Abstract
Nanoparticles of colloidal silver (AgNano) can influence gene expression. Concerning trials of AgNano application in poultry nutrition, it is useful to reveal whether they affect the expression of genes crucial for bird development. AgNano were administered to broiler chickens as a water solution in two concentrations (10 and 20 ppm). After dissection of the birds, breast muscles and hearts were collected. Gene expression of FGF2 and VEGFA on the mRNA and protein levels were evaluated using quantitative polymerase chain reaction and enzyme-linked immunosorbent assay methods. The results for gene expression in the breast muscle revealed changes on the mRNA level (FGF2 was up-regulated, P < 0.05) but not on the protein level. In the heart, 20 ppm of silver nanoparticles in drinking water increased the expression of VEGFA (P < 0.05), at the same time decreasing FGF2 expression both on the transcriptional and translational levels. Changes in the expression of these genes may lead to histological changes, but this needs to be proven using histological and immunohistochemical examination of tissues. In general, we showed that AgNano application in poultry feeding influences the expression of FGF2 and VEGFA genes on the mRNA and protein levels in growing chicken.
Keywords: Silver nanoparticles, VEGFA, FGF2, Muscle, Heart, Chicken
Background
Nanoparticles of colloidal silver (AgNano) are small enough to penetrate into the cell and subsequently into the nucleus. Direct interaction between them and DNA molecules or DNA-related proteins leads to alterations in gene expression profiles [1]. Clear evidence exists that AgNano can modify gene expression in animal models both in vivo and in vitro[2-5]. Taking into account the first trials of silver nanoparticle application in poultry production as an antimicrobial and nutritional agent [6], it is necessary to evaluate their effects on gene expression. From a nutritional point of view, it would be interesting to investigate genes with a crucial meaning for the muscle development in birds. We have focused on two genes, namely basic fibroblast growth factor 2 (FGF2) and vascular endothelial growth factor A (VEGFA).
FGF2 consists of low and high molecular weight protein isoforms, localised to different cellular compartments, indicating unique biological activity in vivo and in vitro[7,8]. It has mitogenic activity on a variety of mesodermal cells, including myoblasts [9]. FGF2 is implicated in early vertebrate embryogenesis [10] and tissue regeneration [11]. The immunolocalisation of FGF2 demonstrates that the spatial pattern of FGF2 localisation is highly specific. In the developing chick embryo, FGF2 has been localised in the myocardium, the somite myotome and limb bud muscles [12]. Experiments in pigs also suggest that FGF2 might be involved in skeletal muscle development during foetal and early postnatal life [13]. In the heart, FGF2 isoforms have distinct roles in many pathological conditions, including cardiac hypertrophy, ischemia-reperfusion injury and atherosclerosis [7].
VEGFA is essential for early development of the vasculature to the extent that inactivation of even a single allele of the VEGFA gene results in embryonic lethality [14,15]; furthermore, VEGFA is essential in regulating postnatal muscle capillarity. Cardiac and skeletal muscles of adult VEGFA-deficient animals exhibit a major intolerance to aerobic exercise [16].
Moreover, VEGFA positively influences dystrophic skeletal muscle repair, improving their vascularity and endogenous regeneration while lowering fibrosis; however, its uncontrolled expression causes unfavourable changes in muscle morphology [17,18]. VEGFA exerts action not only locally. It can mobilise endothelial progenitor cells from the bone marrow even to distant sites of neovascularisation [19].
Nutrition, in the case of poultry, can also comprise in ovo feeding that can stimulate intestinal development by enhancing villi development, thus increasing the intestinal capacity to digest and absorb nutrients, which provides a basis for muscle growth [20]. Moreover, both VEGFA and FGF2 injected into the vitelline vein of the chicken embryo stimulate myocardial vascularization, each of the proteins in a different way [21].
Few studies can be found on FGF2 and VEGFA expression, and their action in chicken embryos [12,21], but there is no information whether AgNano can affect expression of these genes in embryonic and growing chicken as well. The purpose of this study was to assess the influence of AgNano delivered in ovo and in vivo on the expression of the abovementioned genes in chicken and to indicate further research directions, which could reveal possible AgNano applications.
Methods
Experiments, conducted during the prenatal and postnatal growth periods of broiler chickens, were carried out in accordance with the Animal Experimentation Act in Denmark (Law No. 726, September 1993).
Nanoparticles
The hydrocolloid of AgNano (Nano-Tech, Warsaw, Poland) was produced by a patented non-explosive high-voltage method from high-purity metals (99.9999%) and high-purity demineralised water. The colloid contained 50 ppm of Ag nanoparticles, with a particle size ranging from 2 to 6 nm, based on transmission electron microscope (TEM) evaluation as described by Chwalibog et al. [22]. Experimental drinking water solutions used in the experiments were prepared by diluting the original concentration of the AgNano solution (50 ppm) in distilled water. The concentrations were selected based on effective dose recorded in weanling pigs (20 ppm of AgNano) [23] and in broilers (10 ppm; LP, unpublished data), and on economically relevant level.
In ovo treatments
Fertile eggs from Ross × Ross 308 breeder hens were obtained from a commercial hatchery and transported to the experimental farm of the University of Copenhagen. Upon arrival, the eggs were numbered, weighed and consequently treated according to the following descriptions: (1) non-injected control (n = 240), (2) AgNano injected (10 ppm of colloidal AgNano, n = 96) and (3) AgNano injected (20 ppm of colloidal AgNano, n = 96). The eggs were injected with 0.3 ml of the AgNano solutions (10 or 20 ppm) into the air sac at the blunt end of the albumen using sterile 1-ml tuberculin syringes with a 27-gauge needle. Before and after the injection, the shell was disinfected with ethanol. Afterwards, the hole was sealed with hypoallergenic tape and the eggs were placed in an incubator. The eggs were incubated for 21 days under standard conditions (temperature 37.8°C, 55% humidity, turned once per hour during the first 18 days, and at a temperature of 37°C and 60% humidity from day 19 until hatching).
In vivo treatments
For the post-hatching experiment, five groups of 48 chickens were formed. Three groups were created from the non-injected control group, and two additional groups were formed from the groups injected with 10 and 20 ppm of AgNano (Figure 1). Chicks for each group were randomly selected.
Figure 1.
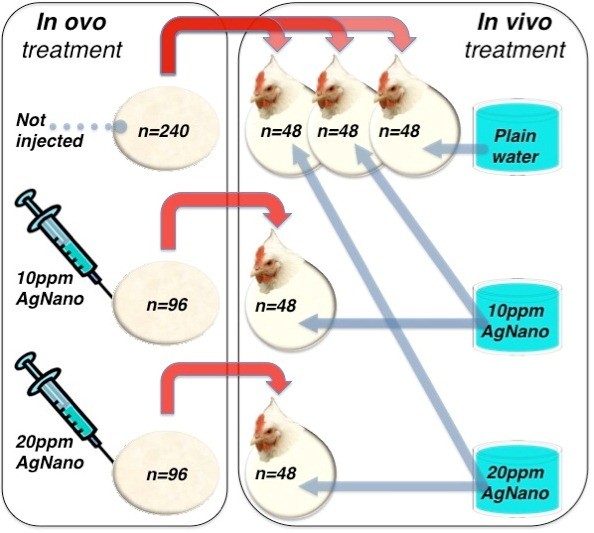
Experimental design. Red arrows indicate random selection of the birds after hatching, from the in ovo part (eggs not injected and injected with AgNano) to the in vivo part (broilers treated with AgNano drinking solution) of the experiment.
After hatching, for the first 6 days, the chicks were kept in pens furnished with a heating lamp. The temperature inside the brooding pen was 30°C to 33°C, and the lighting program was 23L:1D (L = light, D = darkness).
The 6-day transition period was applied to assure that the chicks get used to the environmental conditions before the actual experiment starts and to exclude a change of these conditions as a potential, additional factor influencing the results.
On day 7, 24 chicks from each group were randomly selected, weighed, leg banded and transferred to metabolic cages (0.5 m × 0.5 m × 0.5 m) with four birds per cage allocated into six replications. The room temperature from 7 to 35 days was 22°C with the following lighting program: days 7 to 13, 20L:4D; days 14 to 20, 21L:3D; days 21 to 27, 22L:2D and days 28 to 35, 23L:1D.
Throughout the experiment, the birds were fed ad libitum with a commercial diet. For the first 6 days after hatching, they obtained a commercial mixture containing 18.5% crude protein, 4.4% crude fat, 5.0% crude fibre, 5.8% ash, 10.3 g/kg lysine, 4.1 g/kg methionine and 3.4 g/kg cysteine. In the following 4 weeks, they were given another mixture containing 17.6% crude protein, 3.3% crude fat, 3.8% crude fibre, 5.2% ash, 8.7 g/kg lysine, 4.1 g/kg methionine and 3.2 g/kg cysteine. Both mixtures were produced by DLG (Axelborg, Copenhagen, Denmark). The birds had free access to water containing 0, 10 or 20 ppm of colloidal AgNano offered for 4 weeks after an initial 6-day-long transition period during the brooding period. The broilers' body weight, feed consumption and water intake were recorded at the beginning and end of each week starting at day 7 until 35 days of age.
Dissection procedure
After 4 weeks, the broilers were weighed and euthanized and the samples were collected. The hearts were weighed, and relative heart weight was calculated as the heart weight divided by the body weight, expressed as percentage.
Tissues were put into labelled cryo-tubes and immediately frozen in liquid nitrogen. Afterwards, all the tubes with frozen organs belonging to the same broiler were placed in one plastic bag labelled with the ID number of the bird and stored in the deep freezer (−80°C) until further analysis.
Gene expression on the mRNA level
Gene expression on the mRNA level was measured in breast muscle and heart samples using quantitative polymerase chain reaction method. The tissues (breast muscle and heart) were homogenised in TRIzol reagent (Invitrogen, Carlsbad, CA, USA) using TissueLyser II (Qiagen, Venlo, Netherlands), and total RNA was extracted according to the manufacturer's instructions. The RNA samples were purified using the SV Total RNA Isolation System (Promega, Fitchburg, WI, USA). The total RNA concentration was measured using a NanoDrop ND 1000 Spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). Two micrograms of total RNA were reverse transcribed using reverse transcriptase (Promega), oligo-dT and random primers (TAG Copenhagen A/S Symbion, Copenhagen, Denmark), after which real-time PCR was performed with cDNAs and gene-specific primer pairs (TAG Copenhagen A/S Symbion; Table 1) mixed with LightCycler®480 SYBR Green I Master (Roche Diagnostics, Basel, Switzerland) using a LightCycler®480 system (Roche Diagnostics). The samples were first denatured for 5 min at 95°C and then amplified using 45 cycles of 10 s at 95°C (denaturation), 10 s at 60°C to 62°C (annealing) and 9 s at 72°C (elongation) followed by quantitation. The reaction was performed in triplicate for each cDNA. For all analyses, relative quantification was applied, and actin beta (ACTB) and elongation factor 1 alpha 2 (EEF1A2) were used as the housekeeping genes.
Table 1.
Primer sequences for the investigated genes
| Gene symbol | Gene IDa | Size (bp) | Primer sequence (5’ to 3’) |
|---|---|---|---|
|
ACTB |
396526 |
ca. 169 |
forward: GTCCACCTTCCAGCAGATGT |
| reverse: ATAAAGCCATGCCAATCTCG | |||
|
EEF1A2 |
419244 |
ca. 85 |
forward: AGCAGACTTTGTGACCTTGCC |
| reverse: TGACATGAGACAGACGGTTGC | |||
|
FGF2 |
396413 |
ca. 151 |
forward: GGCACTGAAATGTGCAACAG |
| reverse: TCCAGGTCCAGTTTTTGGTC | |||
|
VEGFA |
395909 |
ca. 194 |
forward: TGAGGGCCTAGAATGTGTCC |
| reverse: TCTTTTGACCCTTCCCCTTT |
aNCBI resources.
Gene expression on the protein level
To estimate protein expression, enzyme-linked immunosorbent assay (ELISA) was applied. Samples from the control group receiving water and groups receiving 20 ppm of AgNano were chosen. Samples of heart and muscle tissues were homogenised in chilled RIPA buffer (Sigma-Aldrich, St. Louis, MO, USA) mixed with protease and phosphatase inhibitors (Protease Inhibitor Cocktail for use with mammalian cell and tissue extracts, DMSO solution, Sigma-Aldrich, and Phosphatase Inhibitor Cocktail 2, Sigma-Aldrich), using TissueLyser II (Qiagen). Then, samples were kept at 4°C for 1 h while slowly mixing. Afterwards, the samples were centrifuged at 4°C (30 min, 14,000 rpm). The supernatant was aliquoted into Eppendorf tubes to avoid frequent freeze/thaw cycles and frozen at −20°C. Each aliquot was used for determining the protein concentration (Total Protein Kit, MicroLowry, Peterson's Modification, Sigma-Aldrich) and for ELISA tests on the same day.
Kits for gallinaceous Fibroblast Growth Factor 2 and Vascular Endothelial Cell Growth Factor A (USCN Life Science, Wuhan, China) were used. Reagents and plates were prepared according to the manufacturer's instructions. The level of absorption was measured using an Infinite M200 microplate reader (TECAN, Crailsheim, Germany) at a wavelength of 450 nm. All samples were measured in duplicate.
Statistical analysis
Analysis of the data was carried out using t-tests and one-way analysis of variance (ANOVA) tests - generalized linear model procedure in SAS 9.2 (SAS Windows, 2002–2008, version 9.2, SAS Institute Inc., Cary, NC, USA). Values differing at P < 0.05 were considered significant.
Results
There were no significant differences in body weight, heart weight and relative heart weight between experimental groups (Table 2).
Table 2.
Body and heart weight of chickens treated with different levels of AgNano
| Weight |
Treatment |
ANOVA |
|||||
|---|---|---|---|---|---|---|---|
| Control | 10 ppm AgNano | 20 ppm AgNano | 10 ppm AgNanob | 20 ppm AgNanob | SEM | Pvalue | |
| Body (kg) |
1.578 |
1.684 |
1.725 |
1.436 |
1.545 |
0.0455 |
0.2853 |
| Heart (g) |
6.52 |
7.00 |
6.80 |
6.14 |
6.23 |
0.299 |
0.8937 |
| Hearta (relative, %) | 0.407 | 0.413 | 0.392 | 0.427 | 0.406 | 0.0124 | 0.9427 |
aValues were calculated as the heart weight divided by the body weight, expressed as percentage; beggs were in ovo injected; the level of significance is P < 0.05. AgNano, nanoparticles of colloidal silver; ANOVA, analysis of variance; SEM, standard error of the mean.
Gene expression
Breast muscle
The expression of FGF2, normalised to ACTB, was higher in all treatment groups than in the control group; however, differences between treatments were not significant. Regarding EEF1A2, significant differences were observed only between the control and the group which obtained the higher dose (20 ppm) of AgNano in the drinking water but had not been injected in ovo (Table 3).
Table 3.
Relative gene expression in the muscle tissue of chickens treated with different levels of AgNano
|
Gene symbol |
Treatment |
ANOVA |
|||||
|---|---|---|---|---|---|---|---|
| Control | 10 ppm AgNano | 20 ppm AgNano | 10 ppm AgNanoc | 20 ppm AgNanoc | SEM | P value | |
|
FGF2a |
0.67† |
1.28‡ |
1.44‡ |
1.05‡ |
1.23‡ |
0.070 |
0.0003 |
|
FGF2b |
0.60† |
0.98 |
1.38‡ |
0.95 |
0.93 |
0.068 |
0.0024 |
|
VEGFAa |
1.39 |
1.08† |
1.03† |
1.13 |
1.60‡ |
0.064 |
0.0057 |
| VEGFAb | 1.28 | 1.02† | 0.95† | 1.01 | 1.28‡ | 0.055 | 0.0072 |
aNormalised to ACTB; bnormalised to EEF1A2; ceggs were in ovo injected; †,‡different symbols indicate statistically significant differences (P < 0.05). AgNano, nanoparticles of colloidal silver; ANOVA, analysis of variance; SEM, standard error of the mean.
When VEGFA was examined, the level of expression in the breast muscle was higher in the group that had been injected in ovo and obtained 20 ppm of AgNano in the drinking water, compared to both non-injected groups receiving different solutions of AgNano in the drinking water. At the same time, the expression of this gene did not differ between the two injected groups and the untreated control group receiving water (Table 3).
Heart
Regarding FGF2 expression levels in the heart, when ACTB was used as the housekeeping gene, a significant difference was observed between the control and the group injected in ovo and treated with 20 ppm of AgNano in the drinking water. The expression of FGF2 normalised to EEF1A2 revealed no significant differences between the experimental groups (Table 4).
Table 4.
Relative gene expression in the heart of chickens treated with different levels of AgNano
|
Gene symbol |
Treatment |
ANOVA |
|||||
|---|---|---|---|---|---|---|---|
| Control | 10 ppm AgNano | 20 ppm AgNano | 10 ppm AgNanoc | 20 ppm AgNanoc | SEM | P value | |
|
FGF2a |
0.97† |
0.85 |
0.79 |
0.84 |
0.69‡ |
0.028 |
0.0443 |
|
FGF2b |
1.47 |
1.32 |
1.34 |
1.15 |
1.31 |
0.054 |
0.5839 |
|
VEGFAa |
0.59 |
0.68 |
0.78 |
0.73 |
0.61 |
0.026 |
0.0888 |
| VEGFAb | 0.75† | 0.93 | 1.12‡ | 0.84 | 1.14‡ | 0.044 | 0.0027 |
aNormalised to ACTB; bnormalised to EEF1A2; ceggs were in ovo injected; †,‡different symbols indicate statistically significant differences (P < 0.05). AgNano, nanoparticles of colloidal silver; ANOVA, analysis of variance; SEM, standard error of the mean.
In the case of VEGFA normalised to ACTB, no significant differences could be reported between the experimental groups. The high level of AgNano (20 ppm) given in the drinking water increased the expression of VEGFA normalised to EEF1A2 in both the injected and non-injected treatment groups (Table 4).
Protein expression
Concerning FGF2 and VEGFA on the protein level, no significant differences were found between treatments in the muscle tissue (Figure 2).
Figure 2.
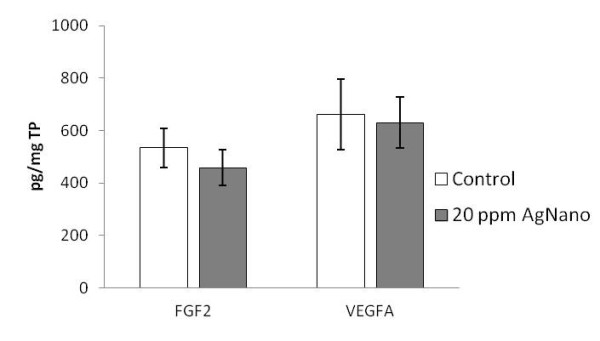
Protein expression in the breast muscle. The vertical axis shows the concentration of the target protein in picograms per milligram of total protein (TP). Error bars indicate standard deviation (SD). The level of significance is P < 0.05.
In the heart, the level of FGF2 protein after AgNano administration was lower, whereas the level of VEGFA protein was higher in the group treated with 20 ppm of AgNano, compared to the control group (Figure 3).
Figure 3.
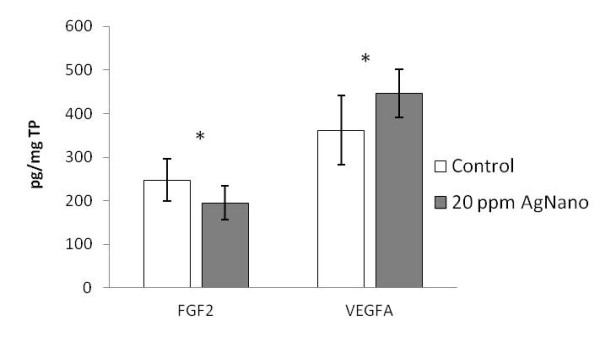
Protein expression in the heart. The vertical axis shows the concentration of the target protein in picograms per milligram of total protein (TP). Error bars indicate standard deviation (SD). Asterisks indicate statistically significant differences (P < 0.05).
Discussion
Due to the tissue specificity of the reference genes [24-27], the results for each examined target gene are not identical relative to the individual reference genes (especially in the heart). However, for both housekeeping genes, the relative levels of target gene expression presented almost the same pattern of changes.
Our results suggest that, in the case of chicken breast muscle tissue, oral AgNano administration influences FGF2 and VEGFA expression on the mRNA level, without leading to changes on the protein level. It could be suspected, at least in the case of FGF2, that such a difference might be caused by an unspecific isoform of this protein. Currently, only low molecular weight recombinant FGF2 protein is available on the market. However, the FGF2 antibody is not differentially specific against the two isoforms [7]. Assuming this, our results could mean that all the histological changes (like multipotent mesenchymal cell differentiation, satellite cell proliferation and angiogenesis) that have been noted in previous in vitro and in vivo studies [28-32] should not be expected in our case, or at least may not be caused directly by changes of FGF2 and VEGFA protein expression. This effect could be considered beneficial in terms of angiogenesis, which is a fundamental process affecting tumour growth and metastasis [19,33-37]. Nevertheless, this remains to be clarified by applying histological and immunohistochemical methods of examination of the tissue.
The opposite effect of AgNano administration was observed in the case of the cardiac muscle, where, without any impact on relative heart weight (Table 2), the expression of both genes was affected.
FGF2 expression on the mRNA level was down-regulated in the in ovo injected group which obtained 20 ppm of AgNano in the drinking water, compared to the control group. Similar results were noted regarding protein expression, where the FGF2 level was lower in the AgNano-treated group compared to the control group. The results obtained for FGF2 in our case are not in agreement with the findings of Bougioukas et al. [38], which suggested possible neovascularisation in the heart caused by this protein. However, according to another study, the expression of this gene remains unchanged during angiogenesis caused by bradycardia, suggesting that FGF2 does not play a direct role in this process [14]. On the other hand, some evidence exists that FGF2 (the high molecular weight isoform) can prevent endothelial cell migration and angiogenesis [39] and promote cardiac myocyte hypertrophy [40-42]. From this point of view, our findings may support possible targeted therapies with AgNano application.
It has been shown that VEGFA mRNA and protein are increased in the muscle during angiogenesis caused by repeated exercise [43-45]. Also, the mechanisms associated with bradycardia provide a signal for the enhancement of VEGFA expression, which is responsible for the myocardial angiogenesis [46]. We have demonstrated that AgNano at a concentration of 20 ppm administered to broiler chickens in the drinking water caused up-regulation of VEGFA expression in the heart on the mRNA and protein level. VEGFA modulates angiogenesis in dystrophic muscle [17,18] and in the heart [21,46]. To reveal whether AgNano might influence the heart angiogenesis in our experiment, resulting in the neovascularisation of the cardiac muscle, histological examination of the tissue should be performed.
Recent data indicate that regulation of VEGFA and FGF2 expression occurs on the transcriptional as well as translational levels, depending on the tissue and cell type [8,33-35,47,48]. Our results support these findings, showing that VEGFA and FGF2 expression can be regulated pre- and post-transcriptionally. Transcriptional and translational regulation of gene expression comprises the cascade of interactions between different biological molecules, including DNA, proteins, mRNAs and miRNAs [49-51]. Our results demonstrate that, in the case of FGF2 expression in the muscle, regulatory mechanisms prevent the increase of the level of the protein product of the gene.
Experimental data show that in ovo treatment can provide a basis for muscle growth and stimulate coronary development [20,21]. However, some of our unpublished data suggest that AgNano injected in ovo at early stages of the embryonic development seem not to evoke long-lasting influence on gene expression, which is prolonged after the hatching period. Therefore, our aim was rather to observe whether in ovo injection could change the effect of AgNano distributed with drinking water. Answering this question, we assumed that indeed, in ovo injection up-regulated both the heart expression of FGF2 as well as the muscle expression of VEGFA in broilers treated with drinking solution of 20 ppm of AgNano. However, the AgNano drinking solution itself (both concentrations) evoked up-regulation of FGF2 expression in the muscle, without enhancing the effect of in ovo injection.
Changes in the expression of investigated genes on the protein level may lead to harmful or beneficial histological changes. When examining silver residues in organs after AgNano administration, our unpublished results have shown minor Ag deposition in the breast muscle tissue and very slight deposition in the hearts of birds treated with a 20-ppm solution of colloidal silver, compared to the control group. However, these results could not inform us regarding the form of Ag deposited in the tissues. Supporting data from histological and TEM examinations of these tissues are needed to elucidate whether AgNano or rather silver ions exert a direct action.
Conclusions
AgNano application in poultry feeding can significantly influence the expression of FGF2 and VEGFA genes on the mRNA and protein levels in growing chicken. In ovo injection of AgNano at early embryonic stages of the development can enhance the effect of AgNano, distributed as a drinking solution, on FGF2 and VEGFA expression. On the protein level, AgNano influence FGF2 and VEGFA expression only in the heart, whereas in the muscle, the level of FGF2 and VEGFA protein remains unchanged. Supporting data from histological and TEM examinations of these tissues are needed to elucidate whether AgNano or rather silver ions exert a direct action on gene expression, and whether AgNano could evoke neovascularisation in the heart.
Abbreviations
ACTB: Actin beta; AgNano: Nanoparticles of colloidal silver; ANOVA: Analysis of variance; EEF1A2: Elongation factor 1 alpha 2; ELISA: Enzyme-linked immunosorbent assay; FGF2: Basic fibroblast growth factor 2; SEM: Standard error of the mean; TEM: Transmission electron microscope; TP: Total protein; VEGFA: Vascular endothelial growth factor A.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
AH carried out the molecular genetic studies and drafted the manuscript. ES conceived the study. LP participated in the design and supervised the experiment. FS participated in the statistical analysis. MG participated in the molecular studies and helped draft the manuscript. AC participated in the design and coordination and helped draft the manuscript. All authors read and approved the final manuscript.
Authors’ informations
AH is a PhD and postdoc at the University of Copenhagen (UC), ES is a PhD, DSc, professor and head of department at Warsaw University of Life Sciences (WULS), LP is a PhD and postdoc at UC, FS is a PhD student at UC, MG is a PhD and postdoc at WULS, and AC is a DSc, professor and head of division at UC.
Contributor Information
Anna Hotowy, Email: hotowy@life.ku.dk.
Ewa Sawosz, Email: ewa_sawosz@sggw.pl.
Lane Pineda, Email: lane@life.ku.dk.
Filip Sawosz, Email: sawosz@life.ku.dk.
Marta Grodzik, Email: margrodka@gmail.com.
André Chwalibog, Email: ac@life.ku.dk.
Acknowledgements
This work was supported by the Danish Agency for Science Technology and Innovation #2106-08-0025.
References
- Singh N, Manshian B, Jenkins GJ, Griffiths SM, Williams PM, Maffeis TG, Wright CJ, Doak SH. NanoGenotoxicology: the DNA damaging potential of engineered nanomaterials. Biomaterials. 2009;30:3891–3914. doi: 10.1016/j.biomaterials.2009.04.009. [DOI] [PubMed] [Google Scholar]
- Ahamed M, Karns M, Goodson M, Rowe J, Hussain SM, Schlager JJ, Hong Y. DNA damage response to different surface chemistry of silver nanoparticles in mammalian cells. ToxicolApplPharmacol. 2008;233:404–410. doi: 10.1016/j.taap.2008.09.015. [DOI] [PubMed] [Google Scholar]
- Sawosz E, Grodzik M, Lisowski P, Zwierzchowski L, Niemiec T, Zielińska M, Szmidt M, Chwalibog A. Influence of hydrocolloids of Ag, Au, and Ag/Cu alloy nanoparticles on the inflammatory state at transcriptional level. Bull Vet Inst Pulawy. 2010;54:81–85. [Google Scholar]
- Bhol KC, Schechter PJ. Topical nanocrystalline silver cream suppresses inflammatory cytokines and induces apoptosis of inflammatory cells in a murine model of allergic contact dermatitis. Br J Dermatol. 2005;152:1235–1242. doi: 10.1111/j.1365-2133.2005.06575.x. [DOI] [PubMed] [Google Scholar]
- Bhol KC, Schechter PJ. Effects of nanocrystalline silver (NPI 32101) in a rat model of ulcerative colitis. Digestive Dis Sci. 2007;52:2732–2742. doi: 10.1007/s10620-006-9738-4. [DOI] [PubMed] [Google Scholar]
- Pineda L, Sawosz E, Hotowy A, Elnif J, Sawosz F, Ali A, Chwalibog A. Effect of nanoparticles of silver and gold on metabolic rate and development of broiler and layer embryos. Comp Biochem Physiol Part A. 2012;161:315–319. doi: 10.1016/j.cbpa.2011.11.013. [DOI] [PubMed] [Google Scholar]
- Liao S, Bodmer J, Pietras D, Azhar M, Doetschman T, Schultz J. Biological functions of the low and high molecular weight protein isoforms of fibroblast growth factor-2 in cardiovascular development and disease. Dev Dyn. 2009;238(2):249–264. doi: 10.1002/dvdy.21677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chlebova K, Bryja V, Dvorak P, Kozubik A, Wilcox WR, Krejci P. High molecular weight FGF2: the biology of a nuclear growth factor. Cell Mol Life Sci. 2009;66:225–235. doi: 10.1007/s00018-008-8440-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clegg CH, Linkhart TA, Olwin BB, Hauschka SD. Growth factor control of skeletal muscle differentiation: commitment to terminal differentiation occurs in GI phase and is repressed by FGF. J Biol Chem. 1986;105:949–956. doi: 10.1083/jcb.105.2.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorey K, Amaya E. FGF signalling: diverse roles during early vertebrate embryogenesis. Development. 2010;137:3731–3742. doi: 10.1242/dev.037689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perl A-K, Gale E. FGF signalling is required for myofibroblast differentiation during alveolar regeneration. Am J Physiol Lung Cell Mol Physiol. 2009;297:L299–L308. doi: 10.1152/ajplung.00008.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph-Silverstein J, Consigli SA, Lyser KM, VerPault C. Basic fibroblast growth factor in the chick embryo: immuolocalization to striated muscle cells and their precursors. J Cell Biol. 1989;108:2459–2466. doi: 10.1083/jcb.108.6.2459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng M, Palin M-F, Veronneau S, LeBel D, Pelletier G. Ontogeny of epidermal growth factor (EGF), EGF receptor (EGFR) and basic fibroblast growth factor (bFGF) mRNA levels in pancreas, liver, kidney, and skeletal muscle of pig. Anim Endocrinol. 1997;14(5):286–294. doi: 10.1016/S0739-7240(97)00025-8. [DOI] [PubMed] [Google Scholar]
- Ferrara N. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am J Physiol Cell Physiol. 2001;280:C1358–C1366. doi: 10.1152/ajpcell.2001.280.6.C1358. [DOI] [PubMed] [Google Scholar]
- Karkkainen MJ, Haiko P, Sainio K, Partanen J, Taipale J, Petrova TV, Jeltsch M, Jackson DG, Talikka M, Rauvala H, Betsholtz C, Alitalo K. Vascular endothelial growth factor C is required for sprouting of the first lymphatic vessels from embryonic veins. Nat Immunol. 2004;5:74–80. doi: 10.1038/ni1013. [DOI] [PubMed] [Google Scholar]
- Olfert IM, Howlett RA, Tang K, Dalton ND, Gu Y, Peterson KL, Wagner PD, Breen EC. Muscle-specific VEGF deficiency greatly reduces exercise endurance in mice. J Physiol. 2009;587:1755–1767. doi: 10.1113/jphysiol.2008.164384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deasy BM, Feduska JM, Payne TR, Li Y, Ambrosio F, Huard J. Effect of VEGF on the regenerative capacity of muscle stem cells in dystrophic skeletal muscle. Mol Ther. 2009;17:1788–1798. doi: 10.1038/mt.2009.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karvinen H, Pasanen E, Rissanen TT, Korpisalo P, Vähäkangas E, Jazwa A, Giacca M, Ylä-Herttuala S. Long-term VEGF-A expression promotes aberrant angiogenesis and fibrosis in skeletal muscle. Gene Ther. 2011;18:1166–1172. doi: 10.1038/gt.2011.66. [DOI] [PubMed] [Google Scholar]
- Hicklin DJ, Ellis LM. Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol. 2005;23:1011–1027. doi: 10.1200/JCO.2005.06.081. [DOI] [PubMed] [Google Scholar]
- Noy Y, Uni Z. Early nutritional strategies. World Poultry Sci J. 2010;66:639–646. doi: 10.1017/S0043933910000620. [DOI] [Google Scholar]
- Tomanek RJ, Lotun K, Clark EB, Suvarna PR, Hu N. VEGF and bFGF stimulate myocardial vascularization in embryonic chick. Am J Physiol. 1998;274:H1620–H1626. doi: 10.1152/ajpheart.1998.274.5.H1620. Heart Circ Physiol 43. [DOI] [PubMed] [Google Scholar]
- Chwalibog A, Sawosz E, Hotowy A, Szeliga J, Mitura S, Mitura K, Grodzik M, Orlowski P, Sokolowska A. Visualization of interaction between inorganic nanoparticles and bacteria or fungi. Int J Nanomedicine. 2010;5:1085–1094. doi: 10.2147/IJN.S13532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fondevila M, Herrer R, Casallas MC, Abecia L, Ducha JJ. Silver nanoparticles as a potential antimicrobial additive for weaned pigs. Anim Feed Sci Tech. 2009;150:259–269. doi: 10.1016/j.anifeedsci.2008.09.003. [DOI] [Google Scholar]
- Barber RD, Harmer DW, Coleman RA, Clark BJ. GAPDH as a housekeeping gene: analysis of GAPDH mRNA expression in a panel of 72 human tissues. Physiol Genomics. 2005;21:389–395. doi: 10.1152/physiolgenomics.00025.2005. [DOI] [PubMed] [Google Scholar]
- Glare EM, Divjak M, Bailey MJ, Walters EH. b-Actin and GAPDH housekeeping gene expression in asthmatic airways is variable and not suitable for normalising mRNA levels. Thorax. 2002;57:765–770. doi: 10.1136/thorax.57.9.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Boever S, Vangestel C, De Backer P, Croubels S, Sys SU. Identification and validation of housekeeping genes as internal control for gene expression in an intravenous LPS inflammation model in chickens. Veterin Immunol Immunopathol. 2008;122:312–317. doi: 10.1016/j.vetimm.2007.12.002. [DOI] [PubMed] [Google Scholar]
- Mahoney DJ, Carey K, Fu MH, Snow R, Cameron-Smith D, Parise G, Tarnopolsky MA. Real-time RT-PCR analysis of housekeeping genes in human skeletal muscle following acute exercise. Physiol Genomics. 2004;18:226–231. doi: 10.1152/physiolgenomics.00067.2004. [DOI] [PubMed] [Google Scholar]
- Yablonka-Reuveni Z, Anderson JE. Satellite cells from dystrophic (mdx) mice display accelerated differentiation in primary cultures and in isolated myofibers. Dev Dyn. 2006;235:203–212. doi: 10.1002/dvdy.20602. [DOI] [PubMed] [Google Scholar]
- Maddalozzo J, Hughes CA, Huang L, Mu Y, Ludemann J, Crawford S. High angiogenic activity in cells isolated from cystic hygroma. Role of bFGF. Arch Otolaryngol Head Neck Surg. 1999;125:45–48. doi: 10.1001/archotol.125.1.45. [DOI] [PubMed] [Google Scholar]
- Grothe C, Brand-Saberi B, Wilting J, Christ B. Fibroblast growth factor receptor 1 in skeletal and heart muscle cells: expression during early avian development and regulation after notochord transplantation. Dev Dyn. 1996;206:310–317. doi: 10.1002/(SICI)1097-0177(199607)206:3<310::AID-AJA8>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- Efthimiadou A, Asimakopoulos B, Nikolettos N, Giatromanolaki A, Sivridis E, Papachristou DN, Kontoleon E. Angiogenic effect of intramuscular administration of basic and acidic fibroblast growth factor on skeletal muscles and influence of exercise on muscle angiogenesis. Br J Sports Med. 2006;40:35–39. doi: 10.1136/bjsm.2005.018754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryan BA, Walshe TE, Mitchell DC, Havumaki JS, Saint-Geniez M, Maharaj AS, Maldonado AE, D'Amore PA. Coordinated vascular endothelial growth factor expression and signalling during skeletal myogenic differentiation. Mol Biol Cell. 2008;19:994–1006. doi: 10.1091/mbc.E07-09-0856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenzon E, Colladel R, Andreuzzi E, Marastoni S, Todaro F, Schiappacassi M, Ligresti G, Colombatti A, Mongia M. MULTIMERIN2 impairs tumor angiogenesis and growth by interfering with VEGF-A/VEGFR2 pathway. Oncogene. 2012;31:3136–3147. doi: 10.1038/onc.2011.487. [DOI] [PubMed] [Google Scholar]
- Kaneda H, Arao T, Matsumoto K, De Velasco MA, Tamura D, Aomatsu K, Kudo K, Sakai K, Nagai T, Fujita Y, Tanaka K, Yanagihara K, Yamada Y, Okamoto I, Nakagawa K, Nishio K. Activin A inhibits vascular endothelial cell growth and suppresses tumour angiogenesis in gastric cancer. Br J Cancer. 2011;105:1210–1217. doi: 10.1038/bjc.2011.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantelmo AR, Cammarota R, Noonan DM, Focaccetti C, Comoglio PM, Prat M, Albini A. Cell delivery of Met docking site peptides inhibit angiogenesis and vascular tumor growth. Oncogene. 2010;29:5286–5298. doi: 10.1038/onc.2010.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grodzik M, Sawosz E, Wierzbicki M, Orlowski P, Hotowy A, Niemiec T, Szmidt M, Mitura K, Chwalibog A. Nanoparticles of carbon allotropes inhibit glioblastoma multiforme angiogenesis in ovo. Int J Nanomed. 2011;6:3041–3048. doi: 10.2147/IJN.S25528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan A, Yu CJ, Chen WJ, Lin FY, Kuo SH, Luh KT, Yang PC. Correlation of total VEGF mRNA and protein expression with histologic type, tumor angiogenesis, patient survival and timing of relapse in non-small-cell lung cancer. Int J Cancer (Pred Oncol) 2000;89:475–483. doi: 10.1002/1097-0215(20001120)89:6<475::AID-IJC2>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- Bougioukas I, Didilis V, Ypsilantis P, Giatromanolaki A, Sivridis E, Lialiaris T, Mikroulis D, Simopoulos C, Bougioukas G. Intramyocardial injection of low-dose basic fibroblast growth factor or vascular endothelial growth factor induces angiogenesis in the infarcted rabbit myocardium. Cardiovasc Pathol. 2007;16:63–68. doi: 10.1016/j.carpath.2006.08.006. [DOI] [PubMed] [Google Scholar]
- Calabrò P, Limongelli G, Riegler L, Maddaloni V, Palmieri R, Golia E, Roselli T, Masarone D, Pacileo G, Golino P, Calabrò R. Novel insights into the role of cardiotrophin-1 in cardiovascular diseases. J Mol Cell Cardiol. 2009;46:142–148. doi: 10.1016/j.yjmcc.2008.11.002. [DOI] [PubMed] [Google Scholar]
- Jiang ZS, Jeyaraman M, Wen GB, Fandrich RR, Dixon IM, Cattini PA, Kardami E. High- but not low-molecular weight FGF-2 causes cardiac hypertrophy in vivo; possible involvement of cardiotrophin-1. J Mol Cell Cardiol. 2007;42:222–233. doi: 10.1016/j.yjmcc.2006.09.002. [DOI] [PubMed] [Google Scholar]
- Frey N, Katus HA, Olson EN, Hill JA. Hypertrophy of the heart: a new therapeutic target? Circulation. 2004;109:1580–1589. doi: 10.1161/01.CIR.0000120390.68287.BB. [DOI] [PubMed] [Google Scholar]
- Santiago JJ, Ma X, McNaughton LJ, Nickel BE, Bestvater BP, Yu L, Fandrich RR, Netticadan T, Kardami E. Preferential accumulation and export of high molecular weight FGF-2 by rat cardiac non-myocytes. Cardiovasc Res. 2011;89:139–147. doi: 10.1093/cvr/cvq261. [DOI] [PubMed] [Google Scholar]
- Wagner PD. Skeletal muscle angiogenesis. A possible role for hypoxia. Adv Exp Med Biol. 2001;502:21–38. [PubMed] [Google Scholar]
- Gustafsson T, Puntschart A, Kaijser L, Jansson E, Sundberg CJ. Exercise-induced expression of angiogenesis-related transcription and growth factors in human skeletal muscle. Am J Physiol. 1999;276:H679–H685. doi: 10.1152/ajpheart.1999.276.2.H679. [DOI] [PubMed] [Google Scholar]
- Gustafsson T, Knutsson A, Puntschart A, Kaijser L, Nordqvist AC, Sundberg CJ, Jansson E. Increased expression of vascular endothelial growth factor in human skeletal muscle in response to short-term one-legged exercise training. Pflugers Arch. 2002;444:752–759. doi: 10.1007/s00424-002-0845-6. [DOI] [PubMed] [Google Scholar]
- Zheng W, Brown MD, Brock TA, Bjercke RJ, Tomanek RJ. Bradycardia-induced coronary angiogenesis is dependent on vascular endothelial growth factor. Circ Res. 1999;85:192–198. doi: 10.1161/01.RES.85.2.192. [DOI] [PubMed] [Google Scholar]
- Manabe I, Shindo T, Nagai R. Gene expression in fibroblasts and fibrosis: involvement in cardiac hypertrophy. Circ Res. 2002;91:1103–1113. doi: 10.1161/01.RES.0000046452.67724.B8. [DOI] [PubMed] [Google Scholar]
- Feliers D, Duraisamy S, Barnes JL, Ghosh-Choudhury G, Kasinath BS. Translational regulation of vascular endothelial growth factor expression in renal epithelial cells by angiotensin II. Am J Physiol Renal Physiol. 2005;288:F521–F529. doi: 10.1152/ajprenal.00271.2004. [DOI] [PubMed] [Google Scholar]
- Lanner F, Rossant J. The role of FGF/Erk signaling in pluripotent cells. Development. 2010;137:3351–3360. doi: 10.1242/dev.050146. [DOI] [PubMed] [Google Scholar]
- Nicoli S, Standley C, Walker P, Hurlstone A, Kevin E, Fogarty KE, Nathan D, Lawson ND. Micro RNA-mediated integration of haemodynamics and VEGF signalling during angiogenesis. Nature. 2010;464(7292):1196–1200. doi: 10.1038/nature08889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cogburn LA, Porter TE, Duclos MJ, Simon J, Burgess SC, Zhu JJ, Cheng HH, Dodgson JB, Burnside J. Functional genomics of the chicken - a model organism. Poultry Sci. 2007;86:2059–2094. doi: 10.1093/ps/86.10.2059. [DOI] [PubMed] [Google Scholar]


