Abstract
The objective of this study was to determine protein quality and hematological properties of infant diets formulated from local food materials. The food materials were obtained locally, fermented, and milled into flour. The flours were mixed as 70% popcorn and 30% African locust bean (FPA), 70% popcorn and 30% bambara groundnut (FPB), and 70% popcorn, 20% bambara groundnut, and 10% African locust bean (FPAB). Proximate analysis, protein quality, hematological properties, and anthropometric measurements of the animals fed with the formulations were investigated. The protein contents of the formulated diets were significantly higher than that of Cerelac (a commercial preparation) (15.75 ± 0.01 g/100 g) and ogi (traditional complementary food) (6.52 ± 0.31 g/100 g). The energy value of FPAB (464.94 ± 1.22 kcal) was higher than those of FPA (441.41 ± 3.05 kcal) and FPB (441.48 ± 3.05 kcal). The biological value (BV) of FPAB (60.20%) was the highest followed by FPB (44.24%) and FPA (41.15%); however, BV of the diets was higher than that of ogi (10.03%) but lower than that of Cerelac (70.43%). Net protein utilization (NPU) of the formulations was 41.16-60.20%, whereas true protein digestibility was 41.05-60.05%. Metabolizable energy (232.98 kcal) and digestible energy (83.69 kcal) of FPAB were the highest, whereas that of FPA had the lowest values. The protein digestibility values corrected for amino acid score of the diets (0.22-0.44) were lower than that of Cerelac (0.52), but higher than that of ogi (0.21). The growth patterns and hematological properties (packed cell volume, red blood cells, hemoglobin, mean corpuscular hemoglobin concentration, mean corpuscular hemoglobin, and mean corpuscular volume) of the formulated diets were higher than those of ogi, but lower than those of Cerelac. In conclusion, we established that the FPAB food sample was rated best in terms of protein quality over the other formulated diets. Therefore, a FPAB blend may be used as a substitute for ogi.
Keywords: Complementary foods, protein quality, hematological properties
Introduction
Adequate child nutrition, which is achieved by consuming a balanced healthy diet and good knowledge of child feeding practices, is a fundamental pillar of health, growth, and development in children [1,2]. Despite the importance of good nutrition, it is evident that many families in developing countries are unable to feed their children an appropriate diet [3,4]. Several studies support the view that poor nutrition during childhood can have detrimental effects on growth and cognitive development, decrease activity levels, and affect social functioning [5-7]. It has been estimated that under-nutrition is directly or indirectly responsible for an increase in morbidity and mortality among children < 5 years of age worldwide [8-11], due to poor complementary feeding practices and low nutrient-dense local complementary foods.
Traditional complementary foods in many developing countries, including Nigeria, are of low nutritive value [12,13], and are characterized by low protein and energy density and high bulk. Cereals form the primary base for most traditional complementary foods in Nigeria and have been implicated in the etiology of protein-energy malnutrition in children during the complementary feeding period [14,15]. The protein content of maize and guinea corn is poor, as they are low in lysine and tryptophan. These two amino acids are indispensable for growth of infants and young children [16]. In view of the nutrition problems associated with the traditional complementary foods in Nigeria, investigated the nutritional quality of complementary foods produced from popcorn, African locust bean, and Bambara groundnut seeds. These food materials were purposely selected because of their nutrient composition and accessibility to many low-income families.
Materials and Methods
Sources of food materials
Popcorn, bambara groundnut, and African locust bean were obtained from a local market in Akure Township Nigeria in March 2011. The control food samples (Cerelac, a commercial product and ogi, a traditional complementary food) were obtained from a reputable supermarket and vendor, respectively.
Food processing
Fermented popcorn flour
Fermentation was performed using microorganisms naturally present on the grain surface. The popcorn seeds were weighed (1 kg), sorted, soaked in hot water, and left for 4 days. The grains were washed with distilled water, wet milled with an attrition mill (locally fabricated grinding machine), sieved with muslin cloth, and allowed to ferment for 3 days. The fermented slurry was decanted, drained, oven dried in a hot air oven at 60℃ (Plus11 Sanyo Gallenkamp PLC, Loughborough, Leicestershire, UK) for 20 hours, re-milled using a Philips laboratory blender (HR2811 model), sieved using a 60 mm mesh sieve (British standard), packed in plastic containers sealed with aluminum foil and stored at room temperature prior to analyses.
Fermented Bambara groundnut flour
The Bambara groundnut seeds were weighed (1 kg), sorted to remove foreign substances, washed with distilled water, and cooked for 1 and half hours. The cooked sample was tightly wrapped in plantain leaves for 3 days to ferment. The fermented bambara groundnut seeds were oven dried at 60℃ (Plus11 Sanyo Gallenkamp PLC) for 20 hours, milled using a Philips laboratory blender (model HR2811), sieved using a 60 mm mesh sieve (British standard), packed in plastic container sealed with aluminum foil, and stored at room temperature prior to analyses.
Fermented African locust bean flour
African locust beans were weighed (1 kg), washed with distilled water, soaked in warm water for 5 days, dehulled manually, and cooked for 1 and half hours. The cooked sample was tightly wrapped in plantain leaves for 1 day to ferment. Fermentation was performed using the microorganisms naturally present on the seed surface. The fermented African locust beans were oven dried at 60℃ (Plus11 Sanyo Gallenkamp PLC) for 20 hours, milled using a Philips laboratory blender, sieved using a 60 mm mesh sieve (British standard), packed in a plastic container sealed with aluminum foil, and stored at room temperature prior to analyses.
Food formulations
The food samples were formulated with reference to infant protein requirements (18 g/day) using Nutrisurvey linear programming (2004) software. The flour samples were mixed as follows Fermented popcorn: African locust bean flour mixe (FPA) (70:30% w/w), fermented popcorn: Bambara groundnut flour mix (FPB) (70:30% w/w), and fermented popcorn: African locust bean: Bambara groundnut flour mixe (FPAB) (70:20:10% w/w). The ogi (corn gruel) and Cerelac (a commercial complementary formula) were used as the control food samples.
Chemical (proximate) analysis
Moisture, ash, crude fat, and crude fiber contents were determined using AOAC methods [17]. Nitrogen was determined by the micro Kjedahl method described by Pearson [18], and nitrogen content was converted to protein by multiplying by 6.25. Carbohydrate was determined as the difference.
Animal husbandry and experimental design
Sixty healthy albino rats (4 weeks of age) were obtained from the Central Animal House, College of Medicine, University of Ibadan, Ibadan. The rats were divided into six groups of 10 animals each. The cages were numbered 1-6. The rats in each group were fed commercial complementary food (Cerelac), local complementary food (ogi), the experimental diets and a basal respectively. The rats were acclimatized on a commercial diet (concentrate) and water ad libitum for 7 days. Thereafter, each of the animal groups was fed with FPA, FPB, FPAB, Cerelac, ogi, or the basal diet with water ad libitum each day for 28 days. Urine was collected from each cage in a small urine container, which contained about 1 cm3 concentrated sulfuric acid. Fecal samples were also collected daily, bulked for each rat, weighed, dried, and milled prior to laboratory analysis. Triplicate samples of feces, urine, and the diets were obtained for nitrogen determination by the Kjedahl method [17].
Weight gained by the animals and nitrogen were used to calculate the following parameters:
Nitrogen retention (NR) (dietary nitrogen retained in the body):
| NR = Ni - (Nf - Nef) - (Nu - Neu) |
Biological value (BV) [19]:
Food efficiency (FE):
Net protein utilization (NPU) (= BV × TD) [19]:
Protein efficiency ratio (PER) [20]:
True protein digestibility (TD) [21]:
Protein rating (PR):
| PR = PER × daily protein intake (g) |
Protein digestibility corrected amino-acid scores (PDCAAS):
 |
Ni, nitrogen intake; Nf, fecal nitrogen; Nef, endogenous fecal nitrogen; Nu, urinary nitrogen; Neu, endogenous urinary nitrogen; EAA, essential amino acids.
Metabolizable energy (ME) and digestible energy (DE) were calculated according to the method described by McCance and Widdowson [22] as modified by Göranzon et al. [23].
| ME = Energy intake - Energy in feces - Energy in urine |
Anthropometric measurements of the albino
Height and weight of the animals were measured at 7 day intervals for 28 days using standard techniques. All measurement was carried out in the morning between 9 am and 12 noon. Weight was measured using a digital weighing scale (Salter, SL20348, London, UK) calibrated to the nearest 0.1 kg. Length was measured using a meter board constructed by and calibrated to the nearest 0.1 cm. The animal was stretched along the meter board with the nostril touching the zero mark and the measurement was taken at the tip of the tail stretched along the meter board to the nearest 0.1 cm. The weight and length of the animals were related using weight-for-length and length-for-age indices to determine nutritional status.
Hematological determinations
Blood collection
On 28 day of the experimental period, all rats were starved for about 3 hours and weighed. Each rat was anaesthetized with chloroform inside a dessicator before been sacrificed. Blood was collected into Bijour bottles containing a speck of dried tetracetic ethylenediamine acid powder, and hematological indices were determined as described by Lambs [24].
Hematological analysis
Packed cell volume (PCV) was estimated by centrifuging about 75 Fl of each blood sample in heparinized capillary tubes in a hematocrit microcentrifuge for 5 minutes. Total red blood cell count (RBC) was determined using normal saline as the diluting fluid. Hemoglobin concentration (HBC) was estimated using the cyanomethemoglobin method and corpuscular hemoglobin concentration (MCHC) mean corpuscular hemoglobin (MCH), and mean corpuscular volume (MCV) were calculated.
Selection criteria for determining the optimal weaning food
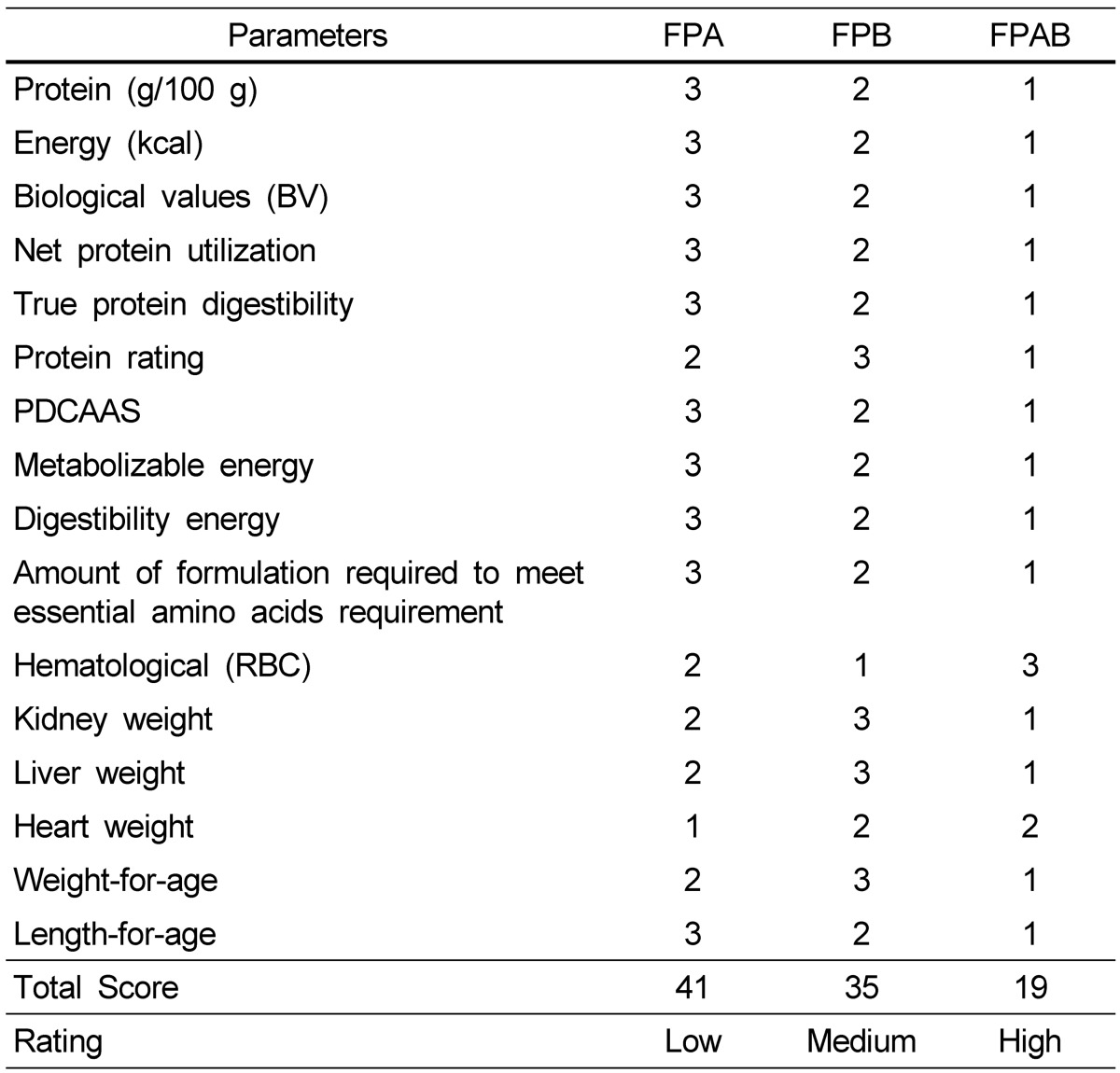
A ranking system using nutritional criteria (Table 1) was devised to determine the optimal blend combination according to the modified method of Griffith et al. [25]. Based on the relative importance and interrelationship of those criteria, ranking is reported on an equal weight basis. Ranking criteria produced identical results. The three blends were ranked from 1 to 3 (best to worst) to objectively determine the best-choice weaning blend. The blend yielding the lowest score was considered to possess the most suitable nutritional characteristics.
Table 1.
Ranking of formulated complementary foods to determine the optimal nutritional profile
PDCAAS, protein digestibility corrected amino acid score; RBC, red blood cells; FPA, fermented popcorn-African locust bean blend; FPB, fermented popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
Statistical analysis
Data were analyzed using SPSS version 15.0 (SPSS, Inc. Chicago, IL, USA). The mean and standard error of triplicate determinations were calculated. An analysis of variance was performed to determine significant differences between the parameters and the means were separated using the new Duncan multiple range test at P < 0.05.
Results
Macronutrient composition of the formulated diets
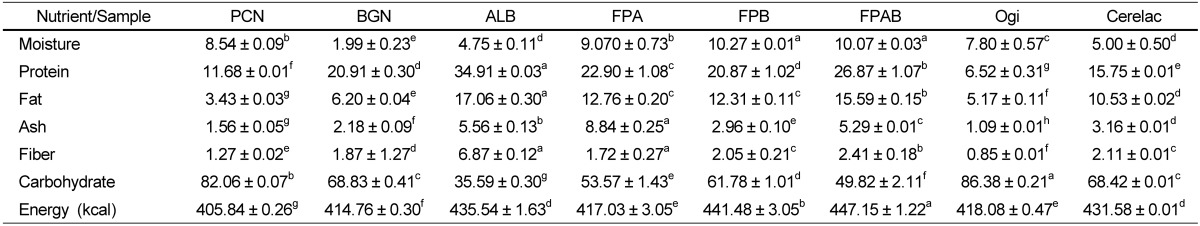
The macronutrient compositions of the raw and formulated complementary food samples are presented in Table 2. The protein content of fermented popcorn, African locust bean, and FPAB (26.87 ± 1.07 g/100 g) was significantly higher when compared with that of FPB (20.87 ± 1.02) and FPA (20.49 ± 1.08 g/100 g) (P < 0.05). The protein contents of the experimental diets were significantly higher than those of Cerelac and ogi (P < 0.05). Fat content of the flour mixes was 12.31 ± 0.11g/100 g for FPB and 15.59 ± 0.15 g/100 g for FPAB; and these values were significantly higher when compared with those of Cerelac and ogi, respectively (P < 0.05). Ash content of the FPA flour mix (8.84 ± 0.25 g/100 g) had the highest value, whereas that of FPB (2.96 ± 0.10 g/100 g) was the lowest; and a significant difference was observed between the experimental diets and control food samples (P < 0.05). The fiber content of the experimental diets ranged from 1.72 ± 0.27 g/100 g for the FPA flour mixe to 2.05 ± 0.21 g/100 g for the FPAB sample. The FPAB flour mix had the highest energy value (447.15 ± 1.22 kcal), whereas the FPA sample had the lowest energy value (417.03 ± 3.05 kcal) and significant differences were observed between the energy values of the experimental diets and control food samples (P < 0.05).
Table 2.
Mean (± standard error) of the macronutrient compositions (g/100 g weight matter) of fermented popcorn, African locust bean and Bambara groundnut flour blends
Mean values with different superscripts in a row are significantly different (P < 0.05).
PCN, raw popcorn; BGN, raw Bambara groundnut; ALB, raw African locust bean; FPA fermented popcorn-African locust bean blend; FPB, popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
Protein quality evaluation of the formulated diets using an animal model
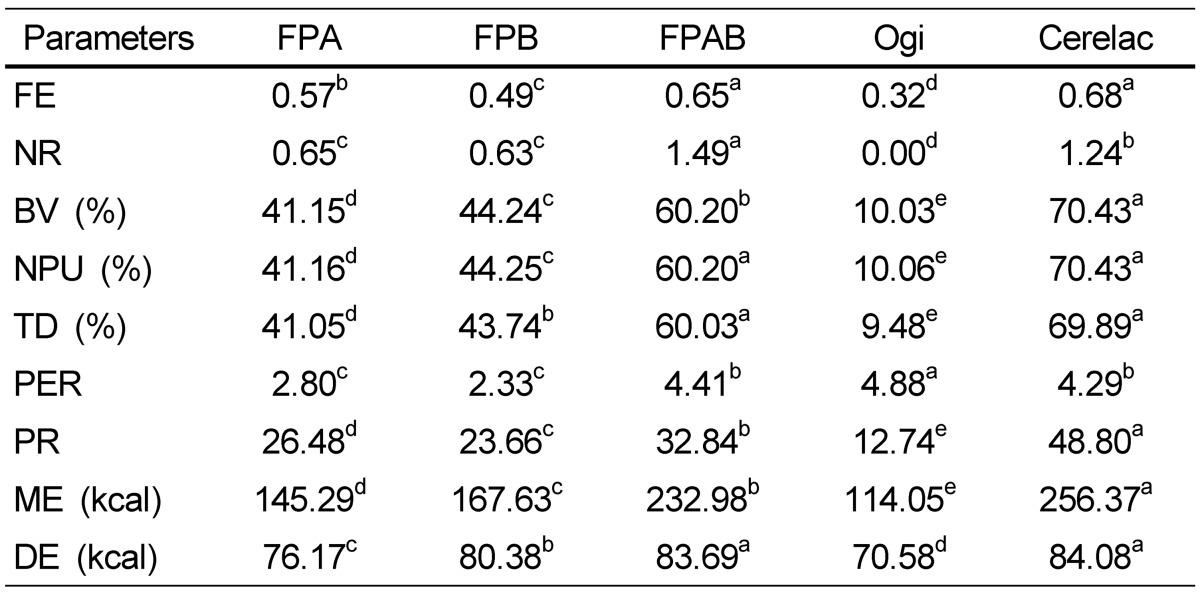
The protein quality, ME, and DE evaluation of the formulated foods and control samples are shown in Table 3. The results showed that the BV of FPAB (60.20%) was higher than those of FPB (44.24%), FPA (41.15%), ogi (10.03%), and Cerelac (50.43%). The NPU value of FPAB was the highest (60.20%) followed by FPB (44.25%), and FPA (41.16%). A significant difference was observed between FPAB and Cerelac (50.43%) and ogi (10.06%). The TD of FPAB was 60.05%, which was higher than that of the other formulated diets and control sample. Similarly, the FPAB protein rating was best compared to those of the remaining formulated diets and ogi, but less than that of Cerelac. ME and DE values of FPAB were higher than those of FPB, FPA, and ogi, but these values were less than that of Cerelac.
Table 3.
Evaluation of protein quality and metabolizable energy of the formulated food samples and control
Mean values with different superscripts in a row are significantly different (P < 0.05).
FE, food efficiency; NR, nitrogen retention; BV, iological value; NPU, net protein utilization; TD, true digestibility; PER, efficiency ratio; PR, protein rating; ME, metabolizable energy; DE, digestibilitle energy.
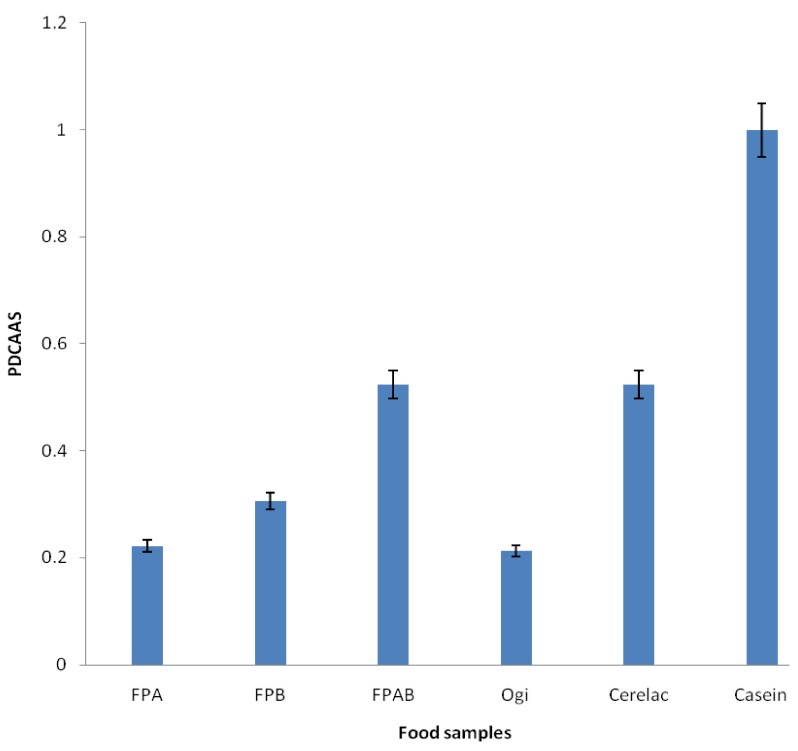
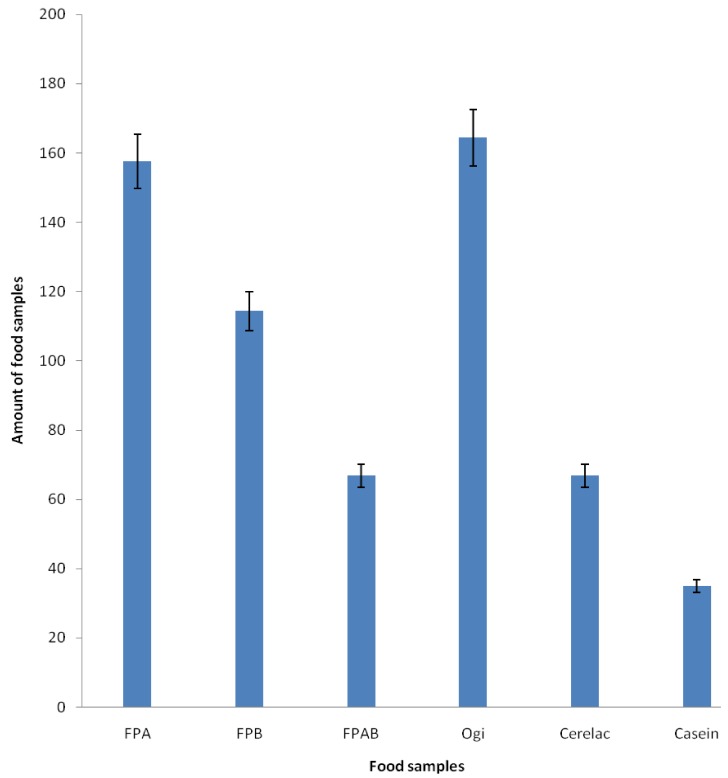
Fig. 1 shows the PDCAAS of the formulated and control food samples. The results showed that FPAB had the highest value (0.44), whereas FPA had the lowest value (0.22). The PDCAAS values of the formulated food samples were lower than those of Cerelac (0.52) and casein (1.0) (a reference standard), but higher than that of ogi (0.21). The amount of food sample required to meet recommended daily allowance of total essential amino acids for infants was lower in FPAB than that in the other formulated samples and ogi, but higher than that of casein and comparable to Cerelac. Amount of formulated diet required to meet the essential amino acid requirement of is presented in Fig. 2. The amount of fermented popcorn-African locust-Bambara groundnut blend (FPAB) required to meet the total amino acid requirement of children was lower when compared with that of fermented popcorn-African locust bean blend (FPA), fermented popcorn-Bambara groundnut blend (FPB) and ogi, but higher than that of cerelac.
Fig. 1.
Comparison of protein digestibility using the corrected amino acid score (PDCAAS) of the formulated food samples, ogi (a traditional complementary food), Cerelac (a commercial formula), and casein (standard) (P < 0.05). FPA, fermented popcorn-African locust bean blend; FPB, fermented popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
Fig. 2.
Amount of the formulated food samples required to meet total essential amino acid requirements compared with that of ogi (a traditional complementary food), Cerelac (a commercial formula), and casein (standard) (P < 0.05). FPA, fermented popcorn-African locust bean blend; FPB, fermented popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
Growth and developmental patterns of animals fed the formulated diets
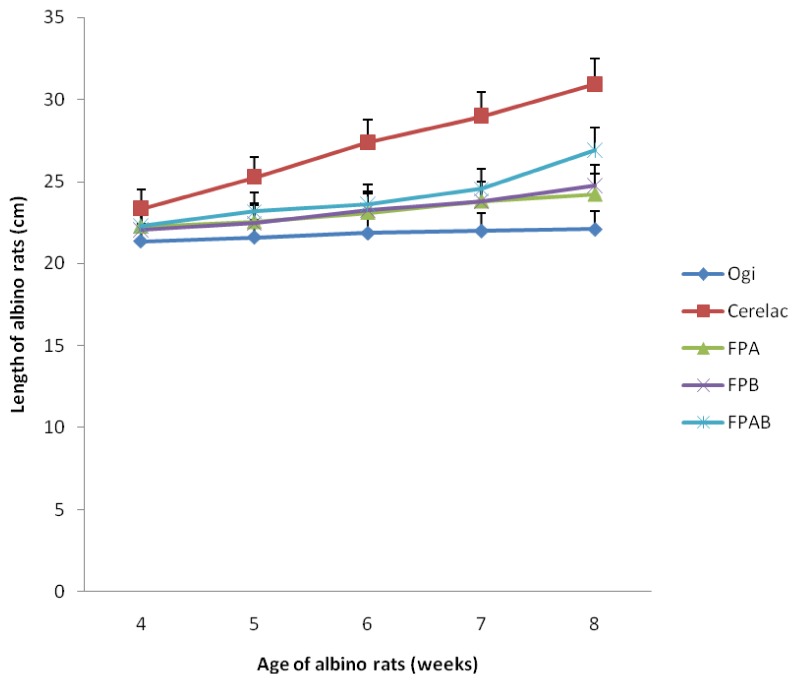
Weight-for-age and length-for-age of albino rats fed the formulated complementary foods compared with rats fed ogi and Cerelac are presented in Figs. 3 and 4. The weight-for-age classification of the animals showed that the growth patterns of animals fed the formulated food samples were better than those of animals fed ogi, but lower when compared with animals fed Cerelac. Similarly, for the length-for-age classification, the growth patterns of animals fedthe formulated diets were better than those fed ogi, but lower when compared with animals fed Cerelac. Animals fed the FPAB diets had the best growth rate comapred to those fed the FPA and FPB diets.
Fig. 3.
Weight-for-age in albino rats fed with the fermented formulated complementary foods compared with rats fed ogi (a traditional complementary food) and Cerelac (a commercial formula) (P < 0.05). FPA, fermented popcorn-African locust bean blend; FPB, fermented popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
Fig. 4.
Lenght-for-age in albino rats fed the fermented formulated complementary foods compared with rats fed ogi (a traditional complementary food) and Cerelac (a commercial formula) (P < 0.05). FPA, fermented popcorn-African locust bean blend; FPB, fermented popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
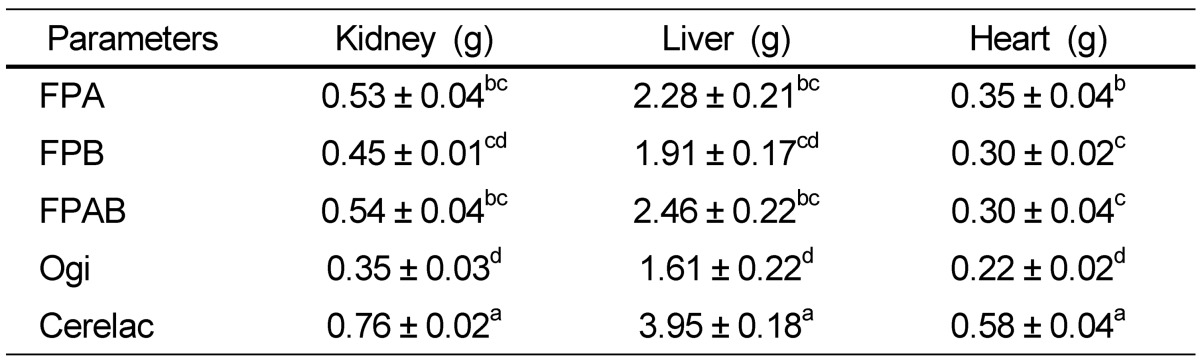
The influence of the diets on the organs of the animals is shown in Table 4. The weight range of the organs in animals fed the formulated diets were as follows: the kidney value was between 0.45 ± 0.01 g for FPB and 0.54 ± 0.04 g for FPAB; for the liver, 1.91 ± 0.17g for FPB and 2.46 ± 0.22 for FPAB, whereas that of the heart was 0.30 ± 0.02 g for FPAB and 0.35 ± 0.04 g for FPA. The weight of the kidneys, liver, and heart of animals fed the formulated diets were significantly higher than those of animals fed ogi, but lower than those fed Cerelac (P < 0.05).
Table 4.
Effect of formulated food samples on organ development in albino rats
Mean values with different superscripts in a row are significantly different (P < 0.05).
FPA, fermented popcorn-African locust bean blend; FPB, fermented popcorn-Bambara groundnut blend; FPAB, fermented popcorn-African locust-Bambara groundnut blend.
Hematological properties of animals fed the formulated diets
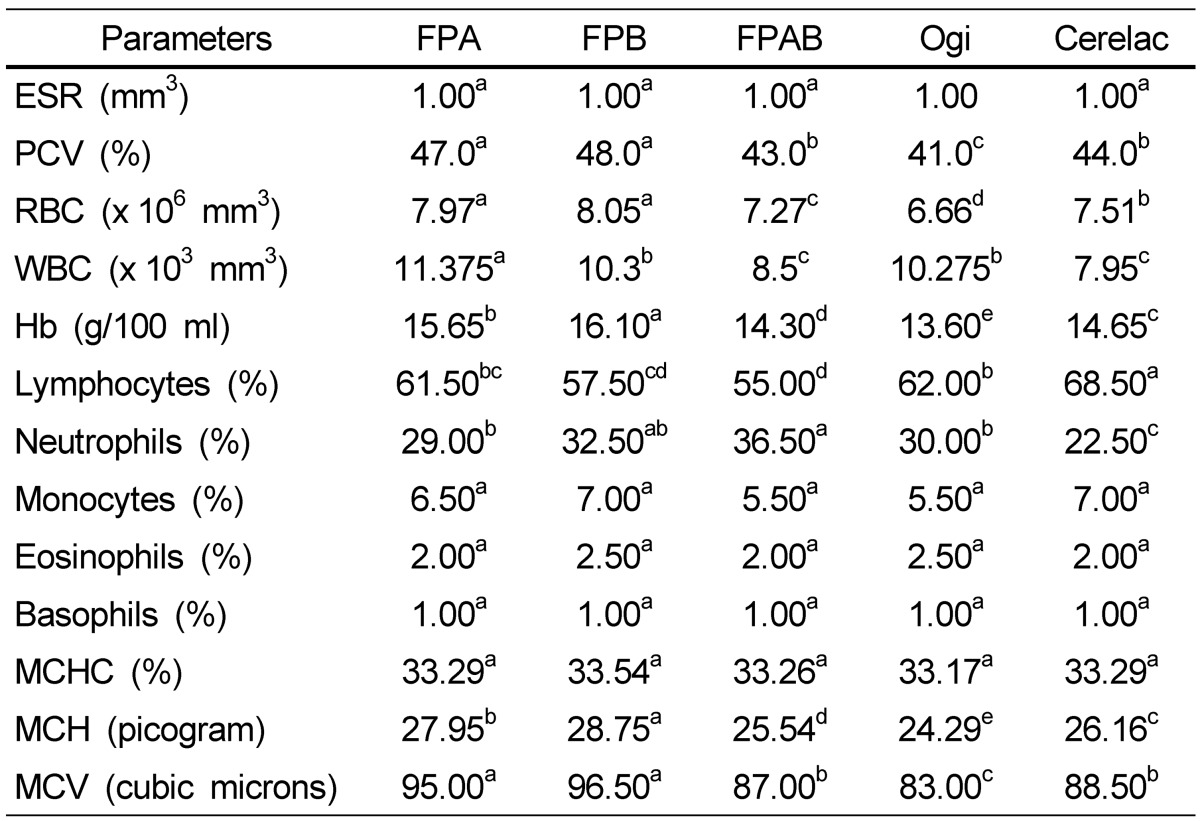
Table 5 shows the hematological properties of albino rats fed the fermented complementary food samples compared with those fed the control food samples. The results showed no significant differences between the erythrocyte sedimentation rate, monocytes, eosinophils, basophils, and MCHC in animals fed any of the foods. However, PCV, RBCs, white blood cells (WBC), hemoglobin (Hb), MCH and MCV of animals fed the FPB and FPA were significantly higher when compared with those fed the FPAB and control food samples (P < 0.05).
Table 5.
Hematological properties of albino rats fed the fermented complementary and control food samples
Mean values with different superscripts in a row are significantly different (P < 0.05).
ESR, erythrocyte sedimentation rate); PCV, packed cell volume; RBC, red blood cells; WBC, white blood cells; Hb, hemoglobin; MCHC, mean cell hemoglobin concentration; MCH, mean cell hemoglobin; MCV, mean cell volume.
Discussion
Poor protein quality and low energy density of complementary foods are major factors affecting infant and young child feeding practices in Nigeria and other developing countries [26,27]. Complementary foods in developing countries contain high levels of carbohydrate with very low or no protein due to the high cost of protein-rich foods and poor knowledge of locally available food materials by nursing mothers [28]. Thus, we investigated the production of complementary foods from locally available cereals and legumes. The findings showed that the protein and energy-density of the food samples were better than those of the traditional complementary food (ogi, a corn gruel) and comparable to a commercial formula (Cerulac). The energy values of the formulated food samples were higher than the minimum desirable level (370 kcal/100 g) for infant complementary foods as recommended by Walker [29]. Adequate quality complementary food is important for infants during normal growth and development [30]. The protein and energy density of complementary food must be sufficient to meet protein and energy needs of a growing infant.
The nutritional quality of formulated food products in terms of BV, TPD, PR, ME and DE were investigated. We established that FPAB, a combination of popcorn, African locust bean, and bambara groundnut was rated higher for these parameters when compared with other formulated food products and ogi, but comparable to those of Cerelac. A combination of cereals with two or more inexpensive plant protein sources such as legumes can be highly beneficial, as the nutritive value of the product improves [28]. The amount of FPAB needed to meet infant essential amino acid requirements was lower than that of the FPA and FPB food samples, but comparable to that of Cerelac. The PDCAAS a nutritional index to predict protein quality and suitability of the formulated diets was higher than that of ogi, but lower than those of Cerelac and casein. This observation could be attributed to the fact that Cerelac is made from milk. However, no significant difference was observed between the PDCAAS of FPAB and that of Cerelac; therefore, the FPAB blend was suitable as a complementary food for infants and young children. The PDCAAS has been recommended to be the most suitable method for routinely evaluating protein quality and highly digestible food products for human nutrition by the FAO/WHO [31]. The PDCAAS method includes the use of young rats for predicting protein digestibility of foods for all ages including the elderly [32].
The RBCs, WBCs, PCV and Hb values were quite high and significant differences were observed between the formulated diets and those of Cerelac and ogi. These high values indicate the adequacy of the formulated diets for promoting good blood health status.
The mean age, weight, and length of the animals were related using weight-for-age and height-for-age nutritional indices. The findings revealed that the animals fed FPAB had better growth rates when compared with those of animals fed the FPA and FPB formulations. The growth patterns of animals fed the experimental diets were significantly better than those of animals fed ogi, but lower than those of Cerelac. The formulated diets promoted growth and development of the organs (kidney, liver and heart) of animals better than that than ogi, but not as well as Cerelac. However, the FPAB food sample promoted growth and development of these organs better than that of the other formulated diets. Considering the nutritional potential of these formulated diets, they appear to be suitable as complementary foods, particularly for children of low-income families who cannot afford animal protein-based complementary foods and who are weaned on ogi.
Quite a number of nutritional studies have reported that most traditional complementary foods in developing countries are low in protein and energy density [27,33-37]. This nutrition problem has led to an increase in the prevalence of protein-energy malnutrition; hence, high rates of morbidity [8-10], mortality [11], impaired intellectual development [38,39]. and growth retardation [40-43]. Thus, a low-cost complementary food that is high in protein and dense in energy is a desirable substitute for expensive imported complementary foods to combat protein-energy malnutrition effectively [44]. Therefore, the formulations in the present study can be considered suitable for preventing protein-energy malnutrition in children.
The FPAB sample was ranked the best when compared with FPB and FPA (Table 1). All formulated diets were better than ogi, but only the FPAB sample was particularly comparable to Cerelac in terms of protein quality; therefore, it may be used as a substitute for traditional complementary food.
References
- 1.Lutter C. Meeting the challenge to improve complementary feeding. SCN News. 2003;(27):4–9. [Google Scholar]
- 2.Lutter CK, Rivera JA. Nutritional status of infants and young children and characteristics of their diets. J Nutr. 2003;133:2941S–2949S. doi: 10.1093/jn/133.9.2941S. [DOI] [PubMed] [Google Scholar]
- 3.Daelmans B, Saadeh R. Global initiatives to improve complementary feeding. SCN News. 2003;(27):10–18. [Google Scholar]
- 4.Anigo KM, Ameh DA, Ibrahim S, Danbauch S. Infant feeding practices and nutritional status of children in North Western Nigeria. Asian J Clin Nutr. 2009;1:12–22. [Google Scholar]
- 5.Pollitt E. Poverty and child development: relevance of research in developing countries to the United States. Child Dev. 1994;65:283–295. [PubMed] [Google Scholar]
- 6.Duncan GJ, Brooks-Gunn J, Klebanov PK. Economic deprivation and early childhood development. Child Dev. 1994;65:296–318. [PubMed] [Google Scholar]
- 7.Kretchmer N, Beard JL, Carlson S. The role of nutrition in the development of normal cognition. Am J Clin Nutr. 1996;63:997S–1001S. doi: 10.1093/ajcn/63.6.997. [DOI] [PubMed] [Google Scholar]
- 8.Brown KH, Black RE, López de Romaña G, Creed de Kanashiro H. Infant-feeding practices and their relationship with diarrheal and other diseases in Huascar (Lima), Peru. Pediatrics. 1989;83:31–40. [PubMed] [Google Scholar]
- 9.Bhandari N, Bahl R, Taneja S, Strand T, Mølbak K, Ulvik RJ, Sommerfelt H, Bhan MK. Substantial reduction in severe diarrheal morbidity by daily zinc supplementation in young North Indian children. Pediatrics. 2002;109:e86. doi: 10.1542/peds.109.6.e86. [DOI] [PubMed] [Google Scholar]
- 10.Kalanda BF, Verhoeff FH, Brabin BJ. Breast and complementary feeding practices in relation to morbidity and growth in Malawian infants. Eur J Clin Nutr. 2006;60:401–407. doi: 10.1038/sj.ejcn.1602330. [DOI] [PubMed] [Google Scholar]
- 11.Edmond KM, Zandoh C, Quigley MA, Amenga-Etego S, Owusu-Agyei S, Kirkwood BR. Delayed breastfeeding initiation increases risk of neonatal mortality. Pediatrics. 2006;117:e380–e386. doi: 10.1542/peds.2005-1496. [DOI] [PubMed] [Google Scholar]
- 12.Brown KH. The importance of dietary quality versus quantity for weanlings in less developed countries: a framework for discussion. Food Nutr Bull. 1991;13:86–94. [Google Scholar]
- 13.Golden MH. The nature of nutritional deficiency in relation to growth failure and poverty. Acta Paediatr Scand Suppl. 1991;374:95–110. doi: 10.1111/j.1651-2227.1991.tb12012.x. [DOI] [PubMed] [Google Scholar]
- 14.Naismith DJ. Kwashiorkor in Western Nigeria: a study of traditional weaning foods, with particular reference to energy and linoleic acid. Br J Nutr. 1973;30:567–576. doi: 10.1079/bjn19730063. [DOI] [PubMed] [Google Scholar]
- 15.Fashakin JB, Ogunsola F. The utilization of local foods in the formulation of weaning foods. J Trop Pediatr. 1982;28:93–96. doi: 10.1093/tropej/28.2.93. [DOI] [PubMed] [Google Scholar]
- 16.Oyenuga VA. Nigeria's Foods and Feeding-Stuffs: Their Chemistry and Nutritive Value. Nigeria: Ibadan University Press; 1968. pp. 234–457. [Google Scholar]
- 17.Association of Official Analytical Chemists (AOAC) Official Methods of Analysis of the Association of Analytical Chemists International. 18th ed. Washington, D. C.: AOAC; 2005. [Google Scholar]
- 18.Pearson D. The Chemical Analysis of Foods. 6th ed. London: Churchill; 1976. pp. 6–7. [Google Scholar]
- 19.Phillips DE, Erye MD, Thompson A, Boulter D. Protein quality in seed meals of Phaseolus vulgaris and heat-stable factors affecting the utilisation of protein. J Sci Food Agric. 1981;32:423–432. doi: 10.1002/jsfa.2740320502. [DOI] [PubMed] [Google Scholar]
- 20.National Academy of Sciences (NAS); National Research Council (NCR) Evaluation of Protein Quality. Washington, D.C.: NAS/NCR; 1963. pp. 23–37. [Google Scholar]
- 21.Dreyer JJ. Biological assessment of protein quality: digestibility of the proteins in certain foodstuffs. S Afr Med J. 1968;42:1304–1313. [PubMed] [Google Scholar]
- 22.McCance RA, Widdowson EM. The Composition of Foods. London: Medical Research Council; 1960. Special Report Series No. 297. [Google Scholar]
- 23.Göranzon H, Forsum E, Thilén M. Calculation and determination of metabolizable energy in mixed diets to humans. Am J Clin Nutr. 1983;38:954–963. doi: 10.1093/ajcn/38.6.954. [DOI] [PubMed] [Google Scholar]
- 24.Lamb GM. Manual of Veterinary Laboratory Technique in Kenya. Kenya: Ciba-Geigy; 1981. pp. 96–102. [Google Scholar]
- 25.Griffith LD, Castell-Perez ME, Griffith ME. Effects of blend and processing method on the nutritional quality of weaning foods made from select cereals and legumes. Cereal Chem. 1998;75:105–112. [Google Scholar]
- 26.Bennett VA, Morales E, González J, Peerson JM, López de Romaña G, Brown KH. Effects of dietary viscosity and energy density on total daily energy consumption by young Peruvian children. Am J Clin Nutr. 1999;70:285–291. doi: 10.1093/ajcn.70.2.285. [DOI] [PubMed] [Google Scholar]
- 27.Mosha TCE, Laswai HS, Tetens I. Nutritional composition and micronutrient status of home made and commercial weaning foods consumed in Tanzania. Plant Foods Hum Nutr. 2000;55:185–205. doi: 10.1023/a:1008116015796. [DOI] [PubMed] [Google Scholar]
- 28.Amankwah EA, Barimah J, Acheampong R, Addai LO, Nnaji CO. Effect of fermentation and malting on the viscosity of maize-soyabean weaning blends. Pak J Nutr. 2009;8:1671–1675. [Google Scholar]
- 29.Walker AF. The contribution of weaning foods to protein-energy malnutrition. Nutr Res Rev. 1990;3:25–47. doi: 10.1079/NRR19900005. [DOI] [PubMed] [Google Scholar]
- 30.McGuire J. Quality versus quantity of infant diets: translating research into action. Food Nutr Bull. 1991;13:132–134. [Google Scholar]
- 31.Food & Agriculture Organization of the United Nations (FAO); World Health Organization (WHO) Protein Quality Evaluation: Report of the Joint FAO/WHO Expert Consultation. Rome: FAO; 1991. FAO Food and Nutrition Paper 51. [Google Scholar]
- 32.Gilani GS, Sepehr E. Protein digestibility and quality in products containing antinutritional factors are adversely affected by old age in rats. J Nutr. 2003;133:220–225. doi: 10.1093/jn/133.1.220. [DOI] [PubMed] [Google Scholar]
- 33.Abbey BW, Nkanga UB. Production of high quality weaning products from Maize-Cowpea-Crayfish mixtures. Nutr Rep Int. 1988;37:951–957. [Google Scholar]
- 34.Michaelsen KF, Friis H. Complementary feeding: a global perspective. Nutrition. 1998;14:763–766. doi: 10.1016/s0899-9007(98)00079-3. [DOI] [PubMed] [Google Scholar]
- 35.Urga K, Narasimha HV. Phytate:zinc and phytate x calcium:zinc molar ratios in selected diets of Ethiopians. Bull Chem Soc Ethiop. 1998;12:1–7. [Google Scholar]
- 36.Hurrell RF. Influence of vegetable protein sources on trace element and mineral bioavailability. J Nutr. 2003;133:2973S–2977S. doi: 10.1093/jn/133.9.2973S. [DOI] [PubMed] [Google Scholar]
- 37.Mbithi-Mwikya S, Van Camp J, Mamiro PR, Ooghe W, Kolsteren P, Huyghebaert A. Evaluation of the nutritional characteristics of a finger millet based complementary food. J Agric Food Chem. 2002;50:3030–3036. doi: 10.1021/jf011008a. [DOI] [PubMed] [Google Scholar]
- 38.Berkman DS, Lescano AG, Gilman RH, Lopez SL, Black MM. Effects of stunting, diarrhoeal disease, and parasitic infection during infancy on cognition in late childhood: a follow-up study. Lancet. 2002;359:564–571. doi: 10.1016/S0140-6736(02)07744-9. [DOI] [PubMed] [Google Scholar]
- 39.Hamadani JD, Fuchs GJ, Osendarp SJ, Khatun F, Huda SN, Grantham-McGregor SM. Randomized controlled trial of the effect of zinc supplementation on the mental development of Bangladeshi infants. Am J Clin Nutr. 2001;74:381–386. doi: 10.1093/ajcn/74.3.381. [DOI] [PubMed] [Google Scholar]
- 40.Dewey KG, Heinig MJ, Nommsen LA, Peerson JM, Lönnerdal B. Growth of breast-fed and formula-fed infants from 0 to 18 months: the DARLING Study. Pediatrics. 1992;89:1035–1041. [PubMed] [Google Scholar]
- 41.Umeta M, West CE, Haidar J, Deurenberg P, Hautvast JG. Zinc supplementation and stunted infants in Ethiopia: a randomised controlled trial. Lancet. 2000;355:2021–2026. doi: 10.1016/S0140-6736(00)02348-5. [DOI] [PubMed] [Google Scholar]
- 42.Rivera JA, Hotz C, González-Cossío T, Neufeld L, García-Guerra A. The effect of micronutrient deficiencies on child growth: a review of results from community-based supplementation trials. J Nutr. 2003;133:4010S–4020S. doi: 10.1093/jn/133.11.4010S. [DOI] [PubMed] [Google Scholar]
- 43.Michaelsen KF, Hoppe C, Roos N, Kaestel P, Stougaard M, Lauritzen L, Mølgaard C, Girma T, Friis H. Choice of foods and ingredients for moderately malnourished children 6 months to 5 years of age. Food Nutr Bull. 2009;30:S343–S404. doi: 10.1177/15648265090303S303. [DOI] [PubMed] [Google Scholar]
- 44.Ijarotimi OS, Jr, Olopade AJ. Determination of amino acid content and protein quality of complementary food produced from locally available food materials in ondo state, Nigeria. Malays J Nutr. 2009;15:87–95. [PubMed] [Google Scholar]