Abstract
Although mesenchymal stem cells (MSCs) have been increasingly trialed to treat a variety of diseases, the underlying mechanisms remain still elusive. In this study, human umbilical cord (UC)-derived MSCs were stimulated by hypoxia, and the membrane microvesicles (MVs) in the supernatants were collected by ultracentrifugation, observed under an electron microscope, and the origin was identified with the flow cytometric technique. The results showed that upon hypoxic stimulus, MSCs released a large quantity of MVs of ∼100 nm in diameter. The MVs were phenotypically similar to the parent MSCs, except that the majority of them were negative for the receptor of platelet-derived growth factor. DiI-labeling assay revealed that MSC-MVs could be internalized into human UC endothelial cells (UC-ECs) within 8 h after they were added into the culture medium. Carboxyfluorescein succinimidyl ester-labeling technique and MTT test showed that MSC-MVs promoted the proliferation of UC-ECs in a dose-dependent manner. Further, MVs could enhance in vitro capillary network formation of UC-ECs in a Matrigel matrix. In a rat hindlimb ischemia model, both MSCs and MSC-MVs were shown to improve significantly the blood flow recovery compared with the control medium (P<0.0001), as assessed by laser Doppler imaging analysis. These data indicate that MV releasing is one of the major mechanisms underlying the effectiveness of MSC therapy by promoting angiogenesis.
Introduction
Mesenchymal stem cells (MSCs) are characterized by their highly proliferative activity, regulatory actions on nearly all the immune cells, and ability to differentiate into at least 3 lineages, including osteoblasts, chondrocytes, and adipocytes [1–3]. MSCs have been discovered in virtually all adult and extraembryonic tissues, including compact bone, tendon, adipose, placenta, and umbilical cord (UC) [4,5], since bone marrow MSCs were first described in detail [1]. Therefore, MSCs have been utilized in many clinical trials for a wide range of therapeutic applications, such as immune-associated disorders, including graft-versus-host disease and autoimmune diseases, bone and cartilage repair, cardiac infarction, neurological degenerative diseases, liver failure, diabetics, and other conditions in which tissue damage is potentially or definitely involved [6–9].
Of note, the transplanted MSCs are unable to survive in vivo as a result of death cues, including hypoxia, inflammatory cytokines, and proapoptotic factors, no matter they are systemically [10–13] or locally administered [14,15]. Therefore, a paracrine mechanism has been considered to explain MSC therapy in the tissue damage in organs such as the heart, liver, brain, lungs, kidneys, and pancreas [16–18]. However, the nature of the factors responsible for the beneficial paracrine effects of MSCs remains elusive.
It has been proposed that death signals might play an active role in the repair of the damaged tissues [19,20], and microvesicles (MVs) released by the injured cells are one of the important actors mediating the crosstalk with the surrounding cells [21–23]. A pioneering work from Camussi and his colleagues demonstrates that MVs derived from human bone marrow MSCs are as effective as MSCs in protecting the kidney from glycerol-induced acute injury in immunodeficient mice [24]. A recent study from the same group proves that MSC-MVs can also protect against ischaemia–reperfusion-induced acute and chronic kidney injury [25]. These investigations suggest that the beneficial effect of MSCs is, at least in part, due to the release of MVs [22,23,26]. To detail the functional activities of MSC-MVs, human UC-MSCs were stimulated by hypoxia, and the angiogenesis-promoting effects of MSC-MVs were investigated. The results here imply that MSC-MVs could promote blood vessel formation both in vitro and in vivo, providing some novel information about the mechanisms of MSC therapy.
Materials and Methods
Cell culture
Human UCs were harvested after obtaining informed consent, according to the Ethics Guidelines for Research Involving Human Subjects or Human Tissue from the General Hospital of Air Force. UC-MSCs were cultured and identified as previously described in our lab [27]. Briefly, UCs were cut into small pieces and digested with collagenase type I. The cells were cultured in minimum essential medium alpha (alpha-MEM) containing 10% fetal bovine serum (FBS) selected from lots (Stem Cell Co.), and the adherent cells were passaged and identified by surface marker analysis and in vitro differentiation assays [27]. MSCs at passages 3–5 were used in all the experiments below.
Meanwhile, UC endothelial cells (UC-ECs) were prepared from the UCs. In brief, the veins were washed twice with phosphate-buffered saline (PBS), and the ECs were flushed out after digestion with 0.25% trypsin for 30 min at room temperature. UC-ECs were seeded onto fibronectin-/gelatin-coated plastic dishes in Dulbecco's modified Eagle medium (DMEM) containing 20% FBS, vascular endothelial growth factor (50 ng/mL), and basic fibroblast growth factor (bFGF, 10 ng/mL). The ECs at passage 2 were used in the experiments as described below.
Hypoxic stimulation and apoptosis detection
UC-MSCs were harvested by enzyme digestion, and the cell viability was >98% as detected by the trypan blue exclusion test. Aliquots of 5×105 cells suspended in alpha-MEM were plated into culture dishes of 150 mm in diameter. The cells were then cultured under the following 4 conditions: (1) with 10% FBS in normoxia; (2) serum free in normoxia; (3) with 10%fetal in hypoxia; and (4) serum free in hypoxia. For normoxic conditions, the cells were cultured in an atmosphere containing 5% CO2, while for hypoxia, the cells were maintained in a gas mixture composed of 94% N2, 5% CO2, and 1% O2. The cultures were maintained for 72 h. The cell supernatants were collected after gentle shaking, and the adherent cells were washed with PBS for twice and digested with 0.05% trypsin and 1 mM ethylenediaminetetraacetic acid for 5 min at room temperature. The digestion was neutralized with the cell supernatants, and the cells were collected by centrifugation for 6 min at a speed of 500 g. After washing with PBS, the cells were reacted with the Annexin V-fluorescent isothiocyanate apoptosis detection kit (Biosea Biotechnology) according to the manufacturer's instructions. At least 10,000 events were collected by FACSCaliber (BD Biosciences), and the data were analyzed with WinMdi 2.9 software.
MV harvest
Aliquots (5×105 cells) of UC-MSCs were plated into 150-mm dishes in alpha-MEM containing 10% FBS (Stem Cell Co.). The serum was ultracentrifuged (100,000 g, 1 h) to eliminate MV contamination before its use. Cell culture was maintained under a hypoxic condition (1% O2) for 72 h. In some cases, UC-MSCs were incubated with DiI dye (5 μM, Sigma) in a serum-free medium for 30 min at 37°C. The cells were washed in PBS and maintained under hypoxia as described above. MVs were isolated from the culture supernatants according to the protocol described previously [28]. Briefly, cell supernatants were centrifuged to discard large debris, followed by filtration through a 0.2-μm Super Membrane (Pall Life Sciences). After additional centrifugation at 100,000 g for 1 h at 4°C, the pellet was washed twice in the apop buffer containing 5 mM KCl, 1 mM MgCl2, and 136 mM NaCl to eliminate protein contamination [29]. MVs were then resuspended in 50 μL apop buffer for one Ultra-Clear™ tube (14×89 mm; Beckman), split into aliquots of 5 μL, and stored at −80°C for the use in the experiments below.
MV identification
The phenotypic profile was determined by a bead-based flow cytometric technique according to the protocol described previously with a slight modification [28]. In brief, MSC-MVs were bound to aldehyde/sulfate latex beads (4 μm; Molecular Probes; Invitrogen) suspended in an MES buffer (0.025 M MES, 0.154 M NaCl, pH 6.0), followed by antibody incubation and data collection by a flow cytometry. Single beads and bead doublets were gated, and the relative fluorescence was analyzed with WinMdi2.9 software [28].
The amount of MVs was determined by measuring total MV-associated proteins, using the Bradford method, and BSA (Sigma-Aldrich) for the standard curve [29]. Also, MVs were observed directly under a transmission electron microscope (Hitachi H-7650), and the photos were taken at a magnification of 10,000.
Internalization of MSC-MVs into UC-ECs
UC-ECs at passage 2 were plated onto fibronectin/gelatin-coated dishes and maintained at 37°C overnight for plastic adherence. DiI-labeled MSC-MVs at a concentration of 10 μg/mL were then added, and the culture was maintained under normoxia or hypoxia as described above. At different time points, the cells were observed with a fluorescent microscopy, and also, UC-ECs were harvested by trypsin digestion, washed with PBS twice at a speed of 500 g for 5 min, and the relative fluorescence intensity was analyzed after the events were collected by FACSCalibur.
Carboxyfluorescein succinimidyl ester-labeling technique
A carboxyfluorescein succinimidyl ester (CFSE) dye dilution assay was used to observe the cell proliferative status as previously reported [30]. Briefly, human UC-ECs were prelabeled by CFSE dye, seeded onto plastic dishes, and cultured in alpha-MEM containing 1% FBS with different doses of MSC-MVs or 10 ng/mL of bFGF. The labeled cells cultured in alpha-MEM containing 5% and 10% FBS were served as the positive controls. The culture was maintained in hypoxia or normoxia for 72 h. The cells were harvested by enzyme digestion, washed in PBS, and the events were collected by FACSCalibur. The data were analyzed by ModFit software (Verity Software House). Using the proliferation wizard basic model of this software, CFSE-labeled cell populations were distributed into 10 generations of cell division after the parent histogram had been curved from the CFSE-labeled cells before culture.
MTT test
MTT test was performed as routinely described. Briefly, aliquots of UC-ECs (2000 cells) were seeded into a 96-well tissue plate. MTT was then added at different time points, and the culture was maintained for 4 h. The formazan was dissolved in dimethyl sulfoxide, and the optical density (OD) at a wavelength of 490 nm was assessed.
Tube formation assay
Aliquots of UV-ECs (1×105) were seeded onto Matrigel-coated wells of a 24-well plate and cultured in 1% fetal calf serum-supplemented DMEM in the presence of MSC-MVs at the indicated concentrations or 10 ng/mL of recombinant human basic fibroblast growth factor (PeproTech) for 24 h. Triple wells were set for each concentration, and the cells were maintained in hypoxia or normoxia. Tube formation was examined by a phase-contrast microscopy (MOTIC AE21), and the number of the network structure was quantified by randomly selecting 5 fields per well as previously described [31,32].
Rat model of hind-limb ischemia
All the animal experiments were carried out according to the approved institutional protocols from the Guide for the Care and Use of Laboratory Animals provided by Beijing Institute of Radiation Medicine. Female Wistar rats, aged 10–12 weeks and weighed around 170 grams, were divided into 6 groups (n=6 for each group). The hindlimb ischemia model was created by permanent ligation of the right femoral artery. The ligation was close to the iliac arteries at the proximal end and to the bifurcation to the saphenous and popliteal arteries at the distal end. Then, the femoral artery was cut between the 2 ligation ends. apop buffer, 106 MSCs, or graded doses of MVs in a total volume of 200 μL were intramuscularly injected 24 h after ligation of the vessels with 5 separate injections into femoral biceps, semitendinous, semimembranous, and adductor muscles.
Laser Doppler blood flow detection
Laser Doppler perfusion imaging was performed in anesthetized rats that had been settled on a heating plate with PeriscanPIM (Perimed), and the blood flow perfusion of region of interest was analyzed with LDPIwin2.3 software. The ratio of right (ischemic) to left (nonischemic) leg was calculated as previously described [33]. The rats that had received the artery ligation without any treatment for 2 days were served as test controls.
Histological examination
After laser Doppler blood flow (LDBF) detection, the adductor muscles from some rats were removed, fixed in 10% formalin, embedded in paraffin, cut into sections of 5 μm in thickness, and stained with hematoxylin and eosin. Histological examination was performed by a pathologist who was blind to the test groups.
Statistical analysis
All values are expressed as the mean±SEM. Analysis of data was completed by analysis of variance with post hoc Fisher's least significant difference to adjust for multiple group comparisons. All statistical analysis was completed using the Statistical Package for the Social Sciences (SPSS). A P-value<0.05 was considered as significant.
Results
UC-MSCs released MVs after hypoxic stimulation
To observe if UC-MSCs could release MVs under hypoxia, the status of UC-MSCs after stimulation was first evaluated by Annexin-V/PI staining. UC-MSCs were cultured under different conditions, and all the cells in culture, including those in the supernatants and those attached relatively loosely and tightly to the plastic, were collected for flow cytometric analysis. The results showed that compared to the cells maintained in a medium containing 10% FBS in normoxia, hypoxia alone did not induce MSC apoptosis, while cells cultured in a serum-free medium under hypoxia had a tendency to adopt the apoptotic pathway (Fig. 1A).
FIG. 1.
Umbilical cord mesenchymal stem cells (UC-MSCs) shed microvesicles (MVs). (A) Representative flow cytometry results show that cells in serum-free medium (SF) under hypoxia (Hypo) were inclined to apoptosis compared with the cells normoxia (Norm). X-axis represents Annexin-V-fluorescent isothiocyanate, and Y-axis indicates propidium iodide (PI). (B) Morphological features of MVs under electron microscope; bar: 100 nm. (C) Identification of MSC-MVs by flow cytometry. Upper panel: washed beads (left) or MV-coated beads after reaction with isotype antibodies (right) were collected, and the single beads (green) and doublets (pink red) were gated for fluorescence analysis. MVs: MSC-MVs, MSCs: the parent MSCs. X-axis: forward scatter corner (FSC). Y-axis: phycoerythrin-conjugated antibodies against the antigens of choice. The positive and negative proportions are indicated. The results are representative of 2 individual experiments. Color images available online at www.liebertpub.com/scd
Subsequently, MSC-MVs were collected by ultracentrifugation, and the total protein contents were assessed. Three individual experiments showed that the total protein content was 402.48±178.12 μg/108 MSCs in normoxia, significantly lower than that of cells in hypoxia (2270.26±574.14 μg/108, P=0.01349) and that of cells upon serum-free stimulation alone (1715.616±516.12 μg/108 cells, P=0.00106). The highest content was collected from MSCs under a serum-free and hypoxic condition (3036.73±375.45 μg/108). It seemed that hypoxia or serum-free culture stimulated MSCs to release MVs.
Under an electron microscope, MSC-MVs took a round morphology with a transparent center, and their diameters were around 80–150 nm, all of which were typical for MVs described elsewhere [28,29] (Fig. 1B). Bead-based flow cytometric analysis showed that MSC-MVs were highly positive for CD29, CD44, and CD73, partially positive for the beta-chain of the receptor for platelet-derived growth factor (PDGFR), and negative for CD31 and CD45. The results here suggested that the MVs were originated from MSCs (Fig. 1C). Interestingly, though the majority of the parent cells were positive for PDGFR-beta, only a small part of MSC-MVs were positive for this receptor.
MSC-MVs could be internalized by UC-ECs
To investigate the possible functions of MSC-MVs, DiI-labeled MVs were added into the UC-EC culture to observe whether these vesicles could be internalized into UC-ECs. MSCs were firstly labeled with DiI dye (Fig. 2A), and MVs were harvested from the labeled MSCs. The fluorescence of UC-ECs in normoxia (Fig. 2B, D) or hypoxia (Fig. 2C, E) was observed at different time points after the addition of DiI-MVs, using a fluorescent microscope and flow cytometry. The results showed that DiI-MVs could be easily internalized into ECs under hypoxic or normoxic conditions, and nearly all the cells became DiI positive after coincubation of 8 h (Fig. 2D, E). Further, it seemed that the internalization processes in hypoxia and normoxia were not obviously different.
FIG. 2.
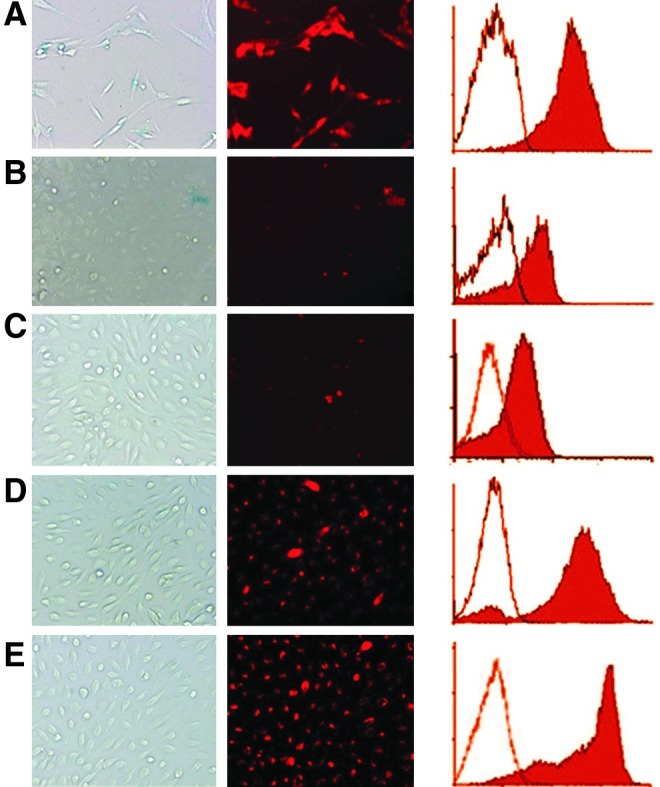
The fluorescence of DiI-labeled MSCs and MSC-MV-treated endothelial cells (ECs). MSCs were labeled with DiI dye (A) and umbilical cord ECs (UC-ECs) were cultured in normoxia (B, D) or hypoxia (C, E) in the presence of DiI-MVs for 2 h (B, C) and 8 h (D, E). The photographs were taken under an ordinary light (left column) or a fluorescent light (middle column). The typical histograms from flow cytometric tests are also indicated (right column). In the histograms, X-axis indicates the relative fluorescence of DiI, and Y-axis represents number of events; the hollow histograms represent the controls, and the solid ones indicate the tested cells. The results are representative of 2 separate experiments. Color images available online at www.liebertpub.com/scd
MSC-MVs promoted the growth of ECs
To observe whether MSC-MVs could stimulate UC-EC growth, CFSE dye dilution assay was used to evaluate how many cells had experienced division, and how many cell cycles they had experienced, after the cells were cultured for 72 h in the presence of graded concentrations of MSC-MVs. Using the proliferation wizard basic model of ModFit software, CFSE-labeled cell populations were distributed into 10 generations of cell division. The higher the generation of a cell, the lower the CFSE fluorescence intensity was (i.e., a shift to the left for the corresponding fluorescent histogram peak). The amplitude or height of each histogram represents the proportion (%) of cells in a given generation. Analysis showed that though UC-ECs from 3 subjects grew at different rates, the results definitely supported the proliferation-promoting effect of MSC-MVs on ECs in a dose-dependent manner (Fig. 3A). Chi-square analysis showed that 1 μg/mL of MSC-MVs was able to stimulate the proliferation of ECs from samples 1 and 2, and MSC-MVs at a concentration of 10 μg/mL promoted the growth of ECs at a comparable rate of cells cultured in the presence of 10 ng/mL of bFGF. Further, the proportions of 50 μg/mL group at generation 4 were significantly greater than those of bFGF and 5% FBS groups (P<0.001 for each sample). The percentages of 100 μg/mL group at generation 4 were higher than those of 10% FBS group (P<0.001 for each sample).
FIG. 3.
MSC-MVs promoted the proliferation of UC-ECs. (A) Carboxyfluorescein succinimidyl ester (CFSE)-labeled human UC-ECs from 3 samples were cultured in the presence of MSC-MVs at graded concentrations for 72 h. The proportions of the cells that had experienced different divisions were analyzed by flow cytometry technique. Cells that were maintained in 1% fetal bovine serum (FBS) were served as sham controls (ctr) and those in the presence of 5% FBS and 10% FBS were served as positive controls. Three main portions are indicated, including histograms with the colors of pink (generation 2), white blue (generation 3), and yellow (generation 4). X-axis represents CFSE intensity, and Y-axis represents the number of the events. (B) MTT assay shows the stimulation effect of MSC-MVs on the proliferation of UC-ECs. Y-axis: optical density at a wavelength of 490 nm. The results here were from ECs of 2 subjects. Color images available online at www.liebertpub.com/scd
To confirm the results above, UC-ECs were cultured in the presence of graded doses of MSC-MVs, and MTT assay was performed at time points of 24, 48, and 72 h. Consistently, the results showed that compared with control cells, ECs grew significantly faster in the presence of MSC-MVs at a concentration of 1 μg/mL (P<0.05 at all the observed time points). After culturing for 72 h, the highest OD490 value was from wells in the presence of MSC-MVs of 100 μg/mL, followed sequentially by 50 μg/mL, 10% FBS, 10 ng/mL bFGF, 5% FBS, MSC-MVs of 10 μg/mL, 1 μg/mL, and medium control. The data here support the conclusion that MSC-MVs stimulate the proliferation of UC-ECs in a dose-dependent manner (Fig. 3B).
MSC-MVs promoted in vitro tube formation of ECs
Tube formation of UC-ECs in Matrigel was observed to evaluate if MSC-MVs promote in vitro angiogenesis under hypoxia and normoxia. Microscopic observation found that tube-like formation of ECs occurred around 6 h of culture, and the number of tube-like structure reached to its maximal at about 16 h. Therefore, the network structure was counted at the time point of 24 h. The results showed that MSC-MVs stimulated tubulogenesis of UC-ECs in hypoxia and normoxia in a dose-dependent manner (Fig. 4A, B). As shown in Fig. 4C, further statistical analysis indicated that 10 μg/mL of MSC-MVs could greatly increase the structure number compared with DMEM alone (P<0.01). Further, those of 100 μg/mL of MVs and bFGF groups were comparable (P>0.05). It was also found that in the presence of MSC-MVs (10 μg/mL), the number of cells in hypoxia was significantly higher than that in normoxia (P=0.008). The results suggested that MSC-MVs promote angiogenesis in vitro, and that this activity seems greater in hypoxia.
FIG. 4.
MSC-MVs promoted tube formation of UC-ECs in a dose-dependent manner. ECs were incubated in Matrigel in the presence of graded doses of MSC-MVs in hypoxia or normoxia. Typical morphology of the tube-like structure is shown in rows (A) (hypoxia) and (B) (normoxia). (C) Y-axis represents the structure number per field, and X-axis indicates different groups. The results are representative of those from 2 individual experiments.
MSC-MVs promoted new vessel formation in a limb ischemic model
To further determine the angiogenic activity of MSC-MVs, a rat hindlimb ischemia model was developed, and MSC-MVs, MSCs, or apop buffer was injected intramuscularly into the ischemic zones. Two weeks after treatment, the LDBF technique was used to evaluate the recovery of the blood flow. The results showed that though partial recovery of the blood flow was observed in sham controls, HU-MSCs or MSC-MVs dramatically improved this process (Fig. 5). To alleviate the errors made by the detection procedure, the ratio of mean blood perfusion in region of interest from the ischemic to that from the nonischemic hindlimb of the same rat was calculated. The results showed that the mean ratio of 10 μg MV group (0.51±0.05) was not significantly different from that of sham control (0.50±0.06, P=0.5390), while that of 50 μg MV rats was significantly increased (0.74±0.06, P<0.0001). The ratio of 100 μg rats (0.92±0.06) was comparable to that of the MSC group (0.95±0.08, P=0.5781). Of note, blood flow recovered to the normal range after 3 weeks in all the tested rats. However, it was found that blood flow recovery was more rapid in MSC- or MSC-MV-treated rats, indicating the angiogenesis-promoting activity of MSCs and MSC-MVs.
FIG. 5.

Representative Laser Doppler images of the rat hindlimbs. Laser Doppler blood flow was performed 2 days after the artery ligation (ctr) without any treatment or 2 weeks after operation (groups as indicated). The colors that correspond to the blood perfusion are shown. Color images available online at www.liebertpub.com/scd
Two weeks after operation, the adductor muscles of the rats were removed and histologically examined. Generally, 2 kinds of typical histological changes could be observed, including complete muscle fiber bunches and inflammatory granulomatosis that occurred due to tissue necrosis. Histological examination showed that small blood vessel tissue necrosis could be readily perceived in the granulomatosis area, and the capillary network could be observed between the muscle fiber bunches. Infiltration of inflammatory cells and necrotic tissues were found in the control rats (Fig. 6A). In the MV- and MSC-treated muscles, the capillary network seemed to be found more easily (Fig. 6B).
FIG. 6.
Typical histological changes of the adductor muscles. (A) Inflammatory granulomatosis; (B) muscle fiber bunches. Arrowhead: small vessels; Arrow: capillaries. Bar: 25 μm. Color images available online at www.liebertpub.com/scd
Discussion
MVs were firstly described as exosomes derived from reticulocytes for eliminating antiquated molecules such as transferrin receptors [34]. Recently, it is recognized that MVs could be shed by all cells in resting and activated conditions [26]. Human bone marrow MSCs have also the ability to produce MVs in the resting state [35]. In the present study, it was proved that human UC-MSCs could discharge MVs either, especially upon hypoxic stimulation. Membrane dye-labeling test showed that the MVs could be incorporated into human UC-ECs, as those into tubular epithelial cells reported previously [35]. The incorporation was rapid, as nearly all ECs became positive for DiI after exposure to MSC-MVs for 8 h. Previous data showed that internalization of MVs derived from human bone marrow MSCs into murine epithelial cells peaked between 12 and 24 h [35], seemingly slower than the observation in this study. The mechanisms underlying internalization remain unclear and should vary depending on the diverse original cell types or even MVs released from the same cell type under different conditions. A recent report suggests that cytoskeleton proteins such as ADP-ribosylation factor 6 on the surface of MVs might contribute to this process [36].
In agreement with a previous report on platelet MVs [32], our results also indicated that like MSCs, MVs from human UC-MSCs promoted the recovery of blood flow in an ischemic model of limbs. The proangiogenic activity of MVs derived from brain tumor cells was also reported [37], and MSC-MVs were found to protect the kidneys in a rat ischemia–reperfusion model [25]. Previous data have also demonstrated that MVs derived from other cell types, including platelets [32], endothelial progenitors [38–41], tumor cells [37,42], and ES-derived MSCs [43], have the proangiogenic activity both in vitro and in vivo.
It is generally accepted that transplanted MSCs promote angiogenesis mainly by the paracrine mechanism [44]. However, it remains largely unknown about which of the compositions released by MSCs contribute to the angiogenesis-promoting activity. Previous data have suggested that MV proteins such as sonic hedgehog [45] and PDGFR [46] might contribute to the proangiogenic activity. However, our results indicated that MSC-MVs were only partially positive for this receptor, appealing further investigations to clarify the complexity. Also, several reports indicate that the horizontal transfer of mRNA and micro-RNAs from MVs to the target cells is the mechanism of action [38–41,43,35,47]. Recent studies point out that miR-126, miR-296, and let-7 micro-RNA family might play a critical role in the angiogenesis-promoting effect of MVs [40,43]. Therefore, it is possibly reasonable to postulate that transfer of mRNA, micro-RNA, and proteins as well may occur, and this process might be attributable to the intricate biological activities of MVs [48].
The findings here that MSC-MVs enhanced the proliferation of ECs might be one of the critical processes in the new vessel formation, as collateral arteries grow from pre-existing anastomoses (arteriogenesis), is the main pathway in the reparative process of arterial occlusion [49]. Nevertheless, the detailed principles on the functionality of MSC-MVs need further investigations to be clarified, and the composition should be analyzed in the coming experiments to find out if the similar mechanisms exist in MSC-MVs as those discovered in the studies with MVs from other cell types.
Further, it still remains unknown about the difference of MVs derived from hypoxia-stimulated MSCs and the resting MSCs. Cells release MVs upon activation and during cell growth. In hypoxia, MSCs proliferate faster than in those in normoxia [50–52]. UC-MSCs were induced to express hypoxia-inducible factors (HIFs) and energy metabolism-associated molecules, including glucose transporter 1, lactate dehydrogenase, and pyruvate dehydrogenase kinase isozyme 1 upon hypoxia stimulation [51,52]. The induction of HIF expression in MSCs might enhance their proangiogenic activity by secreting vascular endothelial growth factor (VEGF) and erythropoietin [53,54], the 2 main HIFs that may involve the neoangiogenesis. Previous data have demonstrated that cancer and ECs in hypoxia can secrete MVs containing angiogenic molecules, including interleukin (IL)-8, VEGF, leukemia inhibitory factor, oncostain M IL-11, matrix metalloproteinase-9 [55], heparin-binding EGF-like growth factor [37], and vascular cell adhesion molecule [56]. Further, recent studies have proved that MVs derived from the injured lungs could induce hematopoietic cells to express pulmonary epithelial cell-specific genes [57,58], and MV sources are the important determinants in the cell fate modulation [59]. Detailed investigations are needed to understand if it is the case for MSC-MVs.
Acknowledgments
The authors are very grateful for the technical assistance of Dr. Shui-Ming Wang from the Department of Pathology, Beijing Institute of Radiation Medicine. They also appreciate the critical manuscript reviewing from Dr. Pei Hsien Tang. This work was partially supported by a grant from the Science and Technology Bureau, Guangzhou Developmental District (No.2009Q-P081, to HS), the National Natural Sciences grants (No. 30871018 and No. 30971068, to GZK), and the National “863” grants (No. 2011AA020101 and No. 2012AA02A, to GZK).
Author Disclosure Statement
All the authors disclose that no competing financial interests exist.
References
- 1.Pittenger MF. Mackay AM. Beck SC. Jaiswal RK. Douglas R. Mosca JD. Moorman MA. Simonetti DW. Craig S. Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 2.Dominici M. Le Blanc K. Mueller I. Slaper-Cortenbach I. Marini F. Krause D. Deans R. Keating A. Prockop D. Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 3.Caplan AI. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol. 2007;213:341–347. doi: 10.1002/jcp.21200. [DOI] [PubMed] [Google Scholar]
- 4.Short BJ. Brouard N. Simmons PJ. Prospective isolation of mesenchymal stem cells from mouse compact bone. Methods Mol Biol. 2009;482:259–268. doi: 10.1007/978-1-59745-060-7_16. [DOI] [PubMed] [Google Scholar]
- 5.Meirelles LS. Chagastelles PC. Nardi NB. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J Cell Sci. 2006;119:2204–2213. doi: 10.1242/jcs.02932. [DOI] [PubMed] [Google Scholar]
- 6.Horwitz EM. Prockop DJ. Fitzpatrick LA. Koo WW. Gordon PL. Neel M. Sussman M. Orchard P. Marx JC. Pyeritz RE. Brenner MK. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfect. Nat Med. 1999;5:309–313. doi: 10.1038/6529. [DOI] [PubMed] [Google Scholar]
- 7.Jones CP. Rankin SM. Bone marrow-derived stem cells and respiratory disease. Chest. 2011;140:205–211. doi: 10.1378/chest.10-2348. [DOI] [PubMed] [Google Scholar]
- 8.Cristofanilli M. Harris VK. Zigelbaum A. Goossens AM. Lu A. Rosenthal H. Sadiq SA. Mesenchymal stem cells enhance the engraftment and myelinating ability of allogeneic oligodendrocyte progenitors in dysmyelinated mice. Stem Cells Dev. 2011;20:2065–2076. doi: 10.1089/scd.2010.0547. [DOI] [PubMed] [Google Scholar]
- 9.Martell K. Trounson A. Baum E. Stem cell therapies in clinical trials: workshop on best practices and the need for harmonization. Cell Stem Cell. 2010;7:451–454. doi: 10.1016/j.stem.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 10.Barbash IM. Chouraqui P. Baron J. Feinberg MS. Etzion S. Tessone A. Miller L. Guetta E. Zipori D et al. Systemic delivery of bone marrow–derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution. Circulation. 2003;108:863–868. doi: 10.1161/01.CIR.0000084828.50310.6A. [DOI] [PubMed] [Google Scholar]
- 11.Devine SM. Cobbs C. Jennings M. Bartholomew A. Hoffman R. Mesenchymal stem cells distribute to a wide range of tissues following systemic infusion into nonhuman primates. Blood. 2003;101:2999–3001. doi: 10.1182/blood-2002-06-1830. [DOI] [PubMed] [Google Scholar]
- 12.Bentzon JF. Stenderup K. Hansen FD. Schroder HD. Abdallah BM. Jensen TG. Kassem M. Tissue distribution and engraftment of human mesenchymal stem cells immortalized by human telomerase reverse transcriptase gene. Biochem Biophys Res Commun. 2005;330:633–640. doi: 10.1016/j.bbrc.2005.03.072. [DOI] [PubMed] [Google Scholar]
- 13.Joo SY. Cho KA. Jung YJ. Kim HS. Park SY. Choi YB. Hong KM. Woo SY. Seoh JY. Ryu KH. Bioimaging for the monitoring of the in vivo distribution of infused mesenchymal stem cells in a mouse model of the graft-versus-host reaction. Cell Biol Int. 2011;35:417–421. doi: 10.1042/CBI20100563. [DOI] [PubMed] [Google Scholar]
- 14.Mangi AA. Noiseux N. Kong D. He H. Rezvani M. Ingwall JS. Dzau VJ. Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts. Nat Med. 2003;9:1195–1201. doi: 10.1038/nm912. [DOI] [PubMed] [Google Scholar]
- 15.Winkler T. von Roth P. Schuman MR. Sieland K. Stoltenburg-Didinger G. Taupitz M. Perka C. Duda GN. Matziolis G. In vivo visualization of locally transplanted mesenchymal stem cells in the severely injured muscle in rats. Tissue Eng Part A. 2008;14:1149–1160. doi: 10.1089/ten.tea.2007.0179. [DOI] [PubMed] [Google Scholar]
- 16.Tolar J. Blanc Le K. Keating A. Blazar BR. Concise review: hitting the right spot with mesenchymal stromal cells. Stem Cells. 2010;28:1446–1455. doi: 10.1002/stem.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lee JW. Fang X. Krasnodembskaya A. Howard JP. Matthay MA. Concise review: mesenchymal stem cells for acute lung injury: role of paracrine soluble factors. Stem Cells. 2011;29:913–919. doi: 10.1002/stem.643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rose RA. Jiang H. Wang X. Helke S. Tsoporis JN. Gong N. Keating SC. Parker TG. Backx PH. Keating A. Bone marrow-derived mesenchymal stromal cells express cardiac-specific markers, retain the stromal phenotype, and do not become functional cardiomyocytes in vitro. Stem Cells. 2008;26:2884–2892. doi: 10.1634/stemcells.2008-0329. [DOI] [PubMed] [Google Scholar]
- 19.Danial NN. Korsmeyer SJ. Cell death: critical control points. Cell. 2004;116:205–219. doi: 10.1016/s0092-8674(04)00046-7. [DOI] [PubMed] [Google Scholar]
- 20.Lo EH. A new penumbra: transitioning from injury into repair after stroke. Nat Med. 2008;14:497–500. doi: 10.1038/nm1735. [DOI] [PubMed] [Google Scholar]
- 21.Quesenberry PJ. Dooner MS. Aliotta JM. Stem cell plasticity revisited: the continuum marrow model and phenotypic changes mediated by microvesicles. Exp Hematol. 2010;38:581–592. doi: 10.1016/j.exphem.2010.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Camussi G. Deregibus MC. Bruno S. Cantaluppi V. Biancone L. Exosomes/microvesicles as a mechanism of cell-to-cell communication. Kidney Int. 2010;78:838–848. doi: 10.1038/ki.2010.278. [DOI] [PubMed] [Google Scholar]
- 23.Camussi G. Deregibus MC. Tetta C. Paracrine/endocrine mechanism of stem cells on kidney repair: role of microvesicle-mediated transfer of genetic information. Curr Opin Nephrol Hypertens. 2010;19:7–12. doi: 10.1097/MNH.0b013e328332fb6f. [DOI] [PubMed] [Google Scholar]
- 24.Bruno S. Grange C. Deregibus MC. Calogero RA. Saviozzi S. Collino F. Morando L. Busca A. Falda M et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol. 2009;20:1053–1067. doi: 10.1681/ASN.2008070798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gatti S. Bruno S. Deregibus MC. Sordi A. Cantaluppi V. Tetta C. Camussi G. Microvesicles derived from human adult mesenchymal stem cells protect against ischaemia-reperfusion-induced acute and chronic kidney injury. Nephrol Dial Transplant. 2011;26:1474–1483. doi: 10.1093/ndt/gfr015. [DOI] [PubMed] [Google Scholar]
- 26.Tetta C. Bruno S. Fonsato V. Deregibus MC. Camussi G. The role of microvesicles in tissue repair. Organogenesis. 2011;7:105–115. doi: 10.4161/org.7.2.15782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Han D. Liu J. Xue M. Zhu L. Yan H. Wang Z. Ding L. Guo Z. Wang H. Clinical analysis of the treatment of spinocerebellar ataxia and multiple system atrophy-cerebellar type with umbilical cord mesenchymal stromal cells. Cytotherapy. 2011;13:913–917. doi: 10.3109/14653249.2011.579958. [DOI] [PubMed] [Google Scholar]
- 28.Thery C. Clayton A. Amigorena S. Raposo G. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. 2006:3.22.1–3.22.29. doi: 10.1002/0471143030.cb0322s30. [DOI] [PubMed] [Google Scholar]
- 29.Distler JHW. Jungel A. Huber C. Seemayer CA. Reich CF., III Gay RE. Michel BA. Fontana A. Gay S. Pisetsky DS. Distler O. The induction of matrix metalloproteinase and cytokine expression in synovial fibroblasts stimulated with immune cell microparticles. Proc Natl Acad Sci. 2005;102:2892–2897. doi: 10.1073/pnas.0409781102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Matera G. Lupi M. Ubezio P. Heterogeneous cell response to topotecan in a CFSE-based proliferation test. Cytometry A. 2004;62:118–128. doi: 10.1002/cyto.a.20097. [DOI] [PubMed] [Google Scholar]
- 31.Combes V. Simon AC. Grau GE. Arnoux D. Camoin L. Sabatier F. Mutin M. Sanmarco M. Sampol J. Dignat-George F. In vitro generation of endothelial microparticles and possible prothrombotic activity in patients with lupus anticoagulant. J Clin Invest. 1999;104:93–102. doi: 10.1172/JCI4985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brilla A. Dashevskya O. Rivob J. Gozalb Y. Varon D. Platelet-derived microparticles induce angiogenesis and stimulate post-ischemic revascularization. Cardiovasc Res. 2005;67:30–38. doi: 10.1016/j.cardiores.2005.04.007. [DOI] [PubMed] [Google Scholar]
- 33.Aoki N. Yokoyama R. Asai N. Ohki M. Ohki Y. KUsubata K. Heissig B. Hattori K. Nakagawa Y. Matsuda T. Adipocyte-derived microvesicles are associated with multiple angiogenic factors and induce angiogenesis in vivo and in vitro. Endocrinology. 2010;151:2567–2576. doi: 10.1210/en.2009-1023. [DOI] [PubMed] [Google Scholar]
- 34.Harding C. Heuser J. Stahi P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97:329–339. doi: 10.1083/jcb.97.2.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Collino F. Deregibus MC. Bruno S. Sterpone L. Aghemo G. Viltono L. Tetta C. Camussi G. Microvesicles derived from adult human bone marrow and tissue specific mesenchymal stem cells shuttle selected pattern of miRNAs. PLoS ONE. 2010;5:e11803. doi: 10.1371/journal.pone.0011803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Afroze SH. Uddin MN. Cao X. Asea A. Gizachew D. Internalization of exogenous ADP-ribosylation factor 6 (Arf6) proteins into cells. Mol Cell Biochem. 2011;354:291–299. doi: 10.1007/s11010-011-0829-y. [DOI] [PubMed] [Google Scholar]
- 37.Svensson KJ. Kucharzewska P. Christianson HC. Sköld S. Löfstedt T. Johansson MC. Mörgelin M. Bengzon J. Ruf W. Belting M. Hypoxia triggers a proangiogenic pathway involving cancer cell microvesicles and PAR-2-mediated heparin-binding EGF signaling in endothelial cells. Proc Natl Acad Sci U S A. 2011;108:13147–13152. doi: 10.1073/pnas.1104261108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Deregibus MC. Cantaluppi V. Calogero R. Lo Iacono M. Tetta C. Biancone L. Bruno S. Bussolati B. Camussi G. Endothelial progenitor cell derived microvesicles activate an angiogenic program in endothelial cells by a horizontal transfer of mRNA. Blood. 2007;110:2440–2448. doi: 10.1182/blood-2007-03-078709. [DOI] [PubMed] [Google Scholar]
- 39.Biancone L. Tetta C. Camussi G. Microvesicles derived from endothelial progenitor cells protect the kidney from ischemia-reperfusion injury by microRNA-dependent reprogramming of resident renal cells. Kidney Int. 2012;82:412–427. doi: 10.1038/ki.2012.105. [DOI] [PubMed] [Google Scholar]
- 40.Cantaluppi V. Biancone L. Figliolini F. Beltramo S. Medica D. Deregibus MC. Galimi F. Romagnoli R. Salizzoni M et al. Microvesicles derived from endothelial progenitor cells enhance neoangiogenesis of human pancreatic islets. Cell Transplant. 2012 doi: 10.3727/096368911×627534. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 41.Ranghino A. Cantaluppi V. Grange C. Vitillo L. Fop F. Biancone L. Deregibus MC. Tetta C. Segoloni GP. Camussi G. Endothelial progenitor cell-derived microvesicles improve neovascularization in a murine model of hindlimb ischemia. Int J Immunopathol Pharmacol. 2012;25:75–85. doi: 10.1177/039463201202500110. [DOI] [PubMed] [Google Scholar]
- 42.Grange C. Tapparo M. Collino F. Vitillo L. Damasco C. Deregibus MC. Tetta C. Bussolati B. Camussi G. Microvesicles released from human renal cancer stem cells stimulate angiogenesis and formation of lung premetastatic niche. Cancer Res. 2011;71:5346–5356. doi: 10.1158/0008-5472.CAN-11-0241. [DOI] [PubMed] [Google Scholar]
- 43.Koh W. Sheng CT. Tan B. Lee QY. Kuznetsov V. Kiang LS. Tanavde V. Analysis of deep sequencing microRNA expression profile from human embryonic stem cells derived mesenchymal stem cells reveals possible role of let-7 microRNA family in downstream targeting of hepatic nuclear factor 4 alpha. BMC Genomics. 2010;11(Suppl. 1):S6. doi: 10.1186/1471-2164-11-S1-S6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kinnaird T. Stabile E. Burnett MS. Lee CW. Barr S. Fuchs S. Epstein SE. Marrow-derived stromal cells express genes encoding a broad spectrum of arteriogenic cytokines and promote in vitro and in vivo arteriogenesis through paracrine mechanisms. Cir Res. 2004;94:678–685. doi: 10.1161/01.RES.0000118601.37875.AC. [DOI] [PubMed] [Google Scholar]
- 45.Benameur T. Soleti R. Porro C. Andriantsitohaina R. Martınez MC. Microparticles carrying sonic hedgehog favor neovascularization through the activation of nitric oxide pathway in mice. PLoS one. 2010;5:e12688. doi: 10.1371/journal.pone.0012688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kim HS. Choi DY. Yun SJ. Choi SM. Kang JW. Jung JW. Hwang D. Kim KP. Kim DW. Proteomic analysis of microvesicles derived from human mesenchymal stem cells. J Proteome Res. 2012;11:839–849. doi: 10.1021/pr200682z. [DOI] [PubMed] [Google Scholar]
- 47.Chen TS. Lai RC. Lee MM. Choo ABH. Lee CN. Lim SK. Mesenchymal stem cell secretes microparticles enriched in pre-microRNAs. Nucleic Acids Res. 2010;38:215–224. doi: 10.1093/nar/gkp857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ratajczak J. Miekus K. Kucia M. Zhang J. Reca R. Dvorak P. Ratajczak MZ. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia. 2006;20:847–856. doi: 10.1038/sj.leu.2404132. [DOI] [PubMed] [Google Scholar]
- 49.Herzog S. Sager H. Khmelevski E. Deylig A. Ito WD. Collateral arteries grow from preexisting anastomoses in the rat hindlimb. Am J Physiol Heart Circ Physiol. 2002;283:H2012–H2020. doi: 10.1152/ajpheart.00257.2002. [DOI] [PubMed] [Google Scholar]
- 50.Hung SP. Ho JH. Shih YR. Lo T. Lee OK. Hypoxia promotes proliferation and osteogenic differentiation potentials of human mesenchymal stem cells. J Orthop Res. 2012;30:260–266. doi: 10.1002/jor.21517. [DOI] [PubMed] [Google Scholar]
- 51.Nekanti U. Dastidar S. Venugopal P. Totey S. Ta M. Increased proliferation and analysis of differential gene expression in human Wharton's jelly-derived mesenchymal stromal cells under hypoxia. Int J Biol Sci. 2010;6:499–512. doi: 10.7150/ijbs.6.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lavrentieva A. Majore I. Kasper C. Hass R. Effects of hypoxic culture conditions on umbilical cord-derived human mesenchymal stem cells. Cell Commun Signal. 2010;89:18–27. doi: 10.1186/1478-811X-8-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang M. Tan J. Coffey A. Fehrenbacher J. Weil BR. Meldrum DR. Signal transducer and activator of transcription 3-stimulated hypoxia inducible factor-1alpha mediates estrogen receptor-alpha-induced mesenchymal stem cell vascular endothelial growth factor production. J Thorac Cardiovasc Surg. 2009;138:163–171. doi: 10.1016/j.jtcvs.2009.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zwezdaryk KJ. Coffelt SB. Figueroa YG. Liu J. Phinney DG. LaMarca HL. Florez L. Morris CB. Hoyle GW. Scandurro AB. Erythropoietin, a hypoxia-regulated factor, elicits a pro-angiogenic program in human mesenchymal stem cells. Exp Hematol. 2007;35:640–652. doi: 10.1016/j.exphem.2007.01.044. [DOI] [PubMed] [Google Scholar]
- 55.Wysoczynski M. Ratajczak MZ. Lung cancer secreted microvesicles: underappreciated modulators of microenvironment in expanding tumors. Int J Cancer. 2009;125:1595–603. doi: 10.1002/ijc.24479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vince RV. Chrismas B. Midgley AW. McNaughton LR. Madden LA. Hypoxia mediated release of endothelial microparticles and increased association of S100A12 with circulating neutrophils. Oxid Med Cell Longev. 2009;2:2–6. doi: 10.4161/oxim.2.1.7611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Aliotta JM. Sanchez-Guijo FM. Dooner GJ. Johnson KW. Dooner MS. Greer KA. Greer D. Pimentel J. Kolankiewicz LM et al. Alteration of marrow gene expression, protein production and engraftment into lung by lung-derived microvesicles. Stem Cells. 2007;9:2245–2256. doi: 10.1634/stemcells.2007-0128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Aliotta JM. Pereira M. Johnson KW. de Paz N. Dooner MS. Puente N. Ayala C. Brilliant K. Berz D et al. Microvesicle entry into marrow cells mediates tissue-specific changes in mRNA by direct delivery of mRNA and induction of transcription. Exp Hematol. 2010;38:233–245. doi: 10.1016/j.exphem.2010.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Aliotta JM. Lee D. Puente N. Faradyan S. Sears EH. Amaral A. Goldberg L. Dooner MS. Pereira M. Quesenberry PJ. Progenitor/stem cells fater determination: interactive dynamics of cell cycle and microsvisicle. Stem Cell Dev. 2012;21:1627–1638. doi: 10.1089/scd.2011.0550. [DOI] [PMC free article] [PubMed] [Google Scholar]






