Abstract
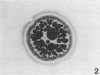
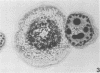
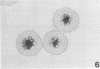
To obtain a wider definition of variation in the virulence of Bacteroides nodosus and in the protective potency of B. nodosus vaccines, we made a comparison of the in vitro characteristics of isolates from clinical infections of sheep and cattle and of certain colony variants observed previously. Three basic colony types were distinguished: papillate or beaded (B)-type colonies were produced by fresh isolates from advanced ovine foot rot; mucoid (M)-type colonies were formed by isolates from noninvasive B. nodosus infections of the interdigital skin of sheep and cattle, and also by cultures of some primary B-type colonies passaged nonselectively in vitro; and circular (C)-type colonies were formed by B. nodosus that eventually predominated in repeatedly passaged liquid subcultures. Each type could be maintained by selective agar subculturing; one strain was thus passaged 40 times as the B-type colony, at which point the organisms induced severe foot rot in experimentally infected sheep. Cultures of M-type colonies were uniformly less pathogenic and those of C-type colony organisms were avirulent. In vitro changes from prototype B-type colonies to M- and C-variants were nonreversible in these experiments, were accelerated in liquid cultures, and wee accompanied by a diminution in elastase activity of the organisms and in their immunoprotective properties against homologous challenge. Strains differed in their stability to these changes; therefore the choice of an appropriate strain and colony type may be an important consideration in studies of B. nodosus virulence and in the development of effective vaccines.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cooper B. S. Differences in morphology of Bacteroides nodosus attributable to culture media. N Z Vet J. 1977 Jan-Feb;25(1-2):16–25. doi: 10.1080/00480169.1977.34341. [DOI] [PubMed] [Google Scholar]
- Depiazzi L. J., Richards R. B. A degrading proteinase test to distinguish benign and virulent ovine isolates of Bacteroides nodosus. Aust Vet J. 1979 Jan;55(1):25–28. doi: 10.1111/j.1751-0813.1979.tb09541.x. [DOI] [PubMed] [Google Scholar]
- Egerton J. R., Burrell D. H. Prophylactic and therapeutic vaccination against ovine foot-rot. Aust Vet J. 1970 Nov;46(11):517–522. doi: 10.1111/j.1751-0813.1970.tb06636.x. [DOI] [PubMed] [Google Scholar]
- Egerton J. R., Merritt G. C. Serology of foot-rot: antibodies against Fusiformis nodosus in normal, affected, vaccinated and passively immunised sheep. Aust Vet J. 1973 Mar;49(3):139–145. doi: 10.1111/j.1751-0813.1973.tb06762.x. [DOI] [PubMed] [Google Scholar]
- Egerton J. R., Parsonson I. M. Benign foot-rot--a specific interdigital dermatitis of sheep associated with infection by less proteolytic strains of Fusiformis nodosus. Aust Vet J. 1969 Aug;45(8):345–349. doi: 10.1111/j.1751-0813.1969.tb06606.x. [DOI] [PubMed] [Google Scholar]
- Egerton J. R., Roberts D. S., Parsonson I. M. The aetiology and pathogenesis of ovine foot-rot. I. A histological study of the bacterial invasion. J Comp Pathol. 1969 Apr;79(2):207–215. doi: 10.1016/0021-9975(69)90007-3. [DOI] [PubMed] [Google Scholar]
- Egerton J. R., Roberts D. S. Vaccination against ovine foot-rot. J Comp Pathol. 1971 Apr;81(2):179–185. doi: 10.1016/0021-9975(71)90091-0. [DOI] [PubMed] [Google Scholar]
- Egerton J. R. Significance of Fusiformis nodosus serotypes in resistance of vaccinated sheep to experimental foot-rot. Aust Vet J. 1974 Feb;50(2):59–62. doi: 10.1111/j.1751-0813.1974.tb05252.x. [DOI] [PubMed] [Google Scholar]
- Egerton J. R. Surface and somatic antigens of Fusiformis nodosus. J Comp Pathol. 1973 Jan;83(1):151–159. doi: 10.1016/0021-9975(73)90038-8. [DOI] [PubMed] [Google Scholar]
- Every D. Purification of pili from Bacteroides nodosus and an examination of their chemical, physical and serological properties. J Gen Microbiol. 1979 Dec;115(2):309–316. doi: 10.1099/00221287-115-2-309. [DOI] [PubMed] [Google Scholar]
- Jephcott A. E., Reyn A., Birch-Andersen A. Neisseria gonorrhoeae 3. Demonstration of presumed appendages to cells from different colony types. Acta Pathol Microbiol Scand B Microbiol Immunol. 1971;79(3):437–439. doi: 10.1111/j.1699-0463.1971.tb00086.x. [DOI] [PubMed] [Google Scholar]
- KELLOGG D. S., Jr, PEACOCK W. L., Jr, DEACON W. E., BROWN L., PIRKLE D. I. NEISSERIA GONORRHOEAE. I. VIRULENCE GENETICALLY LINKED TO CLONAL VARIATION. J Bacteriol. 1963 Jun;85:1274–1279. doi: 10.1128/jb.85.6.1274-1279.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellogg D. S., Jr, Cohen I. R., Norins L. C., Schroeter A. L., Reising G. Neisseria gonorrhoeae. II. Colonial variation and pathogenicity during 35 months in vitro. J Bacteriol. 1968 Sep;96(3):596–605. doi: 10.1128/jb.96.3.596-605.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraus S. J., Brown W. J., Arko R. J. Acquired and natural immunity to gonococcal infection in chimpanzees. J Clin Invest. 1975 Jun;55(6):1349–1356. doi: 10.1172/JCI108054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laing E. A., Egerton J. R. The occurrence, prevalence and transmission of Bacteroides nodosus infection in cattle. Res Vet Sci. 1978 May;24(3):300–304. [PubMed] [Google Scholar]
- McGee Z. A., Dourmashkin R. R., Gross J. G., Clark J. B., Taylor-Robinson D. Relationship of pili to colonial morphology among pathogenic and nonpathogenic species of Neisseria. Infect Immun. 1977 Feb;15(2):594–600. doi: 10.1128/iai.15.2.594-600.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ris D. R. The complement fixation test for the diagnosis of Brucella ovis infection in sheep. N Z Vet J. 1974 Aug;22(8):143–146. doi: 10.1080/00480169.1974.34152. [DOI] [PubMed] [Google Scholar]
- Roberts D. S., Foster W. H., Kerry J. B., Calder H. A. An alum-treated vaccine for the control of foot-rod in sheep. Vet Rec. 1972 Oct 28;91(18):428–429. doi: 10.1136/vr.91.18.428. [DOI] [PubMed] [Google Scholar]
- Short J. A., Thorley C. M., Walker P. D. An electron microscope study of Bacteroides nodusus: ultrastructure of organisms from primary isolates and different colony types. J Appl Bacteriol. 1976 Jun;40(3):311–315. doi: 10.1111/j.1365-2672.1976.tb04179.x. [DOI] [PubMed] [Google Scholar]
- Skerman T. M., Cairney I. M. Experimental observations on prophylactic and therapeutic vaccination against foot-rot in sheep. N Z Vet J. 1972 Nov;20(11):205–211. doi: 10.1080/00480169.1972.34052. [DOI] [PubMed] [Google Scholar]
- Skerman T. M. Determination of some in vitro growth requirements of Bacteroides nodosus. J Gen Microbiol. 1975 Mar;87(1):107–119. doi: 10.1099/00221287-87-1-107. [DOI] [PubMed] [Google Scholar]
- Stewart D. J. The role of elastase in the differentiation of Bacteroides nodosus infections in sheep and cattle. Res Vet Sci. 1979 Jul;27(1):99–105. [PubMed] [Google Scholar]
- Stewart D. J. The role of various antigenic fractions of Bacteroides nodosus in eliciting protection against foot-rot in vaccinated sheep. Res Vet Sci. 1978 Jan;24(1):14–19. [PubMed] [Google Scholar]
- Swanson J., Kraus S. J., Gotschlich E. C. Studies on gonococcus infection. I. Pili and zones of adhesion: their relation to gonococcal growth patterns. J Exp Med. 1971 Oct 1;134(4):886–906. doi: 10.1084/jem.134.4.886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorley C. M. A simplified method for the isolation of Bacteroides nodusus from ovine foot-rot and studies on its colony morphology and serology. J Appl Bacteriol. 1976 Jun;40(3):301–309. doi: 10.1111/j.1365-2672.1976.tb04178.x. [DOI] [PubMed] [Google Scholar]
- Walker P. D., Short J., Thomson R. O., Roberts D. S. The fine structure of Fusiformis nodosus with special reference to the location of antigens associated with immunogenicity. J Gen Microbiol. 1973 Aug;77(2):351–361. doi: 10.1099/00221287-77-2-351. [DOI] [PubMed] [Google Scholar]