Abstract
The Ca2+-ATPase of the plasma membrane (PM) of germinating radish (Raphanus sativus L.) seeds was purified by calmodulin (CaM)-affinity chromatography using a batch procedure. PM purified by aqueous two-phase partitioning was solubilized with n-dodecyl β-d-maltoside and applied to a CaM-agarose matrix. After various washings with decreasing Ca2+ concentrations, the Ca2+-ATPase was eluted with 5 mm ethylenediaminetetraacetate (EDTA). The EDTA-eluted fraction contained about 25% of the loaded Ca2+-ATPase activity, with a specific activity 70-fold higher than that of the starting PM fraction. The EDTA-eluted fraction was highly enriched in a 133-kD polypeptide, which was identified as the PM Ca2+-ATPase by 125I-CaM overlay and fluorescein-isothiocyanate labeling. The PM Ca2+-ATPase cross-reacted with an antiserum against a putative Ca2+-ATPase of the Arabidopsis thaliana chloroplast envelope.
Ca2+-pumping ATPases play a crucial role in maintaining Ca2+ homeostasis and in restoring it after the increase of cytosolic Ca2+ concentration brought about by different stimuli. Given the limited capacity of internal stores, the PM Ca2+-ATPase is particularly important in long-term control, especially to extrude Ca2+ entering the cell due to the opening of PM Ca2+ channels (Briskin, 1990; Evans et al., 1991; De Michelis et al., 1992; Poovaiah and Reddy, 1993; Ranjeva et al., 1993; Askerlund and Sommarin, 1996).
The PM Ca2+-ATPase is a P-type ATPase that catalyzes an H+/Ca2+ exchange (Rasi-Caldogno et al., 1987; Briskin, 1990; Evans et al., 1991; De Michelis et al., 1992; Askerlund and Sommarin, 1996). The PM Ca2+-ATPase activity is stimulated by CaM, which binds to an autoinhibitory domain of the enzyme, causing a strong increase in the Vmax and a decrease in the apparent Km for free Ca2+ (Malatialy et al., 1988; Robinson et al., 1988; Williams et al., 1990; Erdei and Matsumoto, 1991; Rasi-Caldogno et al., 1992, 1993, 1995; De Michelis et al., 1993; Kurosaki and Kaboraki, 1994). All of these characteristics resemble those of the PM Ca2+-ATPase of mammals (Carafoli, 1991). However, in contrast to mammals, plant PM-type Ca2+-ATPases are also present in endomembranes, and are often more abundant in endomembranes than in the PM (for review, see Evans, 1994; Askerlund and Sommarin, 1996). Moreover, the Ca2+-ATPase of the PM of plant cells has a very high affinity for CaM, so that stimulation by exogenous CaM is low or undetectable unless endogenous CaM is stripped by drastic treatments with Ca2+-chelating agents (Williams et al., 1990; Evans et al., 1992; Rasi-Caldogno et al., 1993).
CaM-stimulated Ca2+-ATPases of endomembranes and of PM have similar biochemical characteristics, and cannot be easily discerned (Hsieh et al., 1991; Askerlund and Evans, 1993; Thomson et al., 1993, 1994; Askerlund, 1996; Askerlund and Sommarin, 1996; Hwang et al., 1997). The main reported differences between the two types of plant CaM-stimulated Ca2+-ATPases are: (a) the sensitivity of the endomembrane enzymes to inhibition by fluorescein derivatives is slightly lower than that of the PM enzyme (Thomson et al., 1993; Bush and Wang, 1995; Askerlund and Sommarin, 1996), and (b) the molecular weight of the PM enzyme is higher than that of endomembrane CaM-stimulated Ca2+-ATPases (Thomson et al., 1993; Askerlund, 1996; Askerlund and Sommarin, 1996; Hwang et al., 1997).
Since the first report by Dieter and Marmè (1981), CaM-stimulated Ca2+-ATPases have been purified from different plant materials by CaM-affinity chromatography, but all of them were localized on endomembranes (Briars et al., 1988; Evans et al., 1989, 1992; Askerlund and Evans, 1992; Theodoulou et al., 1994; Askerlund, 1996; Hwang et al., 1997).
Germinating radish seeds are an experimental system particularly suitable for studying the PM-localized Ca2+-ATPase (Rasi-Caldogno et al., 1995). In fact, the endomembrane system is poorly developed, and all of the Ca2+-ATPase activity is localized on the PM (Rasi-Caldogno et al., 1987, 1989). Furthermore, highly purified PM vesicles are easily obtained by the aqueous two-phase partitioning technique, with quite a high yield (De Michelis et al., 1991; Rasi-Caldogno et al., 1995). Ca2+-ATPase activity in the PM is among the highest reported and can be easily monitored both as nucleoside-triphosphate-dependent Ca2+ transport and as Ca2+-dependent ITPase activity (Carnelli et al., 1992).
Here we show that affinity chromatography on CaM-agarose with a batch procedure purifies the PM Ca2+-ATPase of radish seeds by 70-fold, with a recovery of about 25%.
MATERIALS AND METHODS
Preparation of PM Vesicles
Methods for radish (Raphanus sativus L. Tondo Rosso Quarantino, Ingegnoli, Milano, Italy) seed germination, microsome extraction, and PM purification were as described previously (Rasi-Caldogno et al., 1995). CaM stripping was performed by incubating the upper phase for 10 min on ice in the presence of 20 mm BTP (1,3-bis[Tris (hydroxymethyl)methylamino]-propane)-Hepes, pH 7.5, 3 mm ITP, 30 mm EDTA, and 0.1 mg mL−1 Brij 58. The samples were diluted with 5 volumes of ice-cold medium containing 0.25 m Suc, 3 mm DTT, 0.1 mg mL−1 Brij 58, 1 mm PMSF, and 1 mm BTP-Hepes, pH 7.0, and the PM was collected by centrifugation at 48,000g for 35 min at 4°C. The pellets were placed in resuspension medium (10% [v/v] glycerol, 0.5 mm DTT, and 1 mm Mops-KOH, pH 7.0) at 6 to 8 mg of membrane proteins per mL, immediately frozen, and kept at −80°C until use.
Solubilization of PM Ca2+-ATPase
To solubilize PM Ca2+-ATPase, PM vesicles were incubated with n-dodecyl β-d-maltoside (4 mg detergent mL−1: 4 mg protein mL−1, unless otherwise specified) for 15 min on ice in a solubilization medium containing 10% (v/v) glycerol, 20 mm Mops-KOH, pH 7.5, 1 mm p-aminobenzamidine, 2 mm DTT, 1.5 mm ITP, 1 mm CaCl2, 1 mm MgSO4, 5 μg mL−1 leupeptin, and 0.25 m KBr, and then centrifuged for 35 min at 110,000g. The supernatant was added with 375 μg mL−1 Brij 58. When necessary, the pellets were placed in resuspension medium with 1 mm CaCl2 added.
CaM-Affinity Chromatography
Five-hundred microliters of CaM-agarose (catalog no. p-4385, Sigma) was transferred to a conical, 2-mL polypropylene tube and preequilibrated with solubilization medium with 0.5 mg mL−1 l-α-phosphatidylcholine and 375 μg mL−1 Brij 58 added. The solubilized PM proteins (approximately 1.2 mL) were applied and kept overnight under gentle rotation at 4°C. After a short centrifugation, the soluble phase (the unbound fraction) was aspirated and replaced with 1.2 mL of washing medium containing 10% (v/v) glycerol, 20 mm Mops-KOH, pH 7.5, 1 mm p-aminobenzamidine, 2 mm DTT, 100 μm CaCl2, 100 μm MgSO4, 5 μg mL−1 leupeptin, and 0.25 m KBr; a second wash was performed in the same medium in absence of CaCl2 and MgSO4. CaM-bound proteins were eluted in 1.2 mL of 10% (v/v) glycerol, 1 mm Mops-KOH, pH 7.5, 1 mm ITP, 375 μg mL−1 Brij 58, and 0.5 mg mL−1 l-α-phosphatidylcholine in the presence of 1 mm EGTA (twice) or 5 mm EDTA. The eluted fractions were added with stoichiometric CaCl2 to neutralize EGTA or EDTA, and immediately used for assay of Ca2+-ATPase activity or frozen in aliquots and kept at −80°C.
Assay of PM Ca2+-ATPase Activity
Unless otherwise specified, the hydrolytic activity of the PM Ca2+-ATPase was measured as Ca2+-dependent Mg-ITP hydrolysis (Carnelli et al., 1992). The assay medium contained 40 mm BTP-Hepes, pH 7.0, 50 mm KCl, 3 mm MgSO4, 0.1 mm ammonium molybdate, 1 mm ITP, 5 μm carbonyl cyanide p-[trifluoromethoxy]phenylhydrazone, 5 μm A23187, 1 μg mL−1 oligomycin, 5 mm (NH4)2SO4, 0.1 mg mL−1 Brij 58, and 1 mm EGTA plus or minus CaCl2 to give a free Ca2+ concentration of 50 μm (De Michelis et al., 1993). CaM was supplied at 20 μg mL−1; incubation was performed at 25°C for 90 min. Ca2+-ATPase activity was determined as the difference between the activity measured in presence of Ca2+ and that measured in its absence.
Treatment of the PM with FITC
The different fractions were diluted 20-fold with water to minimize interference by ITP (Rasi-Caldogno et al., 1995) and then incubated in 50 μm CaCl2 and 20 mm BTP-Hepes, pH 7.0, in the presence of 5 μm FITC from a freshly prepared 75 μm solution in N,N-dimethylformamide. After incubation for 15 min at 25°C, the samples were precipitated in 10% (v/v) TCA for 2 h at 0°C, and centrifuged for 30 min at 80,000g. The pellets were washed with water, centrifuged again, and placed in resuspension medium.
Protein Assay
Protein was assayed according to the method of Markwell et al. (1978). The EDTA-eluted fraction was first precipitated with 10% (v/v) TCA as decribed above, to avoid interference by l-α-phosphatidylcholine.
SDS-PAGE and Western Analysis
SDS-PAGE was performed according to the method of Laemmli (1970). The different fractions were incubated for 5 min on ice in a cocktail of protease inhibitors (Rasi-Caldogno et al., 1995), and then solubilized for 60 min at 25°C in 4% SDS, 3% β-mercaptoethanol, 20% (v/v) glycerol, 1 mm EDTA, and 20 mm H3PO4 adjusted to pH 2.4 with Tris. Proteins from different fractions (0.2–80 μg per lane) were loaded onto gel (7.5% Tris-Gly gel with 4% stacking gel, catalog no. 161-0900, Bio-Rad). After electrophoresis, the gel was stained using the silver-impregnation method (catalog no. AG-5, Sigma) or blotted as described by Rasi-Caldogno et al. (1995). Immunodetection of FITC-labeled proteins (Rasi-Caldogno et al., 1995) was performed with an anti-fluorescein rabbit IgG (H+L) fraction (catalog no. A-889, Molecular Probes, Sunnyvale, CA) and with a second antibody coupled to alkaline phosphatase (catalog no. A9919, Sigma). Immunodetection with an antiserum against the peptide encoded by PEA1 cDNA (kindly supplied by N.E. Hoffman, Carnegie Institution of Washington, Stanford, CA) was as described by Huang et al. (1993a). 125I-CaM overlay was also as described previously (Rasi-Caldogno et al., 1995).
Statistics
Data are from one experiment representative of at least three experiments performed on PM Ca2+-ATPase purified on three separate occasions. Assays of PM Ca2+-ATPase activity were run with three replicates; se of the assay did not exceed ± 4%.
RESULTS
Purification of the PM Ca2+-ATPase by CaM-Agarose-Affinity Chromatography
Previous work had shown that the PM Ca2+-ATPase is very sensitive to inactivation by detergents (Graf and Weiler, 1990; Kasai and Muto, 1991; Carnelli et al., 1992). In a first set of experiments we have thus compared the ability of different detergents to solubilize the Ca2+-ATPase from PM purified from radish seedlings in active form. With Triton X-100 or 3-([cholamidopropyl]dimethylammonio-)1-propanesulfonate no more than 10% of the PM Ca2+-ATPase activity could be recovered in the soluble fraction, even when phosphatidylcholine was added to the solubilization medium (Graf and Weiler, 1990; Hwang et al., 1997). Similar results were obtained with octylglucopyranoside, methylglucamide, and polyoxyethylene 8-myristyl ether (data not shown). Only solubilization with n-dodecyl β-d-maltoside (1:1 mg of detergent:mg of protein) yielded about 70% of the PM Ca2+-ATPase activity in the soluble fraction (Table I); varying the protein and detergent concentration between 1 and 10 mg mL−1 or the addition of phosphatidylcholine to the solubilization medium had no major effect on Ca2+-ATPase solubilization (data not shown).
Table I.
Purification of the PM Ca2+-ATPase by CaM-agarose affinity chromatography
| Fraction | Protein | Ca2+-ATPase
Activity
|
|||||
|---|---|---|---|---|---|---|---|
| Total
|
Specific
|
||||||
| −CaM | +CaM | ΔCaM | −CaM | +CaM | ΔCaM | ||
| mg | nmol Pi min−1 | μmol min−1 mg−1 protein | |||||
| PM | 5.7 | 96 | 260 | 164 (+171%) | 0.017 | 0.045 | 0.028 |
| Solubilized PM | 4.1 | 90 | 200 | 110 (+122%) | 0.022 | 0.049 | 0.027 |
| CaM-agarose | |||||||
| Unbound | 3.6 | 80 | 90 | 10 (+12%) | 0.022 | 0.025 | 0.003 |
| Wash no. 1 | NDa | 11 | 12 | 1 (+9%) | |||
| Wash no. 2 | ND | 4 | 4 | ||||
| 1 mm EGTA, eluate no. 1 | ND | 9 | 11 | 2 (+22%) | |||
| 1 mm EGTA, eluate no. 2 | ND | 9 | 12 | 3 (+33%) | |||
| 5 mm EDTA eluate | 0.02 | 16 | 58 | 42 (+260%) | 0.8 (×47) | 2.9 (×65) | 2.1 (×75) |
PM proteins were solubilized with n-dodecyl β-d-maltoside (4:4 mg detergent mL−1: mg protein mL−1) and purified by CaM-agarose-affinity chromatography as described in Methods. The first wash was performed in the presence of 100 μm CaCl2 and 100 μm MgSO4; the second one was performed in the absence of added divalent cations. Data in the brackets represent the percent stimulation by CaM (20 μg mL−1). Results are from one experiment, which is representative of more than 10 experiments.
ND, Not determined.
Given the low concentration of Ca2+-ATPase in the PM, for the purification of the enzyme by affinity chromatography with CaM-agarose, we chose to adopt a batch procedure that allows recovery of the eluted fractions in relatively small volumes (details are given in Methods). Preliminary experiments showed that no Ca2+-ATPase activity could be recovered unless phosphatidylcholine was included in the elution medium (data not shown; see also Hwang et al., 1997).
Table I shows the results of a typical purification procedure. Upon overnight incubation of PM protein solubilized with n-dodecyl β-d-maltoside (4:4 mg of detergent:mg of protein) with CaM-agarose, the bulk of CaM-stimulated Ca2+-ATPase activity bound to the matrix: the unbound fraction contained the bulk of protein and most of the CaM-independent Ca2+-ATPase activity measured in the solubilized fraction, but stimulation by CaM of the Ca2+-ATPase activity was virtually undetectable.
Subsequent washes of the matrix with decreasing concentrations of Ca2+ released only traces of Ca2+-ATPase activity, which was virtually insensitive to CaM. Also, elution of CaM-bound proteins with 1 mm EGTA released very low amounts of Ca2+-ATPase activity, also only slightly stimulated by CaM.
EDTA (5 mm) eluted a substantial amount of CaM-stimulated Ca2+-ATPase activity: in 10 purifications performed independently, the activity in the EDTA-eluted fraction, measured in the presence of CaM, was 25 ± 2% of the loaded activity. The CaM-agarose purification procedure determined a marked increase of CaM stimulation of the PM Ca2+-ATPase activity. In the experiment shown in Table I, CaM stimulation in the EDTA-eluted fraction was about 260% (compared with 120% in the solubilized PM), but in some preparations CaM stimulation of the Ca2+-ATPase in the EDTA-eluted fraction was up to 600% (see Fig. 1 and Table II). The source of this variability will be discussed below. The EDTA-eluted fraction contained only about 0.5% of the loaded proteins, so that it was 65-fold enriched in Ca2+-ATPase activity (and 75-fold in CaM-dependent Ca2+-ATPase activity) compared with the native PM.
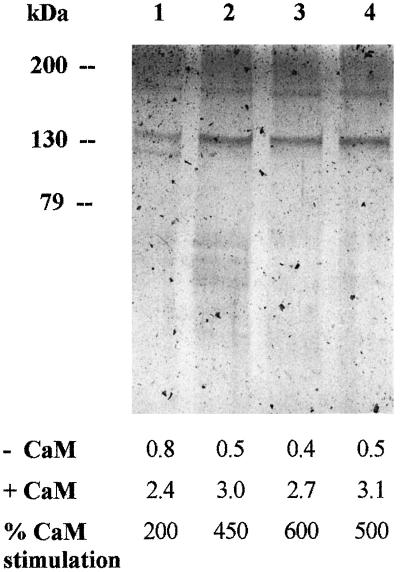
Figure 1.
Silver stain of EDTA-eluted fractions from different purifications with different stimulation by CaM. Each lane was loaded with similar Ca2+-ATPase activity (approximately 0.2 μg of proteins per lane). Values below the picture represent the Ca2+-ATPase activities, expressed in micromoles per minute per milligram of protein.
Table II.
Substrate specificity of the partially purified PM Ca2+-ATPase activity
| Assay Conditions | Nucleoside
Triphosphatase Activity
|
|
|---|---|---|
| ATP | ITP | |
| nmol min−1 mg−1 protein | ||
| −Ca2+ | 0.27 | 0.25 |
| +Ca2+ | 0.70 (0.43) | 0.64 (0.39) |
| +Ca2+ + CaM | 3.26 (2.99) | 3.04 (2.79) |
Assays were performed as described in “Materials and Methods,” in the presence of 1 mm ATP or ITP; values in parentheses represent the Ca2+-dependent activities.
In the experiment described in Table I, the PM Ca2+-ATPase activity was assayed as Ca2+-dependent ITPase activity, the procedure routinely used with native PM vesicles, to avoid interference due to the simultaneous activity of the much more abundant H+-ATPase (Rasi-Caldogno et al., 1989; Carnelli et al., 1992). Table II shows that in the EDTA-eluted fraction the nucleoside triphosphatase activity measured in the absence of Ca2+ was also extremely low when ATP was supplied as a substrate, indicating that the fraction was devoid of H+-ATPase activity.
Figure 1 shows the results of the electrophoretic separation of the EDTA-eluted fraction from four independent purification procedures. The EDTA-eluted fractions were highly enriched in a band of about 133 kD; two more bands at 180 and 120 kD were also visible. The intensity of the 120-kD band compared with the 133-kD band, was quite variable from experiment to experiment and was inversely related to the extent of CaM stimulation of Ca2+-ATPase activity.
Identification of the PM Ca2+-ATPase in the EDTA-Eluted Fraction
The molecular mass (133 kD) of the most abundant band in the EDTA-eluted fraction closely matches that previously reported for the intact Ca2+-ATPase in native PM, which was identified by different methods (Rasi-Caldogno et al., 1995).
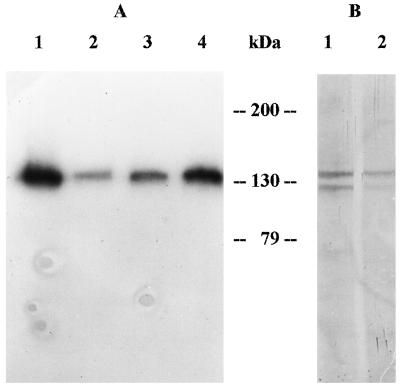
The Ca2+-ATPase is the major protein labeled by CaM overlay of western blots of PM proteins of radish seedlings (Rasi-Caldogno et al., 1995). Figure 2A shows the CaM overlay of a western blot of the main fractions of the CaM-agarose purification procedure. 125I-CaM heavily labeled a 133-kD band in the solubilized PM fraction (lane 1) and in the EDTA-eluted lane (lane 4); labeling was much weaker both in the fraction that did not bind to CaM-agarose (lane 2), and in the EGTA-eluted fraction (lane 3).
Figure 2.
A, Labeling of the PM Ca2+-ATPase of different fractions of the CaM-agarose purification procedure by 125I-CaM. Proteins were from solubilized PM (lane 1), unbound fraction (lane 2), EGTA-eluted fraction (lane 3), and EDTA-eluted fraction (lane 4). Each lane contained protein solubilized from 10 μL of the relevant fraction. B, Labeling of the PM Ca2+-ATPase of native PM (lane 1, 70 μg of proteins) and of the EDTA-eluted fraction (lane 2, 0.2 μg proteins) by FITC and immunodetection on western analysis with an anti-FITC antiserum.
The PM Ca2+-ATPase is extremely sensitive to inhibition by fluorescein derivatives (Giannini et al., 1987; Rasi-Caldogno et al., 1987, 1989, 1995; Olbe and Sommarin, 1991), which act as competitive inhibitors with the nucleoside triphosphates (De Michelis et al., 1993). Treatment of the PM with low concentrations of FITC selectively labels the Ca2+-ATPase, which can be easily detected on western analysis with an anti-FITC antiserum (Rasi-Caldogno et al., 1995). Figure 2B shows the western blot of native PM and of the EDTA-eluted fraction, labeled with FITC under selective conditions. In agreement with previous observations, FITC labeled two major bands of 133 and 120 kD in native PM (lane 1), which have been identified, respectively, as the intact Ca2+-ATPase and a product of its proteolysis lacking the CaM-binding domain (Rasi-Caldogno et al., 1995). In the EDTA-eluted fraction (lane 2), FITC labeled the same two bands, but the signal was much stronger for the 133-kD protein. Conversely, in the fraction that did not bind to CaM-agarose, FITC labeling was much stronger for the lower-molecular-mass band (data not shown).
The first plant cDNA encoding a polypeptide with high homology with the PM Ca2+-ATPase of mammals is PEA1, which encodes a putative Ca2+-ATPase of the plastid envelope of Arabidopsis thaliana (Huang et al., 1993b). Figure 3 shows the western blot of the major fractions of our purification procedure labeled with an antiserum against a portion of the protein encoded by PEA1 (kindly supplied by N.E. Hoffman). The antiserum labeled two bands of about 133 and 120 kD in all of the fractions tested. The 133-kD band was the more heavily labeled in the EDTA-eluted fraction (lane 4), whereas the fraction that did not bind to CaM-agarose (lane 3) was most enriched for the 120-kD protein.
Figure 3.
Immunodecoration of the main fractions of the CaM-agarose purification procedure, with an antiserum against a putative Ca2+-ATPase of the A. thaliana plastid envelope. Lane 1 was loaded with proteins from native PM (70 μg), lane 2 with proteins from solubilized PM (32 μg), lane 3 with proteins from the unbound fraction (28 μg), and lane 4 with proteins from the EDTA-eluted fraction (0.23 μg).
DISCUSSION
In this paper we report the first purification, to our knowledge, of the Ca2+-ATPase of the PM of plant cells. To achieve this goal we have used PM purified from germinating radish seedlings (Rasi-Caldogno et al., 1987, 1989, 1995).
The PM Ca2+-ATPase tightly bound to the CaM-agarose matrix and was not substantially eluted by EGTA (even when the concentration was increased up to 10 mm, data not shown), which has been widely used to elute the plant CaM-stimulated Ca2+-ATPases of endomembranes (Askerlund and Evans, 1992; Theodoulou et al., 1994; Askerlund, 1996; Hwang et al., 1997; Malmstrom et al., 1997). The tight binding of the PM Ca2+-ATPase to the CaM-agarose matrix suggests a high affinity of the enzyme for CaM. This result is in agreement with the previously reported observation that in native PM the Ca2+-ATPase is only slightly stimulated by exogenous CaM unless endogenous CaM is stripped by extensive washings with Ca2+ chelators (Williams et al., 1990; Rasi-Caldogno et al., 1993). In contrast, stimulation of the Ca2+-ATPase activity by exogenous CaM is easily detected in endomembrane fractions (Hsieh et al., 1991; Askerlund and Evans, 1992; Bush and Wang, 1995; Askerlund, 1996; Askerlund and Sommarin, 1996; Hwang et al., 1997). The affinity for CaM may thus be a crucial difference between the PM Ca2+-ATPase and Ca2+-ATPases of endomembranes, and is worthy of further investigation.
Washing of the column with 5 mm EDTA eluted about 25% of the loaded activity and as much as 40% of the CaM-dependent activity. The EDTA-eluted fraction, which was about 70-fold enriched in Ca2+-ATPase activity, was highly enriched in a 133-kD polypeptide, which was labeled by 125I-CaM overlay, as well as by treatment with low concentrations of FITC. All of these characteristics closely match those determined for the PM Ca2+-ATPase in native PM (Rasi-Caldogno et al., 1995). A second band of about 120 kD was present in variable amounts in the EDTA-eluted fraction. This band, which was enriched in the fraction that did not bind to CaM-agarose, and which was labeled by FITC but not by 125I-CaM overlay, has been previously identified as a proteolytic product of the PM Ca2+-ATPase (Rasi-Caldogno et al., 1995). Its presence in the purified Ca2+-ATPase fraction probably reflects the activity of a co-purifying protease activity; accordingly, it was most abundant in the fractions in which the Ca2+-ATPase activity was less stimulated by CaM (see Fig. 1B). Attempts to minimize proteolysis by means of various protease inhibitors have thus far been unsuccessful.
The yield and specific activity of the partially purified PM Ca2+-ATPase agree with the best reports on the endomembrane- localized, CaM-stimulated Ca2+-ATPase (Dieter and Marmè, 1981; Briars et al., 1988; Evans et al., 1989, 1992; Askerlund and Evans, 1992; Theodoulou et al., 1994; Askerlund, 1996; Hwang et al., 1997).
The PM Ca2+-ATPase cross-reacts with an antiserum against a putative chloroplast envelope Ca2+-ATPase of A. thaliana (Huang et al., 1993b), which is highly homologous to the tonoplast Ca2+-ATPase of cauliflower (Malmstrom et al., 1997), as well as to mammalian PM Ca2+-ATPases (Huang et al., 1993b). This result confirms that the same family of CaM-stimulated Ca2+-ATPases is expressed in different membranes of plant cells (Evans, 1994; Askerlund and Sommarin, 1996).
Availability of a purified Ca2+-ATPase of the PM is an essential prerequisite for cloning its gene. In fact, despite biochemical evidence indicating that CaM-stimulated Ca2+-ATPases are the most abundant in plant cells, screening of cDNA libraries with heterologous probes yielded only clones of putative Ca2+-ATPases homologous to the sarcoplasmic reticulum Ca2+-ATPase of mammals (Perez-Prat et al., 1992; Wimmers et al., 1992). This is probably due to the fact that heterologous probes mostly used for searching for P-type ATPases are based on the highly conserved nucleotide-binding site, which is likely to be variant in plant CaM-stimulated Ca2+-ATPases. In fact, plant CaM-stimulated Ca2+-ATPases differ from other P-type Ca2+-ATPases for their ability to use GTP or ITP as alternative substrates and for their very high sensitivity to inhibition by fluorescein derivatives (Rasi-Caldogno et al., 1987, 1989; Williams et al., 1990; Olbe and Sommarin, 1991; Carnelli et al., 1992; De Michelis et al., 1993), which act as competitive inhibitors with respect to the nucleoside-triphospate substrate (De Michelis et al., 1993).
Only very recently (Malmstrom et al., 1997) has a gene encoding the cauliflower tonoplast Ca2+-ATPase been identified using probes derived from the partial amino acid sequence of the purified enzyme: the deduced amino acid sequence shows high similarity to the mammalian PM Ca2+-ATPase, but has the striking characteristic that the putative CaM-binding domain appears localized in the N-terminal rather than in the C-terminal domain (Malmstrom et al., 1997). The putative chloroplast envelope Ca2+-ATPase, which is similar to the mammalian PM Ca2+-ATPase, lacks the C-terminal CaM-binding domain and has an extended N-terminal domain (Huang et al., 1993b); PM-type Ca2+-ATPases lacking the C-terminal CaM-binding domain have also been identified in the tonoplast of Saccharomyces cerevisiae (Cunningham and Fink, 1994) and Dictyostelium discoideum (Moniakis et al., 1995).
It will be interesting to find out whether displacement of the CaM-binding domain is a common feature of nonanimal PM-type Ca2+-ATPases, or if it is typical of the endomembrane-localized members of this family. Unfortunately, preliminary attempts to sequence the 133-kD polypeptide in our purified PM Ca2+-ATPase from radish seedlings have failed due to N-terminal blockage. Work is in progress to obtain partial amino acid sequences from tryptic fragments that are suitable for designing specific probes to isolate the PM Ca2+-ATPase cDNA.
ACKNOWLEDGMENTS
We wish to thank Dr. N.E. Hoffman (Carnegie Institution of Washington, Stanford, CA) for the generous gift of the antiserum against the putative plastid envelope Ca2+-ATPase.
Abbreviations:
- Brij 58
polyoxyethylene-20-cetyl ether
- CaM
calmodulin
- FITC
fluorescein isothiocyanate
- PM
plasma membrane
Footnotes
This work was supported by a grant from the Italian Ministry for University and Scientific and Technologic Research (40% quote).
LITERATURE CITED
- Askerlund P. Modulation of an intracellular calmodulin-stimulated Ca2+-pumping ATPase in cauliflower by trypsin. Plant Physiol. 1996;110:913–922. doi: 10.1104/pp.110.3.913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Askerlund P, Evans DE. Reconstitution and characterization of a calmodulin-stimulated Ca2+-pumping ATPase purified from Brassica oleracea L. Plant Physiol. 1992;100:1670–1681. doi: 10.1104/pp.100.4.1670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Askerlund P, Evans DE. Plant Physiol Biochem. 1993;31:787–791. [Google Scholar]
- Askerlund P, Sommarin M (1996) Calcium efflux tranporters in higher plants. In M Smallwood, JP Knox, DJ Bowles, eds, Membranes: Specialized Functions in Plants. BIOS Scientific Publishers Ltd., Oxford, UK, pp 281–299
- Briars SA, Kessler F, Evans DE. The calmodulin-stimulated ATPase of maize coleoptiles is a 140000-Mr polypeptide. Planta. 1988;176:283–285. doi: 10.1007/BF00392457. [DOI] [PubMed] [Google Scholar]
- Briskin DP. Ca2+-traslocating ATPase of the plant plasma membrane. Plant Physiol. 1990;94:397–400. doi: 10.1104/pp.94.2.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bush DS, Wang T. Diversity of calcium-efflux transporters in wheat aleurone cells. Planta. 1995;197:19–30. [Google Scholar]
- Carafoli E. Calcium pump of the plasma membrane. Physiol Rev. 1991;71:129–153. doi: 10.1152/physrev.1991.71.1.129. [DOI] [PubMed] [Google Scholar]
- Carnelli A, De Michelis MI, Rasi-Caldogno F. Plasma membrane Ca-ATPase of radish seedlings. I. Biochemical characteristics using ITP as a substrate. Plant Physiol. 1992;98:1196–1201. doi: 10.1104/pp.98.3.1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham KW, Fink GR. Calcineurin-dependent growth control in Saccharomyces cerevisiae mutants lacking PMC1, a homolog of plasma membrane Ca2+-ATPases. J Cell Biol. 1994;124:351–363. doi: 10.1083/jcb.124.3.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Michelis MI, Carnelli A, Rasi-Caldogno F. The Ca2+ pump of the plasma membrane of Arabidopsis thaliana: characteristics and sensitivity to fluorescein derivatives. Bot Acta. 1993;106:20–25. [Google Scholar]
- De Michelis MI, Rasi-Caldogno F, Pugliarello M, Olivari C. Fusicoccin binding to its plasma membrane receptor and the activation of the plasma membrane H+-ATPase. II. Stimulation of the H+-ATPase in a plasma membrane fraction purified by phase-partitioning. Bot Acta. 1991;104:265–271. [Google Scholar]
- De Michelis MI, Rasi-Caldogno F, Pugliarello MC. The plasma membrane Ca2+ pump: potential role in Ca2+ homeostasis. In: Karssen CM, Van Loon LC, Vreugdenhil D, editors. Progress in Plant Growth Regulation. Dordrecht, The Netherlands: Kluwer Academic Publishers; 1992. pp. 675–685. [Google Scholar]
- Dieter P, Marmè D. A calmodulin dependent microsomal ATPase from corn (Zea mays L.) FEBS Lett. 1981;125:245–248. [Google Scholar]
- Erdei L, Matsumoto H. Activation of the Ca2+-Mg2+-ATPase by Ca-EGTA in plasmalemma from the roots of cucumber. Biochem Physiol Pflanzen. 1991;187:189–195. [Google Scholar]
- Evans DE. Calmodulin-stimulated calcium pumping ATPases located at higher plant intracellular membranes: a significant divergence from other eukaryotes? Physiol Plant. 1994;90:420–426. [Google Scholar]
- Evans DE, Askerlund P, Boyce JM, Briars SA, Coates J, Cooke DT, Theodouolu FL (1992) Studies on higher plant calmodulin stimulated ATPase. In DT Cooke, DT Clarkson, eds, Transport and Receptor Proteins of Plant Membranes. Plenum Press, New York, pp 39–53
- Evans DE, Briars SA, Williams LE. Active calcium transport by plant cell membranes. J Exp Bot. 1991;42:285–303. [Google Scholar]
- Evans DE, Dewey FM, Briars SA. The calcium pumping ATPase of the plant plasma membrane. In: Dainty J, De Michelis MI, Marrè E, Rasi-Caldogno F, editors. Plant Membrane Transport: The Current Position. Amsterdam, The Netherlands: Elsevier; 1989. pp. 231–236. [Google Scholar]
- Giannini JL, Gildensoph LH, Reynolds-Niesman, Briskin DP. Calcium transport in sealed vesicles from red beet (Beta vulgaris L.) storage tissue. II. Characterization of 45Ca uptake into plasma membrane vesicles. Plant Physiol. 1987;85:1137–1142. doi: 10.1104/pp.85.4.1129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graf P, Weiler EW. Functional reconstitution of an ATP-driven Ca2+-transport system from the plasma membrane of Commelina communis L. Plant Physiol. 1990;94:634–640. doi: 10.1104/pp.94.2.634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh WL, Pierce WS, Sze H. Calcium pumping ATPases in vesicles from carrot cells. Stimulation by calmodulin or phosphatidylserine, and formation of a 120 kilodalton phosphoenzyme. Plant Physiol. 1991;97:1535–1544. doi: 10.1104/pp.97.4.1535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L, Berkelman T, Franklin AE, Hoffman NE. Primary structure and characterization of an Arabidopsis thaliana calnexin-like protein. J Biol Chem. 1993a;268:6560–6566. [PubMed] [Google Scholar]
- Huang L, Berkelman T, Franklin AE, Hoffman NE. Characterization of a gene encoding a Ca2+-ATPase-like protein in the plastid envelope. Proc Natl Acad Sci USA. 1993b;90:10066–10070. doi: 10.1073/pnas.90.21.10066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang I, Ratterman DM, Sze H. Distinction between endoplasmic reticulum-type and plasma membrane-type Ca2+ pumps. Partial purification of a 120-kilodalton Ca2+-ATPase from endomembranes. Plant Physiol. 1997;113:535–548. doi: 10.1104/pp.113.2.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasai M, Muto S. Solubilization and reconstitution of Ca2+ pump from corn leaf plasma membrane. Plant Physiol. 1991;96:565–570. doi: 10.1104/pp.96.2.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurosaki F, Kaburaki H. Calmodulin-dependency of a Ca2+-pump at the plasma membrane of cultured carrot cells. Plant Sci. 1994;104:23–30. [Google Scholar]
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Malatialy L, Greppin H, Penel C. Calcium uptake by tonoplast and plasma membrane vesicles from spinach leaves. FEBS Lett. 1988;233:196–200. [Google Scholar]
- Malmstrom S, Askerlund P, Palmgren MG. A calmodulin-stimulated Ca2+-ATPase from plant vacuolar membranes with a putative regulatory domain at its N-terminus. FEBS Lett. 1997;400:324–328. doi: 10.1016/s0014-5793(96)01448-2. [DOI] [PubMed] [Google Scholar]
- Markwell MAK, Haas SM, Bicher LL, Tolbert NE. A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem. 1978;87:206–210. doi: 10.1016/0003-2697(78)90586-9. [DOI] [PubMed] [Google Scholar]
- Moniakis J, Coukell MB, Forer A. Molecular cloning of an intracellular P-type ATPase from Dyctiostelium that is up-regulated in calcium-adapted cells. J Biol Chem. 1995;270:28276–28281. doi: 10.1074/jbc.270.47.28276. [DOI] [PubMed] [Google Scholar]
- Olbe M, Sommarin M. ATP-dependent Ca2+ transport in wheat root plasma membrane vesicles. Physiol Plant. 1991;83:535–543. [Google Scholar]
- Perez-Prat E, Naramisham MI, Binze ML, Botella MA, Chen Z, Valpuesta V, Bressan RA, Hasegawa PM. Induction of a putative Ca2+-ATPase mRNA in NaCl-adapted cells. Plant Physiol. 1992;100:1471–1478. doi: 10.1104/pp.100.3.1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poovaiah BW, Reddy ASN. Calcium and signal transduction in plants. CRC Crit Rev Plant Sci. 1993;12:185–211. doi: 10.1080/07352689309701901. [DOI] [PubMed] [Google Scholar]
- Ranjeva R, Thulaeu P, Schroeder JI. Signal transduction and calcium channels in higher plants. Curr Opin Biotechnol. 1993;4:172–176. [Google Scholar]
- Rasi-Caldogno F, Carnelli A, De Michelis MI. Plasma membrane Ca-ATPase of radish seedlings. II. Regulation by calmodulin. Plant Physiol. 1992;98:1202–1206. doi: 10.1104/pp.98.3.1202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasi-Caldogno F, Carnelli A, De Michelis MI. Controlled proteolysis activates the plasma membrane Ca2+ pump of higher plants. Plant Physiol. 1993;103:385–390. doi: 10.1104/pp.103.2.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasi-Caldogno F, Carnelli A, De Michelis MI. Identification of the plasma membrane Ca2+-ATPase and of its autoinhibitory domain. Plant Physiol. 1995;108:105–113. doi: 10.1104/pp.108.1.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasi-Caldogno F, Pugliarello MC, De Michelis MI. The Ca2+-transport ATPase of plant plasma membrane catalyzes a nH+/Ca2+ exchange. Plant Physiol. 1987;83:994–1000. doi: 10.1104/pp.83.4.994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasi-Caldogno F, Pugliarello MC, Olivari C, De Michelis MI. Identification and characterization of the Ca2+-ATPase which drives active transport of Ca2+ at the plasma membrane of radish seedlings. Plant Physiol. 1989;90:1429–1434. doi: 10.1104/pp.90.4.1429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson C, Larsson C, Buckhout TJ. Identification of a calmodulin-stimulated (Ca2+ + Mg2+)-ATPase in a plasma membrane fraction isolated from maize (Zea mays) Physiol Plant. 1988;72:177–194. [Google Scholar]
- Theodoulou FL, Dewey FM, Evans DE. Calmodulin stimulated ATPase of maize cells: functional reconstitution, monoclonal antibodies and subcellular localization. J Exp Bot. 1994;45:1553–1564. [Google Scholar]
- Thomson LJ, Hall JL, Williams LE. A study of the effect of inhibitors of the animal sarcoplasmic/endoplasmic reticulum-type calcium pumps on the primary Ca2+-ATPases of red beet. Plant Physiol. 1994;104:1295–1300. doi: 10.1104/pp.104.4.1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomson LJ, Xing T, Hall JL, Williams LE. Investigation of the calcium-transporting ATPases at the endoplasmic reticulum and plasma membrane of red beet (Beta vulgaris) Plant Physiol. 1993;102:553–564. doi: 10.1104/pp.102.2.553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams LE, Schueler SB, Briskin DP. Further characterization of the red beet plasma membrane Ca2+-ATPase using GTP as an alternative substrate. Plant Physiol. 1990;92:747–754. doi: 10.1104/pp.92.3.747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wimmers LE, Ewing NN, Bennett AB. Higher plant Ca2+-ATPase: primary structure and regulation of mRNA abundance by salt. Proc Natl Acad Sci USA. 1992;89:9205–9209. doi: 10.1073/pnas.89.19.9205. [DOI] [PMC free article] [PubMed] [Google Scholar]