Abstract
Dystrophic epidermolysis bullosa is an inherited bullous dermatosis caused by the COL7A1 gene mutation in autosomal dominant or recessive mode. COL7A1 gene encodes type VII collagen – the main component of the anchoring fibrils at the dermal–epidermal junction. Besides the 730 mutations reported, we identified two novel COL7A1 gene mutations in a Chinese family, which caused recessive dystrophic epidermolysis bullosa (RDEB). The diagnosis was established histopathologically and ultrastructurally. After genomic DNA extraction from the peripheral blood sample of all subjects (5 pedigree members and 136 unrelated control individuals), COL7A1 gene screening was performed by polymerase chain reaction amplification and direct DNA sequencing of the whole coding exons and flanking intronic regions. Genetic analysis of the COL7A1 gene in affected individuals revealed compound heterozygotes with identical novel mutations. The maternal mutation is a 2-bp deletion at exon 8 (c.1006_1007delCA), leading to a subsequent reading frame-shift and producing a premature termination codon located 48 amino acids downstream in exon 9 (p.Q336EfsX48), consequently resulting in the truncation of 2561 amino acids downstream. This was only present in two affected brothers, but not in the other unaffected family members. The paternal mutation is a 1-bp deletion occurring at the first base of intron 65 (c.IVS5568+1delG) that deductively changes the strongly conserved GT dinucleotide at the 5′ donor splice site, results in subsequent reading-through into intron 65, and creates a stop codon immediately following the amino acids encoded by exon 65 (GTAA→TAA). This is predicted to produce a truncated protein lacking of 1089 C-terminal amino acids downstream. The latter mutation was found in all family members except one of the two unaffected sisters. Both mutations were observed concurrently only in the two affected brothers. Neither mutation was discovered in 136 unrelated Chinese control individuals. This study reveals novel disease-causing mutations in the COL7A1 gene.
Introduction
Dystrophic epidermolysis bullosa (DEB) is an inherited bullous dermatosis characterized by high skin fragility. The phenotype of this disease is variable [1]. Common symptoms include skin blisters, scars, pseudosyndactyly, and onychodystrophy caused by slight trauma. In severe patients, mutilating scarring of the hands and feet, and squamous cell carcinoma may develop, possibly inducing death.
DEB is the consequence of COL7A1 gene (MIM *120120) mutation in autosomal dominant (DDEB, MIM#131750) or recessive (RDEB, MIM#226600) fashion [2]. COL7A1 gene encodes type VII collagen, the main component of the anchoring fibrils at the sub-lamina densa of the dermo-epidermal junction [3].
More than 730 mutations of COL7A1 gene have been reported, demonstrating the high heterogeneity associated with the disease [4]. Here we report two novel mutations in the COL7A1 gene resulting in RDEB in two brothers from a Chinese family.
Materials and Methods
Ethics Statement
All the analyses have been approved by the ethics committee of the Sichuan Academy of Medical Sciences & Sichuan Provincial People’s Hospital, and all clinical investigations have been conducted according to the principles expressed in the Declaration of Helsinki, with patients’ written informed consent.
Patients and Clinic
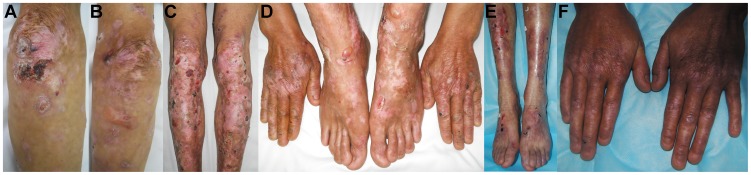
The proband was a 42-year-old man suffering from blisters, blood blisters, and scarring formation primarily on the dorsal side of the hands, feet, elbows, tibialis, and knees after slight trauma since birth. Some hypopigmentation could also be seen in the lesion area. No lesions were found on the trunk or mucous membrane. The nails on his fingers and toes never grew (Fig. 1A, B, C, and D). There was no extracutaneous involvement. The proband’s 33-year-old younger brother (Fig. 1E, F) – but not his two sisters, his father, and all relatives – had the similar experience and lesions (Fig. 2). The proband’s parents were not consanguine. His mother, who died of leukemia, was reported to have no bullous dermatosis in her lifetime.
Figure 1. Clinical manifestations of the two RDEB patients.
Blisters, blood blisters, scarring formation, and hypopigmentation on the dorsal side of the elbows (A, B), tibialis, knees (C), hands, and feet could be seen in the proband. Nails of fingers and toes were all absent (D). Similar lesions could be seen in the proband’s younger brother (E, F).
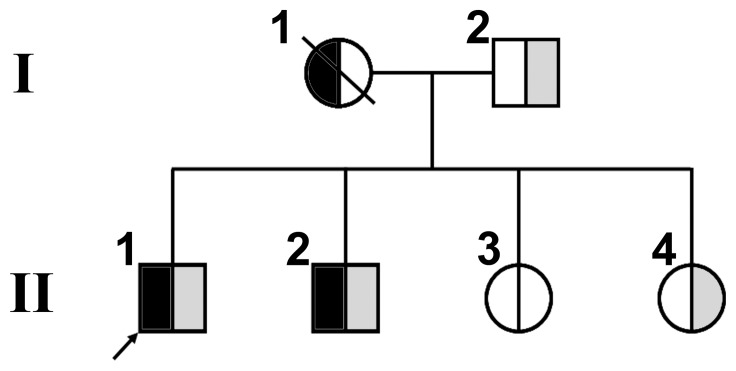
Figure 2. Pedigree of the RDEB family.
The segregation of the mutations of c.1006_1007delCA on exon 8 (half black symbols) and c.IVS5568+1delG at the donor splicing site of intron 65 (half gray symbols) on the COL7A1 gene is shown. The arrow refers to the proband.
Biopsy of the proband was performed for histopathologic examination, direct immunofluorescence assay, and transmission electron microscopy. Blood routine and serum biochemical assay of the two patients, their unaffected father, and two sisters were also investigated.
Mutation Screening
Genome DNA was extracted from the peripheral leucocytes from EDTA-anticoagulated peripheral blood samples of the two patients, their unaffected father, and two sisters, followed by polymerase chain reaction (PCR) with 72 pairs of primers in two patients. PCR primers were designed to include flanking intronic sequences of each exon according to published protocols [5]. Specifically, the two pairs of primer with which the mutations were found were: (L) 5′ CTTGGCTGGGCAAGATAAAG 3′ and (R) 5′CAGCCCACATCTCTCACTCA 3′ for exon 8, and (L) 5′ATGCAGGCACACCCTTAGAC 3′ and (R) 5′ GGGTCAGAGGTTGCAGAAAA 3′ for exon 65 and intron 65. For PCR amplification, 2.0 µl (25 ng/µl) of genome DNA was used as template in 20 µl of reaction mixture containing 1.6 µl dNTPs (2.5 mmol/L), 1.2 µl Mg2+ (25 mmol/L), 1 µl of each primer (10 µmol/L), and 0.4 µl AmpliTaq Gold polymerase (5 U/µl). PCR conditions were 95°C for 5 min followed by 95°C for 30 s, 57–60°C for 30 s, and 72°C for 1 min (35 cycles), and finally extended at 72°C for 10 min. The PCR products, examined by 2% agarose gel electrophoresis, were 362 bp (for exon 8) and 410 bp (for exon 65) in size, respectively.
The PCR products were purified by using QIA quick Gel Extraction Kit (QIAGEN, Valencia, CA, USA), and sequenced by BigDye Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems) on ABI 3100 Genetic Analyzer (Applied Biosystems). The mutation locations were determined by BLAST comparing the results with the original sequence in GenBank.
Mutation Verification
To verify the novel mutations found in the two patients, genome DNA of their remaining three family members and 136 normal controls unrelated to this family and disease were also amplified with primers for exon 8 and exon 65 and sequenced.
Results
Clinical Findings
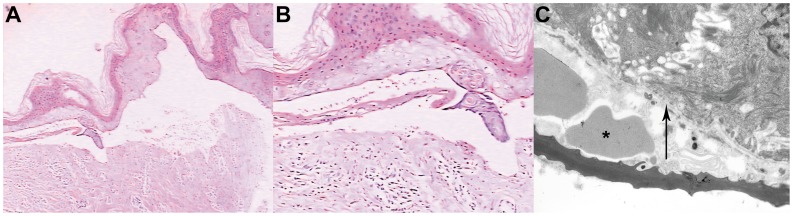
Histopathologic examination of the proband indicated a keratinocyte degeneration and necrosis. subepidermal bulla with infiltration of a few inflammatory cells (Fig. 3A, B). Direct immunofluorescence assay showed negative stain of IgG, IgA, IgM, complement C3, and C4 at the dermo-epidermal junction (data not shown). Under transmission electron microscope, the split was located at the sub-lamina densa where some red blood cells could be seen (Fig. 3C). The diagnosis of RDEB (generalized, other type) was made according to Fine JD et al. [6]. The results of blood routine and serum biochemistry examination of the two patients, their two sisters, and father were all within the normal range.
Figure 3. Histopathologic and ultrastructural features of the RDEB proband’s skin lesion.
Hematoxylin and eosin stain (H&E) of the skin lesion indicated a keratinocytes degeneration and necrosis, subepidermal bulla with infiltration of a few inflammatory cells (A, magnification×40; B×100). Under transmission electron microscope, the split was located at the sub-lamina densa where some red blood cells could be seen (asterisk) (C). The arrow refers to the lamina densa. Magnification×10000.
Sequencing Analysis
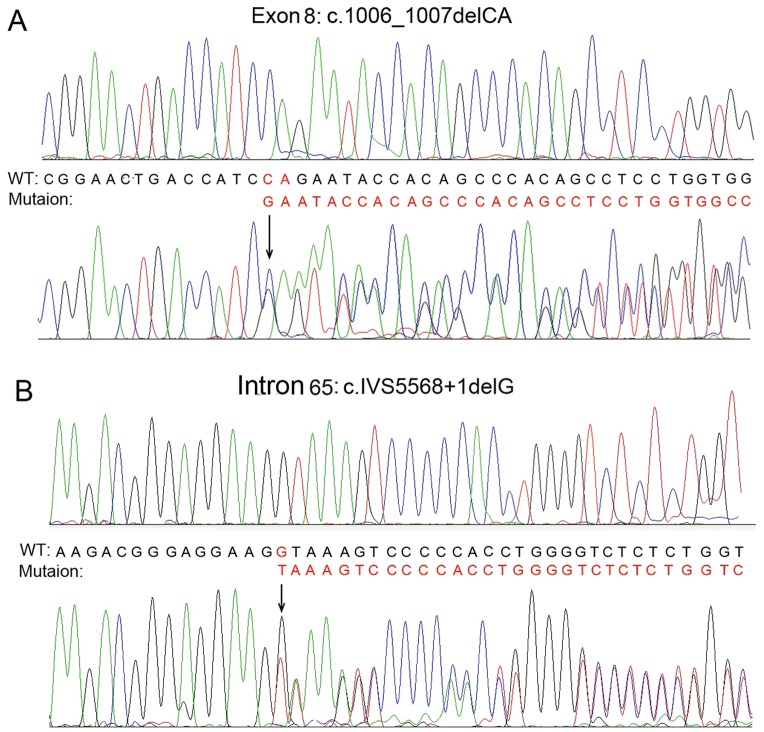
DNA sequencing of exons and flanking introns of the two patients disclosed that both the brothers were compound heterozygotes with identical mutations. One of the two mutations found in the two patients, but not their father and two sisters, was located at exon 8 with c.1006_1007delCA (Fig. 4A). The other novel mutation in the other chain of the alleles in the two patients, their younger sister, and father was c.IVS5568+1delG located at position -1 of the donor splicing site of intron 65 (Fig. 4B). Both the two mutations coexisted simultaneously only in the two brothers. None of the 136 controls unrelated to this family, without bullous dermatosis, was found to be the carrier of either of the two mutations.
Figure 4. Result of DNA sequencing of COL7A1 mutation in the affected brothers.
It shows compound heterozygosity of the mutations of c.1006_1007delCA (exon 8) (A) and c.IVS5568+1delG (intron 65) (B).
Discussion
DEB may be inherited in autosomal dominant or autosomal recessive pattern, both of which are induced by COL7A1 gene mutation. DDEB and RDEB are divided into 6 and 7 subtypes, respectively [6]. Our study focuses on the latter. Comparatively speaking, the clinical phenotype of RDEB is more serious than that of DDEB.
COL7A1 gene is located in chromosome 3p21.31. It is primarily expressed in stratified squamous epithelia, such as the skin, the mucous membranes, and the cornea of the eye [7]. Among all genes described so far, COL7A1 gene is the second largest, containing 118 exons [5], [7], [8]. Its 8.8 Kb coding region of the 9.2 Kb transcript encodes pro- 1 collagen (320 kDa) with distinct structural domains. This collagen, namely the type VII collagen, is composed of 2294 amino acids. The main structure of the molecule consists of a collagenous triple helix (145 kDa) encoded by exons 29–112, and an amino terminus (NC-1) (145 kDa) encoded by exons 2–28, as well as a carboxyl terminus (NC-2) encoded by exons 113–118 (30 kDa) [9]. The protein encoded by COL7A1 gene is essential for the connection of the dermis and the epidermis.
1 collagen (320 kDa) with distinct structural domains. This collagen, namely the type VII collagen, is composed of 2294 amino acids. The main structure of the molecule consists of a collagenous triple helix (145 kDa) encoded by exons 29–112, and an amino terminus (NC-1) (145 kDa) encoded by exons 2–28, as well as a carboxyl terminus (NC-2) encoded by exons 113–118 (30 kDa) [9]. The protein encoded by COL7A1 gene is essential for the connection of the dermis and the epidermis.
The mutation of the COL7A1 gene may result in DEB, in either autosomal dominant or autosomal recessive fashion. So far, more than 730 mutations on the COL7A1 gene have been revealed to be involved in DEB, most of which are glycine substitution mutations located at the triple helix domain [10]. It has been reported that approximately 75% of the DDEB mutations occur in exons 73–75 [11], while RDEB are caused by mutations on both alleles that result in either null alleles or out-of-frame mutations from insertions/deletions, single-base substitutions, and splice junction alterations [12]–[16]. Known RDEB-induced mutation loci are scattered throughout the genome of the COL7A1 gene, with no obvious hot spot [17]–[28]. Similar circumstances also exist in Chinese RDEB patients [29]–[31]. Although most RDEB were induced by compound heterozygous mutations, the majority of severe mutilating RDEB were caused by homozygous mutations leading to premature translational termination [32].
In this study, the maternal mutation is a 2-bp deletion at exon 8 of the COL7A1 gene (c.1006_1007delCA), leading to a subsequent reading frame-shift and substitution of the codon of CAG, which encodes glutamine, by GAA, which encodes glutamic acid (p.Q336EfsX48). This accordingly generates a premature termination codon at 48 amino acids downstream of exon 9, resulting in the truncation of 2561 amino acids downstream, which include part of the NC-1 (from fibronectin 2 domain), all the collagenous triple helix, and the NC-2 (Fig. 5). As a consequence, the type VII collagen chain of the amino-terminus that functions as the adhesion site to the laminin 5 at the lamina densa, became defective, thus markedly affecting the adhesive function of the anchoring fibrils.
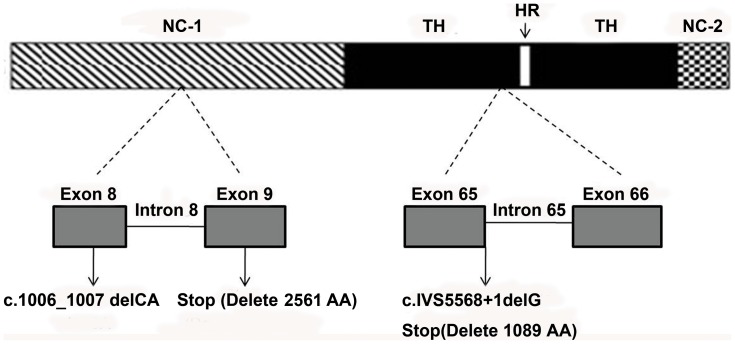
Figure 5. Schematic diagram of the consequences of the mutations identified in the RDEB pedigree.
The mutation on exon 8 (c.1006_1007delCA), standing at NC-1 of the COL7A1 gene, leads to a premature termination codon (stop) located 48 amino acids downstream in exon 9, resulting in truncation of 2561 amino acids downstream, which include part of the NC-1, all the collagenous triple helix, and the NC-2. The mutation discovered on intron 65 (c.IVS5568+1delG), located at the collagenous triple helix domain of the COL7A1 gene, immediately creates a stop codon, consequently producing a truncated protein lacking part of 1089 C-terminal amino acids downstream. Abbreviations: TH, triple helix; HR, hinge region; NC-1, amino terminus; NC-2, carboxyl terminus.
The paternal mutation is a 1-bp deletion at the first basepair of intron 65 (GTAA→TAA), resulting in subsequent read-through intron 65 and immediately creating a stop codon after the amino acids encoded by exon 65 (c.IVS5568+1delG). Due to inactivation of the strongly conserved GT dinucleotide at the 5′ donor splice site, it is predicted to produce a truncated protein lacking part of 1089 C-terminal amino acids downstream (Fig. 5).
As the carriers of either mutation, the patients’ younger sister and their father did not show any phenotype. Considering the fact that the two brothers with compound heterozygosis showed clinical RDEB, and their father was the carrier of the mutation c.IVS5568+1delG, we could presume that the patients’ asymptomatic mother should be the carrier of the mutation c.1006_1007delCA, although we could not detect it since she was deceased (Fig. 2).
The severity of RDEB may be related to the position of the stop codon; however, the presence of some functional protein may also appears to be the important factor in ameliorating the disease severity. Mutations occurring within the central collagenous domain allow preservation of the noncollagenous extensions, which can serve adhesive functions. Furthermore, it has been reported that the heterozygous carriers, which contain only about half the normal amount of anchoring fibrils in their skin, are clinically normal [33]. This may be part of the reason that the carriers of c.1006_1007delCA mutation (patients’ mother) or c.IVS5568+1delG mutation (patients’ father and younger sister) did not demonstrate any phenotype. Also, it has been clarified that the same mutation on COL7A1 gene may result in a different phenotype of either dominant or recessive DEB [16], [34], [35], strongly suggesting that not only the mutation location, but also the nature of the mutation functionally affects helix formation, protein folding, thermal stability, intracellular transport, secretion, and assembly into anti-parallel dimmers or anchoring fibrils [18], [36], [37].
Neither mutation existed in 136 normal controls unrelated to this family, indicating that the mutations found in the two patients were not genetic polymorphism. Identification of two novel mutations in the human COL7A1 gene is based on comparisons with the Human Genome Mutation Database. Despite the high heterogeneity in the COL7A1 gene, the mutations found in this study are new mutations reported for the first time. Although over 730 mutations have been identified in the COL7A1 gene, every novel mutation identified will provide a clue for early diagnosis of the disease in the future, and this compound mutation is the first mutation identified in the COL7A1 gene in dystrophic epidermolysis bullosa in the western Chinese population. So the identification of this novel mutation is not only important for the family in the study, but also useful to study the pathogenesis of the disease in the future.
Funding Statement
This study was supported by the research grants from Sichuan Provincial Bureau of Health (#416.001.004.051), Sichuan Academy of Medical Sciences & Sichuan Provincial People’s Hospital (#303.005.002.209.051) (XJC); grant from the Department of Science and Technology of Sichuan Province, China (2011JTD0020, 2010FZ0076 (ZLY). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Christiano AM, Suga Y, Greenspan DS, Ogawa H, Uitto J (1995) Premature termination codons on both alleles of the type VII collagen gene (COL7A1) in three brothers with recessive dystrophic epidermolysis bullosa. J Clin Invest 95: 1328–1334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Christiano AM, Greenspan DS, Hoffman GG, Zhang X, Tamai Y, et al. (1993) A missense mutation in type VII collagen in two affected siblings with recessive dystrophic epidermolysis bullosa. Nat Genet 4: 62–66. [DOI] [PubMed] [Google Scholar]
- 3. Varki R, Sadowski S, Uitto J, Pfendner E (2007) Epidermolysis bullosa. II. Type VII collagen mutations and phenotype-genotypecorrelations in the dystrophic subtypes. J Med Genet 44: 181–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wertheim-Tysarowska K, Sobczynska-Tomaszewska A, Kowalewski C, Skronski M, Swieckowski G, et al. (2012) The COL7A1 mutation database. Hum Mutat 33: 327–331. [DOI] [PubMed] [Google Scholar]
- 5. Christiano AM, Hoffman GG, Zhang X, Xu Y, Tamai Y, et al. (1997) Strategy for identification of sequence variants in COL7A1 and a novel 2-bpdeletion mutation in recessive dystrophic epidermolysis bullosa. Hum Mutat 10: 408–414. [DOI] [PubMed] [Google Scholar]
- 6. Fine JD, Eady RA, Bauer EA, Bauer JW, Bruckner-Tuderman L, et al. (2008) The classification of inherited epidermolysis bullosa (EB): Report of the ThirdInternational Consensus Meeting on Diagnosis and Classification of EB. J Am Acad Dermatol 58: 931–950. [DOI] [PubMed] [Google Scholar]
- 7. Christiano AM, Greenspan DS, Lee S, Uitto J (1994) Cloning of human type VII collagen. Complete primary sequence of the alpha 1(VII)chain and identification of intragenic polymorphisms. J Biol Chem 269: 20256–20262. [PubMed] [Google Scholar]
- 8. Cuadrado-Corrales N, Sanchez-Jimeno C, Garcia M, Escamez MJ, Illera N, et al. (2010) A prevalent mutation with founder effect in Spanish Recessive DystrophicEpidermolysis Bullosa families. BMC Med Genet 11: 139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Christiano AM, Hoffman GG, Chung-Honet LC, Lee S, Cheng W, et al. (1994) Structural organization of the human type VII collagen gene (COL7A1), composed of more exons than any previously characterized gene. Genomics 21: 169–179. [DOI] [PubMed] [Google Scholar]
- 10. Leverkus M, Ambach A, Hoefeld-Fegeler M, Kohlhase J, Schmidt E, et al. (2011) Late-onset inversa recessive dystrophic epidermolysis bullosa caused by glycinesubstitutions in collagen type VII. Br J Dermatol 164: 1104–1106. [DOI] [PubMed] [Google Scholar]
- 11. Mallipeddi R, Bleck O, Mellerio JE, Ashton GH, Eady RA, et al. (2003) Dilemmas in distinguishing between dominant and recessive forms of dystrophic epidermolysis bullosa. Br J Dermatol 149: 810–818. [DOI] [PubMed] [Google Scholar]
- 12. Christiano AM, Fine JD, Uitto J (1997) Genetic basis of dominantly inherited transient bullous dermolysis of thenewborn: a splice site mutation in the type VII collagen gene. J Invest Dermatol 109: 811–814. [DOI] [PubMed] [Google Scholar]
- 13. Cserhalmi-Friedman PB, McGrath JA, Mellerio JE, Romero R, Salas-Alanis JC, et al. (1998) Restoration of open reading frame resulting from skipping of an exon with aninternal deletion in the COL7A1 gene. Lab Invest 78: 1483–1492. [PubMed] [Google Scholar]
- 14. Mellerio JE, Salas-Alanis JC, Amaya-Guerra M, Tamez E, Ashton GH, et al. (1999) A recurrent frameshift mutation in exon 19 of the type VII collagen gene (COL7A1) in Mexican patients with recessive dystrophic epidermolysis bullosa. Exp Dermatol 8: 22–29. [DOI] [PubMed] [Google Scholar]
- 15. Gardella R, Castiglia D, Posteraro P, Bernardini S, Zoppi N, et al. (2002) Genotype-phenotype correlation in italian patients with dystrophic epidermolysis bullosa. J Invest Dermatol 119: 1456–1462. [DOI] [PubMed] [Google Scholar]
- 16. Gardella R, Zoppi N, Zambruno G, Barlati S, Colombi M (2002) Different phenotypes in recessive dystrophic epidermolysis bullosa patients sharing the same mutation in compound heterozygosity with two novel mutations in the type VII collagen gene. Br J Dermatol 147: 450–457. [DOI] [PubMed] [Google Scholar]
- 17. Cserhalmi-Friedman PB, Karpati S, Horvath A, Christiano AM (1997) Identification of a glycine substitution and a splice site mutation in the type VII collagen gene in a proband with mitis recessive dystrophic epidermolysis bullosa. Arch Dermatol Res 289: 640–645. [DOI] [PubMed] [Google Scholar]
- 18. Hovnanian A, Rochat A, Bodemer C, Petit E, Rivers CA, et al. (1997) Characterization of 18 new mutations in COL7A1 in recessive dystrophic epidermolysis bullosa provides evidence for distinct molecular mechanisms underlying defective anchoring fibril formation. Am J Hum Genet 61: 599–610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Gardella R, Zoppi N, Ferraboli S, Marini D, Tadini G, et al. (1999) Three homozygous PTC mutations in the collagen type VII gene of patients affected by recessive dystrophic epidermolysis bullosa: analysis of transcript levels in dermal fibroblasts. Hum Mutat 13: 439–452. [DOI] [PubMed] [Google Scholar]
- 20. Ryoo YW, Kim BC, Lee KS (2001) Characterization of mutations of the type VII collagen gene (COL7A1) in recessive dystrophic epidermolysis bullosa mitis (M-RDEB) from three Korean patients. J Dermatol Sci 26: 125–132. [DOI] [PubMed] [Google Scholar]
- 21. Ishiko A, Masunaga T, Ota T, Nishikawa T (2004) Does the position of the premature termination codon in COL7A1 correlate with the clinical severity in recessive dystrophic epidermolysis bullosa. Exp Dermatol 13: 229–233. [DOI] [PubMed] [Google Scholar]
- 22. Masse M, Cserhalmi-Friedman PB, Falanga V, Celebi JT, Martinez-Mir A, et al. (2005) Identification of novel type VII collagen gene mutations resulting in severe recessive dystrophic epidermolysis bullosa. Clin Exp Dermatol 30: 289–293. [DOI] [PubMed] [Google Scholar]
- 23. Fassihi H, Lu L, Wessagowit V, Ozoemena LC, Jones CA, et al. (2006) Complete maternal isodisomy of chromosome 3 in a child with recessive dystrophic epidermolysis bullosa but no other phenotypic abnormalities. J Invest Dermatol 126: 2039–2043. [DOI] [PubMed] [Google Scholar]
- 24. Kraemer L, Wajid M, Christiano AM (2006) A glycine substitution in the COL7A1 gene causes mild RDEB in a Pakistani family. Eur J Dermatol 16: 615–619. [PubMed] [Google Scholar]
- 25. Titeux M, Mejia JE, Mejlumian L, Bourthoumieu S, Mirval S, et al. (2006) Recessive dystrophic epidermolysis bullosa caused by COL7A1 hemizygosity and a missense mutation with complex effects on splicing. Hum Mutat 27: 291–292. [DOI] [PubMed] [Google Scholar]
- 26. Yonei N, Ohtani T, Furukawa F (2006) Recessive dystrophic epidermolysis bullosa: case of non-Hallopeau-Siemens variant with premature termination codons in both alleles. J Dermatol 33: 802–805. [DOI] [PubMed] [Google Scholar]
- 27. Dang N, Murrell DF (2008) Mutation analysis and characterization of COL7A1 mutations in dystrophic epidermolysis bullosa. Exp Dermatol 17: 553–568. [DOI] [PubMed] [Google Scholar]
- 28. Cho JW, Nakano H, Lee KS (2009) Type VII collagen gene mutations (c.8569G>T and c.4879G>A) result in the moderately severe phenotype of recessive dystrophic epidermolysis bullosa in a Korean patient. J Korean Med Sci 24: 256–261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lin GT, Chen SK, Liu CS, Wang WH (2000) Splice site mutation in the type VII collagen gene (COL7A1) in a Taiwanese family with recessive dystrophic epidermolysis bullosa. J Formos Med Assoc 99: 693–697. [PubMed] [Google Scholar]
- 30. Jiang W, Sun Y, Li S, Chen XX, Bu DF, et al. (2005) Two novel heterozygous mutations in COL7A1 in a Chinese patient with recessive dystrophic epidermolysis bullosa of Hallopeau-Siemens type. Br J Dermatol 152: 1357–1359. [DOI] [PubMed] [Google Scholar]
- 31. Huang L, Wong YP, Burd A (2011) A novel homozygous splice site mutation in COL7A1 in a Chinese patient with severe recessive dystrophic epidermolysis bullosa and squamous cell carcinoma. Int J Dermatol 50: 52–56. [DOI] [PubMed] [Google Scholar]
- 32. Galehdari H, Mohammadian G, Azmoon S, Salehi B, Pedram M (2010) A novel COL7A1 gene mutation in an Iranian individual suffering dystrophic epidermolysis bullosa. J Mol Diagn 12: 377–379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Christiano AM, Anhalt G, Gibbons S, Bauer EA, Uitto J (1994) Premature termination codons in the type VII collagen gene (COL7A1) underlie severe, mutilating recessive dystrophic epidermolysis bullosa. Genomics 21: 160–168. [DOI] [PubMed] [Google Scholar]
- 34. Almaani N, Liu L, Dopping-Hepenstal PJ, Lai-Cheong JE, Wong A, et al. (2011) Identical glycine substitution mutations in type VII collagen may underlie both dominant and recessive forms of dystrophic epidermolysis bullosa. Acta Derm Venereol 91: 262–266. [DOI] [PubMed] [Google Scholar]
- 35. Uitto J (2011) In this issue: glycine substitution mutations in the COL7A1 gene: implications for inheritance of dystrophic epidermolysis bullosa - dominant vs. recessive. Acta Derm Venereol 91: 259–261. [DOI] [PubMed] [Google Scholar]
- 36. Shimizu H, Hammami-Hauasli N, Hatta N, Nishikawa T, Bruckner-Tuderman L (1999) Compound heterozygosity for silent and dominant glycine substitution mutations in COL7A1 leads to a marked transient intracytoplasmic retention of procollagen VII and a moderately severe dystrophic epidermolysis bullosa phenotype. J Invest Dermatol 113: 419–421. [DOI] [PubMed] [Google Scholar]
- 37. Woodley DT, Hou Y, Martin S, Li W, Chen M (2008) Characterization of molecular mechanisms underlying mutations in dystrophic epidermolysis bullosa using site-directed mutagenesis. J Biol Chem 283: 17838–17845. [DOI] [PMC free article] [PubMed] [Google Scholar]