Abstract
Object
While deep brain stimulation (DBS) has proven to be an effective treatment for many symptoms of Parkinson’s disease (PD), a deterioration of axial symptoms frequently occurs, particularly for speech and swallowing. These unfavorable effects of DBS may depend on the site of stimulation. We made quantitative measures of jaw velocity to compare the relative effectiveness of DBS in the globus pallidus internus (GPi) or the subthalamic nucleus (STN).
Design
Randomized, double-blind, and longitudinal, with matched healthy control subjects
Methods
The peak velocities of self-scaled and externally-scaled jaw movements were studied in 27 PD patients before and after 6 months of bilateral DBS in the GPi or the STN. A mixed-effects model was used to identify differences in jaw velocity before DBS surgery (baseline) while off and on levodopa therapy, and after 6 months of DBS (postoperative) during four treatment conditions (off- and on-levodopa states with and without DBS).
Results
Self-scaled jaw velocity was impaired by the DBS procedure in STN; velocity was significantly decreased across all postoperative conditions compared to either the off- and on-levodopa baseline conditions. In contrast, the GPi group postoperatively was generally faster than the baseline off levodopa state. Turning the DBS off and on had no effect on jaw velocity in either group. Unlike baseline, levodopa therapy postoperatively no longer increased jaw velocity in either group, and this lack of effect was not related to postoperative changes in dose. The externally-scaled jaw velocity was little affected by PD, but DBS still slightly affected performance, with the STN group significantly slower than the GPi group for most conditions.
Conclusions
Our results suggest that either the electrode implant in STN or the subsequent period of continuous STN stimulation negatively affected voluntary jaw velocity, including the loss of the preoperative levodopa induced improvement. While the GPi group showed some improvement in voluntary jaw velocity postoperatively, their performance during the combination of DBS and levodopa was not different than their best medical management presurgery. The results have implications for DBS target selection, particularly for those patients with oromotor dysfunctions.
Keywords: Globus pallidus, subthalamic nucleus, mandible, speech
Introduction
Deep brain stimulation (DBS) in the globus pallidus internus (GPi) or the subthalamic nucleus (STN) is a widely used and accepted procedure for the treatment of medication-refractory Parkinson’s disease (PD), especially if the PD is severe or associated with complications due to long-term levodopa therapy.1,6,24,32,45 A recent, randomized clinical trial of bilateral DBS in GPi or STN revealed that after six months, DBS was more successful than the best medical management of PD,42 and that after two years, DBS in either site was similarly effective in the treatment of PD motor symptoms.9 However, DBS may not be an effective treatment for all PD motor symptoms. In particular, DBS in STN but not GPi has been associated with a progressive deterioration of axial symptoms, including speech and dysphagia.13,18,26,24,27,32,34,46 In contrast, other studies have found that bilateral DBS in STN can improve voluntary force of the lips and tongue10 and can improve the pharyngeal stage but not the oral stage of deglutition.3
The reasons for the adverse effects on speech and swallowing with DBS in STN are not yet clear. Speech and swallowing include many automatic muscle actions that are likely controlled by brain stem neurons. While both STN and GPi have reciprocal connections with the brain stem peduncolopontine nucleus (PPN), which plays a role in axial motor control, STN projects directly and via substantia nigra pars reticular (SNr) to the PPN.12,22,37 Models of PD also suggest a difference in function, with the GPi projections increasing inhibition and the STN projections providing excitatory input to PPN.22,28
Previous studies showed that PD results in decrements in jaw movement17,30 and that DBS can affect jaw velocity, although differential effects of DBS site are not known.31 The purpose of this study was to examine the effects of DBS site on bradykinetic jaw movements that are either self- or externally-scaled in PD patients.
Methods
Study Population
Twenty-seven patients with idiopathic PD participated. A movement disorders specialist (author PH) selected the PD subjects, who were recruited through the Portland and Seattle Veterans Administration Medical Centers (VAMC) and Oregon Health & Science University (OHSU), according to the inclusion and exclusion criteria of a large multi-center clinical trial (Veterans Affairs Cooperative Studies Program #468).9,42 Table 1 shows the demographics of the PD subjects.
TABLE 1.
| Demographics | STN | GPi |
|---|---|---|
| Number | 14 | 13 |
| Males | 13 | 12 |
| Age (yrs)* | 63.8±6.3 | 65.5±8.6 |
| PD duration (yrs)* | 16.8±6.2 | 15.1±10.2 |
| Hoehn/Yahr* | ||
| Off levodopa | 3.6±0.7 | 3.5±0.8 |
| On levodopa | 2.3±0.7 | 2.8±0.6 |
| Levodopa equivalents (mg)* | 1289±652 | 1306±667 |
Values represent means ± standard deviation
We also tested 27 age-gender matched (mean±SD: 63.9±7.4 yrs) control subjects who had no history or symptoms of neurological disease. All PD and control subjects had adequate natural or restored dentition and no tooth or jaw pain. All subjects signed an informed consent for the protocols, which were approved by the VAMC and OHSU Institutional Review Boards, and are in agreement with the Declaration of Helsinki.
Mandibular movement testing
Mandibular kinematics were obtained using a kinesiograph (Model K7, MyoTronics, Tukwila, WA). The kinematics were obtained while the subjects performed oromotor tasks while sitting upright in a dental chair, looking straight ahead, with minimum rotation of the neck. The kinesiography electrically tracked a 5 × 10 mm magnet that was taped securely at the midline of the chin about 1 cm below the lower lip. The three-dimensional coordinates of the mandible were determined by changes in the electromagnetic fields using a pair of magnet sensors mounted on tight-fitting eyeglass frames worn by the subject. The kinesiograph has a linear response (±10%) between 5 and 45 mm for vertical and horizontal movements, as calibrated by an Optotrack Motion Analysis System (Northern Digital, Waterloo, Canada).
Data were collected during self-scaled (voluntary) and externally-scaled (automatic) jaw movement tasks. Examples of the vertical velocities and positions for both tasks are shown in Figures 1A and 2A. For the self-scaled task, the subjects were instructed to open and close their mouth at their own initiation, amplitude, and pace. The main dependent variables were the peak opening and closing velocities, with data collected for five opening-closing cycles per subject.
Fig. 1.
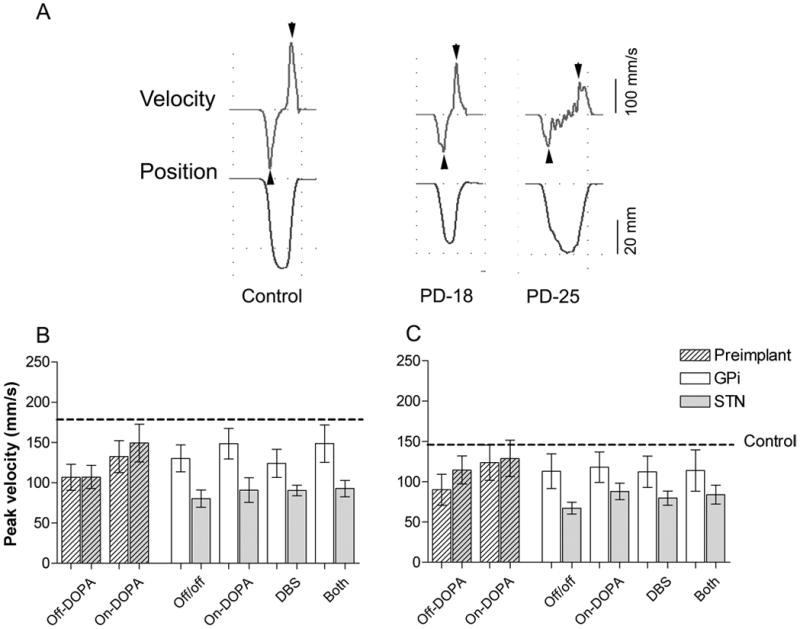
Self-scaled opening and closing jaw movements. A. The vertical velocity (upper trace) and vertical position (lower trace) kinematics during a single opening (down) and closing (up) jaw movement for a representative control subject and two representative PD subjects, one of whom (PD-25) had jaw tremors. Arrowheads indicate peak velocity. Histograms of the mean± standard error of the peak opening (B) and closing (C) velocities for the GPi and STN groups during various treatment conditions before and after DBS implant. Dashed line: control mean (open: 180±8.3; close: 149±9.2 mm/s).
Fig. 2.
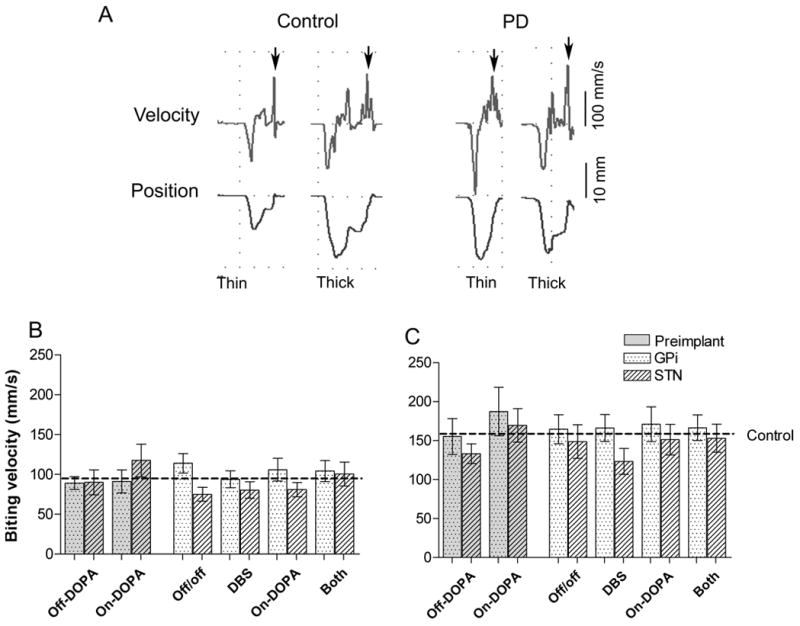
Externally-scaled jaw movements during the carrot biting task. A. The vertical velocity (upper trace) and vertical position (lower trace) kinematics elicited by thin and thick carrot stimuli for a representative control and PD subject. The velocity and vertical amplitude kinematics fluctuate during the positioning of the carrot stimulus on the molar surface prior to the breakage of the carrot piece, which is evident by the last major peak velocity (arrow) preceding the return of vertical position to baseline. Histograms of the mean± standard error of the biting velocity for the thin (B) and thick (C) stimuli for the GPi and STN groups during various treatment conditions before and after the DBS electrode implant. Jaw opening: down; jaw closing: up. Dashed line: control mean (thin: 96.7±5.5; thick: 159±6.8 mm/s)
For the externally-scaled task, jaw velocities were scaled based on peripheral sensory information from the size of food between the teeth. This task was based on a study29 that showed the thickness and not the hardness of a food sample significantly influenced the biting velocity (i.e., a thick food sample elicits a faster biting velocity than a thin sample). The subjects were instructed to bite on 1.5 × 1.5 cm pieces of raw carrots that differed only in thickness: 1.5 cm (thick) and 0.5 cm (thin). Five thick and five thin food stimuli were given in a random order. The experimenter placed the food stimulus on the subject’s tongue, such that the subject did not see or feel the food stimulus before it was placed in the mouth. The subjects were required to move the food stimulus with their tongue onto their molar teeth of the preferred chewing side, bite through the food stimulus in a single, ballistic motion, and then expelled the food stimulus into a paper cup. The dependent variable was the biting velocity.
Surgery
The PD subjects were assigned randomly (following a double-blind method) into either STN (N=14) or GPi (N=13) groups; the PD subjects and experimenters remained unaware of the stimulation site for the duration of the multi-center study. The surgical procedure was done by a neurosurgeon (author KB) with extensive experience with DBS. The PD subjects underwent a bilateral surgical implantation of DBS electrodes (Medtronic, 3387) within one month of site assignment. Electrodes were inserted bilaterally through two pre-coronal sulcus burr holes. The initial electrode-tip targets were based on standard coordinates referenced to the mid-point of the anterior and posterior commissures (STN: x=12, y=−4, z=−4 mm; GPi: x=20–21, y=2, z=−4 mm). Intra-operative microelectrode recordings were performed to confirm target localization, and corrections to the implant site were made accordingly. One week after electrode implantation, a single- (Medtronic, Soletra) or dual-channel (Medtronic, Kinetra) internal pulse generator (IPG) was surgically implanted in the infra-clavicular area.
After the DBS stimulator was turned on, a movement disorders neurologist (PH) periodically adjusted the IPG parameters to empirically achieve optimized PD symptom control and an absence of marked side effects, as well as made appropriate changes in the PD pharmacologic therapy. The mean amplitude of the DBS was 3.28 V (range 2.2 to 4.4 V), with 70% of the subjects having a 90 μm pulse width (60, 120, or 150 μm pulse widths were used in two subjects each), and at a rate of 185 Hz for 71% of the subjects (the rest of the subjects were evenly divided between 130 and 150 Hz). The antiparkinsonian medications were converted to the Levodopa Equivalent Daily Dose (LEDD) according procedures of Nutt et al.25
Procedure
Oromotor testing was done within one month before and six months after the bilateral electrode implants. The PD subjects were assessed using the motor Unified Parkinson’s Disease Rating Scale (UPDRS III)7 and the H&Y scale.15 For the preimplant, baseline testing, the PD subjects were evaluated after a 10 to 12 hr overnight withdrawal (practical off) of antiparkinsonian medications (Off-DOPA), and then again 45 to 60 minutes after taking their levodopa medication(On-DOPA). Each subject self-rated their On-DOPA state, and in a few cases an additional dose of levodopa was required to achieve a 70–100% on-state. At 6 months postoperatively, the PD subjects were tested during four different treatment conditions on the same oromotor tasks. Following the overnight medication withdrawal, the subjects were randomly assigned either to testing first with the DBS turned on (DBS) or off (Off/off), and then tested the second time with the reverse treatment conditions. A 30 minute rest period was given between turning on or off the DBS. The subjects then took their levodopa medicationand after 45 to 60 minutes were tested a third time according to a random schedule of either levodopa only (On-DOPA) or the combination of levodopa and DBS (BOTH). After a rest period, the treatment conditions were reversed and the fourth oromotor testing was done. All testing was completed within two hours after the PD subjects received their levodopa medication. Neither the subject nor the experimenter knew the stimulation conditions, except when subjects had obvious tremor suppression with DBS.
As a control for possible fatigue or learning effects, all control subjects had two test sessions (comparable to the off- and on-levodopa therapy for the PD subjects) and 14 control subjects did four repetitions of the tasks (comparable to the four postoperative treatment conditions of the PD subjects). No significant differences were evident in the jaw velocities between the two repetitions or among the four repetitions of tasks.
Statistical analysis
A linear mixed-model was developed to determine the effects of the following factors: group (control, STN or GPi), DBS (turned off or on), levodopa state (Off-DOPA or On-DOPA), and DBS surgery (baseline or postoperative) on the dependent variables (self-scaled opening and closing peak jaw velocities, and externally-scaled jaw biting peak velocity). To account for between-subject variability, subject number was a random effects factor in the model. The distributions of the dependent measures, for each group in each condition, when log-transformed were not different to normal (by the Shapiro-Wilk test) and the variance was homogenous (by Levene’s test). An auto-regressive covariance structure was used, since it provided the lowest Bayesian Information criterion for the model.44
Contrast tests within the mixed-model were made of hypotheses related to five key questions: (1) Are the baseline jaw velocities of the PD subjects while off- and on-levodopa different than the control group, and does levodopa increase jaw velocity? (2) Is there a “DBS procedure effect” (surgery and 6 month of DBS) where the baseline jaw velocity while Off-DOPA is different than the postoperative Off/off condition, and is any difference related to the STN or GPi implant sites? (3) Does DBS in STN or GPi without levodopa therapy change jaw velocity in comparison to the Off/off condition? (4) Are there differences in the jaw velocities in terms of the usual PD therapeutic treatments between baseline On-DOPA condition and the 6 month postoperative combination of DBS in STN or GPi and levodopa therapy (BOTH)? (5) Does the DBS in STN or GPi change the therapeutic effectiveness of levodopa on jaw velocity? For this comparison, LEDD was a covariate to compare baseline levodopa induced improvement in velocity (Off-DOPA versus On-DOPA) to postoperative levodopa improvement (Off/off versus On-DOPA). T-tests were used for selected comparisons, and non-parametric comparisons were made of the secondary dependent measures of the facial and speech items of the UPDRS-III, which did not have a normal distribution. The statistical analyses were performed using SAS 9.2 (SAS Institute Inc., Cary, NC). The data are reported as means ± standard error.
Results
Baseline
No significant baseline differences were found between the STN and GPi groups in the demographics and clinical measures (Tables 1 and 2). The 27 PD subjects showed a highly significant (P<0.01) improvement in their baseline UPDRS-III total motor scores between off and on levodopa treatment, with similar improvement for both the STN (P<0.01) and GPi (P<0.01) groups.
Table 2.
| Site | Baseline | 6-mo Postoperative | ||||
|---|---|---|---|---|---|---|
| Off-DOPA | On-DOPA | Off/off | On-DOPA | DBS | Both | |
| UPDRS Total Motor | ||||||
| STN | 49.4±3.4 | 25.8±3.7 | 58.3±3.4 | 35.2±4.5 | 33.3±4.8 | 20.2±2.3 |
| GPi | 42.7±4.2 | 22.7±3.6 | 41.3±3.2 | 26.3±2.5 | 29.0±2.0 | 19.8±2.7 |
| UPDRS Speech (Item # 18) | ||||||
| STN | 1.5±0.2 | 0.6±0.1 | 2.1±0.2 | 1.2±0.2 | 1.6±0.2 | 1.4±0.2 |
| GPi | 1.3±0.2 | 1.1±0.2 | 1.8±0.2 | 1.2±0.2 | 1.5±0.2 | 1.4±0.2 |
| UPDRS Facial expression (Item # 19) | ||||||
| STN | 1.9±0.2 | 0.7±0.1 | 2.4±0.3 | 1.4±0.3 | 1.5±0.2 | 1.0±0.2 |
| GPi | 1.7±0.3 | 1.2±0.3 | 2.2±0.2 | 1.3±0.2 | 1.8±0.3 | 1.1±0.3 |
Self-scaled velocity
At baseline, both the STN and GPi groups were significantly slower (P<0.01) than the control group, with the opening and closing peak velocities of the STN and GPi groups were not significantly different in the Off-DOPA state (Fig. 1). Levodopa therapy significantly increased the opening velocity for both the STN and GPi groups (P<0.01 and P<0.05, respectively), although the On-DOPA level for GPi group was still significantly slower (P<0.01) than the control level. Levodopa therapy also significantly increased the closing velocity but only for the GPi group (P<0.01), which resulted in the closing velocities of both groups in the On-DOPA state being similar to the control subjects.
A marked, negative DBS procedure effect was evident at the 6-month postoperative test (Fig. 1). Compared to the baseline Off-DOPA condition, the STN group had significantly slower opening (P<0.01) and closing (P<0.01) velocities during all postoperative treatment conditions. The STN group during the Off/off condition had a 25% decrease in opening velocity and 41% decrease in closing velocity compared to the baseline Off-DOPA condition, both of which were significantly (P<0.01) different from the baseline. The opposite effect was evident during the Off/off condition for the GPi group, which showed a 21% increased opening velocity and a 25% increased closing velocity compared to the baseline Off-DOPA condition. Both opening and closing improvements were significant (P<0.05) for the GPi group.
At the 6-month test, levodopa therapy no longer produced a significant improvement for both sites. The lack of significant postoperative improvement with levodopa therapy may not necessarily be related to postoperative decrease in medication. The STN group had a significant (P<0.05, paired t test) reduction of 371 mg LEDD at 6-months from the baseline level, whereas the GPi group had a non-significant (P=0.13, paired t tests) average increase of 236 mg LEDD from the baseline. LEDD was used as a covariate in the mixed model and the comparison between the Off/off and On-DOPA conditions postoperatively revealed no significant change for either jaw opening (p=0.12) or closing (P=0.74) for either DBS site. This suggests that the reduced LEDD in the STN group at the 6-month test was not responsible for the reduced effects of levodopa therapy.
Since most PD patients typically receive a combination of DBS and levodopa therapy (the BOTH condition), we compared the effectiveness of the best medical management at baseline (On-DOPA) with the postoperative BOTH condition. For the STN group, the opening velocity during the BOTH condition was significantly (P<0.05) slower than the baseline On-DOPA condition, whereas the GPi group showed no difference between conditions (P=0.73). Similar results were evident for the jaw closing velocity.
Externally-scaled biting velocity
The kinematics of the externally-scaled task showed an initial fast opening velocity, followed by fluctuations in position and velocity, which were associated with the placement of the food stimulus on the molars, and a final, consistent peak biting velocity that occurred just before molar occlusion (Fig. 2A). The biting velocity was regulated by the thickness of food stimulus, with the thick stimulus eliciting a significantly faster biting velocity than the thin stimulus for all groups (control group: 69% faster, P<0.01; GPi group: 77% faster, P<0.01; and STN group: 48% faster, P<0.01, paired t tests).
The PD had no affect on the baseline elicited velocities, with no significant differences among the control, STN, or GPi groups for either the thin or thick elicited velocities. However, compared Off-DOPA, baseline levodopa therapy significantly (P<0.05) increased the thin stimulus elicited biting velocity in the STN group and increased the thick stimulus elicited velocity for the both GPi (P<0.05) and the STN (P<0.01) groups. At the 6-month test, similar to the self-scaled jaw velocity, the externally-scaled velocity was significantly worsened by the DBS procedure in STN but not in GPi. The STN group had significantly (P<0.01) slower velocities than the GPi group across treatment conditions, whereas the GPi group had a significantly (P<0.01) faster thin-elicited velocity during Off/off versus the baseline Off-DOPA conditions. However, neither the thick- nor the thin-elicited velocities showed any significant differences among the postoperative conditions for either the STN or GPi groups. This included the loss of the baseline positive effects of the On-DOPA state. For best medical management, a comparison between the baseline On-DOPA and the postoperative BOTH conditions also did not reveal any significantly differences between groups for either the thick or thin stimuli.
UPDRS-III
Table 2 shows the UPDRS measurements for the signs and symptoms of PD for total motor disability and for the subscores of speech and facial expression for the two stimulation sites during all treatment conditions. The UPDRS total motor scores revealed that the STN and GPi groups were not significantly different at baseline while either off or on levodopa (P=0.11 and P=0.47, respectively), and that levodopa significantly (P<0.001) improved the scores of both groups. A comparison between the baseline Off-DOPA and postoperative Off/off conditions revealed a non-significant (P=0.075)14% decrement in the motor score for the STN group, whereas the GPi group showed a 3% improvement. The STN and GPi groups were similar for the other postoperative treatment conditions. Both STN and GPi groups, in comparison to the Off/off condition, showed significant (P<0.01) improvement during DBS, On-DOPA, or BOTH conditions. However, in terms of best therapy, no significant differences were evident between the baseline On-DOPA and the postoperative BOTH conditions for either STN (P=0.214) or GPi (P=0.613).
While no UPDRS-III item is a specific measure of mandibular motor control, some indication of mandibular function is reflected in the scores for the speech and facial expression items. At baseline, the scores for both speech and facial expression were not significantly different between the STN and GPi groups while Off-DOPA, but when on levodopa therapy, only the STN group showed a significantly improved in speech (P<0.01, Mann-Whitney) and facial expression (P<0.001). Postoperatively, the speech and facial expression scores during Off/off condition for both the STN and GPi groups were slightly worse than during the baseline Off-DOPA condition, although these baseline-postoperative differences were not significant. Compared to the Off/off condition, DBS in STN produced a significant (P<0.05, Mann-Whitney) improvement in the facial expression, and levodopa therapy in the STN group also significantly improved both speech (P<0.05) and facial expression (P<0.05). However, in terms of best therapy, the speech scores for the STN group during the postoperative BOTH condition were significantly (P<0.05, Mann-Whitney) worse than during the baseline On-DOPA condition. For the GPi group, no significant changes were found between the Off/off condition and either the DBS or the On-DOPA conditions; no significant differences occurred between the BOTH and the baseline On-DOPA conditions.
Discussion
The major finding of this study was that DBS in STN resulted in a decreased in voluntary jaw opening and closing velocities across all postoperative treatment conditions and that the decrease was unrelated to the levodopa state. Conversely, the GPi group showed improvements postoperatively in voluntary jaw movement for most conditions relative to the preoperative baseline conditions. The externally-scaled task, which was little affected by PD, showed some effects of the DBS procedure on the elicited velocity that were generally similar to the effects on the voluntary jaw velocity. Postoperatively, the STN group had a slower elicited velocity than the control and GPi groups, the GPi group showed a slight increase in velocity, and the levodopa induced increase in velocity during baseline was lost at the 6-month test.
The results of this project are strengthened by the double-blinded protocol that was stringently followed throughout the many years of the study; by the dependent measure of peak jaw velocity, which could be precisely and objectively assessed during repeated testing and during two different tasks; and by the random assignment of the treatment conditions. The self-and externally-scaled jaw movements were well practiced, everyday activities, which minimized possible effects of learning, and the self-scaled tasks had no specific criterion accuracy, which reduced issues about possible error corrections. The PD and control subjects also did not know what were the dependent measures of the tasks, which lessen possible placebo effects. However, the self- and externally-scaled jaw tasks may not be as distinct as initially considered, since jaw opening and closing requires the coordinated excitation and inhibition of the same group of craniomandibular muscles that are controlled directly and indirectly by cortical and brain stem neurons and regulated by the synchronous activation of a large number sensory fibers.23 The experimental design may also not have allowed sufficient time between turning on and off the DBS, given the delayed response of various symptoms with DBS range from almost immediate to hours.39
An initial concern was whether the STN and GPi groups were truly comparable preoperatively, and then whether there was a general overall change in one group more than the other postoperatively. The total UPDRS motor scores showed that STN and GPi groups while off and on levodopa were similar preoperatively, but the two groups appeared different at six month postoperatively during the Off/off condition, with the STN group having a 14% decline and the GPi group with a 3% improvement in their PD motor scores when compared to the baseline Off-DOPA condition. Although this baseline-postoperative difference for STN group did not reach significance, it suggests that the electrode implant procedure or six months of electrical stimulation had negatively affected the STN group more than the GPi group. A multi-center study also found a significant decline in the UPDRS motor scores for the DBS in STN and an improvement for the GPi group when comparing the baseline Off-DOPA and 24 month postoperative Off/off conditions (Follett et al., 2010). Moro et al., (2010) also reported differences between STN and GPi groups at 3 month and 5–6 years postoperatively, with a slight decline in STN scores and considerable improve in GPi scores.
In the present study, the STN and GPi groups had generally similar UPDRS motor scores during the DBS, On-DOPA and BOTH postoperative conditions, and, in comparison to the Off/off condition, had improved motor scores during DBS and levodopa therapy, which was consistent with previous work.9,24 In contrast to the UPDRS motor scores, however, the voluntary jaw velocity in the STN group was slower postoperatively than the baseline not only during the Off/off condition but during all postoperative conditions; further neither the DBS nor levodopa therapy increased the jaw velocity. This suggests jaw velocity is affected in ways that are not reflected in the general UPDRS motor scores.
Since most postoperative conditions showed a similar jaw velocity, there are several possible explanations for the results. The decline in postoperative jaw velocity in the STN group may be related to a progressive deterioration of the PD, which was also evident in a 15% decline in the total motor UPDRS scores for this group while off levodopa. While no control PD group was used to measure disease progression without DBS treatment, disease progression seems unlikely to account for all the changes, since the GPi group showed either no change or an improved jaw velocity and little change in their total motor UPDRS scores. Another possibility is the spread of DBS current to adjacent corticofugal fibers. Klostermann et al.18 suggested that current spread with DBS in STN might account for a decrease in speech velocity. Tripoliti et al.40 also showed that speech intelligibility can deteriorate during high voltage (4V) DBS in STN, which was suggested to be due to current spread to nearby pathways. In this study, however, current spread is an unlikely major factor, since turning the DBS on and off with or without levodopa had no significant effect on the jaw movements, even though turning on DBS did improve the UPDRS total scores for both groups.
Another possible explanation for the postoperative decline in the STN group is that the surgical implant of the DBS electrodes and the microelectrode recordings produced a microlesion, with transitory change in symptoms.5,19 However, other research has shown improvements in motor scores without medications that occurred immediately after surgery were still present six months later.21 Okun et al.26 suggested that an STN lesion could account for a worsening of verbal fluency in PD patients, since the decline in postoperative performance at seven months postoperatively appeared to be independent of DBS or the levodopa. St. George et al. (unpublished data)in a postural control study, which included some of the same PD subjects as the present study, also found a decrement in posture at six months postoperative in the STN, but not the GPi, group.
The relatively small size of the STN, which is much smaller than the GPi, and the existence of multiple fiber pathways within the STN and in the adjacent internal capsule have been noted as a possible limitation of DBS in STN.41 The postmortem studies of DBS electrode implants in various structures reveal a consistent picture of a relatively small tissue damage (astrogliosis, tissue vacuolation, and some cell loss) surrounding the electrodes and usually within 1 mm of the electrode tip.11,14,16 Further, neurophysiological mapping of the STN in PD patients receiving DBS reveals that the representations of face and the oromandibular muscles were in the middle of the dorsolateral region with the arm and leg representations located slightly ventral.33 Consequently, if a lesion of the STN produced the decrement in jaw velocity, then it is surprising that the lesion did not also affect the areas representing the extremities.35 Unfortunately, data on the localization of electrode position within the nuclei were not available for our subjects.
The lack of effect of DBS or levodopa after surgery may be due to effects of long-term electrical stimulation that continue after the DBS is turned off. A recent study of STN unit activity in PD patients receiving therapeutic parameters of DBS revealed that about 50% of the neurons changed their spontaneous firing pattern even when the DBS was terminated;2 other human and animal studies using different parameters of stimulation have reported changes of STN neuronal activity after the end of stimulation.8,43 Long-term effects of DBS in STN may also occur in the anterograde- or retrograde-activated structures. For example, the unit activity in the SNr of patients receiving DBS in STN was reduced with a change in the pattern of activity when the DBS was turned off,20 which corresponds to inhibition of SNr neurons in the rat after high-frequency STN stimulation.38 Thus, continuous DBS may be changing the long-term, functional dynamics of the basal ganglia and its target structures,2 although additional studies are needed where unit activity is analyzed after months of continuous DBS and where any change in unit activity is correlated specifically to changes in jaw velocity.
A major part of the experimental design of this study was to differentiate changes in jaw bradykinesia during self-scaled versus externally-scaled jaw movements. Our hypothesis was that the self-scaled movements likely involve basal ganglia connections to the cortex whereas the externally-scaled movements are driven by peripheral feedback involving the brain stem. Unpredictably, PD affected only the voluntary jaw velocity, with no effect on the externally-scaled jaw velocity. However, levodopa treatment significantly increased both self- and externally-scaled jaw velocities, even when the baseline jaw velocity was within normal limits. DBS in either STN or GPi produced similar effects for both self- and externally-scaled jaw velocities, although the magnitude of the effect was much greater for the self-scaled velocity. The similarities of the levodopa and DBS effects suggest that both tasks involve some of the same anatomical circuits. Consequently, even though the two sizes of the food stimuli elicited significantly different biting velocities, it is likely that biting the food stimulus included some cortical control, which would account for the similarity of effects.
Jaw velocity is only one parameter of the complex coordination of face, oral, pharyngeal, and respiratory muscle groups that occur during speech and various oromotor actions. Thus, jaw velocity can be part of a diverse range of symptoms that frequently occur with PD related speech impairments.36 However, in the present study, the clinical UPDRS subscores for speech and facial expression provided little insight to the role of jaw velocity in their dysfunction. While baseline subscores were similar for the STN and GPi groups while off levodopa, the STN group had significant improvement for both speech and facial expression items while On-DOPA, even though the random assignment of patients to the stimulation sites should have minimized any response differences between the groups. Postoperatively, both groups tended to worsen on the items during the Off/off condition than during the preoperative Off-DOPA condition, whereas the jaw velocity was only worse for the STN group. Also, unlike the results for jaw velocity, the speech and facial expression subscores improved for the STN group during levodopa treatment compared to Off/off condition, and the speech subscore improved while on DBS. However, similar to jaw velocity, the speech subscore during the combination of the DBS and levodopa for the STN group was worse than baseline On-DOPA condition. This clinical result is consistent with previous studies, where DBS in STN while on medication has been associated speech impairment or an exacerbation of a pre-DBS speech disorder,24,32,34 although the underlying mechanisms have not been identified.
How the effects of DBS on jaw velocity relate to clinical measures of speech and oromotor is not yet clear, although Tripoliti et al.40 have suggested slowed jaw movements is one of several altered neuromuscular systems that can lead to imprecise articulation. However, it should come as no surprise that DBS may have differential effects among the participating oromotor neuromuscular systems. While this study showed a decline in jaw velocity (increased bradykinesia) associated with DBS in STN, other work has shown an improvement of the acoustic parameters related to glottal vibration and vocal tremor,4 and an increase in force production in the lips and tongue,10 although no comparisons were made in these studies with preimplant, baseline levels. The challenge of future studies will be to identify the DBS targets and stimulation parameters that can best treat the uniquely affected oromotor impairment of each PD patient.
Conclusion
DBS in STN, but not in GPi, significantly worsened jaw velocity six months after surgery. This result is consistent with clinical speech impairments, as well as worsening of postural control after six months of DBS in the STN (unpublished data). Our results suggest that either the electrode implant in STN or the subsequent period of continuous STN stimulation negatively affected voluntary jaw velocity, including the loss of the preoperative levodopa induced improvement. These results suggest that patients presenting with oral, speech or swallowing dysfunction might be better suited for DBS in GPi than in STN, especially if the dysfunction is related to mandibular motor control. Unlike the STN group, the GPi group showed some improvement in voluntary jaw velocity postoperatively, with similar performance while on DBS and levodopa, which corresponds to the performance evident during best medical management presurgically.
Acknowledgments
This project was supported by the National Institute on Aging grant AG19706 and in part by the Parkinson’s Alliance.
We would like to thank Triana Nagel for subject scheduling and data collection, and Ngoc Nguyen and Dina Medhi for assistance with data entry.
References
- 1.Benabid A, Chabardés S, Seigneuret E. Deep-brain stimulation in Parkinson’s disease: long-term efficacy and safety What happened this year? Cur Opin Neurol. 2005;18:623–630. doi: 10.1097/01.wco.0000186839.53807.93. [DOI] [PubMed] [Google Scholar]
- 2.Carlson JD, Cleary DR, Cetas JS, Heinricher MM, Burchiel KJ. Deep brain stimulation does not silence neurons in subthalamic nucleus in Parkinson’s patients. J Neurophysiol. 2010;103:962–967. doi: 10.1152/jn.00363.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ciucci MR, Barkmeier-Kraemer JM, Scherman SJ. Subthalamic nucleus deep brain stimulation improves deglutition in Parkinson’s disease. Mov Disord. 2008;23:676–683. doi: 10.1002/mds.21891. [DOI] [PubMed] [Google Scholar]
- 4.D’Alatri L, Paludetti G, Contarino MF, Galla S, Marchese MR, Bentivoglio AR. Effects of bilateral subthalamic nucleus stimulation and medication on parkinsonian speech impairment. J Voice. 22:365–372. 200. doi: 10.1016/j.jvoice.2006.10.010. [DOI] [PubMed] [Google Scholar]
- 5.Deuschl G, Herzog J, Kleiner-Fisman G, Kubu C, Lozano AM, Lyons KE, et al. Deep brain stimulation: postoperative issues. Mov Disord Suppl. 2006;14:S219–237. doi: 10.1002/mds.20957. [DOI] [PubMed] [Google Scholar]
- 6.Deuschl G, Schade-Brittinger C, Krack P, Volkmann J, Schäfer H, Bötzel K, et al. A randomized trial of deep-brain stimulation for Parkinson’s disease. N Engl J Med. 2006;355:896–908. doi: 10.1056/NEJMoa060281. [DOI] [PubMed] [Google Scholar]
- 7.Fahn S, Elton R. Members of the UPDRS development committee: Unified Parkinson’s Disease Rating Scale. In: Fahn S, Marsden C, Calne D, Goldstein M, editors. Recent Developments in Parkinson’s disease. Florham Park, NJ: Macmillan Healthcare Information; 1987. pp. 153–164. [Google Scholar]
- 8.Filali M, Hutchison WD, Palter VN, Lozano AM, Dostrovsky JO. Stimulation-induced inhibition of neuronal firing in human subthalamic nucleus. Exp Brain Res. 2004;156:274–281. doi: 10.1007/s00221-003-1784-y. [DOI] [PubMed] [Google Scholar]
- 9.Follett KA, Weaver FM, Stern M, Hur K, Harris CL, Luo P, et al. Pallidal versus subthalamic deep-brain stimulation for Parkinson’s disease. N Engl J Med. 2010;362:2077–2091. doi: 10.1056/NEJMoa0907083. [DOI] [PubMed] [Google Scholar]
- 10.Gentil M, Garcia-Ruiz P, Benebid A-L. Effects of bilateral deep-brain stimulation on oral control of patients with Parkinsonism. Eur Neurol. 2000;44:147–152. doi: 10.1159/000008224. [DOI] [PubMed] [Google Scholar]
- 11.Haberler C, Alesch F, Mazal PR, Pilz P, Jellinger K, Pinter MM, et al. No tissue damage by chronic deep brain stimulation in Parkinson’s disease. Ann Neurol. 2000;48:372–376. [PubMed] [Google Scholar]
- 12.Hammond C, Rouzaire-Dubois B, Feger J, Jackson A, Crossman AR. Anatomical and electrophysiological studies on the reciprocal projections between the subthalamic nucleus and nucleus tegmenti pedunculopontinus in the rat. Neuroscience. 1983;9:41–52. doi: 10.1016/0306-4522(83)90045-3. [DOI] [PubMed] [Google Scholar]
- 13.Hariz MI, Rehncrona S, Quinn NP, Speelman JD, Wensing C. Multicenter study on deep brain stimulation in Parkinson’s disease: an independent assessment of reported adverse events at 4 years. Mov Disord. 2008;23:416–421. doi: 10.1002/mds.21888. [DOI] [PubMed] [Google Scholar]
- 14.Henderson JM, Pell M, O’Sullivan DJ, McCusker EA, Fung VS, Hedges P, et al. Postmortem analysis of bilateral subthalamic electrode implants in Parkinson’s disease. Mov Disord. 2002;17 :133–137. doi: 10.1002/mds.1261. [DOI] [PubMed] [Google Scholar]
- 15.Hoehn MM, Yahr MD. Parkinsonism: onset, progression and mortality. Neurology. 1967;17:427–442. doi: 10.1212/wnl.17.5.427. [DOI] [PubMed] [Google Scholar]
- 16.Jarraya B, Bonnet AM, Duyckaerts C, Houeto JL, Cornu P, Hauw JJ, et al. Parkinson’s disease, subthalamic stimulation, and selection of candidates: a pathological study. Mov Disord. 2003;18:1517–1520. doi: 10.1002/mds.10607. [DOI] [PubMed] [Google Scholar]
- 17.Karlsson S, Persson M, Johnels B. Levodopa induced ON-OFF motor fluctuations in Parkinson’s disease related to rhythmical masticatory jaw movements. J Neurol Neurosurg Psychiatry. 1992;55:304–307. doi: 10.1136/jnnp.55.4.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Klostermann F, Ehlen F, Vesper J, Nubel K, Gross M, Marzinzik F, et al. Effects of subthalamic deep brain stimulation on dysarthrophonia in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2008;79:522–529. doi: 10.1136/jnnp.2007.123323. [DOI] [PubMed] [Google Scholar]
- 19.Maltête D, Derrey S, Chastan N, Debono B, Gérardin E, Fréger P, et al. Microsubthalamotomy: an immediate predictor of long-term subthalamic stimulation efficacy in Parkinson disease. Mov Disord. 2008;23:1047–1050. doi: 10.1002/mds.22054. [DOI] [PubMed] [Google Scholar]
- 20.Maltête D, Jodoin N, Karachi C, Houeto JL, Navarro S, Cornu P, et al. Subthalamic stimulation and neuronal activity in the substantia nigra in Parkinson’s disease. J Neurophysiol. 2007;97:4017–4022. doi: 10.1152/jn.01104.2006. [DOI] [PubMed] [Google Scholar]
- 21.Mann JM, Foote KD, Garvan CW, Fernandez HH, Jacobson CE, 4th, Rodriguez RL, et al. Brain penetration effects of microelectrodes and DBS leads in STN or GPi. J Neurol Neurosurg Psychiatry. 2009;80:794–797. doi: 10.1136/jnnp.2008.159558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mena-Segovia J, Bolam JP, Magill PJ. Pedunculopontine nucleus and basal ganglia: distant relatives or part of the same family. Trends Neurosci. 2004;27:585–588. doi: 10.1016/j.tins.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 23.Miller AJ. Craniomandibular muscles: Their role in function and form. Boca Raton, FL: CPR Press; 2000. [Google Scholar]
- 24.Moro E, Lozano AM, Pollak P, Agid Y, Rehncrona S, Volkmann J, et al. Long-term results of a multicenter study on subthalamic and pallidal stimulation in Parkinson’s disease. Mov Disord. 2010;25:578–86. doi: 10.1002/mds.22735. [DOI] [PubMed] [Google Scholar]
- 25.Nutt JG, Burchiel KJ, Comella CL, Jankovic J, Lang AE, Laws ER, Jr, et al. Randomized, double-blind trial of glial cell line-derived neurotrophic factor (GDNF) in PD. Neurology. 2003;60 :69–73. doi: 10.1212/wnl.60.1.69. [DOI] [PubMed] [Google Scholar]
- 26.Okun MS, Fernandez HH, Wu SS, Kirsch-Darrow L, Bowers D, Bova F, et al. Cognition and mood in Parkinson’s disease in subthalamic nucleus versus globus pallidus interna deep brain stimulation: the COMPARE trial. Ann Neurol. 2009;65:586–595. doi: 10.1002/ana.21596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Østergaard K, Sunde NA. Evolution of Parkinson’s disease during 4 years of bilateral deep brain stimulation of the subthalamic nucleus. Mov Disord. 2006;21:624–631. doi: 10.1002/mds.20776. [DOI] [PubMed] [Google Scholar]
- 28.Pahapill PA, Lozano AM. The pedunculopontine nucleus and Parkinson’s disease. Brain. 2000;123 (Pt 9):1767–1783. doi: 10.1093/brain/123.9.1767. [DOI] [PubMed] [Google Scholar]
- 29.Peyron MA, Maskawi K, Woda A, Tanguay R, Lund JP. Effects of food texture and sample thickness on mandibular movement and hardness assessment during biting in man. J Dent Res. 1997;76:789–795. doi: 10.1177/00220345970760031201. [DOI] [PubMed] [Google Scholar]
- 30.Robertson LT, Hammerstad JP. Jaw movement dysfunction related to Parkinson’s disease and partially modified by levodopa. J Neurol Neurosurg Psychiatry. 1996;60:41–50. doi: 10.1136/jnnp.60.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Robertson LT, Horak FB, Anderson VC, Burchiel KJ, Hammerstad JP. Assessments of axial motor control during deep brain stimulation in parkinsonian patients. Neurosurgery. 2001;48:544–551. doi: 10.1097/00006123-200103000-00017. [DOI] [PubMed] [Google Scholar]
- 32.Rodriguez-Oroz MC, Obeso JA, Lang AE, Houeto JL, Pollak P, Rehncrona S, et al. Bilateral deep brain stimulation in Parkinson’s disease: a multicentre study with 4 years follow-up. Brain. 2005;128(Pt 10):2240–2249. doi: 10.1093/brain/awh571. [DOI] [PubMed] [Google Scholar]
- 33.Rodriguez-Oroz MC, Rodriguez M, Guridi J, Mewes K, Chockkman V, Vitek J, et al. The subthalamic nucleus in Parkinson’s disease: somatotopic organization and physiological characteristics. Brain. 2001;124(Pt 9):1777–1790. doi: 10.1093/brain/124.9.1777. [DOI] [PubMed] [Google Scholar]
- 34.Romito LM, Contarino MF, Vanacore N, Bentivoglio AR, Scerrati M, Albanese A. Replacement of dopaminergic medication with subthalamic nucleus stimulation in Parkinson’s disease: long-term observation. Mov Disord. 2009;24:557–63. doi: 10.1002/mds.22390. [DOI] [PubMed] [Google Scholar]
- 35.Santens P, De Letter M, Van Borsel J, De Reuck J, Caemaert Lateralized effects of subthalamic nucleus stimulation on different aspects of speech in Parkinson’s disease. Brain Lang. 2003;87:253–258. doi: 10.1016/s0093-934x(03)00142-1. [DOI] [PubMed] [Google Scholar]
- 36.Sapir S, Pawlas AA, Ramig LO. Voice and speech abnormalities in Parkinson’s disease: Relation to severity of motor impairment, duration of disease, medication, depression, gender, and age. J Med Speech-Lang Path. 2001;9:213–226. [Google Scholar]
- 37.Semba K, Fibiger HC. Afferent connections of the laterodorsal and the pedunculopontine tegmental nuclei in the rat: a retro- and antero-grade transport and immunohistochemical study. J Comp Neurol. 1992;323:387–410. doi: 10.1002/cne.903230307. [DOI] [PubMed] [Google Scholar]
- 38.Shen KZ, Johnson SW. Complex EPSCs evoked in substantia nigra reticulata neurons are disrupted by repetitive stimulation of the subthalamic nucleus. Synapse. 2008;62:237–242. doi: 10.1002/syn.20488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Temperli P, Ghika J, Villemure JG, Burkhard PR, Bogousslavsky J, Vingerhoets FJ. How do parkinsonian signs return after discontinuation of subthalamic DBS? Neurology. 2003;60:78–81. doi: 10.1212/wnl.60.1.78. [DOI] [PubMed] [Google Scholar]
- 40.Tripoliti E, Zrinzo L, Martinez-Torres I, Tisch S, Frost E, Borrell E, et al. Effects of contact location and voltage amplitude on speech and movement in bilateral subthalamic nucleus deep brain stimulation. Mov Disord. 2008;23:2377–2383. doi: 10.1002/mds.22296. [DOI] [PubMed] [Google Scholar]
- 41.Walter BL, Vitek JL. Surgical treatment for Parkinson’s disease. Lancet Neurol. 2004;3:719–728. doi: 10.1016/S1474-4422(04)00934-2. [DOI] [PubMed] [Google Scholar]
- 42.Weaver FM, Follett K, Stern M, Hur K, Harris C, Marks WJ, Jr, et al. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. CSP 468 Study Group. JAMA. 2009;301:63–73. doi: 10.1001/jama.2008.929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Welter ML, Houeto JL, Bonnet AM, Bejjani PB, Mesnage V, Dormont D, et al. Effects of high-frequency stimulation on subthalamic neuronal activity in parkinsonian patients. Arch Neurol. 2004;61:89–96. doi: 10.1001/archneur.61.1.89. [DOI] [PubMed] [Google Scholar]
- 44.West BT, Welch KB, Galecki AT. Linear Mixed Models: A practical guide using statistical software. Boca Raton, FL: Chapman & Hall/CRC Press; 2007. [Google Scholar]
- 45.Williams A, Gill S, Varma T, Jenkinson C, Quinn N, Mitchell R, et al. Deep brain stimulation plus best medical therapy versus best medical therapy alone for advanced Parkinson’s disease (PD SURG trial): a randomised, open-label trial. Lancet Neurol. 2010;9:581–591. doi: 10.1016/S1474-4422(10)70093-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zahodne LB, Okum MS, Foote KD, Fernandez HH, Rodriguez RL, Wu SS, et al. Greater improvement in quality of life following unilateral deep brain stimulation in the globus plallidus as compared to the subthalamic nucleus. J Neurol. 2009;256:1321–1329. doi: 10.1007/s00415-009-5121-7. [DOI] [PMC free article] [PubMed] [Google Scholar]


