Abstract
Connective tissue growth factor (CTGF/CCN2) is a cysteine-rich matricellular secreted protein that regulates diverse cell functions including adhesion, migration, proliferation, differentiation, survival, senescence and apoptosis. In the pancreas, CTGF/CCN2 regulates critical functions including β cell replication during embryogenesis, stimulation of fibrogenic pathways in pancreatic stellate cells during pancreatitis, and regulation of the epithelial and stromal components in pancreatic ductal adenocarcinoma. This article reviews the evidence establishing CTGF/CCN2 as an important player in pancreatic physiology and pathology, highlighting the specific cell types that are involved in each process and the importance of CTGF/CCN2 as a component of autocrine or paracrine signaling within or between these various cells. Translational applications, including the potential for CTGF/CCN2-based therapies in diabetes, fibrosis, or cancer are discussed.
Keywords: Connective tissue growth factor, pancreatic stellate cell, islets, β cell, acinar cell, desmoplasia, fibrosis, pancreatic ductal adenocarcinoma, matricellular, CCN, CCN2, CTGF, TGF-β
1. Introduction
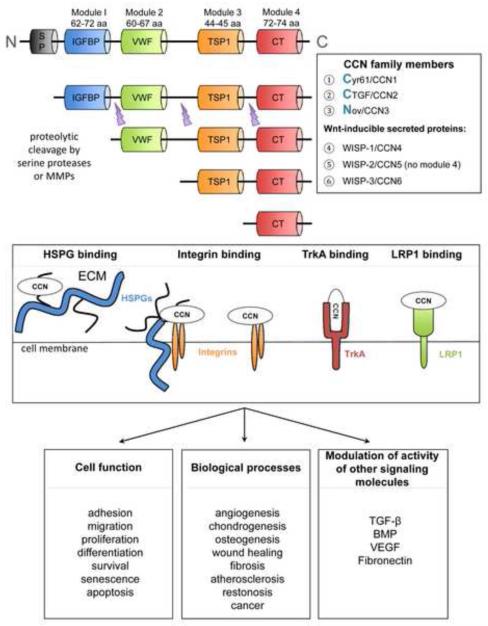
Connective tissue growth factor (CTGF/CCN2) is a prototypical member of the “CCN” family whose members were first recognized in the mid-1990’s and identified based on their synthesis in response to mitogenic signals (serum, growth factors, oncogenes) or their aberrant expression in transformed cells [1]. “CCN” is an acronym adopted by consensus among those working in the field [2] that refers to the first three such members to be described, specifically Cysteine-rich 61 (cyr-61 or CCN1), CTGF (or CCN2), and Nephroblastoma overexpressed (nov or CCN3). The other three family members were identified based on their induction by Wnts (regulators of cell proliferation and differentiation that bind Frizzled receptors and low density lipoprotein receptor-related protein (LRP) 5/6 co-receptors) and are termed Wnt-inducible secreted proteins (WISP-1 or CCN4; WISP-2 or CCN5; and WISP-3 or CCN6). All six proteins are highly evolutionarily conserved and share substantial homology with each other. Structurally, the proteins are organized in a very complex manner and comprise four individual cysteine-rich modules (Figure 1), each of which is encoded by a separate exon that was likely shuffled from other genes [3]. Module 1 contains an insulin-like growth factor binding domain, module 2 contains a von Willebrand factor type C, module 3 contains a thrombospondin type 1 repeat, and module 4 contains a cysteine knot motif. The inter-modular regions are susceptible to proteolytic cleavage by serine proteases or matrix metalloproteases, a process that can result in the liberation of CCN fragments which comprise at least one intact module and which are biologically active [1, 4] (Figure 1).
Fig. 1. Structural and functional properties of CCN proteins.
Primary CCN translational products comprise a signal peptide and four modules that can be differentially liberated by proteolysis. Through their contextual interactions with cell surface receptors, co-receptors, and signaling molecules, CCN proteins regulate multiple cellular functions and biological processes.
Although the primary translational products of CCN proteins contain N-terminal signal peptides and some of the proteins have been detected in cell culture conditioned medium or body fluids [1, 4], the notion of CCN members as simply being secreted proteins is over-simplistic. In fact a very complex picture has emerged regarding the manner in which cell function is regulated by CCN proteins and this derives from the key observations that they (i) are heparin-binding and sequestered with heparan sulfate proteoglycans (HSPG) on cell surfaces and in extracellular matrix (ECM); (ii) interact with a diverse variety of cell surface integrins in a cell-, module-, HSPG-, and/or context-specific manner to regulate a broad range of cellular responses; and (iii) modulate the activity of other bioactive molecules (e.g. transforming growth factor-beta (TGF-β), bone morphogenic protein, vascular endothelial growth factor). Thus CCN proteins are now recognized as matricellular proteins which function as integral but non-structural components of the ECM that modulate cellular responses to other molecular cues in the pericellular environment by interacting with integrins, HSPG or other receptors on the cell surface such as LRP or the TrkA neurotrophin receptor [4-10] (Figure 1).
CCN proteins regulate many aspects of cell function including adhesion, migration, proliferation, differentiation, survival, senescence and apoptosis [4-7]. While the “opposite” nature of some of these responses may appear paradoxical, these outcomes reflect the fact that matricellular proteins function contextually as a function of other molecular cues in the surrounding environment. That being said, CCN proteins have emerged as critical regulators of important biological processes including angiogenesis, chondrogenesis, osteogenesis, wound healing, fibrosis, atherosclerosis, restonisis, and cancer [4-6] (Figure 1). An essential role during embryonic development has been shown by targeted gene deletions in mice for (i) CYR61/CCN1 which show numerous lethal angiogenic and vascular deficiencies [11, 12]; (ii) CTGF/CCN2 which exhibit widespread chondrodysplasia, defective endochondral ossification, reduced growth plate angiogenesis and pulmonary hypoplasia [13, 14], as well as deficiencies in endocrine cell lineage allocation, islet morphogenesis, and embryonic β cell proliferation in the pancreas [15] (see below); such mice die shortly after birth because the skeletal malformations lead to compromised lung function; and (iii) NOV/CCN3 which exhibit defects in the axial and appendicular skeleton, malformation of joints, cardiac septal defects and early onset of cataracts due to lens degeneration [16]. A loss of function mutation causing WISP-3/CCN6 deficiency in humans results in progressive pseudorheumatoid dysplasia, an autosomal recessive disease of the joint in juveniles [17]. While these studies of “global” deficiency have emphasized the importance of individual CCN proteins in cardiovascular and skeletal development in particular, more refined studies have begun to emerge that have allowed conditional knock-outs and/or cell-specific gene deletion or over-expression to be developed to more precisely establish the function of CTGF/CCN2 in other organ systems and/or at later stages of life. Such is the case for recent studies that have determined the role of CTGF/CCN2 in specific cell types during pancreatic development [18], as will be discussed below.
2. Cell biology of the pancreas
The pancreas has both endocrine and exocrine functions. The endocrine component is organized as the islets of Langerhans which comprise 1-2% of the pancreatic mass and contain several cell types that secrete distinct products: glucagon is secreted by α cells (15-20%), insulin and amylin by β cells (65-80%), somatostatin by δ cells (3-10%), pancreatic polypeptide by PP cells (3-5%), and ghrelin by ε cells (< 1%) [19]. The endocrine cells are in intimate proximity to the vasculature either by direct apposition or extension of cytoplasmic processes to the endothelial cells that form an extensive network of blood capillaries, allowing for efficient uptake of secreted proteins into the circulation. The exocrine component is comprised of acinar cells which are organized into clusters (acini) that are responsible for the synthesis, storage and secretion of inactive proforms of digestive enzymes into the lumen of each acinus from where, after activation, they empty into the duodenum by a series of interconnecting epithelial ducts. Other cells include pancreatic stellate cells (PSC) which are located in peri-acinar and periductular locations, contain vitamin A in lipid droplets in their cytoplasm, and stain for selective markers such as desmin and glial fibrillary acidic protein [20]. PSC are normally quiescent but play a critical role in the wound healing response by undergoing a process of transient activation after acute injury whereby they become highly motile, proliferative, myofibroblastic, and produce fibrillar collagens which are deposited at the site of damage to form a provisional ECM that is a critical scaffold for cell repopulation and tissue repair. However, in a setting of chronic injury, PSC activation persists unabated and this phenomenon contributes significantly to pancreatic pathogenesis because fibrillar collagens are unrelentingly deposited in the interstitial spaces, resulting in extensive fibrosis and tissue dysfunction [21, 22]. Many aspects of PSC activation are believed to be recapitulated in vitro when PSC that have been isolated from normal pancreatic tissue are cultured on tissue culture plastic for 1-2 weeks, a procedure that results in their progressive autonomous differentiation into myofibroblast-like cells that express increasing levels of αSMA, laminin, fibronectin, and collagen types I and III. These culture-induced changes have become an important in vitro model for probing the molecular mechanisms of PSC activation and fibrogenic pathways [20, 23-26].
Of the CCN proteins studied to date, a wealth of information has been gathered with respect to the role of CTGF/CCN2 in regulating the functions of many cell types in the pancreas. Moreover, data have accrued which support an essential role for CTGF/CCN2 in distinct aspects of pancreatic biology including embryonic development, would healing, inflammation, fibrosis, and cancer. In this context, the pancreas is one of a few organs in which a detailed picture has begun to emerge regarding the contribution of CTGF/CCN2 to regulating normal and pathological processes and it is illustrative of the many diverse biological roles of CTGF/CCN2 within a single organ system.
3. CTGF/CCN2 in pancreatic development
Pancreatic development is initiated by branching morphogenesis of dorsal and ventral evaginations from the posterior foregut endoderm which later fuse to become a single organ. Endocrine progenitor cells arise via notch/delta signaling in ductal epithelial cells, resulting in their delamination from the epithelium and differentiation into proliferative hormone-producing cells organized as islets [27, 28]. This developmental process, which is initiated on embryonic Day 9.5 (E9.5) in the mouse, is tightly orchestrated by a complex network of transcription factors [28, 29], including neurogenin 3 which plays a central role in specifying the endocrine component [30] (Figure 2). Since there is a deficiency in functional β cell mass in type 1 or type 2 diabetes [31, 32], considerable attention has become focused on understanding the mechanisms by which β cell specification is developmentally determined in the hope that key regulators of this process may be identified that prove to have therapeutic value in the diabetic patient [28], and it is this aspect which has been the driving force behind many of the CTGF/CCN2 studies performed to date. Diabetes continues to be a major medical challenge: in 2011, 8% of the US population was diagnosed with type 2 diabetes and the numbers of patients in all age groups are rising in the face of an obesity epidemic fuelled by lack of physical activity and poor diet.
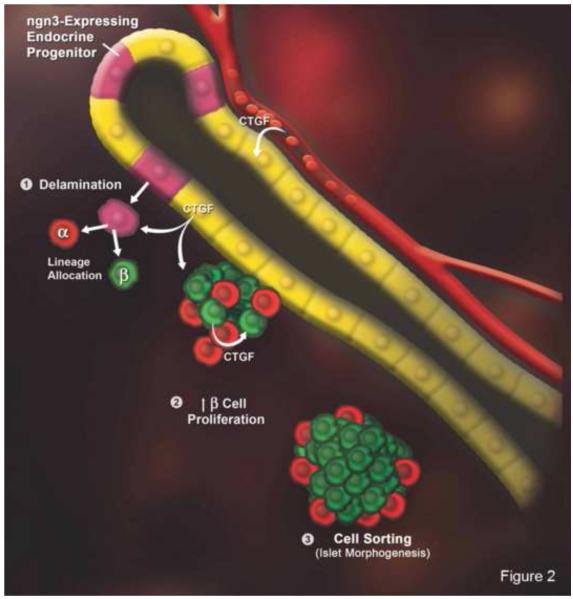
Fig. 2. CTGF/CCN2 in islet development.
Neurogenin 3-positive (ngn3) endocrine progenitors within ductal epithelium delaminate and undergo differentiation into α, β, δ, ε or PP cells in response to signals from adjacent vascular endothelium (1). At late gestation, β-cell proliferation increases (2), islets separate from ducts, and endocrine cells undergo sorting (3). CTGF/CCN2 is produced by ductal epithelium, endothelial cells, and embryonic β-cells. Global CTGF/CCN2 inactivation increase progenitor allocation to the β-cell lineage and decreased β-cell proliferation, resulting in decreased β-cell mass. Also, islet sorting is impaired resulting in a “mixed islet” phenotype. Reproduced by permission from Crawford et al. [15], Copyright 2009, The Endocrine Society.
A role for CTGF/CCN2 in pancreatic development and morphogenesis was first suggested by its detection in the mouse pancreas at E14.5 [33] and its reduced expression in dysmorphic islets of transgenic mice that over-expressed the cut-homeodomain transcription factor hepatic nuclear factor 6 in the pancreatic endocrine cell lineage [34]. Further support came from studies of the CTGF/CCN2 gene which revealed the presence of an enhancer sequence which contains binding sites for multiple transcription factors involved in pancreatic development, including neurogenin 3 which represses expression of CTGF/CCN2 (and WISP-1/CCN4) in pancreatic progenitor cells [35]. Detailed localization studies, including careful CTGF/CCN2 immunohistochemistry of normal mouse specimens and X-gal staining for CTGF/CCN2-β-glactosidase fusion protein in mice containing a lacZ-tagged CTGF/CCN2 null allele, revealed that CTGF/CCN2 expression was correlated with the timing of islet morphogenesis, being produced transiently during late embryogenesis (E18.5) and up to Day 1 of post-natal life (P1) in β cells, with no production in islet cells during adult life nor by α cells at any stage [15]. Demonstration of a functional role for CTGF/CCN2 in the endocrine pancreas was revealed in pan-CTGF/CCN2 null embryos which, as compared to wild-type mice, exhibited less mature islet-associated ductal tissue and contained a higher α:β cell ratio at E13.5-18.5, the latter of which was attributed to decreased β cell proliferation downstream of enhanced levels of the cell cycle inhibitor, p27 [15] (Figure 2). While CTGF/CCN2 homozygous null mice do not survive after birth [14], CTGF/CCN2 heterozygous mice are viable and as adults exhibit pancreatic defects that include irregularly shaped islets and islets that contain proportionately more α cells than their wild type counterparts [15]. However, unlike the islets of CTGF-deficient embryos at E18.5 which contain fewer β cells than wild type mice (see above), those of adults undergo compensatory hypertrophy resulting in a β cell area which is comparable in size to those of wild types [15]. Although these animals did demonstrate a ~20% decrease in insulin production, they were functionally uncompromised as shown by their normal blood glucose levels and glucose clearance when challenged with a 12 week high-fat diet [15].
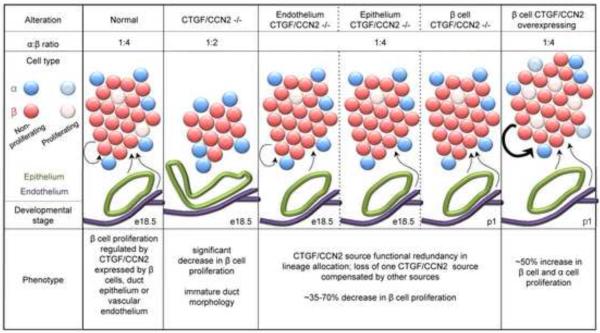
Whereas the above studies showed that CTGF/CCN2 was required for islet morphogenesis and β cell proliferation, the cellular source of CTGF/CCN2 driving this response could not be definitely ascertained in this approach because, in addition to its production by islets during late embryogenesis (see above), CTGF/CCN2 was found to be produced, albeit at declining levels, by pancreatic mesenchyme at E12.5 [15] and, further, to be expressed at high persistent levels in pancreatic ductular and vascular structures throughout embryonic, neonatal and adult life [15], with its detection apparent in pancreatic bud epithelium as early as E10.5 [18]. However, cell-specific inducible mouse knockout models developed to begin to address this question have elegantly shown that β cells, endothelial cells or epithelial cells are each biologically significant sources of CTGF/CCN2 that individually drive β cell proliferation during embryogenesis and function redundantly with one another in their CTGF-dependent promotion of lineage allocation and islet morphogenesis [18] (Figure 3). Overexpression of CTGF/CCN2 in mouse embryonic β cells, achieved by activation of a CTGF/CCN2 transgene in β cells at E9.5, resulted in islets that were of greater mass and which contained more α and β cells at P1 than their wild type counterparts, even though overall pancreatic size or vascularity was not changed (Figure 3). This effect was shown to arise through the stimulation of α or β cell proliferation by CTGF/CCN2 rather than driving an increase in endocrine progenitors [18]. In contrast, induction of the CTGF/CCN2 transgene in β cells for 1 week in mice that were 3 weeks old (weanling) or for 1-5 weeks in mice that were 7 weeks old (adult) failed to stimulate β cell proliferation, islet mass, or glucose homeostasis [36].
Fig. 3. Models of pancreatic CTGF/CCN2 inactivation or overexpression.
β-cell proliferation during late gestation is regulated by CTGF/CCN2 derived from β-cells themselves as well as epithelial or endothelial cells. Loss from all three sources reduces β-cell proliferation (see also Figure 2) whereas this effect is less evident after cell-specific CTGF/CCN2 inactivation due to compensatory mechanisms from the remaining CTGF/CCN2-producing cell types. CTGF/CCN2 overexpression in β-cells stimulates proliferation of both α- and β-cells.
Taken together, these results show that islet morphogenesis and function is exquisitely regulated by CTGF/CCN2 during embryogenesis, and that is it both required and sufficient to induce proliferation of embryonic β cells, while contributing to embryonic α cell function as well (Figures 2,3). Whereas the influence of embryonic CTGF/CCN2 on these parameters is evident after birth (e.g. at P1), this does not appear to be the case for CTGF/CCN2 that is expressed by β cells during postnatal independent life in as much as the β cells in more mature animals appear refractory to its presence, at least with respect to their proliferative rate and glucose regulation. Although the mechanistic basis for this difference is unclear, the data nonetheless support the concept that developmental regulation of CTGF/CCN2 expression may be a viable strategy for increasing the β cell population and islet function and, as such, it may be beneficial in emerging cell-based therapies for diabetes that involve pancreatic stem cell differentiation or β cell expansion [36].
4. CTGF/CCN2 in pancreatitis and fibrosis
In the pancreas, fibrosis is a major feature of desmoplasia (a stromal reaction characteristic of pancreatic cancer; see below) or chronic pancreatitis (CP). Together with acute pancreatitis (AP), CP is associated with long-term heavy alcohol consumption in Western society. In both forms of the disease, the fundamental pathological event is acinar cell injury leading to cellular autodigestion by proteases, the synthesis and export of which are usually tightly controlled. AP is a recurrent inflammation resulting in pain, edema, hemorrhage, acinar cell vacuolation, necrosis, and increased serum amylase and lipase. These features exist in CP which is further characterized by progressive destruction of acinar cells, accumulation of fibroblasts and ECM, calcification of pancreatic ducts, and exocrine or endocrine insufficiency resulting in, respectively, maldigestion or diabetes. Of the ~3500 US deaths due to pancreatitis each year, approximately 85% are caused by AP and 15% by CP. In the latter group, 45% of the deaths are due to alcohol. While AP and CP may be considered two discrete entities, CP may arise as a result of relapsing severe episodes of AP. This necrosis-fibrosis model is supported by studies of patients with alcoholic pancreatitis which showed that the incidence and severity of AP may result in progression to CP [37].
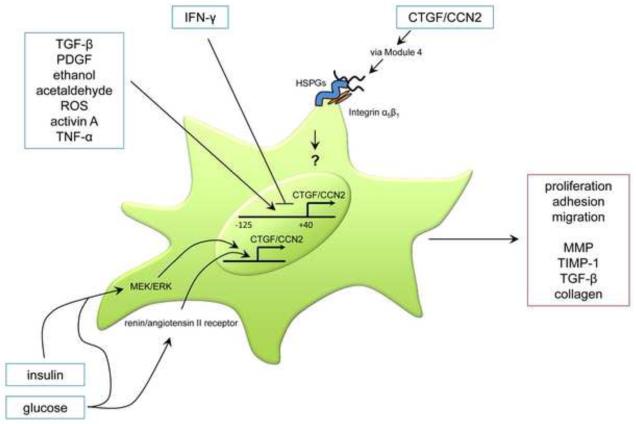
As in many other organ systems [38, 39], over-expression of TGF-β is strongly associated with inflammation and fibrosis in the pancreas. Studies of AP from human clinical samples and rodent models showed that TGF-β and its receptors are over-expressed at the time of injury and inflammation in mild edematous AP and the more severe acute necrotizing pancreatitis (ANP), the latter of which also shows a second wave of TGF-β expression that is correlated with ECM synthesis and tissue repair at sites of necrosis [40-44]. Transgenic mice that overexpress TGF-β1 in the pancreas start to exhibit a progressive accumulation of ECM within 14 days of birth which replaces the exocrine tissue and results in significant acinar loss and pancreatic fibrosis by 11 months of age [45]. The in vivo relevance of TGF-β as an inducer of pancreatic fibrosis has also been shown in a mouse model in which TGF-β was administered against a background of cerulein-induced acute pancreatitis [46]. However, attention has become focused on pancreatic CTGF/CCN2 because of extensive studies in other organ systems that have firmly established CTGF/CCN2 as a TGF-β-inducible factor that plays a dynamic role in driving fibrosis and which mediates the pro-fibrotic properties of TGF-β [38, 47]. In ANP in human tissues or experimental rodent models, CTGF/CCN2 expression occurs mainly in the remaining viable acinar or ductal cells [48]. In CP, CTGF/CCN2 mRNA expression is correlated with the degree of fibrosis and is produced at the injury site by acinar cells or fibroblastic cells, the latter of which produce the highest level of CTGF/CCN2 and likely include a population of activated PSC [49]. The involvement of activated PSC as the principal producers of CTGF/CCN2 has been definitely shown in a mouse model of alcohol-induced CP in which CTGF/CCN2 was co-localized to myofibroblasts that were positive for αSMA and collagen and which localized within areas of fibrosis [50].
Pancreatic fibrosis, driven by CTGF/CCN2 over-expression in β cells of adult mice and characterized by peri-ductular and peri-vascular collagen deposition was recently described [36] but the underlying mechanisms and role of PSC have yet to be reported. On the other hand, mice that were transgenic for pancreatic TGF-β showed an association between CTGF/CCN2 and increased fibronectin production or PSC proliferation [51] and there is now a growing body of evidence that CTGF/CCN2, acting via autocrine or paracrine pathways, plays an important role in pathways of PSC activation and fibrogenesis. Primary mouse or rat PSC maintained in culture produce CTGF/CCN2 [23, 25, 26, 52-55] and its levels increase over time as the cells become progressively activated in vitro [23, 26]. In transiently activated PSC, CTGF/CCN2 mRNA or protein levels are enhanced by treatment of the cells with TGF-β, tumor necrosis factor-α (TNF-α), activin A or ethanol, the latter acting after its metabolism to acetaldehyde and production of oxidant stress [23, 25, 54] (Figure 4). The region -125 to +40 of the CTGF/CCN2 promoter is required for basal activity in cultured PSC and promoter activity is stimulated by TGF-β, platelet-derived growth factor, ethanol, acetaldehyde, activin A, or TNF-α but is inhibited by interferon-gamma [23, 54]. In PSC that have been exposed to high glucose concentrations, the renin-angiotensin system becomes activated and drives expression and secretion of ECM proteins and inhibits collagen degradation [55] (Figure 4). Under these conditions, the PSC also show enhanced expression of CTGF/CCN2 that, along with other matrix proteins, can be reversed by candesartan or ramiprilat which, respectively, block the angiotensin II receptor or angiotensin-converting enzyme [55]. Glucose- or insulin-stimulated CTGF/CCN2 expression in PSC is attenuated by U1026, an inhibitor of ERK1/2 signaling that also blocks cell proliferation [53], supporting a role for CTGF/CCN2 in mediating the additive effects of hyperglycemia and hyperinsulinemia on PSC activation and proliferation that are associated with islet-restricted fibrosis in type 2 diabetes.
Fig. 4. Principal pathways of CTGF/CCN2 production and action in activated PSC.
CTGF/CCN2 functions in activated PSC following injury, being produced in response to diverse extracellular signals as well as regulating PSC responses via interactions with integrins/HSPG. In chronic injury, PSC are perpetually activated leading to excess collagen production downstream of CTGF/CCN2.
In addition to producing CTGF/CCN2, cultured PSC also respond when exogenously exposed to this molecule, resulting in enhanced adhesion, migration, proliferation, phosphorylation of ERK1/2 and STAT3, and expression of matrix metalloprotease-9, tissue inhibitor of metalloproteases-1, TGF-β or collagen [23, 24, 54] (Figure 4). CTGF/CCN2-dependent adhesion or migration of activated PSC is mediated by specific motifs in module 4 that bind to cell surface HSPG or integrin α5β1, the latter of which is also required for CTGF/CCN2-induced collagen synthesis [23, 24]. Down-regulation of CTGF/CCN2 expression by small interfering RNA causes a reduction in the rate of PSC proliferation, demonstrating that endogenous CTGF/CCN2 can act as autocrine regulator of PSC activation [54]. These data have highlighted the possibility that antagonism of CTGF/CCN2 production or action in PSC may provide novel therapeutic approaches in pancreatic fibrosis.
5. CTGF/CCN2 in pancreatic cancer
Each year in the US, there are more than 30,000 diagnoses of pancreatic ductal adenocarcinoma (PDAC), a highly aggressive disease with poor prognosis and a 5-year survival rate of less than 4%. PDAC is the 4th leading cause of cancer-related mortality in the West, with the majority (75-85%) of patients presenting with non-resectable tumors at the time of diagnosis. In one approach, CTGF/CCN2 has been studied as a product of the tumor cells which are characterized by activation mutations in the K-ras proto-oncogene during a pre-invasive state termed pancreatic intra-epithelial neoplasia (PanIN) and, in invasive PDAC, a high frequency of inactivation of tumor suppressor genes such as p53, p16, Smad4 or the type II TGF-β receptor, the latter two of which suggest an important role of aberrant TGF-β signaling in PDAC progression. In another approach, studies have focused on the contribution of CTGF/CCN2 to the desmoplastic reaction which involves the intense production of an interstitial stroma that can contribute up to 80% of the tumor mass and comprises fibroblasts, myofibrobalsts, inflammatory cells and PSC that are associated with a highly expanded ECM comprising collagens, fibronectin, laminin, and proteoglycans. This reaction may constitute a physical barrier that restricts cancer spread but increasingly it is considered a reservoir of ECM-associated growth factors that functions dynamically to sustain tumor growth and increase its invasive potential [56].
Clinical specimens of pancreatic cancer express elevated levels of CTGF/CCN2 mRNA that are 40-60-fold greater than normal pancreas and well correlated with the extent and intensity of desmoplasia [57, 58]. In a study of 2 pancreatic cancer patients, CTGF/CCN2 mRNA appeared to be localized exclusively in the tumor cells themselves [59] and increased CTGF/CCN2 in tumor cells as compared to stromal cells was reported in two other studies of 30 or 58 PDAC patients [60, 61]. On the other hand, CTGF/CCN2 expression in other PDAC samples was shown to occur in both the tumor and stroma but with preferential localization to stromal fibroblasts, including PSC, and this finding was recapitulated in a mouse xenograft model [57, 58]. Culture-activated PSC contain CTGF/CCN2 mRNA in higher abundance than a variety of cultured pancreatic tumor cell lines which express variable but nonetheless lower amounts of the transcript [57]. Whereas TGF-β promotes CTGF/CCN2 mRNA production in PSC [57], TGF-β receptor mediated signaling is frequently impaired in pancreatic tumor cells as mentioned above. Studies of CTGF/CCN2 expression and promoter activity have established that MEK/ERK drives basal CTGF/CCN2 production in Panc-1 pancreatic tumor cells in a Smad-independent fashion [62] as well as in other pancreatic cancer cell lines that have defective Smad signaling [63]. On the other hand, in pancreatic tumor cell lines that are TGF-β-responsive (Panc-1, HPAF), CTGF/CCN2 production is dependent on the type I TGF-β receptor and MEK/ERK [63]. These data support the notion that that the production of CTGF/CCN2 via TGF-β-dependent or -independent mechanisms is likely stimulated via mutational activation of ras, which commonly occurs in PDAC [64, 65] and is a major driver of the ERK/MEK signaling pathway [62, 63]. Further, in an inducible mouse model of PDAC, generated by knockout of the type II TGF-β receptor in combination with overexpression of K-ras, there was strong stromal expression of CTGF/CCN2, especially in areas adjacent to the tumor whereas no such staining was observed in the stroma of mice that over-expressed K-ras alone and developed PanIN (see above) [66], highlighting the combinatorial effects of activated ras and defective TGF-β tumor signaling as a trigger for stromal CTGF/CCN2 production and development of a more aggressive tumor type (Table 1). In follow-up studies, induction of CTGF/CCN2 in stromal cells, which were shown to confer a growth advantage on PDAC in vivo [67], was attributed to the production by the tumor cells of Cxc chemokines and this was reinforced by the finding that Cxcr2 inhibitors decreased stromal CTGF/CCN2 expression, reduced tumor progression and increased overall survival [67] (Table 1). Thus, rather than being functionally segregated, substantial molecular cross-talk exists between the stromal and tumor compartments in PDAC, and complex autocrine or paracrine pathways of CTGF/CCN2 regulation and action have begun to be identified that are dynamically regulated by Cxcr2-dependent chemokines downstream of ras/TGF-β receptors, and which directly regulate PDAC progression.
Table 1.
Principal actions of CTGF/CCN2 in animal models of PDAC
| Alteration | Model | Animals | Therapy | Outcome | Refs |
|---|---|---|---|---|---|
| CTGF knockdown |
Subcutaneous xenografts of Panc-1 cells stably expressing CTGF/CCN2 shRNA |
Nude male mice 8-10 weeks |
Xenografted Panc-1 cells with the highest CTGF/CCN2 knockdown did not form tumors |
60 | |
| Orthotopically implanted pancreatic tumors from Panc-1 xenogra fts stably expressing CTGF/CCN2 shRNA |
Nude male mice 8-10 weeks |
1. Smaller tumors 2. Significant increa se in survival |
60 | ||
| Subcutaneous xenografts of Mia PaCa-2 cells over- expressing CTGF/CCN2 |
Nude male mice 6-8 weeks |
FG-3019 (CTGF/CCN2 m Ab) bi-weekly injection (40 mg/kg) when tumor was150-200 mm3 |
Tumor growth inhibited through induction of apoptosis |
71 | |
| Subcutaneous xenografts of Panc-1 or Su86. 86 cells with over- expression of endogenous CTGF/CCN2 |
Nude male mice 6-8 weeks |
FG-3019 (CTGF/CCN2 m Ab) bi-weekly i.p. injection (140 mg/kg) for 6 weeks |
1. Mean tumor size decreased by > 50% in Panc-1 xenografts as compared to controls, and inhibited lymph node metastasis 2. Tumor size decrease by<75% in Su86.86 xenografts |
71 | |
| Orthotopically implanted pancreatic tumors from Panc-1 xenogra fts over-expressing CTGF/CCN2 |
Nude male mice 6-8 weeks |
FG-3019 (CTGF/CCN2 mAb) started 2 weeks post-implantation ; bi-weekly i.p injections (20 mg/kg) for 6 weeks |
1 Significantly smaller tumors as compared to controls 2. Reduced mesenteric lymph node metastasis 3. Reduced angiogenesis |
70 | |
| CTGF over- expression |
Orthotopically implanted pancreatic tumors from Panc-1 xenogra fts over-expressing CTGF/CCN2 |
Nude male mice 8-10 weeks |
1. Enhanced growth and metastasis 2. Significantly decreased survival times |
60 | |
| Subcutaneous xenografts of Mia PaCa-2 cells over- expressing CTGF/CCN2 |
Nude male mice 6-8 weeks |
1. Significantly enhanced tumor growth directly proportional to CTGF/CCN2 protein produced 2. Increased proliferation 3. Reduced apoptosis |
71 | ||
| Kras over- expression with type II TGF-β receptor knockdown |
−Kras + Tgfbr2KO | Ptf1acre/+; Tgfbr2flox/flox; LSL-KrasG12D/+ C57Bl/6/DBA/2/129/S yJae |
1. Development of PDAC with strong stromal CTGF/CCN2 expression. 2. Over-expression of K- ras alone causes PanIN but not PDAC |
66 | |
| Kras + Tgfbr2KO | Ptf1acre/+; Tgfbr2flox/flox; LSL-KrasG12D/+ C57Bl/6/DBA/2/129/S yJae |
SB225002 or Repertaxin (Cxcr2 inhibitors) |
1. Decreased CTGF/CCN2 expression 2. Significantly reduced tumor volume |
67 |
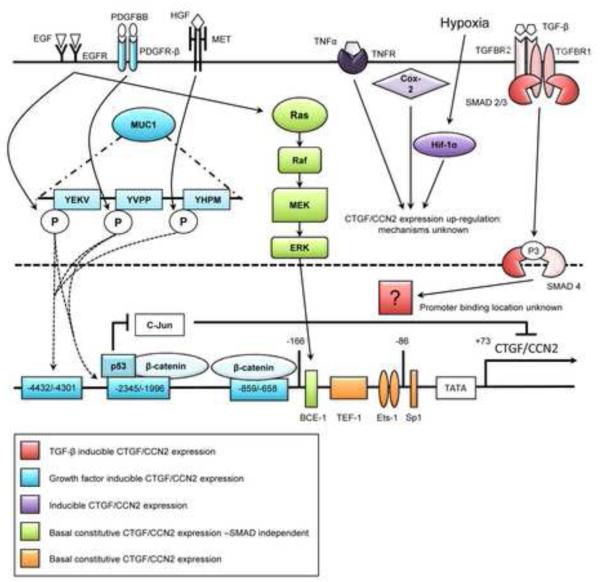
Other pathways of CTGF/CCN2 gene transcription have been described in PDAC, including their induction as immediate early genes in human pancreatic tumor cells in response to epidermal growth factor or transforming growth factor-alpha which, unlike the response of the same cells to TGF-β, do not cause enhanced collagen production [58]. Also, CTGF/CCN2 expression in pancreatic cancer cells in response to serum growth factors is enabled by high expression of cell surface mucin (MUC1) which is overexpressed in PDAC and involved in PDAC invasion and metastasis [68]. Mechanistically, this effect occurs via differential phosphorylation of the cytoplasmic tail of MUC1 which allows it to interact with various transcription factors and reorganize their association with the CTGF/CCN2 promoter. Specifically, MUC1 enhances the association of β-catenin or mutant p53 with the CTGF/CCN2 promoter to drive CTGF/CCN2 expression while at the same time preventing c-jun, a transcriptional repressor, from binding to the same upstream elements [68]. This mode of action is broadly used by MUC1 to regulate the activity of a diverse variety of transcription factors for many target genes and is proposed to drive formation of a reactive tumor micro-environment - of which CTGF/CCN2 was thus identified to be an important component in PDAC. The major mechanisms of CTGF/CCN2 transcriptional regulation in PDAC tumor cells are summarized in Figure 5.
Fig. 5. Key pathways regulating CTGF/CCN2 production in PDAC tumor cells.
Growth factors, cytokines, or environmental cues drive CTGF/CCN2 production via activation of ras, Smad or as yet undefined mechanisms. Downstream interactions with transcription response elements in the CTGF/CCN2 promoter increase CTGF/CCN2production, an outcome that can be amplified by MUC1.
As well as potentially driving aspects of the desmoplastic reaction via its promotion of fibrogenic signaling pathways in PSC (see above), CTGF/CCN2 has been identified in a metastasis-related gene cluster in PDAC neoplastic epithelium [59, 69]. Incubation of Panc-1 cells in vitro with recombinant CTGF/CCN2 stimulated cell proliferation in monolayer culture as well invasion in a Matrigel assay [70]. Anchorage-independent growth of pancreatic tumor cells in vitro was reduced after knock-down of CTGF/CCN2 mRNA expression [60] and, conversely, was enhanced after transfection of the cells with human CTGF/CCN2 cDNA - the latter effect being blocked by FG-3019, a neutralizing CTGF/CCN2 monoclonal antibody [71]. Unlike addition of exogenous CTGF/CCN2 [70], forced over- or under-expression of CTGF/CCN2 expression in pancreatic tumor cells was not associated with altered rates of cell proliferation in monolayer culture [60, 71], highlighting the importance of contextual cues for specific CTGF/CCN2 responses. After subcutaneous injection in mice, PaCa-2 clones expressing differing amounts of human CTGF/CCN2 spawned tumors that were directly proportional in size to the CTGF/CCN2 protein level produced [71] and, similarly, silencing of CTGF/CCN2 gene expression in Panc-1 cells resulted in their inability to form solid subcutaneous tumors in mice [60] (Table 1). Several studies have shown that CTGF/CCN2 expression in pancreatic tumor models in vivo is associated with increased proliferation and decreased apoptosis of the tumor cell [60, 71] (Table 1). Notably, CTGF/CCN2-expressing tumor cells are present in hypoxic regions of experimental tumors in vivo or in clinical samples and this has been attributed to the ability of CTGF/CCN2 to confer protection against hypoxia-mediated apoptosis [60]. Also, NS-398, a non-steroidal anti-inflammatory drug that selectively targets cyclo-oxygenase-2, was shown to inhibit growth and proliferation in pancreatic tumor cell lines and to induce their apoptosis while at the same time causing significant down-regulation of CTGF/CCN2 expression, although mechanistic studies to functionally link these observations were not reported [72].
Definitive evidence for a functional role of CTGF/CCN2 in driving PDAC, at least in animal models, came from two landmark studies in which FG-3019 was shown to reduce tumor growth, metastasis and angiogenesis in mice either injected subcutaneously with CTGF-transfected Panc-1 cells [71] or which were implanted intra-pancreatically with fragments of tissue from subcutaneous tumors derived from wild-type Panc-1 cells [70] (Table1). While these promising outcomes may translate to novel therapeutic approaches that target CTGF/CCN2 production or action in the PDAC patient population, further work is needed to clarify whether FG-3019 exerts its inhibitory action by acting on the tumor cells, stromal cells, or both. Moreover, it should be emphasized that entirely different conclusions regarding the role of CTGF/CCN2 in human pancreatic cancer were made based on the findings that CTGF/CCN2 expression patterns were correlated with lower grade (more highly differentiated) PDAC and improved patient survival [57, 61]. Curiously, CTGF/CCN2 mRNA was preferentially localized to the stroma in one of these studies [57] and CTGF/CCN2 protein to the tumor cells in the other study [61]. While the apparent positive prognostic value of CTGF/CCN2 levels in PDAC highlighted in these reports is provocative, follow-up studies that include mechanistic and functional approaches will need to be undertaken to determine the validity of the findings and to reconcile them with the other data discussed above.
6. CTGF/CCN2 in pancreatic inflammation
Emerging data in the CCN field, especially from studies of CYR61/CCN1 and NOV/CCN3, have begun to highlight this family of proteins as important components of inflammatory responses [73-75] and several studies appear to support a role for CTGF/CCN2 in pancreatic inflammation. For example, in human or rat ANP, CTGF/CCN2 production is correlated with that of TGF-β and follows a bi-phasic pattern in which their first peak of expression is co-incident with inflammatory cell infiltration [48]. Rather than being produced by inflammatory cells, CTGF/CCN2 expression occurs in the remaining ductular cells, acinar cells and fibroblasts of the injured tissue suggesting that it might either contribute to the inflammatory reaction or be produced as a consequence of it [48]. These possibilities are supported, respectively, by the findings that CTGF/CCN2 treatment of activated rat PSC in vitro causes increased expression of the pro-inflammatory cytokines interleukin-1β or interleukin-6 [54], or that CTGF/CCN2 production occurs downstream of Cxcr2-dependent chemokines in experimental PDAC [67]. While these observations are intriguing, these studies were not designed to specifically address the role of CTGF/CCN2 in pancreatic inflammation and this question will need to be directly addressed in future research.
7. Summary
A substantial body of evidence has shown unequivocally that CTGF/CCN2 plays critical roles in pancreatic development, fibrosis, and cancer. In vitro approaches coupled with elegant in vivo models have provided definitive evidence for epithelial-, endothelial-, or β cell-derived CTGF/CCN2 in driving embryonic β cell replication, for the regulation of PSC fibrogenic pathways by CTGF/CCN2, and for the involvement of CTGF/CCN2 in PDAC and its associated desmoplasia including a chemokine-dependent CTGF/CCN2 axis that regulates tumor-stromal interactions. Although additional studies are needed to understand the mechanisms by which pancreatic CTGF/CCN2 is regulated and the signaling pathways that it controls, strategies that attenuate or augment CTGF/CCN2 circuitry in the pancreas hold promise as innovative treatments for several major pancreatic diseases for which there are currently inadequate therapies, including diabetes, pancreatitis, and cancer.
Acknowledgements
This work was supported by NIH grant R01 AA015554 awarded to D.R.B.
9. Abbreviations
- ANP
acute necrotizing pancreatitis
- AP
acute pancreatitis
- CP
chronic pancreatitis
- CTGF/CCN2
connective tissue growth factor
- CYR61/CCN1
cysteine-rich 61
- ECM
extracellular matrix
- HSPG
heparan sulfate proteoglycan
- LRP
low density lipoprotein receptor-related protein
- MUC1
mucin
- NOV/CCN3
nephroblastoma overexpressed
- PanIN
pancreatic intra-epithelial neoplasia
- PDAC
pancreatic ductal adenocarcinoma
- PSC
pancreatic stellate cells
- TGF-β
transforming growth factor beta
- TNF-α
tumor necrosis factor alpha
- WISP-1/CCN4
Wnt-inducible secreted protein-1
Biography
David R. Brigstock received his Ph.D. from Cambridge University, UK in 1988 and then completed a 3-year post-doctoral fellowship at Children’s Hospital, Boston MA. In 1991, he joined the Department of Surgery at Nationwide Children’s Hospital and The Ohio State University, Columbus OH where he has remained to the present day. In the mid-1990’s he discovered the occurrence of bioactive low mass forms of CTGF/CCN2, a finding that was instrumental in mapping functional domains in the CTGF/CCN2 molecule. His current research interests are supported by funding from NIH and focus on the role of CTGF/CCN2 in the function of hepatic or pancreatic stellate cells. He serves on the Scientific Board of the International CCN Society (http://ccnsociety.com), which fosters interest in all aspects of CCN biology 
Alyssa Charrier received her B.S. from Brigham Young University, Provo, UT in 2004. She then worked at Amgen for several years before joining The Ohio State University as a graduate student in the Molecular, Cellular and Developmental Biology Program. Her research projects focus on the role of CTGF/CCN2 in regulating inflammation and fibrosis in alcoholic pancreatitis. 
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
11. References
- [1].Brigstock DR. The connective tissue growth factor/cysteine-rich 61/nephroblastoma overexpressed (CCN) family. Endocr Rev. 1999;20:189–206. doi: 10.1210/edrv.20.2.0360. [DOI] [PubMed] [Google Scholar]
- [2].Brigstock DR, Goldschmeding R, Katsube KI, et al. Proposal for a unified CCN nomenclature. Mol Pathol. 2003;56:127–8. doi: 10.1136/mp.56.2.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Bork P. The modular architecture of a new family of growth regulators related to connective tissue growth factor. FEBS Lett. 1993;327:125–30. doi: 10.1016/0014-5793(93)80155-n. [DOI] [PubMed] [Google Scholar]
- [4].Rachfal AW, Brigstock DR. Structural and functional properties of CCN proteins. Vitam Horm. 2005;70:69–103. doi: 10.1016/S0083-6729(05)70003-0. [DOI] [PubMed] [Google Scholar]
- [5].Chen CC, Lau LF. Functions and mechanisms of action of CCN matricellular proteins. Int J Biochem Cell Biol. 2009;41:771–83. doi: 10.1016/j.biocel.2008.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Lau LF. CCN1/CYR61: the very model of a modern matricellular protein. Cell Mol Life Sci. 2011;68:3149–63. doi: 10.1007/s00018-011-0778-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Lau LF, Lam SC. The CCN family of angiogenic regulators: the integrin connection. Exp Cell Res. 1999;248:44–57. doi: 10.1006/excr.1999.4456. [DOI] [PubMed] [Google Scholar]
- [8].Gao R, Brigstock DR. Low density lipoprotein receptor-related protein (LRP) is a heparin-dependent adhesion receptor for connective tissue growth factor (CTGF) in rat activated hepatic stellate cells. Hepatol Res. 2003;27:214–20. doi: 10.1016/s1386-6346(03)00241-9. [DOI] [PubMed] [Google Scholar]
- [9].Kawata K, Kubota S, Eguchi T, et al. Role of low-density lipoprotein receptor related protein 1 (LRP1) in CCN2/connective tissue growth factor (CTGF) protein transport in chondrocytes. J Cell Sci. 2012 [Google Scholar]
- [10].Wahab NA, Weston BS, Mason RM. Connective tissue growth factor CCN2 interacts with and activates the tyrosine kinase receptor TrkA. J Am Soc Nephrol. 2005;16:340–51. doi: 10.1681/ASN.2003100905. [DOI] [PubMed] [Google Scholar]
- [11].Mo FE, Lau LF. The matricellular protein CCN1 is essential for cardiac development. Circ Res. 2006;99:961–9. doi: 10.1161/01.RES.0000248426.35019.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Mo FE, Muntean AG, Chen CC, Stolz DB, Watkins SC, Lau LF. CYR61 (CCN1) is essential for placental development and vascular integrity. Mol Cell Biol. 2002;22:8709–20. doi: 10.1128/MCB.22.24.8709-8720.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Baguma-Nibasheka M, Kablar B. Pulmonary hypoplasia in the connective tissue growth factor (Ctgf) null mouse. Dev Dyn. 2008;237:485–93. doi: 10.1002/dvdy.21433. [DOI] [PubMed] [Google Scholar]
- [14].Ivkovic S, Yoon BS, Popoff SN, et al. Connective tissue growth factor coordinates chondrogenesis and angiogenesis during skeletal development. Development. 2003;130:2779–91. doi: 10.1242/dev.00505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Crawford LA, Guney MA, Oh YA, et al. Connective tissue growth factor (CTGF) inactivation leads to defects in islet cell lineage allocation and beta-cell proliferation during embryogenesis. Mol Endocrinol. 2009;23:324–36. doi: 10.1210/me.2008-0045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Heath E, Tahri D, Andermarcher E, Schofield P, Fleming S, Boulter CA. Abnormal skeletal and cardiac development, cardiomyopathy, muscle atrophy and cataracts in mice with a targeted disruption of the Nov (Ccn3) gene. BMC Dev Biol. 2008;8:18. doi: 10.1186/1471-213X-8-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Hurvitz JR, Suwairi WM, Van Hul W, et al. Mutations in the CCN gene family member WISP3 cause progressive pseudorheumatoid dysplasia. Nat Genet. 1999;23:94–8. doi: 10.1038/12699. [DOI] [PubMed] [Google Scholar]
- [18].Guney MA, Petersen CP, Boustani A, et al. Connective tissue growth factor acts within both endothelial cells and beta cells to promote proliferation of developing beta cells. Proc Natl Acad Sci U S A. 2011;108:15242–7. doi: 10.1073/pnas.1100072108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Prado CL, Pugh-Bernard AE, Elghazi L, Sosa-Pineda B, Sussel L. Ghrelin cells replace insulin-producing beta cells in two mouse models of pancreas development. Proc Natl Acad Sci U S A. 2004;101:2924–9. doi: 10.1073/pnas.0308604100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Apte MV, Haber PS, Applegate TL, et al. Periacinar stellate shaped cells in rat pancreas: identification, isolation, and culture. Gut. 1998;43:128–33. doi: 10.1136/gut.43.1.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Apte M, Pirola R, Wilson J. The fibrosis of chronic pancreatitis: new insights into the role of pancreatic stellate cells. Antioxid Redox Signal. 2011;15:2711–22. doi: 10.1089/ars.2011.4079. [DOI] [PubMed] [Google Scholar]
- [22].Haber PS, Keogh GW, Apte MV, et al. Activation of pancreatic stellate cells in human and experimental pancreatic fibrosis. Am J Pathol. 1999;155:1087–95. doi: 10.1016/S0002-9440(10)65211-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Gao R, Brigstock DR. Connective tissue growth factor (CCN2) in rat pancreatic stellate cell function: integrin alpha5beta1 as a novel CCN2 receptor. Gastroenterology. 2005;129:1019–30. doi: 10.1053/j.gastro.2005.06.067. [DOI] [PubMed] [Google Scholar]
- [24].Gao R, Brigstock DR. A novel integrin alpha5beta1 binding domain in module 4 of connective tissue growth factor (CCN2/CTGF) promotes adhesion and migration of activated pancreatic stellate cells. Gut. 2006;55:856–62. doi: 10.1136/gut.2005.079178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Lawrencia C, Charrier A, Huang G, Brigstock DR. Ethanol-mediated expression of connective tissue growth factor (CCN2) in mouse pancreatic stellate cells. Growth Factors. 2009;27:91–9. doi: 10.1080/08977190902786319. [DOI] [PubMed] [Google Scholar]
- [26].Shinji T, Ujike K, Ochi K, et al. Establishment of a novel collagenase perfusion method to isolate rat pancreatic stellate cells and investigation of their gene expression of TGF-beta1, type I collagen, and CTGF in primary culture or freshly isolated cells. Acta Med Okayama. 2002;56:211–8. doi: 10.18926/AMO/31682. [DOI] [PubMed] [Google Scholar]
- [27].Pan FC, Wright C. Pancreas organogenesis: from bud to plexus to gland. Dev Dyn. 2011;240:530–65. doi: 10.1002/dvdy.22584. [DOI] [PubMed] [Google Scholar]
- [28].Rojas A, Khoo A, Tejedo JR, Bedoya FJ, Soria B, Martin F. Islet cell development. Adv Exp Med Biol. 2010;654:59–75. doi: 10.1007/978-90-481-3271-3_4. [DOI] [PubMed] [Google Scholar]
- [29].Kim SK, MacDonald RJ. Signaling and transcriptional control of pancreatic organogenesis. Curr Opin Genet Dev. 2002;12:540–7. doi: 10.1016/s0959-437x(02)00338-6. [DOI] [PubMed] [Google Scholar]
- [30].Gradwohl G, Dierich A, LeMeur M, Guillemot F. Neurogenin 3 is required for the development of the four endocrine cell lineages of the pancreas. Proc Natl Acad Sci U S A. 2000;97:1607–11. doi: 10.1073/pnas.97.4.1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes. 2003;52:102–10. doi: 10.2337/diabetes.52.1.102. [DOI] [PubMed] [Google Scholar]
- [32].Yoon KH, Ko SH, Cho JH, et al. Selective beta-cell loss and alpha-cell expansion in patients with type 2 diabetes mellitus in Korea. J Clin Endocrinol Metab. 2003;88:2300–8. doi: 10.1210/jc.2002-020735. [DOI] [PubMed] [Google Scholar]
- [33].Surveyor GA, Brigstock DR. Immunohistochemical localization of connective tissue growth factor (CTGF) in the mouse embryo between days 7.5 and 14.5 of gestation. Growth Factors. 1999;17:115–24. doi: 10.3109/08977199909103520. [DOI] [PubMed] [Google Scholar]
- [34].Wilding Crawford L, Tweedie Ables E, Oh YA, Boone B, Levy S, Gannon M. Gene expression profiling of a mouse model of pancreatic islet dysmorphogenesis. PLoS One. 2008;3:e1611. doi: 10.1371/journal.pone.0001611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Kapasa M, Serafimidis I, Gavalas A, Kossida S. Identification of phylogenetically conserved enhancer elements implicated in pancreas development in the WISP1 and CTGF orthologs. Genomics. 2008;92:301–8. doi: 10.1016/j.ygeno.2008.06.003. [DOI] [PubMed] [Google Scholar]
- [36].Gunasekaran U, Hudgens CW, Wright BT, Maulis MF, Gannon M. Differential regulation of embryonic and adult beta cell replication. Cell Cycle. 2012;11 doi: 10.4161/cc.20545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Ammann RW, Muellhaupt B. Progression of alcoholic acute to chronic pancreatitis. Gut. 1994;35:552–6. doi: 10.1136/gut.35.4.552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Leask A, Abraham DJ. TGF-beta signaling and the fibrotic response. FASEB J. 2004;18:816–27. doi: 10.1096/fj.03-1273rev. [DOI] [PubMed] [Google Scholar]
- [39].Verrecchia F, Mauviel A. Transforming growth factor-beta and fibrosis. World J Gastroenterol. 2007;13:3056–62. doi: 10.3748/wjg.v13.i22.3056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Friess H, Lu Z, Riesle E, et al. Enhanced expression of TGF-betas and their receptors in human acute pancreatitis. Ann Surg. 1998;227:95–104. doi: 10.1097/00000658-199801000-00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Konturek PC, Dembinski A, Warzecha Z, et al. Comparison of epidermal growth factor and transforming growth factor-beta1 expression in hormone-induced acute pancreatitis in rats. Digestion. 1998;59:110–9. doi: 10.1159/000007483. [DOI] [PubMed] [Google Scholar]
- [42].Menke A, Yamaguchi H, Gress TM, Adler G. Extracellular matrix is reduced by inhibition of transforming growth factor beta1 in pancreatitis in the rat. Gastroenterology. 1997;113:295–303. doi: 10.1016/s0016-5085(97)70107-0. [DOI] [PubMed] [Google Scholar]
- [43].Muller-Pillasch F, Gress TM, Yamaguchi H, Geng M, Adler G, Menke A. The influence of transforming growth factor beta 1 on the expression of genes coding for matrix metalloproteinases and tissue inhibitors of metalloproteinases during regeneration from cerulein-induced pancreatitis. Pancreas. 1997;15:168–75. doi: 10.1097/00006676-199708000-00009. [DOI] [PubMed] [Google Scholar]
- [44].Riesle E, Friess H, Zhao L, et al. Increased expression of transforming growth factor beta s after acute oedematous pancreatitis in rats suggests a role in pancreatic repair. Gut. 1997;40:73–9. doi: 10.1136/gut.40.1.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Lee MS, Gu D, Feng L, et al. Accumulation of extracellular matrix and developmental dysregulation in the pancreas by transgenic production of transforming growth factor-beta 1. Am J Pathol. 1995;147:42–52. [PMC free article] [PubMed] [Google Scholar]
- [46].Van Laethem JL, Robberecht P, Resibois A, Deviere J. Transforming growth factor beta promotes development of fibrosis after repeated courses of acute pancreatitis in mice. Gastroenterology. 1996;110:576–82. doi: 10.1053/gast.1996.v110.pm8566606. [DOI] [PubMed] [Google Scholar]
- [47].Leask A, Abraham DJ. The role of connective tissue growth factor, a multifunctional matricellular protein, in fibroblast biology. Biochem Cell Biol. 2003;81:355–63. doi: 10.1139/o03-069. [DOI] [PubMed] [Google Scholar]
- [48].di Mola FF, Friess H, Riesle E, et al. Connective tissue growth factor is involved in pancreatic repair and tissue remodeling in human and rat acute necrotizing pancreatitis. Ann Surg. 2002;235:60–7. doi: 10.1097/00000658-200201000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].di Mola FF, Friess H, Martignoni ME, et al. Connective tissue growth factor is a regulator for fibrosis in human chronic pancreatitis. Ann Surg. 1999;230:63–71. doi: 10.1097/00000658-199907000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Charrier AL, Brigstock DR. Connective tissue growth factor production by activated pancreatic stellate cells in mouse alcoholic chronic pancreatitis. Lab Invest. 2010;90:1179–88. doi: 10.1038/labinvest.2010.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Vogelmann R, Ruf D, Wagner M, Adler G, Menke A. Effects of fibrogenic mediators on the development of pancreatic fibrosis in a TGF-beta1 transgenic mouse model. Am J Physiol Gastrointest Liver Physiol. 2001;280:G164–72. doi: 10.1152/ajpgi.2001.280.1.G164. [DOI] [PubMed] [Google Scholar]
- [52].Fitzner B, Brock P, Nechutova H, et al. Inhibitory effects of interferon-gamma on activation of rat pancreatic stellate cells are mediated by STAT1 and involve down-regulation of CTGF expression. Cell Signal. 2007;19:782–90. doi: 10.1016/j.cellsig.2006.10.002. [DOI] [PubMed] [Google Scholar]
- [53].Hong OK, Lee SH, Rhee M, et al. Hyperglycemia and hyperinsulinemia have additive effects on activation and proliferation of pancreatic stellate cells: possible explanation of islet-specific fibrosis in type 2 diabetes mellitus. J Cell Biochem. 2007;101:665–75. doi: 10.1002/jcb.21222. [DOI] [PubMed] [Google Scholar]
- [54].Karger A, Fitzner B, Brock P, et al. Molecular insights into connective tissue growth factor action in rat pancreatic stellate cells. Cell Signal. 2008;20:1865–72. doi: 10.1016/j.cellsig.2008.06.016. [DOI] [PubMed] [Google Scholar]
- [55].Ko SH, Hong OK, Kim JW, et al. High glucose increases extracellular matrix production in pancreatic stellate cells by activating the renin-angiotensin system. J Cell Biochem. 2006;98:343–55. doi: 10.1002/jcb.20797. [DOI] [PubMed] [Google Scholar]
- [56].Korc M. Pancreatic cancer-associated stroma production. Am J Surg. 2007;194:S84–6. doi: 10.1016/j.amjsurg.2007.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Hartel M, Di Mola FF, Gardini A, et al. Desmoplastic reaction influences pancreatic cancer growth behavior. World J Surg. 2004;28:818–25. doi: 10.1007/s00268-004-7147-4. [DOI] [PubMed] [Google Scholar]
- [58].Wenger C, Ellenrieder V, Alber B, et al. Expression and differential regulation of connective tissue growth factor in pancreatic cancer cells. Oncogene. 1999;18:1073–80. doi: 10.1038/sj.onc.1202395. [DOI] [PubMed] [Google Scholar]
- [59].Iacobuzio-Donahue CA, Ryu B, Hruban RH, Kern SE. Exploring the host desmoplastic response to pancreatic carcinoma: gene expression of stromal and neoplastic cells at the site of primary invasion. Am J Pathol. 2002;160:91–9. doi: 10.1016/S0002-9440(10)64353-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Bennewith KL, Huang X, Ham CM, et al. The role of tumor cell-derived connective tissue growth factor (CTGF/CCN2) in pancreatic tumor growth. Cancer Res. 2009;69:775–84. doi: 10.1158/0008-5472.CAN-08-0987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Mantoni TS, Schendel RR, Rodel F, et al. Stromal SPARC expression and patient survival after chemoradiation for non-resectable pancreatic adenocarcinoma. Cancer Biol Ther. 2008;7:1806–15. doi: 10.4161/cbt.7.11.6846. [DOI] [PubMed] [Google Scholar]
- [62].Pickles M, Leask A. Analysis of CCN2 promoter activity in PANC-1 cells: regulation by ras/MEK/ERK. J Cell Commun Signal. 2007;1:85–90. doi: 10.1007/s12079-007-0008-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Kwon S, Munroe X, Crawley SC, et al. Expression of connective tissue growth factor in pancreatic cancer cell lines. Int J Oncol. 2007;31:693–703. [PubMed] [Google Scholar]
- [64].Klimstra DS, Longnecker DS. K-ras mutations in pancreatic ductal proliferative lesions. Am J Pathol. 1994;145:1547–50. [PMC free article] [PubMed] [Google Scholar]
- [65].Rozenblum E, Schutte M, Goggins M, et al. Tumor-suppressive pathways in pancreatic carcinoma. Cancer Res. 1997;57:1731–4. [PubMed] [Google Scholar]
- [66].Ijichi H, Chytil A, Gorska AE, et al. Aggressive pancreatic ductal adenocarcinoma in mice caused by pancreas-specific blockade of transforming growth factor-beta signaling in cooperation with active Kras expression. Genes Dev. 2006;20:3147–60. doi: 10.1101/gad.1475506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [67].Ijichi H, Chytil A, Gorska AE, et al. Inhibiting Cxcr2 disrupts tumor-stromal interactions and improves survival in a mouse model of pancreatic ductal adenocarcinoma. J Clin Invest. 2011;121:4106–17. doi: 10.1172/JCI42754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Behrens ME, Grandgenett PM, Bailey JM, et al. The reactive tumor microenvironment: MUC1 signaling directly reprograms transcription of CTGF. Oncogene. 2010;29:5667–77. doi: 10.1038/onc.2010.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Ryu B, Jones J, Hollingsworth MA, Hruban RH, Kern SE. Invasion-specific genes in malignancy: serial analysis of gene expression comparisons of primary and passaged cancers. Cancer Res. 2001;61:1833–8. [PubMed] [Google Scholar]
- [70].Aikawa T, Gunn J, Spong SM, Klaus SJ, Korc M. Connective tissue growth factor-specific antibody attenuates tumor growth, metastasis, and angiogenesis in an orthotopic mouse model of pancreatic cancer. Mol Cancer Ther. 2006;5:1108–16. doi: 10.1158/1535-7163.MCT-05-0516. [DOI] [PubMed] [Google Scholar]
- [71].Dornhofer N, Spong S, Bennewith K, et al. Connective tissue growth factor-specific monoclonal antibody therapy inhibits pancreatic tumor growth and metastasis. Cancer Res. 2006;66:5816–27. doi: 10.1158/0008-5472.CAN-06-0081. [DOI] [PubMed] [Google Scholar]
- [72].Youns M, Efferth T, Hoheisel JD. Transcript profiling identifies novel key players mediating the growth inhibitory effect of NS-398 on human pancreatic cancer cells. Eur J Pharmacol. 2011;650:170–7. doi: 10.1016/j.ejphar.2010.10.026. [DOI] [PubMed] [Google Scholar]
- [73].Jun JI, Lau LF. Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. Nat Rev Drug Discov. 2011;10:945–63. doi: 10.1038/nrd3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [74].Kular L, Pakradouni J, Kitabgi P, Laurent M, Martinerie C. The CCN family: a new class of inflammation modulators? Biochimie. 2011;93:377–88. doi: 10.1016/j.biochi.2010.11.010. [DOI] [PubMed] [Google Scholar]
- [75].Le Dreau G, Kular L, Nicot AB, et al. NOV/CCN3 upregulates CCL2 and CXCL1 expression in astrocytes through beta1 and beta5 integrins. Glia. 2010;58:1510–21. doi: 10.1002/glia.21025. [DOI] [PubMed] [Google Scholar]