Abstract
We review the evidence that there are two types of disc degeneration. ‘Endplate-driven’ disc degeneration involves endplate defects and inwards collapse of the annulus, has a high heritability, mostly affects discs in the upper lumbar and thoracic spine, often starts to develop before age 30 years, usually leads to moderate back pain, and is associated with compressive injuries such as a fall on the buttocks. ‘Annulus-driven’ disc degeneration involves a radial fissure and/or a disc prolapse, has a low heritability, mostly affects discs in the lower lumbar spine, develops progressively after age 30 years, usually leads to severe back pain and sciatica, and is associated with repetitive bending and lifting. The structural defects which initiate the two processes both act to decompress the disc nucleus, making it less likely that the other defect could occur subsequently, and in this sense the two disc degeneration phenotypes can be viewed as distinct.
Keywords: ageing, biomechanics, degeneration, intervertebral disc, phenotype
Introduction
Intervertebral discs are pads of fibrocartilage which lie between the vertebral bodies of the spine (Fig. 1). With advancing age, they frequently exhibit degenerative changes which are a major cause of back pain. In this discussion paper we summarise the evidence that there are two distinct types of intervertebral disc degeneration, and suggest why this is important clinically.
Fig. 1.
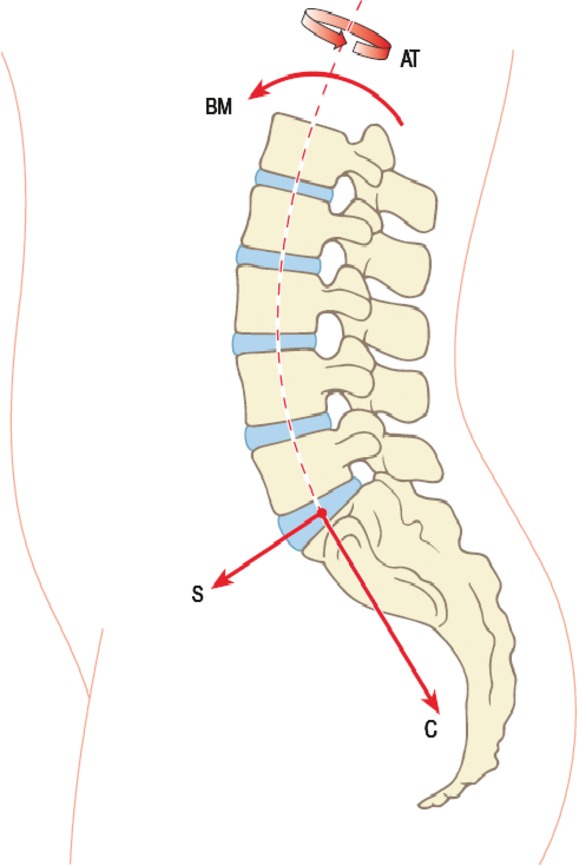
Intervertebral discs, shown here in blue, lie between the vertebral bodies of the spine. Compressive (C) and shear (S) forces on the spine act perpendicular to, and parallel to, the mid-plane of each disc A bending moment (BM) causes the spine to bend, and an axial torque (AT) causes it to rotate about its long axis. Adapted with permission from Adams et al. (2012).
Research into disc degeneration is expanding rapidly, as large population studies have shown a strong dose-related association between intervertebral disc degeneration and back pain or sciatica (Bendix et al. 2008; Cheung et al. 2009; de Schepper et al. 2010). The association is notoriously variable, however, because some previous studies on smaller selected groups found little or no association between some aspects of disc degeneration and pain (Boden et al. 1990; Jensen et al. 1994; Boos et al. 1995). Evidently, certain features of disc degeneration are often painful, but not in all cases, and other features are rarely if ever painful. Gross features of disc degeneration that are associated with pain include the presence of fissures in the annulus fibrosus, structural changes to the vertebral body endplates, and collapse of the annulus leading to gross disc narrowing (Videman et al. 2003; Cheung et al. 2009; de Schepper et al. 2010). Other features associated with painful discs include the ingrowth and sensitisation of nociceptive nerves (Coppes et al. 1990; Freemont et al. 1997; Olmarker, 2008), processes which are themselves variable and poorly understood. In contrast, several biochemical changes in degenerated discs, such as proteoglycan and water loss, appear to be more or less inevitable with advancing age, and have little or no direct association with pain (Jensen et al. 1994; Boos et al. 1995; Videman et al. 2003; Bendix et al. 2008).
The etiology of disc degeneration appears to be as variable as its relationship with pain, with identified risk factors including age (Miller et al. 1988), genetic inheritance (Battie et al. 2008), heavy lifting (Videman et al. 1995b) and smoking cigarettes (Battie et al. 1991). A study comparing identical and non-identical twins suggested that approximately 75% of the variance in ‘who has a degenerated disc’ is attributable to genetic inheritance (Sambrook et al. 1999). However, this percentage refers to middle-aged women; the genetic influence is less in a population of men of mixed age (Battie et al. 1995) and falls to approximately 35% if only the lower lumbar discs are considered (Battie et al. 2008), suggesting that genetic influences interact with age and the mechanical environment.
Accordingly, it is becoming increasingly apparent that ‘disc degeneration’ is too general a term to be of much use when considering either the etiology of the condition, or its likely clinical consequences. The condition needs to be divided up into distinctive ‘phenotypes’, each of which may have a different etiology and a different clinical outcome (Battie et al. 2008).
We propose that ‘disc degeneration’ can usefully be divided into two distinct categories, as indicated in Table 1. According to this scheme, one type of disc degeneration arises from endplate defects and occurs mostly in the upper lumbar and thoracic spine; the other arises from annulus fissures and occurs primarily at L4–5 and L5–S1. The following sections summarise the evidence that each phenotype has distinct risk factors, etiology and clinical presentation, and suggests why they should be considered to be largely independent of each other. But first, it will be helpful to identify the age-related changes that occur in all ageing discs and which are not essential features of either disc degeneration ‘phenotype’.
Table 1.
Comparison between two disc degeneration phenotypes
| Type A: Endplate-driven | Type B: Annulus-driven |
|---|---|
| Associated with endplate defects | Associated with annulus fissures |
| Upper lumbar and thoracic spine | Lower lumbar spine |
| Higher heritability | Lower heritability |
| Often occurs before age 30 years | Rarely occurs before age 30 years |
| Moderate association with pain | Strong association with pain |
| Pain from endplate/vertebral body | Pain from peripheral annulus/nerve root |
| Caused by spinal compression | Caused by spinal bending |
Age-related changes that affect all intervertebral discs
Disc cell density decreases throughout growth, as metabolite transport difficulties increase, but once full size is reached, there is no further decrease in cellularity (Liebscher et al. 2011). At skeletal maturity, cell density in the nucleus is < 25% of that in the outer annulus (Hastreiter et al. 2001), presumably because metabolite transport is better in the disc periphery (Ferguson et al. 2004). With subsequent ageing, the number of active non-senescent cells does show some decrease, especially in the nucleus, and this leads to reduced concentrations of proteoglycans and hence water (Adams & Roughley, 2006). Decreasing cell activity also reduces the turn-over of disc collagens (Sivan et al. 2008), so that collagen fibres become thicker, and less inter-woven (Schollum et al. #b300). The process of non-enzymatic glycation gradually makes old cartilaginous tissues stiffer and more easily injured (DeGroot et al. 2004) and, as a side-effect of this process, old intervertebral discs often become yellow or brown in colour. The result is a fibrous, discoloured and dehydrated nucleus surrounded by an annulus with thickened discoloured lamellae.
Functionally, these biochemical changes in the disc matrix cause the hydrostatic pressure in the nucleus to fall and the region of fluid-like behaviour to shrink (Adams et al. 1996b). Reduced nucleus volume and pressure allows the annulus to bulge radially outwards, rather like a ‘flat tyre’ (Brinckmann & Grootenboer, 1991), as it resists an increasing proportion of the compressive force acting on the disc. Corresponding inwards bulging of inner lamellae would require adjacent lamellae to separate, against the resistance of translamellar bridging structures (Schollum et al. 2009), or else some individual lamellae would have to undergo very high radial strains.
None of the above age-related changes is essentially pathological, and the MRI appearance of a slightly bulging ‘grey’ disc, which indicates reduced water content (Marinelli et al. 2009), is not associated with back pain (Jensen et al. 1994; Boos et al. 1995; Videman et al. 2003; Bendix et al. 2008). However, old discs are more vulnerable to injury because of non-enzymatic glycation, and because their decreasing water content leads to compressive stresses being concentrated in certain regions of the annulus rather than being distributed evenly, as in younger discs (Adams et al. 1996b).
‘Disc degeneration’ can be likened to accelerated ageing
The biochemical and functional changes of ageing are exaggerated (or accelerated) in some discs, particularly those in the lower lumbar spine. We have suggested previously that the essential non-reversible step that causes this acceleration is structural damage to the disc (Adams & Roughley, 2006). The mechanism is outlined in Fig. 2: structural damage to an ageing annulus or endplate creates regions within the disc of low and high compressive stresses (Fig. 3), both of which are known to inhibit disc cell metabolism (Ishihara et al. 1996). Stress concentrations cause the disruption to propagate, and the ability of disc cells to repair the disrupted matrix is reduced by the altered matrix stresses. The result is a vicious circle of frustrated healing and repeated re-injury which characterise disc degeneration (Adams & Roughley, 2006). This explanation is compatible with all of the major known risk factors: genetic inheritance and ageing weaken the disc matrix, mechanical loading disrupts its structure, and inadequate metabolite transport prevents full healing. Disc cells could spontaneously degrade their own matrix, but entirely cell-based theories of disc degeneration struggle to explain why certain regions of certain discs usually show advanced focal changes. Decreasing numbers of small holes in the vertebral endplate have been linked to disc degeneration (Benneker et al. 2005), but more recent evidence suggests that disc metabolite transport actually increases with age and degeneration, as the endplates become more permeable (Rodriguez et al. 2011), more porous (Rodriguez et al. 2012) and, in some cases, more disrupted (Rajasekaran et al. 2004). This suggests that inadequate nutrition is not an important initiating factor in the degenerative process.
Fig. 2.
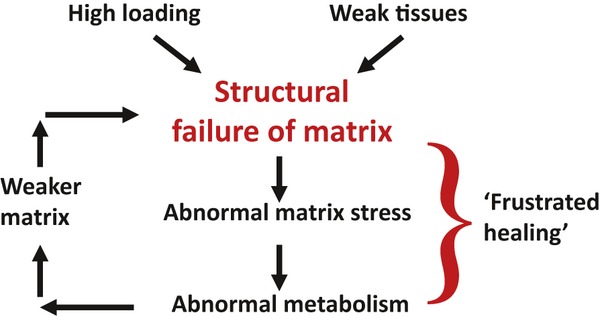
Schematic suggesting how structural damage to the extracellular matrix of an intervertebral disc leads to progressive degeneration. See text for details.
Fig. 3.
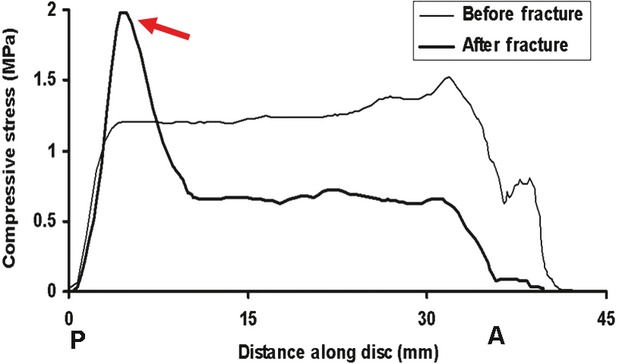
Profiles of vertically-acting compressive stress measured along the sagittal midline of a cadaveric intervertebral disc which was loaded in compression. A, P: anterior, posterior. Measurements were repeated after a vertebral endplate was fractured by compressive overload. Endplate damage decompresses the central and anterior regions of the disc and generates a high concentration of compressive stress in the posterior annulus (arrow). Endplate damage also transfers some compressive load-bearing to the neural arch (Luo et al. 2007).
The next two sections describe how two distinct disc degeneration phenotypes can arise, depending on the nature of the initiating structural damage.
Disc degeneration Type A: endplate-driven
Features
This type of disc degeneration is characterized by a damaged endplate, by circumferential tears between the lamellae of the annulus, and by internal bulging or collapse of the annulus into the nucleus. Outwards bulging of the peripheral annulus is usually slight, and there may be inflammatory changes in the adjacent (damaged) vertebral body. Vertical migration of nucleus pulposus through a damaged endplate and subsequent calcification can create a ‘Schmorl's node’ in the adjacent vertebral body. Typical images of endplate-driven degeneration are shown in Fig. 4.
Fig. 4.
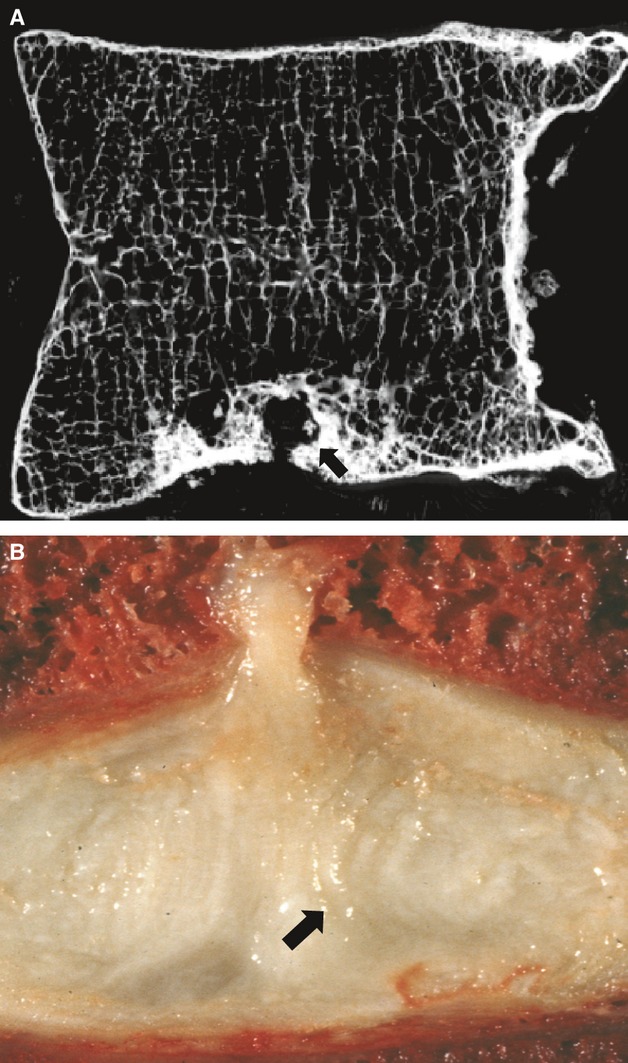
Images of endplate-driven disc degeneration. (A) Microradiograph of a 5 mm-thick section of a lumbar vertebral body showing a Schmorl's node (arrow) in its lower endplate. (B) Mid-sagittal section through a cadaveric spine showing a vertical herniation of nucleus pulposus through the disc's superior endplate. Herniation was caused by compressive loading. Note that the inner lamellae of the annulus have collapsed into the nucleus cavity (arrow).
Epidemiology
Internal collapse of the annulus is not easy to detect in living people, although MRI can be used for this purpose in cadaveric specimens (Gunzburg et al. 1992). More typically, internal collapse is inferred from the presence of an endplate defect coupled with a reduction in disc space height. This type of disc degeneration is relatively rare in the lower lumbar spine, where Schmorl's nodes are least common (Pfirrmann & Resnick, 2001; Williams et al. 2007; Mok et al. 2010) and are only weakly associated with disc degeneration (Hilton et al. 1976; Mok et al. 2010). Endplate injuries often go undetected by radiographs (Jiang et al. 2010) but are more apparent on MRI scans, especially if there is an associated Schmorl's node (Hamanishi et al. 1994). Even smaller endplate irregularities can be detected by cadaveric dissection, and are reported to occur in 76% of spines aged 13–96 years (Hilton et al. 1976). These minor defects also are more common in the thoracic and upper lumbar spine, where they are strongly related to disc degeneration (Hilton et al. 1976).
Etiology
Sportsmen who subject their backs to impact loading often have vertebral damage in conjunction with disc degeneration (Sward et al. 1991) and many of those who suffer endplate damage in adolescence later develop degenerative changes in adjacent discs (Kerttula et al. 2000). Another study found that vertebral injury did not lead to disc degeneration 40 years later (Moller et al. 2007) but the influence of the injury may have been obscured by 40 years of ‘wear and tear’. Artificially-induced endplate damage in pigs leads to adjacent disc degeneration, including internal disruption of the annulus (Holm et al. 2004). Mechanical experiments on cadaveric spines can explain the underlying mechanism: a damaged endplate bulges into the adjacent vertebra, increasing the volume available to the nucleus and therefore causing nucleus pressure to fall immediately (Fig. 3), typically by 30–50% (Adams et al. 1993, 2000). Compressive load-bearing is shifted onto the neural arch and onto the annulus, so that high concentrations of compressive stress can appear in the disc, especially in the posterior annulus. Subsequent cyclic loading in compression can then cause the inner lamellae of the annulus to collapse into the decompressed nucleus region (Adams et al. 2000), as shown in Fig. 4B. Finite element models can explain this mechanism in terms of known materials and geometric properties of the disc (Shirazi-Adl, 1992). The effect is reduced in highly hydrated young discs which function somewhat like a water-bed, equalizing pressure even when the endplate is damaged (Adams et al. 1993). Internal annulus collapse is probably facilitated by prior delamination, arising from high interlaminar shear stresses (Goel et al. 1995), which are capable of breaking the translamellar bridging structures (Schollum et al. 2009). Endplate-driven degeneration could possibly be initiated without any overload event if accumulating trabecular microdamage just behind the endplate (Vernon-Roberts & Pirie, 1973) should lead to the nucleus gradually causing the endplate to bulge into the vertebral body (Twomey & Taylor, 1985). Vertebral body size and compressive strength increase at lower lumbar levels (Brinckmann et al. 1989), presumably to match the increased compressive loading applied to them in life.
Not all endplate defects are necessarily mechanical in origin: some occur early in life and may represent a congenital weakness arising from the site of the embryonic notochord. This could explain why Schmorl's nodes are highly heritable (Williams et al. 2007), why their prevalence increases little beyond the age of 30 years (Hamanishi et al. 1994), and why they are associated with the moderate (rather than advanced) disc degeneration normally seen near the thoracolumbar junction (Pfirrmann & Resnick, 2001).
Clinical implications
Endplates are innervated (Bailey et al. 2011) and can be painful (Peng et al. 2009), although bone tends to heal relatively quickly, especially in young spines. As a result, Schmorl's nodes carry only a twofold increased risk of a history of back pain (Hamanishi et al. 1994; Williams et al. 2007). Inflammatory (‘Modic’) changes within the vertebral body often accompany endplate defects (Modic et al. 1988) and are themselves associated with back pain (Kjaer et al. 2005; Albert & Manniche, 2007; Jensen et al. 2008). Internal disc collapse can generate high concentrations of compressive stress within the annulus (Adams et al. 2000), and these have been associated with pain in living patients (McNally et al. 1996). In the long term, increased load-bearing by the neural arch following disc decompression and narrowing (Dunlop et al. 1984; Pollintine et al. 2004) probably leads to osteoarthritis in the apophyseal joints (Butler et al. 1990; Robson-Brown et al. 2008), and to back pain. Painful endplate lesions can be identified in vivo using discography (Peng et al. 2009).
Disc degeneration Type B: annulus-driven
Features
This type of disc degeneration is most common in the lower lumbar spine, where it is associated with severe disc height loss (Kjaer et al. 2005). Radial fissures progress outwards from the nucleus, usually posteriorly or posterolaterally (Hirsch & Schajowicz, 1953; Osti et al. 1992) as shown in Fig. 5. Radial fissures are associated with nucleus degeneration (Osti et al. 1992; Jensen et al. 1994) and with disc radial bulging (Yu et al. 1988) but it is not clear which comes first. If nucleus pulposus material migrates down a radial fissure, it can be detected as a ‘High Intensity Zone’ (Aprill & Bogduk, 1992), which is visible on MRI scans. Another type of annulus fissure, the peripheral ‘rim lesion’, consists of focal circumferential avulsions of the peripheral annulus, sometimes with sclerosis and osteophytosis of the adjacent bone. When simulated on animals, they lead to progressive disc degeneration (Osti et al. 1990) but in humans they occur mostly in the anterior annulus (Hilton & Ball, 1984; Osti et al. 1992) and have minimal effect on nucleus pressure (Przybyla et al. 2006) and so are unlikely to be major causes of progressive and painful disc degeneration.
Fig. 5.
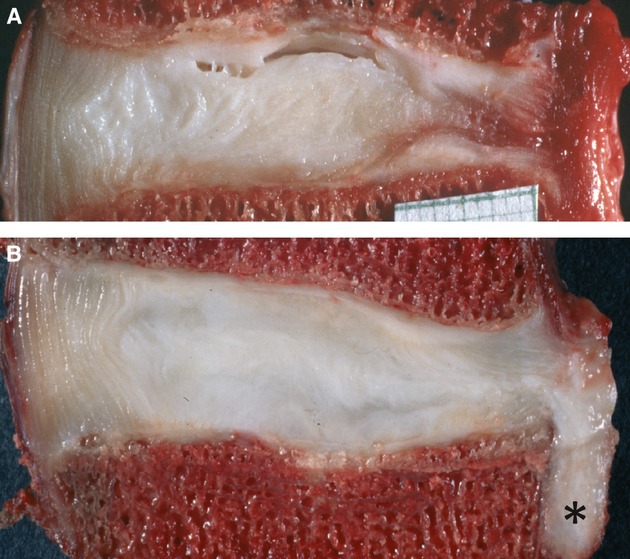
Images of annulus-driven disc degeneration: two mid-sagittal sections through lower lumbar cadaveric intervertebral discs (posterior on the right). (A) There is a complete radial fissure in the posterior annulus, with blood in its peripheral margins. (B) Nucleus pulposus tissue (*) has herniated through this radial fissure in response to severe loading in bending and compression (Adams et al. 2000).
Disc prolapse
When radial fissures allow gross migration of nucleus relative to annulus, to the extent that the disc periphery is affected, then the disc can be said to be ‘prolapsed’ (or ‘herniated’). Displaced tissue is mostly nucleus (Moore et al. 1996), as shown in Fig. 5B, but may include cartilaginous endplate, especially in older people (Harada & Nakahara, 1989; Brock et al. 1992). We suggest that disc prolapse should be considered one possible endpoint of the ‘annulus-driven’ type of disc degeneration. As indicated below under ‘Etiology’, this endpoint can be reached gradually over many years, or accelerated by severe repetitive loading, or it can represent an injury. Different types of disc prolapse are recognized in the surgical literature: in a ‘protrusion’, the annulus bulges markedly and focally; in an ‘extrusion’ the annulus is ruptured, but any expelled nucleus is still attached to the rest of the nucleus (Fig. 5B); and in a ‘sequestration’, the expelled disc tissue is no longer attached to the rest of the disc. A disc with a generally bulging annulus is sometimes referred to as prolapsed, but this should be recognized as a normal consequence of ageing (rather like a bulging waistline).
Epidemiology
Radial fissure formation is most common in the lower lumbar spine, posterior to the nucleus, and becomes steadily more prevalent with age (Hirsch & Schajowicz, 1953; Hilton et al. 1980; Videman & Nurminen, 2004). Annulus bulging also tends to be greatest here (Videman et al. 1995a). Posterior disc bulging shows little correlation with age or other signs of spinal degeneration (Videman et al. 1995a), is rare throughout the thoracic spine (Niemelainen et al. 2008), and has a reduced heritability compared with other features of disc degeneration (Videman et al. 2008). Conversely, Schmorl's nodes are relatively rare in the lower lumbar spine (and) where they are only weakly associated with disc degeneration (Mok et al. 2010). Disc prolapse is associated with asymmetrical apophyseal joints (‘facet tropism’) but only at the L4–5 level in adults (Lee et al. 2006).
Etiology
The process of radial fissure formation can be simulated in cadaveric and animal discs by various combinations of repetitive mechanical loading which cause the annulus lamellae to rupture and allow nucleus material to migrate radially towards the disc periphery (Adams & Hutton, 1985; Callaghan & McGill, 2001; Veres et al. 2009). This happens only if the disc is subjected to a substantial component of bending, as it would be when the spine is flexed, because bending is able to stretch and weaken the annulus on the side away from the bending so that it fails before the vertebral endplate. If the applied stresses are exceptionally high, then disc prolapse can occur as a sudden injury, even if the disc is apparently normal for its age (Adams & Hutton, 1982; Adams et al. 2000; Veres et al. 2009). If a radial fissure is created artificially in the annulus, it leads to progressive cell-mediated degenerative changes in both humans (Carragee et al. 2009) and animals (Osti et al. 1990).
In living people, the lower lumbar discs are subjected to the highest bending moments (Adams & Dolan, 1991) and the relatively thin posterior annulus of these discs then makes them vulnerable to injury. As indicated above, the increased compressive force acting on the lower lumbar discs is unlikely to be a major etiological factor because the increased force is applied over a larger disc area. High anterior shear forces act on the lower lumbar spine, where the discs are inclined to the horizontal plane, but this shear force is largely resisted by the apophyseal joints so that little of it acts on the discs (Cyron & Hutton, 1980).
Mathematical models indicate that the risk of disc prolapse is increased if the disc is not already degenerated (Shirazi-Adl, 1989, 1992; Schmidt et al. 2007). Non-degenerated discs have a hydrated nucleus exhibiting a high fluid pressure (Sato et al. 1999) that acts to rupture the annulus in tension. The importance of nucleus pressure can be demonstrated by attempting to create disc prolapse in cadaveric specimens after first subjecting the disc to sustained compressive loading in order to reduce nucleus water content (McMillan et al. 1996) and pressure (Adams et al. 1996a): it then becomes more difficult to create a disc prolapse (Adams et al. 1987). Extrapolating to living people, it would appear that radial fissure formation is more likely to progress in the early morning, when nucleus water content and pressure are highest as a result of the spine being relatively unloaded overnight (Botsford et al. 1994).
Clinical implications
Severe back pain is strongly associated with radial fissures that reach the disc periphery (Moneta et al. 1994; Videman et al. 2003; Videman & Nurminen, 2004; Peng et al. 2005, 2006). The innervation of the annulus is normally confined to the peripheral 3 mm (Palmgren et al. 1999) although nerves can grow further in along radial fissures (Peng et al. 2005; Fagan et al. 2010), which comprise a protected micro-environment of low compressive stress and low proteoglycan content (Melrose et al. 2002; Stefanakis et al. 2012). Intense pain arising from the peripheral annulus has been demonstrated by pain-provocation and pain-blocking studies (Kuslich et al. 1991; Schwarzer et al. 1995) and can be attributed to sensitisation of nerves in disrupted and inflamed tissue, augmented by chemicals diffusing from internally displaced nucleus pulposus (Olmarker, 2008). Sciatica has long been associated with disc prolapse arising from activities such as bending and lifting (Kelsey et al. 1984; Seidler et al. 2003; Kaila-Kangas et al. 2009) and it can be attributed to physical and chemical compromise of a spinal nerve root by displaced disc tissue (Kuslich et al. 1991; Ozaktay et al. 2006; Goupille et al. 2007).
A final common pathway for disc degeneration?
Although the two types of disc degeneration have different origins, we suggest that both can progress along a final common pathway leading to complete disc failure. Annulus bulging progresses under compressive loading, regardless of whether the lamellae are bulging outwards or inwards, and the disc can also bulge vertically into the adjacent vertebral bodies if the bones become osteopaenic. These deformations progressively reduce nucleus pressure (Adams et al. 1996b; Sato et al. 1999) and hence annulus height (Brinckmann & Grootenboer, 1991). Degenerated human discs lose height at a rate of approximately 3% per year (Hassett et al. 2003), whereas normal discs lose <1% per year (Videman et al. 2008). Annulus height largely determines the separation of adjacent neural arches, so disc-narrowing causes severe compressive loading of the apophyseal joints (Dunlop et al. 1984; Pollintine et al. 2004), which in turn can lead to osteoarthritis (Butler et al. 1990; Robson-Brown et al. 2008). Disc height loss also slackens intervertebral ligaments (Adams et al. 1987), leading to segmental instability (Zhao et al. 2005) and subsequent re-stabilisation by means of vertebral body marginal osteophytes (Al-Rawahi et al. 2011).
Comparisons between the two types of disc degeneration
In the lower lumbar spine, discs are higher anteriorly than posteriorly, have a relatively thin posterior annulus (from inner to outer), and are usually orientated at a considerable angle to the horizontal. These characteristics would help them to dissipate energy from a sudden compressive overload event (such as a fall on the buttocks) and yet make them intrinsically more vulnerable to injury in forwards bending (Adams & Hutton, 1982). Hence the lower lumbar discs are more often injured in bending, and the upper lumbar and thoracic discs in compression. Furthermore, recent (unpublished) results from our own laboratory show that the percent reduction in nucleus pressure that follows endplate damage is least in the lower lumbar discs, presumably because the extra volume created by endplate damage is only a small proportion of existing nucleus volume in these large discs. So, even if a lower lumbar disc does suffer a compressive endplate injury, the resulting nucleus decompression will not be severe, and the risk of degeneration will be less than at higher spinal levels.
Endplate-driven degeneration tends to occur at a younger age because underlying structural defects in the endplate can be congenital in origin. Annulus-driven degeneration tends to increase throughout life because it depends more on accumulating fatigue damage than on congenital defects, and because age-related biochemical changes make the annulus more vulnerable to mechanical loading.
Clinical consequences of endplate-driven degeneration may be relatively less severe because underlying defects in the bony endplate and vertebral body are capable of healing (Vernon-Roberts & Pirie, 1973). In contrast, the relatively poor healing potential of the annulus (Adams et al. 2010) may lead to repeated injury, a magnified inflammatory response (Ulrich et al. 2007), and more severe pain from the peripheral annulus and/or nerve root.
How distinct are these two types of disc degeneration?
The concept of two distinct routes to disc degeneration is supported by a large MRI population study from Hong Kong. This shows that associations between Schmorl's nodes (an indicator of endplate damage) and disc degeneration are much stronger at L1–L3 than at L4–S1, and that discs adjacent to Schmorl's nodes are less likely than normal to show evidence of radial fissures or prolapse (Mok et al. 2010). The two phenotypes also exhibit distinct (though overlapping) age-profiles, as discussed above. Perhaps the strongest evidence for distinct etiologies, however, comes from cadaveric experiments which show that endplate fracture and disc prolapse both cause a major decompression of the nucleus pulposus, as compressive load-bearing is transferred to the annulus and neural arch (Adams et al. 2012). High nucleus pressure is essential to simulate disc prolapse in the laboratory (Adams et al. 1987) and it facilitates endplate fracture also, so it appears that if a disc sustains one type of initiating injury it will be less likely to sustain the other in the near future. In this sense the two types of disc degeneration can be considered distinct, rather than two ends of a continuous spectrum.
However, it is impossible to separate the phenotypes entirely, especially in the mid-lumbar spine. Schmorl's nodes can be found in vertebrae adjacent to prolapsed discs, especially in older people (Hamanishi et al. 1994), and it would be reasonable to suppose that an incident that was severe enough to injure an ageing annulus might also damage an adjacent osteopaenic endplate, because vertebrae appear to weaken more quickly with age compared with the adjacent discs (Skrzypiec et al. 2007). This may explain the gradual appearance of Modic changes (and back pain) in some patients with pre-existing sciatica (Albert & Manniche, 2007).
Concluding remarks
The evidence reviewed above suggests that there are two distinct routes towards disc degeneration. More research is required to add weight to this suggestion, and perhaps to extend the number of distinct disc degeneration phenotypes beyond two. This should assist in defining risk factors more precisely, and in developing more effective strategies for prevention and treatment.
Acknowledgments
Related research by the authors is funded in the UK by Action Medical Research, BackCare, and The Annett Charitable Trust.
Conflict of interest
The authors declare that they have no conflict of interest.
References
- Adams MA, Dolan P. A technique for quantifying the bending moment acting on the lumbar spine in vivo. J Biomech. 1991;24:117–126. doi: 10.1016/0021-9290(91)90356-r. [DOI] [PubMed] [Google Scholar]
- Adams MA, Hutton WC. Prolapsed intervertebral disc. A hyperflexion injury 1981 Volvo award in basic science. Spine. 1982;7:184–191. [PubMed] [Google Scholar]
- Adams MA, Hutton WC. Gradual disc prolapse. Spine. 1985;10:524–531. doi: 10.1097/00007632-198507000-00006. [DOI] [PubMed] [Google Scholar]
- Adams MA, Roughley PJ. What is intervertebral disc degeneration, and what causes it? Spine. 2006;31:2151–2161. doi: 10.1097/01.brs.0000231761.73859.2c. [DOI] [PubMed] [Google Scholar]
- Adams MA, Dolan P, Hutton WC. Diurnal variations in the stresses on the lumbar spine. Spine. 1987;12:130–137. doi: 10.1097/00007632-198703000-00008. [DOI] [PubMed] [Google Scholar]
- Adams MA, McNally DS, Wagstaff J, et al. Abnormal stress concentrations in lumbar intervertebral discs following damage to the vertebral body: a cause of disc failure. Eur Spine J. 1993;1:214–221. doi: 10.1007/BF00298362. [DOI] [PubMed] [Google Scholar]
- Adams MA, McMillan DW, Green TP, et al. Sustained loading generates stress concentrations in lumbar intervertebral discs. Spine. 1996a;21:434–438. doi: 10.1097/00007632-199602150-00006. [DOI] [PubMed] [Google Scholar]
- Adams MA, McNally DS, Dolan P. ‘Stress’ distributions inside intervertebral discs. The effects of age and degeneration. J Bone Joint Surg Br. 1996b;78:965–972. doi: 10.1302/0301-620x78b6.1287. [DOI] [PubMed] [Google Scholar]
- Adams MA, Freeman BJ, Morrison HP, et al. Mechanical initiation of intervertebral disc degeneration. Spine. 2000;25:1625–1636. doi: 10.1097/00007632-200007010-00005. [DOI] [PubMed] [Google Scholar]
- Adams MA, Stefanakis M, Dolan P. Healing of a painful intervertebral disc should not be confused with reversing disc degeneration: implications for physical therapies for discogenic back pain. Clin Biomech (Bristol, Avon) 2010;25:961–971. doi: 10.1016/j.clinbiomech.2010.07.016. [DOI] [PubMed] [Google Scholar]
- Adams MA, Bogduk N, Burton K, et al. The Biomechanics of Back Pain. 3rd edn. Edinburgh: Churchill Livingstone; 2012. [Google Scholar]
- Albert HB, Manniche C. Modic changes following lumbar disc herniation. Eur Spine J. 2007;16:977–982. doi: 10.1007/s00586-007-0336-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Rawahi M, Luo J, Pollintine P, et al. Mechanical function of vertebral body osteophytes, as revealed by experiments on cadaveric spines. Spine. 2011;36:770–777. doi: 10.1097/BRS.0b013e3181df1a70. [DOI] [PubMed] [Google Scholar]
- Aprill C, Bogduk N. High-intensity zone: a diagnostic sign of painful lumbar disc on magnetic resonance imaging. Br J Radiol. 1992;65:361–369. doi: 10.1259/0007-1285-65-773-361. [DOI] [PubMed] [Google Scholar]
- Bailey JF, Liebenberg E, Degmetich S, et al. Innervation patterns of PGP 9.5-positive nerve fibers within the human lumbar vertebra. J Anat. 2011;218:263–270. doi: 10.1111/j.1469-7580.2010.01332.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Battie MC, Videman T, Gill K, et al. 1991 Volvo Award in clinical sciences. Smoking and lumbar intervertebral disc degeneration: an MRI study of identical twins. Spine. 1991;16:1015–1021. [PubMed] [Google Scholar]
- Battie MC, Videman T, Gibbons LE, et al. 1995 Volvo award in clinical sciences. Determinants of lumbar disc degeneration. A study relating lifetime exposures and magnetic resonance imaging findings in identical twins. Spine. 1995;20:2601–2612. [PubMed] [Google Scholar]
- Battie MC, Videman T, Levalahti E, et al. Genetic and environmental effects on disc degeneration by phenotype and spinal level: a multivariate twin study. Spine. 2008;33:2801–2808. doi: 10.1097/BRS.0b013e31818043b7. [DOI] [PubMed] [Google Scholar]
- Bendix T, Kjaer P, Korsholm L. Burned-out discs stop hurting: fact or fiction? Spine. 2008;33:E962–E967. doi: 10.1097/BRS.0b013e31818804b3. [DOI] [PubMed] [Google Scholar]
- Benneker LM, Heini PF, Alini M, et al. 2004 Young investigator award winner: vertebral endplate marrow contact channel occlusions and intervertebral disc degeneration. Spine. 2005;30:167–173. doi: 10.1097/01.brs.0000150833.93248.09. [DOI] [PubMed] [Google Scholar]
- Boden SD, Davis DO, Dina TS, et al. Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects. A prospective investigation. J Bone Joint Surg Am. 1990;72:403–408. [PubMed] [Google Scholar]
- Boos N, Rieder R, Schade V, et al. 1995 Volvo award in clinical sciences. The diagnostic accuracy of magnetic resonance imaging, work perception, and psychosocial factors in identifying symptomatic disc herniations. Spine. 1995;20:2613–2625. doi: 10.1097/00007632-199512150-00002. [DOI] [PubMed] [Google Scholar]
- Botsford DJ, Esses SI, Ogilvie-Harris DJ. In vivo diurnal variation in intervertebral disc volume and morphology. Spine. 1994;19:935–940. doi: 10.1097/00007632-199404150-00012. [DOI] [PubMed] [Google Scholar]
- Brinckmann P, Grootenboer H. Change of disc height, radial disc bulge, and intradiscal pressure from discectomy. An in vitro investigation on human lumbar discs. Spine. 1991;16:641–646. doi: 10.1097/00007632-199106000-00008. [DOI] [PubMed] [Google Scholar]
- Brinckmann P, Biggemann M, Hilweg D. Prediction of the compressive strength of human lumbar vertebrae. Clin Biomech. 1989;4(Suppl 2) doi: 10.1016/0268-0033(89)90071-5. [DOI] [PubMed] [Google Scholar]
- Brock M, Patt S, Mayer HM. The form and structure of the extruded disc. Spine. 1992;17:1457–1461. doi: 10.1097/00007632-199212000-00003. [DOI] [PubMed] [Google Scholar]
- Butler D, Trafimow JH, Andersson GB, et al. Discs degenerate before facets. Spine. 1990;15:111–113. doi: 10.1097/00007632-199002000-00012. [DOI] [PubMed] [Google Scholar]
- Callaghan JP, McGill SM. Intervertebral disc herniation: studies on a porcine model exposed to highly repetitive flexion/extension motion with compressive force. Clin Biomech (Bristol, Avon) 2001;16:28–37. doi: 10.1016/s0268-0033(00)00063-2. [DOI] [PubMed] [Google Scholar]
- Carragee EJ, Don AS, Hurwitz EL, et al. 2009 ISSLS prize winner: does discography cause accelerated progression of degeneration changes in the lumbar disc: a ten-year matched cohort study. Spine. 2009;34:2338–2345. doi: 10.1097/BRS.0b013e3181ab5432. [DOI] [PubMed] [Google Scholar]
- Cheung KM, Karppinen J, Chan D, et al. Prevalence and pattern of lumbar magnetic resonance imaging changes in a population study of one thousand forty-three individuals. Spine. 2009;34:934–940. doi: 10.1097/BRS.0b013e3181a01b3f. [DOI] [PubMed] [Google Scholar]
- Coppes MH, Marani E, Thomeer RT, et al. Innervation of annulus fibrosis in low back pain [letter] [published erratum appears in Lancet 1990 Aug 4;336(8710):324] Lancet. 1990;336:189–190. doi: 10.1016/0140-6736(90)91723-n. [DOI] [PubMed] [Google Scholar]
- Cyron BM, Hutton WC. Articular tropism and stability of the lumbar spine. Spine. 1980;5:168–172. doi: 10.1097/00007632-198003000-00011. [DOI] [PubMed] [Google Scholar]
- DeGroot J, Verzijl N, Wenting-Van Wijk MJ, et al. Accumulation of advanced glycation end products as a molecular mechanism for aging as a risk factor in osteoarthritis. Arthritis Rheum. 2004;50:1207–1215. doi: 10.1002/art.20170. [DOI] [PubMed] [Google Scholar]
- Dunlop RB, Adams MA, Hutton WC. Disc space narrowing and the lumbar facet joints. J Bone Joint Surg Br. 1984;66:706–710. doi: 10.1302/0301-620X.66B5.6501365. [DOI] [PubMed] [Google Scholar]
- Fagan AB, Sarvestani G, Moore RJ, et al. Innervation of anulus tears: an experimental animal study. Spine. 2010;35:1200–1205. doi: 10.1097/BRS.0b013e3181c02812. [DOI] [PubMed] [Google Scholar]
- Ferguson SJ, Ito K, Nolte LP. Fluid flow and convective transport of solutes within the intervertebral disc. J Biomech. 2004;37:213–221. doi: 10.1016/s0021-9290(03)00250-1. [DOI] [PubMed] [Google Scholar]
- Freemont AJ, Peacock TE, Goupille P, et al. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet. 1997;350:178–181. doi: 10.1016/s0140-6736(97)02135-1. [DOI] [PubMed] [Google Scholar]
- Goel VK, Monroe BT, Gilbertson LG, et al. Interlaminar shear stresses and laminae separation in a disc. Finite element analysis of the L3–L4 motion segment subjected to axial compressive loads. Spine. 1995;20:689–698. [PubMed] [Google Scholar]
- Goupille P, Mulleman D, Paintaud G, et al. Can sciatica induced by disc herniation be treated with tumor necrosis factor alpha blockade? Arthritis Rheum. 2007;56:3887–3895. doi: 10.1002/art.23051. [DOI] [PubMed] [Google Scholar]
- Gunzburg R, Parkinson R, Moore R, et al. A cadaveric study comparing discography, magnetic resonance imaging, histology, and mechanical behavior of the human lumbar disc. Spine. 1992;17:417–426. doi: 10.1097/00007632-199204000-00007. [DOI] [PubMed] [Google Scholar]
- Hamanishi C, Kawabata T, Yosii T, et al. Schmorl's nodes on magnetic resonance imaging. Their incidence and clinical relevance. Spine. 1994;19:450–453. doi: 10.1097/00007632-199402001-00012. [DOI] [PubMed] [Google Scholar]
- Harada Y, Nakahara S. A pathologic study of lumbar disc herniation in the elderly. Spine. 1989;14:1020–1024. doi: 10.1097/00007632-198909000-00017. [DOI] [PubMed] [Google Scholar]
- Hassett G, Hart DJ, Manek NJ, et al. Risk factors for progression of lumbar spine disc degeneration: the Chingford Study. Arthritis Rheum. 2003;48:3112–3117. doi: 10.1002/art.11321. [DOI] [PubMed] [Google Scholar]
- Hastreiter D, Ozuna RM, Spector M. Regional variations in certain cellular characteristics in human lumbar intervertebral discs, including the presence of alpha-smooth muscle actin. J Orthop Res. 2001;19:597–604. doi: 10.1016/S0736-0266(00)00069-3. [DOI] [PubMed] [Google Scholar]
- Hilton RC, Ball J. Vertebral rim lesions in the dorsolumbar spine. Ann Rheum Dis. 1984;43:302–307. doi: 10.1136/ard.43.2.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilton RC, Ball J, Benn RT. Vertebral end-plate lesions (Schmorl's nodes) in the dorsolumbar spine. Ann Rheum Dis. 1976;35:127–132. doi: 10.1136/ard.35.2.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilton RC, Ball J, Benn RT. Annular tears in the dorsolumbar spine. Ann Rheum Dis. 1980;39:533–538. doi: 10.1136/ard.39.6.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirsch C, Schajowicz F. Studies on structural changes in the lumbar annulus fibrosus. Acta Orthop Scand. 1953;22:184–231. doi: 10.3109/17453675208989006. [DOI] [PubMed] [Google Scholar]
- Holm S, Holm AK, Ekstrom L, et al. Experimental disc degeneration due to endplate injury. J Spinal Disord Tech. 2004;17:64–71. doi: 10.1097/00024720-200402000-00012. [DOI] [PubMed] [Google Scholar]
- Ishihara H, McNally DS, Urban JP, et al. Effects of hydrostatic pressure on matrix synthesis in different regions of the intervertebral disk. J Appl Physiol. 1996;80:839–846. doi: 10.1152/jappl.1996.80.3.839. [DOI] [PubMed] [Google Scholar]
- Jensen MC, Brant-Zawadzki MN, Obuchowski N, et al. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331:69–73. doi: 10.1056/NEJM199407143310201. [DOI] [PubMed] [Google Scholar]
- Jensen TS, Karppinen J, Sorensen JS, et al. Vertebral endplate signal changes (Modic change): a systematic literature review of prevalence and association with non-specific low back pain. Eur Spine J. 2008;17:1407–1422. doi: 10.1007/s00586-008-0770-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang G, Luo J, Pollintine P, et al. Vertebral fractures in the elderly may not always be ‘osteoporotic’. Bone. 2010;47:111–116. doi: 10.1016/j.bone.2010.03.019. [DOI] [PubMed] [Google Scholar]
- Kaila-Kangas L, Leino-Arjas P, Karppinen J, et al. History of physical work exposures and clinically diagnosed sciatica among working and nonworking Finns aged 30 to 64. Spine. 2009;34:964–969. doi: 10.1097/BRS.0b013e31819b2c92. [DOI] [PubMed] [Google Scholar]
- Kelsey JL, Githens PB, White AA, III, et al. An epidemiologic study of lifting and twisting on the job and risk for acute prolapsed lumbar intervertebral disc. J Orthop Res. 1984;2:61–66. doi: 10.1002/jor.1100020110. [DOI] [PubMed] [Google Scholar]
- Kerttula LI, Serlo WS, Tervonen OA, et al. Post-traumatic findings of the spine after earlier vertebral fracture in young patients: clinical and MRI study. Spine. 2000;25:1104–1108. doi: 10.1097/00007632-200005010-00011. [DOI] [PubMed] [Google Scholar]
- Kjaer P, Leboeuf-Yde C, Korsholm L, et al. Magnetic resonance imaging and low back pain in adults: a diagnostic imaging study of 40-year-old men and women. Spine. 2005;30:1173–1180. doi: 10.1097/01.brs.0000162396.97739.76. [DOI] [PubMed] [Google Scholar]
- Kuslich SD, Ulstrom CL, Michael CJ. The tissue origin of low back pain and sciatica: a report of pain response to tissue stimulation during operations on the lumbar spine using local anesthesia. Orthop Clin North Am. 1991;22:181–187. [PubMed] [Google Scholar]
- Lee DY, Ahn Y, Lee SH. The influence of facet tropism on herniation of the lumbar disc in adolescents and adults. J Bone Joint Surg Br. 2006;88:520–523. doi: 10.1302/0301-620X.88B4.16996. [DOI] [PubMed] [Google Scholar]
- Liebscher T, Haefeli M, Wuertz K, et al. Age-related variation in cell density of human lumbar intervertebral disc. Spine. 2011;36:153–159. doi: 10.1097/BRS.0b013e3181cd588c. [DOI] [PubMed] [Google Scholar]
- Luo J, Skrzypiec DM, Pollintine P, et al. Mechanical efficacy of vertebroplasty: influence of cement type, BMD, fracture severity, and disc degeneration. Bone. 2007;40:1110–1119. doi: 10.1016/j.bone.2006.11.021. [DOI] [PubMed] [Google Scholar]
- Marinelli NL, Haughton VM, Munoz A, et al. T2 relaxation times of intervertebral disc tissue correlated with water content and proteoglycan content. Spine. 2009;34:520–524. doi: 10.1097/BRS.0b013e318195dd44. [DOI] [PubMed] [Google Scholar]
- McMillan DW, Garbutt G, Adams MA. Effect of sustained loading on the water content of intervertebral discs: implications for disc metabolism. Ann Rheum Dis. 1996;55:880–887. doi: 10.1136/ard.55.12.880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNally DS, Shackleford IM, Goodship AE, et al. In vivo stress measurement can predict pain on discography. Spine. 1996;21:2580–2587. doi: 10.1097/00007632-199611150-00007. [DOI] [PubMed] [Google Scholar]
- Melrose J, Roberts S, Smith S, et al. Increased nerve and blood vessel ingrowth associated with proteoglycan depletion in an ovine anular lesion model of experimental disc degeneration. Spine. 2002;27:1278–1285. doi: 10.1097/00007632-200206150-00007. [DOI] [PubMed] [Google Scholar]
- Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine. 1988;13:173–178. [PubMed] [Google Scholar]
- Modic MT, Steinberg PM, Ross JS, et al. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology. 1988;166:193–199. doi: 10.1148/radiology.166.1.3336678. [DOI] [PubMed] [Google Scholar]
- Mok FP, Samartzis D, Karppinen J, et al. ISSLS prize winner: prevalence, determinants, and association of Schmorl nodes of the lumbar spine with disc degeneration: a population-based study of 2449 individuals. Spine. 2010;35:1944–1952. doi: 10.1097/BRS.0b013e3181d534f3. [DOI] [PubMed] [Google Scholar]
- Moller A, Maly P, Besjakov J, et al. A vertebral fracture in childhood is not a risk factor for disc degeneration but for Schmorl's nodes: a mean 40-year observational study. Spine. 2007;32:2487–2492. doi: 10.1097/BRS.0b013e3181573d6a. [DOI] [PubMed] [Google Scholar]
- Moneta GB, Videman T, Kaivanto K, et al. Reported pain during lumbar discography as a function of anular ruptures and disc degeneration. A re-analysis of 833 discograms. Spine. 1994;19:1968–1974. doi: 10.1097/00007632-199409000-00018. [DOI] [PubMed] [Google Scholar]
- Moore RJ, Vernon-Roberts B, Fraser RD, et al. The origin and fate of herniated lumbar intervertebral disc tissue. Spine. 1996;21:2149–2155. doi: 10.1097/00007632-199609150-00018. [DOI] [PubMed] [Google Scholar]
- Niemelainen R, Battie MC, Gill K, et al. The prevalence and characteristics of thoracic magnetic resonance imaging findings in men. Spine. 2008;33:2552–2559. doi: 10.1097/BRS.0b013e3181852860. [DOI] [PubMed] [Google Scholar]
- Olmarker K. Puncture of a lumbar intervertebral disc induces changes in spontaneous pain behavior: an experimental study in rats. Spine. 2008;33:850–855. doi: 10.1097/BRS.0b013e31816b46ca. [DOI] [PubMed] [Google Scholar]
- Osti OL, Vernon-Roberts B, Fraser RD. 1990 Volvo Award in experimental studies. Anulus tears and intervertebral disc degeneration. An experimental study using an animal model. Spine. 1990;15:762–767. doi: 10.1097/00007632-199008010-00005. [DOI] [PubMed] [Google Scholar]
- Osti OL, Vernon-Roberts B, Moore R, et al. Annular tears and disc degeneration in the lumbar spine. A post-mortem study of 135 discs. J Bone Joint Surg Br. 1992;74:678–682. doi: 10.1302/0301-620X.74B5.1388173. [DOI] [PubMed] [Google Scholar]
- Ozaktay AC, Kallakuri S, Takebayashi T, et al. Effects of interleukin-1 beta, interleukin-6, and tumor necrosis factor on sensitivity of dorsal root ganglion and peripheral receptive fields in rats. Eur Spine J. 2006;15:1529–1537. doi: 10.1007/s00586-005-0058-8. [DOI] [PubMed] [Google Scholar]
- Palmgren T, Gronblad M, Virri J, et al. An immunohistochemical study of nerve structures in the anulus fibrosus of human normal lumbar intervertebral discs. Spine. 1999;24:2075–2079. doi: 10.1097/00007632-199910150-00002. [DOI] [PubMed] [Google Scholar]
- Peng B, Wu W, Hou S, et al. The pathogenesis of discogenic low back pain. J Bone Joint Surg Br. 2005;87:62–67. [PubMed] [Google Scholar]
- Peng B, Hou S, Wu W, et al. The pathogenesis and clinical significance of a high-intensity zone (HIZ) of lumbar intervertebral disc on MR imaging in the patient with discogenic low back pain. Eur Spine J. 2006;15:583–587. doi: 10.1007/s00586-005-0892-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng B, Chen J, Kuang Z, et al. Diagnosis and surgical treatment of back pain originating from endplate. Eur Spine J. 2009;18:1035–1040. doi: 10.1007/s00586-009-0938-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfirrmann CW, Resnick D. Schmorl nodes of the thoracic and lumbar spine: radiographic-pathologic study of prevalence, characterization, and correlation with degenerative changes of 1,650 spinal levels in 100 cadavers. Radiology. 2001;219:368–374. doi: 10.1148/radiology.219.2.r01ma21368. [DOI] [PubMed] [Google Scholar]
- Pollintine P, Przybyla AS, Dolan P, et al. Neural arch load-bearing in old and degenerated spines. J Biomech. 2004;37:197–204. doi: 10.1016/s0021-9290(03)00308-7. [DOI] [PubMed] [Google Scholar]
- Przybyla A, Pollintine P, Bedzinski R, et al. Outer annulus tears have less effect than endplate fracture on stress distributions inside intervertebral discs: relevance to disc degeneration. Clin Biomech. 2006;21:1013–1019. doi: 10.1016/j.clinbiomech.2006.07.003. [DOI] [PubMed] [Google Scholar]
- Rajasekaran S, Babu JN, Arun R, et al. ISSLS prize winner: a study of diffusion in human lumbar discs: a serial magnetic resonance imaging study documenting the influence of the endplate on diffusion in normal and degenerate discs. Spine. 2004;29:2654–2667. doi: 10.1097/01.brs.0000148014.15210.64. [DOI] [PubMed] [Google Scholar]
- Robson-Brown K, Pollintine P, Adams MA. Biomechanical implications of degenerative joint disease in the apophyseal joints of human thoracic and lumbar vertebrae. Am J Phys Anthropol. 2008;136:318–326. doi: 10.1002/ajpa.20814. [DOI] [PubMed] [Google Scholar]
- Rodriguez AG, Slichter CK, Acosta FL, et al. Human disc nucleus properties and vertebral endplate permeability. Spine. 2011;36:512–520. doi: 10.1097/BRS.0b013e3181f72b94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez AG, Rodriguez-Soto AE, Burghardt AJ, et al. Morphology of the human vertebral endplate. J Orthop Res. 2012;30:280–287. doi: 10.1002/jor.21513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sambrook PN, MacGregor AJ, Spector TD. Genetic influences on cervical and lumbar disc degeneration: a magnetic resonance imaging study in twins. Arthritis Rheum. 1999;42:366–372. doi: 10.1002/1529-0131(199902)42:2<366::AID-ANR20>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Sato K, Kikuchi S, Yonezawa T. In vivo intradiscal pressure measurement in healthy individuals and in patients with ongoing back problems. Spine. 1999;24:2468–2474. doi: 10.1097/00007632-199912010-00008. [DOI] [PubMed] [Google Scholar]
- de Schepper EI, Damen J, van Meurs JB, et al. The association between lumbar disc degeneration and low back pain: the influence of age, gender, and individual radiographic features. Spine. 2010;35:531–536. doi: 10.1097/BRS.0b013e3181aa5b33. [DOI] [PubMed] [Google Scholar]
- Schmidt H, Kettler A, Rohlmann A, et al. The risk of disc prolapses with complex loading in different degrees of disc degeneration – a finite element analysis. Clin Biomech (Bristol, Avon) 2007;22:988–998. doi: 10.1016/j.clinbiomech.2007.07.008. [DOI] [PubMed] [Google Scholar]
- Schollum ML, Robertson PA, Broom ND. A microstructural investigation of intervertebral disc lamellar connectivity: detailed analysis of the translamellar bridges. J Anat. 2009;214:805–816. doi: 10.1111/j.1469-7580.2009.01076.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schollum ML, Robertson PA, Broom ND. How age influences unravelling morphology of annular lamellae - a study of interfibre cohesivity in the lumbar disc. J Anat. 2010;216:310–319. doi: 10.1111/j.1469-7580.2009.01197.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarzer AC, Aprill CN, Derby R, et al. The prevalence and clinical features of internal disc disruption in patients with chronic low back pain [see comments] Spine. 1995;20:1878–1883. doi: 10.1097/00007632-199509000-00007. [DOI] [PubMed] [Google Scholar]
- Seidler A, Bolm-Audorff U, Siol T, et al. Occupational risk factors for symptomatic lumbar disc herniation; a case–control study. Occup Environ Med. 2003;60:821–830. doi: 10.1136/oem.60.11.821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirazi-Adl A. Strain in fibers of a lumbar disc. Analysis of the role of lifting in producing disc prolapse. Spine. 1989;14:96–103. doi: 10.1097/00007632-198901000-00019. [DOI] [PubMed] [Google Scholar]
- Shirazi-Adl A. Finite-element simulation of changes in the fluid content of human lumbar discs. Mechanical and clinical implications. Spine. 1992;17:206–212. [PubMed] [Google Scholar]
- Sivan SS, Wachtel E, Tsitron E, et al. Collagen turnover in normal and degenerate human intervertebral discs as determined by the racemization of aspartic acid. J Biol Chem. 2008;283:8796–8801. doi: 10.1074/jbc.M709885200. [DOI] [PubMed] [Google Scholar]
- Skrzypiec D, Tarala M, Pollintine P, et al. When are intervertebral discs stronger than their adjacent vertebrae? Spine. 2007;32:2455–2461. doi: 10.1097/BRS.0b013e3181573b87. [DOI] [PubMed] [Google Scholar]
- Stefanakis M, Al-Abassi M, Harding I, et al. Annulus fissures are mechanically and chemically conducive to the ingrowth of nerves and blood vessels. Spine. 2012 doi: 10.1097/BRS.0b013e318263ba59. in press. [DOI] [PubMed] [Google Scholar]
- Sward L, Hellstrom M, Jacobsson B, et al. Disc degeneration and associated abnormalities of the spine in elite gymnasts. A magnetic resonance imaging study. Spine. 1991;16:437–443. doi: 10.1097/00007632-199104000-00009. [DOI] [PubMed] [Google Scholar]
- Twomey L, Taylor J. Age changes in lumbar intervertebral discs. Acta Orthop Scand. 1985;56:496–499. doi: 10.3109/17453678508993043. [DOI] [PubMed] [Google Scholar]
- Ulrich JA, Liebenberg EC, Thuillier DU, et al. ISSLS prize winner: repeated disc injury causes persistent inflammation. Spine. 2007;32:2812–2819. doi: 10.1097/BRS.0b013e31815b9850. [DOI] [PubMed] [Google Scholar]
- Veres SP, Robertson PA, Broom ND. The morphology of acute disc herniation: a clinically relevant model defining the role of flexion. Spine. 2009;34:2288–2296. doi: 10.1097/BRS.0b013e3181a49d7e. [DOI] [PubMed] [Google Scholar]
- Vernon-Roberts B, Pirie CJ. Healing trabecular microfractures in the bodies of lumbar vertebrae. Ann Rheum Dis. 1973;32:406–412. doi: 10.1136/ard.32.5.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Videman T, Nurminen M. The occurrence of anular tears and their relation to lifetime back pain history: a cadaveric study using barium sulfate discography. Spine. 2004;29:2668–2676. doi: 10.1097/01.brs.0000146461.27105.2b. [DOI] [PubMed] [Google Scholar]
- Videman T, Battie MC, Gill K, et al. Magnetic resonance imaging findings and their relationships in the thoracic and lumbar spine. Insights into the etiopathogenesis of spinal degeneration. Spine. 1995a;20:928–935. doi: 10.1097/00007632-199504150-00009. [DOI] [PubMed] [Google Scholar]
- Videman T, Sarna S, Battie MC, et al. The long-term effects of physical loading and exercise lifestyles on back-related symptoms, disability, and spinal pathology among men. Spine. 1995b;20:699–709. doi: 10.1097/00007632-199503150-00011. [DOI] [PubMed] [Google Scholar]
- Videman T, Battie MC, Gibbons LE, et al. Associations between back pain history and lumbar MRI findings. Spine. 2003;28:582–588. doi: 10.1097/01.BRS.0000049905.44466.73. [DOI] [PubMed] [Google Scholar]
- Videman T, Battie MC, Parent E, et al. Progression and determinants of quantitative magnetic resonance imaging measures of lumbar disc degeneration: a five-year follow-up of adult male monozygotic twins. Spine. 2008;33:1484–1490. doi: 10.1097/BRS.0b013e3181753bb1. [DOI] [PubMed] [Google Scholar]
- Videman T, Gibbons LE, Battie MC. Age- and pathology-specific measures of disc degeneration. Spine. 2008;33:2781–2788. doi: 10.1097/BRS.0b013e31817e1d11. [DOI] [PubMed] [Google Scholar]
- Williams FM, Manek NJ, Sambrook PN, et al. Schmorl's nodes: common, highly heritable, and related to lumbar disc disease. Arthritis Rheum. 2007;57:855–860. doi: 10.1002/art.22789. [DOI] [PubMed] [Google Scholar]
- Yu SW, Haughton VM, Sether LA, et al. Anulus fibrosus in bulging intervertebral disks. Radiology. 1988;169:761–763. doi: 10.1148/radiology.169.3.3186998. [DOI] [PubMed] [Google Scholar]
- Zhao F, Pollintine P, Hole BD, et al. Discogenic origins of spinal instability. Spine. 2005;30:2621–2630. doi: 10.1097/01.brs.0000188203.71182.c0. [DOI] [PubMed] [Google Scholar]


