Synopsis
Previous studies have shown that the presence of one P450 enzyme can affect the function of another P450. The goal of this study was to determine if P450 enzymes are capable of forming homomeric complexes that affect P450 function. To address this problem, the catalytic activities of several P450s were examined in reconstituted systems containing NADPH-cytochrome P450 reductase (POR) and a single P450. CYP2B4-, CYP2E1-, and CYP1A2-mediated activities were measured as a function of POR concentration using reconstituted systems containing different concentrations of P450. Although CYP2B4-dependent activities could be explained by a simple Michaelis Menten interaction between POR and CYP2B4, both CYP2E1 and CYP1A2 activities generally produced a sigmoidal response as a function of [POR]. Interestingly, the non-Michaelis behavior of CYP1A2 could be converted to a simple mass-action response by increasing the ionic strength of the buffer. Next, physical interactions between CYP1A2 enzymes were demonstrated in reconstituted systems by chemical crosslinking and in cellular systems by bioluminescence resonance energy transfer (BRET). Crosslinking data were consistent with the kinetic responses in that both were similarly modulated by increasing the ionic strength of the surrounding solution. Taken together, these results show that CYP1A2 forms CYP1A2•CYP1A2 complexes that exhibit altered catalytic activity.
Keywords: Cytochrome P450, Homomeric P450•P450 complexes, Protein-protein interactions, enzyme kinetics, Protein cross-linking, Bioluminescence resonance energy transfer
INTRODUCTION
Cytochrome P450 plays a major role in the metabolism of a wide variety of drugs and other xenobiotics as well as numerous endogenous compounds. Although the interactions between NADPH-cytochrome P450 reductase (POR) and P450 necessary for monooxygenase function are well characterized [1-6], the potential for P450 enzymes to interact with each other has only been examined more recently [7-10]. Many of these P450•P450 interactions have been shown to significantly alter monooxygenase function.
Several laboratories have now reported that one P450 enzyme can influence the behavior of another P450 [7-15]. Our laboratory demonstrated that the catalytic activity of CYP2B4-dependent 7-pentoxyresorufin (7PR) metabolism was inhibited by the addition of CYP1A2 to the POR/CYP2B4 reconstituted system [16]. Inhibition of this CYP2B4 activity by CYP1A2 was dependent on the substrate used [8;16], was more pronounced when POR was subsaturating, and appeared to be related to a high affinity association between CYP1A2 and POR. When CYP1A2 function was examined in the same reconstituted systems, the presence of CYP2B4 synergistically stimulated CYP1A2 activities. Again, the response was more pronounced at subsaturating POR concentrations.
Subsequent reports showed that this interaction was not simply competition between CYP1A2 and CYP2B4 for POR, but actually involved formation of a CYP1A2•CYP2B4 complex [7;11;13]. In mixed reconstituted systems (containing CYP1A2, CYP2B4 and POR), POR appears to bind with high affinity to the CYP1A2 moiety of the CYP1A2•CYP2B4 complex, whereas the POR•CYP1A2 complex has a much lower binding affinity in simple reconstituted systems containing a single P450 [6;7;11]. These results support the concept that the catalytic behavior of a P450 enzyme is significantly influenced by the presence of another P450.
P450•P450 complexes were also reported for CYP1A2•CYP2E1. Again the CYP1A2•CYP2E1 complex bound POR with high affinity [12]. B o th CYP1A2•CYP2E1 complexes [12], and the CYP1A2•CYP2B4 complexes mentioned above [11] were sensitive to alterations in ionic strength, as the inhibitory/synergistic effects on enzyme activities were relieved using higher concentrations of HEPES buffer. These data are consistent with the heteromeric P450•P450 complexes being stabilized by electrostatic interactions, where an increase in the ionic strength of the surrounding solvent is capable of disrupting these P450•P450 complexes. Interestingly, evidence for significant interactions that affected monooxygenase function was not found with mixed reconstituted systems containing CYP2B4 and CYP2E1 [12]. Taken together from this limited data set, P450•P450 interactions capable of influencing monooxygenase function were observed when CYP1A2 was one of the protein components, but were less likely when this P450 was not involved.
The above results are consistent with CYP1A2 facilitating the formation of P450•P450 complexes. This raises the question, “If CYP1A2 supports the formation of heteromeric P450 complexes, then is CYP1A2 capable of forming complexes with other CYP1A2 enzymes?” Despite its simplicity, little is known about the potential for a particular P450 enzyme to interact with itself within a simple reconstituted system containing POR and a single P450. The goal of this study is to determine if CYP1A2 is capable of forming homomeric complexes that influence P450 function. The results support the idea that homomeric CYP1A2 complexes do exist, and are consistent with the concept that at least some P450 enzymes exist in the endoplasmic reticulum as dimers or higher order complexes.
EXPERIMENTAL
Chemicals
Dilauroylphosphatidylcholine (DLPC), 7-ethoxyresorufin (7-ER), resorufin, nicotinamide adenine dinucleotide phosphate reduced tetrasodium salt (NADPH), glucose-6-phosphate, glucose-6-phosphate dehydrogenase and formaldehyde were purchased from Sigma-Aldrich (St. Louis, MO). Benzphetamine (BZP) was a gift from Upjohn (Kalamazoo, MI). 7-ethoxy-4-trifluoromethylcoumarin (7-EFC), and 7-hydroxy-4-trifluoromethylcoumarin (7-HFC) were obtained from Molecular Probes (Eugene, OR). Escherichia coli C41 cells were purchased through Avidis SA (Biopole Clermont-Limagne, France).
HEK293T/17 cells (CRL-11268) were purchased from ATCC (Manassas, VA). Lipofectamine 2000, Dulbecco's Modified Eagle Medium (DMEM), and phosphate-buffered saline (PBS) were purchased from Invitrogen (Carlsbad, CA). Coelenterazine 400a was purchased from Gold Biotechnology (St. Louis, MO), and coelenterazine h was purchased from Promega (Madison, WI). The BRET vectors were obtained from Perkin Elmer (Waltham, MA). BRET measurements were performed on a TriStar LB 941 plate reader (Berthold Technologies, Bad Wildbad, Germany).
Enzymes
Cytochrome P450 2B4 (CYP2B4) was expressed in Escherichia coli C41 and purified according to standard procedures with minor modifications [17]. Recombinant rabbit NADPH cytochrome P450 reductase (plasmid: pSC-POR, provided by Lucy Waskell (Univ. Michigan); constructed from plasmid pCWori-rabbit POR, utilizing a T7 promoter) was expressed in Escherichia coli C41, solubilized and purified according to a modification of previously described methods [18;19]. CYP1A2 was isolated from βNF treated rabbit liver microsomes as previously described [20]. CYP2E1 was expressed and purified according to [21].
Measurement of Monooxygenase Activities
Catalytic activities of CYP1A2, CYP2E1 and CYP2B4 were determined using reconstituted systems where POR and CYP1A2, CYP2E1 or CYP2B4 were combined in DLPC. The reconstituted systems were preincubated at room temperature for 2 h before the addition of the other assay components [22]. After preincubation, the enzyme activities were measured at 37°C. All fluorimetric products were monitored on an SpectraMax M5 plate reader (Molecular Devices, Sunnyvale, CA).
For 7-ethoxyresorufin-O-dealkylation (EROD) activity, the final assay conditions were: the reconstituted system [containing varying concentrations of P450 (ranging from 0.025 μM to 0.2 μM), varying concentrations of POR (ranging from 0.025 μM to 0.4 μM) and 8 μM DLPC], and 4.5 μM substrate, in 50 mM HEPES buffer (pH 7.5). Reactions were initiated by the addition of NADPH to a final concentration of 0.5 mM. The accumulation of resorufin was monitored fluorometrically at an excitation wavelength of 530 nm and an emission wavelength of 585 nm. A hydroxyresorufin standard curve was calculated each time to determine the level of product formation [23;24].
For 7-EFC O-deethylation, the final assay conditions were: the reconstituted system [as described in the preceding paragraph], and substrate (0.1 mM 7-EFC), in 100 mM potassium phosphate buffer (pH 7.25). Reactions were initiated by the addition of NADPH to a final concentration of 0.5 mM. The fluorescent product, 7-HFC, was monitored over a 2-min time course using excitation/emission wavelength of 410 nm/510 nm [25;26].
Demethylation of p-nitroanisole (pNA) was examined by directly measuring the formation of the product p-nitrophenol at 405 nm. The final assay conditions were: the reconstituted system [as described above], and pNA (0.4 mM), in 100 mM potassium phosphate buffer (pH 7.25). Reactions were initiated by the addition of NADPH to a final concentration of 0.5 mM. Formation of p-nitrophenol was determined using an extinction coefficient of 12.9 mM-1(cm)-1 as described [27;28].
A fluorometric method was used to determine BZP and N-nitrosodimethylamine (NDMA) demethylation activity [29;30]. The final assay conditions were: the reconstituted system [containing P450 (ranging from 0.05 μM to 0.4 μM), POR (ranging from 0.05 μM to 0.8 μM), 16 μM DLPC, as described above], and substrate (either 1 mM benzphetamine or 2 mM NDMA) in 100 mM potassium phosphate buffer (pH 7.25). Reactions were initiated by the addition of NADPH to a final concentration of 0.5 mM. After incubation at 37°C for times ranging from 0-8 min, the reaction was terminated with 0.11 volumes of a mixture of 25% ZnSO4 (w/v) and 0.5 M semicarbazide (10:1). The samples were combined with 0.1 volumes of saturated Ba(OH)2, placed on ice for 10 min, and then centrifuged at 14,000 g for 10 min. An aliquot (0.5 ml) of supernatant was mixed with 0.5 ml of Nash reagent [0.3% (v/v) acetic acid, 0.2% (v/v) acetylacetone, 15.4% (w/v) ammonium acetate]. The formaldehyde product was measured fluorometrically (excitation wavelength 410 nm, emission wavelength 510 nm) and was quantified using a standard curve. The initial rates were determined from the time course.
Modeling of the data collected from enzymatic assays
The kinetic data for the hyperbolic curves were fit using a global analysis of a mass action binding of POR and P450 using the Dynafit program (Biokin, Ltd.; Pullman, WA), which applies the Levenberg-Marquardt algorithm to generate a least squares nonlinear regression fit of the data. The data were fit using a single KD for the POR•P450 complex and a single kcat for each activity, according to the following equations.
| Equation 1 |
| Equation 2 |
Where “A” represents the P450 enzyme, and “R” represents POR. KAR is the dissociation constant for formation of the POR•P450 complex, kj is the kcat for the reaction, and “P” represents the product formed. Each of the assays described in this manuscript was performed under conditions of saturating substrate concentration; therefore, A and AR actually represent the substrate bound forms of these enzymes, with the assumption being that once product is released from the enzyme, it is rapidly replaced by another substrate molecule. The kcat value is a characteristic of the ability of the substrate-bound AR complex to turnover a substrate and generate product. This value will vary for different substrates. The Dynafit program used is included in Supplemental Data (Program A).
The Dynafit program determines the KAR and kcat values that provide the best fit of the data at all of the P450 concentrations (i.e. all of the curves within a Figure), assuming a Michaelis Menten type model for substrate binding and catalysis. Consequently, the data analysis generates a single KAR and kcat value, where the only difference among the curves is the P450 concentration.
Demonstration of CYP1A2•CYP1A2 complexes by crosslinking with BS3
Reconstituted systems of DLPC, CYP1A2, and/or POR were prepared as described above. The conditions used for these reconstituted systems were selected to assess the aggregation of enzymes (by chemical cross-linking) at different concentrations of POR, salt, and lipid. Before incubating at room temperature for 2 hr, all reconstituted systems, containing variable amounts of enzymes and lipid as described in Results, were diluted to a final P450 (or POR, if P450 was not present in the system) concentration of 11.5 μM with buffer containing 50 mM HEPES (pH 7.4), 100 mM NaCl, 5 mM EDTA, and 20 % glycerol (v/v). The samples were stirred briefly with a 10 μL pipet tip before incubating for 2 hr at room temperature. After the 2 hr incubation, 10 pmol of P450 from each reconstituted system was diluted to a volume of 48 μL with 50 mM HEPES buffer (pH 7.5). The incubations without CYP1A2 contained 0.2 μM POR and 32 μM DLPC. The samples were heated to 37°C for 5 min before cross linking was initiated with 2 μL of 6.25 mM aqueous bis(sulfosuccinimidyl) suberate (BS3) (Pierce-Thermo, Rockford, IL). After adding BS3, the samples were stirred briefly with a pipet tip and incubated at 37°C for 20 sec. The reactions were terminated with 25 μL of 3x loading buffer containing 75 mM Tris (pH 6.8), 9 % sodium dodecyl sulfate (SDS) (w/v), 0.015 % bromophenol blue, 36 % sucrose, 750 mM glycine, and 1 M NaCl. The loading buffer was heated at 37°C for 30 min before use to dissolve the buffer components. Immediately after adding the 3x loading buffer, the samples were vortexed and immersed in ice for at least 15 min and stored at 4°C. The protein samples were denatured by the addition of β-mercaptoethanol, boiled for 30 min; and 15 μL of each was loaded in individual lanes of a 4-12% SDS-polyacrylamide gel. Polyacrylamide gel electrophoresis (PAGE) was performed at 4°C by initially running the gel at 50 volts for 30 min. After the 50 volt run, the samples were run for an additional 1.5 hr at 200 volts. Gels were then transferred to nitrocellulose membranes, and immune blotted using anti-CYP1A2 and anti-POR.
Detection of CYP1A2•CYP1A2 complexes using Bioluminescence Resonance Energy Transfer
Vectors for bioluminescence resonance energy transfer (BRET) assays were generated by ligating rabbit CYP1A2 cDNA into pRluc-N2 and pGFP2-N1 vectors. A vector for the expression of unlabeled CYP1A2 was generated by inserting a stop codon into the pGFP2-N1/CYP1A2 vector using site-directed mutagenesis. Both Rluc and GFP constructs were fused to the C-terminal region of the CYP1A2 gene in order to minimize interference with membrane binding of CYP1A2.
HEK293T/17 cells were maintained in DMEM supplemented with 10% fetal bovine serum under a humidified 37°C atmosphere of air +5% CO2. One day prior to transfection, cells were seeded onto 6-well plates at 7.5 × 105 cells/well. Cells were transiently transfected with Lipofectamine 2000, and media was replaced with fresh DMEM 4 hours after transfection.
Twenty-four hours after transfection, cells were washed and resuspended in 750 μl PBS per well. Aliquots (100 μl/well) of the suspension were pipeted into both a black, clear-bottom 96-well plate for GFP quantification and a white, opaque 96-well plate for Rluc quantification and BRET measurement. The BRET ratio was determined by measuring the ratio of GFP fluorescence (at 510 nm) to Rluc luminescence (at 410 nm) immediately following the addition of coelenterazine 400a to a final concentration of 5 μM. BRET measurement was performed on three wells for each sample. Untransfected cells were used to normalize GFP and Rluc signals, and the resulting BRET ratio for each sample was normalized to that of cells expressing the Rluc construct alone.
The ratio of GFP- to Rluc-tagged protein was determined by comparing the GFP fluorescence and coelenterazine h-induced Rluc luminescence to that of a GFP-Rluc fusion protein, which was assumed to express GFP and Rluc at a 1:1 ratio. GFP fluorescence was measured at 510 nm with a SpectraMax M5 plate reader. Rluc expression levels were determined by measuring coelenterazine h-induced luminescence in a TriStar LB 941 plate reader at 480 nm.
RESULTS
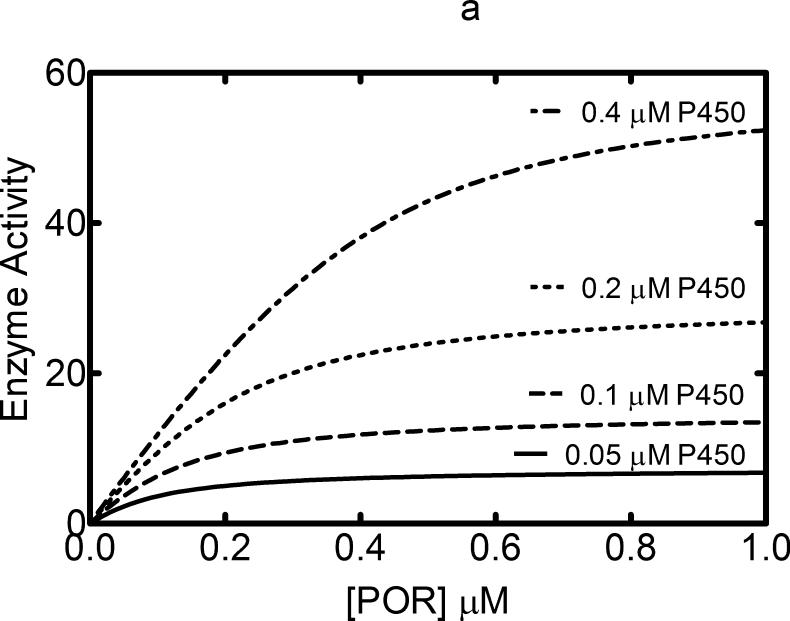
Formation of homomeric P450 complexes would be a function not only of the affinity of the proteins for each other, but also of the concentration of the P450s (i.e. more complexation would be expected at higher P450 concentrations due to more protein crowding in the membrane). Therefore, the behavior of a P450 that does not form complexes can be readily predicted theoretically. If a P450 enzyme-mediated catalytic activity is measured as a function of POR, classical hyperbolic curves are expected at each P450 concentration if the activity follows Michaelis-Menten kinetics (Figure 1a). As the P450 concentration is increased, there is a shift to the right in the POR concentration necessary to achieve saturation, and an increase in the maximal activity that is proportional to the P450 concentration. However, if P450s exist as a complex, and either the activity of the P450•P450 complex or its affinity for POR differs from that of the P450 monomer, then deviations from this predicted behavior would be expected.
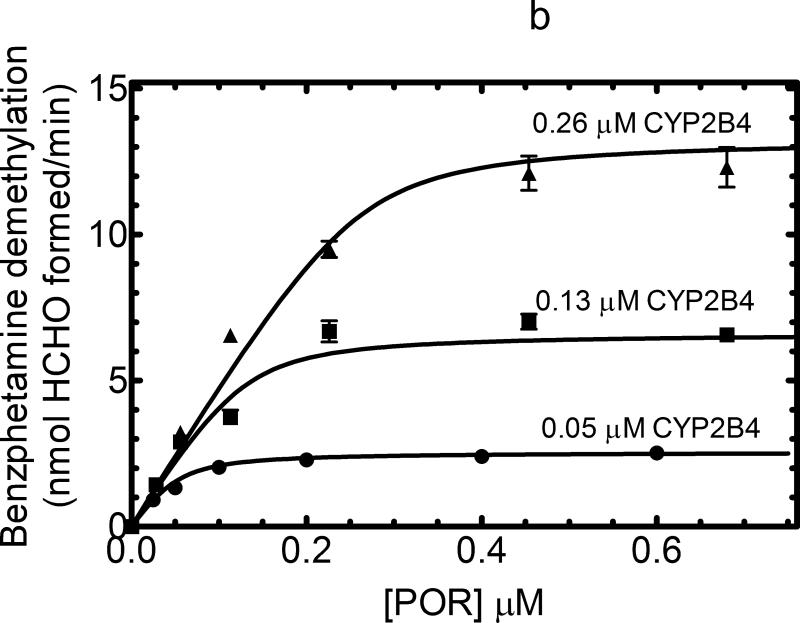
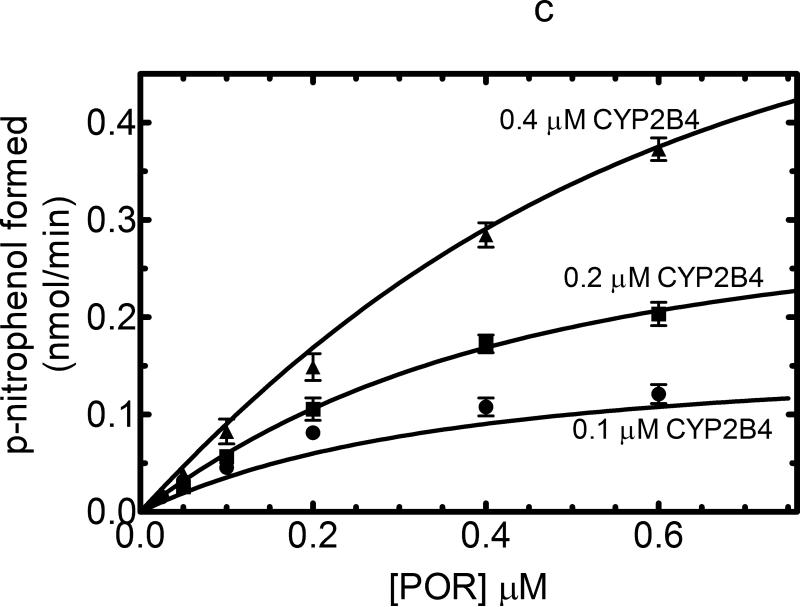
Figure 1. Effect of CYP2B4 concentration on the formation of homomeric P450•P450 complexes.
(A) The theoretical model of describing the titration of a P450 enzyme activity as a function of POR at a series of P450 concentrations. A P450 protein that does not form homomeric complexes and interacts with POR based on simple mass-action behavior, the changes in activity can be predicted according to the Michaelis Menten saturation curves shown. The hyperbolic nature of the curves will be unaffected by the concentration of P450. (B) The effect of CYP2B4 concentration on CYP2B4-mediated benzphetamine metabolism as a function of POR concentration. CYP2B4-mediated benzphetamine demethylation was measured as a function of POR concentration using reconstituted systems containing POR (ranging from 0 – 0.7 μM) and CYP2B4 (ranging from 0.05 – 0.4 μM) in 32 μM DLPC. The data were fit to a model assuming a simple mass-action interaction between POR and CYP2B4 generating the curves shown. The kinetic constants derived from the best-fit were KDapp = 0.013 μM and kcat = 51 nmol (min)-1(nmol CYP2B4)-1. The data represent the mean ± SEM for 4 determinations. (C) The effect of CYP2B4 concentration of CYP2B4-mediated p-nitroanisole demethylation as a function of POR concentration. The metabolism of p-nitroanisole was examined under similar conditions as described in (B). Again, the data was fit to a simple mass action model with a KDapp = 0.29 μM and kcat = 1.7 nmol (min)-1(nmol CYP2B4)-1. The data represent the mean ± SEM for 3 determinations.
The ability of three different rabbit P450 enzymes, CYP2B4, CYP2E1, and CYP1A2, were examined to determine if these proteins formed kinetically-detectable complexes. When examined as a function of POR concentration, CYP2B4-dependent benzphetamine N-demethylation produced a hyperbolic response as expected for a simple interaction between POR and P450 monomers (Figure 1b). All three curves can be fit to a typical Michaelis function having a KD of 0.013 μM for the POR•CYP2B4 complex and a kcat of 51 nmol (min)-1 (nmol CYP2B4)-1. Qualitatively similar results were obtained when the substrate p-nitroanisole (pNA) was used (Figure 1c). CYP2B4-dependent pNA demethylation also showed results consistent with a typical Michaelis Menten reaction between monomeric POR and CYP2B4; however, the KD for the POR•CYP2B4 complex was 20-fold larger than that found for benzphetamine. The data do not provide any evidence for the formation of CYP2B4•CYP2B4 complexes that affect the function of this enzyme. However, these results demonstrate that substrate can have a profound effect on the ability of a P450 to interact with POR [31;32].
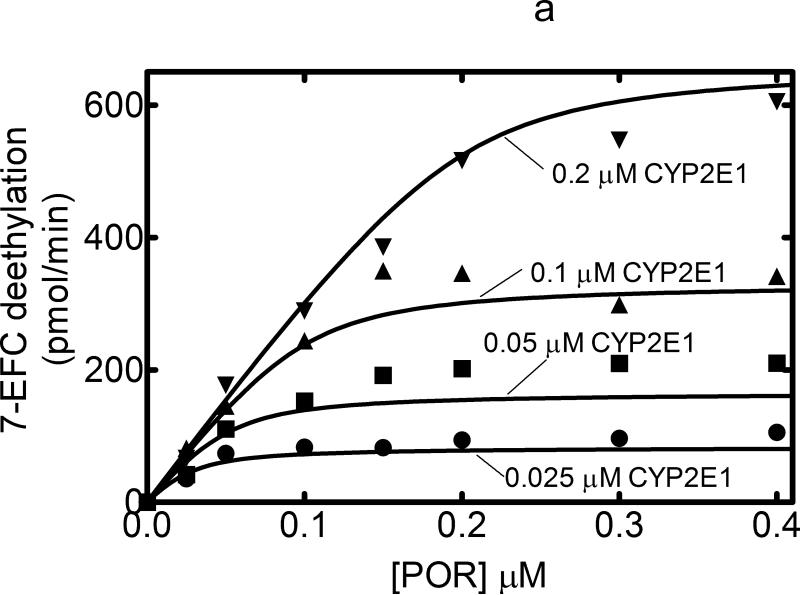
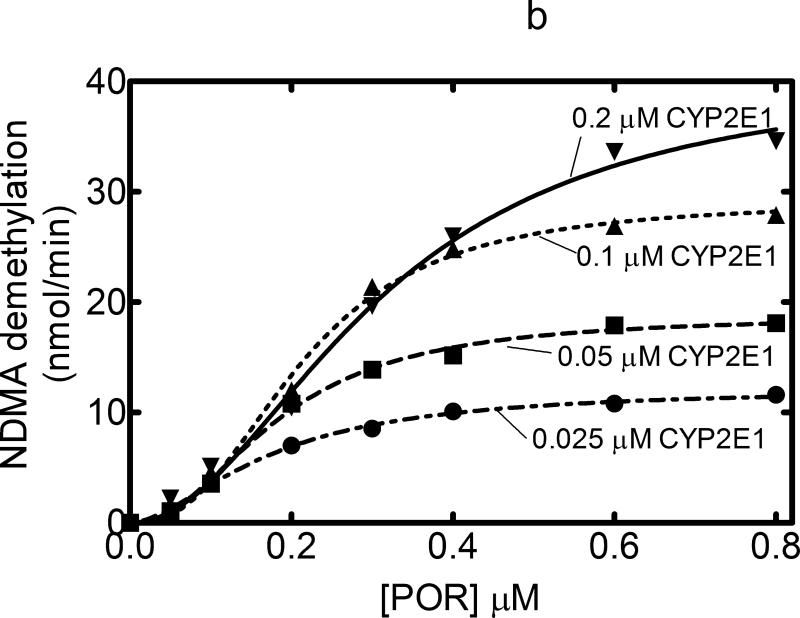
CYP2E1-dependent 7-EFC O-deethylation activity measured as a function of POR concentration also yielded typical hyperbolic curves with a KD equal to 0.011 μM (Figure 2a). It was consistent with simple interaction between monomeric CYP2E1 and POR expected for Michaelis Menten enzymes, suggesting that any CYP2E1•CYP2E1 complexes that may be formed do not appear to alter POR binding to the P450s. In contrast, a different response was observed when CYP2E1-mediated NDMA demethylation was examined. Rather than the hyperbolic response observed with 7-EFC demethylation with CYP2E1 and with the CYP2B4-mediated reactions, NDMA demethylation was shown to generate a sigmoidal response as a function of POR concentration (Figure 2b). Interestingly, at low POR concentrations, the overall reaction rate was independent of CYP2E1 concentration. This lack of a hyperbolic response suggests that a more complex interaction between POR and CYP2E1, and the type of response is sensitive to the substrate used.
Figure 2. Measurement of CYP2E1 activities as a function of [POR]: Effect of CYP2E1 concentration.
Titration curves for CYP2E1-dependent activities were measured as a function of [POR] to estimate the ability of POR to associate with CYP2E1. These titration curves were measured at several different CYP2E1 concentrations (0.025, 0.05, 0.1, and 0.2 μM). Reconstituted systems were prepared as described in Experimental and Figure 1. (A) Effect on 7-ethoxytrifluorocoumarin metabolism. The 7-EFC deethylation data was fit to a simple Michaelis Menten model with the KDPOR = 0.011 μM and a kcat = 3.31 nmol (min)-1(nmol CYP2E1)-1. (B) Effect on N-nitrosodimethylamine demethylation. POR titrations of this CYP2E1-dependent activity generated results that were better fit to a sigmoidal curve than to a simple Michaelis Menten model (r2 = 0.994 vs. 0.97, respectively, at 0.2 μM CYP2E1).
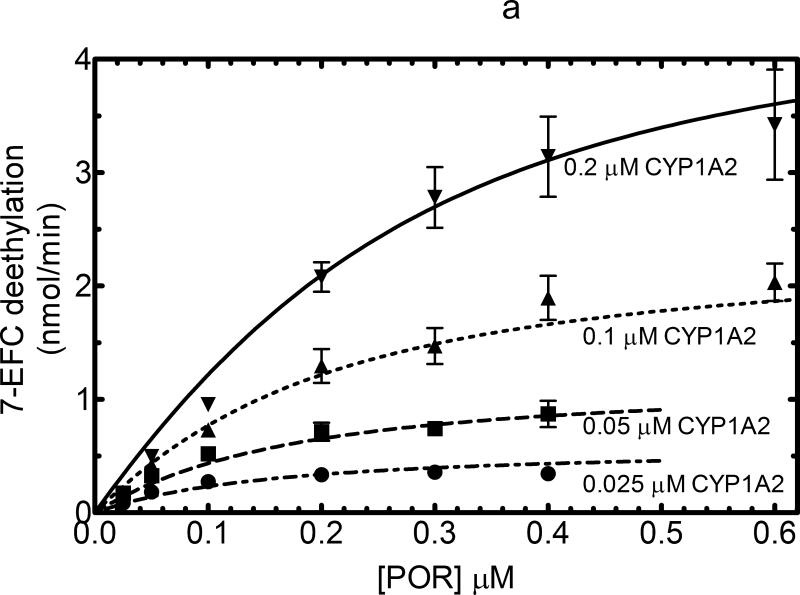
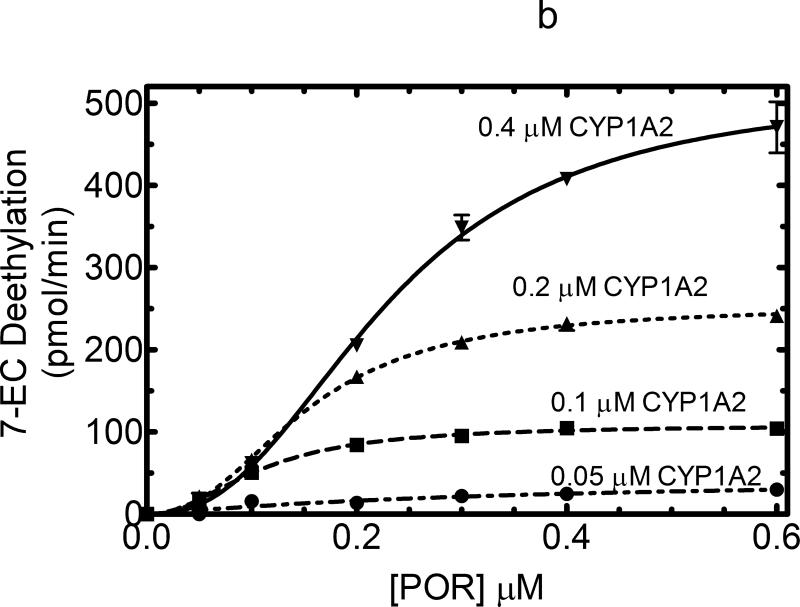
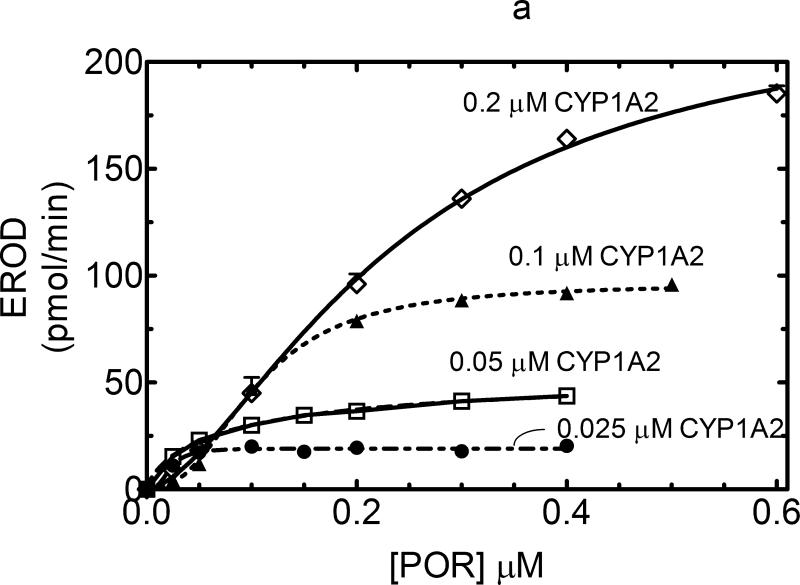
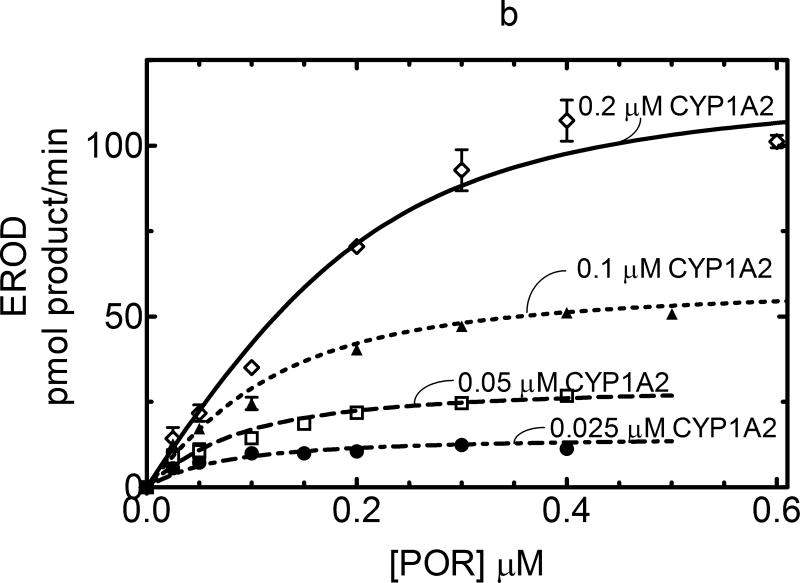
The responsiveness of CYP1A2 as a function of POR concentration was similar to that observed with CYP2E1. 7-EFC metabolism produced a hyperbolic response that was proportional to the P450 concentration and was consistent with a simple mass action binding of POR to CYP1A2 (Figure 3a). The KD for this process was found to be 0.012μM. In contrast, CYP1A2-dependent 7-ethoxycoumarin O-dealkylation (7-EC) activity exhibited a sigmoidal response, suggesting that a more complex interaction than can be explained by a simple mass action interaction of POR with monomeric CYP1A2 must be invoked to explain the data (Figure 3b). A similar sigmoidal response was observed when 7-ethoxyresorufin demethylation was examined (Figure 4a). Interestingly, at lower concentrations of CYP1A2 (0.025 and 0.05 μM), the catalytic activities as a function of POR concentration yielded hyperbolic curves, consistent with simple interaction between monomeric CYP1A2 and POR expected for Michaelis Menten enzymes. However, at higher concentrations of CYP1A2 (0.1 and 0.2 μM), the sigmoidal response was observed, leading to activities at subsaturating POR (at 0.025 and 0.05 μM) that were independent of the CYP1A2 concentration.
Figure 3. Measurement of CYP1A2 activities as a function of [POR]: Effect of CYP1A2 concentration.
Titration curves for CYP1A2-dependent activities were measured as a function of [POR] to estimate the ability of POR to associate with CYP1A2. These titration curves were measured at several different CYP1A2 concentrations (0.05, 0.1, 0.2 and 0.4 μM). Reconstituted systems were prepared as described in Experimental and Figure 1. The data represent the mean ± SEM for 3 determinations. (A) Effect on 7-ethoxytrifluorocoumarin metabolism. The 7-EFC deethylation data was fit to a simple Michaelis Menten model with the KDPOR = 0.14 μM and a kcat = 2.37 nmol (min)-1(nmol CYP1A2)-1. (B) Effect on 7-ethoxycoumarin deethylation. POR titrations of this CYP1A2-dependent activity generated data that were better fit to a sigmoidal curve than to a simple Michaelis Menten model (r2 = 0.99 vs. 0.95, respectively, at 0.2 μM CYP1A2).
Figure 4. Effect of ionic strength on CYP1A2-mediated EROD as a function of [POR].
Preparation of the reconstituted systems is described in Experimental. Each data point represents the mean ± SEM for 3 determinations. (A) CYP1A2-mediated EROD at low ionic strength. EROD activity was measured as a function of [POR] in 50 mM HEPES buffer, pH 7.5. Similar to the data shown in Figure 3b, POR titrations of this CYP1A2-mediated activity were sigmoidal in nature. The correlation coefficient r2 based on a sigmoidal fit of the upper curve (at 0.2 μM CYP1A2) was 0.995 – when fitted to a hyperbolic function, the correlation coefficient was only 0.98. (B) CYP1A2-mediated EROD at high ionic strength. EROD activity was measured as a function of [POR] in 50 mM HEPES buffer, pH 7.5 containing 300 mM KCl. Elevation of the ionic strength of the buffer generated a hyperbolic response that could be described using a KDPORD = 0.057 μM and a kcat = 0.60 nmol (min)-1(nmol CYP1A2)-1.
These deviations from Michaelis Menten behavior were increasingly observed at elevated P450 concentrations when the P450 proteins were more crowded into the membrane. This raised the potential for the sigmoidal behavior to be the result of homomeric P450•P450 interactions. Our laboratory previously reported that heteromeric CYP1A2•CYP2B4 and CYP1A2•CYP2E1 [11;12] complexes could be disrupted by increasing the ionic strength of the buffer. Therefore, if the sigmoidal behavior observed at elevated CYP1A2 concentrations was also due to electrostatic interactions, then an increase in ionic strength should lead to disruption of these homomeric complexes and allow the kinetics to revert to a hyperbolic response that can be described by a simple Michaelis Menten type interaction between POR and CYP1A2. As shown in Figure 4b, the inclusion of 300 mM KCl in the buffer caused a change in the kinetic response to a hyperbolic response that could be explained by a single KD value for POR binding, results consistent with the disruption of CYP1A2•CYP1A2 complexes. A similar response was observed when 7-pentoxyresorufin dealkylation was examined (supplemental Figure A).
Detection of CYP1A2•CYP1A2 Complexes by Chemical Crosslinking
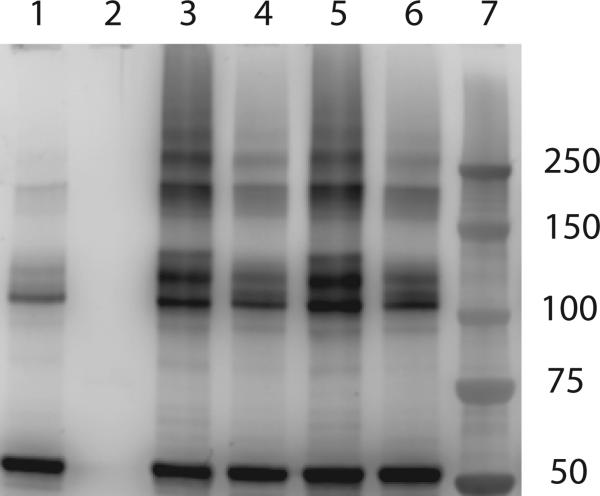
The kinetic data show that CYP1A2 and CYP2E1 exhibit non-Michaelis behavior, particularly at high P450:lipid ratios. If these kinetic effects are indeed the result of homomeric P450 complexes that are stabilized by electrostatic interactions, we would expect to be able to cross-link these P450s and potentially alter the degree of complexation by changing the ionic strength. For these experiments we focused on CYP1A2, which has been shown to readily form complexes with CYP2B4 [7;11;13] and CYP2E1 [12] in addition to the non-Michaelis Menten behavior shown in Figs. 3 and 4. In these experiments we prepared several reconstituted systems in DLPC, crosslinked the proteins with bis(sulfosuccinimidyl) suberate (BS3), separated the proteins using polyacrylamide gels and immune blotted with anti-CYP1A2 and anti-POR. Crosslinking with BS3 (Figure 5) shows that CYP1A2 forms several complexes in simple reconstituted systems ranging in size from about 100 kD to greater than 250 kD, both in the absence or presence of POR (lanes 3 and 5). Overall, crosslinking produced complexes in the 100 kDa, 200 kDa, 250 kDa, and 300 kDa ranges, representing dimers, tetramers, pentamers, and hexamers. Interestingly, we did not observe a single band in the 100 kDa region, representative of a CYP1A2•CYP1A2 dimer, but several bands in the 100-125kDa region. This is possibly due to the formation of dimeric complexes that were crosslinked in different orientations. In separate experiments the addition of substrate alone did not appear to significantly affect complex formation (not shown).
Figure 5. Detection of CYP1A2•CYP1A2 complexes by chemical crosslinking with BS3.
Reconstituted systems containing 0.2 μM CYP1A2 in 32 μM DLPC (lanes 3 & 4) and 0.025 μM POR, 0.2 μM CYP1A2 in 32 μM DLPC (lanes 5 & 6) were subjected to chemical crosslinking using BS3 in 50 mM HEPES buffer pH 7.4 (lanes 3 & 5) or 50 mM HEPES buffer pH 7.4, containing 300 mM KCl (lanes 4 &6). The samples were subjected to SDS-polyacrylamide gel electrophoresis and then blotted using anti-CYP1A2. Lane descriptions: lane 1 – CYP1A2 not crosslinked, lane 2 – POR not crosslinked, lane 3 – CYP1A2/DLPC (1:160) crosslinked in 50 mM HEPES (i.e. low ionic strength), lane 4 –CYP1A2/DLPC (1:160) crosslinked in HEPES + 300 mM KCl (i.e. high ionic strength), lane 5 – POR/CYP1A2/DLPC (0.125:1:160) crosslinked in HEPES (i.e. low ionic strength), and lane 6 – POR/CYP1A2/DLPC (0.125:1:160) crosslinked in HEPES + 300 mM KCl (i.e. high ionic strength), lane 7 – molecular weight standards. This experiment has been replicated with separate reconstituted systems on three occasions. Representative data is shown.
When the reconstituted systems were placed in a high ionic strength buffer (containing 300 mM KCl), changes in the sizes of the complexes were observed (Figure 5, lanes 4 and 6). Generally, there were decreases in the intensity of the complexes, which is consistent with the disruption of CYP1A2•CYP1A2 complexes with increasing ionic strength. Ionic strength also caused a decrease in the formation of POR-containing complexes in a manner similar to that shown for CYP1A2 (supplemental data Figure B). These results are consistent with the kinetic data, where an increase in the ionic strength shifted the sigmoidal activity versus [POR] curves to hyperbolas that follow Michaelis Menten behavior.
Detection of CYP1A2•CYP1A2 Complexes using Bioluminescence Resonance Energy Transfer
Resonance energy transfer methods have been used more recently to measure the formation of complexes among membrane-bound proteins. These methods have been used to detect the formation of both homomeric and heteromeric P450 complexes [9;33-35] as well as other proteins such as heme oxygenase [36;37]. The potential for CYP1A2 to form homomeric complexes was examined by bioluminescence resonance energy transfer (BRET) where the C-terminal region of the CYP1A2 gene was fused with Renilla luciferase (Rluc) and another CYP1A2 gene was fused with green fluorescent protein (GFP). These two fusion proteins were then transfected into HEK293T cells at varying CYP1A2-GFP:CYP1A2-Rluc ratios, the total amount of transfected DNA remaining constant.
As a first step, it was important to demonstrate that the transfected fusion proteins localized in the endoplasmic reticulum. Evidence for the co-localization of the CYP1A2-GFP fusion protein with the ER-marker DSred-ER is shown in supplemental Figure C.
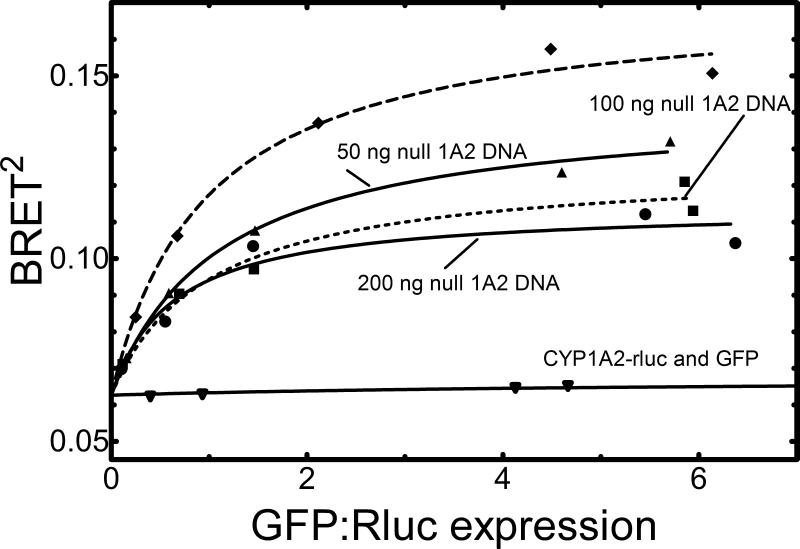
Next, the formation of CYP1A2•CYP1A2 complexes was established using BRET. Both the CYP1A2-Rluc and CYP1A2-GFP genes were transfected into HEK293T cells at a series of CYP1A2-GFP:CYP1A2-Rluc ratios. Twenty four hours after transfection of the fusion proteins, the luciferase substrate coelenterazine 400a was added to the HEK293T cells, generating a luminescent signal at 410 nm. In the event that a complex between CYP1A2 molecules occurs, there is energy transfer from the luminescent signal to the CYP1A2-GFP fusion protein leading to a fluorescent signal at 510 nm. The BRET signal represents a ratio of the 510 nm fluorescence/410 nm luminescence. At low CYP1A2-GFP:CYP1A2-Rluc ratios, the BRET signal is low, due to the low levels of GFP-construct limiting the potential for energy transfer; however, as the relative amounts of the GFP-containing construct are increased, there was a substantial increase in the total BRET (Figure 6, upper curve). The total BRET appeared to saturate at high levels of GFP, which is consistent with the formation of a specific CYP1A2•CYP1A2 complex. Transfection of CYP1A2-Rluc with a naked GFP vector showed only a minimal increase in the total BRET (Figure 6, lower curve).
Figure 6. Demonstration of CYP1A2•CYP1A2 complex formation using BRET: Disruption of the BRET signal by co-transfection with native CYP1A2.
Two CYP1A2 fusion proteins (CYP1A2-Rluc & CYP1A2-GFP) were co-transfected into HEK293T cells at varying ratios of the Rluc- and GFP-containing constructs holding the total amount of transfected DNA constant at 300 ng. Formation of the CYP1A2•CYP1A2 complex is shown in the upper curve. The control curve was generated by co-transfection of CYP1A2-Rluc fusion protein and GFP (lower curve). The middle curves represent the effect of cotransfection of an unlabelled CYP1A2 construct (lacking either the Rluc- of GFP-tags). The amount of unlabeled CYP1A2 DNA transfected into the HEK293T cells was 50 ng (▲), 100 ng (■), and 200 ng (●). This experiment has been replicated three times, with the figure showing the data from one of these experiments.
As an additional demonstration of the specificity of this response, transfection of unlabeled-CYP1A2 (i.e. a CYP1A2 DNA containing neither Rluc nor GFP) was able to compete with the CYP1A2 fusion proteins and diminish the total BRET response. This response, as measured by the maximal BRET at high GFP:Rluc ratios, was dependent on the amount of unlabeled-CYP1A2 transfected into the cells, where increasing unlabeled-CYP1A2 caused a greater inhibition of the total response (Figure 6, middle curves).
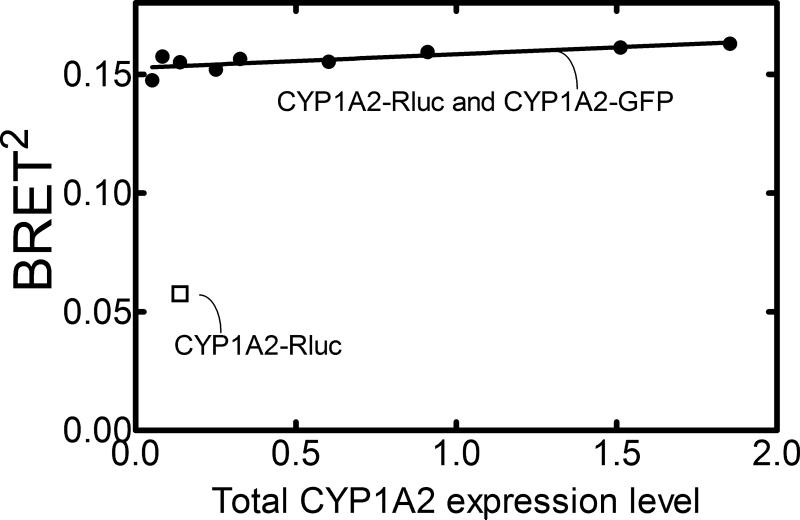
There is a potential concern that the transfection of unlabeled-CYP1A2 would affect the BRET response, by decreasing the total amount of expression of the GFP- and Rluc-fusion proteins. However, such a decrease in protein expression will not affect the maximal BRET response for a high affinity complex. As shown in Figure 7, the total amount of transfected DNA was varied by a factor of more than 35-fold. Regardless of the substantial decrease in the expressed protein, the BRET response was not significantly reduced. These results demonstrate that the decrease in BRET response (shown in Figure 6) was not an artifact of a decrease in expression of the CYP1A2-GFP and CYP1A2-Rluc fusion proteins. In addition, the results illustrate that CYP1A2 forms a high affinity complex, and are not simply the result of protein overcrowding in the membrane. If the complexes were due to protein overcrowding, a significant decrease in the BRET response would be expected with a decrease in protein expression, due to dissociation of the BRET-sensitive complexes [38]. Taken together, these results demonstrate that P450 enzymes are capable of forming homomeric complexes that can modulate monooxygenase function, that these are high affinity complexes, and that they occur not only in reconstituted systems, but also in living cells.
Figure 7. Effect of quantity of DNA transfected on the BRET response: Interaction of CYP1A2-GFP with CYP1A2-Rluc.
HEK293T cells were transfected with different quantities of the CYP1A2-GFP and CYP1A2-Rluc constructs, while maintaining about a 2:1 ratio of the GFP:Rluc fusion proteins (which would give about a maximal BRET response – see Figure 6). Total protein expression was estimated using the Rluc and GFP emissions after each was normalized to the luminescence and fluorescence signals of the GFP-Rluc fusion protein. Total CYP1A2 expression was varied more than 35-fold. Each of the data points (●) represent different amounts of protein expression in the HEK293T cells. The ratio of the CYP1A2-GFP:CYP1A2-Rluc for each point was (from left to right) 1.9, 2.0, 3.2, 3.2,1.8, 2.1, 2.3, 2.2, and 2.0. The open square (□) represents a control transfection where CYP1A2-Rluc and naked GFP constructs were transfected into the cells.
DISCUSSION
It is now generally accepted that cytochromes P450 form complexes in membranes and proteoliposomes [7;13-16;39-41]. Furthermore, it has been shown that in many cases, the physical interaction of different types of P450s can alter the catalytic activities of the associating enzymes [7;10;12-14;39]. Both inhibition and activation of metabolism by P450s have been reported to result from P450-P450 interactions.
The original hypothesis that P450-P450 interactions could influence enzyme function was formulated by comparing substrate metabolism by a single P450 to mixed reconstituted systems containing mixtures of P450s. Thus, the early evidence for these effects was derived from kinetic studies pointing to heteromeric interactions between two different P450 enzymes. However, most of the physical evidence for complex formation (aggregation) of P450 (derived from techniques such as cross-linking [42], rotational diffusion studies using optical [43;44], fluorescent [9], or electroparamagnetic resonance [45] analysis, analytical ultracentrifugation [46-48], gel filtration chromatography [42;49;50], and electron microscopy [47;49]) was demonstrated using a single P450. This raises the question of whether homomeric complexation also influences enzyme function.
Only a single previous study has implicated a homomeric interaction that influences catalytic activity [51]. This study used a global mathematical analysis to fit kinetic data on the metabolism of p-nitrophenol by CYP2E1 at varying concentrations of both the P450 and POR. Their data suggested the existence of the following types of P450 aggregates: 1) The P450/P450 homodimer, 2) the POR/P450 binary complex, 3) a quaternary complex of two PORs and two P450s; and 4) a putative ternary complex of two P450s and one POR. The best fit of the kinetic data was consistent with only the binary POR•CYP2E1 complex being catalytically active – higher order complexes were inactive.
In the present study, the potential for homomeric complex formation to affect P450 function was examined using three P450 enzymes, CYP2B4, CYP2E1 and CYP1A2. The kinetic data obtained using CYP2B4 could be explained by a simple mass action complex being formed between POR and CYP2B4. Although these results do not preclude homomeric complex formation, the results demonstrate that the CYP2B4•CYP2B4 complexes formed do not appear to affect function. The ability of CYP2B4 to form complexes has been shown by cross-linking of reconstituted systems containing only lipid and CYP2B4 (Supplemental Figure D). Despite the tendency for these proteins to form complexes, monooxygenase activities were not affected – at least for the substrates tested.
Both CYP1A2 and CYP2E1 exhibited an apparent cooperative response when several, but not all activities were examined as a function of [POR] concentration. Interestingly, an increase in the ionic strength of the surrounding buffer was capable of disrupting these CYP1A2•CYP1A2 complexes and caused the conversion of the sigmoidal kinetics to a hyperbolic response.
It is important to emphasize that our data suggest the functional effects resulting from homomeric P450 interactions are largely substrate-dependent. CYP1A2-mediated metabolism of 7-EC, PROD, and EROD were clearly influenced by its self-aggregation, as evidenced by the significant sigmoidal curves when activity vs. [POR] was plotted. However, metabolism of 7-EFC by CYP1A2 fit reasonably well to simple, Michaelis-Menten kinetics (Figure 3c). Similar results were obtained with CYP2E1 activities (Figure 2).
The kinetic evidence for homomeric complex formation was corroborated by experiments demonstrating that CYP1A2 forms physical complexes. There are two lines of evidence to support this contention. First, cross-linking data showed that dimers and higher order complexes were readily detected when the proteins were incorporated into membranes prepared from dilauroylphosphatidylcholine (Figure 5). These experiments also showed that these CYP1A2•CYP1A2 complexes were disrupted by increasing the ionic strength of the assay system. These results are consonant with the kinetic data where the “sigmoidal” kinetics in 7ER, 7PR, and 7-EC metabolism at low ionic strength were converted to hyperbolic responses that could be explained by Michaelis Menten behavior at high ionic strength. The results support the formation of kinetically unique homomeric complexes that are disrupted by elevated ionic strength.
A second line of support for the formation of physical CYP1A2•CYP1A2 complexes comes from analysis of the BRET data. These experiments provide evidence for the formation of a high affinity complex between CYP1A2 molecules. The specificity of these complexes was reinforced by the ability of unlabelled CYP1A2 to disrupt the BRET signal.
Given the tendencies of these enzymes to associate in proteoliposomes, it seems likely that homomeric and heteromeric P450 interactions also occur when there are no observable functional effects. However, for reasons that have not been elucidated, it seems that some substrates influence the system in a way that limits the ability of POR to interact with the P450, whereas other substrates do not impede the ability of POR to deliver electrons. Another possibility is that substrates change the affinities by which P450s bind to themselves or to other P450s. In cases where substrates weaken P450•P450 binding affinities, POR may be able to displace P450 enzymes from the aggregates and readily deliver electrons. In cases, where the substrates strengthen the P450-P450 binding affinities, the access of POR to the P450s within the aggregate may be limited.
Previous studies also have revealed that the functional effects attributable to heteromeric P450 interactions are specific to the types of P450 being studied. For instance, testosterone-6β-hydroxylation by CYP3A4 was activated by the presence of CYP1A enzymes but was not affected by the presence of CYP2C9, CYP2D6, or CYP2E1 [39]. In this study, we have presented data supporting the possibility that rabbit CYP2B4 does not form homomeric complexes that influence its ability to carry out substrate metabolism. While this assumption is clearly tentative because of the limited number of compounds tested, CYP2B4-mediated metabolism could be modeled by simple, Michaelis-Menten kinetic with both substrates, unlike the results obtained with CYP1A2 where sigmoidal curves were observed. This sigmoidicity was much more apparent when CYP1A2 concentrations in the membrane were elevated leading to protein crowding and greater opportunities for P450•P450 interactions.
Interestingly, two substrates did have dramatically different effects on the apparent binding affinity of POR and CYP2B4. The apparent Km for POR and CYP2B4 was 13 nM in the presence of benzphetamine and 290 nM in the presence of p-nitrophenol. The putative effects of substrates on the binding of P450s and POR are variable. Using systems in which CYP2B4 and POR were reconstituted in separate lipid vesicles, Backes and Eyer [31] clearly showed that the rate of association of the enzymes was strongly influenced by substrate. Consistent with our current data, the earlier study showed that the rates of association were slightly stimulated by p-nitroanisole but greatly stimulated by benzphetamine relative to the rate in the absence of substrate [31]. Later studies presented findings that showed the effects of substrate on the binding affinity of POR varied widely among different types of P450 [32;50]. In other words, substrates affected the POR-binding affinities of some types of P450 but not others [52]. Similarly, work from the same lab showed that substrates did not affect the binding of POR to a variety of P450s that were affixed to the wells of a polystyrene plate [52]. However, from seeing the varying effects of substrates for P450 enzymes in our study, it could be questioned whether more comprehensive sampling would have revealed that the POR binding to all P450s could be influenced by some compounds but not others. Furthermore, it can be questioned whether the nature of binding between the proteins is profoundly altered after affixing one of the enzymes to the wells of a polystyrene plate. Conceivably, the influence of substrate on the binding of POR and P450 may be manifest when the proteins are reconstituted in a lipid milieu but not when the enzymes are affixed to the plate.
This study provides compelling evidence that the homomeric aggregation of P450s can influence the rate of substrate metabolism by a P450 enzyme, and that the functional effects of homomeric P450 complex formation are both substrate- and P450 type-specific. These homomeric complexes appear to be sensitive to alterations in the ionic strength where the non-Michaelis Menten kinetics observed at low ionic strength can be converted to typical hyperbolic kinetics at elevated salt concentrations. The kinetic data correlated well with studies showing that physical CYP1A2•CYP1A2 complexes exist both in reconstituted systems (chemical crosslinking) and in cellular systems (BRET). Finally, the sensitivity of the kinetic data to ionic strength effects were consistent with those obtained by chemical crosslinking showing a decrease in the formation of CYP1A2•CYP1A2 complexes at elevated ionic strength.
The data demonstrating the existence of homomeric P450 complexes are supportive of previous reports of heteromeric P450 complexes that affect monooxygenase function. These heteromeric interactions have been reported not only in reconstituted systems [7;11-13] but also in cellular systems where P450s are expressed [34;35;53]. Additionally, heteromeric interactions that affect P450 function were shown to occur in rabbit liver microsomes [54]. Although we have been able to show that these complexes occur, we currently do not know the actual size of the complexes in vivo. Cross-linking data shows that P450s can form not only dimers, but also larger complexes (Figure 5). Consequently, we currently do not know the size of the average complex, or even whether they contain other non-P450 proteins as constituents. Additionally, we have several lines of evidence that these complexes occur in vivo. First, the kinetic data in rabbit liver microsomes providing evidence for heteromeric P450•P450 interactions is consistent with the effects reported using defined reconstituted systems [54]. Second, the BRET data shows that CYP1A2•CYP1A2 complexes can form when expressed in HEK293T cells (Figure 6), and that these complexes are not the result of protein over-expression (Figure 7). Taken together, the data strongly supports the idea that P450s exist in the ER membrane as a mixture of homomeric and heteromeric complexes. Changes in the relative amounts of a particular P450 enzyme, such as alterations caused by exposure to a P450 inducer, would be expected to change the composition of these complexes – and potentially lead to alterations in monooxygenase function.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by grants from the National Institute of Environmental Health Sciences (R01 ES004344 & P42 ES013648).
Abbreviations
- CYP; P450
cytochrome P450
- BRET
bioluminescence resonance energy transfer
- EROD
7-ethoxyresorufin-O-dealkylation
- 7-ER
7-ethoxyresorufin
- 7-EFC
7-ethoxy-4-trifluromethylcoumarin
- 7-HFC
7-hydroxy-4-trifluoromethylcoumarin
- BZP
benzphetamine
- NDMA
N-nitrosodimethylamine
- pNA
p-nitroanisole
- NADPH
reduced nicotinamide adenine dinucleotide phosphate
- POR
NADPH – cytochrome P450 oxidoreductase
- DLPC
L-α-dilauroyl-sn-glycero-3-phosphocholine
REFERENCES
- 1.Lu AY, Strobel HW, Coon MJ. Hydroxylation of benzphetamine and other drugs by a solubilized form of cytochrome P-450 from liver microsomes: lipid requirement for drug demethylation Biochem. Biophys.Res.Commun. 1969;36:545–551. doi: 10.1016/0006-291x(69)90339-8. [DOI] [PubMed] [Google Scholar]
- 2.Lu AYH, Coon MJ. Role of hemoprotein P-450 in fatty acid omega-hydroxylation in a soluble enzyme system from liver microsomes. J.Biol.Chem. 1968;243:1331–1332. [PubMed] [Google Scholar]
- 3.Miwa GT, Lu AYH. The association of cytochrome P-450 and NADPH-cytochrome P-450 reductase in phospholipid membranes. Arch.Biochem.Biophys. 1984;234:161–166. doi: 10.1016/0003-9861(84)90337-0. [DOI] [PubMed] [Google Scholar]
- 4.Miwa GT, West SB, Huang MT, Lu AYH. Studies on the association of cytochrome P-450 and NADPH- cytochrome c reductase during catalysis in a reconstituted hydroxylating system. J.Biol.Chem. 1979;254:5695–5700. [PubMed] [Google Scholar]
- 5.Backes WL. NADPH-Cytochrome P450 Reductase: Function. In: Schenkman JB, Greim H, editors. Cytochrome P450. Springer-Verlag; Berlin: 1993. pp. 15–34. [Google Scholar]
- 6.Reed JR, Backes WL. Formation of P450.P450 complexes and their effect on P450 function. Pharmacol.Ther. 2012;133:299–310. doi: 10.1016/j.pharmthera.2011.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Backes WL, Batie CJ, Cawley GF. Interactions among P450 enzymes when combined in reconstituted systems: formation of a 2B4-1A2 complex with a high affinity for NADPH-cytochrome P450 reductase. Biochem. 1998;37:12852–12859. doi: 10.1021/bi980674a. [DOI] [PubMed] [Google Scholar]
- 8.Backes WL, Kelley RW. Organization of multiple cytochrome P450s with NADPH-cytochrome P450 reductase in membranes. Pharmacology & Therapeutics. 2003;98:221–233. doi: 10.1016/s0163-7258(03)00031-7. [DOI] [PubMed] [Google Scholar]
- 9.Davydov DR, Petushkova NA, Bobrovnikova EV, Knyushko TV, Dansette P. Association of cytochromes P450 1A2 and 2B4: are the interactions between different P450 species involved in the control of the monooxygenase activity and coupling? Adv.Exp.Med.Biol. 2001;500:335–338. doi: 10.1007/978-1-4615-0667-6_53. [DOI] [PubMed] [Google Scholar]
- 10.Hazai E, Kupfer D. Interactions between CYP2C9 and CYP2C19 in reconstituted binary systems influence their catalytic activity: possible rationale for the inability of CYP2C19 to catalyze methoxychlor demethylation in human liver microsomes. Drug Metab Dispos. 2005;33:157–164. doi: 10.1124/dmd.104.001578. [DOI] [PubMed] [Google Scholar]
- 11.Kelley RW, Reed JR, Backes WL. Effects of ionic strength on the functional interactions between CYP2B4 and CYP1A2. Biochem. 2005;44:2632–2641. doi: 10.1021/bi0477900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kelley RW, Cheng D, Backes WL. Heteromeric Complex Formation between CYP2E1 and CYP1A2: Evidence for the Involvement of Electrostatic Interactions. Biochem. 2006;45:15807–15816. doi: 10.1021/bi061803n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Reed JR, Eyer M, Backes WL. Functional interactions between cytochromes P450 1A2 and 2B4 require both enzymes to reside in the same phospholipid vesicle: evidence for physical complex formation. J.Biol.Chem. 2010;285:8942–8952. doi: 10.1074/jbc.M109.076885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Subramanian M, Zhang H, Tracy TS. CYP2C9-CYP3A4 protein-protein interactions in a reconstituted expressed enzyme system. Drug Metab.Dispos. 2010;38:1003–1009. doi: 10.1124/dmd.109.030155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Subramanian M, Low M, Locuson CW, Tracy TS. CYP2D6-CYP2C9 protein-protein interactions and isoform-selective effects on substrate binding and catalysis. Drug Metab Dispos. 2009;37:1682–1689. doi: 10.1124/dmd.109.026500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cawley GF, Batie CJ, Backes WL. Substrate-dependent competition of different P450 isozymes for limiting NADPH-cytochrome P450 reductase. Biochem. 1995;34:1244–1247. doi: 10.1021/bi00004a018. [DOI] [PubMed] [Google Scholar]
- 17.Saribas AS, Gruenke L, Waskell L. Overexpression and Purification of the Membrane-Bound Cytochrome P450 2B4. Protein Expression and Purification. 2001;21:303–309. doi: 10.1006/prep.2000.1377. [DOI] [PubMed] [Google Scholar]
- 18.Yasukochi Y, Masters BS. Some properties of a detergent-solubilized NADPH-cytochrome c(cytochrome P-450) reductase purified by biospecific affinity chromatography. J.Biol.Chem. 1976;251:5337–5344. [PubMed] [Google Scholar]
- 19.Shen AL, Porter TD, Wilson TE, Kasper CB. Structural analysis of the FMN binding domain of NADPH-cytochrome P-450 oxidoreductase by site-directed mutagenesis. J Biol.Chem. 1989;264:7584–7589. [PubMed] [Google Scholar]
- 20.Coon MJ, Van Der Hoeven TA, Dahl SB, Haugen DA. Methods in Enzymology. Academic Press; 1984. Two Forms of Liver Microsomal Cytochrome P-450, P-450LM2 and P-450LM4 (Rabbit Liver) pp. 109–123. [DOI] [PubMed] [Google Scholar]
- 21.Cheng D, Kelley RW, Cawley GF, Backes WL. High-level expression of recombinant rabbit cytochrome P450 2E1 in Escherichia coli C41 and its purification. Protein Expression and Purification. 2004;33:66–71. doi: 10.1016/j.pep.2003.08.009. [DOI] [PubMed] [Google Scholar]
- 22.Causey KM, Eyer CS, Backes WL. Dual role of phospholipid in the reconstitution of cytochrome P- 450 LM2-dependent activities. Mol.Pharmacol. 1990;38:134–142. [PubMed] [Google Scholar]
- 23.Beebe LE, Roberts ES, Fornwald LW, Hollenberg PF, Alworth WL. Mechanism-based inhibition of mouse P4502b-10 by selected arylalkynes. Biochem.Pharmacol. 1996;52:1507–1513. doi: 10.1016/s0006-2952(96)00525-4. [DOI] [PubMed] [Google Scholar]
- 24.Burke MD, Thompson S, Elcombe JR, Halpert J, Haaparanta T, Mayer RT. Ethoxy-. Pentoxy-and Benzyloxyphenoxazones and Homologues: A Series of Substrates to Distinguish Between Different Induced Cytochromes P-450. Biochem.Pharmacol. 1985;34:3337–3345. doi: 10.1016/0006-2952(85)90355-7. [DOI] [PubMed] [Google Scholar]
- 25.Roberts ES, Ballou DP, Hopkins NE, Alworth WL, Hollenberg PF. Mechanistic studies of 9-ethynylphenanthrene-inactivated cytochrome P450 2B1. Arch.Biochem.Biophys. 1995;323:303–312. doi: 10.1006/abbi.1995.9960. [DOI] [PubMed] [Google Scholar]
- 26.Hayashi S, Omata Y, Sakamoto H, Hara T, Noguchi M. Purification and characterization of a soluble form of rat liver NADPH-cytochrome P-450 reductase highly expressed in Escherichia coli. Protein Expr.Purif. 2003;29:1–7. doi: 10.1016/s1046-5928(03)00023-8. [DOI] [PubMed] [Google Scholar]
- 27.Cheng D, Reed JR, Harris D, Backes WL. Inhibition of CYP2B4 by the mechanism-based inhibitor 2-ethynylnaphthalene: Inhibitory potential of 2EN is dependent on the size of the substrate. Arch.Biochem.Biophys. 2007;462:28–37. doi: 10.1016/j.abb.2007.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Netter KJ, Seidel G. An adaptively stimulated O-demethylating system in rat liver microsomes and its kinetic properties. J.Pharmacol.Exp.Ther. 1964;146:61–65. [PubMed] [Google Scholar]
- 29.de Andrade JB, Bispo MS, Reboucas MV, Carvalho MLSM, Pinheiro HLC. Spectrofluorimetric determination of formaldehyde in liquid samples. Am.Lab. 1996;28:56–58. [Google Scholar]
- 30.Hanna IH, Reed JR, Guengerich FP, Hollenberg PF. Expression of human cytochrome P450 2B6 in Escherichia coli: characterization of catalytic activity and expression levels in human liver. Arch.Biochem.Biophys. 2000;376:206–216. doi: 10.1006/abbi.2000.1708. [DOI] [PubMed] [Google Scholar]
- 31.Backes WL, Eyer CS. Cytochrome P-450 LM2 reduction. Substrate effects on the rate of reductase-LM2 association. J.Biol.Chem. 1989;264:6252–6259. [PubMed] [Google Scholar]
- 32.Eyer CS, Backes WL. Relationship between the rate of reductase-cytochrome P450 complex formation and the rate of first electron transfer. Arch.Biochem.Biophys. 1992;293:231–240. doi: 10.1016/0003-9861(92)90390-i. [DOI] [PubMed] [Google Scholar]
- 33.Hu G, Johnson EF, Kemper B. CYP2C8 exists as a dimer in natural membranes. Drug Metab.Dispos. 2010;38:1976–1983. doi: 10.1124/dmd.110.034942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ozalp C, Szczesna-Skorupa E, Kemper B. Bimolecular fluorescence complementation analysis of cytochrome p450 2c2, 2e1, and NADPH-cytochrome p450 reductase molecular interactions in living cells. Drug Metab Dispos. 2005;33:1382–1390. doi: 10.1124/dmd.105.005538. [DOI] [PubMed] [Google Scholar]
- 35.Szczesna-Skorupa E, Mallah B, Kemper B. Fluorescence Resonance Energy Transfer Analysis of Cytochromes P450 2C2 and 2E1 Molecular Interactions in Living Cells. J.Biol.Chem. 2003;278:31269–31276. doi: 10.1074/jbc.M301489200. [DOI] [PubMed] [Google Scholar]
- 36.Hwang HW, Lee JR, Chou KY, Suen CS, Hwang MJ, Chen C, Shieh RC, Chau LY. Oligomerization is crucial for the stability and function of heme oxygenase-1 in the endoplasmic reticulum. J.Biol.Chem. 2009;284:22672–22679. doi: 10.1074/jbc.M109.028001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marohnic CC, Huber WJ, Iii, Patrick CJ, Reed JR, McCammon K, Panda SP, Martasek P, Backes WL, Masters BS. Mutations of human cytochrome P450 reductase differentially modulate heme oxygenase-1 activity and oligomerization. Arch.Biochem.Biophys. 2011;513:42–50. doi: 10.1016/j.abb.2011.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.James JR, Oliveira MI, Carmo AM, Iaboni A, Davis SJ. A rigorous experimental framework for detecting protein oligomerization using bioluminescence resonance energy transfer. Nat.Methods. 2006;3:1001–1006. doi: 10.1038/nmeth978. [DOI] [PubMed] [Google Scholar]
- 39.Yamazaki H, Gillam EMJ, Dong MS, Johnson WW, Guengerich FP, Shimada T. Reconstitution of recombinant cytochrome P450 2C10(2C9) and comparison with cytochrome P450 3A4 and other forms: Effects of cytochrome P450-P450 and cytochrome P450-b5 interactions. Arch.Biochem.Biophys. 1997;342:329–337. doi: 10.1006/abbi.1997.0125. [DOI] [PubMed] [Google Scholar]
- 40.Davydov DR, Fernando H, Baas BJ, Sligar SG, Halpert JR. Kinetics of dithionite-dependent reduction of cytochrome P450 3A4: heterogeneity of the enzyme caused by its oligomerization. Biochem. 2005;44:13902–13913. doi: 10.1021/bi0509346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Davydov DR, Sineva EV, Sistla S, Davydova NY, Frank DJ, Sligar SG, Halpert JR. Electron transfer in the complex of membrane-bound human cytochrome P450 3A4 with the flavin domain of P450BM-3: the effect of oligomerization of the heme protein and intermittent modulation of the spin equilibrium. Biochim.Biophys.Acta. 2010;1797:378–390. doi: 10.1016/j.bbabio.2009.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Myasoedova KN, Berndt P. Cytochrome P-450LM2 oligomers in proteoliposomes. FEBS Lett. 1990;275:235–238. doi: 10.1016/0014-5793(90)81479-8. [DOI] [PubMed] [Google Scholar]
- 43.Kawato S, Gut J, Cherry RJ, Winterhalter KH, Richter C. Rotation of cytochrome P-450. I. Investigations of protein-protein interactions of cytochrome P-450 in phospholipid vesicles and liver microsomes. J.Biol.Chem. 1982;257:7023–7029. [PubMed] [Google Scholar]
- 44.Gut J, Richter C, Cherry RJ, Winterhalter KH, Kawato S. Rotation of cytochrome P-450. II. Specific interactions of cytochrome P-450 with NADPH-cytochrome P-450 reductase in phospholipid vesicles. J.Biol.Chem. 1982;257:7030–7036. [PubMed] [Google Scholar]
- 45.Schwarz D, Pirrwitz J, Ruckpaul K. Rotational diffusion of cytochrome P-450 in the microsomal membrane-evidence for a clusterlike organization from saturation transfer electron paramagnetic resonance spectroscopy. Arch.Biochem.Biophys. 1982;216:322–328. doi: 10.1016/0003-9861(82)90217-x. [DOI] [PubMed] [Google Scholar]
- 46.Guengerich FP, Holladay LA. Hydrodynamic characterization of highly purified and functionally active liver microsomal cytochrome P-450. Biochem. 1979;18:5442–5449. doi: 10.1021/bi00591a029. [DOI] [PubMed] [Google Scholar]
- 47.Tsuprun VL, Myasoedova KN, Berndt P, Sograf ON, Orlova EV, Chernyak VY, Archakov AI, Skulachev VP. Quaternary structure of the liver microsomal cytochrome P-450. FEBS Lett. 1986;205:35–40. doi: 10.1016/0014-5793(86)80861-4. [DOI] [PubMed] [Google Scholar]
- 48.Autor AP, Kaschnitz RM, Heidema JK, Coon MJ. Sedimentation and other properties of the reconstituted liver microsomal mixed-function oxidase system containing cytochrome P-450, reduced triphosphopyridine nucleotide-cytochrome P-450 reductase, and phosphatidylcholine. Mol.Pharmacol. 1973;9:93–104. [PubMed] [Google Scholar]
- 49.Myasoedova KN, Tsuprun VL. Cytochrome P-450: hexameric structure of the purified. LM4 form FEBS Lett. 1993;325:251–254. doi: 10.1016/0014-5793(93)81083-c. [DOI] [PubMed] [Google Scholar]
- 50.French JS, Guengerich FP, Coon MJ. Interactions of cytochrome P-450, NADPH-cytochrome P-450 reductase, phospholipid, and substrate in the reconstituted liver microsomal enzyme system. J.Biol.Chem. 1980;255:4112–4119. [PubMed] [Google Scholar]
- 51.Jamakhandi AP, Kuzmic P, Sanders DE, Miller GP. Global analysis of protein-protein interactions reveals multiple CYP2E1-reductase complexes. Biochem. 2007;46:10192–10201. doi: 10.1021/bi7003476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shimada T, Mernaugh RL, Guengerich FP. Interactions of mammalian cytochrome P450, NADPH-cytochrome P450 reductase, and cytochrome b(5) enzymes. Arch.Biochem.Biophys. 2005;435:207–216. doi: 10.1016/j.abb.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 53.Tan YZ, Patten CJ, Smith P, Yang CS. Competitive interactions between cytochromes P450 2A6 and 2E1 for NADPH-cytochrome P450 oxidoreductase in the microsomal membranes produced by a baculovirus expression system. Arch.Biochem.Biophys. 1997;342:82–91. doi: 10.1006/abbi.1997.9995. [DOI] [PubMed] [Google Scholar]
- 54.Cawley GF, Zhang S, Kelley RW, Backes WL. Evidence supporting the interaction of CYP2B4 and CYP1A2 in microsomal preparations. Drug Metab Dispos. 2001;29:1529–1534. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.