Abstract
T cell function is dependent on store-operated Ca2+ influx that is activated by the stromal interaction molecules (STIM) 1 and 2. We show that mice with T-cell specific deletion of STIM1 or STIM2 are protected from experimental autoimmune encephalomyelitis (EAE), a mouse model of multiple sclerosis. While STIM1- and STIM2-deficient T cells could be successfully primed by autoantigen, they failed to produce the proinflammatory cytokines IL-17 and IFN-γ. STIM1-deficient T cells showed reduced expression of IL- 23R, required for Th17 cell homeostasis, and had impaired chemokine dependent T cell migration caused by a lack of chemokine-induced Ca2+ influx. Autoantigen-specific STIM1- or STIM2-deficient T cells failed to expand and accumulate in the CNS and lymph nodes following adoptive transfer to passively induce EAE, suggesting that autoantigen-specific restimulation or homeostasis of STIM1- and STIM2-deficient T cells are impaired. Combined deletion of both STIM1 and STIM2, previously shown to impair Treg cell development and function, completely protected mice from EAE. This indicates that, in the absence of Ca2+ influx, autoreactive T cells are severely dysfunctional rendering Treg dispensable for the prevention of CNS inflammation. Our findings demonstrate that both STIM1 and STIM2 are critical for T cell function and autoimmunity in vivo.
Keywords: EAE, MS, Th17, STIM1, STIM2, calcium, CRAC, Ca2+, T cells, autoimmunity
Introduction
Multiple sclerosis (MS) is an autoimmune disease characterized by focal demyelination of the central nervous system (CNS). MS plaques are characterized by inflammatory infiltrates of autoreactive T cells and macrophages. The pathophysiology of disease has been studied extensively in experimental autoimmune encephalomyelitis (EAE), an animal model of MS, which is induced by immunization of mice or rats with myelin self antigens such as myelin oligodendrocyte glycoprotein (MOG) or adoptive transfer of encephalitogenic T cells [1]. CD4+ Th1 cells secreting interferon (IFN)-γ were long considered to be the predominant disease inducing T cell subset in EAE. This view has been challenged by the finding that Ifng-deficient mice showed more severe EAE than wild-type littermates [2–4]. More recently, Th17 cells have emerged as an important encephalitogenic T cell subset causing CNS inflammation based on evidence from animal models and patients with MS [5–8].
Th17 cells produce the pro-inflammatory cytokines IL-17A, IL-17F and IL-22 [9] and mice lacking IL-17 expression were partially resistant to the induction of EAE [10]. IL-17 levels were increased in cells isolated from MS lesions [11] and in the cerebrospinal fluid of MS patients [12, 13]. IL-17 secreted by Th17 cells induces the production of proinflammatory cytokines and chemokines by a number of cells such as fibroblasts, endothelial cells, epithelial cells and macrophages [7] resulting in the recruitment of, for instance, neutrophils and monocytes [14] and disruption of the blood brain barrier [13]. Differentiation of Th17 cells from naïve CD4+ T cells depends on a number of molecules including the transcription factor RORγt which functions as the master regulator of Th17 differentiation. Mucosal T cells from RORγt-deficient mice failed to produce IL-17 and to induce colitis indicating that RORγt is critical for the differentiation of autoreactive Th17 cells [15, 16]. In addition, IL-6 induces the production of IL-21 and thereby expression of IL-23 receptor. The interaction of IL-23 with IL-23R is thought to be important for terminal Th17 differentiation and homeostasis [5, 17, 18].
Activation of T cells requires Ca2+ influx, which results in expression of many cytokine and chemokine genes [19]. Ca2+ influx in T cells is mediated by specialized Ca2+ channels in the plasma membrane, so-called Ca2+-release-activated-Ca2+ (CRAC) channels, which are activated by depletion of ER Ca2+ stores following antigen binding to the TCR in a process termed store-operated Ca2+ entry (SOCE) [20]. ORAI1 is the ion channel pore forming subunit of the CRAC channel, which is activated by stromal interaction molecules (STIM) 1 and 2, single transmembrane proteins located in the endoplasmic reticulum (ER) membrane. Following depletion of ER calcium stores, Ca2+ dissociates from STIM1 and STIM2 resulting in a conformational change in the N termini of these proteins, their translocation to the plasma membrane and activation of ORAI1 CRAC channels [21]. ORAI1, STIM1 and STIM2 were shown to be critical for CRAC channel function and SOCE [22]. T cells of mice lacking Stim1 or Orai1 show severe defects in SOCE and production of IL-2, IL-4 and IFN-γ [23, 24] consistent with a similar lack of cytokine gene expression in immunodeficient patients with mutations in ORAI1 or STIM1 [22, 25, 26]. STIM2 is involved predominantly in maintaining basal cytoplasmic Ca2+ levels [27] and is necessary to sustain SOCE for several hours following TCR stimulation [24]. Accordingly, murine T cells lacking STIM2 show impaired cytokine gene expression [24] but the role of STIM2 in T cell function and immune responses in vivo has not been demonstrated yet. Combined deletion of both Stim1 and Stim2 results in impaired development and function of regulatory T cells and is associated with myelolymphoproliferative disease in mice [24].
In this study, we investigated whether STIM1 and STIM2 in T cells are required for induction of T cell mediated autoimmune disease. Mice with T cell specific deletion of Stim1 or both Stim1 and Stim2 were protected from induction of EAE, whereas lack of Stim2 significantly attenuated disease severity. Resistance to EAE in Stim1fl/fl Cd4-Cre and Stim2fl/fl Cd4-Cre mice was characterized by severely impaired effector T cell functions such as production of proinflammatory cytokines IL-17 and IFN-γ STIM1 and STIM2-deficient T cells failed to expand and to accumulate in the CNS and lymph nodes, a defect that is associated with impaired IL-23R expression on Th17-differentiated cells. STIM1 deficiency is associated with abolished chemokine dependent Ca2+ signaling and reduced chemotaxis of T cells. These studies demonstrate a crucial role for STIM1, STIM2 and SOCE in the function of autoreactive T cells.
Results
Mice with T cell specific deletion of Stim1, Stim2 or both Stim1 and Stim2 are resistant to induction of EAE
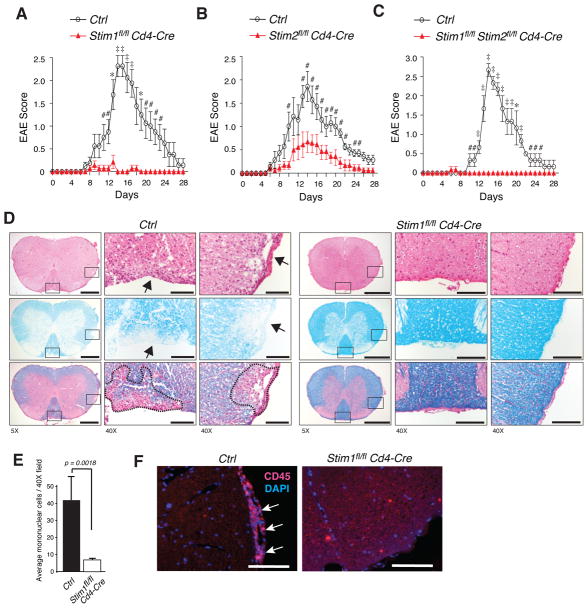
To understand whether SOCE is required for autoreactive T cell function and in the pathophysiology of autoimmune and inflammatory disease, we investigated the susceptibility of conditional knockout mice with T cell-specific deletion of Stim1, Stim2 or both Stim1 and Stim2 to develop experimental autoimmune encephalomyelitis (EAE). EAE was induced in Stim1fl/fl Cd4-Cre, Stim2fl/fl Cd4-Cre, Stim1fl/fl Stim2fl/fl Cd4-Cre and wild-type control mice by immunization with MOG35-55 peptide in complete Freund's adjuvant (CFA). Disease onset in wild-type mice occurred on average 9.2 (± 0.46) days after immunization and peaked around day 14, with a mean maximum disease score of 2.19 (Fig. 1A-C, Table 1). By contrast, mice with T cell specific deletion of STIM1 were almost completely resistant to EAE induction with a mean disease score of 0.13. Disease incidence in these mice was 16.7% and the highest EAE score observed in an individual Stim1fl/fl Cd4-Cre mouse was 1.0 (Fig. 1A, Table 1). Deletion of STIM2 in T cells resulted in attenuated severity of disease with a mean disease score of 0.75 and a disease incidence of 66.7% (Fig. 1B, Table 1), which is consistent with a defect in sustained Ca2+ responses in T cells lacking STIM2 [24]. The course of disease in terms of onset and duration, however, was not altered in STIM2-deficient mice compared to wild-type mice. Significantly decreased severity of EAE in Stim2fl/fl Cd4-Cre mice is in contrast to normal disease severity in Stim2−/− mice (despite a moderate delay in EAE onset) reported recently [28]. In summary, we find that abolishing either STIM1 or STIM2 expression in T cells is sufficient to protect mice from EAE, demonstrating for the first time an important role for STIM2 in T cell function in vivo.
Figure 1. Mice with T-cell specific deletion of STIM1, STIM2 or both STIM1 and STIM2 are resistant to EAE.
EAE was induced by immunization of (A) Stim1fl/fl Cd4-Cre (n=7), (B) Stim2fl/fl Cd4-Cre (n=9), (C) Stim1fl/flStim2fl/fl Cd4-Cre (n=6) and wild-type control (Stim1fl/fl or Stim2fl/fl, n=8 in A, n=7 in B, n=3 in C) mice with MOG35-55. Shown are mean±SEM EAE scores. #, p<0.05; *, p<0.005; ‡, p<0.001. The statistical significance of differences between Ctrl and Stim deficient mice was calculated using Student's t-test. D, H&E and luxol fast blue stains of spinal cord sections from Ctrl (Stim1fl/fl) and Stim1fl/fl Cd4-Cre mice 14 days after immunization with MOG35-55. The higher magnification panels (40X) correspond to the boxed regions in the low magnification panels (5X). Areas of demyelination are indicated by arrows and dashed lines. Scale bars, 500 μm (5x), 100 μm (40x). E, Mean±SEM mononuclear cell numbers from 3 spinal cord sections per mouse (3 mice per group) on day 14 after EAE induction. One 40X field per section was analyzed. F, CD45+ lymphocytes (red) and nuclei (blue) in the spinal cords of Ctrl and Stim1fl/fl Cd4-Cre mice on day 14 after EAE induction. Magnification 40X; scale bars, 100 μm.
Table 1.
Summary of EAE in STIM-deficient mice
| Genotype | Incidence | Average age of onset1 (±SEM) | Maximum EAE score (±SEM) |
|---|---|---|---|
| Ctrl (Stim1fl/fl or Stim2fl/fl) | 20/212 (95.2%) | 9.2 (±0.46) | 2.19 (±0.18) |
| Stim1fl/fl Cd4-Cre | 2/12 (16.7%) | 8.50 (±0.50) | 0.13 (±0.09) |
| Stim2 fl/fl Cd4-Cre | 6/9 (66.7%) | 9.17 (±1.25) | 0.75 (±0.27) |
| Stim1fl/fl Stim2fl/fl Cd4-Cre | 1/6 (16.7%) | 6 | 0.08 (±0.08) |
Days after MOG35-55 immunization
Numbers indicate affected mice / total number of mice tested; includes animals which have been sacrificed for isolation of cells or histological analysis at the peak of disease symptoms.
Notably, Stim1fl/fl Stim2fl/fl Cd4-Cre mice were as protected from EAE as STIM1-deficient mice with a disease incidence of 16.7% and a mean disease score of 0.08 (Fig. 1C, Table 1). We had shown previously that mice with T cell specific deletion of both STIM1 and STIM2 have severely reduced numbers and function of Treg and show a propensity to develop an autoinflammatory, myelolymphoproliferative phenotype [24]. The complete protection of these mice from EAE despite the paucity of functional Treg suggests that the lack of STIM1 and STIM2 impairs autoreactive effector T cell functions so severely that Treg become dispensable for controlling CNS inflammation. In the following, we therefore focused our studies on mice with T cell specific deletion of either STIM1 or STIM2 alone, deliberately excluding the effects of impaired Treg activity in Stim1fl/fl Stim2fl/fl Cd4-Cre mice. Taken together, we show here that both STIM1 and STIM2 are critical for autoreactive T cell function in vivo.
Stim1fl/fl Cd4-Cre mice lack signs of CNS inflammation and lymphocyte infiltration
Absence of clinical signs of EAE in Stim1fl/fl Cd4-Cre mice is consistent with the lack of detectable leukocyte infiltration in the CNS of these mice in contrast to extensive infiltration of CD45+ lymphocytes in the perivascular and submeningeal areas of the spinal cord of wild-type mice (Fig. 1D-F). In contrast to wild-type mice, areas of demyelination were not detected in MOG immunized Stim1fl/fl Cd4-Cre mice (Fig. 1D). These findings are consistent with a recently published report showing that Stim1−/− bone marrow chimeric mice are protected from EAE and clearly demonstrate that SOCE in autoreactive T cells is required for induction of CNS inflammation and demyelination.
STIM1-deficient T cells are primed by MOG35-55 peptide but fail to produce IL-17 and IFN-γ in response to autoantigen stimulation
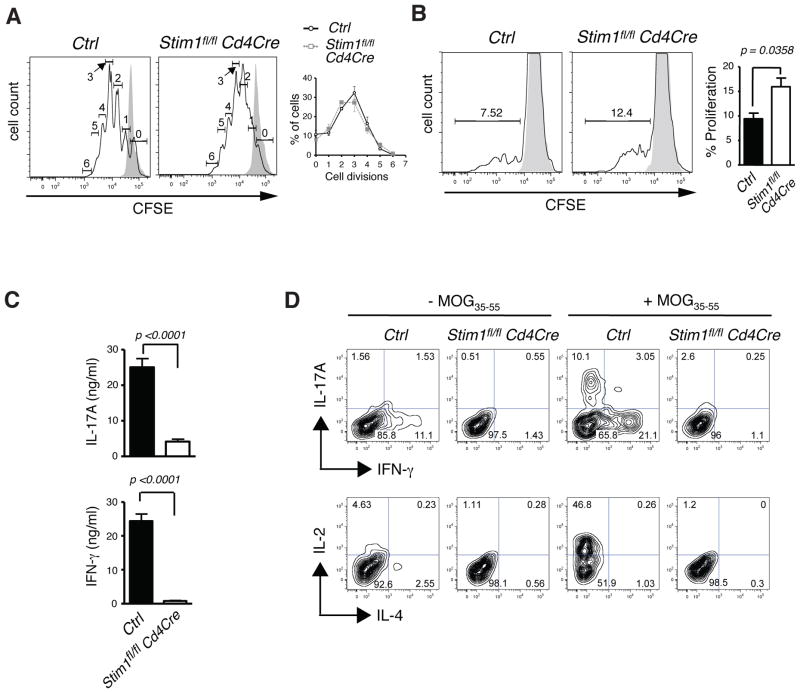
Potential causes for the inability of STIM-deficient T cells to induce EAE are (i) a defect in priming of T cells by MOG35-55 peptide expressed on antigen presenting cells, (ii) a failure of STIM-deficient T cells to differentiate into proinflammatory Th1 or Th17 cells, (iii) a failure of successfully primed T cells to produce proinflammatory cytokines, (iv) a defect in the expansion of autoreactive T cells or (v) a defect in infiltration of effector T cells into the CNS. To distinguish between these possibilities, we first tested the ability of STIM1-deficient T cells to be activated by TCR stimulation. Activation of T cells from Stim1fl/fl Cd4-Cre mice by TCR crosslinking with anti-CD3 / anti-CD28 antibodies in vitro induced proliferation in STIM1-deficient T cells that was comparable to that of wild-type T cells (Fig. 2A). Furthermore, we found normal to moderately increased autoantigen specific proliferation of STIM1-deficient T cells when CD4+ T cells isolated from MOG35-55 immunized Stim1fl/fl Cd4-Cre or wild-type mice were restimulated with MOG35-55 in vitro (Fig. 2B). These results indicate that priming of MOG35-55 specific T cells had occurred in STIM1-deficient mice in vivo and that SOCE is dispensable for the initial proliferation of encephalitogenic T cells.
Figure 2. Successful priming but impaired IFN-γ and IL-17 expression in MOG35-55-specific T cells lacking STIM1.
A, Naive CD4+ T cells isolated from wild-type Ctrl (Stim1fl/fl) or Stim1fl/fl Cd4-Cre mice were loaded with CFSE and stimulated with αCD3 / αCD28 antibodies for 3 days. Representative histogram (solid black line, stimulated cells; shaded area, unstimulated; numbers indicate the number of cell divisions) and mean±SEM cell percentages per cell division of two mice per group. B-D, EAE was induced in Ctrl (Stim1fl/fl ) and Stim1fl/fl Cd4-Cre mice with MOG35-55 and 12 days later, CD4+ T cells were isolated from the draining lymph nodes. B, The isolated CD4+ T cells were labeled with CFSE and stimulated with 50 μg/ml MOG35-55 peptide in the presence of irradiated (20 Gy) syngeneic splenocytes for three days. Representative histograms (solid black line, stimulated cells; shaded area, unstimulated) and mean±SEM percentages of proliferating T cells of three mice per group. C, The isolated CD4+ T cells were cultured with irradiated syngeneic splenocytes and 50 μg/ml MOG35-55 for 3 days and IL-17A and IFN-γ levels determined by ELISA; mean±SEM of four mice per group. D, The isolated CD4+ T cells were cultured with 10 ng/ml IL-23 for 3 days in the presence (-) or absence (+) of 50 μg/ml MOG35-55 and then stimulated with PMA/ ionomycin for 6 hours before detection of intracellular cytokines. Density plots are representative of three independent experiments.
In human and mouse T cells SOCE is required for production of several cytokine genes such as IL-2 and IFN-γ [19, 24] but the role of Ca2+ influx in expression of proinflammatory Th17 cytokines has not been studied in detail. Because Th17 cells are an important encephalitogenic CD4+ T cell subset in EAE [5–8], we asked whether antigen-primed STIM1-deficient T cells are able to produce IL-17A. CD4+ T cells from MOG35-55 immunized wild-type mice produced robust amounts of IL-17A and IFN-γ after restimulation with MOG35-55 for 3 days in vitro whereras T cells from Stim1fl/fl Cd4-Cre mice were strongly impaired in their expression of both cytokines (Fig. 2C). A similar defect in IL-17A production was observed in STIM1-deficient but not wild-type control T cells that were isolated from MOG35-55 treated mice 14 days after immunization, restimulated with MOG35-55 peptide and cultured in vitro for 3 days in the presence of IL- 23 to promote terminal Th17 differentiation. The subsequent stimulation of STIM1-deficient Th17 cells with PMA/ ionomycin failed to induce significant expression of IL-17A (Fig. 2D). This defect is not specific to Th17 cells as lack of STIM1 also impaired production of IFN-γ and IL-2. Taken together, these data show that STIM1 and SOCE are required for antigen specific expression of IL-17A and IFN-γ in T cells that were successfully primed with autoantigen.
Impaired IL-17 expression in STIM1-deficient Th17-differentiated cells
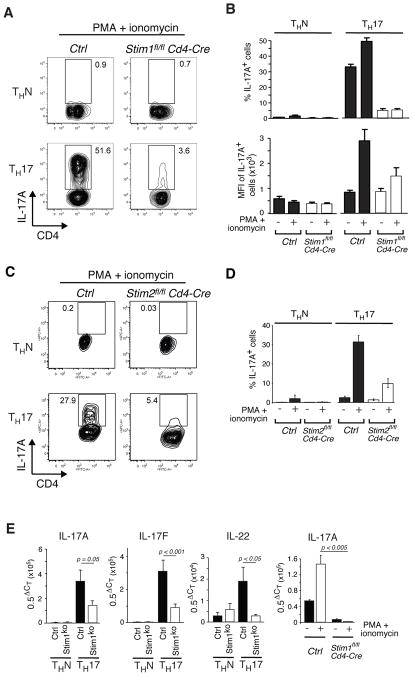
The failure of MOGspecific STIM1-deficient T cells to produce IL-17A could be due to a defect in the differentiation of naïve CD4+ T cells into Th17 effector cells. To evaluate this possibility we differentiated naïve CD4+ T cells from wildtype, Stim1fl/fl Cd4-Cre and Stim2fl/fl Cd4-Cre mice under Th17 conditions in vitro. Th17 cells (but not cells in non-polarizing condtions (ThN cells)) from wild-type mice showed robust induction of IL-17A expression upon restimulation with PMA/ionomycin (Fig. 3A-D). By contrast, the number of IL-17A+ cells and the level of IL-17A expression per cell were severely reduced in STIM1-deficient Tcells that were restimulated with PMA/ ionomycin (Fig. 3A, B). A similar defect in Th17 cytokine expression was observed in T cells from Stim1fl/fl Cd4-Cre mice at the mRNA level. Transcript levels of IL-17A, IL-17F and IL-22 in non-stimulated cells were significantly reduced (Fig. 3E, left panels) and induction of IL-17A expression upon restimulation was strongly impaired (Fig. 3E, right panel). Th17-differentiated cells from Stim2fl/fl Cd4-Cre mice showed a similar defect in IL-17 production that was only slightly less pronounced than that in STIM1-deficient Th17-differentiated cells (Fig. 3C, D). This finding is consistent with the role of STIM2 in maintaining SOCE and nuclear translocation of NFAT, which is required for expression of cytokines such as IFN-γ [24]. Taken together, Th17 cytokine expression is severely compromised in the absence of either STIM1 or STIM2 even under cell culture conditions in vitro that strongly favor the development of Th17 cells.
Figure 3. Impaired production of IL-17 in STIM1- and STIM2-deficient Th17 cells.
CD4+ T cells isolated from the lymph nodes of Ctrl (Stim1fl/fl), Stim1fl/fl Cd4-Cre and Stim2fl/fl Cd4-Cre mice were cultured under ThN or Th17 conditions for 3 days. A, C, Representative cytometry plots of intracellular IL-17A staining after PMA/ionomycin stimulation for 6 hours. B, D, Percentage and (B) mean fluorescent intensity (MFI) of IL-17A producing cells in the presence/absence of PMA/ionomycin stimulation; mean±SEM from 3-10 repeat experiments. E, mRNA levels of IL-17A, IL-17F and IL-22 in unstimulated Th17 and ThN cells (3 panels on the left) and IL-17A mRNA levels in PMA/ionomycin (6h)-stimulated Th17 cells (right panel) as determined by quantitative real-time PCR. Mean±SEM from 6-11 (unstimulated) and 3 (stimulated) repeat PCR experiments performed in triplicates. The statistical significance was calculated using Student's t-test. Abbreviations: Stim1KO, Stim1fl/fl Cd4-Cre; CT, threshold cycle.
Normal expression of Th17 cell lineage specific transcription factors but reduced levels of IL-23R in STIM1-deficient T cells
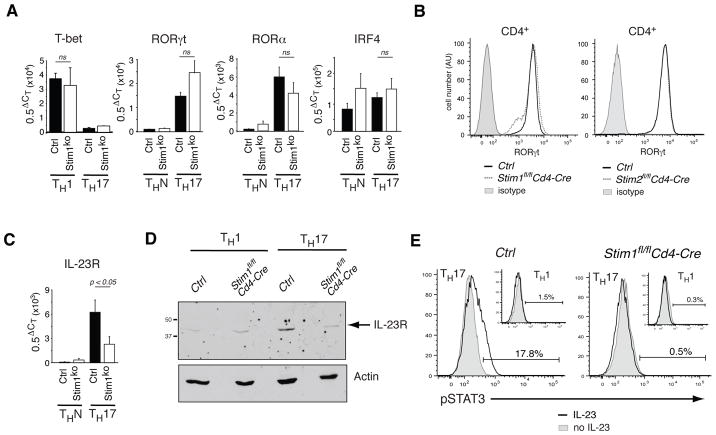
To exclude that IL-17 expression was impaired because CD4+ T cells fail to differentiate into Th17 cells in the absence of SOCE, we assessed the expression of transcription factors, cytokines and cytokine receptors that are essential for Th17 differentiation in T cells from Stim1fl/fl Cd4-Cre mice. T cells from RORγt−/− and IRF4−/− mice fail to differentiate into Th17 cells and both mouse strains are resistant to induction of EAE [15, 29]. STIM1-deficient and wild-type CD4+ T cells cultured under Th17-polarizing conditions in vitro expressed comparable amounts of the Th17 lineage-specific transcription factors RORγt and RORα as well as IRF4, a transcription factor that is important for both Th2 and Th17 differentiation (Fig. 4A). mRNA expression of RORγt and RORα was restricted to Th17 cells and not observed in CD4+ T cells cultured under non-polarizing conditions (Fig. 4A). In addition, protein expression of RORγt was comparable to that in wild-type T cells in both STIM1- and STIM2-deficient T cells (Fig. 4B). Of note is the comparable expression of the Th1 specific transcription factor T-bet in STIM1-deficient and wild-type T cells differentiated under Th1 conditions despite the severe defect in IFN-γ expression reported earlier (Fig. 4A)[24]. Collectively, normal expression of Th1 and Th17 specific transcription factors T-bet, RORγt and RORα suggests that the initial differentiation of naive CD4+ T cells into proinflammatory Th subsets is intact in the absence of STIM1 and SOCE. Defects in the Ca2+ dependent expression of cytokines such as IL-21 or cytokine receptors could, however, negatively affect differentiation and homeostasis of STIM1-deficient Th17 cells [30].
Figure 4. Normal levels of Th17 lineage-specific transcription factors but reduced expression of IL-23R in STIM1-deficient Th17 cells.
CD4+ T cells isolated from the lymph nodes of Ctrl (Stim1fl/fl and Stim2fl/fl), Stim1fl/fl Cd4-Cre and Stim2fl/fl Cd4-Cre mice were cultured under non-polarizing (ThN), Th1 or Th17 conditions for 3 days. A, T-bet, RORγt, RORα and IRF4 mRNA expression as measured by real-time PCR; mean±SEM of 4–10 real-time PCR experiments (performed in triplicates) per gene from three independent cDNAs. CT, threshold cycle; ns, non-significant (Student's t-test). B, Representative histograms of RORγt protein expression in CD4+ Th17-differentiated cells as measured by flow cytometry. Results are representative of ≥3 repeat experiments. C, IL-23R mRNA expression measured by real-time PCR. Mean±SEM of 10 repeat PCR experiments (performed in triplicates) from three independent cDNAs. Stim1KO, Stim1fl/fl Cd4-Cre. D, IL-23R levels as determined by Western blot; data are representative of 3 repeat experiments. E, Th1 and Th17 cells from Stim1fl/fl Cd4-Cre and Ctrl (Stim1f/f) mice were incubated with or without IL-23 for 30 min, fixed and stained with anti-STAT3 (pY705) antibody and pSTAT3 levels were determined by flow cytometry. Data are representative of 2 repeat experiments using 3 mice per group.
Consistent with this idea, mRNA and protein expression levels of the receptor for IL-23 (IL-23R) were significantly reduced in Th17-differentiated cells lacking STIM1 compared to wild-type control cells (Fig. 4 C, D). Impaired IL-23R expression resulted in impaired IL-23-mediated STAT3 phosphorylation in STIM1-deficient Th17 cells (Fig. 4E). Reduced IL-23R expression in STIM1-deficient T cells may interfere with Th17 cell homeostasis and expansion of IL-17-producing encephalitogenic T cells as IL-23 was shown to maintain IL-17 production in Th17 cells including encephalitogenic Th17 cells [31].
STIM1 and STIM2 are required for expansion of encephalitogenic T cells and their accumulation in the CNS and lymphoid organs
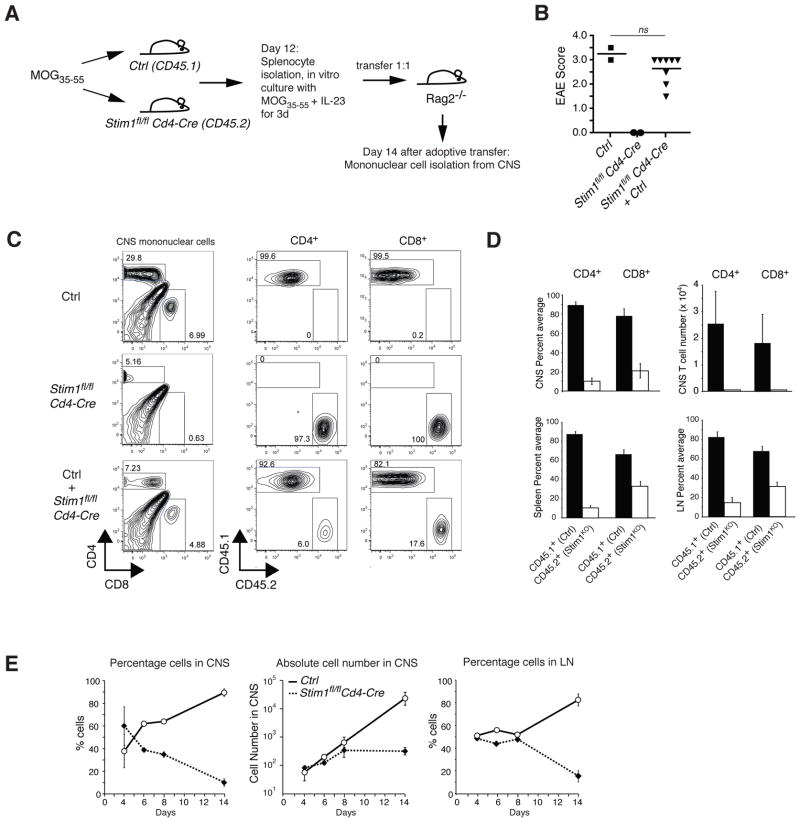
While the defect in Th17 cell effector function in the absence of STIM1 provides a reasonable explanation for the resistance of Stim1fl/fl Cd4-Cre mice to EAE, it was surprising to see a complete absence of STIM1-deficient T cells in the CNS of MOG35-55 treated mice (Fig. 1D-F) despite apparently normal priming and proliferation of STIM1-deficient T cells (Fig. 2A, B). IL-17 itself was shown to disrupt tight junctions between endothelial cells of the blood brain barrier thus directly promoting infiltration of encephalitogenic T cells into the CNS[13]. Impaired IL-17 production in STIM1-deficient T cells could be responsible –at least in part – for the absence of T cells in the CNS of Stim1fl/fl Cd4-Cre mice. To test whether MOG35-55-specific T cells lacking STIM1 can expand in vivo and infiltrate the brain once the blood brain barrier is breached during CNS inflammation, we adoptively cotransferred T cells from CD45.2+ Stim1fl/fl Cd4-Cre mice and CD45.1+ wild-type mice immunized with MOG35-55 into Rag2−/− mice (Fig. 5A). Transfer of mononuclear cells from MOG35-55 immunized wild-type mice that were restimulated in vitro with MOG and IL-23 to boost Th17 differentiation resulted in EAE in recipient mice (Fig. 5B). By contrast, mice that had received mononuclear cells from MOG immunized Stim1fl/fl Cd4-Cre mice only did not develop EAE. Adoptive cotransfer of a 1:1 mixture of wildtype and STIM1-deficient mononuclear cells caused EAE with disease scores similar to those observed after transfer of wild-type T cells alone. Surprisingly, analysis of T cells in the brain and spinal cord of recipient Rag2−/− mice at the peak of disease (day 14 after transfer) showed that the large majority (>90%) of CNS-infiltrating CD4+ T cells were of wild-type donor origin whereas the percentage and absolute number of STIM1-deficient T cells in the brain and spinal cord was strongly reduced (Fig. 5C, D). The predominance of wildtype T cells and paucity of STIM1-deficient T cells was, however, not specific to the CNS but was also observed in the spleen and lymph nodes (Fig. 5D). Cotransfer of T cells isolated from Stim2fl/fl Cd4-Cre and wild-type mice resulted in a similar enrichment of wild-type T cells in both the CNS and peripheral lymphoid organs (Supporting Information Fig. 1). These findings suggested that STIM1- and STIM2-deficient T cells die or fail to proliferate when transferred in vivo or that they have a defect in migrating to the CNS and secondary lymphoid organs.
Figure 5. Impaired ability of encephalitogenic STIM1-deficient T cells to expand and accumulate in the CNS and lymphoid organs.
A, Schematic of the protocol used in (B-E). Mononuclear cells were harvested from the CNS of CD45.1+ Ctrl mice or CD45.2+ Stim1fl/fl Cd4-Cre mice 12 days after immunization with MOG35-55 and restimulated in vitro with MOG35-55 and IL-23 for 3 days; 2 x 107 of the stimulated cells were transferred either separately or together into Rag2−/− mice. The recipient mice were analysed 14 days after transfer. B, Clinical EAE scores in the recipient Rag2−/− mice . Symbols represent individual mice; horizontal line, the mean; ns, not significant (Student's t-test). C, CD45.1 and CD45.2 expression on CD4+ and CD8+ T cells present in the mononuclear cell population isolated from the CNS. Density plots are representative of three independent experiments. D, Percentages of CD45.1+ and CD45.2+ CD4+ and CD8+ T cells in the CNS, spleen and lymph nodes (LN) and cell numbers of CD45.1+ and CD45.2+ CD4+ and CD8+ T cells in the CNS; mean±SEM of 8 mice. Stim1KO, Stim1fl/fl Cd4-Cre. E, Similar experiment as described in B-D, but mononuclear cells were recovered from the CNS and lymph nodes of Rag2−/− recipient mice on days 4, 6, 8 and 14 after adoptive co-transfer and analyzed for CD45.1 and CD45.2 expression. Shown are the mean±SEM percentages and absolute numbers of T cells from Ctrl (Stim1fl/fl) and Stim1fl/fl Cd4-Cre mice in the CNS and lymph nodes. Data shown are from one cotransfer experiment with two mice analyzed per time point.
Excessive death of T cells lacking SOCE as a cause for reduced numbers of STIM1-deficient T cells in the CNS or lymphoid organs is unlikely as ratios of CD45.1+ wild-type and CD45.2+ STIM1-deficient T cells remained close to a 1:1 ratio for at least 8 days after cell transfer (Fig. 5E, left and right panels). Absolute numbers of STIM1-deficient cells in the CNS increased moderately during this time period at a rate that was only slightly reduced compared to wild-type T cells (Fig. 5E, middle panel). By contrast, 14 days after transfer, absolute numbers of CD45.1+ wild-type T cells in the CNS had increased dramatically whereas numbers of CD45.2+ STIM1-deficient T cells remained constant (Fig. 5E, middle panel) suggesting that T cells lacking SOCE fail to properly expand in vivo or migrate to the CNS and lymphoid organs. The reduced number of STIM1-deficient T cells 14 days after transfer is unlikely to be due to a general proliferation defect or excessive cell death. T cells lacking STIM1 proliferate– at least over a short period of time (3 days) –as well or better than wild-type T cells in vitro (Fig. 2A, B) and in vivo when adoptively transferred into an allogeneic recipient (Supporting Information Fig. 3). STIM1-deficient T cells differentiated under Th17 conditions for 8 days in vitro are viable but fail to expand compared to wildtype control cells. This defect is Th17 specific and is not observed in STIM1-deficient Th1 cells (Supporting Information Fig. 2). A specific defect in the expansion of STIM1-deficient Th17 cells in vitro (Supporting Information Fig. 2C) and in vivo (Fig. 5E) may be related to the reduced IL-23R expression we observed in Th17-differentiated cells as IL-23 is critical for Th17 homeostasis [18].
Chemokine receptor mediated Ca2+ influx and migration are impaired in STIM1- deficient T cells
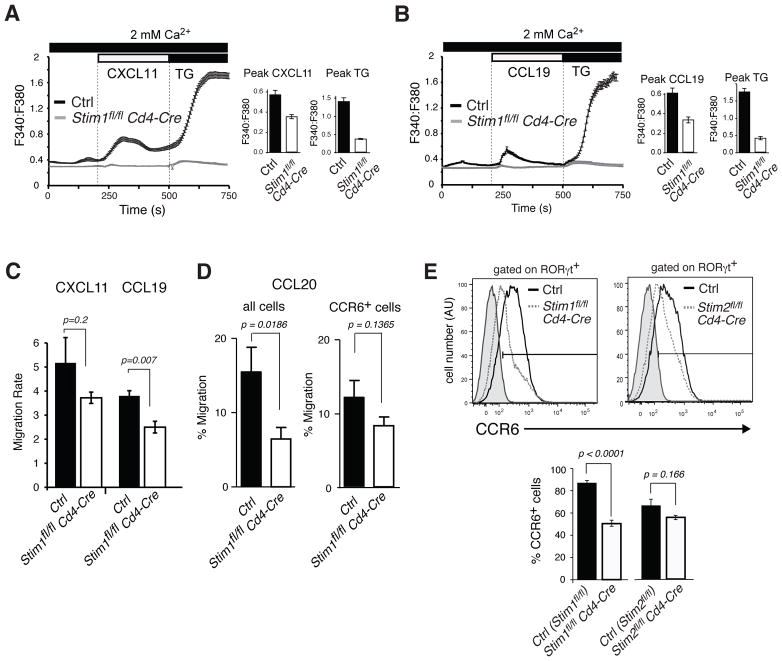
To determine whether STIM1-deficient T cells may have a defect in their ability to migrate, we investigated chemokine dependent Ca2+ influx and chemotaxis. Chemokine binding to G-protein coupled chemokine receptors activates PLCβ resulting in production of inositol 1,4,5-triphosphate (IP3). T cells from PLCβ2β3−/− mice were shown to have impaired Ca2+ influx and a defect in migration when stimulated with the CXCR4 ligand SDF1α (CXCL12) [32]. We find that Ca2+ influx in response to chemokine stimulation is store-operated and depends on STIM1 as T cells from Stim1fl/fl Cd4-Cre mice lack CXCL11 and CCL19 mediated Ca2+ influx in contrast to wild-type T cells (Fig. 6A, B) despite normal expression levels of the chemokine receptors CXCR3 and CCR7 (Supporting Information Fig. 4). Impaired chemokine induced SOCE in STIM1-deficient T cells is associated with a moderate defect in chemotaxis in response to CXCL11 and CCL19 stimulation (Fig. 6C). A similar migration defect of STIM1-deficient T cells was observed in response to CCL20, which binds to the chemokine receptor CCR6. CCR6 is predominantly expressed on Th17 cells. Infiltration of encephalitogenic T cells into the CNS during EAE has recently been suggested to depend on CCR6 because mice lacking expression of CCR6 were protected from EAE and lacked T cells in the CNS [33–35]. We find that CCL20 dependent migration of Th17-differentiated cells lacking STIM1 was impaired compared to wild-type T cells (Fig. 6D, left panel). In addition, expression levels of CCR6 were reduced on Th17-differentiated cells from Stim1fl/fl Cd4-Cre mice compared to wild-type controls (Fig. 6E). When chemotaxis was compared in CCR6+ T cells only, STIM1-deficient CCR6+ Tcells showed moderately reduced chemotaxis compared to CCR6+ wild-type T cells (Fig. 6D, right panel), suggesting that impaired CCL20 dependent chemotaxis of STIM1-deficient T cells is due to both reduced CCR6 expression and partially impaired signaling through CCR6. As PLCβ activation and Ca2+ influx are signaling mechanisms common to all chemokine receptors, these findings indicate that SOCE is required for chemokine signaling and migration of T cells in response to a wide range of chemokine signals. Abolished SOCE in the absence of STIM1 is likely to contribute to the low numbers of T cells in the CNS and lymphoid organs of Stim1fl/fl Cd4-Cre mice.
Figure 6. Impaired chemokine-receptor mediated Ca2+ influx and migration in STIM1-deficient T cells.
A-B,CD4+ T cells from Ctrl (Stim1fl/fl) and Stim1fl/fl Cd4-Cre mice were cultured in vitro for 6 days under ThN conditions. The T cells were then loaded with 1 μM Fura2-AM, stimulated with (A) 100 nM CXCL11 or (B) 30 nM CCL19 in 2 mM extracellular Ca2+ and intracellular [Ca2+]i levels were measured. Thapsigargin (TG, 1 μM) stimulation was used to fully activate SOCE. A representative plot and mean±SEM of peak [Ca2+]i of 4–6 (Ctrl) and 2–4 (Stim1fl/fl Cd4-Cre) repeat experiments are shown. C-D, CD4+ T cells from wild-type and Stim1fl/fl Cd4-Cre mice were cultured under (C) ThN and (D) Th17 conditions for 6 and 3 days respectively. T cell migration towards (C) CXCL11 or CCL19 and (D) CCL20 was determined by transwell assay. Mean±SEM of (C) migration rates and (D) percentages of migrated cells of 4 (CCL19), 8 (CXCL11) and 4 (CCL20) repeat experiments performed in triplicate and corrected for unspecific chemokinesis in the absence of chemokine. E, CD4+ T cells of Ctrl (Stim1fl/fl or Stim2fl/fl), Stim1fl/fl Cd4-Cre and Stim2fl/fl Cd4-Cre mice were cultured under Th17-polarizing conditions for 3 days, stained with anti-RORγt and either anti- CCR6 or isotype control antibodies and analyzed by flow cytometry. Representative histograms of CCR6 expression and mean±SEM of the percentages of CCR6+ T cells of 7 (Stim1fl/fl and Stim1fl/fl Cd4-Cre) and 3 (Stim2fl/fl and Stim2fl/fl Cd4-Cre) independent experiments. Student's t-test was used for calculation of statistical significance.
Discussion
In this study we show that two essential regulators of store-operated Ca2+ entry in T cells, STIM1 and STIM2, are required for the function of autoreactive T cells and their ability to induce EAE. T cell specific deletion of STIM1 renders mice almost completely resistant to EAE; a similar albeit less complete protection was observed in mice lacking STIM2 in T cells. An important role for STIM1 and STIM2 in EAE was recently reported in a study demonstrating that Stim1−/− bone marrow chimeric mice are resistant to EAE whereas severity of EAE in complete Stim2−/− mice was comparable to control mice despite moderately delayed disease onset [28]. The latter finding is in contrast to a more pronounced protective effect we observed in mice with T cell specific deletion of STIM2. Given the use of bone marrow chimeric mice in the latter study, the lack of STIM proteins in a variety of immune cell types including T cells and macrophages can potentially contribute to the protection from EAE despite the fact that dendritic cells appeared to function normally [28]. We here propose that T cell specific deletion of STIM1 or STIM2 is sufficient to protect mice from EAE. We show that STIM proteins are required for several aspects of effector T cell function during EAE including (i) production of proinflammatory cytokines, (ii) expression of IL-23R on Th17 cells, (iii) expansion of encephalitogenic T cells, and (iv) chemokine dependent migration of T cells.
First, STIM1 and STIM2 are required for expression of the proinflammatory Th1 and Th17 cytokines IFN-γ and IL-17. STIM1-deficient T cells from MOG immunized mice are unable to produce IL-17 in response to restimulation with MOG antigen in vitro. This defect is not due to impaired priming of STIM1-deficient T cells in secondary lymphoid organs as T cells from Stim1fl/fl CD4Cre mice proliferated normally in response to restimulation with MOG. This finding suggests a differential requirement for STIM1 and SOCE during the priming and effector phases of a T cell response. A weak Ca2+ signal in response to TCR stimulation –resulting for instance from depletion of ER Ca2+ stores which is intact in STIM1-deficient T cells [24] – may be sufficient to prime T cells and allow for their initial proliferation. This is consistent with normal proliferative responses of STIM1-deficient T cells upon anti-CD3 stimulation in vitro (Fig. 2A)[24, 36, 37] and allogeneic stimulation in vivo (Supporting Information Fig. 3). By contrast, cytokine gene expression in effector T cells is likely to require stronger, more sustained Ca2+ signals provided by SOCE. Importantly, even when STIM1- and STIM2-deficient T cells were biased to differentiate into Th17 cells in the presence of IL-6 and TGF-β in vitro they failed to produce IL-17 (Fig. 3A-D).
The defect in IL-17 production in T cells from Stim1fl/fl CD4Cre mice is not specific to Th17 cytokines as STIM1-deficient T cells also lacked expression of IFN-γ, IL-2 and IL-4 (Fig. 2D) [24]. In Th1 and Th2 cells, Ca2+ influx is required for activation of the transcription factor NFAT which interacts with T-bet and GATA-3 to induce production of IFN-γ and IL-4, respectively (reviewed in [38]). NFAT has recently been demonstrated to be able to mediate IL-17A expression, presumably by binding to a newly identified NFAT binding site in the IL-17A promoter [39]. Lack of IL-17 and IFN-γ production in STIM1-deficient T cells does not seem to result from impaired initial T helper cell differentiation as the expression of lineage specific transcription factors such as RORγt, RORα and T-bet was normal in STIM1-deficient Th17 and Th1 cells, respectively.
By contrast, we observed reduced expression of IL-23R in Th17-differentiated cells from STIM1-deficient mice. IL-23 was shown to maintain IL-17 production in Th17 cells including encephalitogenic Th17 cells [31], to be required for terminal Th17 differentiation and to act as a survival factor for Th17 cells [5, 17, 18]. Mice lacking the p19 subunit of IL-23 or IL-23R showed reduced IL-17 production and were resistant to induction of EAE [17, 40]. We speculate that reduced expression of IL-23R in STIM1-deficient T cells may interfere with homeostasis of Th17 cells. This could explain why STIM1-deficient encephalitogenic T cells from MOG immunized mice fail to expand in vivo compared to wild-type T cells when adoptively transferred to Rag2-/- mice (Fig. 5E) and why Th17 but not Th1 cells from Stim1fl/fl CD4Cre mice fail to proliferate in vitro (Supporting Information Fig. 2C).
Strongly impaired production of both IL-17 and IFN-γ in T cells from STIM1 and STIM2-deficient mice is likely to contribute to their resistance to EAE. Although the role of Th1 cells in the pathophysiology of murine EAE has been called into question after the finding that Ifng−/− mice are susceptible to EAE [2, 41, 42], recent data suggest that both Th1 and Th17 cell contribute to different aspects of EAE pathogenesis [43–45]. It therefore seems likely that the combined lack of both IFN-γ and IL-17A production in STIM-deficient T cells has a synergistic effect in protecting animals from autoimmune CNS inflammation.
Finally, a defect in chemokine dependent migration in STIM1-deficient T cells may contribute to protection against EAE. Chemokine signaling has been implicated in the pathogenesis of EAE as several chemokine receptor-deficient mice including Ccr2−/−, Ccr6−/− and Ccr7−/− mice are protected from EAE [33, 46–48]. Resistance to EAE in some of these mice was attributed to altered T cell priming and generation of proinflammatory T cells in the periphery [34, 48] while impaired T cell infiltration into the CNS is responsible for resistance to EAE in Ccr6−/− mice despite normal differentiation into Th17 cells in the periphery [33]. We observed a partial defect in the expression of CCR6 in Th17-differentiated STIM1-deficient cells in vitro and impaired T cell migration towards the CCR6 ligand CCL20. This part of a more general defect in chemokine signaling and migration in the absence of STIM1. Chemokine receptors, like other G-protein coupled receptors, activate PLCβ resulting in the production of IP3, release of Ca2+ from ER stores and induction of SOCE. In contrast to wild-type T cells, STIM1-deficient T cells lacked chemokine induced Ca2+ influx and showed a partial defect in T cell migration. We speculate that abolished Ca2+ signals downstream of not just one but multiple chemokine receptors in STIM1-deficient T cells contributes their inability to migrate to the CNS and peripheral lymphoid organs.
Combined T cell specific deletion of both STIM1 and STIM2 completely protected mice from EAE to an extent greater than that observed in either Stim1fl/fl Cd4-Cre or Stim2fl/fl Cd4-Cre mice alone, indicating that both genes contribute to autoreactive T cell function. We had previously shown that deletion of both STIM1 and STIM2 impairs Treg cell development resulting in a severe autoinflammatory phenotype characterized by lymphadenopathy, splenomegaly and myelolymphocytic infiltration of solid organs [24]. Limited autoimmunity was also observed in human patients that lacked STIM1 expression and had reduced numbers of Treg [25]. Protection of Stim1fl/fl Stim2fl/fl Cd4-Cre mice from EAE despite ~ 90% reduction in Treg cell numbers and severely compromised Treg cell function [24] suggests that effector T cells functions are so severely impaired in the absence Ca2+ influx mediated by STIM1 and STIM2 that Treg are not required for controlling CNS inflammation. Myelin specific Treg were shown to accumulate in the CNS during EAE but failed to prevent the onset of disease, presumably because encephalitogenic effector T cells suppressed their function via secretion of IL-6 and TNFα [49]. Interestingly, we had observed substantial TNFα expression in STIM1/STIM2-deficient CD4+ T cells whereas production of other cytokines such as IL- 2 was strongly impaired [24], suggesting that STIM1/STIM2-deficient encephalitogenic Teff cells may retain the ability to further suppress the already incapacitated Treg pool in Stim1fl/fl Stim2fl/fl Cd4-Cre mice.
A role for STIM1 and STIM2 has been described for other cell types known to be important for EAE besides T cells including neurons and endothelial cells. Neurons in Stim2−/− mice were protected from hypoxic cell death [50] and STIM1 was reported to mediate store-operated Ca2+ entry and proliferation of endothelial cells [51]. There is some evidence that Ca2+ signals in endothelial cells are involved in regulating the integrity and formation of adherens and tight junctions between endothelial cells in the lung and CNS, respectively. In the lung vasculature endothelial cells are linked predominantly by adherens junctions. Neurohumoral inflammatory mediators induce Ca2+ influx in endothelial cells and increase endothelial cell permeability and intercellular gap formation [52]. Ca2+ influx may also be implicated in the formation and maintenance of tight junctions between endothelial cells [53]. In light of these findings, it is noteworthy that mice with T-cell specific deletion of STIM2 in our study were more protected from EAE compared to Stim2−/− mice that lack STIM2 in all tissues including oligodendrocytes and endothelial cells [28]. Lack of STIM2 apparently does not protect oligodendrocytes – the main target of encephalitogenic T cells in EAE and –MS and neurons from cell death during EAE which is in contrast to protection of Stim2−/− mice from hypoxemia induced neuronal death [50]. In addition, absence of STIM2 in endothelial cells of Stim2−/− mice does not seem to afford additional protection from EAE compared to Stim2fl/fl Cd4-Cre mice. Increased EAE severity in Stim2−/− mice compared to Stim2fl/fl Cd4-Cre mice might in fact point to a role for STIM2 in endothelial tight junction formation. This is speculative, however, as neither expression nor function of STIM2 in endothelial cells and oligodendrocytes have been analyzed in vivo. Our finding that T-cell specific deletion of STIM2 protects mice from EAE demonstrates an essential role of STIM2 for T cell function in vivo.
Evidence for an important role of Ca2+ signals in the pathogenesis of EAE also comes from experiments in which inhibition of K+ channels in T cells – required for maintaining a negative membrane potential and providing the driving force for Ca2+ influx[54] –resulted in attenuation of EAE in rats and mice [55, 56]. Furthermore, inhibition of the Ca2+ dependent phosphatase calcineurin with the immunosuppressant cyclosporin A was shown to have beneficial effects in the treatment of MS in human patients although significant adverse effects prevented its use in most cases [57]. These findings demonstrate that Ca2+ influx and Ca2+ dependent signaling in T cells are essential for the function of autoreactive T cells. Since most if not all Ca2+ influx in T cells is store-operated and dependent on STIM1 and STIM2 function, inhibition of this signaling pathway may be beneficial for the treatment of autoimmune diseases such as MS.
Materials and Methods
Mice
Stim1fl/fl, Stim2fl/fl and Stim1fl/flStim2fl/fl Cd4-Cre mice were described previously [24]. Rag2−/− mice were from Taconic (Hudson, NY). All mice were housed under specific pathogen-free conditions and used in accordance with a protocol approved by the Institutional Animal Care and Use Committee at NYU Medical Center.
EAE
EAE was induced as described [58]. Briefly, mice were immunized with 200 μg MOG35-55 peptide (Anaspec, Fremont, CA) emulsified in complete Freund’s adjuvans (CFA, Pierce, Thermo Scientific, Rockford, IL). On day 0 and 2 after immunization, mice were injected with 200 ng pertussis toxin (List Biological Laboratories, Campbell, CA). For passive induction of EAE, mice were immunized with 200 μg MOG35-55 emulsified in CFA. 12 days later, cells were isolated from lymph nodes and spleen and stimulated with 50 μg/ml MOG35-55 in the presence of 10 ng/ml IL-23 (eBioscience) for 3 days. Viable lymphocytes were isolated by Ficoll-Paque centrifugation and 1x107 cells from wild-type and Stim1fl/fl Cd4-Cre mice were injected separately or at a 1:1 ratio into Rag2−/− mice. On days 0 and 2 after cell transfer, recipient mice received 200 ng pertussis toxin. The severity of EAE was monitored and evaluated on a scale from 0–5 [58]: 0 = no disease; 0.5 = partially limp tail; 1 = paralyzed tail; 2 = hind limb weakness; 3 = hind limb paralysis; 4 = hind and fore limb paralysis; 5 = moribundity and death.
Histology and immunohistochemistry
Spinal cord serial sections, cut at 5 μm, were stained with hematoxylin/eosin and Luxol fast blue using standard methods. Images were acquired using a Zeiss Axioskop 40 microscope (Carl Zeiss MicroImaging, Thornwood, NY) and ProgRes image capture software (JENOPTIK Optical Systems, Easthampton, MA). Immunohistochemistry was performed as described [59]. Briefly, sections were incubated with biotin-conjugated rat-anti-mouse CD45 antibody (30-F11, BD Bioscience) and horseradish peroxidase (HRP)-conjugated streptavidin (Jackson Immunoresearch, West Grove, PA). Biotin-tyramide (Perkin Elmer) was used to amplify the fluorescence signal with streptavidin-Alexa 594 (Invitrogen). Images were acquired using a Zeiss AxioImager M1 equipped with an AxioCam MRm digital camera and analyzed using AxioVision software (Carl Zeiss MicroImaging).
T cell isolation and stimulation
Unless otherwise indicated, CD4+ T cells were isolated from draining lymph nodes and spleen of mice by negative selection using antibody-coated magnetic beads (Invitrogen). For cytokine measurements, CD4+ T cells isolated from spleen and lymph nodes of MOG immunized mice were stimulated with 50 μg/ml MOG35-55 either together with irradiated (20 Gy) splenocytes from syngeneic C57BL/6 mice or 10 ng/ml IL-23 as indicated.
Th differentiation in vitro
For in vitro differentiation of CD4+ T cells into ThN, Th1 or Th17 cells, 1x106 negatively selected CD4+ T cells were stimulated with 0.25 μg/ml anti-CD3 (145-2C11, eBioscience) and 1 μg/ml anti-CD28 (37.51, eBioscience) in goat-anti-hamster IgG (0.12 mg/ml, MP Biomedicals) pre-coated 6-well plates. For ThN cells, no further supplements were added; for Th1 cells, 10 ng/ml IL-12 (Peprotech) and 5 μg/ml anti-IL-4 (11B11, eBioscience) were added; for Th17 cells, 10 μg/ml anti-IFN-γ (XMG1.2, eBioscience), 10 μg/ml anti-IL-4, 20 ng/ml mouse IL-6 (Peprotech, Rocky Hill, NJ) and 0.5 ng/ml human TGF-β1 (Peprotech) were added. T cells were cultured for 3 days in Iscove’s modified Dulbecco’s medium (IMDM, Cellgro, Mediatech, Manassas, VA) containing 2mM L-glutamine, 50 μM β-mercaptoethanol, 100 U/ml penicillin, 100 μg/ml streptomycin and 10% FBS.
Proliferation assays
CD4+ T cells isolated from MOG35-55-immunized mice were labeled with 4 μM CFSE (Invitrogen) at room temperature for 5 min according to manufacturer’s instructions. Cells were stimulated for 3 days with 50 μg/ml MOG35-55 in the presence of irradiated (20 Gy) splenocytes from C57BL/6 mice. Alternatively, CD4+ T cells were stimulated with 0.25 μg/ml anti-CD3 and 1 μg/ml anti-CD28 antibodies on goat-anti-hamster IgG pre-coated plates. For flow cytometry, CFSE labeled cells were stained with AlexaFluo 647-conjugated anti-mouse CD4 antibody (L3T4).
Intracellular cytokine measurements and flow cytometry
For cytokine analysis, T cells were stimulated with 10 nM PMA and 1 μM ionomycin for 6 h; 5 μM Brefeldin A was added during the last 2 hrs of stimulation. Following Fc block with anti-CD16/32 (clone 93, eBioscience) cells were incubated with FITC-conjugated anti-mouse CD4 (L3T4) and fixed / permeabilized either with 4% paraformaldehyde / 0.5% saponin or commercial "Foxp3 staining buffer" (eBioscience). Cells were stained with the following antibodies (all from eBioscience): Alexa Fluor 647-conjugated anti-mouse IL-17A (eBioTC11-18H10.1), PE-conjugated anti-mouse IFN-γ (XMG1.2), APC-conjugated anti-mouse IL-2 (JES6-5H4), PE-conjugated anti-mouse IL-4 (11B11), PE-RORγt (clone AFKJS-9). For chemokine receptor analysis, cells were stained with rat anti-mouse CCR6 (Alexa Fluor 647, BD Pharmingen) and analyzed using a LSRII cytometer (BD Biosciences) and FlowJo software (Treestar, Ashland, OR).
Isolation of mononuclear cells from the CNS
Mice were anesthetized with ketamin/xylazine, perfused intracardially with 1x PBS and sacrificed immediately. Brain and spinal cord were homogenized in 1x PBS, passed through a 70 μm cell strainer and mononuclear cells isolated by Percoll (Sigma) density gradient centrifugation according to the manufacturer’s protocol.
STAT3 phosphorylation
Th1 and Th17 cells were differentiated for 3 days and incubated with 5 ng/ml of IL-23 (eBioscience) for 30 min at 37°C in 10% CO2. Cells were fixed in 4% paraformaldehyde, permeabilized with 90% methanol (-20oC) and washed with 1xPBS / 1% BSA. Following Fc block with anti-CD16/32, cells were incubated with anti-mouse CD4 (L3T4, eFluor 450-conjugated, eBioscience) and anti-mouse STAT3 (pY705, clone 4/P-STAT3, PE-conjugated, BD Biosciences).
ELISA
CD4+ cells were isolated from MOG35-55-immunized mice and stimulated for 3 days with 50 μg/ml MOG35-55 in the presence of syngeneic irradiated (20 Gy) splenocytes. On day 3, cell culture supernatants were analyzed for IL-17A and IFN-γ using Ready-SET-Go ELISA kits (eBioscience) according to manufacturer’s instructions.
Migration assays
5x105 CD4+ T cells differentiated in vitro into ThN or Th17 cells were resuspended in RPMI-1640 (0.5% FBS) and added to the upper compartment of a transwell chamber (Costar, Corning Incorporated). The bottom compartment contained either medium alone or medium plus CCL20 (500 ng/ml), CXCL11 (100 nM) or CCL19 (30 nM) (all from Peprotech). Cells were incubated for 2 h (CXCL11, CCL19) or 4 h (CCL20) at 37 °C, 5% CO2. Cells migrated to the bottom compartment were counted directly (CXCL11, CCL19) or first stained with anti-CCR6 antibody (CCL20) and then analyzed and counted by flow cytometry. Migration rates were calculated by dividing the number of cells migrated towards the chemokine by the number of cells migrated towards medium alone.
Realtime PCR
Quantitative RT-PCR was performed as described [60]. Briefly, cDNA was synthesized with SuperScript II reverse transcriptase (Invitrogen) from 150 ng total RNA of ThN- or Th17-differentiated T cells. Gene-specific cDNAs were amplified using the Maxima SYBR Green qPCR Master Mix (Fermentas, Glen Burnie, MD) and an iCycler thermocycler (BioRad). Threshold cycles for each transcript (CT) were normalized to GAPD (ΔCT). Gene expression is shown as 0.5ΔCT. Real-time PCR were performed in triplicates. Primer sequences can be found in Supporting Information.
Western blots were performed as described [60]. Briefly, total cell lysates from Stim1fl/fl Cd4-Cre and Ctrl (Stim1fl/fl) Th1 and Th17 cells were separated by SDS-PAGE, transferred to nitrocellulose membrane and incubated over night at 4°C with rabbit polyclonal anti-IL23R antibody (Millipore). Blots were reprobed with anti-Actin antibody (Santa Cruz) to control for equal loading.
Ca2+ imaging
Intracellular Ca2+ concentrations [Ca2+]i were measured as described [60]. Briefly, ThN cells were loaded with 1 μM fura-2/AM (Invitrogen), attached to poly-L-lysine-coated coverslips and stimulated in 2 mM extracellular Ca2+ Ringer's solution (155 mM NaCl, 4.5 mM KCl, 1 mM MgCl2, 2 mM CaCl2, 10 mM D-glucose, 5 mM Na- Hepes) with chemokines (30 nM CCL19, 100 nM CXCL11, Peprotech, Rocky Hill, NJ) or 1 μM thapsigargin (EMD Biosciences, San Diego, CA). Ca2+ signals were analyzed by time-lapse digital imaging on an IX81 epifluorescence microscope (Olympus) using Slidebook imaging software v4.2 (Olympus).
Statistical analysis was performed using the unpaired, two-tailed Student’s t test.
Supplementary Material
Acknowledgments
We thank Dr. J. Lafaille for valuable discussions and members of the Lafaille lab and C.-S. Tay for technical assistance. This work was funded by NIH grant AI066128 to S.F.
Abbreviations used in this paper
- CRAC
calcium release activated calcium
- EAE
experimental autoimmune encephalomyelitis
- MS
multiple sclerosis
- SOCE
store-operated calcium entry
- STIM
stromal interaction molecule
Footnotes
Conflict-of-interest. S.F. is scientific co-founder of and advisor to Calcimedica, a company that seeks to develop CRAC inhibitors.
References
- 1.Ben-Nun A, Wekerle H, Cohen IR. The rapid isolation of clonable antigen-specific T lymphocyte lines capable of mediating autoimmune encephalomyelitis. Eur J Immunol. 1981;11:195–199. doi: 10.1002/eji.1830110307. [DOI] [PubMed] [Google Scholar]
- 2.Ferber IA, Brocke S, Taylor-Edwards C, Ridgway W, Dinisco C, Steinman L, Dalton D, Fathman CG. Mice with a disrupted IFN-gamma gene are susceptible to the induction of experimental autoimmune encephalomyelitis (EAE) J Immunol. 1996;156:5–7. [PubMed] [Google Scholar]
- 3.Krakowski M, Owens T. Interferon-gamma confers resistance to experimental allergic encephalomyelitis. Eur J Immunol. 1996;26:1641–1646. doi: 10.1002/eji.1830260735. [DOI] [PubMed] [Google Scholar]
- 4.Willenborg DO, Fordham S, Bernard CC, Cowden WB, Ramshaw IA. IFN-gamma plays a critical down-regulatory role in the induction and effector phase of myelin oligodendrocyte glycoprotein-induced autoimmune encephalomyelitis. J Immunol. 1996;157:3223–3227. [PubMed] [Google Scholar]
- 5.Bettelli E, Korn T, Oukka M, Kuchroo VK. Induction and effector functions of T(H)17 cells. Nature. 2008;453:1051–1057. doi: 10.1038/nature07036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sutton C, Brereton C, Keogh B, Mills KH, Lavelle EC. A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J Exp Med. 2006;203:1685–1691. doi: 10.1084/jem.20060285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Aranami T, Yamamura T. Th17 Cells and autoimmune encephalomyelitis (EAE/MS) Allergol Int. 2008;57:115–120. doi: 10.2332/allergolint.R-07-159. [DOI] [PubMed] [Google Scholar]
- 8.Hofstetter H, Gold R, Hartung HP. Th17 Cells in MS and Experimental Autoimmune Encephalomyelitis. Int MS J. 2009;16:12–18. [PubMed] [Google Scholar]
- 9.Zhou L, Littman DR. Transcriptional regulatory networks in Th17 cell differentiation. Curr Opin Immunol. 2009;21:146–152. doi: 10.1016/j.coi.2009.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y. IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol. 2006;177:566–573. doi: 10.4049/jimmunol.177.1.566. [DOI] [PubMed] [Google Scholar]
- 11.Lock C, Hermans G, Pedotti R, Brendolan A, Schadt E, Garren H, Langer-Gould A, Strober S, Cannella B, Allard J, Klonowski P, Austin A, Lad N, Kaminski N, Galli SJ, Oksenberg JR, Raine CS, Heller R, Steinman L. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat Med. 2002;8:500–508. doi: 10.1038/nm0502-500. [DOI] [PubMed] [Google Scholar]
- 12.Ishizu T, Osoegawa M, Mei FJ, Kikuchi H, Tanaka M, Takakura Y, Minohara M, Murai H, Mihara F, Taniwaki T, Kira J. Intrathecal activation of the IL-17/IL-8 axis in opticospinal multiple sclerosis. Brain. 2005;128:988–1002. doi: 10.1093/brain/awh453. [DOI] [PubMed] [Google Scholar]
- 13.Kebir H, Kreymborg K, Ifergan I, Dodelet-Devillers A, Cayrol R, Bernard M, Giuliani F, Arbour N, Becher B, Prat A. Human TH17 lymphocytes promote blood-brain barrier disruption and central nervous system inflammation. Nat Med. 2007;13:1173–1175. doi: 10.1038/nm1651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aggarwal S, Gurney AL. IL-17: prototype member of an emerging cytokine family. J Leukoc Biol. 2002;71:1–8. [PubMed] [Google Scholar]
- 15.Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, Cua DJ, Littman DR. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126:1121–1133. doi: 10.1016/j.cell.2006.07.035. [DOI] [PubMed] [Google Scholar]
- 16.Leppkes M, Becker C, Ivanov II, Hirth S, Wirtz S, Neufert C, Pouly S, Murphy AJ, Valenzuela DM, Yancopoulos GD, Becher B, Littman DR, Neurath MF. RORgamma-expressing Th17 cells induce murine chronic intestinal inflammation via redundant effects of IL-17A and IL-17F. Gastroenterology. 2009;136:257–267. doi: 10.1053/j.gastro.2008.10.018. [DOI] [PubMed] [Google Scholar]
- 17.McGeachy MJ, Chen Y, Tato CM, Laurence A, Joyce-Shaikh B, Blumenschein WM, McClanahan TK, O'Shea JJ, Cua DJ. The interleukin 23 receptor is essential for the terminal differentiation of interleukin 17-producing effector T helper cells in vivo. Nat Immunol. 2009;10:314–324. doi: 10.1038/ni.1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Elson CO, Cong Y, Weaver CT, Schoeb TR, McClanahan TK, Fick RB, Kastelein RA. Monoclonal anti-interleukin 23 reverses active colitis in a T cell-mediated model in mice. Gastroenterology. 2007;132:2359–2370. doi: 10.1053/j.gastro.2007.03.104. [DOI] [PubMed] [Google Scholar]
- 19.Feske S, Giltnane J, Dolmetsch R, Staudt LM, Rao A. Gene regulation mediated by calcium signals in T lymphocytes. Nat Immunol. 2001;2:316–324. doi: 10.1038/86318. [DOI] [PubMed] [Google Scholar]
- 20.Feske S. Calcium signalling in lymphocyte activation and disease. Nat Rev Immunol. 2007;7:690–702. doi: 10.1038/nri2152. [DOI] [PubMed] [Google Scholar]
- 21.Cahalan MD. STIMulating store-operated Ca(2+) entry. Nat Cell Biol. 2009;11 :669–677. doi: 10.1038/ncb0609-669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Feske S. ORAI1 and STIM1 deficiency in human and mice: roles of store-operated Ca2+ entry in the immune system and beyond. Immunol Rev. 2009;231:189–209. doi: 10.1111/j.1600-065X.2009.00818.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gwack Y, Srikanth S, Oh-Hora M, Hogan PG, Lamperti ED, Yamashita M, Gelinas C, Neems DS, Sasaki Y, Feske S, Prakriya M, Rajewsky K, Rao A. Hair loss and defective T- and B-cell function in mice lacking ORAI1. Mol Cell Biol. 2008;28:5209–5222. doi: 10.1128/MCB.00360-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oh-Hora M, Yamashita M, Hogan PG, Sharma S, Lamperti E, Chung W, Prakriya M, Feske S, Rao A. Dual functions for the endoplasmic reticulum calcium sensors STIM1 and STIM2 in T cell activation and tolerance. Nat Immunol. 2008;9:432–443. doi: 10.1038/ni1574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Picard C, McCarl CA, Papolos A, Khalil S, Luthy K, Hivroz C, LeDeist F, Rieux-Laucat F, Rechavi G, Rao A, Fischer A, Feske S. STIM1 mutation associated with a syndrome of immunodeficiency and autoimmunity. N Engl J Med. 2009;360:1971–1980. doi: 10.1056/NEJMoa0900082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Feske S, Gwack Y, Prakriya M, Srikanth S, Puppel SH, Tanasa B, Hogan PG, Lewis RS, Daly M, Rao A. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature. 2006;441:179–185. doi: 10.1038/nature04702. [DOI] [PubMed] [Google Scholar]
- 27.Brandman O, Liou J, Park WS, Meyer T. STIM2 is a feedback regulator that stabilizes basal cytosolic and endoplasmic reticulum Ca2+ levels. Cell. 2007;131:1327–1339. doi: 10.1016/j.cell.2007.11.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schuhmann MK, Stegner D, Berna-Erro A, Bittner S, Braun A, Kleinschnitz C, Stoll G, Wiendl H, Meuth SG, Nieswandt B. Stromal interaction molecules 1 and 2 are key regulators of autoreactive T cell activation in murine autoimmune central nervous system inflammation. J Immunol. 2010;184:1536–1542. doi: 10.4049/jimmunol.0902161. [DOI] [PubMed] [Google Scholar]
- 29.Brustle A, Heink S, Huber M, Rosenplanter C, Stadelmann C, Yu P, Arpaia E, Mak TW, Kamradt T, Lohoff M. The development of inflammatory T(H)-17 cells requires interferon-regulatory factor 4. Nat Immunol. 2007;8:958–966. doi: 10.1038/ni1500. [DOI] [PubMed] [Google Scholar]
- 30.Hermann-Kleiter N, Baier G. NFAT pulls the strings during CD4+ T helper cell effector functions. Blood. 2010;115:2989–2997. doi: 10.1182/blood-2009-10-233585. [DOI] [PubMed] [Google Scholar]
- 31.Langrish CL, Chen Y, Blumenschein WM, Mattson J, Basham B, Sedgwick JD, McClanahan T, Kastelein RA, Cua DJ. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J Exp Med. 2005;201:233–240. doi: 10.1084/jem.20041257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bach TL, Chen QM, Kerr WT, Wang Y, Lian L, Choi JK, Wu D, Kazanietz MG, Koretzky GA, Zigmond S, Abrams CS. Phospholipase cbeta is critical for T cell chemotaxis. J Immunol. 2007;179:2223–2227. doi: 10.4049/jimmunol.179.4.2223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reboldi A, Coisne C, Baumjohann D, Benvenuto F, Bottinelli D, Lira S, Uccelli A, Lanzavecchia A, Engelhardt B, Sallusto F. C-C chemokine receptor 6-regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat Immunol. 2009;10:514–523. doi: 10.1038/ni.1716. [DOI] [PubMed] [Google Scholar]
- 34.Liston A, Kohler RE, Townley S, Haylock-Jacobs S, Comerford I, Caon AC, Webster J, Harrison JM, Swann J, Clark-Lewis I, Korner H, McColl SR. Inhibition of CCR6 function reduces the severity of experimental autoimmune encephalomyelitis via effects on the priming phase of the immune response. J Immunol. 2009;182:3121–3130. doi: 10.4049/jimmunol.0713169. [DOI] [PubMed] [Google Scholar]
- 35.Yamazaki T, Yang XO, Chung Y, Fukunaga A, Nurieva R, Pappu B, Martin-Orozco N, Kang HS, Ma L, Panopoulos AD, Craig S, Watowich SS, Jetten AM, Tian Q, Dong C. CCR6 regulates the migration of inflammatory and regulatory T cells. J Immunol. 2008;181:8391–8401. doi: 10.4049/jimmunol.181.12.8391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Beyersdorf N, Braun A, Vogtle T, Varga-Szabo D, Galdos RR, Kissler S, Kerkau T, Nieswandt B. STIM1-independent T cell development and effector function in vivo. J Immunol. 2009;182:3390–3397. doi: 10.4049/jimmunol.0802888. [DOI] [PubMed] [Google Scholar]
- 37.Baba Y, Nishida K, Fujii Y, Hirano T, Hikida M, Kurosaki T. Essential function for the calcium sensor STIM1 in mast cell activation and anaphylactic responses. Nat Immunol. 2008;9:81–88. doi: 10.1038/ni1546. [DOI] [PubMed] [Google Scholar]
- 38.Macian F. NFAT proteins: key regulators of T-cell development and function. Nat Rev Immunol. 2005;5:472–484. doi: 10.1038/nri1632. [DOI] [PubMed] [Google Scholar]
- 39.Gomez-Rodriguez J, Sahu N, Handon R, Davidson TS, Anderson SM, Kirby MR, August A, Schwartzberg PL. Differential expression of interleukin-17A and -17F is coupled to T cell receptor signaling via inducible T cell kinase. Immunity. 2009;31:587–597. doi: 10.1016/j.immuni.2009.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, Lucian L, To W, Kwan S, Churakova T, Zurawski S, Wiekowski M, Lira SA, Gorman D, Kastelein RA, Sedgwick JD. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature. 2003;421:744–748. doi: 10.1038/nature01355. [DOI] [PubMed] [Google Scholar]
- 41.Renno T, Krakowski M, Piccirillo C, Lin JY, Owens T. TNF-alpha expression by resident microglia and infiltrating leukocytes in the central nervous system of mice with experimental allergic encephalomyelitis. Regulation by Th1 cytokines. J Immunol. 1995;154:944–953. [PubMed] [Google Scholar]
- 42.Merrill JE, Kono DH, Clayton J, Ando DG, Hinton DR, Hofman FM. Inflammatory leukocytes and cytokines in the peptide-induced disease of experimental allergic encephalomyelitis in SJL and B10. PL mice. Proc Natl Acad Sci U S A. 1992;89:574–578. doi: 10.1073/pnas.89.2.574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kroenke MA, Carlson TJ, Andjelkovic AV, Segal BM. IL-12-and IL-23-modulated T cells induce distinct types of EAE based on histology, CNS chemokine profile, and response to cytokine inhibition. J Exp Med. 2008;205:1535–1541. doi: 10.1084/jem.20080159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Stromnes IM, Cerretti LM, Liggitt D, Harris RA, Goverman JM. Differential regulation of central nervous system autoimmunity by T(H)1 and T(H)17 cells. Nat Med. 2008;14:337–342. doi: 10.1038/nm1715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.El-Behi M, Rostami A, Ciric B. Current Views on the Roles of Th1 and Th17 Cells in Experimental Autoimmune Encephalomyelitis. J Neuroimmune Pharmacol. 2010 doi: 10.1007/s11481-009-9188-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fife BT, Huffnagle GB, Kuziel WA, Karpus WJ. CC chemokine receptor 2 is critical for induction of experimental autoimmune encephalomyelitis. J Exp Med. 2000;192:899–905. doi: 10.1084/jem.192.6.899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Izikson L, Klein RS, Charo IF, Weiner HL, Luster AD. Resistance to experimental autoimmune encephalomyelitis in mice lacking the CC chemokine receptor (CCR)2. J Exp Med. 2000;192:1075–1080. doi: 10.1084/jem.192.7.1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kuwabara T, Ishikawa F, Yasuda T, Aritomi K, Nakano H, Tanaka Y, Okada Y, Lipp M, Kakiuchi T. CCR7 ligands are required for development of experimental autoimmune encephalomyelitis through generating IL-23-dependent Th17 cells. J Immunol. 2009;183:2513–2521. doi: 10.4049/jimmunol.0800729. [DOI] [PubMed] [Google Scholar]
- 49.Korn T, Reddy J, Gao W, Bettelli E, Awasthi A, Petersen TR, Backstrom BT, Sobel RA, Wucherpfennig KW, Strom TB, Oukka M, Kuchroo VK. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nat Med. 2007;13:423–431. doi: 10.1038/nm1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Berna-Erro A, Braun A, Kraft R, Kleinschnitz C, Schuhmann MK, Stegner D, Wultsch T, Eilers J, Meuth SG, Stoll G, Nieswandt B. STIM2 regulates capacitive Ca2+ entry in neurons and plays a key role in hypoxic neuronal cell death. Sci Signal. 2009;2:ra67. doi: 10.1126/scisignal.2000522. [DOI] [PubMed] [Google Scholar]
- 51.Abdullaev IF, Bisaillon JM, Potier M, Gonzalez JC, Motiani RK, Trebak M. Stim1 and Orai1 mediate CRAC currents and store-operated calcium entry important for endothelial cell proliferation. Circ Res. 2008;103:1289–1299. doi: 10.1161/01.RES.0000338496.95579.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cioffi DL, Barry C, Stevens T. Store-operated calcium entry channels in pulmonary endothelium: the emerging story of TRPCS and Orai1. Adv Exp Med Biol. 2010;661:137–154. doi: 10.1007/978-1-60761-500-2_9. [DOI] [PubMed] [Google Scholar]
- 53.Ye J, Tsukamoto T, Sun A, Nigam SK. A role for intracellular calcium in tight junction reassembly after ATP depletion-repletion. Am J Physiol. 1999;277:F524–532. doi: 10.1152/ajprenal.1999.277.4.F524. [DOI] [PubMed] [Google Scholar]
- 54.Chandy KG, Wulff H, Beeton C, Pennington M, Gutman GA, Cahalan MD. K+ channels as targets for specific immunomodulation. Trends Pharmacol Sci. 2004;25:280–289. doi: 10.1016/j.tips.2004.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Reich EP, Cui L, Yang L, Pugliese-Sivo C, Golovko A, Petro M, Vassileva G, Chu I, Nomeir AA, Zhang LK, Liang X, Kozlowski JA, Narula SK, Zavodny PJ, Chou CC. Blocking ion channel KCNN4 alleviates the symptoms of experimental autoimmune encephalomyelitis in mice. Eur J Immunol. 2005;35:1027–1036. doi: 10.1002/eji.200425954. [DOI] [PubMed] [Google Scholar]
- 56.Rangaraju S, Chi V, Pennington MW, Chandy KG. Kv1. 3 potassium channels as a therapeutic target in multiple sclerosis. Expert Opin Ther Targets. 2009;13:909–924. doi: 10.1517/14728220903018957. [DOI] [PubMed] [Google Scholar]
- 57.Rudge P, Koetsier JC, Mertin J, Mispelblom Beyer JO, Van Walbeek HK, Clifford Jones R, Harrison J, Robinson K, Mellein B, Poole T, et al. Randomised double blind controlled trial of cyclosporin in multiple sclerosis. J Neurol Neurosurg Psychiatry. 1989;52:559–565. doi: 10.1136/jnnp.52.5.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Stromnes IM, Goverman JM. Active induction of experimental allergic encephalomyelitis. Nat Protoc. 2006;1:1810–1819. doi: 10.1038/nprot.2006.285. [DOI] [PubMed] [Google Scholar]
- 59.Collins MK, Tay CS, Erlebacher A. Dendritic cell entrapment within the pregnant uterus inhibits immune surveillance of the maternal/fetal interface in mice. J Clin Invest. 2009;119:2062–2073. doi: 10.1172/JCI38714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Feske S, Prakriya M, Rao A, Lewis RS. A severe defect in CRAC Ca2+ channel activation and altered K+ channel gating in T cells from immunodeficient patients. J Exp Med. 2005;202:651–662. doi: 10.1084/jem.20050687. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.