Abstract
Cigarette smoking contributes to the development of cancer, and pathogenesis of other diseases. Many chemicals have been identified in cigarettes that have potent biological properties. Nicotine is especially known for its role in addiction and plays a role in other physiological effects of smoking and tobacco use. Recent studies have provided compelling evidence that, in addition to promoting cancer, nicotine also plays a pathogenic role in systems, such as the lung, kidney, heart, and liver. In many organ systems, nicotine modulates fibrosis by altering the functions of fibroblasts. Understanding the processes modulated by nicotine holds therapeutic potential and may guide future clinical and research decisions. This review discusses the role of nicotine in the general fibrogenic process that governs fibrosis and fibrosis-related diseases, focusing on the cellular mechanisms that have implications in multiple organ systems. Potential research directions for the management of nicotine-induced fibrosis, and potential clinical considerations with regard to nicotine-replacement therapy (NRT) are presented.—Jensen, K., Nizamutdinov, D., Guerrier, M., Afroze, S., Dostal, D., Glaser, S. General mechanisms of nicotine-induced fibrogenesis.
Keywords: cigarette smoking, fibroblast, reactive oxygen species, collagen, NRT
Nicotine is an addictive substance in cigarette smoke (CS), chewing tobacco, and nicotine replacement therapy (NRT), which has effects on mood, appetite, and task performance through the central nervous system (CNS) (1, 2). Nicotine is a tertiary amine consisting of a pyridine and pyrrolidine ring that is rapidly absorbed through the skin, alveoli of the lungs, and gastrointestinal epithelium prior to entering the bloodstream (2–4). Nicotine is distributed throughout the body by the circulatory system and alters the physiological processes of cells that express its receptors (2–4). The liver extensively metabolizes nicotine to form numerous metabolites. Typically, in humans, 70-80% of nicotine is metabolized to form cotinine. This is catalyzed by cytochrome P-450 2A6 (CYP2A6) and results in aldehyde formation on the fourth carbon (carbon oxidation) of the pyrrolidine ring (5, 6). Cotinine itself may undergo further transformation into trans-3′-hydroxycotinine and be excreted into the urine by the kidneys, or, similar to nicotine, stimulate its own biological effects (7).
Nicotine exerts its biological actions by binding to nicotinic acetylcholine receptors (nAChRs) located on the cell membrane. NAChRs are pentameric proteins formed by the assembly of nAChR subunits: α1–α10, β1–β4, γ, δ, and ε (8). The combination of the various subunits gives rise to nicotine's differential effects on the activation of signaling mechanisms. NAChRs are nonselective ligand-gated ion channels that allow several different positively charged ions, such as potassium, sodium, and calcium, to cross the cell membrane (8). Recently, the α7-nAChR has been the primary focus of many studies due to its pronounced effects on proliferation and the pathogenesis of several disease states (9). The binding of nicotine to the α7-nAChR receptor triggers an influx of calcium across the plasma membrane, thereby activating a downstream signaling mechanism that contributes to dysregulated growth, angiogenesis, release of growth factors, and modulation of the microenvironment (10).
Numerous studies have linked CS to a number of pathologies, such as cancer, and diseases of the cardiovascular and respiratory systems (11–13). However, it is not well understood to what extent nicotine alone contributes to these disease processes. Studies in various cell culture and animal models indicate that nicotine is a potent agent and stimulates biological responses leading to disease. For example, there is increasing evidence that nicotine, through the α7-nAChR pathway, triggers the release of growth factors that contribute to the pathogenesis of cancer (10, 14). In a nude mouse xenograft tumor model, nicotine promoted the growth of colon cancer (15). The researchers postulated that the β-adrenergic receptor-dependent growth-promoting effects of nicotine might be due to either nicotine-induced simulation of local catecholamine release by tumor cells or catecholamine release by the adrenal glands (16). These findings illustrate the fundamental concept that the release of neuroendocrine factors stimulated by nicotine may contribute to the activation of other pathogenic processes in animal models.
OVERVIEW OF FIBROGENIC MECHANISMS
Fibrosis is the pathophysiological response to chronic injury that results in excessive deposition of extracellular-matrix (ECM) proteins and scarring. In each organ system, the fibrogenic process has unique characteristics related to the function of the organ and the microenvironment created by the organ-specific epithelium. Yet key steps are common to the majority of fibrotic diseases. Fibrogenesis has been described as a stepwise process, as follows: damage to epithelial/endothelial barriers; release of transforming growth factor-β1 (TGF-β1); recruitment of inflammatory cells; production of reactive oxygen species (ROS); activation of collagen-producing cells; and ECM-dependent activation of myofibroblast cells (17). There has been long-standing interest in elucidating the process of fibrogenesis on a cellular level and identifying the environmental factors controlling the progression of fibrosis. Numerous studies over the past decade have provided compelling evidence for the involvement of nicotine in the modulation of fibroblast activation and function (18, 19). In this review, we critically discuss evidence indicating that nicotine plays a role in one or more profibrotic mechanisms. In general, collagen and related ECM molecules are the end product of these fibrogenic processes. Therefore, nicotine may stimulate the creation of a microenvironment that potentiates the activation of fibroblast function. Although the steps of fibrogenesis are often studied independently, these mechanisms are interrelated and may occur simultaneously, or in a different order. For example, some fibrotic diseases may rely more heavily on the activation of one process over another.
NICOTINE PROMOTES DAMAGE TO EPITHELIAL/ENDOTHELIAL BARRIERS
Damage to epithelial and endothelial cells plays a pivotal role in the activation of fibrogenesis. In vascular disorders, such as systemic sclerosis, damage to the endothelial bed activates the production of proinflammatory cytokines, TGF-β1 production, tissue hypoxia, and platelet aggregation, which contribute to the development and progression of fibrosis (20, 21). Chronic epithelial damage and/or apoptosis followed by tissue healing can lead to the release of inflammatory factors that trigger immune-cell infiltration and activation of local fibroblasts. Nicotine can promote both epithelial and endothelial cell damage (Fig. 1). In a recent in vivo study, nicotine directly affected the ionic homeostasis of lung and gastrointestinal tract epithelial cells and also provoked an inflammatory reaction that contributed to cellular damage (22). The researchers postulated that the epithelial damage observed in the nicotine-treated rats was due to alterations in Na+ and Cl− conductance (22). Chronic treatment with nicotine also stimulated increased eosinophil number in the lamina propria, edema, and increased damage to basal cells in the trachea (22). These effects were found in both the trachea and gastrointestinal tract. However, they were more prominent in the trachea (22). Another group demonstrated that nicotine has toxic effects in the proximal tubules of the kidney and causes to renal damage (14, 23). In a mouse model of ischemia reperfusion injury, nicotine-induced epithelial damage was associated with morphological changes in the renal epithelium, increased expression kidney injury molecule-1, and elevated creatinine levels (23). Nicotine alone was sufficient to stimulate increased expression of markers of oxidative stress and augment fibrotic and inflammatory pathways (23). In addition, there was increased expression of the classic epithelial-mesenchymal transition (EMT) markers vimentin, fibronectin, and α-smooth muscle actin (α-SMA) in the renal epithelium (14). Taken together, these studies indicate that chronic nicotine exposure may facilitate progression from acute kidney injury to chronic kidney disease (14, 23). These in vivo studies were complemented by several in vitro studies that demonstrated nicotine-induced toxicity in epithelial cells of various organs, such as the lungs and oral mucosa (24–26).
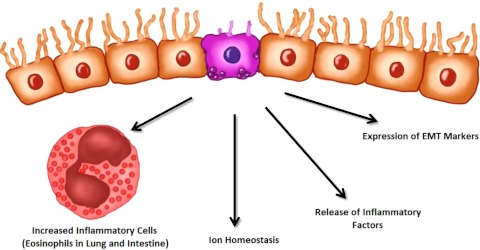
Figure 1.
Nicotine causes damage to epithelial cells. In various systems, nicotine has been shown either to directly cause damage to epithelial barriers or to potentiate the effects of injury. This results in increased release of inflammatory factors, expression of epithelial-mesenchymal transition (EMT) markers, and a change in ion homeostasis representative of epithelial dysfunction. In the lung and intestine, increased eosinophils accompany these changes.
The mechanisms regulating nicotine-induced endothelial cell damage are not well understood. A study in an in vivo model suggested that nicotine promoted the growth of atherosclerotic plaques through the activation of multiple mechanisms (27). Nicotine also regulated endothelial cell gene expression, which promoted plaque growth and angiogenesis in mice (27). Consistent with these findings, in vitro treatment of primary human coronary endothelial cells with nicotine for 24 h increased expression of nitric oxide synthase, angiotensin-I-converting enzyme, tissue-type plasminogen activator, and vascular cell adhesion molecule-1, which are factors known to promote the development of atherosclerotic plaques (28).
NICOTINE STIMULATES THE PRODUCTION AND RELEASE OF TGF-β1
TGF-β1 is a critical cytokine involved in wound healing, repair, and differentiation, in many cell types and tissues (29). TGF-β1 stimulates the production of ECM proteins (including collagen in parenchymal cells, fibroblasts, and inflammatory cells) and plays a key role in the regulation of fibrosis (30, 31). Various in vivo models of fibrosis have demonstrated that nicotine activates TGF-β1 and stimulates fibroblast functions in an autocrine fashion (Fig. 2). In a canine model of atrial fibrosis, nicotine significantly up-regulated TGF-β1 and TGF-β receptor II (TGF-βRII) expression, and stimulated atrial fibroblast proliferation and collagen deposition (18). Introduction of microRNA-133 (miR-133) and miR-590 abolished the fibrogenic effects, or, conversely, siRNA knockdown of miR-133 and miR-590 increased fibrogenesis (18). In addition, nicotine decreased expression of miR-133 and miR-590, which was negated by pretreatment with a specific α7-nAChR antagonist. These findings suggest that nicotine-induced α7-nAChR signaling was responsible for the activation of atrial fibroblasts through microRNA-dependent regulation of the TGF-β1 pathway (18). Another in vivo study demonstrated that nicotine increased the expression of TGF-β1, which was responsible for F-actin reorganization, and expression of vimentin, fibronectin, and α-SMA in the mouse kidney (14). Nicotine stimulates fibrogenesis and activates secretion of collagen from human primary liver cultures. An in vitro study on human hepatic stellate cells (HSCs) demonstrated that nicotine-activated collagen synthesis and TGF-β1 (32). In this study, healthy human liver cells treated with nicotine exhibited increased expression of a variety of proteins, including collagen I and TGF-β1 (32). A general antagonist for nAChRs abolished these effects, indicating a direct role for nicotine in the activation of the expression of profibrotic proteins (32).
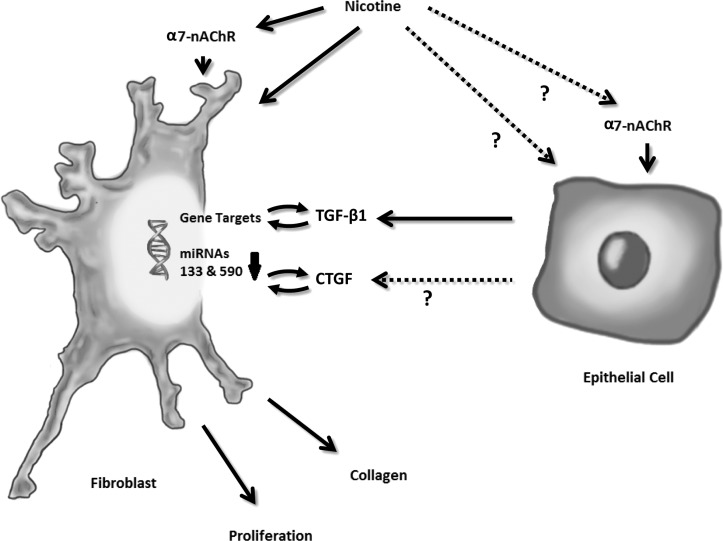
Figure 2.
Nicotine-stimulated release of TGF-β1 and CTGF. Nicotine stimulates the release of TGF-β1 either directly or through the α7-nAChR. Nicotine is postulated to trigger TGF-β1 release in other cell types, such as hepatocytes and other epithelial cells, in a similar way.
Another key cytokine known for its involvement in fibrogenesis is connective tissue growth factor (CTGF). The functions of CTGF parallel those of TGF-β1, including the stimulation of ECM remodeling and collagen synthesis. CTGF also plays a crucial role in the chemotaxis and mitogenesis of fibroblasts (33). In a recent study, nicotine increased CTGF in human gingival cells and collagen production in the periodontal ligament (34). The findings suggested that nicotine triggered direct cellular damage, induced secretion of CTGF, and activation of fibrogenesis in an autocrine fashion (34).
NICOTINE RECRUITS INFLAMMATORY CELLS
Recruitment of infiltrating inflammatory cells is a key event leading to fibrosis, and a pathogenic result of acute and chronic tissue injury. Changes in the activation status and the number of inflammatory cells play a vital role in a number of diseases associated with smoking. Therefore, it is relevant to discern the effects of nicotine on both the recruitment and activation of inflammatory cells and to identify inflammatory mediators/cytokines involved. In several systems, the effects of nicotine are controversial and appear concentration-dependent. Furthermore, this illustrates the concept that nicotine's actions are pleiotropic and variable.
Inflammatory cytokines, including the prime neutrophil chemoattractant interleukin 8 (IL-8), leukotriene B4 (LTB4), monocyte chemotactic protein-1 (MCP-1), and a number of other factors attract inflammatory cells of the neutrophil and monocyte-macrophage lineage (31). In tobacco-induced emphysema, these cells pool in the pulmonary circulation of cigarette smokers and secrete proteases that cause irreversible destruction of the alveolar walls of the lungs (35, 36). A number of in vitro and in vivo studies have described nicotine's profound effects on neutrophils during emphysema (Fig. 3). Yet the precise mechanism whereby nicotine induces emphysema is controversial. Nearly 30 years ago, Totti et al. (37) reported that nicotine was a chemotactic for neutrophils in vitro. In this study, neutrophils obtained from human blood exhibited a dose-dependent migratory response to nicotine (37). In a subsequent study, high concentrations of nicotine suppressed neutrophil chemotaxis. Interestingly, nicotine at this high dose increased neutrophil degranulation and eicosanoid generation (38). Despite contradictory findings, it is notable that both studies agreed that nicotine contributes to profound changes in neutrophil function that lead to emphysema. Recent in vitro studies have focused on evaluating the mechanisms whereby nicotine stimulates neutrophil activity. In one study, nicotine activated F-actin formation and intracellular Ca2+ release in neutrophils and was postulated to play a role in neutrophil migration and/or degranulation during pulmonary disease (39). Another in vitro study in a human myeloblast/promyelocyte cell line demonstrated that nicotine at both low and high doses caused increased elastase gene and protein expression (40). In human bronchial epithelial cells, nicotine up-regulated the IL-8 gene (and 260 other genes) downstream of Ca2+ signaling (41). Inhibition of extracellular signal-related kinase (ERK) and c-Jun N-terminal kinase (JNK) mitogen-associated protein kinases (MAPKs) significantly inhibited the nicotine-induced IL-8 response (41). Although the findings of this study have not been supported by in vivo data, another in vitro study has reported that nicotine increases IL-8 expression in gingival cells following the activation of Ca2+ signaling (42). Recently, several in vivo studies have sought to confirm that nicotine stimulated the activation of neutrophilic recruitment. In a mouse model of lung inflammation, nicotine enhanced the localization of neutrophils to inflammatory sites created by treatment with zymosan (activator of Toll-like receptor 2; ref. 43). On the other hand, nicotine alone triggered only a weak response. Together, these in vivo data explain, in part, the clinical finding that neutrophil numbers increase in the lower airways of cigarette smokers, where an elastase/antielastase imbalance is more prevalent (43). The mechanism responsible for neutrophilic recruitment is poorly understood, and it should be mentioned that nicotine's relationship to IL-8 secretion levels was reversed in an in vitro arthritis model (44). Here, nicotine decreased IL-8 secretion in response to TNF-α in synoviocytes of the joint (44). These findings clearly indicate that nicotine has pleiotropic effects depending on the cell type and disease process (44).
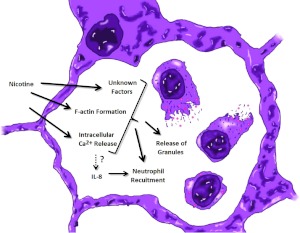
Figure 3.
Nicotine stimulates neutrophilic functions in emphysema. Nicotine increases the recruitment of neutrophils to the alveolar space and release of granules.
In addition to neutrophils, other inflammatory cells types, such as macrophages, dendritic cells (DCs), and lymphocytes, respond to nicotine. Macrophage activation by nicotine has been assessed in both in vitro and in vivo models. In one study, peritoneal macrophages from mice were treated with increasing concentrations of nicotine to evaluate its effects on superoxide anion generation, lipid peroxidation, protein oxidation, and antioxidant activity (45). Superoxide radical and lipid/protein peroxidation increased with increasing doses of nicotine (45). Conversely, the antioxidant status decreased (45). This suggests a ROS-mediated activation of an inflammatory reaction by macrophages (45). In vivo, a decrease in the antioxidants glutathione, catalase, superoxide dismutase, glutathione peroxidase, and glutathione reductase in the liver and spleen was observed after intraperitoneal administration of nicotine (45). Several similar reports published by this group of scientists have demonstrated macrophage activation by nicotine and is discussed in the following section.
Reports on the effect of nicotine on DCs are conflicting. Most studies tend to agree that nicotine has potent effects on DC regulation. However, nicotine has been observed to both activate and suppress DCs. In a mouse model of atherosclerosis, nicotine strongly activated DCs and the cell-mediated T-helper 1 (TH1) cell response (46). However, other studies have presented data demonstrating the opposite effect for nicotine (47, 48). The concentration of nicotine, and costimulation by other factors, could be responsible for these differences. Clearly, more studies are required to understand the exact mechanisms triggered by nicotine in DCs and during cell-mediated immunity.
NICOTINE ACTIVATES ROS PRODUCTION
ROS production has a significant effect on different organs and plays roles in physiological (i.e., immune cells digest foreign particles using ROS) and pathological (i.e., excess ROS can lead to cell damage) conditions (49). ROS are strong modulators of inflammatory processes in the CNS, development of atherosclerosis in vessels, stroke injury, renal epithelial damage, pulmonary cell damage, and critical factors in cancer development (50). The key step in the synthesis of ROS is the formation of superoxide by nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (51). In some organs, this function is carried out by NADPH homologs (51). Antioxidants play a role in reducing lipid peroxides throughout the body. In certain systems such as the liver, endogenous production of antioxidants is a powerful tool for defense against toxin exposure.
Nicotine induces oxidative stress in a number of cell types, including epithelial cells, macrophages, fatty liver cells, and mesangial cells (Fig. 4). Nicotine-stimulated ROS production has been linked to the damage of epithelial cells in an in vivo model of chronic kidney disease (CKD; ref. 23). In this model, nicotine exposure produced ROS through NADPH oxidase in the proximal tubules of the kidney. Chronic nicotine administration not only increased ROS by itself, but also exacerbated acute renal injury, and stimulated the progression into CKD (23). The activation of JNK and activator protein (AP-1) were shown to be involved in this process (23). Since pJNK and AP-1 gene targets are increased in smokers with CKD, nicotine is thought contribute to CKD through this mechanism (23). In an in vitro study, nicotine-induced injury and ROS in mesangial cells of the kidney via NADPH oxidase (52). In a mouse model of type 2 diabetes, nicotine worsened diabetic nephropathy through increased NADPH oxidase homologue-NOX4 activity and production of ROS (53). However, these effects could be reduced by the inclusion of exercise (53).
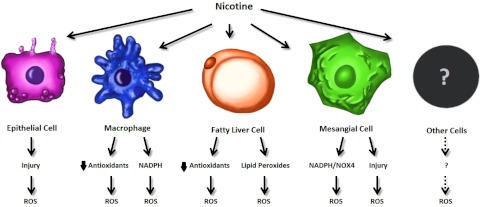
Figure 4.
Proposed mechanisms for the production of ROS in various cells types. Nicotine causes epithelial cell damage that results in the production of ROS. Nicotine also triggers the production of ROS by the up-regulation of NADPH in macrophages. Nicotine causes increases in lipid peroxides and decreased GSH in fatty liver disease. Nicotine also triggers the production of ROS through injury and up-regulation of NADPH and NOX4 in mesangial cells. Nicotine is implicated in ROS generation in other cell types and future research directions. Overlap of these mechanisms (not shown) likely occurs. Some of these mechanisms (i.e., decreased GSH) are suspected in many systems.
ROS production by inflammatory cells is an essential mediator of nicotine toxicity. Nicotine induces ROS production in macrophages in vitro and in vivo. Elevation of nicotine concentration correlated with increased lipid peroxidation and decreased antioxidant activity of peritoneal macrophages (45). These antioxidants included glutathione, catalase, superoxide dismutase, glutathione peroxidase, and glutathione reductase (45). These effects were assessed in vivo by examining the antioxidant status of the liver and spleen after intraperitoneal administration of nicotine (45). This same group demonstrated that nicotine induces activation of macrophages and superoxide-mediated oxidative damage in several studies that aimed to identify compounds that reduce the oxidative stress caused by nicotine (54, 55). Finally, nicotine increased the expression of markers for oxidative damage in the systemic circulation of rats, as well as in lymphocytes in culture (56, 57).
Nicotine has also been shown to induce ROS production in the liver. Physiologically, the liver deals with toxins that produce lipid peroxidation in the pathogenesis of several liver diseases. For example, lipid peroxidation plays a key role in alcoholic liver disease (58). Diets depleted in unsaturated fats and high in saturated fats lower the peroxidation of lipids and alcohol-induced damage (59, 60). In vivo models have provided pertinent information about nicotine's effects on lipid peroxidation. In addition, nicotine exacerbated lipid peroxidation and reduced ROS-scavenging enzymes in rats fed a high-fat diet (61). In addition, nicotine decreased the overall activity of scavenging enzymes (61). Interestingly, in vivo studies have shown that the antioxidants glutathione (GTH), S-allyl cysteine sulfoxide, and vitamin E protect against nicotine-induced lipid peroxidation in the liver, kidney and lung (62, 63). The effects of nicotine on lipid peroxide levels were further enhanced by nicotine's ability to reduce the levels of antioxidants. Nicotine by itself or in combination with ethanol decreased GSH activity in the liver and lungs (64). Furthermore, the combination of nicotine and ethanol also increased lipid peroxidation (64). Helen et al. (65) suggested that antioxidant depletion occurs via the high levels of superoxide anion and hydrogen peroxide production induced by nicotine.
NICOTINE ACTIVATES COLLAGEN-PRODUCING CELLS
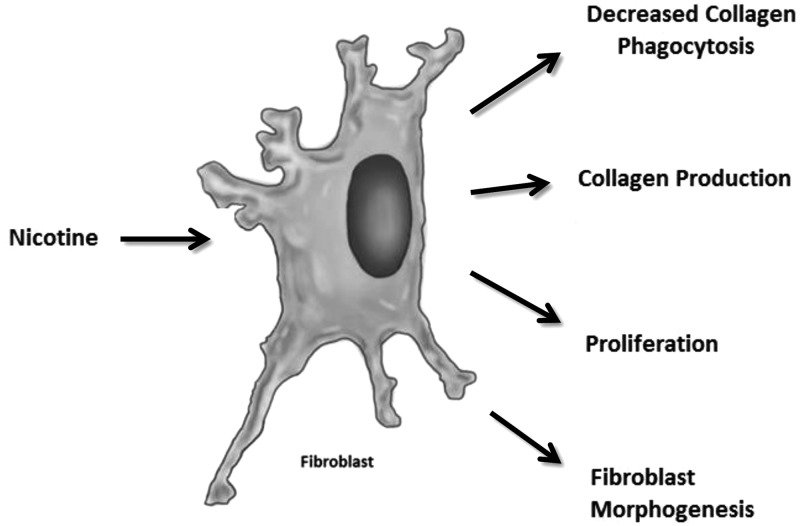
Collagen, a chief component of the ECM, is synthesized primarily by resident fibroblasts during fibrogenesis (17). In general, fibroblasts require a stimulus such as TGF-β1 to activate the secretion of collagen. In fibroblasts, increased collagen synthesis is accompanied by a rapid myofibroblastic change, which can be recognized by the elevation of α-smooth muscle actin (α-SMA), vimentin, and ECM protein expression levels (66). Several studies have shown that CS contributes to cardiac fibrosis by increasing the deposition of collagen (11, 67–70). Specifically, collagen accumulation in the atrial myocardium correlates with pack-year history of cigarette abuse and nicotine base concentration (70). A number of in vitro and in vivo studies have demonstrated that nicotine induces changes in collagen deposition via fibroblasts (Fig. 5). In vitro, stimulation with nicotine for 24 h caused a 7-fold increase in collagen III production in primary cultures of human atrial myocardial cells (70). Another group has shown in multiple reports that nicotine alters the differentiation and activation status of lung fibroblasts in vitro (19, 71–73). Exposure of embryonic lung fibroblasts in culture to nicotine for 7 d resulted in decreased expression of the key lipogenic markers parathyroid hormone-related peptide (PTHrP) and peroxisomal proliferator-activated receptor γ (PPARγ), and it increased expression of the myogenic marker α-SMA (71). This together with a decrease in triglyceride uptake suggested differentiation of normal lung fibroblasts to a myofibroblast phenotype during lung disease (71). On the signaling level, nicotine altered PTHrP-receptor binding, resulting in decreased cyclic AMP (cAMP)/PKA signaling, which contributed to the change in fibroblast phenotype (71). Lipogenic transdifferentiation into a myofibroblastic phenotype has been hypothesized to contribute to increased collagen production by fibroblasts (71). A number of reports in vivo have demonstrated that nicotine increases collagen and fibroblast activation in various systems, such as the heart, lung, gingiva, prostate, and joints (18, 34, 72, 74, 75). Collectively, it is not surprising that nicotine alters the morphogenesis and differentiation of fibroblasts, since nicotine stimulates the transdifferentiation process in other disease models, such as gastric cancer (76).
Figure 5.
Nicotine increases collagen production through its actions on fibroblasts. Nicotine increases collagen production in fibroblasts and reduces the phagocytosis of collagen. Nicotine has a proliferative effect on fibroblasts. Nicotine stimulates the morphogenesis of fibroblasts into a myofibroblastic phenotype.
The phagocytosis of collagen is another fibroblast function altered by nicotine (77). An in vitro study demonstrated that fibroblasts from patient gingiva and cultures stimulated with nicotine had reduced internalization of collagen-coated beads (77). In vivo, nicotine inhibited the phagocytosis of collagen dose dependently, and areas of oral submucosal fibrosis exhibited less phagocytosis than normal gingiva from the same patient (77).
While nicotine promotes fibroblast activation and collagen synthesis in many organs, there are some exceptions. For example, nicotine decreased collagen expression and ECM synthesis in two in vivo models of bone remodeling and tendon-bone healing (78, 79). In this setting, inflammation may be responsible for the decreased healing observed during nicotine treatment (79). Another study demonstrated in vivo that scar tissue healing was unaffected by nicotine (80). These processes are not well understood and require further investigation into the signaling mechanisms induced by nicotine.
SUMMARY AND FUTURE DIRECTIONS
The understanding of the processes activated by nicotine has considerably expanded to include the activation of fibrogenesis. The growing volume of data from different systems suggests activation of general components of the fibrogenic process and calls for more focus on the subject. To date, the data support nicotine's fibrogenic actions in cardiac, renal, lung, oral, and other organs and is currently under evaluation in the liver. A deeper understanding of the specific, and/or differential mechanisms by which nicotine stimulates the fibrosis of various organ systems, will provide a foundation for the development of novel therapeutic paradigms. The effects of nicotine that have been reported from in vitro studies should be confirmed in animal models. In particular, nicotine dramatically increases the levels of circulating catecholamines and other hormones that could have serious consequences on various organ systems. At the same time, there is much to be gained from in vitro cell culture models, from the standpoint that elucidating the intracellular signaling mechanisms and cellular processes may have utility in multiple organ systems. Since nicotine is metabolized to cotinine in vivo, studies in animal models may have a confounding contribution due to the effects of circulating cotinine and other nicotine metabolites. In addition, there is a clear need for extensive studies regarding the effects of nicotine in the kidney and vascular system, since smoking is a risk factor for diseases in these organ systems.
While smoking is prohibited for patients with a number of diseases, including primary biliary cirrhosis, little is known about nicotine's contribution to their pathogenesis. Liver cirrhosis is a disease that is potentiated by smoking. An understanding of the protective mechanisms activated in the liver during nicotine injury would be essential, especially since the liver is the site for nicotine metabolism. Furthermore, the liver has a high capacity to adapt to increased loads of oxidative stress by increasing antioxidants and wound healing. Activation of HSCs plays a key role in the progression of liver diseases. Preliminary in vitro studies in HSCs suggest that nicotine is fibrogenic and plays a role in HSC activation through nAChRs (32). The ability of the liver to adapt to injury and activate repair mechanisms to return to its normal function is not shared by all organs. For example, the lung easily reacts to nicotine to produce the irreversible pathology seen in emphysema.
CLINICAL PERSPECTIVES
Cigarette smoking is responsible for over 435,000 deaths each year in the United States (81). Cigarette smoke contains many biologically active components. However, nicotine is responsible for its addictive properties, and an estimated 21% of Americans smoke (81, 82). NRT is the most widely used treatment for promoting smoking cessation (83). NRT has been approved by the Food and Drug Administration and is considered a safe and effective aid in quitting smoking. Epidemiological studies of the nicotine patch primarily reported non-life-threatening side effects (84, 85). At first-glance, this appears to contradict the large number of cell culture and animal studies reporting nicotine's disease-causing mechanisms. However, consideration should be given to the fact that the long-term nicotine patch utilization has not been studied. That being said there is a critical need for epidemiological research to evaluate long-term NRT usage. Clinical investigators should monitor patients using NRT for extended periods of time, especially if they are at risk for cancer or fibrotic/inflammatory diseases.
Acknowledgments
Portions of this work were supported by the Scott and White Hospital, Department of Internal Medicine; a Scott and White Research Advancement award; a U.S. Department of Veteran's Affairs Career Development Award-2; and a U.S. National Institutes of Health RO1 grant (DK081442) to S.G.
The authors thank Dr. Gianfranco Alpini for his kind suggestions and comments on the manuscript.
Footnotes
- α-SMA
- α-smooth muscle actin
- CNS
- central nervous system
- CS
- cigarette smoke
- CTGF
- connective tissue growth factor
- C
- dendritic cell
- ECM
- extracellular matrix
- HSC
- hepatic stellate cell
- IL
- interleukin
- JNK
- c-Jun N-terminal kinase
- miR
- microRNA
- nAChR
- nicotinic acetylcholine receptor
- NADPH
- nicotinamide adenine dinucleotide phosphate
- NRT
- nicotine-replacement therapy
- ROS
- reactive oxygen species
- TGF-βI
- transforming growth factor-βI
- TGF-βRII
- transforming growth factor-β receptor II
REFERENCES
- 1. Cardenas L., Tremblay L. K., Naranjo C. A., Herrmann N., Zack M., Busto U. E. (2002) Brain reward system activity in major depression and comorbid nicotine dependence. J. Pharmacol. Exp. Ther. 302, 1265–1271 [DOI] [PubMed] [Google Scholar]
- 2. Benowitz N. L. (1988) Drug therapy. Pharmacologic aspects of cigarette smoking and nicotine addition. N. Engl. J. Med. 319, 1318–1330 [DOI] [PubMed] [Google Scholar]
- 3. Cahill K., Stead L. F., Lancaster T. (2011) Nicotine receptor partial agonists for smoking cessation. Cochrane Database Syst. Rev. CD006103. [DOI] [PubMed] [Google Scholar]
- 4. Hukkanen J., Jacob P., 3rd, Benowitz N. L. (2005) Metabolism and disposition kinetics of nicotine. Pharmacol. Rev. 57, 79–115 [DOI] [PubMed] [Google Scholar]
- 5. Hucker H. B., Gillette J. R., Brodie B. B. (1960) Enzymatic pathway for the formation of cotinine, a major metabolite of nicotine in rabbit liver. J. Pharmacol. Exp. Ther. 129, 94–100 [PubMed] [Google Scholar]
- 6. Ejaz S., Lim C. W. (2005) Toxicological overview of cigarette smoking on angiogenesis. Environ. Toxicol. Pharmacol. 20, 335–344 [DOI] [PubMed] [Google Scholar]
- 7. Nakajima M., Yamamoto T., Nunoya K., Yokoi T., Nagashima K., Inoue K., Funae Y., Shimada N., Kamataki T., Kuroiwa Y. (1996) Role of human cytochrome P4502A6 in C-oxidation of nicotine. Drug Metab. Dispos. 24, 1212–1217 [PubMed] [Google Scholar]
- 8. Millar N. S., Harkness P. C. (2008) Assembly and trafficking of nicotinic acetylcholine receptors (review). Mol. Membr. Biol. 25, 279–292 [DOI] [PubMed] [Google Scholar]
- 9. Egleton R. D., Brown K. C., Dasgupta P. (2008) Nicotinic acetylcholine receptors in cancer: multiple roles in proliferation and inhibition of apoptosis. Trends Pharmacol. Sci. 29, 151–158 [DOI] [PubMed] [Google Scholar]
- 10. Schuller H. M. (2009) Is cancer triggered by altered signalling of nicotinic acetylcholine receptors? Nat. Rev. Cancer 9, 195–205 [DOI] [PubMed] [Google Scholar]
- 11. Benowitz N. L., Gourlay S. G. (1997) Cardiovascular toxicity of nicotine: implications for nicotine replacement therapy. J. Am. Coll. Cardiol. 29, 1422–1431 [DOI] [PubMed] [Google Scholar]
- 12. Yang I. A., Relan V., Wright C. M., Davidson M. R., Sriram K. B., Savarimuthu Francis S. M., Clarke B. E., Duhig E. E., Bowman R. V., Fong K. M. (2011) Common pathogenic mechanisms and pathways in the development of COPD and lung cancer. Expert Opin. Therap. Targets 15, 439–456 [DOI] [PubMed] [Google Scholar]
- 13. Costa F., Soares R. (2009) Nicotine: a pro-angiogenic factor. Life Sci. 84, 785–790 [DOI] [PubMed] [Google Scholar]
- 14. Arany I., Reed D. K., Grifoni S. C., Chandrashekar K., Booz G. W., Juncos L. A. (2012) A novel U-STAT3-dependent mechanism mediates the deleterious effects of chronic nicotine exposure on renal injury. Am. J. Physiol. Renal Physiol 302, F722–F729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Wong H. P., Yu L., Lam E. K., Tai E. K., Wu W. K., Cho C. H. (2007) Nicotine promotes colon tumor growth and angiogenesis through beta-adrenergic activation. Toxicol. Sci. 97, 279–287 [DOI] [PubMed] [Google Scholar]
- 16. Haass M., Kubler W. (1997) Nicotine and sympathetic neurotransmission. Cardiovasc. Drugs Ther. 10, 657–665 [DOI] [PubMed] [Google Scholar]
- 17. Kisseleva T., Brenner D. A. (2008) Mechanisms of fibrogenesis. Exp. Biol. Med. (Maywood) 233, 109–122 [DOI] [PubMed] [Google Scholar]
- 18. Shan H., Zhang Y., Lu Y., Zhang Y., Pan Z., Cai B., Wang N., Li X., Feng T., Hong Y., Yang B. (2009) Downregulation of miR-133 and miR-590 contributes to nicotine-induced atrial remodelling in canines. Cardiovasc. Res. 83, 465–472 [DOI] [PubMed] [Google Scholar]
- 19. Krebs M., Sakurai R., Torday J. S., Rehan V. K. (2010) Evidence for in vivo nicotine-induced alveolar interstitial fibroblast-to-myofibroblast transdifferentiation. Exp. Lung. Res. 36, 390–398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Lafyatis R. (2006) Targeting fibrosis in systemic sclerosis. Endocr. Metab. Immune Disord. Drug Targets 6, 395–400 [DOI] [PubMed] [Google Scholar]
- 21. Jain M., Varga J. (2006) Bosentan for the treatment of systemic sclerosis-associated pulmonary arterial hypertension, pulmonary fibrosis and digital ulcers. Expert Opin. Pharmacother. 7, 1487–1501 [DOI] [PubMed] [Google Scholar]
- 22. Roomans G. M., Vanthanouvong V., Dragomir A., Kozlova I., Wroblewski R. (2002) Effects of nicotine on intestinal and respiratory epithelium. J. Submicrosc. Cytol. Pathol. 34, 381–388 [PubMed] [Google Scholar]
- 23. Arany I., Grifoni S., Clark J. S., Csongradi E., Maric C., Juncos L. A. (2011) Chronic nicotine exposure exacerbates acute renal ischemic injury. Am. J. Physiol. Renal Physiol. 301, F125–F133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Zia S., Ndoye A., Nguyen V. T., Grando S. A. (1997) Nicotine enhances expression of the alpha 3, alpha 4, alpha 5, and alpha 7 nicotinic receptors modulating calcium metabolism and regulating adhesion and motility of respiratory epithelial cells. Res. Commun. Mol. Pathol. Pharmacol. 97, 243–262 [PubMed] [Google Scholar]
- 25. Prabhulkar S., Li C. Z. (2010) Assessment of oxidative DNA damage and repair at single cellular level via real-time monitoring of 8-OHdG biomarker. Biosens. Bioelectron. 26, 1743–1749 [DOI] [PubMed] [Google Scholar]
- 26. Caldeira E. J., Carvalho C. A., Padovani C. R., Camilli J. A., Garcia P. J., Cagnon V. H. (2007) Morphological alterations in the epithelium of the oral mucosa of rats (Rattus norvegicus) submitted to long-term systemic nicotine treatment. Arch. Oral. Biol. 52, 83–89 [DOI] [PubMed] [Google Scholar]
- 27. Heeschen C., Jang J. J., Weis M., Pathak A., Kaji S., Hu R. S., Tsao P. S., Johnson F. L., Cooke J. P. (2001) Nicotine stimulates angiogenesis and promotes tumor growth and atherosclerosis. Nat. Med. 7, 833–839 [DOI] [PubMed] [Google Scholar]
- 28. Zhang S., Day I., Ye S. (2001) Nicotine induced changes in gene expression by human coronary artery endothelial cells. Atherosclerosis 154, 277–283 [DOI] [PubMed] [Google Scholar]
- 29. Sporn M. B., Roberts A. B., Wakefield L. M., Assoian R. K. (1986) Transforming growth factor-beta: biological function and chemical structure. Science 233, 532–534 [DOI] [PubMed] [Google Scholar]
- 30. Jensen K., Marzioni M., Munshi M. K., Afroze S., Alpini G., Glaser S. (2012) Autocrine regulation of biliary pathology by activated cholangiocytes. Am. J. Physiol. Gastrointest. Liver Physiol. 302, G473–G483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bataller R., Brenner D. A. (2005) Liver fibrosis. J. Clin. Invest. 115, 209–218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Soeda J., Morgan M., McKee C., Mouralidarane A., Lin C., Roskams T., Oben J. A. (2012) Nicotine induces fibrogenic changes in human liver via nicotinic acetylcholine receptors expressed on hepatic stellate cells. Biochem. Biophys. Res. Commun. 417, 17–22 [DOI] [PubMed] [Google Scholar]
- 33. Frazier K., Williams S., Kothapalli D., Klapper H., Grotendorst G. R. (1996) Stimulation of fibroblast cell growth, matrix production, and granulation tissue formation by connective tissue growth factor. J. Invest. Dermatol. 107, 404–411 [DOI] [PubMed] [Google Scholar]
- 34. Takeuchi H., Kubota S., Murakashi E., Zhou Y., Endo K., Ng P. S., Takigawa M., Numabe Y. (2010) Nicotine-induced CCN2: from smoking to periodontal fibrosis. J. Dent. Res. 89, 34–39 [DOI] [PubMed] [Google Scholar]
- 35. MacNee W., Wiggs B., Belzberg A. S., Hogg J. C. (1989) The effect of cigarette smoking on neutrophil kinetics in human lungs. N. Engl. J. Med. 321, 924–928 [DOI] [PubMed] [Google Scholar]
- 36. Sharafkhaneh A., Hanania N. A., Kim V. (2008) Pathogenesis of emphysema: from the bench to the bedside. Proc. Am. Thorac. Soc. 5, 475–477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Totti N., 3rd, McCusker K. T., Campbell E. J., Griffin G. L., Senior R. M. (1984) Nicotine is chemotactic for neutrophils and enhances neutrophil responsiveness to chemotactic peptides. Science 223, 169–171 [DOI] [PubMed] [Google Scholar]
- 38. Seow W. K., Thong Y. H., Nelson R. D., MacFarlane G. D., Herzberg M. C. (1994) Nicotine-induced release of elastase and eicosanoids by human neutrophils. Inflammation 18, 119–127 [DOI] [PubMed] [Google Scholar]
- 39. Ryder M. I. (1994) Nicotine effects on neutrophil F-actin formation and calcium release: implications for tobacco use and pulmonary diseases. Exp. Lung Res. 20, 283–296 [DOI] [PubMed] [Google Scholar]
- 40. Armstrong L. W., Rom W. N., Martiniuk F. T., Hart D., Jagirdar J., Galdston M. (1996) Nicotine enhances expression of the neutrophil elastase gene and protein in a human myeloblast/promyelocyte cell line. Am. J. Respir. Crit. Care Med. 154, 1520–1524 [DOI] [PubMed] [Google Scholar]
- 41. Tsai J. R., Chong I. W., Chen C. C., Lin S. R., Sheu C. C., Hwang J. J. (2006) Mitogen-activated protein kinase pathway was significantly activated in human bronchial epithelial cells by nicotine. DNA Cell Biol. 25, 312–322 [DOI] [PubMed] [Google Scholar]
- 42. Kashiwagi Y., Yanagita M., Kojima Y., Shimabukuro Y., Murakami S. (2012) Nicotine up-regulates IL-8 expression in human gingival epithelial cells following stimulation with IL-1β or P. gingivalis lipopolysaccharide via nicotinic acetylcholine receptor signalling. Arch. Oral. Biol. 57, 483–490 [DOI] [PubMed] [Google Scholar]
- 43. Nowak D., Ruta U., Piasecka G. (1990) Nicotine increases human polymorphonuclear leukocytes chemotactic response—a possible additional mechanism of lung injury in cigarette smokers. Exp. Pathol. 39, 37–43 [DOI] [PubMed] [Google Scholar]
- 44. Zhou Y., Zuo X., Li Y., Wang Y., Zhao H., Xiao X. (2012) Nicotine inhibits tumor necrosis factor-α induced IL-6 and IL-8 secretion in fibroblast-like synoviocytes from patients with rheumatoid arthritis. Rheumatol. Int. 32, 97–104 [DOI] [PubMed] [Google Scholar]
- 45. Mahapatra S. K., Das S., Bhattacharjee S., Gautam N., Majumdar S., Roy S. (2009) In vitro nicotine-induced oxidative stress in mice peritoneal macrophages: a dose-dependent approach. Toxicol. Mech. Methods 19, 100–108 [DOI] [PubMed] [Google Scholar]
- 46. Aicher A., Heeschen C., Mohaupt M., Cooke J. P., Zeiher A. M., Dimmeler S. (2003) Nicotine strongly activates dendritic cell-mediated adaptive immunity: potential role for progression of atherosclerotic lesions. Circulation 107, 604–611 [DOI] [PubMed] [Google Scholar]
- 47. Yanagita M., Kobayashi R., Kojima Y., Mori K., Murakami S. (2012) Nicotine modulates the immunological function of dendritic cells through peroxisome proliferator-activated receptor-gamma upregulation. Cell. Immunol. 274, 26–33 [DOI] [PubMed] [Google Scholar]
- 48. Nouri-Shirazi M., Guinet E. (2003) Evidence for the immunosuppressive role of nicotine on human dendritic cell functions. Immunology 109, 365–373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Ciobica A., Hritcu L., Artenie V., Padurariu M. (2009) [The effects of some cholinergic drugs on cognitive processes and oxidative stress in rat]. Rev. Med. Chir. Soc. Med. Nat. Iasi 113, 832–837 [PubMed] [Google Scholar]
- 50. Hemnani T., Parihar M. S. (1998) Reactive oxygen species and oxidative DNA damage. Indian J. Physiol. Pharmacol. 42, 440–452 [PubMed] [Google Scholar]
- 51. Geiszt M., Kopp J. B., Varnai P., Leto T. L. (2000) Identification of renox, an NAD(P)H oxidase in kidney. Proc. Natl. Acad. Sci. U. S. A. 97, 8010–8014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Jaimes E. A., Tian R. X., Raij L. (2007) Nicotine: the link between cigarette smoking and the progression of renal injury? Am. J. Physiol. Heart Circ. Physiol. 292, H76–H82 [DOI] [PubMed] [Google Scholar]
- 53. Hua P., Feng W., Ji S., Raij L., Jaimes E. A. (2010) Nicotine worsens the severity of nephropathy in diabetic mice: implications for the progression of kidney disease in smokers. Am. J. Physiol. Renal Physiol. 299, F732–F739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Kar Mahapatra S., Chakraborty S. P., Majumdar S., Bag B. G., Roy S. (2009) Eugenol protects nicotine-induced superoxide mediated oxidative damage in murine peritoneal macrophages in vitro. Eur. J. Pharmacol. 623, 132–140 [DOI] [PubMed] [Google Scholar]
- 55. Mahapatra S. K., Chakraborty S. P., Das S., Roy S. (2009) Methanol extract of Ocimum gratissimum protects murine peritoneal macrophages from nicotine toxicity by decreasing free radical generation, lipid and protein damage and enhances antioxidant protection. Oxid. Med. Cell. Longev. 2, 222–230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Sudheer A. R., Muthukumaran S., Kalpana C., Srinivasan M., Menon V. P. (2007) Protective effect of ferulic acid on nicotine-induced DNA damage and cellular changes in cultured rat peripheral blood lymphocytes: a comparison with N-acetylcysteine. Toxicol. In Vitro 21, 576–585 [DOI] [PubMed] [Google Scholar]
- 57. Sudheer A. R., Muthukumaran S., Devipriya N., Devaraj H., Menon V. P. (2008) Influence of ferulic acid on nicotine-induced lipid peroxidation, DNA damage and inflammation in experimental rats as compared to N-acetylcysteine. Toxicology 243, 317–329 [DOI] [PubMed] [Google Scholar]
- 58. Muriel P. (2009) Role of free radicals in liver diseases. Hepatol. Int. 3, PMC2790593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Morimoto M., Zern M. A., Hagbjork A. L., Ingelman-Sundberg M., French S. W. (1994) Fish oil, alcohol, and liver pathology: role of cytochrome P450 2E1. Proc. Soc. Exp. Biol. Med. 207, 197–205 [DOI] [PubMed] [Google Scholar]
- 60. Nanji A. A., Zhao S., Sadrzadeh S. M., Dannenberg A. J., Tahan S. R., Waxman D. J. (1994) Markedly enhanced cytochrome P450 2E1 induction and lipid peroxidation is associated with severe liver injury in fish oil-ethanol-fed rats. Alcohol Clin. Exp. Res. 18, 1280–1285 [DOI] [PubMed] [Google Scholar]
- 61. Ashakumary L., Vijayammal P. L. (1996) Effect of nicotine on antioxidant defence mechanisms in rats fed a high-fat diet. Pharmacology 52, 153–158 [DOI] [PubMed] [Google Scholar]
- 62. Dey S. K., Roy S. (2010) Role of reduced glutathione in the amelioration of nicotine-induced oxidative stress. Bull. Environ. Contam. Toxicol. 84, 385–389 [DOI] [PubMed] [Google Scholar]
- 63. Helen A., Krishnakumar K., Vijayammal P. L., Augusti K. T. (2003) A comparative study of antioxidants S-allyl cysteine sulfoxide and vitamin E on the damages induced by nicotine in rats. Pharmacology 67, 113–117 [DOI] [PubMed] [Google Scholar]
- 64. Husain K., Scott B. R., Reddy S. K., Somani S. M. (2001) Chronic ethanol and nicotine interaction on rat tissue antioxidant defense system. Alcohol 25, 89–97 [DOI] [PubMed] [Google Scholar]
- 65. Helen A., Krishnakumar K., Vijayammal P. L., Augusti K. T. (2000) Antioxidant effect of onion oil (Allium cepa. Linn) on the damages induced by nicotine in rats as compared to alpha-tocopherol. Toxicol. Lett. 116, 61–68 [DOI] [PubMed] [Google Scholar]
- 66. Wynn T. A. (2007) Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J. Clin. Invest. 117, 524–529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Yashima M., Ohara T., Cao J. M., Kim Y. H., Fishbein M. C., Mandel W. J., Chen P. S., Karagueuzian H. S. (2000) Nicotine increases ventricular vulnerability to fibrillation in hearts with healed myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 278, H2124–H2133 [DOI] [PubMed] [Google Scholar]
- 68. Hayashi H., Omichi C., Miyauchi Y., Mandel W. J., Lin S. F., Chen P. S., Karagueuzian H. S. (2003) Age-related sensitivity to nicotine for inducible atrial tachycardia and atrial fibrillation. Am. J. Physiol. Heart Circ. Physiol. 285, H2091–H2098 [DOI] [PubMed] [Google Scholar]
- 69. Goette A., Lendeckel U., Klein H. U. (2002) Signal transduction systems and atrial fibrillation. Cardiovasc. Res. 54, 247–258 [DOI] [PubMed] [Google Scholar]
- 70. Goette A., Lendeckel U., Kuchenbecker A., Bukowska A., Peters B., Klein H. U., Huth C., Rocken C. (2007) Cigarette smoking induces atrial fibrosis in humans via nicotine. Heart 93, 1056–1063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Rehan V. K., Wang Y., Sugano S., Romero S., Chen X., Santos J., Khazanchi A., Torday J. S. (2005) Mechanism of nicotine-induced pulmonary fibroblast transdifferentiation. Am. J. Physiol. Lung Cell. Mol. Physiol. 289, L667–L676 [DOI] [PubMed] [Google Scholar]
- 72. Rehan V. K., Wang Y., Sugano S., Santos J., Patel S., Sakurai R., Boros L. G., Lee W. P., Torday J. S. (2007) In utero nicotine exposure alters fetal rat lung alveolar type II cell proliferation, differentiation, and metabolism. Am. J. Physiol. Lung Cell. Mol. Physiol. 292, L323–L333 [DOI] [PubMed] [Google Scholar]
- 73. Rehan V. K., Sakurai R., Wang Y., Santos J., Huynh K., Torday J. S. (2007) Reversal of nicotine-induced alveolar lipofibroblast-to-myofibroblast transdifferentiation by stimulants of parathyroid hormone-related protein signaling. Lung 185, 151–159 [DOI] [PubMed] [Google Scholar]
- 74. Sekhon H. S., Keller J. A., Proskocil B. J., Martin E. L., Spindel E. R. (2002) Maternal nicotine exposure upregulates collagen gene expression in fetal monkey lung. Association with alpha7 nicotinic acetylcholine receptors. Am. J. Respir. Cell Mol. Biol. 26, 31–41 [DOI] [PubMed] [Google Scholar]
- 75. Carvalho C. A., Pissolato M., Candido E. M., Liberti E. A., Cagnon V. H. (2012) Morphological alterations in the prostate stroma of rats submitted to chronic nicotine treatment. Microsc. Res. Tech. 75, 46–53 [DOI] [PubMed] [Google Scholar]
- 76. Shin V. Y., Jin H. C., Ng E. K., Sung J. J., Chu K. M., Cho C. H. (2010) Activation of 5-lipoxygenase is required for nicotine-mediated epithelial-mesenchymal transition and tumor cell growth. Cancer Lett. 292, 237–245 [DOI] [PubMed] [Google Scholar]
- 77. Shieh D. H., Chiang L. C., Lee C. H., Yang Y. H., Shieh T. Y. (2004) Effects of arecoline, safrole, and nicotine on collagen phagocytosis by human buccal mucosal fibroblasts as a possible mechanism for oral submucous fibrosis in Taiwan. J. Oral. Pathol. Med. 33, 581–587 [DOI] [PubMed] [Google Scholar]
- 78. Yamano S., Berley J. A., Kuo W. P., Gallucci G. O., Weber H. P., Sukotjo C. (2010) Effects of nicotine on gene expression and osseointegration in rats. Clin. Oral Implants Res. 21, 1353–1359 [DOI] [PubMed] [Google Scholar]
- 79. Galatz L. M., Silva M. J., Rothermich S. Y., Zaegel M. A., Havlioglu N., Thomopoulos S. (2006) Nicotine delays tendon-to-bone healing in a rat shoulder model. J. Bone Joint Surg. Am. 88, 2027–2034 [DOI] [PubMed] [Google Scholar]
- 80. Biondo-Simoes Mde L., Tetilla M. R., Biondo-Simoes R., Martin M. M., Repka J. C., Zanato D. (2009) [The influence of nicotine on the density of collagen in cutaneous scars in rats]. Rev. Col. Bras. Cir. 36, 425–430 [DOI] [PubMed] [Google Scholar]
- 81. Centers for Disease Control and Prevention (2006) Tobacco use among adults—United States, 2005. MMWR Morb. Mortal. Wkly. Rep. 55, 1145–1148 [PubMed] [Google Scholar]
- 82. Grief S. N. (2011) Nicotine dependence: health consequences, smoking cessation therapies, and pharmacotherapy. Prim. Care 38, 23–39, v [DOI] [PubMed] [Google Scholar]
- 83. Molyneux A. (2004) Nicotine replacement therapy. BMJ 328, 454–456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Mills E. J., Wu P., Lockhart I., Wilson K., Ebbert J. O. (2010) Adverse events associated with nicotine replacement therapy (NRT) for smoking cessation. A systematic review and meta-analysis of one hundred and twenty studies involving 177,390 individuals. Tob. Induc. Dis. 8, 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Murray R. P., Connett J. E., Zapawa L. M. (2009) Does nicotine replacement therapy cause cancer? Evidence from the Lung Health Study. Nicotine Tob. Res. 11, 1076–1082 [DOI] [PMC free article] [PubMed] [Google Scholar]