Abstract
A better understanding of the molecular mechanisms of signaling by the neurotransmitter serotonin is required to assess the hypothesis that defects in serotonin signaling underlie depression in humans. Caenorhabditis elegans uses serotonin as a neurotransmitter to regulate locomotion, providing a genetic system to analyze serotonin signaling. From large-scale genetic screens we identified 36 mutants of C. elegans in which serotonin fails to have its normal effect of slowing locomotion, and we molecularly identified eight genes affected by 19 of the mutations. Two of the genes encode the serotonin-gated ion channel MOD-1 and the G-protein-coupled serotonin receptor SER-4. mod-1 is expressed in the neurons and muscles that directly control locomotion, while ser-4 is expressed in an almost entirely non-overlapping set of sensory and interneurons. The cells expressing the two receptors are largely not direct postsynaptic targets of serotonergic neurons. We analyzed animals lacking or overexpressing the receptors in various combinations using several assays for serotonin response. We found that the two receptors act in parallel to affect locomotion. Our results show that serotonin functions as an extrasynaptic signal that independently activates multiple receptors at a distance from its release sites and identify at least six additional proteins that appear to act with serotonin receptors to mediate serotonin response.
Keywords: serotonin, serotonin receptors, Caenorhabditis elegans, extrasynaptic neurotransmitter signaling, locomotion behavior
DEPRESSION is hypothesized to involve dysfunction of the neurotransmitter serotonin (Cowen 2008). Understanding the molecular mechanism of serotonin signaling is complicated by the fact that the human brain expresses 14 types of serotonin receptors, one of which is a serotonin-gated ion channel, and the rest of which are G-protein-coupled receptors (Millan et al. 2008). An additional challenge to understanding serotonin signaling is the fact that serotonin can act locally at synapses where it is released or diffuse away and act at distant receptors. While classical neurotransmitters such as GABA and glutamate appear to function mainly locally at synapses, serotonin can diffuse several microns from its release sites at concentrations sufficient to activate its receptors (Bunin and Wightman 1998). Furthermore, serotonin receptors are often localized at nonsynaptic sites (Kia et al. 1996). These observations suggest that serotonin might act predominantly as an extrasynaptic signal to activate several receptor types on cells distant from its release sites and that the combined action of these several receptors somehow coordinates appropriate responses to serotonin. The details of how such action might occur remain unclear.
Caenorhabditis elegans uses serotonin as a neurotransmitter (Horvitz et al. 1982; Chase and Koelle 2007) and provides a model system with the potential to make important contributions to the study of serotonin signaling. First, C. elegans allows the use of forward genetic screens to identify the proteins beyond serotonin receptors that mediate serotonin response. Second, the known synaptic wiring of the C. elegans nervous system (White et al. 1986) provides the opportunity to directly test whether serotonin acts at synapses or extrasynaptically.
Previous studies of C. elegans have shown that, of the precisely 302 neurons found in an adult hermaphrodite, the NSM, HSN, ADF, VC4/5, AIM, and RIH neurons contain serotonin, although the AIM, RIH, and possibly also the VC4/5 neurons do not synthesize serotonin themselves but rather take up serotonin made by the other neurons (Horvitz et al. 1982; Sawin et al. 2000; Jafari et al. 2011). Animals with mutations in the enzymes that synthesize serotonin or in which the NSMs have been ablated are defective for the strong reduction in locomotion behavior, known as enhanced slowing, that occurs when food-deprived animals encounter food (Sawin et al. 2000). This observation suggests that the NSMs release serotonin to slow locomotion. While enhanced slowing is only one of several behaviors known to be controlled by serotonin in C. elegans (Chase and Koelle 2007), it is amenable to genetic analysis, and direct genetic screens for mutants defective in enhanced slowing behavior have succeeded in isolating mutants defective for serotonin signaling (Sawin et al. 2000). However, assays for this behavior are relatively tedious. In a second approach, mutants carrying gene knockouts for all five C. elegans serotonin receptor homologs have been tested for defects in the ability of serotonin to slow locomotion (Hapiak et al. 2009). Several of the receptor knockouts cause defects in this behavior, but basic issues about the functions of these receptors have remained unresolved. How do the receptors act together to slow locomotion? Do they act at sites distant from serotonin release sites? Do the various receptors act in the same or different cells? What other signaling proteins act with the receptors to mediate serotonin response?
In this study, we developed genetic screens for C. elegans mutants defective for the effects of serotonin on locomotion. We identified 36 mutants, found the relevant molecular lesions in 19 of them, and thus identified eight proteins required for serotonin response. Among these proteins were two serotonin receptors, MOD-1 and SER-4. Since understanding how the remaining signaling proteins affect serotonin response first requires understanding the function of the two receptors, we analyzed how MOD-1 and SER-4 affect locomotion. Our results suggest that the two receptors act in parallel in different cells to mediate different effects of extrasynaptic serotonin on locomotion.
Materials and Methods
Nematode culture
C. elegans strains were maintained at 20° under standard conditions, and double- and triple-mutant strains were generated using standard genetic techniques (Brenner 1974). All strains and transgenes used in this study are described in Supporting Information, File S5, Table S1, and Table S2.
Genetic screens
In a clonal screening strategy, fourth-stage larval (L4) wild-type hermaphrodites were mutagenized (Brenner 1974) using ethyl methanesulfonate (EMS), and F1 progeny were grown from eggs harvested after bleaching the P0 gravid adults. First larval-stage F1 animals were diluted to 1/100 µl in S medium (Sulston and Hodgkin 1988) supplemented with penicillin and streptomycin (final concentrations 100 units/ml and 100 µg/ml, respectively; Gibco 15140-122), nysatin (final concentration 100 units/ml; Sigma N1638), and Escherichia coli strain HB101 diluted to a final OD550 = 2.4. A 12-channel pipette was used to distribute 50 µl/well into 96-well plates. Wells containing >1 F1 were marked and not screened. Plates were placed in a plastic container with a loose-fitting lid containing wet paper towels and incubated for 6 days at 20° until mixed-stage F2 progeny were present but the cultures were not yet starved. A total of 100 µl of 45 mM serotonin (5-hydroxytryptamine creatine sulfate complex; Sigma H-7752) was added to each well. The supersaturated 45-mM serotonin stock solution was prepared by adding serotonin powder to S medium lacking trace metals, heating in a water bath to 80° for 5 min, vortexing, cooling to room temperature, and using within 30 min.
For nonclonal genetic screens, mod-5(n3314) L4 hermaphrodites were mutagenized using EMS or the Mosl transposon (Boulin and Bessereau 2007). Five P0 animals were transferred to each of several 10-cm diameter NGM plates (Brenner 1974) and allowed to produce F1 progeny for 3–5 days at 20°. F1 progeny were bleached to collect eggs. Approximately 300 eggs from each 10-cm plate were spotted to 6-cm-diameter NGM plates and grown at 20° for 2 days. These F2 progeny were transferred to 24-well cell culture plates containing 20 or 60 mM serotonin dissolved in M9 buffer (Sulston and Hodgkin 1988) and assayed for movement 5 min later. Moving animals were transferred from wells using a mouth pipette to individual 6-cm NGM plates, and their progeny were retested for serotonin resistance.
Measurement of the serotonin-resistance phenotype in liquid
Serotonin-resistance measurements in Table 1 and Figure 2A were performed in M9 buffer in microtiter wells containing serotonin. Serotonin was prepared as a 45-mM stock solution in M9 by heating to 80°, vortexing, and cooling as described above. Animals were isolated at the late L4 stage based on the morphology of the developing vulva and cultured for 24 hr at 20° to produce young adults. Animals (typically 25–33 animals per well) were transferred with a platinum-wire worm pick to microtiter wells containing 50 µl M9 buffer. A 12-channel pipette was used to add serotonin stock solution to the final desired concentration, and 20 min later wells were scored for the number of animals “moving,” defined as having smooth swimming movements of the entire body. Animals showing only movements of the head or only stiff or jerky movements of ≤50% of the body were scored as “not moving.”
Table 1 . Isolation and characterization of serotonin-resistant mutants.
| Serotonin resistance mutationa | Chromosomeb | Genetic backgroundc | Serotonin in screen (mM)d | Mutagen | Serotonin in assay (mM)e | % moving in assayf |
|---|---|---|---|---|---|---|
| None | Wild type | 30 | 4 | |||
| None | mod-5 | 5 | 4 | |||
| None | mod-5 | 15 | 1 | |||
| None | mod-5 | 30 | 0 | |||
| goa-1(vs115) | I | Wild type | 30 | EMS | 30 | 98 |
| goa-1(vs134) | I | Wild type | 30 | EMS | 30 | 74 |
| goa-1(n4093) | I | mod-5 | 20 | Mos1 | 5 | 58 |
| goa-1(n4402) | I | mod-5 | 60 | EMS | 15 | 15 |
| goa-1(n4405) | I | mod-5 | 60 | EMS | 15 | 88 |
| goa-1(n4439) | I | mod-5 | 60 | EMS | 15 | 100 |
| goa-1(n4492) | I | mod-5 | 60 | EMS | 15 | 100 |
| goa-1(n4493) | I | mod-5 | 60 | EMS | 15 | 95 |
| goa-1(n4494) | I | mod-5 | 60 | EMS | 15 | 83 |
| eat-16(n4403) | I | mod-5 | 60 | EMS | 15 | 83 |
| abts-1(n4094) | I | mod-5 | 20 | Mos1 | 5 | 90 |
| n3498 | I | mod-5 | 20 | EMS | 5 | 69 |
| n3792 | I | mod-5 | 20 | EMS | 5 | 43 |
| n4404 | I | mod-5 | 60 | EMS | 15 | 72 |
| n3461 | II | mod-5 | 20 | EMS | 5 | 86 |
| n3488 | II | mod-5 | 20 | EMS | 5 | 40 |
| n3495 | II | mod-5 | 20 | EMS | 5 | 35 |
| n3775 | II | mod-5 | 20 | EMS | 5 | 80 |
| ser-4(vs122) | III | Wild type | 30 | EMS | 30 | 66 |
| emb-9(vs114) | III | Wild type | 30 | EMS | 30 | 20 |
| flp-1(n4491) | IV | mod-5 | 60 | EMS | 15 | 83 |
| flp-1(n4495) | IV | mod-5 | 60 | EMS | 15 | 100 |
| mod-1(vs107) | V | Wild type | 30 | EMS | 30 | 61 |
| mod-1(n3791) | V | mod-5 | 20 | EMS | 30 | 100 |
| mod-1(n4054) | V | mod-5 | 20 | Mos1 | 5 | 86 |
| n3510 | V | mod-5 | 20 | EMS | 30 | 45 |
| n3774 | V | mod-5 | 20 | EMS | 30 | 28 |
| n3799 | V | mod-5 | 20 | EMS | 10 | 34 |
| elpc-3(vs119) | V | wild type | 30 | EMS | 30 | 70 |
| n3477 | X | mod-5 | 20 | EMS | 5 | 49 |
| vs113 | Unmapped | Wild type | 30 | EMS | 30 | 12 |
| vs108 | Unmapped | Wild type | 30 | EMS | 30 | 15 |
| vs111 | Unmapped | Wild type | 30 | EMS | 30 | 16 |
| vs116 | Unmapped | Wild type | 30 | EMS | 30 | 19 |
| vs120 | Unmapped | Wild type | 30 | EMS | 30 | 20 |
| vs121 | Unmapped | Wild type | 30 | EMS | 30 | 21 |
Allele designations without gene names are given for the 17 mutations not assigned to a gene.
Six weak mutations at bottom (“Unmapped”) have not been genetically mapped and thus are not assigned to a chromosome.
mod-5 denotes that the genetic screen and assays were performed in the mod-5(n3314) genetic background.
Concentration of serotonin in buffer used in the genetic screen that initially isolated the mutation.
Concentration of serotonin in M9 buffer in which mutant animals were assayed. The strains listed as “None” carried no serotonin resistance mutations and serve as controls.
Data averaged from two to three separate assays of 20–40 animals each.
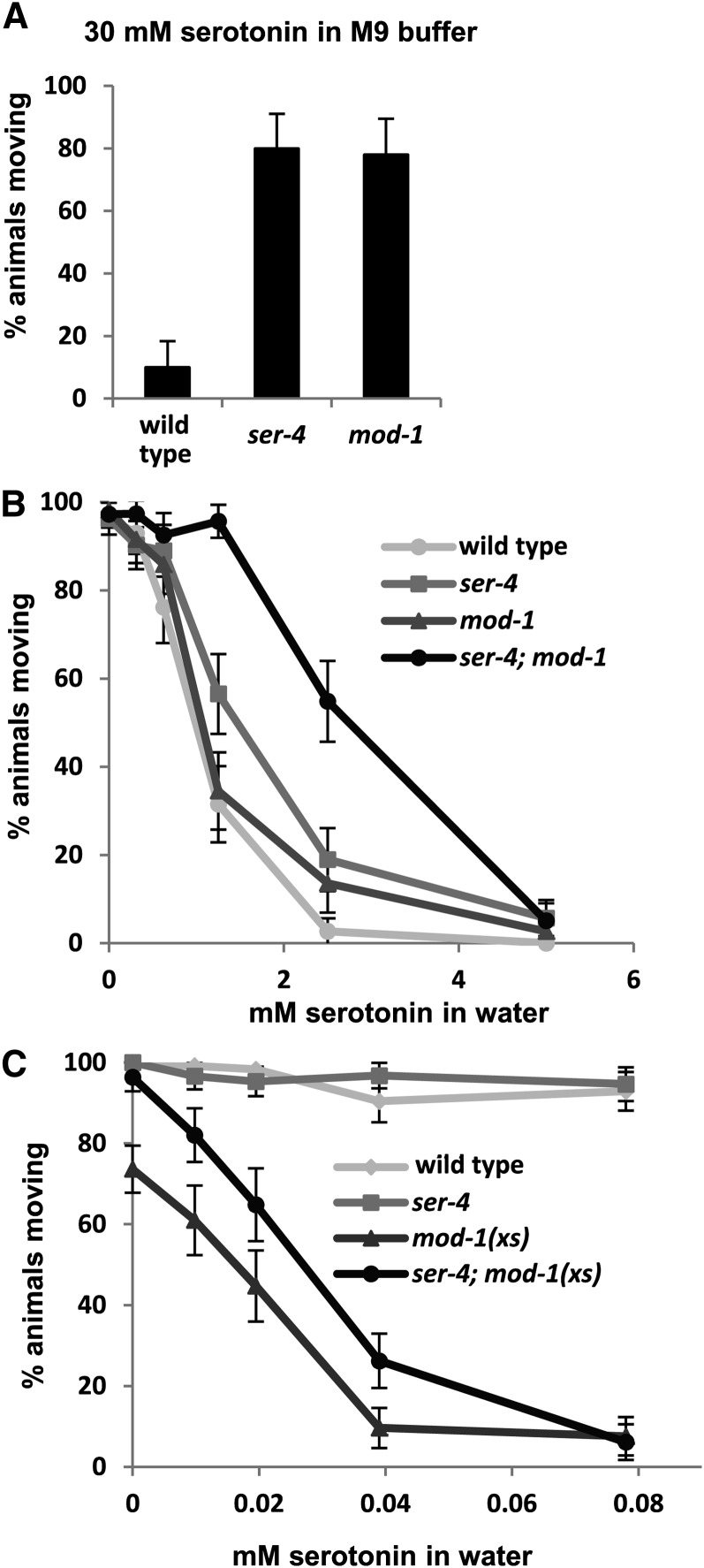
Figure 2 .
Effects of MOD-1 and SER-4 on the ability of serotonin to paralyze C. elegans in liquid. Young adult animals were placed in microtiter wells containing serotonin at the indicated concentrations and 20 min later were scored for movement. (A) Effects of ser-4 or mod-1 deletion mutations on serotonin response when assayed in M9 buffer. (B) Effects of these mutations on serotonin response, alone or in combination, when assayed in water. (C) A MOD-1 overexpressor transgene, mod-1(xs), produces serotonin hypersensitivity in a wild-type or ser-4 mutant background. Assays are in water as in B, but note the ∼100-fold change in scale of the x-axis. Shown are the percentages of animals paralyzed from samples of n = 50 (A) or n >100 (B and C) for each genotype and concentration, with error bars representing 95% confidence intervals.
Serotonin-resistance measurements shown in Figure 2, B and C were performed in an analogous fashion, except that the young adult animals were picked directly into microtiter wells containing various concentrations of serotonin dissolved in water and were scored for movement 20 min later. We found that ∼30-fold less serotonin is sufficient to paralyze C. elegans when it is applied in water rather than in M9 buffer, analogous to the similarly large increase in effectiveness of exogenous dopamine on C. elegans when it is applied in water rather than M9 buffer (Schafer and Kenyon 1995). We thus performed quantitative assays of response to serotonin in water or on agar dissolved in pure water (see below) so that serotonin could be used to affect behavior at concentrations below its solubility limit, producing more reliable assays.
Measurement of serotonin resistance after optogenetic activation of endogenous serotonin release
B broth inoculated with OP50 bacteria (Sulston and Hodgkin 1988) was cultured overnight at 37° without shaking, and just before use 4 µl of 100 mM all-trans retinal (Spectrum Chemical, #R3041) in 100% ethanol was added per 1 ml. Standard NGM plates were seeded with 200 µl of this mix. Plates were incubated in a light-tight box overnight at room temperature to allow a bacterial lawn to grow. Control plates were made with no trans-retinal. Twenty-four hours before the assay, late L4 animals were picked onto freshly prepared retinal or control plates and incubated at 20° overnight in the dark. Thirty minutes prior to assay, unseeded NGM assay plates were ringed with an osmotic barrier to prevent worms from leaving the agar surface by placing a 70-µl drop of 4M fructose on the edge of each plate and tilting the plate around in a circle. For the assay, individual worms not on the OP50 lawn were picked to separate fructose-ringed NGM plates using a bare pick. Ten minutes after transfer, each plate was moved to a Zeiss M2BIO fluorescence dissecting scope, handling the plates gently and leaving the lids in place to avoid mechanically stimulating the animals. Animals were illuminated with 1.2 mW/mm2 of 480 nm light, as measured with a Solar Meter model 9.4 blue light radiometer (Solartech Inc.). The number of body bends was counted during the first 30 sec of blue light exposure. A body bend was defined as “a maximum flexure of the body just behind the pharynx in the opposite direction as the previously scored bend.” ≥15 animals per condition were tested with the experimenter blinded to the genotype, and the exp was repeated three times. Figure 4 shows the results from one representative experiment.
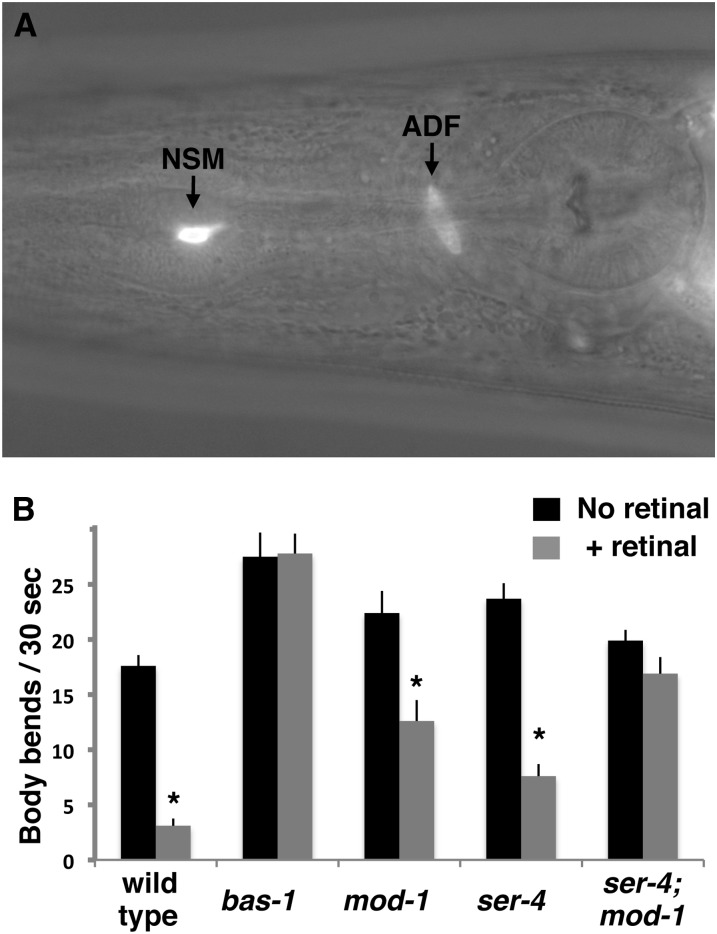
Figure 4 .
SER-4 and MOD-1 receptors are required for control of locomotion by optogenetically released endogenous serotonin. (A) Image of the head region of an animal carrying the ljIs102 transgene, which expresses ChR2::YFP in serotonergic neurons. A Nomarski optics image showing the anatomy of the head is superimposed on an image of yellow fluorescence. Labeled arrows point to YFP fluorescence in the NSM and ADF neurons. Each neuron type is a bilaterally symmetric pair of cells, of which only the left member is visible in this micrograph. (B) Optogenetically stimulated serotonin release slows locomotion dependent on the MOD-1 and SER-4 serotonin receptors. Young adults of the indicated genotypes grown either with or without the all-trans retinal cofactor required for ChR2 function were illuminated with blue light, and the number of body bends during the first 30 sec of illumination was counted. In addition to the mutations indicated, all animals in this experiment carried a mutation in lite-1 (Edwards et al. 2008) to block the endogenous response to blue light of C. elegans. Asterisks indicate significant slowing (P < 0.05, one-tailed t-test) in all-trans retinal-treated animals relative to the controls without all-trans retinal.
Locomotion-slowing assays
Slowing assays were performed as described by Sawin et al. (2000). Measurements shown are averaged from assays repeated over 2–9 days with samples totaling at least 20 animals for the no-food assays and at least 40 animals for the + food assays.
Gene identification
Serotonin-resistance mutations were mapped to regions of individual chromosomes using a combination of conventional marker mutations and single-nucleotide polymorphisms (Fay 2006). For mutations that mapped to regions including genes known to mutate to the serotonin-resistant phenotype (goa-1, eat-16, ser-4, mod-1, and flp-1), we performed complementation tests with mutations in these genes and also amplified the candidate genes from the mutant strains and used DNA sequencing to identify mutant lesions (Table S1). The mutation emb-9(vs114), in addition to causing weak serotonin resistance, caused a temperature-sensitive lethal phenotype that facilitated mapping and complementation tests. elpc-3(vs119) was mapped to an ∼40-kb interval using single-nucleotide polymorphisms, and all genes within that interval were amplified and then sequences were determined from the mutant strain to identify the elpc-3 lesion. For the three Mos1 transposon insertion alleles, we used inverse PCR amplification of genomic DNA flanking the insert and DNA sequencing to determine that the insertions were in goa-1, mod-1, and abts-1.
Fluorescence microscopy
C. elegans was immobilized with 10 mM levamisole, and a Zeiss LSM 710 confocal microscope was used to obtain Z-stack images. The 3D reconstructions seen in Figure 3, Figure S1, and File S3 were created using Volocity software (Improvision).
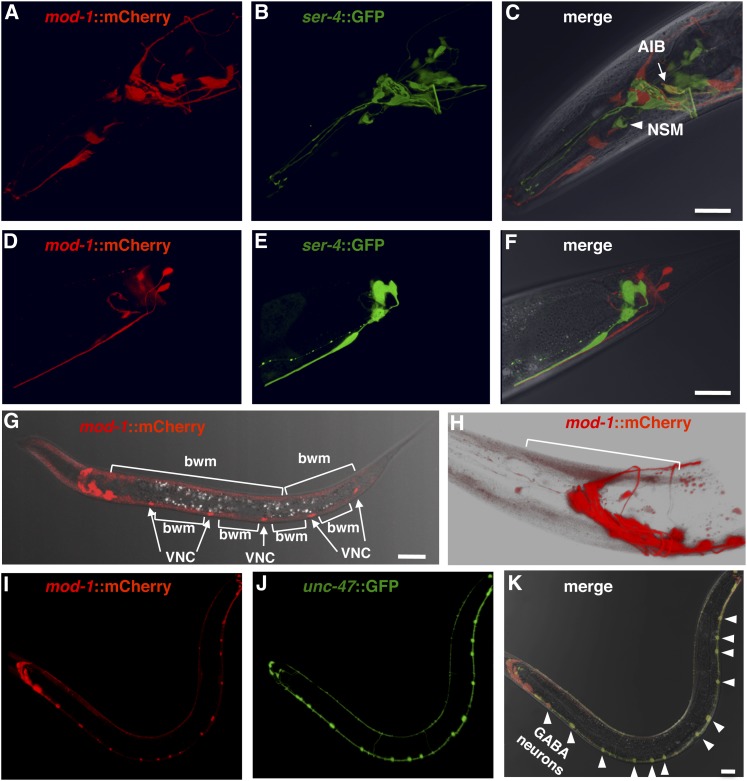
Figure 3 .
Expression patterns of mod-1 and ser-4 reporter transgenes are mostly distinct. Shown are confocal micrographs of transgenic animals in which the mod-1 promoter drives expression of the red fluorescent protein mCherry and the ser-4 promoter drives expression of green fluorescent protein (GFP). Red or green fluorescence and a merge of both superimposed on a brightfield image of the animal are shown, as indicated. (A–C) Adult head region. Arrow indicates AIB, the only cell type that reliably coexpressed both reporters. Arrowhead indicates the serotonergic NSM neurons. The double-labeling pattern seen in C is shown in more detail in File S3, a movie of a rotating three-dimensional confocal image of the head region. (D–F) Adult tail region. (G) mod-1:: mCherry fluorescence in an L3 larva superimposed on a brightfield image of the animal. Arrows point to mCherry-labeled ventral cord neurons (VNC), while brackets indicate labeled body-wall muscles (bwm). (H) mod-1::mCherry expression in adult head. The white background used allows weak red fluorescence in body-wall muscle to be visualized. Bracket indicates an individual muscle cell. (I–K) Lower magnification images of a young adult expressing mod-1::mCherry and an unc-47::GFP transgene that labels GABAergic neurons. Arrowheads in K point to the double-labeled GABA neurons of the ventral nerve cord that are most visible in this individual. Bars, 20 µm.
Results
Genetic screens for mutants with defects in response to exogenous serotonin
When C. elegans is placed in liquid medium it continuously flexes its body (File S1), and we found that addition of serotonin reduces these movements in a dose-dependent manner such that high concentrations eliminate almost all body bends except for small movements of the head (File S2).
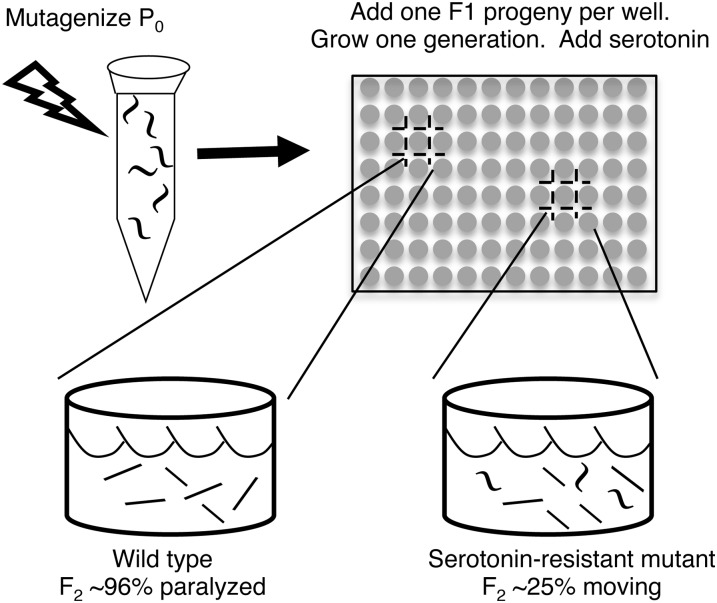
We performed several genetic screens for serotonin-resistant mutants that continue to move at high doses of exogenous serotonin. In one “clonal” strategy, schematized in Figure 1, individual F1 progeny of mutagenized animals were cultured for one generation in separate microtiter wells so that each well contained a brood of F2 animals sharing the same mutations. Serotonin (30 mM) was added to the cultures, and wells containing >20% serotonin-resistant animals were identified. We thus screened 6000 wells, representing 12,000 mutagenized haploid genomes, and identified 12 serotonin-resistant mutants.
Figure 1 .
Schematic diagram of one of four genetic screens used to isolate serotonin-resistant mutants of C. elegans. Individual F1 progeny of mutagenized animals, each heterozygous for different mutations, were cultured in microtiter wells. Each F1 self-fertilized to produce a brood of F2 progeny. Serotonin (30 mM) was added to paralyze ∼96% of animals in most wells (bottom left). Rare wells (bottom right) had ∼25% F2 progeny that continued to move vigorously because they were homozygous for a mutation causing serotonin resistance.
We performed additional screens in which F1 progeny were cultured in groups rather than individually, and their F2 progeny were screened en masse for individuals that were not paralyzed by 20 or 60 mM serotonin. This “nonclonal” strategy allowed animals representing many more mutagenized genomes to be screened but was complicated by the fact that, since even high concentrations of serotonin do not paralyze 100% of wild-type animals, a large number of false positives might be initially isolated that would then have to be discarded during subsequent retesting. To minimize this problem, the nonclonal screens were performed using a strain mutant for the serotonin reuptake transporter (SERT) MOD-5 (Ranganathan et al. 2001). SERT acts to terminate serotonin signaling by removing serotonin from the extracellular space, so a SERT-defective mutant has elevated serotonin signaling. We found that serotonin at concentrations >20 mM can paralyze nearly 100% of mod-5 mutant animals. The nonclonal screens used either a chemical mutagen (a total 42,000 mutagenized haploid genomes from screens performed at 20 or 60 mM serotonin) or Mos1 transposon mutagenesis (63,000 mutagenized haploid genomes screened at 60 mM serotonin). We thus isolated 24 more mutants. Table 1 lists details of all 36 serotonin-resistant mutants identified.
Serotonin-resistance mutations identify eight proteins, including two serotonin receptors, that affect response to exogenous serotonin
To identify the genes affected by the serotonin-resistance mutations, we performed a combination of genetic mapping, complementation tests, inverse PCR amplification in the case of transposon-insertion mutations, and PCR amplification of candidate genes followed by DNA sequence determination in the case of chemically induced mutations. We thus mapped 30 of the 36 mutations to specific chromosomes and subsequently identified the molecular lesions causing serotonin resistance in 19 of the mutants. In each case of a gene identified by a single mutation from our serotonin-resistance screens, we obtained a deletion or other independently isolated mutation for that gene and verified that it induced a similar serotonin-resistance phenotype.
The 19 serotonin-resistance mutations identified eight different proteins required for serotonin response (Table 2). MOD-1 is a serotonin-gated chloride channel (Ranganathan et al. 2000), while SER-4 is a G-protein-coupled serotonin receptor (Olde and McCombie 1997). GOA-1 is the C. elegans ortholog of the major neural G protein Gαo (Lochrie et al. 1991) and is a member of the Gαi/o class of G proteins thought to be activated by SER-4 (Olde and McCombie 1997). Serotonin resistance of MOD-1, SER-4, and GOA-1 mutants has been noted previously (Ségalat et al. 1995; Ranganathan et al. 2000; Hapiak et al. 2009) and suggests that serotonin acts directly on two different receptors to inhibit locomotion.
Table 2. Genes identified by serotonin-resistance mutations.
| Gene | No. of mutations isolated | Protein encoded |
|---|---|---|
| mod-1 | 3 | Serotonin-gated ion channel |
| ser-4 | 1 | G-protein-coupled serotonin receptor |
| goa-1 | 9 | Neural G protein α-subunit Gαo |
| eat-16 | 1 | Regulator of G protein signaling |
| abts-1 | 1 | Bicarbonate chloride exchanger |
| flp-1 | 2 | Neuropeptide |
| elpc-3 | 1 | Lysine acetyl transferase |
| emb-9 | 1 | Basement membrane collagen |
Five additional proteins that affect serotonin response were identified in our screens. EAT-16 is a G-protein regulator known to function with GOA-1 (Hajdu-Cronin et al. 1999) and thus could affect SER-4/GOA-1 signaling. ABTS-1 is a transmembrane chloride transporter that controls the chloride gradient across the plasma membrane (Bellemer et al. 2011) and thus could affect the function of the serotonin-gated chloride channel MOD-1. We note, however, that we isolated an abts-1 mutant in a screen performed using the mod-5 mutant background, and we have not detected any effect of abts-1 mutations on serotonin response in a wild-type background (data not shown). FLP-1 is a neuropeptide previously known to affect serotonin response (Nelson et al. 1998), ELPC-3 is the catalytic subunit of a lysine acetylase protein complex (Chen et al. 2009; Solinger et al. 2010), and EMB-9 is a collagen that is a component of basement membranes (Graham et al. 1997).
Our results suggest MOD-1 and SER-4 are serotonin receptors that inhibit C. elegans locomotion, and we propose that the other proteins detected in our genetic screens affect signaling by MOD-1 and/or SER-4. To lay the foundation for understanding of how all of the proteins mediate serotonin response, we first focused on how the two receptors affect locomotion.
SER-4 and MOD-1 act in parallel to mediate response to exogenous serotonin
We quantitated the effects of ser-4 and mod-1 null mutations on the ability of exogenous serotonin to paralyze C. elegans. When assayed under conditions similar to those of our genetic screens, in M9 buffer, the receptor single mutants were strongly serotonin resistant, with ∼80% of the mutant animals continuing to move at a serotonin concentration sufficient to paralyze all but 10% of the wild type (Figure 2A). While systematically varying the assay conditions, we found that the salts present in nematode growth media strongly inhibit serotonin response, so that ∼30-fold less serotonin was sufficient to paralyze animals if the assay was performed in water instead. By assaying in water, for the first time we were able to observe responses of C. elegans to exogenous serotonin at concentrations of serotonin below its solubility limit, greatly improving the reliability our serotonin response assays. We generated serotonin dose-response curves by placing young adults into microtiter wells with various concentrations of serotonin in water and analyzed the ability of serotonin to paralyze mutants lacking the receptors individually or in combination. Wild-type animals were paralyzed in a dose-dependent fashion by serotonin in water (Figure 2B). In this assay condition, the ser-4 and mod-1 single null mutants showed only weak serotonin resistance, but the ser-4; mod-1 double mutant showed much stronger serotonin resistance.
In a second approach to understanding the functional relationship between the two receptors, we generated a strain carrying a chromosomally integrated transgene with multiple copies of a mod-1 genomic clone, which presumably overexpresses MOD-1. We found that these mod-1(xs) animals were hypersensitive to paralysis by exogenous serotonin in water, in that mod-1(xs) animals were paralyzed by serotonin at a concentration ∼30-fold lower than that required to paralyze the wild type (Figure 2C). The mod-1(xs) transgene also caused a baseline paralysis of ∼25% of the animals in this assay even when no exogenous serotonin was added, possibly because of endogenous serotonin signaling through overexpressed MOD-1. When the mod-1(xs) transgene was combined with the ser-4(null) mutation, this baseline paralysis was suppressed, but the ser-4; mod-1(xs) strain still showed hypersensitivity to exogenous serotonin similar to that of the mod-1(xs) strain, as the slopes of the dose-response curves for the two strains were similar. Thus MOD-1 can mediate the ability of exogenous serotonin to paralyze animals even in the absence of SER-4.
Our results indicate that SER-4 and MOD-1 are the principal receptors that mediate the ability of serotonin to slow locomotion, as they are the only serotonin receptors so far identified by our genetic screen, and at least in high-salt buffer, null mutations for either receptor lead to strong serotonin resistance. When assayed in water, the ser-4; mod-1 double null mutant can still be paralyzed by sufficiently high concentrations of exogenous serotonin, so additional receptor(s) must also mediate the ability of serotonin to slow of locomotion in this condition. SER-4 and MOD-1 appear to function in parallel to each other to mediate slowing of locomotion since null mutants for the individual receptors are not as serotonin resistant as is the ser-4; mod-1 double mutant.
mod-1 and ser-4 are expressed in almost completely non-overlapping sets of cells
To further clarify the relationship between SER-4 and MOD-1, we examined the expression patterns of the mod-1 and ser-4 genes. Because the specific muscles, motor neurons, and interneurons that control C. elegans locomotion have been well characterized (Von Stetina et al. 2006), understanding precisely which of these cells express the receptors serves as a starting point for understanding how each receptor affects locomotion. Previous studies have generated transgenic animals that express the green fluorescent protein (GFP) from the mod-1 or ser-4 promoters and have reported the identification of some cells that express each reporter (Ranganathan et al. 2000; Tsalik et al. 2003; Carnell et al. 2005; Harris et al. 2009). However, these studies do not entirely agree, probably because of the weak and variable expression from the extrachromosomal transgenes used. We used chromosomally integrated mod-1:mCherry and ser-4::GFP transgenes that gave strong stable expression and allowed us to directly compare the expression of mod-1 and ser-4 reporters in the same animals.
We found that the mod-1 and ser-4 reporter transgenes were expressed in almost non-overlapping sets of cells (Figure 3, A–F, and File S3). In agreement with previous work (Ranganathan et al. 2000; Tsalik et al. 2003; Carnell et al. 2005; Harris et al. 2009), we found that mod-1 and ser-4 are each expressed in a subset of head and tail neurons. Using double-transgenic animals, we found that there was only one type of cell that reliably coexpressed both reporter transgenes (Figure 3C and File S3). The coexpressing cells were a bilaterally symmetric pair of head neurons previously identified as either RIB (Tsalik et al. 2003) or AIB (Harris et al. 2009), two interneuron types that have very similar cell-body positions and process morphology. Harris et al. (2009) reported that mod-1 functions in AIB, and our analysis of the cell-body position of the double-labeled cell is also most consistent with AIB. RIB and AIB are among the many head interneurons that directly or indirectly synapse onto the command interneurons to control whether C. elegans moves forward or backward (White et al. 1986; Von Stetina et al. 2006). We identified some cells that had not previously been reported to express mod-1 or ser-4 reporters. Specifically, we saw mod-1::mCherry expression in RME neurons (Figure S1A) and ser-4::GFP expression in the serotonergic NSM neurons (Figure 3C) and in the vm2 egg-laying muscles (Figure S1B).
The mod-1::mCherry reporter was expressed in muscles and neurons that directly control locomotion. By examining larvae at the L3 stage or younger, we saw that the mod-1 reporter was expressed strongly in all body-wall muscles, the cells that directly execute body bends during C. elegans locomotion (Figure 3G). We then found that in L4 larvae and adults this body-wall muscle expression was much reduced, but still detectable (Figure 3H). It was previously reported (Ranganathan et al. 2000) that a mod-1 reporter was expressed in unidentified cells of the ventral nerve cord, which contains motor neurons that synapse onto the body-wall muscles. We also saw ventral cord neurons expressing mod-1::mCherry (Figure 3I). To identify these cells, we generated strains that carried the mod-1::mCherry transgene plus an unc-47::GFP transgene to label the GABAergic motor neurons (Figure 3J) or an unc-17::GFP transgene to label the cholinergic motor neurons (data not shown). MOD-1 expression overlapped perfectly in the ventral cord with the unc-47::GFP-expressing neurons, with mod-1::mCherry expressed in both VD and DD classes of GABAergic motor neurons (Figure 3K). These motor neurons release GABA directly onto the body-wall muscles to help generate body bends during locomotion (Von Stetina et al. 2006). We note that the mod-1 reporter is not expressed in all 26 GABAergic neurons; in the head, the only overlap between the mod-1 and unc-47 reporter expression is in RME interneurons (Figure S1A).
In summary, the ser-4 reporter transgene expression was restricted to cells, including several types of head interneurons, that likely affect locomotion only indirectly. In contrast, the mod-1 expression pattern included the body-wall muscles and GABAergic ventral cord neurons that directly control locomotion. The only overlap in the mod-1 and ser-4 expression patterns appeared to be in the AIB head interneurons.
MOD-1 and SER-4 serotonin receptors together mediate effects of optogenetically released endogenous serotonin on locomotion
MOD-1 and SER-4 are required for exogenous serotonin to paralyze C. elegans. Do MOD-1 and SER-4 have a normal physiological role in the control of locomotion by endogenously released serotonin? We initially addressed this question by optogenetically stimulating neurotransmitter release from the serotonergic neurons of C. elegans and measuring whether MOD-1 and SER-4 affect the resulting locomotion response.
Our optogenetic assays used a chromosomally integrated transgene, ljIs102 (Ezcurra et al. 2011), to express the blue-light-activated cation channel, channelrhodopsin-2 (ChR2) fused to the yellow fluorescent protein (YFP). A fragment of the promoter for the tph-1 gene (Sze et al. 2000) was used to drive expression of ChR2::YFP specifically in serotonergic NSM and ADF neurons (Figure 4A). We performed all of our ChR2 assays in genetic backgrounds mutant for the C. elegans endogenous blue light receptor LITE-1 (Edwards et al. 2008), so that any locomotion responses to blue light should be entirely caused by ChR2 activation.
Upon stimulation by blue light to activate ChR2 and depolarize NSM and ADF, animals were nearly paralyzed (File S4). To quantitate this locomotion response, we counted the number of body bends during the first 30 sec after turning on blue light illumination. To further ensure that the effects measured were caused by ChR2 activity, we assayed animals grown either in the presence or absence of all-trans retinal, a cofactor required for ChR2 activity. We found that ChR2 activation of NSM and ADF caused a 5.7-fold decrease in body bends (Figure 4B). This slowing of locomotion appeared to depend absolutely on release of serotonin because it was absent in a mutant for bas-1, which encodes an enzyme required for serotonin and dopamine biosynthesis. We note that in this experiment we were unable to use a tph-1 mutation, which would block only serotonin and not dopamine biosynthesis, because the ChR2 transgene lies on chromosome II too close to the tph-1 gene to allow the necessary strain to be constructed. However, since the NSM and ADF neurons make serotonin and not dopamine, the bas-1 result strongly suggests that ChR2 stimulation caused slowing via release of serotonin.
Our optogenetic assays showed that mod-1 and ser-4 together are required for the effects of endogenous serotonin on locomotion (Figure 4B). In the mod-1 or ser-4 mutant backgrounds, ChR2 activation caused only a 1.8- or 3.1-fold decrease in body bends, respectively. In a genetic background mutant for both mod-1 and ser-4, ChR2 activation caused little if any reduction in body bends (P > 0.05).
MOD-1 and SER-4 serotonin receptors together mediate hyper-enhanced slowing, an effect of endogenous serotonin on locomotion
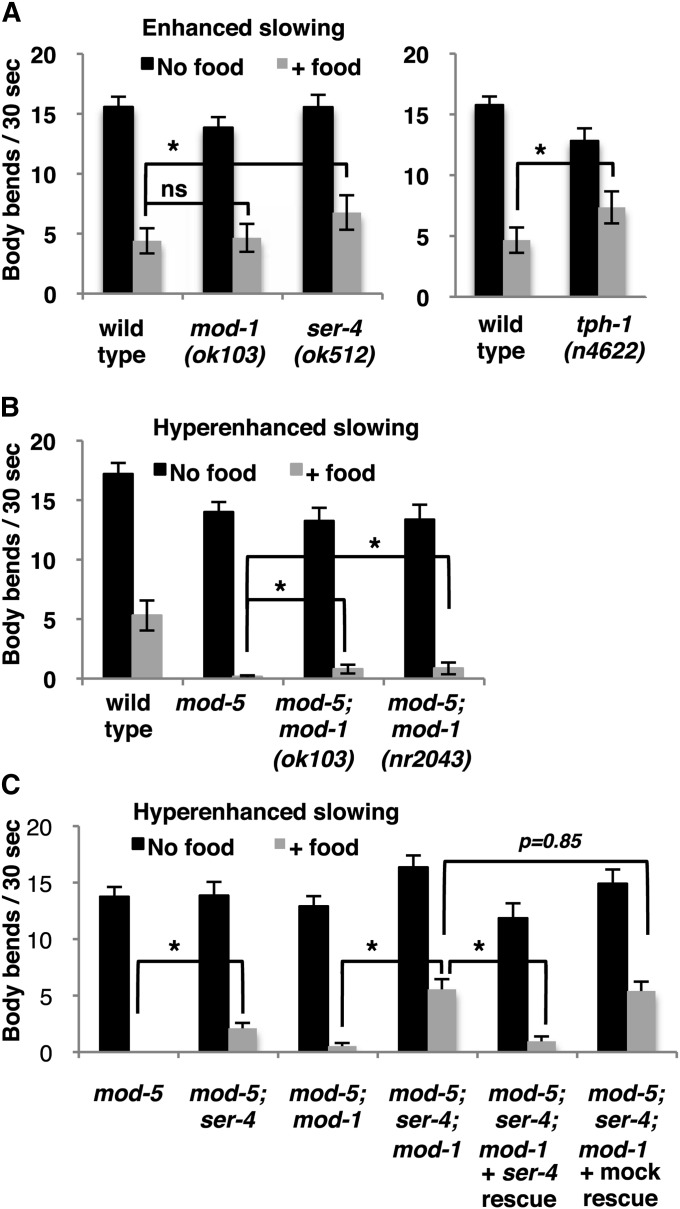
The optogenetic assay showed that MOD-1 and SER-4 can mediate effects of endogenously released serotonin on locomotion. For a more physiological assay of endogenous serotonin signaling, we analyzed how SER-4 and MOD-1 mutations affect a locomotion-slowing behavior known to depend on natural release of endogenous serotonin. C. elegans slows when it encounters food in the form of a lawn of E. coli bacteria on an agar plate. If the animals are well fed, they show a modest “basal slowing,” which depends on the release of the neurotransmitter dopamine (Sawin et al. 2000). If the animals assayed are food deprived for 30 min prior to being presented with food, they show a stronger “enhanced slowing” response, in which the additional slowing depends on release of serotonin from the NSM and other serotonergic neurons (Sawin et al. 2000). Finally, an even more dramatic hyper-enhanced slowing response occurs in a null mutant for the serotonin reuptake transporter MOD-5: since MOD-5 activity terminates serotonin signaling, exaggerated responses to serotonin are seen in mod-5 mutants (Ranganathan et al. 2001). As shown in Figure 5B, food-deprived wild-type animals slowed their locomotion by ∼75% when they encountered food, demonstrating enhanced slowing, while food-deprived mod-5 animals were almost completely paralyzed when they encountered food, demonstrating hyper-enhanced slowing.
Figure 5 .
Effects of SER-4 and MOD-1 receptors on enhanced and hyper-enhanced slowing behavioral responses dependent on endogenous serotonin. (A) Enhanced slowing was measured by first food-depriving young adults for 30 min, transferring the animals to agar plates with or without food (a lawn of bacteria), and scoring locomotion by measuring the rate of body bends. mod-1(ok103) (shown) had no significant defect (P = 0.76, Student’s t-test), and two other mod-1 alelles gave similar results (data not shown). The ser-4 deletion mutation caused a modest but significant (P = 0.01) defect in enhanced slowing. As a positive control, the serotonin-deficient mutant tph-1(n4622) was assayed and failed to slow as much as did the wild type (P = 0.02). (B and C) Hyper-enhanced slowing was measured by using the same assay on animals carrying mutations in the MOD-5 serotonin reuptake transporter. (B) Upon exposure to food, wild-type animals slowed, while mod-5 mutants demonstrated hyper-enhanced slowing by almost completely stopping all body bends. Two independent mod-1 deletion mutants were partially defective for hyper-enhanced slowing. (C) The ser-4 deletion mutation caused defective hyper-enhanced slowing and added to the defect of the mod-1(ok103) mutant. This additional defect was rescued by a ser-4 transgene but not by a mock ser-4 transgene containing a frameshift mutation that blocks SER-4 expression. Brackets with asterisks indicate significant differences (P < 0.05, Student’s t-test). ns, not significant.
mod-1 was originally defined by Ranganathan et al. (2000) based on a mutant strain that was both serotonin resistant and defective in the enhanced slowing response. Using the serotonin-resistant phenotype, these investigators mapped and cloned the mod-1 gene and found it to encode a serotonin-gated chloride channel. Subsequent work has indicated that mod-1 mutants are not defective in the enhanced slowing response. First, the enhanced-slowing defect of the original mod-1 strain carrying the n3034 mutation proved to be conferred by a locus genetically separable from the mod-1 mutation as defined by the serotonin-resistant phenotype (D. Omura, personal communication). This observation indicated that the enhanced-slowing defect in this mod-1 strain was caused by a background mutation, now named n4954. Second, we have been unable to reproduce the enhanced-slowing defects reported by Ranganathan et al. (2000) for the mod-1(ok103) and mod-1(nr2043) strains. For example, Figure 5A shows our data for mod-1(ok103) animals. We do not know why the observations of Ranganathan et al. (2000) concerning the mod-1(ok103) and mod-1(nr2043) strains differ from our more recent observations.
We saw a very weak enhanced-slowing defect for ser-4 (Figure 5A) and also, at most, a weak enhanced-slowing defect in the mod-1; ser-4 double mutant (data not shown). To help understand if the enhanced-slowing response truly depends on serotonin, we measured enhanced slowing in a null allele for the serotonin biosynthesis gene tph-1, mutations of which were not available at the time of the previously published studies of enhanced slowing (Figure 5A, right). In the absence of food, this tph-1 deletion mutant moved somewhat more slowly than did the wild type. Despite this complication, the tph-1 mutant failed to slow in the presence of food to the low rate of body bends seen in the wild type (P = 0.002) and thus did show the expected enhanced-slowing defect.
Since the hyper-enhanced slowing response shows more dramatic effects of endogenous serotonin on locomotion than does the enhanced-slowing response, we tested how ser-4 and mod-1 null mutations affected hyper-enhanced slowing. Whereas food-deprived mod-5 single mutants were essentially paralyzed when they encountered food, two independent deletion alleles of mod-1 were significantly defective in this hyper-enhanced slowing response (Figure 5B). The ser-4 deletion mutant was also defective in hyper-enhanced slowing, and combining ser-4 and mod-1 deletions together produced yet a stronger defect (Figure 5C). Since we did not have a second independent deletion allele of ser-4, we used a ser-4 rescuing transgene to check if the effect of this deletion on enhanced slowing was indeed caused by the loss of ser-4 function. We found that the ser-4 transgene rescued the mod-5; ser-4; mod-1 triple-mutant phenotype in the slowing assay to the phenotype of the mod-5; mod-1 double mutant. A mock ser-4 transgene, with a frameshift mutation in the ser-4 coding sequences, showed no such rescue (Figure 5C).
Our results show that MOD-1 and SER-4 together mediate hyper-enhanced slowing, a behavioral effect of endogenous serotonin release.
Discussion
A genetic system for discovery and functional analysis of serotonin-signaling proteins
We developed large-scale genetic screens to isolate mutants with defects in serotonin response. Many past studies of serotonin signaling in C. elegans have analyzed gene knockouts for worm homologs of serotonin-signaling proteins already known from studies in vertebrates. For example, mutants defective in enzymes required for serotonin biosynthesis and for serotonin receptor homologs have revealed a number of behaviors that depend on serotonin (Sze et al. 2000; Carnell et al. 2005; Dempsey et al. 2005; Hobson et al. 2006; Dernovici et al. 2007; Hapiak et al. 2009; Harris et al. 2009). However, the greatest potential for C. elegans genetics to illuminate mechanisms of serotonin signaling likely lies in genetic screens to discover new serotonin-signaling proteins not previously known from vertebrate studies.
We chose locomotion as a serotonin-regulated behavior in C. elegans suitable for analysis by large-scale genetic screens. The simplest approach to such screens, applying serotonin to descendants of mutagenized animals and screening for animals that were not paralyzed, would have produced an overwhelming number of false-positive isolates that did not carry serotonin-resistance mutations because even high concentrations of serotonin do not paralyze 100% of wild-type animals. We developed two alternative approaches: clonal genetic screens and nonclonal screens using a mutation in the MOD-5 serotonin reuptake transporter in the genetic background. These approaches allowed us to screen animals representing 117,000 mutagenized haploid genomes and to isolate 36 mutants resistant to the paralytic effects of exogenous serotonin. Although serotonin is used as a neurotransmitter in perhaps all organisms with a nervous system, to our knowledge no other large-scale systematic screens for mutants with serotonin-signaling defects have been performed.
We developed new assays for measuring the effects of serotonin on locomotion to assess how specific mutations affect serotonin response. We discovered that C. elegans responds to exogenous serotonin at ∼30-fold lower concentrations when it is dissolved in media lacking any salts instead of in the C. elegans media that have previously been used for serotonin response assays. This finding allowed us to perform more reliable assays in which serotonin was used at concentrations below its solubility limit. Even when applied in water, exogenous serotonin must be presented at concentrations >100 μM to affect wild-type C. elegans locomotion, raising concern that the responses seen might not be related to the physiological effects of endogenous serotonin. For this reason, we developed a new serotonin response assay based on channelrhodopsin-2 stimulation of endogenous serotonin release. We also used hyper-enhanced slowing behavior as a further assay of endogenous serotonin signaling. mod-1 and ser-4 mutations caused defects in all four assays that we used: response to exogenous serotonin in high or low salt liquid, the optogenetic assay, and the hyper-enhanced slowing assay. The strongest defects were seen when both receptors were mutated, and in some assays mutants lacking only a single receptor failed to show significant defects. Our results suggest mutations in other genes isolated based on defects for response to exogenous serotonin likely also affect endogenous serotonin signaling. However, any mutation that affects only one receptor-signaling pathway might need to be combined with a second mutation affecting the other receptor-signaling pathway to reveal effects on endogenous serotonin signaling.
Eight proteins required for serotonin control of locomotion in C. elegans
Our genetic screens identified eight genes required for serotonin response. Three of these genes encode the G-protein-coupled serotonin receptor SER-4, its potential target Gαo protein GOA-1, and the G-protein regulator EAT-16, previously known to indirectly affect Gαo signaling (Hajdu-Cronin et al. 1999). Two more genes encode the serotonin-gated chloride channel MOD-1 and the chloride transporter ABTS-1, previously known to control chloride channel function by affecting the chloride gradient across the plasma membrane (Bellemer et al. 2011).
How the remaining three proteins identified in our screens might affect serotonin response is less clear. EMB-9 is a basement membrane collagen (Graham et al. 1997). The NSM neurosecretory neurons release serotonin directly onto a basement membrane surrounding the pharynx, so the serotonin must traverse the basement membrane to reach target cells outside the pharynx (Axäng et al. 2008). Perhaps exogenous serotonin also enters C. elegans through its pharynx by traversing this same basement membrane, and EMB-9 affects diffusion of serotonin through the basement membrane.
The flp-1 gene encodes several FMRFamide-related neuropeptides. Previous studies have shown that flp-1 mutations decrease the ability of exogenous serotonin to slow locomotion (Nelson et al. 1998) and that flp-1 appears to potentiate the ability of both endogenous and exogenous serotonin to stimulate egg-laying behavior (Waggoner et al. 2000). flp-1 is expressed in a subset of head interneurons (Nelson et al. 1998), but the mechanism by which FLP-1 released from these cells directly or indirectly affects serotonin response in the locomotion and egg-laying systems of C. elegans remains to be determined.
The final protein identified by our genetic screens is ELPC-3, a subunit of a multisubunit complex known as elongator based on its initial discovery in yeast as a factor involved in transcriptional elongation (Otero et al. 1999). The ELPC-3 subunit has lysine acetylase activity and is highly evolutionarily conserved among all eukaryotes, with >65% identity among orthologs in various species from yeast to humans (Wittschieben et al. 1999). In animals, elongator is found in the nervous system, and mutations in the human elongator subunit Elp1 cause the neurodegenerative disease familial dysautonomia (Slaugenhaupt and Gusella 2002). Elongator is found in both the nucleus and the cytosol and has been shown to acetylate tubulin in C. elegans (Solinger et al. 2010). Elongator has not previously been implicated in serotonin signaling. However, a genome-wide association study found an association of a polymorphism upstream of the Elp3 gene with suicide ideation during treatment with an antidepressant that acts as a selective serotonin reuptake inhibitor (Perroud et al. 2010). We suggest that elongator positively regulates serotonin signaling by acetylating and thus activating another protein that directly mediates serotonin signaling. Alternatively, elongator might very indirectly affect serotonin signaling since, for example, it is required for proper modification of transfer RNA in C. elegans (Chen et al. 2009) and might cause subtle defects in protein translation in neurons.
Our genetic screens have not yet reached a saturation scale. Nevertheless, as shown in Table 2, we recovered nine alleles of the Gαo gene goa-1, despite the fact that strong loss-of-function goa-1 mutants are relatively sick and slow growing. The relatively high rate of isolating goa-1 mutants is likely because of their strong serotonin-resistance phenotype. Five genes were identified in our screens by only single alleles. Null mutants for these five genes are relatively healthy and show serotonin resistance similar to that seen in the screen isolates, and the genes are not particularly small, so there is no obvious reason why alleles for these genes should be rare. Of the 36 serotonin-resistant mutants that we isolated, 17 have not yet been assigned to genes, and at least 11 of the 17 show a serotonin-resistance phenotype sufficiently strong to allow genetic mapping. We expect that further analysis of these mutants will identify additional serotonin-signaling genes.
Two receptors act in parallel to allow extrasynaptic serotonin to inhibit locomotion
We have initiated analysis of the serotonin-signaling proteins identified in our genetic screens with studies of the serotonin receptors. Because the other serotonin-signaling proteins presumably affect signaling by one or more serotonin receptor, understanding how serotonin receptors control locomotion should provide the basis for assigning the remaining signaling proteins to the receptors that they regulate or to which they respond.
MOD-1 and SER-4 are the principal receptors that slow locomotion in response to exogenous serotonin and to the presumably high concentrations of endogenous serotonin released by optogenetic stimulation of the NSM and ADF neurons or in the hyper-enhanced slowing assay. MOD-1 and SER-4 were the only serotonin receptors identified in our genetic screens for defects in response to exogenous serotonin, and knocking out both of these receptors together eliminated slowing in response to optogenetic release of endogenous serotonin (Figure 4). The ser-4; mod-1 double knockout might also completely eliminate the serotonin-induced hyper-enhanced slowing response: the residual slowing that occurs in the double knockout in this assay (Figure 5) might be entirely caused by the dopamine-induced basal slowing response. We note, however, that mod-1 and ser-4 knockouts had little or no effect on enhanced slowing, a more natural behavioral effect of presumably lower levels of endogenous serotonin. Furthermore, application of high concentrations of exogenous serotonin caused residual slowing of ser-4; mod-1 double null mutants (Figure 2), again suggesting that other serotonin receptors also contribute to the effects of serotonin on locomotion. In addition to SER-4 and MOD-1, there are three other serotonin receptor homologs in C. elegans, and one report suggests that the SER-1 receptor has a significant effect on slowing in response to exogenous serotonin (Dernovici et al. 2007). However, another study did not find a strong effect of SER-1 (Hapiak et al. 2009), and we found that a ser-1 null mutation individually or in combination with mod-1 or ser-4 mutations had little effect in our assays for response to exogenous serotonin (data not shown). Thus existing assays show little effect of other serotonin receptors on locomotion compared to the strong effects of MOD-1 and SER-4.
Our genetic and expression-pattern studies suggest SER-4 and MOD-1 act independently of each other to slow locomotion. In one assay of response to exogenous serotonin and two assays of response to endogenously released serotonin, we found that ser-4; mod-1 double null mutants had more severe defects in serotonin response than did the single mutants lacking only one receptor. These results are consistent with the hypothesis that SER-4 and MOD-1 act independently and in parallel to mediate slowing in response to serotonin. In agreement with this hypothesis, ser-4 and mod-1 appear to be expressed in almost entirely non-overlapping sets of cells, with ser-4 expressed in many head interneurons and mod-1 expressed in different interneurons as well as in the GABAergic motor neurons and body-wall muscles that directly cause locomotion. Either SER-4 and MOD-1 act in parallel in these different cells, or they act in parallel in the only two cells in which they are coexpressed, the AIB interneurons, which synapse onto command interneurons and thus could mediate the effects of serotonin on locomotion. The precise sites of action and mechanism by which the two receptors act to slow locomotion remain to be resolved.
Expression of the SER-4 and MOD-1 serotonin receptors does not appear to be limited to cells that receive synapses from serotonergic neurons. There are six pairs of neurons that contain serotonin—NSM, ADF, HSN, RIH, AIM, and VC4/5 (Sawin et al. 2000; Duerr et al. 2001; Jafari et al. 2011)—and they form significant synapses onto 20 other types of neurons and onto the vm2 egg-laying muscle cells (White et al. 1986). We found that the vm2 muscles express ser-4::GFP (Figure S1B). While we have not identified all of the neurons that express our ser-4 and mod-1 reporters, the neurons that we have identified do not correspond to any of the 20 neuron types postsynaptic to serotonergic neurons. For example, the GABAergic motor neurons of the ventral cord and the body-wall muscles that express mod-1 do not receive synapses from serotonin neurons and are quite distant from any serotonin neurons. Thus, serotonin might diffuse from its release sites to act on distant cells. Consistent with this idea, previous work has shown that serotonin from the NSM and ADF neurons travels extrasynaptically to the RIH and AIM neurons, which use the serotonin uptake transporter MOD-5 to accumulate serotonin (Jafari et al. 2011). Furthermore, the neurons with by far the largest serotonin content in C. elegans are the NSM neurons, which are neurosecretory cells with no postsynaptic partners. The NSMs appear to be the principal neurons that release serotonin to cause slowing of locomotion (Sawin et al. 2000). Studies of vertebrate brain are also consistent with the idea that serotonin diffuses and signals extrasynaptically (Bunin and Wightman 1998).
Our results lead to a model in which the NSM neurosecretory cells release serotonin extrasynaptically to diffuse across a basement membrane with an EMB-9 collagen component that facilitates diffusion across the membrane. The serotonin continues to diffuse to distant target cells and acts in parallel on different serotonin receptors, likely found on different cells. Signaling through the G-protein-coupled receptor SER-4 and its target G protein GOA-1, potentially in head interneurons, would provide one mechanism to slow locomotion. Signaling via the serotonin-gated chloride channel MOD-1, potentially on the GABAergic motor neurons and/or body-wall muscles, would provide a second independent mechanism to slow locomotion. The remaining serotonin response proteins identified in our genetic screens could act to facilitate either the MOD-1- or SER-4-signaling pathways. It should be possible using the assays that we describe to determine for each new C. elegans serotonin-signaling protein which receptor pathway is affected. It would then be possible to determine if the mammalian homologs of these proteins play corresponding roles in serotonin signaling in the brain and if deficits in any of these proteins are involved in human neuropsychiatric disorders, such as depression.
Supplementary Material
Acknowledgments
We thank Bill Schafer and the members of his laboratory for strains carrying the ser-4::GFP transgene ljIs570 and the tph-1::Chr2-YFP transgene ljIs102; Nirada Koonrugsa for identifying vs114 as an allele of emb-9; and Jessica Tanis for generating the mod-1 transgene vsIs123. We also thank the Caenorhabditis Genetics Center for additional strains and the Yale Center for Cellular and Molecular Imaging for use of the LSM 710 microscope. This work was supported by National Institutes of Health grants NS036918 (to M.R.K.) and GM24663 (to H.R.H.). H.R.H. is an Investigator of the Howard Hughes Medical Institute.
Footnotes
Communicating editor: D. I. Greenstein
Literature Cited
- Axäng C., Rauthan M., Hall D. H., Pilon M., 2008. Developmental genetics of the C. elegans pharyngeal neurons NSML and NSMR. BMC Dev. Biol. 8: 38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellemer A., Hirata T., Romero M. F., Koelle M. R., 2011. Two types of chloride transporters are required for GABAA receptor-mediated inhibition in C. elegans. EMBO J. 30: 1852–1863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulin T., Bessereau J. L., 2007. Mosl-mediated insertional mutagenesis in Caenorhabditis elegans. Nat. Protoc. 2: 1276–1287 [DOI] [PubMed] [Google Scholar]
- Brenner S., 1974. The genetics of Caenorhabditis elegans. Genetics 77: 71–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bunin M. A., Wightman R. M., 1998. Quantitative evaluation of 5-hydroxytryptamine (serotonin) neuronal release and uptake: an investigation of extrasynaptic transmission. J. Neurosci. 18: 4854–4860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carnell L., Illi J., Hong S. W., McIntire S. L., 2005. The G-protein-coupled serotonin receptor SER-1 regulates egg laying and male mating behaviors in Caenorhabditis elegans. J. Neurosci. 25: 10671–10681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chase D. L., Koelle M. R., 2007. Biogenic amine neurotransmitters in C. elegans (February 20, 2007), in WormBook, edited by The C. elegans Research Community, doi/10.1895/wormbook.1.132.1, http://www.wormbook.org
- Chen C., Tuck S., Byström A. S., 2009. Defects in tRNA modification associated with neurological and developmental dysfunctions in Caenorhabditis elegans elongator mutants. PLoS Genet. 5: e1000561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowen P. J., 2008. Serotonin and depression: Pathophysiological mechanism or marketing myth? Trends Pharmacol. Sci. 29: 433–436 [DOI] [PubMed] [Google Scholar]
- Dempsey C. M., Mackenzi S. M., Gargus A., Blanco G., Sze S. J., 2005. Serotonin (5HT), fluoxetine, imipramine and dopamine target distinct 5HT receptor signaling to modulate Caenorhabditis elegans egg-laying behavior. Genetics 169: 1425–1436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dernovici S., Starc T., Dent J. A., Ribeiro P., 2007. The serotonin receptor SER-1 (5HT2ce) contributes to the regulation of locomotion in Caenorhabditis elegans. Dev. Neurobiol. 67: 189–204 [DOI] [PubMed] [Google Scholar]
- Duerr J. S., Gaskin J., Rand J. B., 2001. Identified neurons in C. elegans coexpress vesicular transporters for acetylcholine and monoamines. Am. J. Physiol. Cell Physiol. 280: C1616–C1622 [DOI] [PubMed] [Google Scholar]
- Edwards S.L., Charlie N. K., Milfort M. C., Brown B. S., Gravlin C. N., et al. , 2008. A novel molecular solution for ultraviolet light detection in Caenorhabditis elegans. PLoS Biol. 6: e198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezcurra M., Tanizawa Y., Swoboda P., Schafer W. R., 2011. Food sensitizes C. elegans avoidance behaviours through acute dopamine signalling. EMBO J. 30: 1110–1122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fay D., 2006. Genetic mapping and manipulation: Chapter 1–introduction and basics (February 17, 2006), WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/wormbook.1.90.1, http://www.wormbook.org
- Graham P. L., Johnson J. J., Wang S., Sibley M. H., Gupta M. C., et al. , 1997. Type IV collagen is detectable in most, but not all, basement membranes of Caenorhabditis elegans and assembles on tissues that do not express it. J. Cell Biol. 137: 1171–1183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajdu-Cronin Y. M., Chen W. J., Patikoglou G., Koelle M. R., Sternberg P. W., 1999. Antagonism between Goα and Gqα in Caenorhabditis elegans: the RGS protein EAT-16 is necessary for Goα signaling and regulates Gqα activity. Genes Dev. 13: 1780–1793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hapiak V. M., Hobson R. J., Hughes L., Smith K., Harris G., et al. , 2009. Dual excitatory and inhibitory serotonergic inputs modulate egg laying in Caenorhabditis elegans. Genetics 181: 153–163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris G. P., Hapiak V. M., Wragg R. T., Miller S. B., Hughes L. J., et al. , 2009. Three distinct amine receptors operating at different levels within the locomotory circuit are each essential for the serotonergic modulation of chemosensation in Caenorhabditis elegans. J. Neurosci. 29: 1446–1456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hobson R. J., Hapiak V. M., Xiao H., Buehrer K. L., Komuniecki P. R., et al. , 2006. SER-7, a Caenorhabditis elegans 5–HT7-like receptor, is essential for the 5-HT stimulation of pharyngeal pumping and egg laying. Genetics 172: 159–169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horvitz H. R., Chalfie M., Trent C., Sulston J. E., Evans P. D., 1982. Serotonin and octopamine in the nematode Caenorhabditis elegans. Science 216: 1012–1014 [DOI] [PubMed] [Google Scholar]
- Jafari G., Xie Y., Kullyev A., Liang B., Sze J. Y., 2011. Regulation of extrasynaptic 5-HT by serotonin reuptake transporter function in 5-HT-absorbing neurons underscores adaptation behavior in Caenorhabditis elegans. J. Neurosci. 31: 8948–8957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kia H. K., Brisorgueil M. J., Hamon M., Calas A., Vergé D., 1996. Ultrastructural localization of 5-hydroxytryptamine1A receptors in the rat brain. J. Neurosci. Res. 46: 697–708 [DOI] [PubMed] [Google Scholar]
- Lochrie M. A., Mendel J. E., Sternberg P. W., Simon M. I., 1991. Homologous and unique G protein alpha subunits in the nematode Caenorhabditis elegans. Cell Regul. 2: 135–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millan M. J., Marin P., Bockaert J., Mannoury la Cour C., 2008. Signaling at G-protein-coupled serotonin receptors: recent advances and future research directions. Trends Pharmacol. Sci. 29: 454–464 [DOI] [PubMed] [Google Scholar]
- Nelson L. S., Rosoff M. L., Li C., 1998. Disruption of a neuropeptide gene, flp-1, causes multiple behavioral defects in Caenorhabditis elegans. Science 281: 1686–1690 [DOI] [PubMed] [Google Scholar]
- Olde B., McCombie W. R., 1997. Molecular cloning and functional expression of a serotonin receptor from Caenorhabditis elegans. J. Mol. Neurosci. 8: 53–62 [DOI] [PubMed] [Google Scholar]
- Otero G., Fellows J., Li Y., de Bizemont T., Dirac A. M., et al, 1999. Elongator, a multisubunit component of a novel RNA polymerase II holoenzyme for transcriptional elongation. Mol. Cell 3: 109–118 [DOI] [PubMed] [Google Scholar]
- Perroud N., Uher R., Ng M. Y., Guipponi M., Hauser J., et al. , 2010. Genome-wide association study of increasing suicidal ideation during antidepressant treatment in the GENDEP project. Pharmacogenomics J. 12: 68–77 [DOI] [PubMed] [Google Scholar]
- Ranganathan R., Cannon S. C., Horvitz H. R., 2000. MOD-1 is a serotonin-gated chloride channel that modulates locomotory behaviour in C. elegans. Nature 408: 470–475 [DOI] [PubMed] [Google Scholar]
- Ranganathan R., Sawin E. R., Trent C., Horvitz H. R., 2001. Mutations in the Caenorhabditis elegans serotonin reuptake transporter MOD-5 reveal serotonin-dependent and- independent activities of fluoxetine. J. Neurosci. 21: 5871–5884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawin E. R., Ranganathan R., Horvitz H. R., 2000. C. elegans locomotory rate is modulated by the environment through a dopaminergic pathway and by experience through a serotonergic pathway. Neuron 26: 619–631 [DOI] [PubMed] [Google Scholar]
- Schafer W. R., Kenyon C. J., 1995. A calcium-channel homologue required for adaptation to dopamine and serotonin in Caenorhabditis elegans. Nature 375: 73–78 [DOI] [PubMed] [Google Scholar]
- Ségalat L., Elkes D. A., Kaplan J. M., 1995. Modulation of serotonin-controlled behaviors by Go in Caenorhabditis elegans. Science 267: 1648–1651 [DOI] [PubMed] [Google Scholar]
- Slaugenhaupt S. A., Gusella J. F., 2002. Familial dysautonomia. Curr. Opin. Genet. Dev. 12: 307–311 [DOI] [PubMed] [Google Scholar]
- Solinger J. A., Paolinelli R., Klöss H., Scorza F. B., Marchesi S., et al. , 2010. The Caenorhabditis elegans Elongator complex regulates neuronal alpha-tubulin acetylation. PLoS Genet. 6: e1000820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sulston J., Hodgkin J., 1988. Methods, pp. 587–606 in The Nematode Caenorhabditis elegans, edited by Wood W. B. and the Community of C. elegans Researchers. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY [Google Scholar]
- Sze J. Y., Victor M., Loer C., Shi Y., Ruvkun G., 2000. Food and metabolic signaling defects in a Caenorhabditis elegans serotonin-synthesis mutant. Nature 403: 560–564 [DOI] [PubMed] [Google Scholar]
- Tsalik E. L., Niacaris T., Wenick A. S., Pau K., Avery L., et al. , 2003. LIM homeobox gene-dependent expression of biogenic amine receptors in restricted regions of the C. elegans nervous system. Dev. Biol. 263: 81–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Von Stetina S. E., Treinin M., Miller D. M., III 2006. The motor circuit. Int. Rev. Neurobiol. 69: 125–167 [DOI] [PubMed] [Google Scholar]
- Waggoner L. E., Hardaker L. A., Golik S., Schafer W. R., 2000. Effect of a neuropeptide gene on behavioral states in Caenorhabditis elegans egg-laying. Genetics 154: 1181–1192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White J. G., Southgate E., Thomson J. N., Brenner S., 1986. The structure of the nervous system of the nematode Caenorhabditis elegans. Philos. Trans. R. Soc. Lond. B Biol. Sci. 314: 1–340 [DOI] [PubMed] [Google Scholar]
- Wittschieben B. O., Otero G., de Bizemont T., Fellows J., Erdjument-Bromage H., et al. , 1999. A novel histone acetyltransferase is an integral subunit of elongating RNA polymerase II holoenzyme. Mol. Cell 4: 123–128 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.