Abstract
The aim of this study was to investigate the molecular mechanism of 8-chloroadenosine 3′,5′-monophosphate (8-Cl-cAMP) in the inhibition of the growth and induction of apoptosis of multiple myeloma (MM) cells. Two MM-derived cell lines, RPMI-8226 and U266, were used. Cell viability, apoptosis induction and mitochondrial transmembrane potential were determined and the expression levels of cell cycle regulatory proteins (Cdk2, cyclin E, p27 and c-myc) and p38 mitogen-activated protein kinase (MAPK) protein were detected. Following treatment with 8-Cl-cAMP, the percentage of apoptotic cells increased in a concentration- and time-dependent manner and the mitochondrial transmembrane potential collapsed to reveal typical apoptotic features. Our data further demonstrated that 8-Cl-cAMP induced progressive phosphorylation of p38 MAPK and that the expression levels of p27 proteins in the MM cells were increased whereas those of c-myc were significantly decreased. Notably, the proapoptotic effect of 8-Cl-cAMP was largely prevented by a p38 MAPK inhibitor. Furthermore, knockdown of p27 was able to decrease the 8-Cl-cAMP-induced apoptosis in the MM cells. These results indicate that 8-Cl-cAMP induced p27-dependent cell cycle arrest and apoptosis in the MM cells, which demonstrates the potential of cAMP-modulating agents for use in the treatment of MM.
Keywords: 8-chloroadenosine 3′,5′-monophosphate; multiple myeloma; apoptosis; p27; cell cycle regulators
Introduction
Multiple myeloma (MM) is an incurable B-cell malignancy characterized by infiltrating, slow-growing plasma cells in the bone marrow that produce monoclonal immunoglobulin molecules (1). MM is more common than Hodgkin disease and acute leukemia and accounts for 1% of all cancers and slightly more than 10% of all hematological cancers. Few effective therapies have existed until recently. Alkylating agents and corticosteroids were the therapy of choice for several decades. A number of novel agents for MM have now become available, including the immunomodulatory drugs thalidomide and lenalidomide, as well as the proteasome inhibitor bortezomib (2–5). Although numerous therapeutic advances, including combined chemotherapy and hematopotietic stem cell transplantation, have been made to improve the survival of patients with MM, a high proportion of patients are not able to expect long term remission due to drug-resistant disease, minimal residual disease or infection. In order to overcome these limitations, certain new promising approaches are being widely studied (6,7).
As with unregulated proliferation, dysregulated apoptosis is also an important event in carcinogenesis. MM cells over-express the apoptotic antagonists bcl-2 and bcl-xL, although they are not overtly hyperproliferative (8,9). The protection against apoptosis may be significant in the expansion and/or resistance of the MM cell clones to chemotherapy. Modulation of apoptosis with adjuvant therapy may have the potential to improve the clinical outcome of MM. In further studies, the safety and efficacy of a combination therapy should be investigated (10).
Cyclic adenosine monophosphate (cAMP), a well-known and ubiquitous chemical messenger, has an antiproliferative effect on the majority of cell types (11,12). The best studied of its derivatives is 8-chloroadenosine 3′,5′-monophosphate (8-Cl-cAMP), which has demonstrated antiproliferative effects in vitro and in vivo and has been evaluated in phase I/II clinical trials (11,13,14). 8-Cl-cAMP is a site-selective analog of cAMP. It has been reported that 8-Cl-cAMP shows a potent growth inhibitory effect and has reverse-transforming activity in cancer cells (15–18). In the current study, we investigated the ability of 8-Cl-cAMP to induce apoptosis in two MM cell lines, RPMI-8226 and U266. Our results indicate that 8-Cl-cAMP-induced cellular apoptosis occurred in a concentration- and time-related manner with mitochondrial transmembrane potential collapse, increased expression levels of p27 and decreased expression levels of c-myc. p27 knockdown was able to decrease the 8-Cl-cAMP-induced apoptosis of the MM cells, indicating that the apoptotic action occured through a p27-dependent pathway.
Materials and methods
The study was approved by the Independent Ethics Committee of Shanghai Ninth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, China.
Reagents and cell culture
8-Cl-cAMP, propidium iodide (PI), rhodamine 123 (Rh123), SB202190 and other reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA). p27 siRNA was designed by Dharmacon (Lafayette, CO, USA). Fetal bovine serum (FBS), RPMI-1640 medium and penicillin-streptomycin were obtained from Gibco-BRL (Gaithersburg, MD, USA). All antibodies were purchased from Santa Cruz Biotechnology, (Santa Cruz, CA, USA. The ECL kit was purchased from Amersham Pharmacia Biotech (Amersham, UK). An Annexin V-FITC apoptosis detection kit, Oligofectamine and a mitochondrial membrane potential detection kit were purchased from Invitrogen (Eugene, OR, USA). The human myeloma cell lines RPMI-8226 and U266 (Shanghai Institute of Hematology, China) were cultured in RPMI-1640 medium supplemented with 10% FBS in a humidified atmosphere of 5% CO2 at 37°C.
Trypan blue exclusion assay
The effect of 8-Cl-cAMP on MM cell viability was measured by the trypan blue exclusion assay (19). RPMI-8226 and U266 cells were collected, mixed with an equal volume of PBS containing 0.4% trypan blue dye and manually counted. Actual cell numbers were calculated by multiplying by the dilution factor and were compared with the initial cell numbers. Cell viability (%) = viable cell numbers/total (viable + dead) cell numbers x 100.
Flow cytometric analysis of nuclear DNA distribution
Cells (2x106) were collected, rinsed and fixed overnight with 70% cold ethanol. They were then rinsed with PBS, treated with 1 mg/ml RNase at 37°C for 30 min and stained with 250 μg/ml PI. The nuclear DNA contents were detected by flow cytometry (Beckman Coulter, Miami, FL, USA). All data were collected, stored and analyzed by MultiCycle software.
Flow cytometric analysis of mitochondrial membrane potential
After washing with PBS twice, 1–2x105 RPMI-8226 cells were incubated with 10 μg/ml Rh123 at 37°C for 30 min. Subsequently, 250 μg/ml PI was injected into cells. Rh123 and PI staining intensities were determined by flow cytometry.
Western blot analysis
At appropriate time-points following treatment with 10 μmol/l 8-Cl-cAMP, the RPMI-8226 cells were collected. Protein extracts (100 μg) were loaded onto a 10% SDS-polyacrylamide gel, electrophoresed and transferred onto nitrocellulose membranes, which were subsequently stained with 0.2% Ponceau red to assure equal protein loading and transfer. After blocking with 10% non-fat milk powder, the membrane was incubated with primary antibody overnight at 4°C. The membrane was then washed with PBS and incubated with horseradish peroxidase-conjugated secondary antibody for 60 min at room temperature. The blots were again washed and the immunocomplex was visualized using the ECL kit.
Transfection of p27 siRNA and cell viability assay
The cells (1x104 cells/well) were seeded in a 96-well plate, incubated for 24 h to enable them to attach to the bottom of the well, and then transfected with 80 nM p27 siRNA or control siRNA using Oligofectamine. Following transfection for 12 h, the cells were treated with 10 μmol/l 8-Cl-cAMP for 48 h. Cell growth was was then determined by measuring the MTT dye absorbance of the living cells (20). Following exposure to 8-Cl-cAMP for the indicated time, 20 μl MTT solution (5 mg/ml in PBS) was added to each well and the plates were incubated for an additional 4 h at 37°C. The MTT solution in the medium was aspirated. To achieve solubilization of the formazan crystals formed in viable cells, 150 μl dimethylsulfoxide (DMSO) was added to each well and the absorbance at 570 nm was measured using a microplate reader (Bio-Rad, Hercules, CA, USA).
Statistical analysis
Each result in this study is representative of at least three separate experiments. Values represent the mean ± standard deviation (SD) of these experiments. The statistical significance was calculated with a Student’s t-test. P<0.05 was considered to indicate a statistically significant result.
Results
8-Cl-cAMP induced MM cell growth inhibition and apoptosis in a concentration- and time-related manner
To investigate the cytotoxic effect of 8-Cl-cAMP on RPMI-8226 and U266 cells, the cell viability following treatment with 8-Cl-cAMP at various concentrations (1, 3, 10, 30 μM) was determined by trypan blue exclusion assay. As shown in Fig. 1B, 8-Cl-cAMP reduced the cell viability of the RPMI-8226 and U266 cells in a concentration-dependent manner. Higher concentrations of 8-Cl-cAMP yielded more effective inhibition. The RPMI-8226 cells were more sensitive than the U266 cells to 8-Cl-cAMP. When treated with 30 μM 8-Cl-cAMP for 72 h, almost all RPMI-8226 cells died (Fig. 1A). The results of Annexin V-FITC and PI staining revealed that 8-Cl-cAMP induced apoptotic and necrotic cell death. The percentage of cell death was 19.27% in the RPMI-8226 cells treated with 30 μM 8-Cl-cAMP for 24 h (Fig. 2A). Since the reduction of mitochondrial transmembrane potential is often associated with apoptosis, we further investigated the effect of 8-Cl-cAMP on mitochondria. The treatment of RPMI-8226 cells with 8-Cl-cAMP resulted in an increase in the percentage of cells with a reduced mitochondrial transmembrane potential and increased apoptosis in a time-dependent manner (Fig. 2B). These features suggest that the 8-Cl-cAMP-induced MM cell death was due to apoptosis.
Figure 1.
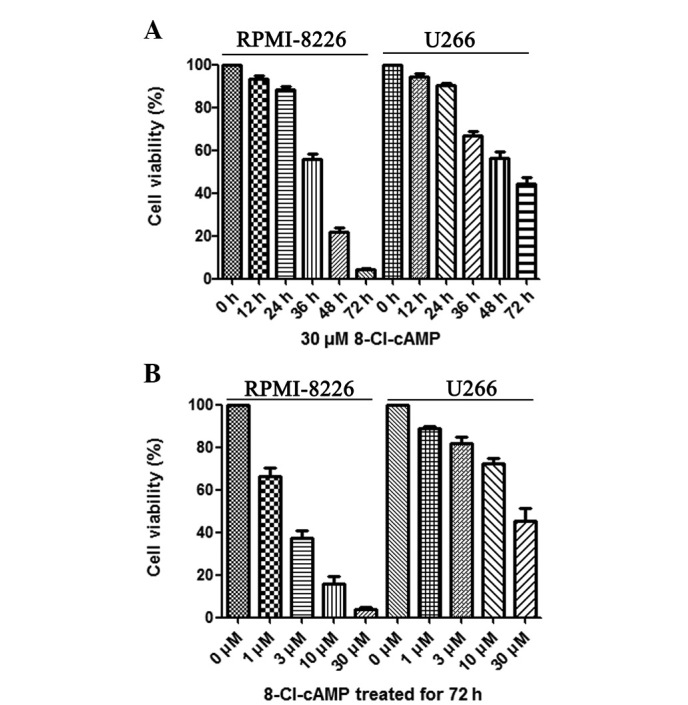
Multiple myeloma (MM) cells were treated with 8-chloroadenosine 3′,5′-monophosphate (8-Cl-cAMP) for (A) various times and (B) at different concentrations. Cell growth was evaluated by trypan blue exclusion assay. Data are expressed as a percentage of the untreated control. Values are expressed as the mean ± SEM (n=3).
Figure 2.
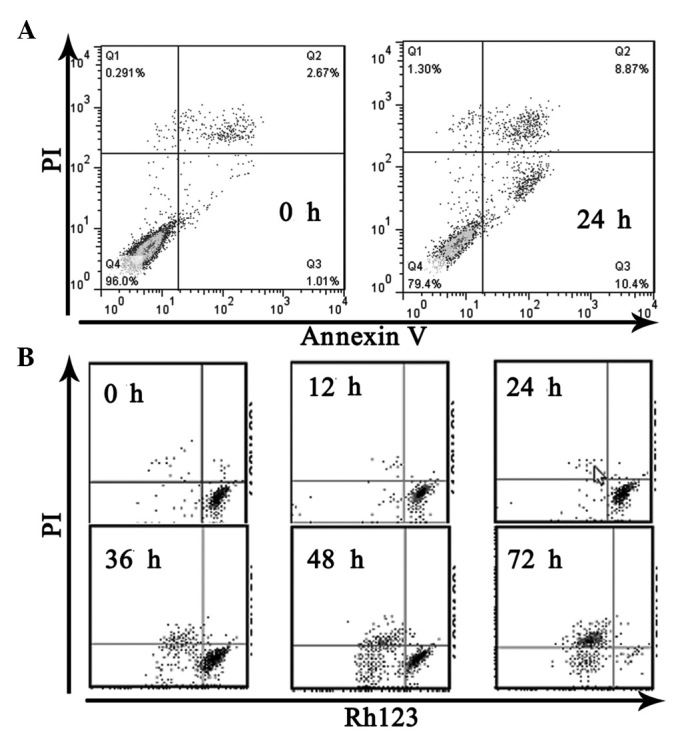
Effects of 8-chloroadenosine 3′,5′-monophosphate (8-Cl-cAMP) on (A) apoptotic cell death and (B) loss of mitochondrial membrane potential in multiple myeloma (MM) cells. PI, propidium iodide.
8-Cl-cAMP induces cell cycle arrest and apoptosis
To elucidate whether the apoptosis induced by 8-Cl-cAMP was due to cell cycle arrest, we evaluated the cell cycle distribution by flow cytometry. Flow cytometric analysis of the cellular DNA contents revealed that the sub-G1 phase ratio rose from 6.5 to 63.6% in the RPMI-8226 cells treated with 30 μM 8-Cl-cAMP for 48 h (Fig. 3A). The expression of the cyclin-dependent kinase (Cdk) inhibitor p27, which is involved in the arrest of the cell cycle in the G1 phase, was also evaluated. p27 levels, not detectable under basal conditions, were found to be increased following exposure to 10 μM 8-Cl-cAMP for 48 h (Fig. 3B). We also monitored the levels of other cell cycle-associated proteins. The levels of Cdk2 and cyclin E almost remained constant, but the expression levels of c-myc protein decreased in the RPMI-8226 cells exposed to 10 μmol/l 8-Cl-cAMP for 48 h (Fig. 3B). These results suggest that the 8-Cl-cAMP-induced cell cycle arrest was mediated by the expression of cell cycle-associated proteins. We also found that poly(ADP-ribose) polymerase (PARP) was cleaved following treatment with 8-Cl-cAMP (Fig. 3B).
Figure 3.
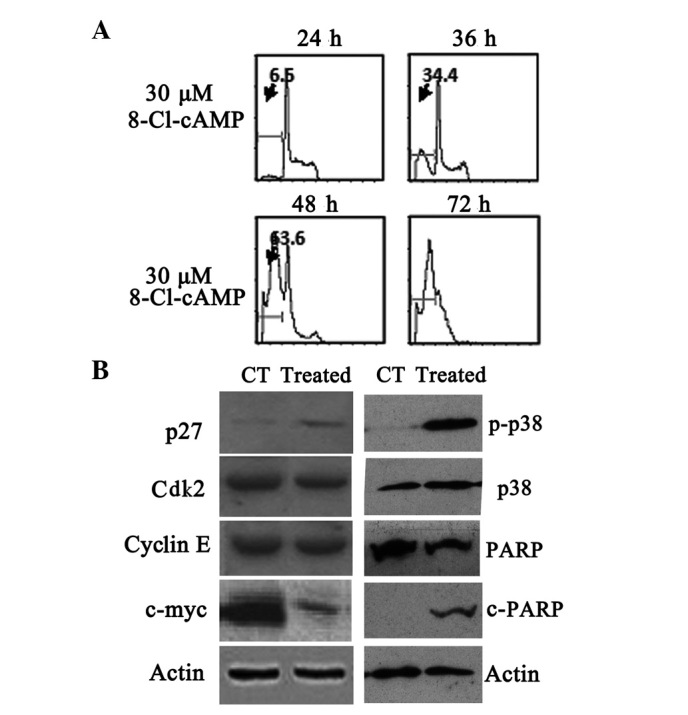
Effects of 8-chloroadenosine 3′,5′-monophosphate (8-Cl-cAMP) on (A) cell cycle distribution and (B) expression of cell cycle associated proteins, p38 mitogen-activated protein kinase (MAPK) and poly(ADP-ribose) polymerase (PARP). CT, control; Cdk2, cyclin-dependent kinase 2.
8-Cl-cAMP induces apoptosis in MM cells through a p27-dependent pathway
Cell cycle associated proteins play a critical role in mediating the cytotoxicity induced by 8-Cl-cAMP, but their targets are unknown. To determine whether higher p27 levels were a critical factor contributing to the 8-Cl-cAMP-induced MM cell apoptosis, we knocked down p27 expression by RNA interference. Immunoblotting revealed that the transient introduction of p27 siRNA into the RPMI-8226 cells markedly reduced p27 expression levels compared with those in their control counterparts (Fig. 4A). The viability of the RPMI-8226 cells in the presence of 10 μM 8-Cl-cAMP was 14.7%, whereas that of the p27-knocked-down RPMI-8226 cells was 74.1% (Fig. 4B). Similarly, flow cytometric analysis revealed that the level of apoptosis of sub-G1 cells increased in the RPMI-8226 cells treated with 10 μM 8-Cl-cAMP, whereas the apoptosis of sub-G1 cells was almost undetectable in the p27-knocked--down RPMI-8226 cells (Fig. 4C). Thus, on the basis of the above results, p27 is functionally linked to the sensitivity of the RPMI-8226 cells to 8-Cl-cAMP-induced apoptosis.
Figure 4.
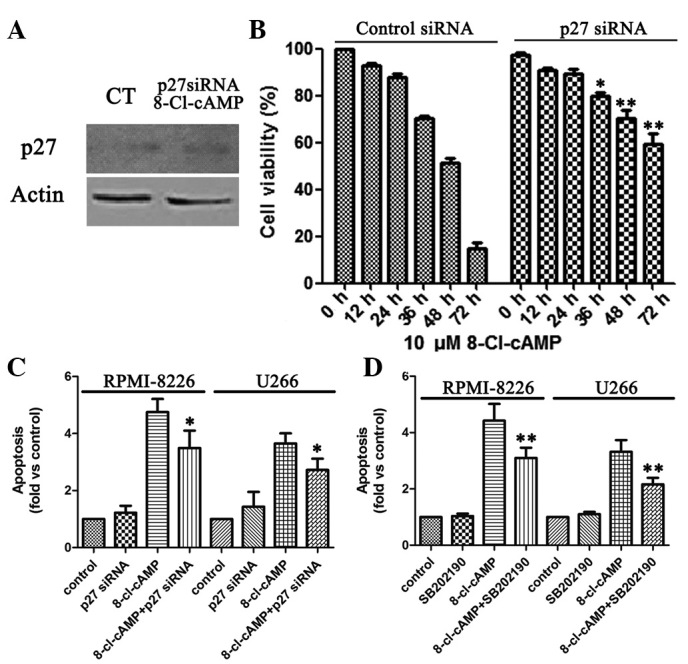
Role of p27 and p38 mitogen-activated protein kinase (MAPK) in 8-chloroadenosine 3′,5′-monophosphate (8-Cl-cAMP)-mediated effects. (A) Western blot analysis of p27 expression in cells treated with 80 nM p27 siRNA. Effects of 8-Cl-cAMP on (B) cell growth and (C) apoptotic cell distribution in multiple myeloma (MM) cells treated with 80 nM p27 siRNA. (D) The pro-apoptotic effect of 8-Cl-cAMP was largely prevented by a p38 MAPK inhibitor. CT, control.
Effect of 8-Cl-cAMP on p38 mitogen-activated protein kinase (MAPK) expression
8-Cl-cAMP has been shown to induce p38 MAPK phosphorylation in HL60 and HeLa cells (15,21). Therefore, we evaluated the effect of 8-Cl-cAMP on p38 MAPK phosphorylation in the MM cells. We identified that the treatment of RPMI-8226 and U266 cells with 8-Cl-cAMP was associated with a progressive increase in p38 MAPK phosphorylation that started after 48 h of incubation (Fig. 3B). Finally, we evaluated whether the pro-apoptotic effect of 8-Cl-cAMP was dependent upon p38 MAPK activation. To this aim, we treated the two cell lines with 8-Cl-cAMP for 48–72 h in the presence or absence of a selective p38 MAPK inhibitor (SB 202190) and analyzed the cell cycle distribution by flow cytometry as described previously. Notably, the inhibition of p38 MAPK largely prevented the pro-apoptotic effect of 8-Cl-cAMP (Fig. 4D). These data strongly suggest that the pro-apoptotic effect of 8-Cl-cAMP on RPMI-8226 and U266 cells is mediated by p38 MAPK.
Discussion
Following the identification of cAMP in 1958 by Rall and Sutherland, research focused for more than a decade on elucidating the role that this secondary messenger played in regulating metabolic pathways, as well as identifying the enzymes responsible for cAMP synthesis and catabolism (22–24). By the 1970s, however, cAMP was implicated as a regulator of cell growth (25–27), and several investigators reported that the elevation of cAMP levels induced proliferation arrest or cell death in susceptible normal or malignant lymphoid populations (28–33). The cAMP signaling pathway has emerged as a key regulator of hematopoietic cell proliferation, differentiation and apoptosis. The 8-chloro derivative of cAMP is a very potent cAMP analog that is under investigation as a potential chemotherapeutic agent. Phase I clinical studies in patients with solid tumors revealed its safety as well as evidence of clinical response (34).
In our study, we identified that 8-Cl-cAMP induced mitochondial transmembrane potential collapse and apoptosis simultaneously in RPMI-8226 cells. Western blot analysis revealed that 8-Cl-cAMP (10 μmol/l)-induced apoptosis was accompanied by upregulation of p27 and downregulation of c-myc in RPMI-8226 cells. p27, also known as Cdk inhibitor 1B, is an enzyme which belongs to the Cip/Kip family of Cdk inhibitor proteins (35). It binds to and prevents the activation of cyclin E-Cdk2 and cyclin D-Cdk4 complexes, and thus controls the cell cycle progression at G1 (36). It is often referred to as a cell cycle inhibitor protein since its major function is to stop or slow down the cell division cycle (35–38). In the current study, we found that 8-Cl-cAMP induced the upregulation of the Cdk inhibitor p27. Therefore, we assessed the significance of p27. Fig. 4 shows that p27 knockdown inhibited the 8-Cl-cAMP-induced decreases in cell viability, cell cycle arrest and cell death. These results indicate that the 8-Cl-cAMP-induced apoptosis in MM cells is p27-dependent, and p27 may be the target of 8-Cl-cAMP.
The proto-oncogene c-myc is involved in the control of cell proliferation, apoptosis and differentiation, and its aberrant expression is frequently observed in human cancer (39). It has been previously reported that small-molecule inhibitors of c-myc suppress proliferation and induce apoptosis of promyelocytic leukemia cells via cell cycle arrest (39,40). Although our data show that 8-Cl-cAMP induced the downregulation of c-myc, the mechanism for this action is not well defined. We also hypothesize that c-myc is significant in the mediation of 8-Cl-cAMP-induced cytotoxicity.
In addition, we identified that the pro-apoptotic effect of 8-Cl-cAMP was accompanied by a progressive increase of p38 MAPK phosphorylation. The p38 MAPK is a key regulator of cell survival (41,42) and has been implicated in the pro-apoptotic effect of 8-Cl-cAMP in HL60 and HeLa cells. In conclusion, our data indicate that 8-Cl-cAMP has a potent antiproliferative effect on MM cell lines through multiple pathways.
Acknowledgments
This study was supported by the Scientific Research Program of the Health Bureau of Shanghai (Grant No. 2010101) and the Shanghai Committee of Science and Technology, China (Grant No. 12ZR1416800). The authors are grateful to all members of the Hematology Department of Shanghai Ninth People’s Hospital for their continuous support and encouragement.
References
- 1.Hallek M, Bergsagel PL, Anderson KC. Multiple myeloma: increasing evidence for a multistep transformation process. Blood. 1998;91:3–21. [PMC free article] [PubMed] [Google Scholar]
- 2.Feng X, Yan J, Wang Y, Zierath JR, Nordenskjöld M, Henter JI, et al. The proteasome inhibitor bortezomib disrupts tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) expression and natural killer (NK) cell killing of TRAIL receptor-positive multiple myeloma cells. Mol Immunol. 2010;47:2388–2396. doi: 10.1016/j.molimm.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 3.Perez LE, Parquet N, Meads M, Anasetti C, Dalton W. Bortezomib restores stroma-mediated APO2L/TRAIL apoptosis resistance in multiple myeloma. Eur J Haematol. 2010;84:212–222. doi: 10.1111/j.1600-0609.2009.01381.x. [DOI] [PubMed] [Google Scholar]
- 4.Iguchi T, Yachide-Noguchi T, Hashimoto Y, Nakazato S, Sagawa M, Ikeda Y, Kisaki M. Novel tubulin-polymerization inhibitor derived from thalidomide directly induces apoptosis in human multiple myeloma cells: possible anti-myeloma mechanism of thalidomide. Int J Mol Med. 2008;21:163–168. [PubMed] [Google Scholar]
- 5.Zonder JA, Crowley J, Hussein MA, Bolejack V, Moore DF, Sr, Whittenberger BF, et al. Lenalidomide and high-dose dexamethasone compared with dexamethasone as initial therapy for multiple myeloma: a randomized Southwest Oncology Group trial (S0232) Blood. 2010;116:5838–5841. doi: 10.1182/blood-2010-08-303487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Offidani M, Leoni P, Corvatta L, Polloni C, Gentili S, Liberati AM, et al. Outcome and toxicity in the modern era of new drugs for multiple myeloma: a reappraisal for comparison with future investigational trials. Clin Lymphoma Myeloma Leuk. 2010;10:353–360. doi: 10.3816/CLML.2010.n.068. [DOI] [PubMed] [Google Scholar]
- 7.Dingli D, Rajkumar SV. How best to use new therapies in multiple myeloma. Blood Rev. 2010;24:91–100. doi: 10.1016/j.blre.2010.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pettersson M, Jernberg-Wiklund H, Larsson LG, Sundström C, Givol I, Tsujimoto Y, Nilsson K. Expression of the bcl-2 gene in human multiple myeloma cell lines and normal plasma cells. Blood. 1992;79:495–502. [PubMed] [Google Scholar]
- 9.Peeters SD, Hovenga S, Rosati S, Vellenga E. Bcl-xl expression in multiple myeloma. Med Oncol. 2005;22:183–190. doi: 10.1385/MO:22:2:183. [DOI] [PubMed] [Google Scholar]
- 10.Caravita T, Siniscalchi A, Tendas A, Cupelli L, Ales M, Perrotti A, et al. Safety and efficacy of a combination therapy with Revlimid, Adriamycin and dexamethasone (RAD) in relapsed/refractory multiple myeloma (MM): a single-centre experience. Ann Hematol. 2011;90:115–116. doi: 10.1007/s00277-010-0967-4. [DOI] [PubMed] [Google Scholar]
- 11.Lucchi S, Calebiro D, de Filippis T, Grassi ES, Borghi MO, Persani L. 8-Chloro-cyclic AMP and protein kinase A I-selective cyclic AMP analogs inhibit cancer cell growth through different mechanisms. PLoS One. 2011;6:e20785. doi: 10.1371/journal.pone.0020785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dumaz N, Marais R. Integrating signals between cAMP and the RAS/RAF/MEK/ERK signalling pathways. Based on the anniversary prize of the Gesellschaft für Biochemie und Molekularbiologie Lecture delivered on 5 July 2003 at the Special FEBS Meeting in Brussels. FEBS J. 2005;272:3491–3504. doi: 10.1111/j.1742-4658.2005.04763.x. [DOI] [PubMed] [Google Scholar]
- 13.Cho-Chung YS. Role of cyclic AMP receptor proteins in growth, differentiation, and suppression of malignancy: new approaches to therapy. Cancer Res. 1990;50:7093–7100. [PubMed] [Google Scholar]
- 14.Propper DJ, Saunders MP, Salisbury AJ, Long L, O’Byrne KJ, Braybrooke JP, et al. Phase I study of the novel cyclic AMP (cAMP) analogue 8-chloro-cAMP in patients with cancer: toxicity, hormonal, and immunological effects. Clin Cancer Res. 1999;5:1682–1689. [PubMed] [Google Scholar]
- 15.Han JH, Ahn YH, Choi KY, Hong SH. Involvement of AMP-activated protein kinase and p38 mitogen-activated protein kinase in 8-Cl-cAMP-induced growth inhibition. J Cell Physiol. 2009;218:104–112. doi: 10.1002/jcp.21573. [DOI] [PubMed] [Google Scholar]
- 16.Bajic V, Stanimirovic Z, Stevanovic J, Spremo-Potparevic B, Zivkovic L, Milicevic Z. Cytogenetic effects of 8-Cl-cAMP on human and animal chromosomes. J BUON. 2009;14:71–77. [PubMed] [Google Scholar]
- 17.Vucić V, Nićiforović A, Adzić M, Radojcić MB, Ruzdijić S. The combination of gamma ionizing radiation and 8-Cl-cAMP induces synergistic cell growth inhibition and induction of apoptosis in human prostate cancer cells. Invest New Drugs. 2008;26:309–317. doi: 10.1007/s10637-007-9101-4. [DOI] [PubMed] [Google Scholar]
- 18.Pesic M, Drabek K, Esler C, Ruzdijic S, Pejanovic V, Pietrzkowski Z. Inhibition of cell growth and proliferation in human glioma cells and normal human astrocytes induced by 8-Cl-cAMP and tiazofurin. Nucleosides Nucleotides Nucleic Acids. 2000;19:963–975. doi: 10.1080/15257770008033036. [DOI] [PubMed] [Google Scholar]
- 19.Li Y, Qu X, Qu J, Zhang Y, Liu J, Teng Y, et al. Arsenic trioxide induces apoptosis and G2/M phase arrest by inducing Cbl to inhibit PI3K/Akt signaling and thereby regulate p53 activation. Cancer Lett. 2009;284:208–215. doi: 10.1016/j.canlet.2009.04.035. [DOI] [PubMed] [Google Scholar]
- 20.Vistica DT, Skehan P, Scudiero D, Monks A, Pittman A, Boyd MR. Tetrazolium-based assays for cellular viability: a critical examination of selected parameters affecting formazan production. Cancer Res. 1991;51:2515–2520. [PubMed] [Google Scholar]
- 21.Ahn YH, Jung JM, Hong SH. 8-Chloro-cyclic AMP-induced growth inhibition and apoptosis is mediated by p38 mitogen-activated protein kinase activation in HL60 cells. Cancer Res. 2005;65:4896–4901. doi: 10.1158/0008-5472.CAN-04-3122. [DOI] [PubMed] [Google Scholar]
- 22.Rall TW, Sutherland EW. Formation of a cyclic adenine ribonucleotide by tissue particles. J Biol Chem. 1958;232:1065–1076. [PubMed] [Google Scholar]
- 23.Butcher RW, Sutherland EW. Adenosine 3′,5′-phosphate in biological materials. I. Purification and properties of cyclic 3′,5′-nucleotide phosphodiesterase and use of this enzyme to characterize adenosine 3′,5′-phosphate in human urine. J Biol Chem. 1962;237:1244–1250. [PubMed] [Google Scholar]
- 24.Robison GA, Butcher RW, Oye I, Morgan HE, Sutherland EW. The effect of epinephrine on adenosine 3′,5′-phosphate levels in the isolated perfused rat heart. Mol Pharmacol. 1965;1:168–177. [PubMed] [Google Scholar]
- 25.Abell CW, Monahan TM. The role of adenosine 3′,5′-cyclic monophosphate in the regulation of mammalian cell division. J Cell Biol. 1973;59:549–558. doi: 10.1083/jcb.59.3.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pastan IH, Johnson GS, Anderson WB. Role of cyclic nucleotides in growth control. Annu Rev Biochem. 1975;44:491–522. doi: 10.1146/annurev.bi.44.070175.002423. [DOI] [PubMed] [Google Scholar]
- 27.Friedman DL. Role of cyclic nucleotides in cell growth and differentiation. Physiol Rev. 1976;56:652–708. doi: 10.1152/physrev.1976.56.4.652. [DOI] [PubMed] [Google Scholar]
- 28.Franks DJ, MacManus JP, Whitfield JF. The effect of prostaglandins on cyclic AMP production and cell proliferation in thymic lymphocytes. Biochem Biophys Res Commun. 1971;44:1177–1183. doi: 10.1016/s0006-291x(71)80210-3. [DOI] [PubMed] [Google Scholar]
- 29.MacManus JP, Whitfield JF, Youdale T. Stimulation by epinephrine of adenyl cyclase activity, cyclic AMP formation, DNA synthesis and cell proliferation in populations of rat thymic lymphocytes. J Cell Physiol. 1971;77:103–116. doi: 10.1002/jcp.1040770112. [DOI] [PubMed] [Google Scholar]
- 30.Ralph P, Hyman R, Epstein R, Nakoinz I, Cohn M. Independence of theta and TL surface antigens and killing by thymidine, cortisol, phytohemagglutinin, and cyclic AMP in a murine lymphoma. Biochem Biophys Res Commun. 1973;55:1085–1091. doi: 10.1016/s0006-291x(73)80006-3. [DOI] [PubMed] [Google Scholar]
- 31.Coffino P, Bourne HR, Tomkins GM. Mechanism of lymphoma cell death induced by cyclic AMP. Am J Pathol. 1975;81:199–204. [PMC free article] [PubMed] [Google Scholar]
- 32.Ponzetti K, King M, Gates A, Anwer MS, Webster CR. Cyclic AMP-guanine exchange factor activation inhibits JNK-dependent lipopolysaccharide-induced apoptosis in rat hepatocytes. Hepat Med. 2010;2010:1–11. doi: 10.2147/HMER.S7673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Moujalled D, Weston R, Anderton H, Ninnis R, Goel P, Coley A, et al. Cyclic-AMP-dependent protein kinase A regulates apoptosis by stabilizing the BH3-only protein Bim. EMBO Rep. 2011;12:77–83. doi: 10.1038/embor.2010.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tortora G, Ciardiello F, Pepe S, Tagliaferri P, Ruggiero A, Bianco C, et al. Phase I clinical study with 8-chloro-cAMP and evaluation of immunological effects in cancer patients. Clin Cancer Res. 1995;1:377–384. [PubMed] [Google Scholar]
- 35.Ogino S, Kawasaki T, Ogawa A, Kirkner GJ, Loda M, Fuchs CS. Cytoplasmic localization of p27 (cyclin-dependent kinase inhibitor 1B/KIP1) in colorectal cancer: inverse correlations with nuclear p27 loss, microsatellite instability, and CpG island methylator phenotype. Hum Pathol. 2007;38:585–592. doi: 10.1016/j.humpath.2006.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Igreja S, Chahal HS, Akker SA, Gueorguiev M, Popovic V, Damjanovic S, et al. Assessment of p27 (cyclin-dependent kinase inhibitor 1B) and aryl hydrocarbon receptor-interacting protein (AIP) genes in multiple endocrine neoplasia (MEN1) syndrome patients without any detectable MEN1 gene mutations. Clin Endocrinol (Oxford) 2009;70:259–264. doi: 10.1111/j.1365-2265.2008.03379.x. [DOI] [PubMed] [Google Scholar]
- 37.Wang B, Ni F, Li L, Wei Z, Zhu X, Wang J, et al. Analysis of cyclin-dependent kinase inhibitor 1B mutation in Han Chinese women with premature ovarian failure. Reprod Biomed Online. 2010;21:212–214. doi: 10.1016/j.rbmo.2010.04.025. [DOI] [PubMed] [Google Scholar]
- 38.Tong Q, Zhang W, Jin S, Li S, Chen Z. The relationship between p27(kip1) expression and the change of radiosensitivity of esophageal carcinoma cells. Scand J Gastroenterol. 2011;46:173–176. doi: 10.3109/00365521.2010.522721. [DOI] [PubMed] [Google Scholar]
- 39.Huang MJ, Cheng YC, Liu CR, Lin S, Liu HE. A small-molecule c-Myc inhibitor, 10058-F4, induces cell-cycle arrest, apoptosis, and myeloid differentiation of human acute myeloid leukemia. Exp Hematol. 2006;34:1480–1489. doi: 10.1016/j.exphem.2006.06.019. [DOI] [PubMed] [Google Scholar]
- 40.Jeong KC, Ahn KO, Yang CH. Small-molecule inhibitors of c-Myc transcriptional factor suppress proliferation and induce apoptosis of promyelocytic leukemia cell via cell cycle arrest. Mol Biosyst. 2010;6:1503–1509. doi: 10.1039/c002534h. [DOI] [PubMed] [Google Scholar]
- 41.Ono K, Han J. The p38 signal transduction pathway: activation and function. Cell Signal. 2000;12:1–13. doi: 10.1016/s0898-6568(99)00071-6. [DOI] [PubMed] [Google Scholar]
- 42.Wada T, Penninger JM. Mitogen-activated protein kinases in apoptosis regulation. Oncogene. 2004;23:2838–2849. doi: 10.1038/sj.onc.1207556. [DOI] [PubMed] [Google Scholar]


