Abstract
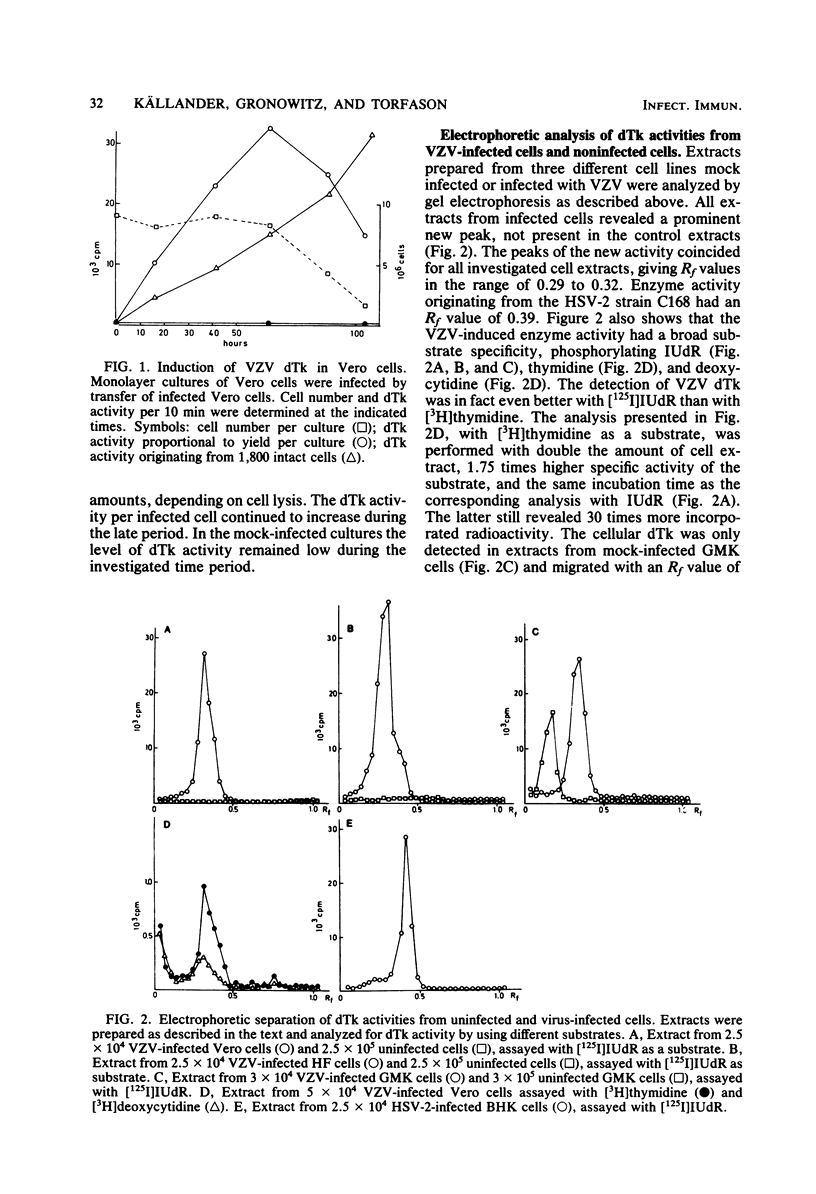
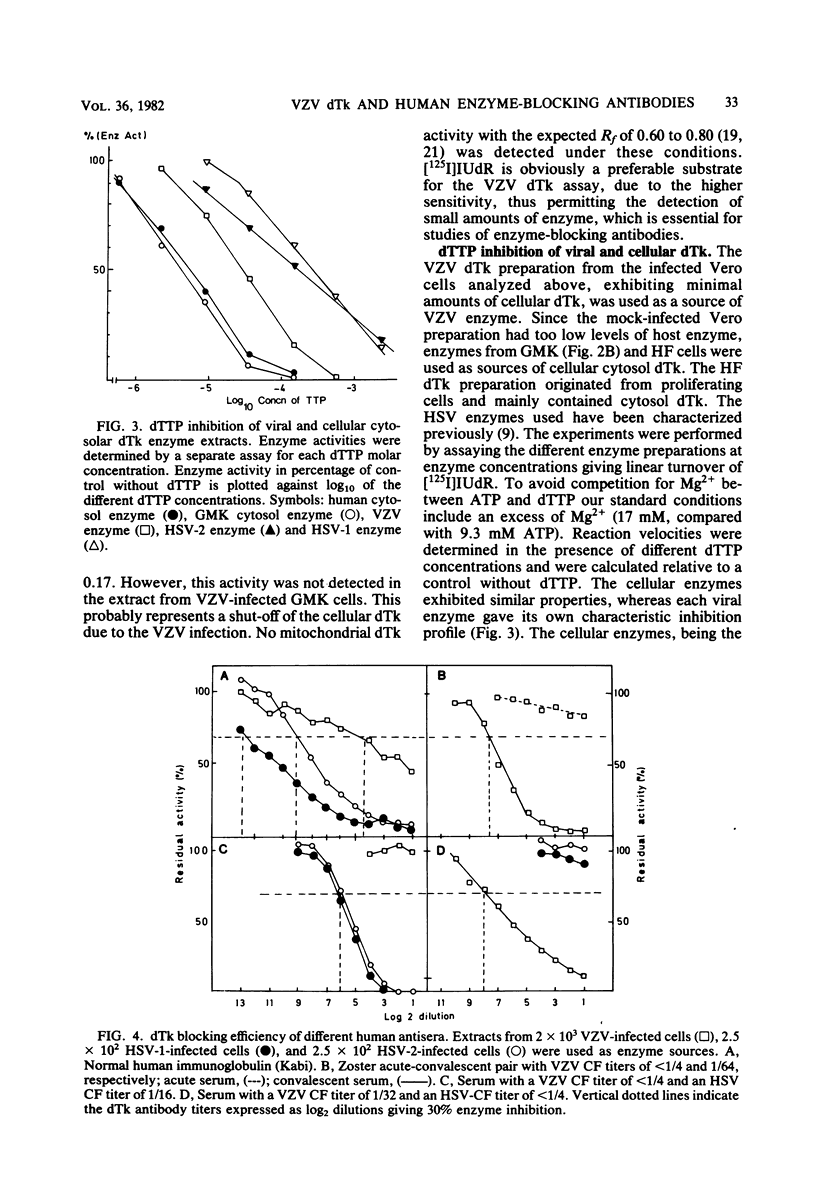
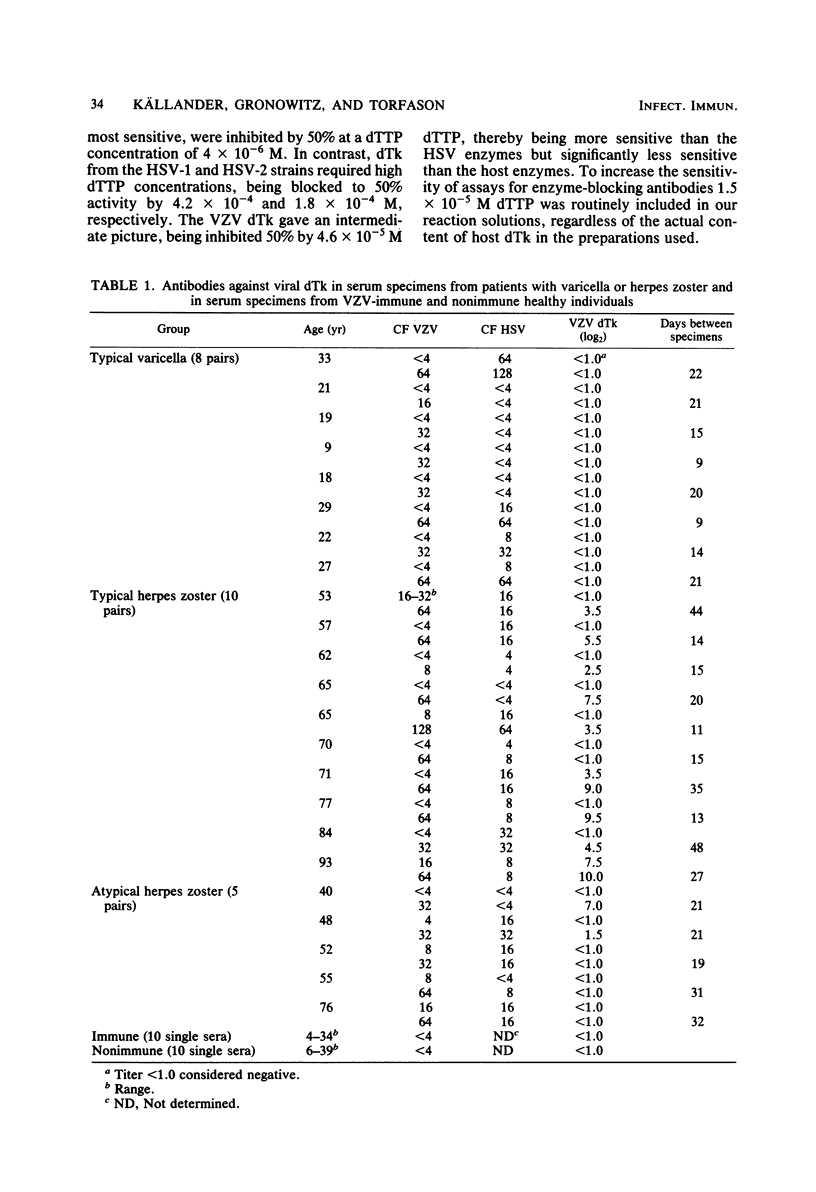
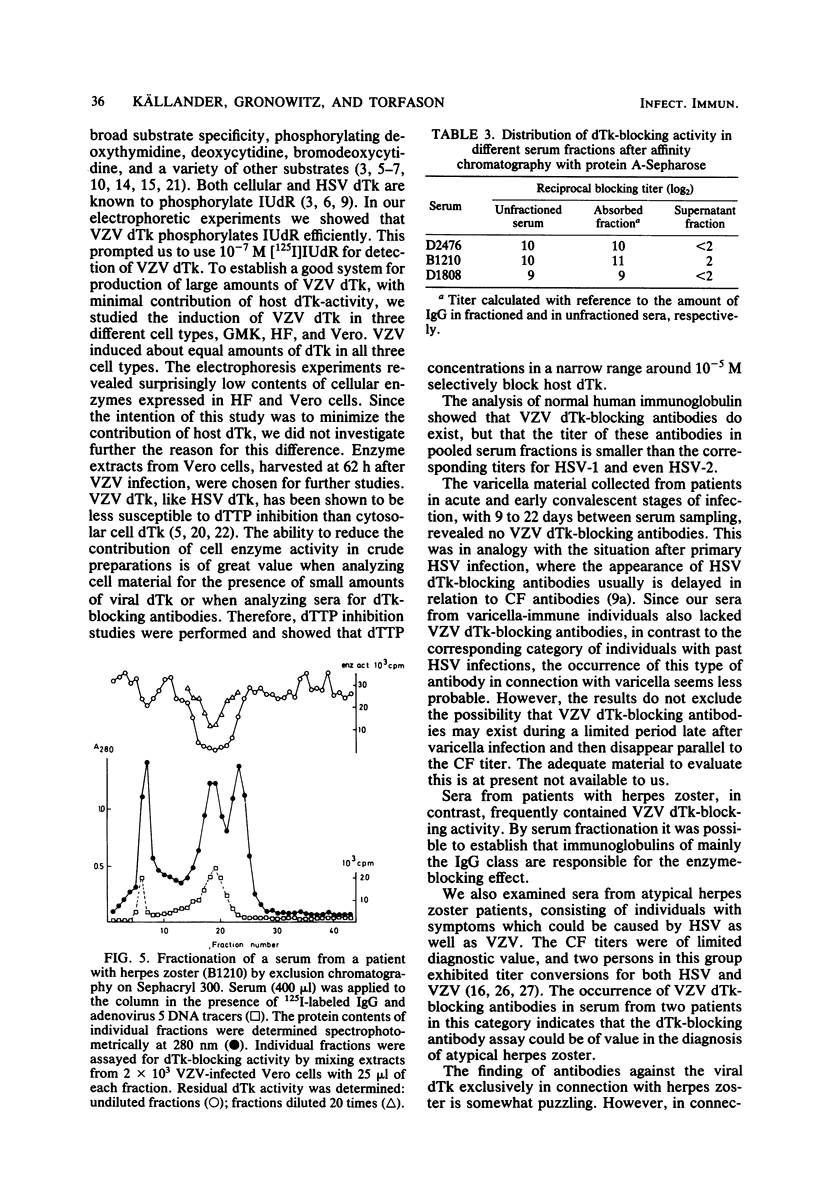
The conditions required for the production of varicella-zoster virus (VSV)-induced deoxythymidine kinase (dTk) have been studied. Extracts from Vero cells harvested 62 h after VZV infection were found to contain VZV-induced dTk activity, with a minimal contribution from the cellular dTk activity. VZV dTK was shown to have a broad substrate specificity phosphorylating both deoxythymidine, deoxycytidine, and iododeoxyuridine. Deoxythymidine triphosphate inhibition studies revealed an intermediate deoxythymidine triphosphate sensitivity when compared with that of the cellular cytosolar enzyme and the deoxythymidine triphosphate-insensitive herpes simplex virus dTk. An assay for VZV dTk-blocking antibodies was developed, with [125I]iododeoxyuridine as a substrate in the presence of a deoxythymidine triphosphate concentration which selectively blocked the dTK of host cell origin. A total of 79 serum samples were studied; these included serum pairs from patients with varicella or herpes zoster and single sera from immune and nonimmune adults. VZV dTk blocking antibodies were detected exclusively in sera from patients with herpes zoster. All serum pairs showing VZV dTK seroconversion also showed a parallel conversion of complement fixation titers. The VZV dTk antibodies were found to be of the immunoglobulin G class. The immunological specificity of VZV dTK was investigated, and no cross-reactivity with herpes simplex virus type 1 or 2 dTk was found.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Buttyan R., Spear P. G. Factors governing expression of the herpes simplex virus gene for thymidine kinase in clonal derivatives of transformed mouse L cells. J Virol. 1981 Jan;37(1):459–472. doi: 10.1128/jvi.37.1.459-472.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camerini-Otero R. D., Zasloff M. A. Nucleosomal packaging of the thymidine kinase gene of herpes simplex virus transferred into mouse cells: an actively expressed single-copy gene. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5079–5083. doi: 10.1073/pnas.77.9.5079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y. C. A rational approach to the development of antiviral chemotherapy: alternative substrates of herpes simplex virus Type 1 (HSV-1) and Type 2 (HSV-2) thymidine kinase (TK). Ann N Y Acad Sci. 1977 Mar 4;284:594–598. doi: 10.1111/j.1749-6632.1977.tb21992.x. [DOI] [PubMed] [Google Scholar]
- Cheng Y. C., Hoffmann P. J., Kung M. P. Presence of antibody against herpes simplex virus-specified thymidine kinase and deoxyribonuclease in human sera. IARC Sci Publ. 1978;(24 Pt 2):899–904. [PubMed] [Google Scholar]
- Cheng Y. C., Tsou T. Y., Hackstadt T., Mallavia L. P. Induction of thymidine kinase and DNase in varicella-zoster virus-infected cells and kinetic properties of the virus-induced thymidine kinase. J Virol. 1979 Jul;31(1):172–177. doi: 10.1128/jvi.31.1.172-177.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobersen M. J., Greer S. Herpes simplex virus type 2 induced pyrimidine nucleoside kinase: enzymatic basis for the selective antiherpetic effect of 5-halogenated analogues of deoxycytidine. Biochemistry. 1978 Mar 7;17(5):920–928. doi: 10.1021/bi00598a028. [DOI] [PubMed] [Google Scholar]
- Dobersen M. J., Jerkofsky M., Greer S. Enzymatic basis for the selective inhibition of varicella-zoster virus by 5-halogenated analogues of deoxycytidine. J Virol. 1976 Nov;20(2):478–486. doi: 10.1128/jvi.20.2.478-486.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grandien M., Appelgren P., Espmark A., Hanngren K. Determination of varicella immunity by the indirect immunofluorescence test in urgent clinical situations. Scand J Infect Dis. 1976;8(2):65–69. doi: 10.3109/inf.1976.8.issue-2.01. [DOI] [PubMed] [Google Scholar]
- Gronowitz J. S., Källander C. F. Occurrence of antibodies against herpes simplex virus thymidine kinase in human sera. J Med Virol. 1981;8(3):177–186. doi: 10.1002/jmv.1890080304. [DOI] [PubMed] [Google Scholar]
- Gronowitz J. S., Källander C. F. Optimized assay for thymidine kinase and its application to the detection of antibodies against herpes simplex virus type 1- and 2-induced thymidine kinase. Infect Immun. 1980 Aug;29(2):425–434. doi: 10.1128/iai.29.2.425-434.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hackstadt T., Mallavia L. P. Deoxypyrimidine nucleoside metabolism in varicella-zoster virus-infected cells. J Virol. 1978 Feb;25(2):510–517. doi: 10.1128/jvi.25.2.510-517.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halliburton I. W., Andrew J. C. DNA polymerase in pseudorabies virus infected cells. J Gen Virol. 1976 Jan;30(1):145–148. doi: 10.1099/0022-1317-30-1-145. [DOI] [PubMed] [Google Scholar]
- Hoffmann P. J., Cheng Y. C. The deoxyribonuclease induced after infection of KB cells by herpes simplex virus type 1 or type 2. I. Purification and characterization of the enzyme. J Biol Chem. 1978 May 25;253(10):3557–3562. [PubMed] [Google Scholar]
- Jamieson A. T., Gentry G. A., Subak-Sharpe J. H. Induction of both thymidine and deoxycytidine kinase activity by herpes viruses. J Gen Virol. 1974 Sep;24(3):465–480. doi: 10.1099/0022-1317-24-3-465. [DOI] [PubMed] [Google Scholar]
- Jamieson A. T., Subak-Sharpe J. H. Biochemical studies on the herpes simplex virus-specified deoxypyrimidine kinase activity. J Gen Virol. 1974 Sep;24(3):481–492. doi: 10.1099/0022-1317-24-3-481. [DOI] [PubMed] [Google Scholar]
- KAPSENBERG J. G. POSSIBLE ANTIGENIC RELATIONSHIP BETWEEN VARICELLA ZOSTER VIRUS AND HERPES SIMPLEX VIRUS. Arch Gesamte Virusforsch. 1964 Sep 9;15:67–73. doi: 10.1007/BF01241422. [DOI] [PubMed] [Google Scholar]
- Keir H. M., Subak-Sharpe H., Shedden W. I., Watson D. H., Wildy P. Immunological evidence for a specific DNA polymerase produced after infection by herpes simplex virus. Virology. 1966 Sep;30(1):154–157. doi: 10.1016/s0042-6822(66)81022-x. [DOI] [PubMed] [Google Scholar]
- Kit S., Leung W. C., Trkula D. Properties of mitochondrial thymidine kinases of parental and enzyme-deficient HeLa cells. Arch Biochem Biophys. 1973 Oct;158(2):503–513. doi: 10.1016/0003-9861(73)90542-0. [DOI] [PubMed] [Google Scholar]
- Kit S. Viral-associated and induced enzymes. Pharmacol Ther B. 1979;4(3):501–585. doi: 10.1016/0163-7258(79)90004-4. [DOI] [PubMed] [Google Scholar]
- Klemperer H. G., Haynes G. R., Shedden W. I., Watson D. H. A virus-specific thymidine kinase in BHK-21 cells infected with herpes simplex virus. Virology. 1967 Jan;31(1):120–128. doi: 10.1016/0042-6822(67)90015-3. [DOI] [PubMed] [Google Scholar]
- Leung W. C., Dubbs D. R., Trkula D., Kit S. Mitochondrial and herpesvirus-specific deoxypyrimidine kinases. J Virol. 1975 Sep;16(3):486–497. doi: 10.1128/jvi.16.3.486-497.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogino T., Otsuka T., Takahashi M. Induction of deoxypyrimidine kinase activity in human embryonic lung cells infected with varicella-zoster virus. J Virol. 1977 Mar;21(3):1232–1235. doi: 10.1128/jvi.21.3.1232-1235.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapp F., Turner N., Schaffer P. A. Biochemical transformation by temperature-sensitive mutants of herpes simplex virus type 1. J Virol. 1980 Jun;34(3):704–710. doi: 10.1128/jvi.34.3.704-710.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawls W. E., Cashon G., Adam E., Ogino T., Duff R., Rapp F. Herpesvirus type 2-induced thymidine kinase and carcinoma of the cervix. Cancer Res. 1974 Feb;34(2):362–366. [PubMed] [Google Scholar]
- Schmidt N. J., Dennis J., Lennette E. H. Complement-fixing reactivity of Varicella-Zoster virus subunit antigens with sera from homotypic infections and heterotypic Herpes simplex virus infections. Infect Immun. 1977 Mar;15(3):850–854. doi: 10.1128/iai.15.3.850-854.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt N. J., Lennette E. H., Magoffin R. L. Immunological relationship between herpes simplex and varicella-zoster viruses demonstrated by complement-fixation, neutralization and fluorescent antibody tests. J Gen Virol. 1969 Apr;4(3):321–328. doi: 10.1099/0022-1317-4-3-321. [DOI] [PubMed] [Google Scholar]
- Schmidt N. J., Lennette E. H., Woodie J. D., Ho H. H. Immunofluorescent staining in the laboratory diagnosis of varicella-zoster virus infections. J Lab Clin Med. 1965 Sep;66(3):403–412. [PubMed] [Google Scholar]
- Smiley J. R. Construction in vitro and rescue of a thymidine kinase-deficient deletion mutation of herpes simplex virus. Nature. 1980 May 29;285(5763):333–335. doi: 10.1038/285333a0. [DOI] [PubMed] [Google Scholar]
- Thouless M. E. Serological properties of thymidine kinase produced in cells infected with type 1 or type 2 herpes virus. J Gen Virol. 1972 Dec;17(3):307–315. doi: 10.1099/0022-1317-17-3-307. [DOI] [PubMed] [Google Scholar]



