The problem
Diabetes is prevalent and chronic (1). A meta-analysis of studies published between 1990 and 2006 showed that in adults, obesity prevalence increased from 13 to 32% between the 1960s and 2004. Currently, 66% of U.S. adults are overweight or obese.
The incidence of diabetes is increasing and afflicting new populations including children and developing societies. Changes in obesity prevalence among children and teens tripled, from nearly 5% to approximately 15% since the 1960s (2). Type 2 diabetes mellitus (T2DM) is a global health problem. In particular, Asia is at the epicenter of the epidemic as these populations develop diabetes at younger ages and at lower BMI levels than people of European origin (3).
Diabetes does not usually diminish over time despite standard treatment. It is a lifelong illness that generally worsens with time and often leads to debilitating complications including cardiovascular disease, neuropathy, retinopathy, and nephropathy (4).
These realities indicate that both our understanding of the disease and our treatment of the disease are inadequate. Current approaches are not working.
Current disease model
It is widely believed that overeating calorie-dense food, particularly a high-fat diet, together with an inactive lifestyle causes obesity. A consequence of obesity is increased circulating lipids and cytokines, indicators of inflammation and inducers of insulin resistance. Obesity and insulin resistance are believed to be the precursors of diabetes in about 20% of subjects. The current standard treatment for diabetes includes diet, exercise, and a biguanide together with agents that increase circulating insulin in an effort to overcome insulin resistance (5).
The term Insulin Resistance Syndrome is used to describe the combination of insulin resistance and compensatory hyperinsulinemia (HI). It is assumed, without evidence, that insulin resistance precedes and causes HI. Insulin resistance purportedly stimulates increased insulin secretion, interpreted as the body’s attempt to overcome the resistance, however, there is no satisfactory explanation for how insulin resistance might stimulate insulin secretion. In addition, there are few naturally occurring or genetic models of primary insulin resistance and few diabetes genes are implicated in insulin resistance. Although this has been the prevalent model under investigation by basic scientists and a major focus for drug development, we have no viable drugs to improve long-term metabolic health. Perhaps the model is not viable and we need to look elsewhere.
It is well documented that average food consumption has increased and that activity has decreased and both correlate with insulin resistance, however, this does not prove or infer causation. There is no evidence that overeating and inactivity are the initiating causes of obesity and diabetes in our population. They may simply coexist or be caused by other yet unidentified factors. Where in the multiple tissue/metabolic interactions shown in Fig. 1 does the trigger lie? Is there only one trigger?
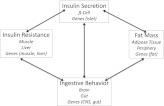
Figure 1.
Potential initiating defects and interactions in obesity/diabetes. Illustration of the interconnectedness among insulin secretion, fat mass, insulin resistance, and ingestive behavior such that a primary defect in any one could lead to the observed alterations in each of the others that occur in the development of T2DM. To illustrate: A β-cell defect that initially caused inappropriate hypersecretion of insulin at basal glucose would lower blood glucose, thereby stimulating eating that would increase fat mass and lead to insulin-induced downregulation of peripheral insulin receptors with ensuing insulin resistance. Alternatively, a defect in sensing hunger or satiety would promote overeating resulting in insulin secretion and increased fat mass with ensuing insulin resistance. On the other hand, excessive fat mass would increase circulating lipids and cytokines to cause insulin resistance and hyperinsulinemia. CNS, central nervous system.
Not all overweight/obese individuals are insulin resistant nor are all insulin resistant individuals overweight/obese (6). Furthermore, there is evidence that alterations in food consumption do not cause sustained weight change. An elegant study by Sims et al. (7) in the 1970s found that experimentally overfed volunteers required nearly 6,000 calories per day to maintain a 20% increase in their body weight and most of these subjects rapidly returned to their normal lower weight at the end of the study. Abundant evidence documents the failure of most dieters to sustain weight loss. In a review of studies of the long-term outcomes of diets to determine if dieting is an effective therapy for obesity, it was found that one-third to two-thirds of dieters regain more weight than they lost on their diets (8). These studies also did not provide consistent evidence that dieting results in significant long-term health improvements (8). Furthermore, the prevalent focus on the evils of dietary fat and their implied causative role in obesity are not well documented by scientific evidence. Indeed, the decrease in fat consumption that has resulted from abundant (nonevidence-based) medical and governmental advice against fat consumption has not decreased the burden of metabolic disease in our society (9).
An alternative perspective is that insulin resistance is not the cause of diabetes, but is rather a beneficial adaptive mechanism to HI and/or hyperlipidemia. Insulin functions to store all fuels. Excess circulating lipids and amino acids can increase insulin secretion in the presence of normal circulating glucose levels. Thus, in the setting of normal blood glucose, insulin elevation caused by fat or amino acids would result in hypoglycemia. This does not happen because glucose transport into muscle and fat is diminished, because it is resistant to the normal ability of insulin to promote glucose transport.
Finding the right model is crucial to finding the right treatment. The therapeutic approach to further increase insulin, as often recommended to T2DM patients, may actually be exacerbating the disease. Resistance may prove to be secondary to another defect or even beneficial by facilitating excess fat storage without causing hypoglycemia that could ensue from “curing” resistance. Pharmacologic attempts to abolish this mechanism have largely failed as metabolic regulatory mechanisms appear able to overcome our efforts, suggesting that the body’s metabolic wisdom trumps ours.
Alternative considerations
Environmental agents
Almost nothing has remained constant during the several decades of the evolution of the diabetes epidemic: environmental temperature, altered chemicals in agricultural practice, the use of drugs in our food animals, and the processing and packaging of our foods (10). These include pesticides, hormones, drugs, food additives, plasticizers, etc. (11). In a study of pregnant woman in the National Health and Nutrition Examination Survey (NHANES), blood levels of a number of environmental pollutants were measured. It was found that certain polychlorinated biphenyls, organochlorine pesticides, perfluorinated chemicals, phenols, polybrominated diphenyl ethers, phthalates, polycyclic aromatic hydrocarbons, and perchlorate were detected in 99–100% of pregnant women (12).
Diabetogenic agents
Some environmental pollutants have been shown to alter β-cell function, although it has not yet been determined whether prevailing environmental concentrations of these types of compounds can be a risk factor for diabetes. An intimation that the general population is exposed to concentrations that may cause diabetes is derived from epidemiological studies (13). However, clear causality between human exposure to pollutants and diabetes has not been established. In contrast to most agents studied, a particularly good case has been made for BPA (bisphenol A) as a potential cause of diabetes (14). BPA is a compound that has been considered a weak estrogen based on its lower binding affinity to nuclear estrogen receptors. It is commonly used in food and beverage container manufacturing. Animal studies have demonstrated that it can interfere with endocrine signaling pathways at low doses during fetal, neonatal, or perinatal periods—as well as in adulthood—and cause impaired β-cell function (14).
Advanced glycation end products, strongly associated with the complications of diabetes, can be formed in vivo but importantly are also derived from dietary components and are known to contribute to increased oxidant stress and inflammation in humans (15).
A new approach
The lack of solid evidence linking environmental pollutants, antibiotics, and growth stimulants present in food animals and food additives to metabolic health is due for the most part to the absence of data evaluating these possibilities.
In the absence of evidence proving that the current focus on overindulgence and idleness as causes of insulin resistance (blaming patients for their disease) is valid, it is suggested that we increase our focus on the thousands of factors that have changed during this epidemic and evaluate alternative models.
There is general consensus that the cause of the epidemic increase in diabetes and obesity involves environmental rather than genetic changes since this has occurred only in the past 50 years. However, the susceptibility to environmental factors may be genetically determined, explaining the variable susceptibility among populations and families. It should be noted that the majority of genes associated with diabetes are β-cell genes (16) whereas many of the obesity genes point toward a role for central neuronal signaling pathways, adipogenesis, or energy metabolism (17).
Alternative models
In the absence of proof of a primary role for insulin resistance in the development of diabetes, what alternative hypotheses are worth testing? It seems rational to focus on modifiable elements in our environment that have changed during the time of the epidemic. There are several broad primary mechanisms, in addition to insulin resistance, that could cause obesity leading to diabetes and are consistent with available data. Each of these mechanisms has the potential to theoretically be the primary cause and lead to the other characteristics of T2DM (Fig. 1).
1. Defective hunger or satiety signals
Excess nutrient ingestion stimulates insulin secretion, fat storage, and consequent insulin resistance. Excess nutrient ingestion can be stimulated through the use of common psychotropic drugs (18), gold thioglucose (19), and also has been documented in genetic defects in leptin or leptin receptor signaling (20).
Excess nutrient ingestion as a target for obesity treatment has been the focus of pharmacological research attempting to identify agents that decrease hunger or increase satiety. The side effects of many centrally acting psychotropic drugs on weight gain (21) prompted the development of drugs to specifically impact eating behavior. This effort has not resulted in a major useful drug (22). Even successful drugs that modulate appetite or satiety have had only modest effects (23) that disappear upon cessation of therapy. In addition, many problems have arisen due to misuse or side effects of these agents.
The possibility that the gut plays a major role in appetite regulation has not been investigated as thoroughly (24). In the clinical narrative of this Bench to Clinic Symposia that follows this “preclinical” perspective (25), the cure of diabetes following gastric bypass surgery supports the possibility of an important and hitherto unrecognized role for the gut in diabetes. Interestingly, ghrelin receptor-null mice eat less food, store less of their consumed calories as fat, use more fat as an energy substrate, and accumulate less body weight than control mice (26). Again, environmental changes that affect secreted gut peptides have not yet been identified, however, there has also been little investigation of environmental agents on gut function or the microbiome (27).
Nevertheless, the possibility that environmental agents that we have not yet studied or identified have a primary effect to increase ingestive behavior or decrease satiety via central or peripheral mechanisms cannot be eliminated.
2. Excessive increase in body fat accumulation
Obesity is strongly associated with hypersecretion of insulin and insulin resistance. Free fatty acids (FFAs) have emerged as an important link between insulin secretion and insulin resistance. FFAs are elevated in obesity. Decreasing FFAs decrease insulin secretion and increase insulin sensitivity implying that FFAs or lipids from excessive adipose tissue mass may explain the increasing incidence of HI and T2DM. Increases in plasma FFAs can account for a large part of HI and insulin resistance in obese patients with T2DM. The mechanisms by which plasma FFAs cause HI and insulin resistance include increased generation of lipid metabolites, reactive oxygen species (ROS), and proinflammatory cytokines (28).
In obese individuals, does the increase of fat mass play a causative role in HI? It would seem not. For example, thiazolidinediones increase adipogenesis and fat mass but actually decrease HI and improve insulin sensitivity (5). In addition, recent studies reveal no improvement in metabolic parameters as a consequence of surgical fat removal diminishing the likelihood of increased fat mass as a primary lesion (29). Other mechanisms are possible, though, such as altered adipokine production induced by environmental agents that have not yet been identified or studied.
3. Mitochondrial DNA and epigenetic changes
Metabolic disease is frequently associated with mitochondrial defects (30,31). Despite this, mitochondrial DNA in diabetes and obesity has been studied in considerably fewer subjects than nuclear DNA, with the exception of rare instances of maternally transmitted diabetes (32). In addition to regulation of mitochondrial function, nuclear DNA expression is modulated by histone phosphorylation and acetylation requiring ATP and acetyl-CoA generated by mitochondria to facilitate increased specific gene expression when energy sources are abundant. Phosphorylation, acetylation, and cellular redox state also regulate most signal transduction pathways and activities of multiple transcription factors involved in many common diseases of aging, including heart disease, diabetes, Parkinson disease, and dementia (33).
In addition, a role for epigenetics cannot be ruled out since DNA methylation and histone modification, both of which serve to regulate gene expression without altering the underlying DNA sequence, may respond to environmental factors (34). Such dysregulation or posttranscriptional modifications of histones in chromatin, including histone lysine methylation, have been implicated in aberrant gene regulation associated with the pathology of diabetes and its complications, and genome-wide studies have shown cell-specific changes in histone methylation under diabetic conditions (35).
Thus, both mitochondrial DNA and epigenetic changes could play a role in the etiology of diabetes through any of the organ systems considered here.
4. A defect in the insulin-secreting β-cell
The hypothesis favored in this perspective is that HI causes obesity leading to diabetes and is induced by exposure to environmental β-cell toxins (Fig. 2). There is published evidence for this, and it is a testable alternative to insulin resistance.
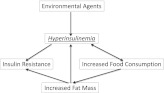
Figure 2.
Model of environmental agent–induced hypersecretion of insulin. In this model, an environmental agent such as monoglyceride or saccharin increases basal insulin secretion. This stimulates food consumption and insulin resistance leading to increased fat mass. Increased fat mass generates fatty acids and cytokines that further enhance both insulin resistance and secretion. In the defect modeled here, the best way to cure the metabolic disease is to prevent HI.
Supporting evidence
Hypersecretion of insulin (HI) by the β-cell in the absence of a stimulatory fuel can increase ingestive behavior through decreased blood glucose. Increased food consumption leads to increased fat stores. HI also can cause insulin resistance through insulin-induced receptor downregulation both in the periphery (36) and in the brain where HI-induced insulin resistance may abbrogate its normal role as a satiety signal (37,38).
Rodents overexpressing the human insulin gene or treated with exogenous insulin develop insulin resistance secondary to HI (39). In contrast, lowering insulin levels with diazoxide increases insulin sensitivity in rodents and humans (40–42).
Screening in our laboratory has identified a number of environmental compounds that increase insulin secretion in β-cells at a nonstimulatory glucose concentration. These include food preservatives, such as monoacylglycerides (43), the artificial sweetener saccharin, and excess iron (10). There have also been reports of other environmental agents that stimulate insulin secretion at basal glucose levels. These include estrogenic compounds including bis-phenol A (14,44), plant extracts (45,46), and certain viruses (47).
Thus, agents that cause primary HI are readily identified and are potential participating initiators of the current diabetes epidemic. The β-cell as the site of primary lesion is consistent with available clinical data since the appearance of HI and insulin resistance are always present together and have not been temporally separated. They each cause the other to occur and each is defined by the other.
There are examples of situations where HI precedes insulin resistance, including studies showing that artificially increasing insulin in the circulation in man and rodents causes insulin resistance and weight gain (39,48,49). In addition, patients with insulinomas are often normoglycemic and insulin resistant, except when fasted (50). Furthermore, inhibition of insulin secretion in HI does not cause hyperglycemia and actually improves weight loss during dieting in obese humans (40,41,51). Further evidence that HI can precede and predict diabetes is derived from studies in which high plasma insulin concentrations predict T2DM in the Pima Indians (52).
It is also well established that gastric bypass surgery, dieting, and exercise all decrease HI although it is not known whether HI or insulin resistance changes first, except following surgery where the decrease in HI precedes the resolution of insulin resistance (53).
Possible mechanism
The initial three agents studied in our laboratory—monoglycerides, artificial sweeteners, and iron—were found to increase insulin secretion at basal glucose through the production of ROS such as superoxide, nitric oxide, and peroxides (10). We determined that ROS could directly stimulate insulin secretion (10,54). We also found that increasing the mitochondrial redox state (NADH/NAD) increased both ROS generation and insulin secretion (43) (Fig. 3).
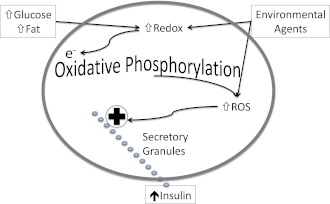
Figure 3.
Mechanism of ROS-induced hypersecretion. Redox increases mitochondrial NADH in response to stimulatory fuels. NADH donates electrons to the respiratory chain to make ATP via oxidative phosphorylation. An increase in ROS production accompanies increased flux through the respiratory chain that stimulates exocytosis of insulin containing secretory granules. Several environmental agents—monoglycerides and saccharin–also generate ROS either directly or through a change in redox, but without a stimulatory fuel. Increasing ROS by either mechanism stimulates insulin secretion.
ROS, NADH, and thiol redox systems in the mitochondria are integrated by the nicotinamide nucleotide transhydrogenase (NNT) that interconverts NADH and NADPH (55), which is a cofactor for reduction of oxidized glutathione and thioredoxin (Fig. 4).
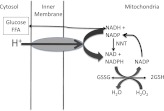
Figure 4.
Communication among cellular redox and ROS systems. NADH, generated from metabolism, can be used to regulate the NADPH/NADP and GSH/GSSG redox states via the NNT. The proton-translocating NNT reduces NADP by NADH and is an inner membrane protein utilizing the electrochemical proton gradient for NADPH generation. This enzyme has an important physiological role in the maintenance of reduced glutathione, essential for scavenging reactive oxygen. ROS stimulates scavenging activities using NADPH and increasing flux through NNT.
Furthermore, the redox state established by the thiol ratios, cysteine-to-cystine and reduced-to-oxidized glutathione (GSH/GSSG), correlates with aging, diabetes, heart disease, and some cancers and regulates intracellular signal transduction and mitochondrial ROS production (56,57). Since the intramitochondrial NADH-to-NAD ratio influences the NADP-to-NADPH ratio and thiol redox states in both mitochondria and cytosol (55), further testing is needed to determine the molecular mechanisms for ROS-mediated insulin secretion as well as the target proteins or transcription factors that are impacted by changes in ROS.
Consistent with an essential and sufficient role for ROS in HI, we also determined that scavenging ROS completely prevented both basal and stimulated insulin secretion (43,54). ROS production and removal are in dynamic balance and both are necessary for secretion. It is highly unlikely that ROS removal would be a viable therapy for HI: It would require a balance sufficient to prevent HI but allow glucose-induced insulin secretion. Environmental agents that have a direct effect within the β-cell can be tested for their effect on ROS production and insulin secretion. It is possible that this knowledge will lead to identification and elimination of causes of the obesity/diabetes epidemic.
Approaches to prevent hypersecretion of insulin
Inhibition of insulin secretion
In addition to essential studies to identify causes of excess ROS production, there are several interesting observations that suggest possible means of reversing HI without impairing fuel-induced insulin secretion. The limited experiments with humans treated with diazoxide to inhibit insulin secretion (41) suggest that it might be feasible to decrease HI without causing hyperglycemia as long as the liver is able to respond with decreased glucose production. This might not be achievable in patients with impaired liver function although it might be possible to inhibit nocturnal HI. Limited evidence supports the concept that blood glucose can be maintained at a wide variety of basal insulin values, but this needs to be rigorously evaluated in humans.
Inhibition of basal HI
Studies with rimonabant, a cannabinoid receptor antagonist, in Zucker rats have documented an interesting ability of this drug to decrease HI without inhibiting the fold increase induced by stimulatory glucose under certain conditions (58). Islets from lean littermates and obese Zucker and Zucker diabetic fatty rats were incubated for 24-h in vitro and exposed to 11 mmol/L glucose and 0.3 mmol/L palmitate (high fat, high carb) with or without rimonabant. Insulin secretion was determined at basal (3 mmol/L) or stimulatory (15 mmol/L) glucose concentrations. As expected, basal secretion was significantly elevated in islets from obese or high fat-, high carb–treated lean rats whereas the fold increase in glucose-stimulated insulin secretion (GSIS) was diminished. Rimonabant decreased basal hypersecretion in islets from obese rats and high fat-, high carb–treated lean rats without decreasing the fold increase in GSIS. However, it decreased GSIS in islets from lean rats without affecting basal secretion. These findings indicate that rimonabant had direct effects on islets to reduce insulin secretion when secretion was elevated above normal levels by diet or in obesity. The observation that it decreased stimulated secretion in islets from lean animals did not translate to any abnormalities in glucose handling in humans (59). The mechanism for this effect has not been established so it is not known whether rimonabant influences basal hypersecretion by decreasing ROS generation in islets or even if it acts via islet cannabinoid receptors. However, it is known that the main effect of rimonabant in humans is to decrease food intake through neural cannabinoid receptors (60). Unfortunately, the drug failed to receive U.S. Food and Drug Administration approval because of depressive side effects. However, it is conceivable that future drugs could be developed with similar mechanisms without central nervous system effects and with greater β-cell specificity.
The arguments presented here support the possibility of improving metabolic health by suppressing HI. A major and most important task is to identify any environmental agents, alone or in combination, that cause HI. It will then be necessary to determine whether blood levels of such compounds are sufficient to explain HI and whether their removal is able to reverse or prevent diabetes.
Conclusions
The problem of continuing increases in obesity and diabetes and their associated comorbidities and societal costs are sufficiently important to encourage serious endeavors to identify the causative agents.
There are alternative models and many environmental changes that have not been investigated but should be. It is our opinion that HI is a likely possible initiating event and determination of its cause and mechanism could identify a means to a cure. However, we cannot rule out other potential mechanisms involving regulation of ingestion or adipocyte mass that provide valid alternative explanations. Further, we cannot rule out simultaneous effects of past or future environmental agents on all of the candidate systems (61).
Acknowledgments
The research from the Corkey laboratory was supported by the following National Institutes of Health grants: DK35914, DK56690, DK74778, and DK46200 (to B.E.C.).
No potential conflicts of interest relevant to this article were reported.
Footnotes
See accompanying article, p. 2438.
References
- 1.Wang Y, Beydoun MA. The obesity epidemic in the United States—gender, age, socioeconomic, racial/ethnic, and geographic characteristics: a systematic review and meta-regression analysis. Epidemiol Rev 2007;29:6–28 [DOI] [PubMed] [Google Scholar]
- 2.Ogden CL, Carroll MD, Curtin LR, Lamb MM, Flegal KM. Prevalence of high body mass index in US children and adolescents, 2007-2008. JAMA 2010;303:242–249 [DOI] [PubMed] [Google Scholar]
- 3.Hu FB. Globalization of diabetes: the role of diet, lifestyle, and genes. Diabetes Care 2011;34:1249–1257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clark CM, Jr, Lee DA. Prevention and treatment of the complications of diabetes mellitus. N Engl J Med 1995;332:1210–1217 [DOI] [PubMed] [Google Scholar]
- 5.Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycaemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2012;55:1577–1596 [DOI] [PubMed] [Google Scholar]
- 6.Reaven GM. Insulin resistance: the link between obesity and cardiovascular disease. Med Clin North Am 2011;95:875–892 [DOI] [PubMed] [Google Scholar]
- 7.Sims EA, Danforth E, Jr, Horton ES, Glennon JA, Bray GA, Salans LB. Experimental obesity in man. A progress report. Isr J Med Sci 1972;8:813–814 [PubMed] [Google Scholar]
- 8.Mann T, Tomiyama AJ, Westling E, Lew AM, Samuels B, Chatman J. Medicare’s search for effective obesity treatments: diets are not the answer. Am Psychol 2007;62:220–233 [DOI] [PubMed] [Google Scholar]
- 9.Taubes G. Nutrition. The soft science of dietary fat. Science 2001;291:2536–2545 [DOI] [PubMed] [Google Scholar]
- 10.Corkey BE. Banting Lecture 2011: hyperinsulinemia: cause or consequence? Diabetes 2012;61:4–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Winter R. A consumer's dictionary of food additives: Descriptions in plain English of more than 12,000 ingredients both harmful and desirable found in foods 7th ed. New York, Random House Digital, Inc., 2009 [Google Scholar]
- 12.Woodruff TJ, Zota AR, Schwartz JM. Environmental chemicals in pregnant women in the United States: NHANES 2003-2004. Environ Health Perspect 2011;119:878–885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hectors TL, Vanparys C, van der Ven K, et al. Environmental pollutants and type 2 diabetes: a review of mechanisms that can disrupt beta cell function. Diabetologia 2011;54:1273–1290 [DOI] [PubMed] [Google Scholar]
- 14.Alonso-Magdalena P, Quesada I, Nadal A. Endocrine disruptors in the etiology of type 2 diabetes mellitus. Nat Rev Endocrinol 2011;7:346–353 [DOI] [PubMed] [Google Scholar]
- 15.Uribarri J, Woodruff S, Goodman S, et al. Advanced glycation end products in foods and a practical guide to their reduction in the diet. J Am Diet Assoc 2010;110:911–916.e12 [DOI] [PMC free article] [PubMed]
- 16.Gloyn AL, Braun M, Rorsman P. Type 2 diabetes susceptibility gene TCF7L2 and its role in beta-cell function. Diabetes 2009;58:800–802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yang W, Kelly T, He J. Genetic epidemiology of obesity. Epidemiol Rev 2007;29:49–61 [DOI] [PubMed] [Google Scholar]
- 18.Garland EJ, Remick RA, Zis AP. Weight gain with antidepressants and lithium. J Clin Psychopharmacol 1988;8:323–330 [PubMed] [Google Scholar]
- 19.Debons AF, Siclari E, Das KC, Fuhr B. Gold thioglucose-induced hypothalamic damage, hyperphagia, and obesity: dependence on the adrenal gland. Endocrinology 1982;110:2024–2029 [DOI] [PubMed] [Google Scholar]
- 20.Farooqi IS, O’Rahilly S. Leptin: a pivotal regulator of human energy homeostasis. Am J Clin Nutr 2009;89:980S–984S [DOI] [PubMed] [Google Scholar]
- 21.Nihalani N, Schwartz TL, Siddiqui UA, Megna JL. Weight gain, obesity, and psychotropic prescribing. J Obes 2011;2011:893629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kang JG, Park CY. Anti-obesity drugs: a review about their effects and safety. Diabetes Metab J 2012;36:13–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ioannides-Demos LL, Piccenna L, McNeil JJ. Pharmacotherapies for obesity: past, current, and future therapies. J Obes 2011;2011:179674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Suzuki K, Simpson KA, Minnion JS, Shillito JC, Bloom SR. The role of gut hormones and the hypothalamus in appetite regulation. Endocr J 2010;57:359–372 [DOI] [PubMed] [Google Scholar]
- 25.Pories WJ, Dohm GL. Diabetes: have we got it all wrong? Hyperinsulinism as the culprit: surgery provides the evidence. Diabetes Care 2012;35:2438–2442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zigman JM, Nakano Y, Coppari R, et al. Mice lacking ghrelin receptors resist the development of diet-induced obesity. J Clin Invest 2005;115:3564–3572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Phillips ML. Gut reaction: environmental effects on the human microbiota. Environ Health Perspect 2009;117:A198–A205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Boden G. Obesity, insulin resistance and free fatty acids. Curr Opin Endocrinol Diabetes Obes 2011;18:139–143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Klein S, Fontana L, Young VL, et al. Absence of an effect of liposuction on insulin action and risk factors for coronary heart disease. N Engl J Med 2004;350:2549–2557 [DOI] [PubMed] [Google Scholar]
- 30.Fisher-Wellman KH, Neufer PD. Linking mitochondrial bioenergetics to insulin resistance via redox biology. Trends Endocrinol Metab 2012;23:142–153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Newsholme P, Gaudel C, Krause M. Mitochondria and diabetes. An intriguing pathogenetic role. Adv Exp Med Biol 2012;942:235–247 [DOI] [PubMed] [Google Scholar]
- 32.Alcolado JC, Laji K, Gill-Randall R. Maternal transmission of diabetes. Diabet Med 2002;19:89–98 [DOI] [PubMed] [Google Scholar]
- 33.Wallace DC, Fan W, Procaccio V. Mitochondrial energetics and therapeutics. Annu Rev Pathol 2010;5:297–348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nolan CJ, Damm P, Prentki M. Type 2 diabetes across generations: from pathophysiology to prevention and management. Lancet 2011;378:169–181 [DOI] [PubMed] [Google Scholar]
- 35.Villeneuve LM, Reddy MA, Natarajan R. Epigenetics: deciphering its role in diabetes and its chronic complications. Clin Exp Pharmacol Physiol 2011;38:401–409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gavin JR, 3rd, Roth J, Neville DM, Jr, de Meyts P, Buell DN. Insulin-dependent regulation of insulin receptor concentrations: a direct demonstration in cell culture. Proc Natl Acad Sci USA 1974;71:84–88 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schwartz MW, Marks JL, Sipols AJ, et al. Central insulin administration reduces neuropeptide Y mRNA expression in the arcuate nucleus of food-deprived lean (Fa/Fa) but not obese (fa/fa) Zucker rats. Endocrinology 1991;128:2645–2647 [DOI] [PubMed] [Google Scholar]
- 38.Chavez M, Kaiyala K, Madden LJ, Schwartz MW, Woods SC. Intraventricular insulin and the level of maintained body weight in rats. Behav Neurosci 1995;109:528–531 [DOI] [PubMed] [Google Scholar]
- 39.Destefano MB, Stern JS, Castonguay TW. Effect of chronic insulin administration on food intake and body weight in rats. Physiol Behav 1991;50:801–806 [DOI] [PubMed] [Google Scholar]
- 40.Alemzadeh R, Jacobs W, Pitukcheewanont P. Antiobesity effect of diazoxide in obese Zucker rats. Metabolism 1996;45:334–341 [DOI] [PubMed] [Google Scholar]
- 41.Alemzadeh R, Langley G, Upchurch L, Smith P, Slonim AE. Beneficial effect of diazoxide in obese hyperinsulinemic adults. J Clin Endocrinol Metab 1998;83:1911–1915 [DOI] [PubMed] [Google Scholar]
- 42.Standridge M, Alemzadeh R, Zemel M, Koontz J, Moustaid-Moussa N. Diazoxide down-regulates leptin and lipid metabolizing enzymes in adipose tissue of Zucker rats. FASEB J 2000;14:455–460 [DOI] [PubMed] [Google Scholar]
- 43.Saadeh M, Ferrante TC, Kane A, Shirihai O, Corkey BE, Deeney JT. Reactive oxygen species stimulate insulin secretion in rat pancreatic islets: studies using mono-oleoyl-glycerol. PLoS ONE 2012;7:e30200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Alonso-Magdalena P, Ropero AB, Soriano S, et al. Bisphenol-A acts as a potent estrogen via non-classical estrogen triggered pathways. Mol Cell Endocrinol 2012;355:201–207 [DOI] [PubMed] [Google Scholar]
- 45.Persaud SJ, Al-Majed H, Raman A, Jones PM. Gymnema sylvestre stimulates insulin release in vitro by increased membrane permeability. J Endocrinol 1999;163:207–212 [DOI] [PubMed] [Google Scholar]
- 46.Hoa NK, Norberg A, Sillard R, et al. The possible mechanisms by which phanoside stimulates insulin secretion from rat islets. J Endocrinol 2007;192:389–394 [DOI] [PubMed] [Google Scholar]
- 47.Szopa TM, Ward T, Dronfield DM, Portwood ND, Taylor KW. Coxsackie B4 viruses with the potential to damage beta cells of the islets are present in clinical isolates. Diabetologia 1990;33:325–328 [DOI] [PubMed] [Google Scholar]
- 48.Larue-Achagiotis C, Goubern M, Laury MC. Concomitant food intake and adipose tissue responses under chronic insulin infusion in rats. Physiol Behav 1988;44:95–100 [DOI] [PubMed] [Google Scholar]
- 49.Del Prato S, Leonetti F, Simonson DC, Sheehan P, Matsuda M, DeFronzo RA. Effect of sustained physiologic hyperinsulinaemia and hyperglycaemia on insulin secretion and insulin sensitivity in man. Diabetologia 1994;37:1025–1035 [DOI] [PubMed] [Google Scholar]
- 50.Fajans SS, Vinik AI. Insulin-producing islet cell tumors. Endocrinol Metab Clin North Am 1989;18:45–74 [PubMed] [Google Scholar]
- 51.Greenwood RH, Mahler RF, Hales CN. Improvement in insulin secretion in diabetes after diazoxide. Lancet 1976;1:444–447 [DOI] [PubMed] [Google Scholar]
- 52.Weyer C, Hanson RL, Tataranni PA, Bogardus C, Pratley RE. A high fasting plasma insulin concentration predicts type 2 diabetes independent of insulin resistance: evidence for a pathogenic role of relative hyperinsulinemia. Diabetes 2000;49:2094–2101 [DOI] [PubMed] [Google Scholar]
- 53.Pories WJ, MacDonald KG, Jr, Morgan EJ, et al. Surgical treatment of obesity and its effect on diabetes: 10-y follow-up. Am J Clin Nutr 1992;55(Suppl.):582S–585S [DOI] [PubMed] [Google Scholar]
- 54.Pi J, Bai Y, Zhang Q, et al. Reactive oxygen species as a signal in glucose-stimulated insulin secretion. Diabetes 2007;56:1783–1791 [DOI] [PubMed] [Google Scholar]
- 55.Hoek JB, Rydström J. Physiological roles of nicotinamide nucleotide transhydrogenase. Biochem J 1988;254:1–10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jones DP. Redox potential of GSH/GSSG couple: assay and biological significance. Methods Enzymol 2002;348:93–112 [DOI] [PubMed] [Google Scholar]
- 57.Jones DP, Park Y, Gletsu-Miller N, et al. Dietary sulfur amino acid effects on fasting plasma cysteine/cystine redox potential in humans. Nutrition 2011;27:199–205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Getty-Kaushik L, Richard AM, Deeney JT, Krawczyk S, Shirihai O, Corkey BE. The CB1 antagonist rimonabant decreases insulin hypersecretion in rat pancreatic islets. Obesity (Silver Spring) 2009;17:1856–1860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Scheen AJ. The endocannabinoid system: a promising target for the management of type 2 diabetes. Curr Protein Pept Sci 2009;10:56–74 [DOI] [PubMed] [Google Scholar]
- 60.Fong TM, Heymsfield SB. Cannabinoid-1 receptor inverse agonists: current understanding of mechanism of action and unanswered questions. Int J Obes (Lond) 2009;33:947–955 [DOI] [PubMed] [Google Scholar]
- 61.Corkey BE, Shirihai O. Metabolic master regulators: sharing information among multiple systems. Trends Endocrinol Metab. 30 August 2012 [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]