Abstract
Human choriocarcinoma is one of the most aggressive malignant tumors characterized by early hematogenous spread to lung and brain tissues, and may be a cause of death in patients. Choriocarcinoma may occur following pregnancy and during implantation; however, trophoblastic invasion in human pregnancy is tightly regulated. The transforming growth factor-beta 1 (TGF-β1) has been suggested to play a role in controlling this process. In this study, we investigated the impact of TGF-β1 on invasion, as well as its sites of action in the TGF-β1/Smad pathway using a JEG-3 choriocarcinoma cell line. Following the treatment of cells with different doses of TGF-β1, cell invasion was observed. We also detected the expression of TGF-β receptor type I (TβR I) and TGF-β receptor type II (TβR II), Smad4, matrix metalloprotease (MMP)-9 and tissue inhibitor of metalloproteinase (TIMP)-1 in JEG-3 cells. Our data demonstrated that TGF-β1 promoted the invasive capability of JEG-3 cells depending on the downregulation of TβR I, TβR II, Smad4 and the upregulation of MMP-9 and TIMP-1. These observations suggest that TGF-β1 may play a critical role in the initiation of the trophoblastic invasion process.
Keywords: transforming growth factor-beta 1, transforming growth factor-beta 1 receptor type I, transforming growth factor-beta 1 receptor type II, Smad4, matrix metalloprotease-9, tissue inhibitors of metalloprotease-1, choriocarcinoma
Introduction
A successful human pregnancy requires embryonic trophocytes to invade into the womb and adhere to the endometrium; however, this kind of invasion is strictly regulated temporospatially (1). The deregulation of the invasion process may cause a series of pregnancy-related diseases, such as hydatidiform mole and invasive mole; however, excessive invasion of trophocytes is significantly associated with the formation of placental choriocarcinoma (2). In this process, one critical step of choriocarcinoma invasion and metastasis is the degradation of the extracellular matrix (ECM), in which the MMP-9/TIMP-1 ratio plays a significant role (3,4).
The main treatment for choriocarcinoma is 5-fluorouracil, which has a very good therapeutic efficacy; however, it may also cause toxic reactions, side effects and drug resistance (5). Since conventional treatments including surgery and chemotherapy often fail, it is necessary to explore the metastasis mechanisms of proliferation and invasiveness, and find a new target for drug therapy.
TGF-β belongs to a growth factor super-family which consists of a highly evolutionary conserved group of secreted cytokines (6). The TGF-β family consists of TGF-β, activins, bone morphogenetic proteins (BMPs), nodals, inhibins and anti-mullerian hormone (AMH). TGF-β1 is the prototypic family member and is secreted as an inactive latent precursor (7,8). It plays a significant role in the control of growth and development, including the regulation of cell proliferation and differentiation, the promotion of ECM formation and suppression of the immune response (9). TGF-β1 exerts its cellular effects through TβR I, TβR II and the Smad transcription factors, which have pivotal roles in intracellular signaling (7,10). Moreover, Smad4 is an essential factor in TGF-β signaling and is a frequently mutated tumor suppressor gene found in human tumors (11–13). Compared with other tumors, fewer studies have explored the role of the TGF-β/Smad signaling pathway in the initiation and development of placental choriocarcinoma. Previous studies have revealed that TGF-β1 inhibits normal cell proliferation, but enhances the growth of tumor cells (14). During the early stages of tumorigenesis, TGF-β1 may be an inhibitor of tumor cells through its suppressive functions; however, following the development of malignance, it may promote tumor cell proliferation (15). We have conducted a series of studies on the choriocarcinoma JEG-3 cell line (16–18), a number of which are in agreement with these findings (15); however, the more exact mechanisms of development of placental choriocarcinoma remain a controversial issue.
In order to explore the potential mechanism of placental choriocarcinoma invasion, we examined the alterations in the invasive capacity of the JEG-3 cell line when exposed to exogenous TGF-β1, and the protein expression levels of MMP-9 and TIMP-1. The mRNA levels for TβR I, TβR II and Smad4 were also detected to investigate the impacts of the TGF-β1/Smad signaling pathway on choriocarcinoma cell proliferation and invasion. These results may provide theoretical and laboratory evidence to improve clinical early diagnosis for placental choriocarcinoma. They may also give rise to novel ideas regarding a target for its early treatment.
Materials and methods
Materials
The human placental choriocarcinoma JEG-3 cell line was obtained from the State Key Laboratory of Reproductive Biology, Institute of Zoology, Chinese Academy of Sciences. The study was approved by the ethics committee of the Natural Science Foundation of Hebei Province and the Education Department of Hebei province, Hebei, China.
JEG-3 cells culture
JEG-3 cells were cultured in an incubator with 5% CO2 at 37°C in Dulbecco’s modified Eagle’s medium (DMEM; Gibco-BRL, Carlsbad, CA, USA) with 10% fetal bovine serum (FBS, Hangzhou Sijiqing Biological Engineering Materials Co., Ltd, Hangzhou, China), 200 mM glutamine, 100 mM sodium pyruvate, 100 μg/ml streptomycin and 100 U/ml penicillin. Cells were subcultured with 0.25% trypsin and 0.02% EDTA when cell growth reached 70–80%, and the density of subcultured cells was at a ratio between 1:2 and 1:4.
Transwell invasion assay
The invasion assay for JEG-3 cells was performed on a 24-well Transwell chamber containing an 8-μm pore size polycarbonate membrane filter with a diameter of 6.5 mm (Corning Inc., Acton, MA, USA). The Transwell chamber was coated with a thin layer of 60 μl growth factor-reduced diluted Matrigel (BD Biosciences, Franklin Lakes, NJ, USA) (19) and incubated at 37°C for 4 h. A total of 500 μl 20% DMEM was placed on the bottom of the Transwell chamber. After a 24 h culture in a serum-free medium, JEG-3 cells were mixed with DMEM substrate containing 1% BSA and adjusted to a concentration of 3x104 cells/ml. Different concentrations of TGF-β1 [0 μg/l (control group), 5 μg/l, 50 μg/l, 100 μg/l and 200 μg/l] were added, respectively, then the cells were plated in the upper chambers for re-culturing at 37°C for 24 h. On removal of the Transwell chambers, cotton swabs were utilized to rub off the upper surface cells and then the chambers were allowed to dry naturally. The polycarbonate membrane was fixed by 4% paraformaldehyde for 10 min, stained with hematoxylin for 5–15 min and finally set on the slide (20). Five fields were randomly selected and the number of cells appearing on the undersurface of the polycarbonate membranes was counted scored under a microscope at ×200 magnification. Cell invasion was performed on five independent occasions.
Reverse transcription (RT)-PCR
JEG-3 cells in the exponential phase were incubated in 6-well plates at an initial concentration of 3×104 cells/ml for 48 h and then cultured with serum-free DMEM for 24 h to obtain cell synchronization. The cells were subsequently treated for 48 h with a series of concentrations of TGF-β1 [0 μg/l (control group), 5 μg/l, 10 μg/l, 25 μg/l, 50 μg/l, 100 μg/l, and 200 μg/l]. Total RNA was isolated with TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The final RNA concentration, integrity and purity were validated, and meet the experiment requirements. RT-PCR was performed following the manufacturer’s recommendations (Takara Biotechnology, Dalian, Liaoning, China). β-actin was used as the internal control (21). The primers used are listed in Table I. Amplification conditions were: 94°C for 2 min, followed by 28 or 30 cycles of 94°C for 30 sec, annealing temperature for 30 sec and 72°C for 1 min. The PCR products were confirmed by electrophoresis in 2% agarose gel at 120V for 30 min. Gel images were taken under UV light using the UVP image analysis system (Shanghai Jiapeng Technology, Shanghai, China) and analyzed with Quantity One software (Bio-Rad, Hercules, CA, USA). Each sample was repeated a minimum of five times. The relative quantity of the specific gene was obtained and normalized to the values of β-actin.
Table I.
PCR primers used in reaction.
| Target | Primer | Sequences (5′-3′) | Product size (bp) | Annealing temperature (°C) |
|---|---|---|---|---|
| TβRI | Forward | GGC CAA ATA TCC CAA ACA GAT | 509 | 60 |
| Reverse | AAT CCA ACT CCT TTG CCC TTA | |||
| TβRII | Forward | GAC TTG ACC TGT TGC CTG TGT | 310 | 59 |
| Reverse | TCA GCA CAC TGT CTT TCA TGC | |||
| Smad4 | Forward | CAG CAT CCA CCA AGT AAT CGT | 587 | 60 |
| Reverse | CTC TCA ATG GCT TCT GTC CTG | |||
| β-actin | Forward | AGC GGG AAA TCG TGC GTG AC | 453 | 58 |
| Reverse | ACA TCT GCT GGA AGG TGG AC |
Western blot assay
The cells were incubated in 6-well plates with an initial concentration of 3×104 cells/ml for 48 h and then were allowed to grow in serum-free DMEM for 24 h. Following cell synchronization, different concentrations of TGF-β1 [0 μg/l (control group), 100 μg/l and 200 μg/l] were added into each well and incubation continued for 48 h. The cells were washed with PBS twice and then collected, degraded and centrifuged at 12,000 rpm for 20 min at 4°C. The supernatants were collected as whole cell protein extracts. The bicinchoninic acid assay was used to determine protein quantity. Samples were mixed with 5X sodium dodecyl sulfate (SDS) loading buffer and denatured for 10 min at 95°C. Equal amounts of extracted proteins were separated on 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to polyvinylidene difluoride (PVDF) membranes. The membranes were blocked in 5% non-fat dried milk in tris-buffered saline Tween-20 (TBST) buffer (10 mmol/l Tris-HCL, 100 mmol/l NaCl and 0.1% Tween-20) and then incubated with the primary antibody at room temperature. After washing with the TBST buffer, the membranes were incubated with the secondary antibody, and then washed with TBST again three times, 5 minutes per wash. Immunoblots were then visualized with Super ECL Plus luminescence fluid (Applygen Technologies Inc., Beijing, China). β-actin was the internal control. The densities of the bands were scanned and calculated with Quantity One software (Bio-Rad). The relative amounts of the specific protein expression (MMP-9 and TIMP-1) were obtained by normalization to the densities of β-actin.
Statistical analysis
All data were expressed as the means ± standard deviation (SD). One-way analysis of variance (ANOVA) was used to compare differences among groups. The SNK-q test was performed to compare the differences between each two groups. P<0.05 was considered to indicate a statistically significant difference. All statistical analyses were performed using SPSS 13.0 software (SPSS Inc., Chicago, IL, USA).
Results
TGF-β1 promotes JEG-3 cell invasion
When JEG-3 cells were treated with TGF-β1 at a series of concentrations (0, 5, 50, 100 and 200 μg/l), the numbers of transmigrated cells significantly increased with an increasing TGF-β1 concentration (P<0.05; Fig. 1). This suggests a dose-dependent effect. The numbers of transmigrated cells displayed significant differences between the experimental groups and the control group, as well as between any other two groups (P<0.05; Fig. 1).
Figure 1.
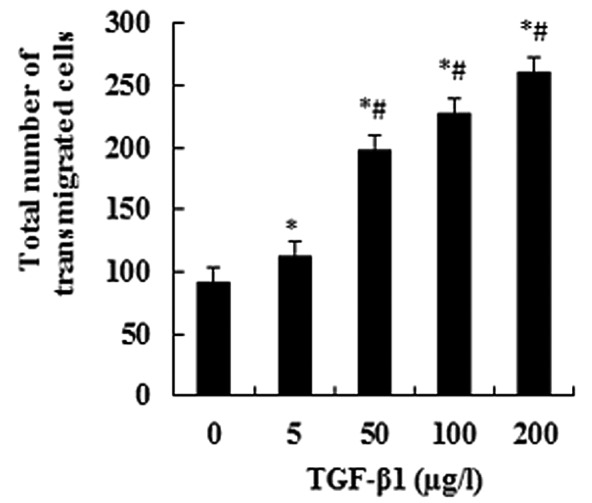
The JEG-3 cells were treated with TGF-β1 for 48 h and their invasive capacity was subsequently detected using the Transwell invasion assay. The total number of transmigrated cells was obtained by counting the number of dyed cells with a light microscope. Exogenous TGF-β1 specifically promoted the invasive capacity of JEG-3 cells. The data are presented as the means ± SD (n=5; *P<0.05 when compared with TGF-β1 0 μg/l; #P<0.05 when compared with any other group except 0 μg/l).
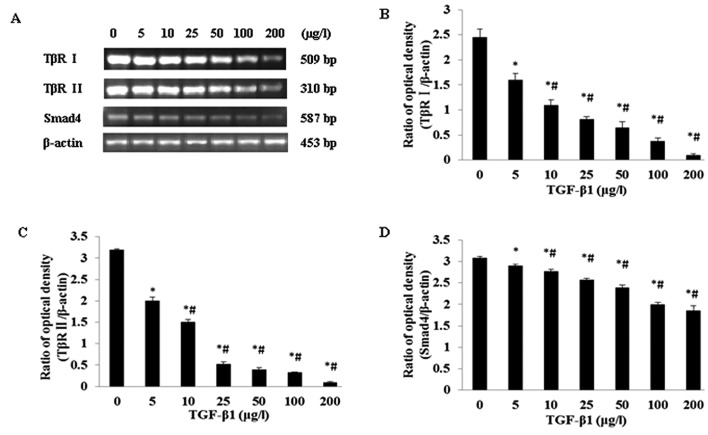
Impact of TGF-β1 on TβR I, TβR II and Smad4 mRNA expression in the JEG-3 cell line
JEG-3 cells were separately treated by a series of TGF-β1 concentrations (0, 5, 10, 25, 50, 100, and 200 μg/l). It was revealed that with increasing TGF-β1 concentrations, the mRNA expression levels for TβR I, TβR II and Smad4 gradually reduced, suggesting a certain dose-dependence (Fig. 2A). The mRNA expression levels for TβR I, TβR II and Smad4 showed a statistically significantly difference (P<0.05) when compared with all other groups (Fig. 2B, C and D).
Figure 2.
Regulatory effects of different doses of TGF-β1 on TβR I, TβR II and Smad4 mRNA expression levels in JEG-3 cells. (A) RT-PCR analysis of TβR I, TβR II and Smad4 mRNA levels in JEG-3 cells cultured in different concentrations of TGF-β1; (B,C,D) Statistical analyses. *P<0.05 when compared with TGF-β1 0 μg/l; #P<0.05 when compared with any other group except 0 μg/l.
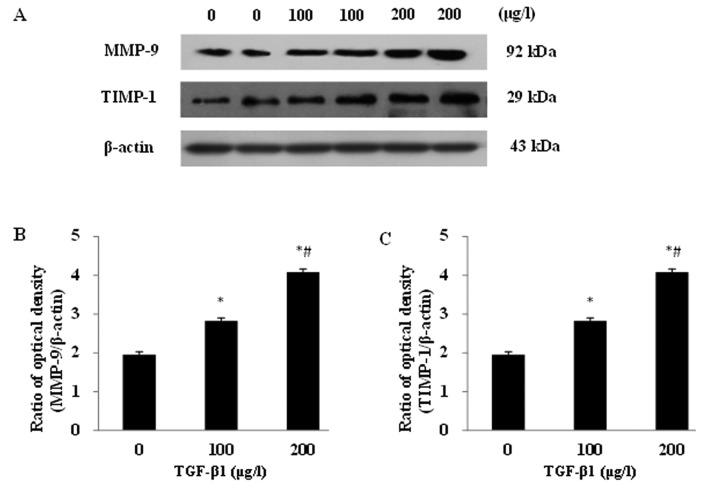
Impacts of TGF-β1 on MMP-9 and TIMP-1 protein expression in the JEG-3 cell line
After incubation with the different concentrations of TGF-β1 (0, 100 and 200 μg/l) for 48 h, significantly increased protein expression levels of MMP-9 and TIMP-1 were detected in JEG-3 cells (Fig. 3A). A dose-dependent response was observed for both proteins. The MMP-9 and TIMP-1 protein expressions displayed significant differences between the experimental groups and the control group, as well as between any other two groups (P<0.05; Fig. 3B and C).
Figure 3.
TGF-β1 upregulated MMP-9 and TIMP-1 protein expressions in JEG-3 with increasing of TGF-β1 concentrations. (A) Western blot assay of MMP-9 and TIMP-1 protein expressions in different concentrations of TGF-β1 in the JEG-3 cells. (B,C) MMP-9 and TIMP-1 protein expression. *P<0.05 when compared with TGF-β1 0 μg/l; #P<0.05 when compared with TGF-β1 100 μg/l.
Discussion
Human choriocarcinoma is the most aggressive malignant tumor characterized by early hematogenous spread to lung and brain tissues and can cause patients’ death. Choriocarcinoma may occur following pregnancy and during implantation; however, trophoblastic invasion in human pregnancy is tightly regulated. TGF-β1 has been found to correlate closely with trophoblastic invasion (22). The expression of TGF-β1 can be detected in almost all types of tumors and exists at an upregulated level in cancer cells, allowing it to inhibit normal cell proliferation yet enhance the growth of tumor cells (23). During the early stage of tumorigenesis, TGF-β1 may be an inhibitor of tumor cells through the suppression of many cell functions; however, following the development of malignance, it promotes tumor cell proliferation (24). TGF-β1 exists within cells as a non-active complex that must be activated in order to exert a biological effect on its major downstream signaling molecule, Smad (25). The signaling pathway begins with TGF-β1 ligands, which bind to a type II receptor, and stimulate the Smad protein to act as a transcription factor and participate in the regulation of target gene expression. If mutations in multiple genes occur in the TGF-β1 signaling pathway, it can lead to TGF-β1 tolerance and loss of function in senescence and apoptosis pathways. This may result in the induction of malignant transformation by TGF-β1. Our previous studies (16,17) revealed that the choriocarcinoma JEG-3 cell line responds to the stimulation of TGF-β1 and may promote JEG-3 cell proliferation; however, the effect of TGF-β1 on invasion and the more exact mechanisms remain a controversial issue. In this study, we found a significantly higher function of TGF-β1 in promoting invasion in the JEG-3 cell line. These findings suggest that TGF-β1 may participate in the development of choriocarcinoma. Our observations concur with the role of TGF-β1 in other malignancies (26,27).
We also found that mRNA expression was detectable for TβR I, TβR II and Smad4 in the choriocarcinoma JEG-3 cell line. With increasing concentrations of exogenous TGF-β1, the expressions of TβR I, TβR II and Smad4 were gradually reduced. This suggests that the TGF-β1/Smad4 transduction system may participate in the initiation process and promote the proliferation and invasion of choriocarcinoma. This function may be correlated with the decreased expression of TβR I, TβR II and common-mediator Smads (Smad4), which limit the downward propagation of the TGF-β1/Smad4 signal and subsequently cause tumor growth inhibition to reduce or even become deficient. Certain animal experiments and clinical studies (28–31) have also revealed that, within the tumor, several types of mutations in the TβR II and Smad4 genes have been identified. The loss of TβR II function may cause tumor cells to deregulate the expression of the receptor protein, obstruct the transmission of the TGF-β1 growth inhibiting signal (32) and subsequently display malignant proliferation and invasion. Moreover, TGF-β1 could also play distinct roles in the Smad4-dependent and -independent signaling pathways (33).
Prior to invasion or metastasis, tumor cells must destroy the adjacent tissues and cells, degrade the basement membrane and then infiltrate the periphery mesenchyme. TGF-β1 may strengthen the capabilities of invasiveness and metastasis through inducing the aberrant tumor microenvironment, participating in the degeneration and rebuilding of the microenvironment and enhancing the production of proteinase, such as MMPs and urokinase (34,35). MMP-9 has the largest molecular mass of all the MMP family members and TIMP-1 is a specific inhibitor of MMP-9. They are involved in the degradation of the extracellular matrix (ECM) and play a role in the invasion and metastasis of tumor cells (3,4). Previous studies (36–38) have suggested that disruption to the ECM during tumor progression is due to an imbalance in the MMP/TIMP ratio. Our study revealed that a significant upregulation of MMP-9 and TIMP-1 protein expression in JEG-3 cells was observed following treatment with TGF-β1 at concentrations of 100 and 200 μg/l. These results were consistent with those of Yudate et al (39). However, the ratio of MMP-9/TIMP-1 was still higher than 1, which implied that the balance of TIMP-1 inhibiting MMP-9 was disturbed, and resulted in the high invasiveness of choriocarcinoma. By integrating these results with our previous findings (17), we believe that the downregulation in the expression of TβR I, TβR II and common mediator Smads may limit the downward propagation of the TGF-β1/Smad4 signal, and promote the tumor’s invasive capability. Simultaneously, the increasing secretion of invasion-related factor MMP-9 may enhance the degradation of the ECM and also promote the invasiveness of tumor cells.
In summary, although some previous studies have demonstrated the critical role of TGF-β1 in the invasiveness and metastasis of placental choriocarcinoma, the detailed biological mechanisms are still unclear. Our data indicate that the role of TGF-β1 in promoting invasion and metastasis may be mediated through TβR I, TβR II, Smads-dependent pathways and the regulation of MMP-9 and TIMP-1. Following the binding of TGF-β1 to the cell membrane type II receptor, it activates the type I receptor and stimulates the downstream molecules, Smad2/Smad3, to bind to Smad4 forming a complex, which subsequently participates in regulating target gene transcription mediated by TGF-β1 in the nucleus. Based on the significance of TGF-β1 in tumorigenesis and metastasis, further clarification of the mechanisms of TGF-β1 in promoting tumor metastasis will have crucial theoretical and clinical significance.
References
- 1.Shih leM, Kuo KT. Power of the eternal youth: Nanog expression in the gestational choriocarcinoma. Am J Pathol. 2008;173:911–914. doi: 10.2353/ajpath.2008.080624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Genest DR, Berkowitz RS, Fisher RA. Gestational trophoblastic disease. In: Tavassoli FA, Devilee P, editors. World Health Organization Classification of Tumours. Pathology and Genetics Tumours of the Breast and Female Genital Organs. IARC Press; Lyon: 2003. pp. 250–254. [Google Scholar]
- 3.Decock J, Thirkettle S, Wagstaff L, Edwards DR. Matrix metalloproteinases: protective roles in cancer. J Cell Mol Med. 2011;15:1254–1265. doi: 10.1111/j.1582-4934.2011.01302.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bourboulia D, Stetler-Stevenson WG. Matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs): Positive and negative regulators in tumor cell adhesion. Semin Cancer Biol. 2010;20:161–168. doi: 10.1016/j.semcancer.2010.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van Trommel NE, Lybol C, Thomas CM, Sweep FC, Massuger LF. Diagnosis and treatment of gestational trophoblastic disease. European Obstetrics & Gynaecology. 2011;6:28–32. [Google Scholar]
- 6.Pardali E, Ten Dijke P. TGFβ signaling and cardiovascular diseases. Int J Biol Sci. 2012;8:195–213. doi: 10.7150/ijbs.3805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heldin CH, Miyazono K, ten Dijke P. TGF-β signalling from cell membrance to nucleus through SMAD proteins. Nature. 1997;390:465–471. doi: 10.1038/37284. [DOI] [PubMed] [Google Scholar]
- 8.Heldin CH, Moustakes A. Role of Smads in TGFβ signaling. Cell Tissue Res. 2012;347:21–36. doi: 10.1007/s00441-011-1190-x. [DOI] [PubMed] [Google Scholar]
- 9.Hyytiäinen M, Pentinen C, Keski-Oja J. Latent TGF-beta binding proteins: extracellular matrix association and roles in TGF-beta activation. Crit Rev Clin Lab Sci. 2004;41:233–264. doi: 10.1080/10408360490460933. [DOI] [PubMed] [Google Scholar]
- 10.ten Dijke P, Arthur HM. Extracellular control of TGFβ signalling in vascular development and disease. Nat Rev Mol Cell Biol. 2007;8:857–869. doi: 10.1038/nrm2262. [DOI] [PubMed] [Google Scholar]
- 11.Vogelstein B, Kinzler KW. Cancer genes and the pathways they control. Nat Med. 2004;10:789–799. doi: 10.1038/nm1087. [DOI] [PubMed] [Google Scholar]
- 12.Bardeesy N, Depinho RA. Pancreatic cancer biology and genetics. Nature Rev Cancer. 2002;2:897–909. doi: 10.1038/nrc949. [DOI] [PubMed] [Google Scholar]
- 13.Lee SH, Jung YS, Chung JY, et al. Novel tumor suppressive function of Smad4 in serum starvation-induced cell death through PAK1-PUMA pathway. Cell Death Dis. 2011;2:e235. doi: 10.1038/cddis.2011.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Grady WM. Transforming growth factor-beta, Smads, and cancer. Clin Cancer Res. 2005;11:3151–3154. doi: 10.1158/1078-0432.CCR-05-0414. [DOI] [PubMed] [Google Scholar]
- 15.Moustakas A, Souchelnytskyi S, Heldin CH. Smad regulation in TGF-beta signal transduction. J Cell Sci. 2001;114:4359–4369. doi: 10.1242/jcs.114.24.4359. [DOI] [PubMed] [Google Scholar]
- 16.Li X, Li Y, Xu Q, Zhao S, Zhao L. Effect of transforming growth factor β1 on proliferation of JEG-3 choriocarcinoma cells and Smad3, 7 mRNA expression. Maternal & Child Health Care of China. 2010;25:1685–1688. [Google Scholar]
- 17.Li X, Li Y, Xu Q, Zhao L, Zhao S, Zhao L. Effect on TGF-β1 on MMP-9 mRNA and TIMP-1 mRNA in choriocarcinoma JEG-3 cell. Shandong Medical Journal. 2009;49:18–20. [Google Scholar]
- 18.Zhang Z, Xu Q, Shi C, Li Y. Interleukin-12 inhibits cell invasion in choriocarcinoma. Int J Mol Med. 2012;30:57–62. doi: 10.3892/ijmm.2012.960. [DOI] [PubMed] [Google Scholar]
- 19.Beristain AG, Zhu H, Leung PC. Regulated expression of ADAMTS-12 in human trophoblastic cells: a role for ADAMTS-12 in epithelical cell invasion? PLoS One. 2011;6:e18473. doi: 10.1371/journal.pone.0018473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nicolson GL. Transfilter cell invasion assays. In: Celis JE, editor. Cell Biology: A Laboratory Handbook. Vol. 1. Academic Press; New York: 2006. pp. 359–362. [Google Scholar]
- 21.Suzuki T, Higgins PJ, Crawford DR. Control selection for RNA quantitation. Biotechniques. 2000;29:332–337. doi: 10.2144/00292rv02. [DOI] [PubMed] [Google Scholar]
- 22.Rama S, Suresh Y, Rao AJ. TGF beta1 induces multiple independent signals to regulate human trophoblastic differentiation: mechanistic insights. Mol Cell Endocrinol. 2003;206:123–136. doi: 10.1016/s0303-7207(03)00202-8. [DOI] [PubMed] [Google Scholar]
- 23.Prud’homme GJ. Pathobiology of transforming growth factor beta in cancer, fibrosis and immunologic disease, and therapeutic considerations. Lab Invest. 2007;87:1077–1091. doi: 10.1038/labinvest.3700669. [DOI] [PubMed] [Google Scholar]
- 24.Joshi A, Cao D. TGF-beta signaling, tumor microenvioronment and tumor progression: the butterfly effect. Front Biosci. 2010;15:180–194. doi: 10.2741/3614. [DOI] [PubMed] [Google Scholar]
- 25.Samanta D, Datta PK. Alterations in the Smad pathway in human cancers. Front Biosci. 2012;17:1281–1293. doi: 10.2741/3986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Davies M, Prime SS, Eveson JW, Price N, Ganapathy A, D’Mello A, Paterson IC. Transforming growth factor-β enhances invasion and metastasis in Ras-transfected human malignant epidermal keratinocytes. Int J Exp Pathol. 2012;93:148–156. doi: 10.1111/j.1365-2613.2011.00806.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shang D, Liu Y, Yang P, Chen Y, Tian Y. TGFBI-promoted adhesion, migration and invasion of human renal cell carcinoma depends on inactivation of von Hippel-Lindau tumor suppressor. Urology. 2012;79:966.e1–7. doi: 10.1016/j.urology.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 28.Zhang B, Halder SK, Kashikar ND, Cho YJ, Datta A, Gorden DL, Datta PK. Antimetastatic role of Smad4 signaling in colorectal cancer. Gastroenterology. 2010;138:969–980. doi: 10.1053/j.gastro.2009.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bornstein S, White R, Malkoski S, et al. Smad4 loss in mice causes spontaneous head and neck cancer with increased genomic instability and inflammation. J Clin Invest. 2009;119:3408–3419. doi: 10.1172/JCI38854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Romero D, Iqlesias M, Vary CP, Quintanilla M. Functional blockade of Smad4 leads to a decrease in beta-catenin levels and signaling activity in human pancreatic carcinoma cells. Carcinogenesis. 2008;29:1070–1076. doi: 10.1093/carcin/bgn054. [DOI] [PubMed] [Google Scholar]
- 31.Lu B, Zhou YN, Li Q, et al. Correlations of TGF-β RII, Smad4 and Smad7 expression to clinicopathologic characteristics and prognosis of gastric cancer. Chinese Journal of Cancer. 2009;2:538–542. [PubMed] [Google Scholar]
- 32.Horie K, Yamashita H, Mogi A, Takenoshita S, Miyazono K. Lack of transforming growth factor-beta type II receptor expression in human retinoblastoma cells. J Cell Physiol. 1998;175:305–313. doi: 10.1002/(SICI)1097-4652(199806)175:3<305::AID-JCP8>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 33.Wang X, Sun W, Zhang C, Ji G, Ge Y, Xu Y, Zhao Y. TGF-β1 inhibits the growth and metastasis of tongue squamous carcinoma cells through Smad4. Gene. 2011;485:160–166. doi: 10.1016/j.gene.2011.06.023. [DOI] [PubMed] [Google Scholar]
- 34.Binker MG, Binker-Cosen AA, Gaisano HY, de Cosen RH, Cosen-Binker LI. TGF-β1 increases invasiveness of SW1990 cells through Rac1/ROS/NF-κB/IL-6/MMP-2. Biochem Biophys Res Commun. 2011;405:140–145. doi: 10.1016/j.bbrc.2011.01.023. [DOI] [PubMed] [Google Scholar]
- 35.Bao J, Wu ZS, Qi Y, Wu Q, Yang F. Expression of TGF-beta1 and the mechanism of invasiveness and metastasis induced by TGF-beta1 in breast cancer. Zhonghua Zhong Liu Za Zhi. 2009;31:679–682. [PubMed] [Google Scholar]
- 36.Singh RD, Haridas N, Patel JB, Shah FD, Shukla SN, Shah PM, Patel PS. Matrixmetallo-proteinases and their inhibitors: correlation with invasion and metastasis in oral cancer. India J Clin Biochem. 2010;25:250–259. doi: 10.1007/s12291-010-0060-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kang H, Jang SW, Ko J. Human sLZIP induces migration and invasion of cervical cancer cells via expression of matrix metalloproteinase-9. J Biol Chem. 2011;286:42072–42081. doi: 10.1074/jbc.M111.272302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zarrabi K, Dufour A, Li J, et al. Inhibition of matrix metalloproteinase 14 (MMP-14)-mediated cancer cell migration. J Biol Chem. 2011;286:33167–33177. doi: 10.1074/jbc.M111.256644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yudate T, Isaka K, Kosugi Y, Koshiishi M, Shiraishi K, Hosaka M, Takayama M. Analysis of the mechanism of trophoblast infiltration. Nihon Sanka Fujinka Gakkai Zasshi. 1996;48:191–198. [PubMed] [Google Scholar]