T-lymphocyte lines specific for the nicotinic acetylcholine receptor from thymuses of myasthenic patients. J Clin Invest. 1988;81(3):902–908. doi: 10.1172/JCI113401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sommer N, Willcox N, Harcourt GC, Newsom-Davis J. Myasthenic thymus and thymoma are selectively enriched in acetylcholine receptor-reactive T cells. Ann Neurol. 1990;28(3):312–319. doi: 10.1002/ana.410280303. [DOI] [PubMed] [Google Scholar]
- 25.Sommer N, Harcourt GC, Willcox N, Beeson D, Newsom-Davis J. Acetylcholine receptor-reactive T lymphocytes from healthy subjects and myasthenia gravis patients. Neurology. 1991;41(8):1270–1276. doi: 10.1212/wnl.41.8.1270. [DOI] [PubMed] [Google Scholar]
- 26.Mygland A, Vincent A, Newsom-Davis J, et al. Autoantibodies in thymoma-associated myasthenia gravis with myositis or neuromyotonia. Arch Neurol. 2000;57(4):527–531. doi: 10.1001/archneur.57.4.527. [DOI] [PubMed] [Google Scholar]
- 27.Kadota Y, Okumura M, Miyoshi S, et al. Altered T cell development in human thymoma is related to impairment of MHC class II transactivator expression induced by interferon-gamma (IFN-gamma) Clin Exp Immunol. 2000;121(1):59–68. doi: 10.1046/j.1365-2249.2000.01256.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Scarpino S, Di Napoli A, Stoppacciaro A, et al. Expression of autoimmune regulator gene (AIRE) and T regulatory cells in human thymomas. Clin Exp Immunol. 2007;149(3):504–512. doi: 10.1111/j.1365-2249.2007.03442.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ströbel P, Rosenwald A, Beyersdorf N, et al. Selective loss of regulatory T cells in thymomas. Ann Neurol. 2004;56(6):901–904. doi: 10.1002/ana.20340. [DOI] [PubMed] [Google Scholar]
- 30•.Howard FM, Jr, Lennon VA, Finley J, Matsumoto J, Elveback LR. Clinical correlations of antibodies that bind, block, or modulate human acetylcholine receptors in myasthenia gravis. Ann NY Acad Sci. 1987;505:526–538. doi: 10.1111/j.1749-6632.1987.tb51321.x. Study examining the frequencies and clinical correlations of binding, blocking and modulating anti-AChR antibodies in 349 patients with MG. [DOI] [PubMed] [Google Scholar]
- 31.Lindstrom J. An assay for antibodies to human acetylcholine receptor in serum from patients with myasthenia gravis. Clin Immunol Immunopathol. 1977;7(1):36–43. doi: 10.1016/0090-1229(77)90027-7. [DOI] [PubMed] [Google Scholar]
- 32.Lindstrom JM, Seybold ME, Lennon VA, Whittingham S, Duane DD. Antibody to acetylcholine receptor in myasthenia gravis. Prevalence, clinical correlates, and diagnostic value. Neurology. 1976;26(11):1054–1059. doi: 10.1212/wnl.26.11.1054. [DOI] [PubMed] [Google Scholar]
- 33.Lefvert AK, Bergström K, Matell G, Osterman PO, Pirskanen R. Determination of acetylcholine receptor antibody in myasthenia gravis: clinical usefulness and pathogenetic implications. J Neurol Neurosurg Psychiatr. 1978;41(5):394–403. doi: 10.1136/jnnp.41.5.394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vincent A, Newsom-Davis J. Acetylcholine receptor antibody as a diagnostic test for myasthenia gravis: results in 153 validated cases and 2967 diagnostic assays. J Neurol Neurosurg Psychiatr. 1985;48(12):1246–1252. doi: 10.1136/jnnp.48.12.1246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Agius MA, Richman DP, Vincent A. Autoantibody testing in the diagnosis and management of autoimmune disorders of neuromuscular transmission and related disorders. In: Kaminski HJ, editor. Myasthenia Gravis and Related Disorders. 2. Humana Press; NJ, USA: 2009. pp. 143–156. [Google Scholar]
- 36.Vincent A, Newsom-Davis J. Acetylcholine receptor antibody characteristics in myasthenia gravis. I Patients with generalized myasthenia or disease restricted to ocular muscles. Clin Exp Immunol. 1982;49(2):257–265. [PMC free article] [PubMed] [Google Scholar]
- 37.Lefvert AK, Björkholm M. Antibodies against the acetylcholine receptor in hematologic disorders: implications for the development of myasthenia gravis after bone marrow grafting. N Engl J Med. 1987;317(3):170. doi: 10.1056/NEJM198707163170315. [DOI] [PubMed] [Google Scholar]
- 38.Lennon VA. Serological diagnosis of myasthenia gravis and Lambert–Eaton myasthenic syndrome. In: Lisak RP, editor. Handbook of Myasthenia Gravis and Myasthenic Syndromes. Marcel Dekker; NY, USA: 1994. pp. 149–164. [Google Scholar]
- 39.McKeon A, Lennon VA, Jacob A, et al. Coexistence of myasthenia gravis and serological markers of neurological autoimmunity in neuromyelitis optica. Muscle Nerve. 2009;39(1):87–90. doi: 10.1002/mus.21197. [DOI] [PubMed] [Google Scholar]
- 40.Chan KH, Lachance DH, Harper CM, Lennon VA. Frequency of seronegativity in adult-acquired generalized myasthenia gravis. Muscle Nerve. 2007;36(5):651–658. doi: 10.1002/mus.20854. [DOI] [PubMed] [Google Scholar]
- 41.Vernino S, Lennon VA. Autoantibody profiles and neurological correlations of thymoma. Clin Cancer Res. 2004;10(21):7270–7275. doi: 10.1158/1078-0432.CCR-04-0735. [DOI] [PubMed] [Google Scholar]
- 42.Tindall RS. Humoral immunity in myasthenia gravis: biochemical characterization of acquired antireceptor antibodies and clinical correlations. Ann Neurol. 1981;10(5):437–447. doi: 10.1002/ana.410100506. [DOI] [PubMed] [Google Scholar]
- 43.Jaretzki A, 3rd, Barohn RJ, Ernstoff RM, et al. Myasthenia gravis: recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Neurology. 2000;55(1):16–23. doi: 10.1212/wnl.55.1.16. [DOI] [PubMed] [Google Scholar]
- 44.Besinger UA, Toyka KV, Hömberg M, Heininger K, Hohlfeld R, Fateh-Moghadam A. Myasthenia gravis: long-term correlation of binding and bungarotoxin blocking antibodies against acetylcholine receptors with changes in disease severity. Neurology. 1983;33(10):1316–1321. doi: 10.1212/wnl.33.10.1316. [DOI] [PubMed] [Google Scholar]
- 45.Oosterhuis HJ, Limburg PC, Hummel-Tappel E, The TH. Anti-acetylcholine receptor antibodies in myasthenia gravis. Part 2 Clinical and serological follow-up of individual patients. J Neurol Sci. 1983;58(3):371–385. doi: 10.1016/0022-510x(83)90096-5. [DOI] [PubMed] [Google Scholar]
- 46.Oosterhuis HJ, Limburg PC, Hummel-Tappel E, Van den Burg W, The TH. Anti-acetylcholine receptor antibodies in myasthenia gravis. Part 3 The effect of thymectomy. J Neurol Sci. 1985;69(3):335–343. doi: 10.1016/0022-510x(85)90144-3. [DOI] [PubMed] [Google Scholar]
- 47.Tindall RS. Humoral immunity in myasthenia gravis: effect of steroids and thymectomy. Neurology. 1980;30(5):554–557. doi: 10.1212/wnl.30.5.557. [DOI] [PubMed] [Google Scholar]
- 48.Vincent A, Newsom-Davis J, Newton P, Beck N. Acetylcholine receptor antibody and clinical response to thymectomy in myasthenia gravis. Neurology. 1983;33(10):1276–1282. doi: 10.1212/wnl.33.10.1276. [DOI] [PubMed] [Google Scholar]
- 49.Zong Y, Zhang B, Gu S, et al. Structural basis of agrin-LRP4-MuSK signaling. Genes Dev. 2012;26(3):247–258. doi: 10.1101/gad.180885.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Punga AR, Lin S, Oliveri F, Meinen S, Rüegg MA. Muscle-selective synaptic disassembly and reorganization in MuSK antibody positive MG mice. Exp Neurol. 2011;230(2):207–217. doi: 10.1016/j.expneurol.2011.04.018. [DOI] [PubMed] [Google Scholar]
- 51.Shiraishi H, Motomura M, Yoshimura T, et al. Acetylcholine receptors loss and postsynaptic damage in MuSK antibody-positive myasthenia gravis. Ann Neurol. 2005;57(2):289–293. doi: 10.1002/ana.20341. [DOI] [PubMed] [Google Scholar]
- 52.Cole RN, Reddel SW, Gervásio OL, Phillips WD. Anti-MuSK patient antibodies disrupt the mouse neuromuscular junction. Ann Neurol. 2008;63(6):782–789. doi: 10.1002/ana.21371. [DOI] [PubMed] [Google Scholar]
- 53.Shigemoto K, Kubo S, Maruyama N, et al. Induction of myasthenia by immunization against muscle-specific kinase. J Clin Invest. 2006;116(4):1016–1024. doi: 10.1172/JCI21545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jha S, Xu K, Maruta T, et al. Myasthenia gravis induced in mice by immunization with the recombinant extracellular domain of rat muscle-specific kinase (MuSK) J Neuroimmunol. 2006;175(1–2):107–117. doi: 10.1016/j.jneuroim.2006.03.016. [DOI] [PubMed] [Google Scholar]
- 55.ter Beek WP, Martínez-Martínez P, Losen M, et al. The effect of plasma from muscle-specific tyrosine kinase myasthenia patients on regenerating endplates. Am J Pathol. 2009;175(4):1536–1544. doi: 10.2353/ajpath.2009.090040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lavrnic D, Losen M, Vujic A, et al. The features of myasthenia gravis with autoantibodies to MuSK. J Neurol Neurosurg Psychiatr. 2005;76(8):1099–1102. doi: 10.1136/jnnp.2004.052415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mori S, Kubo S, Akiyoshi T, et al. Antibodies against muscle-specific kinase impair both presynaptic and postsynaptic functions in a murine model of myasthenia gravis. Am J Pathol. 2012;180(2):798–810. doi: 10.1016/j.ajpath.2011.10.031. [DOI] [PubMed] [Google Scholar]
- 58.Mori S, Yamada S, Kubo S, et al. Divalent and monovalent autoantibodies cause dysfunction of MuSK by distinct mechanisms in a rabbit model of myasthenia gravis. J Neuroimmunol. 2012;244(1–2):1–7. doi: 10.1016/j.jneuroim.2011.12.005. [DOI] [PubMed] [Google Scholar]
- 59.Klooster R, Plomp JJ, Huijbers MG, et al. Muscle-specific kinase myasthenia gravis IgG4 autoantibodies cause severe neuromuscular junction dysfunction in mice. Brain. 2012;135(Pt 4):1081–1101. doi: 10.1093/brain/aws025. [DOI] [PubMed] [Google Scholar]
- 60.Kawakami Y, Ito M, Hirayama M, et al. Anti-MuSK autoantibodies block binding of collagen Q to MuSK. Neurology. 2011;77(20):1819–1826. doi: 10.1212/WNL.0b013e318237f660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hatanaka Y, Hemmi S, Morgan MB, et al. Nonresponsiveness to anticholinesterase agents in patients with MuSK-antibody-positive MG. Neurology. 2005;65(9):1508–1509. doi: 10.1212/01.wnl.0000183145.91579.74. [DOI] [PubMed] [Google Scholar]
- 62.Zhu D, Yang Z, Luo Z, Luo S, Xiong WC, Mei L. Muscle-specific receptor tyrosine kinase endocytosis in acetylcholine receptor clustering in response to agrin. J Neurosci. 2008;28(7):1688–1696. doi: 10.1523/JNEUROSCI.4130-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63•.Guptill JT, Sanders DB, Evoli A. Anti-MuSK antibody myasthenia gravis: clinical findings and response to treatment in two large cohorts. Muscle Nerve. 2011;44(1):36–40. doi: 10.1002/mus.22006. Report of clinical features of two large cohorts of MuSK MG patients. [DOI] [PubMed] [Google Scholar]
- 64.Evoli A, Tonali PA, Padua L, et al. Clinical correlates with anti-MuSK antibodies in generalized seronegative myasthenia gravis. Brain. 2003;126(Pt 10):2304–2311. doi: 10.1093/brain/awg223. [DOI] [PubMed] [Google Scholar]
- 65.Ohta K, Shigemoto K, Kubo S, et al. MuSK antibodies in AChR Ab-seropositive MG vs AChR Ab-seronegative MG. Neurology. 2004;62(11):2132–2133. doi: 10.1212/01.wnl.0000129274.12702.92. [DOI] [PubMed] [Google Scholar]
- 66.Sanders DB, El-Salem K, Massey JM, McConville J, Vincent A. Clinical aspects of MuSK antibody positive seronegative MG. Neurology. 2003;60(12):1978–1980. doi: 10.1212/01.wnl.0000065882.63904.53. [DOI] [PubMed] [Google Scholar]
- 67.Suzuki S, Utsugisawa K, Nagane Y, Satoh T, Kuwana M, Suzuki N. Clinical and immunological differences between early and late-onset myasthenia gravis in Japan. J Neuroimmunol. 2011;230(1–2):148–152. doi: 10.1016/j.jneuroim.2010.10.023. [DOI] [PubMed] [Google Scholar]
- 68.Sanders DB, Juel VC. MuSK-antibody positive myasthenia gravis: questions from the clinic. J Neuroimmunol. 2008;201–202:85–89. doi: 10.1016/j.jneuroim.2008.05.032. [DOI] [PubMed] [Google Scholar]
- 69.Leite MI, Ströbel P, Jones M, et al. Fewer thymic changes in MuSK antibody-positive than in MuSK antibody-negative MG. Ann Neurol. 2005;57(3):444–448. doi: 10.1002/ana.20386. [DOI] [PubMed] [Google Scholar]
- 70••.Bartoccioni E, Scuderi F, Minicuci GM, Marino M, Ciaraffa F, Evoli A. Anti-MuSK antibodies: correlation with myasthenia gravis severity. Neurology. 2006;67(3):505–507. doi: 10.1212/01.wnl.0000228225.23349.5d. Anti-MuSK antibodies were measured in 83 serum samples from 40 patients; a correlation between antibody levels and disease severity was found. Anti-MuSK antibody concentrations were often reduced by immunosuppression but not after thymectomy. [DOI] [PubMed] [Google Scholar]
- 71.Niks EH, van Leeuwen Y, Leite MI, et al. Clinical fluctuations in MuSK myasthenia gravis are related to antigen-specific IgG4 instead of IgG1. J Neuroimmunol. 2008;195(1–2):151–156. doi: 10.1016/j.jneuroim.2008.01.013. [DOI] [PubMed] [Google Scholar]
- 72.Strauss AJ, Kemp PG., Jr Serum autoantibodies in myasthenia gravis and thymoma: selective affinity for I-bands of striated muscle as a guide to identification of antigen(s) J Immunol. 1967;99(5):945–953. [PubMed] [Google Scholar]
- 73.Marx A, Willcox N, Leite MI, et al. Thymoma and paraneoplastic myasthenia gravis. Autoimmunity. 2010;43(5–6):413–427. doi: 10.3109/08916930903555935. [DOI] [PubMed] [Google Scholar]
- 74.Romi F, Gilhus NE, Varhaug JE, Myking A, Skeie GO, Aarli JA. Thymectomy and anti-muscle autoantibodies in late-onset myasthenia gravis. Eur J Neurol. 2002;9(1):55–61. doi: 10.1046/j.1468-1331.2002.00352.x. [DOI] [PubMed] [Google Scholar]
- 75.Wang K, McClure J, Tu A. Titin: major myofibrillar components of striated muscle. Proc Natl Acad Sci USA. 1979;76(8):3698–3702. doi: 10.1073/pnas.76.8.3698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yamamoto AM, Gajdos P, Eymard B, et al. Anti-titin antibodies in myasthenia gravis: tight association with thymoma and heterogeneity of nonthymoma patients. Arch Neurol. 2001;58(6):885–890. doi: 10.1001/archneur.58.6.885. [DOI] [PubMed] [Google Scholar]
- 77.Romi F, Skeie GO, Aarli JA, Gilhus NE. The severity of myasthenia gravis correlates with the serum concentration of titin and ryanodine receptor antibodies. Arch Neurol. 2000;57(11):1596–1600. doi: 10.1001/archneur.57.11.1596. [DOI] [PubMed] [Google Scholar]
- 78.Skeie GO, Mygland A, Aarli JA, Gilhus NE. Titin antibodies in patients with late onset myasthenia gravis: clinical correlations. Autoimmunity. 1995;20(2):99–104. doi: 10.3109/08916939509001933. [DOI] [PubMed] [Google Scholar]
- 79.Romi F, Skeie GO, Vedeler C, Aarli JA, Zorzato F, Gilhus NE. Complement activation by titin and ryanodine receptor autoantibodies in myasthenia gravis. A study of IgG subclasses and clinical correlations. J Neuroimmunol. 2000;111(1–2):169–176. doi: 10.1016/s0165-5728(00)00394-5. [DOI] [PubMed] [Google Scholar]
- 80.Skeie GO, Aarli JA, Matre R, et al. Titin antibody positive myasthenia gravis patients have a cellular immune response against the main immunogenic region of titin. Eur J Neurol. 1997;4:131–137. doi: 10.1111/j.1468-1331.1997.tb00318.x. [DOI] [PubMed] [Google Scholar]
- 81.Romi F, Bø L, Skeie GO, Myking A, Aarli JA, Gilhus NE. Titin and ryanodine receptor epitopes are expressed in cortical thymoma along with costimulatory molecules. J Neuroimmunol. 2002;128(1–2):82–89. doi: 10.1016/s0165-5728(02)00145-5. [DOI] [PubMed] [Google Scholar]
- 82.Skeie GO, Freiburg A, Kolmerer B, et al. Titin transcripts in thymomas. J Autoimmun. 1997;10(6):551–557. doi: 10.1006/jaut.1997.0162. [DOI] [PubMed] [Google Scholar]
- 83.Coronado R, Morrissette J, Sukhareva M, Vaughan DM. Structure and function of ryanodine receptors. Am J Physiol. 1994;266(6 Pt 1):C1485–C1504. doi: 10.1152/ajpcell.1994.266.6.C1485. [DOI] [PubMed] [Google Scholar]
- 84.Mygland A, Aarli JA, Matre R, Gilhus NE. Ryanodine receptor antibodies related to severity of thymoma associated myasthenia gravis. J Neurol Neurosurg Psychiatr. 1994;57(7):843–846. doi: 10.1136/jnnp.57.7.843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Romi F, Aarli JA, Gilhus NE. Myasthenia gravis patients with ryanodine receptor antibodies have distinctive clinical features. Eur J Neurol. 2007;14(6):617–620. doi: 10.1111/j.1468-1331.2007.01785.x. [DOI] [PubMed] [Google Scholar]
- 86.Mygland A, Tysnes OB, Aarli JA, Matre R, Gilhus NE. IgG subclass distribution of ryanodine receptor autoantibodies in patients with myasthenia gravis and thymoma. J Autoimmun. 1993;6(4):507–515. doi: 10.1006/jaut.1993.1042. [DOI] [PubMed] [Google Scholar]
- 87.Skeie GO, Mygland A, Treves S, Gilhus NE, Aarli JA, Zorzato F. Ryanodine receptor antibodies in myasthenia gravis: epitope mapping and effect on calcium release in vitro. Muscle Nerve. 2003;27(1):81–89. doi: 10.1002/mus.10294. [DOI] [PubMed] [Google Scholar]
- 88.Mygland A, Tysnes OB, Matre R, Aarli JA, Gilhus NE. Anti-cardiac ryanodine receptor antibodies in thymoma-associated myasthenia gravis. Autoimmunity. 1994;17(4):327–331. doi: 10.3109/08916939409010673. [DOI] [PubMed] [Google Scholar]
- 89.Skeie GO, Lunde PK, Sejersted OM, Mygland A, Aarli JA, Gilhus NE. Myasthenia gravis sera containing antiryanodine receptor antibodies inhibit binding of [3H]-ryanodine to sarcoplasmic reticulum. Muscle Nerve. 1998;21(3):329–335. doi: 10.1002/(sici)1097-4598(199803)21:3<329::aid-mus6>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 90.Ohta M, Ohta K, Itoh N, Kurobe M, Hayashi K, Nishitani H. Anti-skeletal muscle antibodies in the sera from myasthenic patients with thymoma: identification of anti-myosin, actomyosin, actin, and alpha-actinin antibodies by a solid-phase radioimmunoassay and a western blotting analysis. Clin Chim Acta. 1990;187(3):255–264. doi: 10.1016/0009-8981(90)90110-e. [DOI] [PubMed] [Google Scholar]
- 91.Yamamoto T, Sato T, Sugita H. Antifilamin, antivinculin, and antitropomyosin antibodies in myasthenia gravis. Neurology. 1987;37(8):1329–1333. doi: 10.1212/wnl.37.8.1329. [DOI] [PubMed] [Google Scholar]
- 92.Skeie GO, Romi F. Paraneoplastic myasthenia gravis: immunological and clinical aspects. Eur J Neurol. 2008;15(10):1029–1033. doi: 10.1111/j.1468-1331.2008.02242.x. [DOI] [PubMed] [Google Scholar]
- 93.Suzuki S, Utsugisawa K, Yoshikawa H, et al. Autoimmune targets of heart and skeletal muscles in myasthenia gravis. Arch Neurol. 2009;66(11):1334–1338. doi: 10.1001/archneurol.2009.229. [DOI] [PubMed] [Google Scholar]
- 94.Suzuki S, Utsugisawa K, Nagane Y, Suzuki N. Three types of striational antibodies in myasthenia gravis. Autoimmune Dis. 2011;2011:740583. doi: 10.4061/2011/740583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Suzuki S, Satoh T, Yasuoka H, et al. Novel autoantibodies to a voltage-gated potassium channel Kv1.4 in a severe form of myasthenia gravis. J Neuroimmunol. 2005;170(1–2):141–149. doi: 10.1016/j.jneuroim.2005.08.017. [DOI] [PubMed] [Google Scholar]
- 96.Romi F, Suzuki S, Suzuki N, Petzold A, Plant GT, Gilhus NE. Anti-voltage-gated potassium channel Kv1.4 antibodies in myasthenia gravis. J Neurol. 2011;259(7):1312–1316. doi: 10.1007/s00415-011-6344-y. [DOI] [PubMed] [Google Scholar]
- 97.Agius MA, Zhu S, Kirvan CA, et al. Rapsyn antibodies in myasthenia gravis. Ann NY Acad Sci. 1998;841:516–521. doi: 10.1111/j.1749-6632.1998.tb10972.x. [DOI] [PubMed] [Google Scholar]
- 98.Agius MA, Zhu S, Aarli JA. Antirapsyn antibodies occur commonly in patients with lupus. Ann NY Acad Sci. 1998;841:525–526. doi: 10.1111/j.1749-6632.1998.tb10974.x. [DOI] [PubMed] [Google Scholar]
- 99.Phillips TM, Manz HJ, Smith FA, Jaffe HA, Cohan SL. The detection of anti-cholinesterase antibodies in myasthenia gravis. Ann NY Acad Sci. 1981;377:360–371. doi: 10.1111/j.1749-6632.1981.tb33744.x. [DOI] [PubMed] [Google Scholar]
- 100.Geen J, Howells RC, Ludgate M, Hullin DA, Hogg SI. The prevalence of anti-acetylcholinesterase antibodies in autoimmune disease. Autoimmunity. 2004;37(8):579–585. doi: 10.1080/08916930400021360. [DOI] [PubMed] [Google Scholar]
- 101.Higuchi O, Hamuro J, Motomura M, Yamanashi Y. Autoantibodies to low-density lipoprotein receptor-related protein 4 in myasthenia gravis. Ann Neurol. 2011;69(2):418–422. doi: 10.1002/ana.22312. [DOI] [PubMed] [Google Scholar]
- 102.Pevzner A, Schoser B, Peters K, et al. Anti-LRP4 autoantibodies in AChR- and MuSK-antibody-negative myasthenia gravis. J Neurol. 2012;259(3):427–435. doi: 10.1007/s00415-011-6194-7. [DOI] [PubMed] [Google Scholar]
- 103••.Zhang B, Tzartos JS, Belimezi M, et al. Autoantibodies to lipoprotein-related protein 4 in patients with double-seronegative myasthenia gravis. Arch Neurol. 2012;69(4):445–451. doi: 10.1001/archneurol.2011.2393. In this study, anti-LRP4 antibodies were detected in 11 of 120 patients with MG without detectable anti-AChR or anti-MuSK antibodies (double seronegative) and in one of 36 patients without anti-AChR antibodies but with anti-MuSK antibodies. No healthy control subjects and only two of the 76 control patients with neurologic disease had anti-LRP4 antibodies. [DOI] [PubMed] [Google Scholar]
- 104.Yang L, Maxwell S, Leite MI, et al. Non-radioactive serological diagnosis of myasthenia gravis and clinical features of patients from Tianjin, China. J Neurol Sci. 2011;301(1–2):71–76. doi: 10.1016/j.jns.2010.10.023. [DOI] [PubMed] [Google Scholar]
- 105.Papp K, Prechl J. The use of antigen microarrays in antibody profiling. Methods Mol Biol. 2012;815:175–185. doi: 10.1007/978-1-61779-424-7_14. [DOI] [PubMed] [Google Scholar]