Summary
Low-density lipoprotein cholesterol (LDL-C) levels and interleukin 28B (IL28B) polymorphism are associated with sustained viral response (SVR) to peginterferon/ribavirin (pegIFN/RBV) for chronic hepatitis C (CHC) infection. IL28B has been linked with LDL-C levels using a candidate gene approach, but it is not known whether other genetic variants are associated with LDL-C, nor how these factors definitively affect SVR. We assessed genetic predictors of serum lipid and triglyceride levels in 1604 patients with genotype 1 (G1) chronic hepatitis C virus (HCV) infection by genome-wide association study and developed multivariable predictive models of SVR. IL28B polymorphisms were the only common genetic variants associated with pretreatment LDL-C level in Caucasians (rs12980275, P = 4.7 × 10−17, poor response IL28B variants associated with lower LDL-C). The association was dependent on HCV infection, IL28B genotype was no longer associated with LDL-C in SVR patients after treatment, while the association remained significant in non-SVR patients (P < 0.001). LDL-C was significantly associated with SVR for heterozygous IL28B genotype patients (P < 0.001) but not for homozygous genotypes. SVR modelling suggested that IL28B heterozygotes with LDL-C > 130 mg/dL and HCV RNA ≤600 000 IU/mL may anticipate cure rates >80%, while the absence of these two criteria was associated with an SVR rate of <35%. IL28B polymorphisms are the only common genetic variants associated with pretreatment LDL-C in G1-HCV. LDL-C remains significantly associated with SVR for heterozygous IL28B genotype patients, where LDL-C and HCV RNA burden may identify those patients with high or low likelihood of cure with pegIFN/RBV therapy.
Keywords: cholesterol, hepatitis C virus, interleukin 28B, lipids, sustained viral response
Introduction
The hepatitis C virus (HCV) is able to co-opt and disrupt host lipid metabolism to facilitate cell entry [1], assembly [2,3], replication and secretion [4–6]. Lower pretreatment serum low-density lipoprotein cholesterol (LDL-C) levels have been associated with poor response to peginterferon/ribavirin (pegIFN/RBV) therapy for patients with chronic hepatitis C (CHC) [7–9]. This lipid-lowering effect of HCV typically corrects after eradication of the virus, supporting the assertion that these clinical effects are virally mediated [7,10,11].
Genome-wide association studies (GWAS) independently identified single nucleotide polymorphisms (SNPs) near the interleukin 28B (IL28B) gene (chromosome 19) that are strongly associated with response to pegIFN/RBV treatment for patients infected with genotype-1 HCV [12–14]. The polymorphisms share a haplotype block around the IL28B gene with the causal variant(s) yet to be identified. In a candidate gene approach, IL28B polymorphism rs12979860 was found to be associated with LDL-C levels in genotype 1 (G1) CHC [15] and has been associated with hepatic steatosis [16,17]. However, the association of other common genetic variants with lipid levels in HCV has not been tested. Furthermore, the relationship between genetic polymorphism and lipid levels during and after treatment and the interactions with sustained viral response (SVR) prediction have not been explored.
We therefore sought to identify whether any other common genetic variants may contribute to serum lipid and triglyceride levels by assessing whole-genome variation by GWAS in the IDEAL pharmacogenomics cohort [18]. The large size and well-characterized nature of the cohort enabled us to analyse LDL-C levels during and after treatment to characterize the host–virus interdependence of this association, which has not previously been studied. Finally, we assessed the clinical utility of LDL-C in the prediction of SVR in the light of IL28B genetic associations, modelling specific clinical parameters to help define how LDL-C may be associated with SVR.
Materials and Methods
Study cohort
The IDEAL trial was a multi-centre, randomized control trial comparing efficacy and adverse events in 3070 treatment naïve patients with CHC (ClinicalTrials.gov number, NCT00081770) and has previously been described [18]. Patients chronically infected with genotype-1 HCV were randomized to one of three treatment arms: peginterferon alfa-2b at standard dose (1.5 μg per kilogram body weight per week); or peginterferon alfa-2b at a lower dose (1.0 μg per kilogram per week), both with ribavirin doses between 800–1400 mg per day; or peginterferon alfa-2a at standard dose (180 μg/week) and ribavirin dose of 1000–1200 mg/day. Study patients with evaluable GWAS samples were predominantly Caucasian males in their forties with mean BMI 28.2 kg/m2 [standard deviation (SD) 4.8] mean HCV RNA of 6.36 log10 IU/mL (SD 0.62) and 11% had advanced fibrosis (METAVIR fibrosis stages 3 or 4) (Table 1).
Table 1. Clinical characteristics of the study population for genome-wide association studies related to serum lipids.
| All | Caucasians | African Americans | Hispanics | |
|---|---|---|---|---|
| N | 1319 | 1017 | 207 | 95 |
| Gender (M/F) | 803/516 | 622/395 | 121/86 | 60/35 |
| Age, years (mean) | 47.4 | 47.2 | 49.7 | 44.3 |
| BMI, kg/m2 (mean) | 28.2 | 27.8 | 29.7 | 29.2 |
| Advanced fibrosis (>F2), n (%) | 148 (11) | 115 (11) | 20 (10) | 13 (14) |
| Steatosis (>0%), n (%) | 810 (61) | 607 (60) | 134 (65) | 69 (73) |
| Activity grade (% with grade 2–3) | 82.6 | 82.0 | 85.5 | 83.2 |
| Baseline viral load (log10) (mean) | 6.36 | 6.36 | 6.36 | 6.15 |
| Baseline fasting blood glucose, mm (mean) | 5.3 | 5.2 | 5.4 | 5.2 |
| Baseline low-density lipoprotein cholesterol, mg/dL (mean) | 103.2 | 104.0 | 100.5 | 98.6 |
| Sustained viral response (%) | 51 | 55 | 26 | 51 |
The IDEAL cohort has previously been analysed for non-genomic, clinical relationships between serum cholesterol, statin use and SVR [9]. A pharmacogenomics cohort from IDEAL consented to DNA testing (n = 1604) and has been analysed for genetic associations with treatment response [12] and ribavirin-induced haemolytic anaemia [19].
From the pharmacogenomics cohort, patients on statin therapy at any time during the study period (n = 46) were excluded from the GWAS analysis to avoid potential confounding. Patients were included if all covariate data were available for the relevant models and GWAS genotyping quality control protocols were satisfied (n = 1319) [12]. For the SVR analysis in addition to further adjusting for self-declared race rather than genetically inferred ancestry and pegIFN dose received, we considered all patients with covariate data on an intention-to-treat basis, irrespective of treatment compliance, ethnicity or statin therapy (n = 1473).
Genetic analysis
Patients were genotyped using the Illumina Human610-quad BeadChip (Illumina, San Diego, CA, USA). After quality control, 97.5% or 565 759 SNPs were analysed with multivariable linear and logistic regression models. The primary association model used single-marker genotype trend tests in three independent ethnic groups (Caucasians, African Americans and Hispanics). Ethnicity was genetically inferred and a modified Eigenstrat method corrected for population substratification [20].
Clinically important covariates adjusted for in the models included age; gender; body mass index (kg/m2); baseline HCV viral load (log10 IU/mL); fibrosis (binary variable as METAVIR stage F0–2 vs F3–4); inflammation (METAVIR grade 0–1 vs 2–3), alanine transaminase (ALT) values, baseline fasting blood glucose levels and respective genetic ancestry subpopulation (or Eigen) sets. Plasma HCV RNA concentrations were measured using the COBAS Taqman assay (lower limit of quantitation of 27 IU/mL; Roche Diagnostics, Indianapolis, IN, USA).
Phenotypes
Fasting levels of total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), LDL-C and triglycerides (TG) were analysed at baseline through GWAS using a linear regression model. Serum lipid measurements were considered as continuous variables, and LDL-C was log transformed to fit a normal distribution.
Serum LDL-C was further analysed by IL28B genotype during treatment at 12 weeks (n = 1385), 24 weeks (n = 1045) and 48 weeks (n = 797), then at 24 weeks follow-up post-treatment (n = 1079). Follow-up LDL-C was analysed based on SVR status, defined as undetectable HCV RNA 24 weeks after treatment completion (or 12 weeks after completion if post-treatment 24 week HCV RNA data were missing).
Multivariable logistic regression models of SVR adjusted for pegIFN alfa-2b dose (low vs standard dose) in addition to the covariates in the GWAS. Patients were considered on an intention to treat basis irrespective of treatment adherence, with ethnicity defined by patient declaration rather than inferred from genotype data as in the GWAS. Patients on statins were reincluded and statin use was considered as a covariate.
Statistical analyses
The Bonferroni method corrected for multiple testing with genome-wide significance predefined at P < 4.4 × 10−8 (α = 0.05). Student's T-test was used to compare LDL levels between IL28B genotype (good vs poor response genotypes) during treatment and when comparing between SVR and non-SVR patients at follow-up. Linear and logistic regression modelling was conducted with PLINK [21], STATA (Stata-Corp, College Station, TX, USA) and SAS (SAS Institute, Cary, NC, USA) software. The study met local Institutional Review Board requirements and the 1975 Declaration of Helsinki guidelines.
Results
Serum lipid and triglyceride genome-wide association studies
IL28B polymorphisms are the only common variants with genome-wide significant association with baseline serum low-density lipoprotein cholesterol levels
Four SNPs demonstrated genome-wide significant association with pretreatment LDL-C (Table 2). These SNPs are in the region of the IL28B gene on chromosome 19 and are all in linkage disequilibrium with the top SNP (Table S1), and all have previously been associated with pegIFN/RBV treatment outcome [12–14].
Table 2. Genome-wide significant associations with pretreatment low-density lipoprotein cholesterol levels.
| Single nucleotide polymorphism | P-value | |||
|---|---|---|---|---|
|
| ||||
| Combined (n = 1319) | Caucasians (n = 1017) | African Americans (n = 207) | Hispanics (n = 95) | |
| rs12980275 | 4.7 × 10−17 | 6.9 × 10−16 | 0.017 | 0.63 |
| rs12979860 | 4.9 × 10−17 | 1.3 × 10−15 | 0.014 | 0.26 |
| rs8099917 | 3.2 × 10−16 | 1.2 × 10−15 | 0.33 | 0.086 |
| rs12972991 | 7.0 × 10−9 | 2.0 × 10−8 | 0.21 | 0.48 |
For the most significant SNP rs12980275, located 2.4 kb downstream from the IL28B gene, the G ‘poor response’ allele was associated with lower serum LDL-C levels across combined ethnic groups (G allele, all ethnicities P = 4.7 × 10−17). In subanalysis by ethnicity, the association was significant in Caucasians (n = 1017, P = 6.9 × 10−16; Table S2), but not genome-wide significant in the smaller cohorts of African American (n = 207, P = 0.017) and Hispanic patients (n = 95, P = 0.63), although there was a consistent direction of effect. Importantly, no other common genetic variants on genome-wide testing were associated with LDL-C on genome-wide testing.
These results likely reflect an IL28B haplotype effect rather than being SNP-specific, given the degree of linkage disequilibrium demonstrated. As the most significant genetic association for LDL-C in the GWAS was from rs12980275, further analysis will focus on this SNP. To minimize confusion in allele description of ‘introducing’ a new IL28B allele, the variant will be discussed in terms of its association with treatment response to pegIFN/RBV as good or poor response. Results will thus be described as ‘good response’ variants corresponding to patients homozygous for the ‘A’ allele at rs12980275 (with equivalent direction of effect for the better known ‘C’ allele at rs12979860; or the ‘T’ allele at rs8099917) and the presence of the G allele (G/G or A/G) corresponding to ‘poor response’.
Low-density lipoprotein cholesterol levels above 130 mg/dL have previously been associated with SVR [9]. For Caucasians, twice as many patients with the good response IL28B genotype had LDL-C above this clinical threshold than poor response genotypes (A/A vs A/G and G/G, 33.4% vs 14.3%, respectively, P = 9.92 × 10−6).
Baseline serum total cholesterol, high-density lipoprotein cholesterol and triglycerides analyses do not demonstrate genome-wide criteria but identify significant candidate genetic associations
No genome-wide significant associations for other lipids or triglyceride levels were found on GWAS. Several previously identified associations satisfy less stringent candidate gene criteria (results for Caucasians presented in Tables S3–S5). For the HDL-C GWAS, the top SNP (rs3764261, P = 5.80 × 10−7) was located upstream from the cholesterylester transfer protein (CETP) gene which has previously been linked to HDL-C levels [22]. For TG, the second ranked SNP was close to the APOA5 gene, a gene previously associated with hypertriglyceridemia (rs6589567, P = 4.00 × 10−6) [23].
Genetic association between IL28B genotype and low-density lipoprotein cholesterol is hepatitis C virus-dependent
Although there were IL28B genotype-based differences in mean LDL-C levels at baseline (P = 6.9 × 10−16), during treatment with exogenous peginterferon there were no significant differences in mean serum LDL-C by IL28B genotype (rs12980275; good response genotype vs poor response genotype, Fig. 1). We found no differences based on either viral response or IL28B genotype when comparing LDL-C levels by on-treatment milestones [early viral responses (EVRs) vs LDL-C at 12 weeks and end of treatment responses vs week 48 LDL-C level, Tables S8 & S9]. However the potentially confounding effect of exogenous interferon [24] was a limitation for this on-treatment analysis relative to the pretreatment and SVR/non-SVR LDL-C analyses.
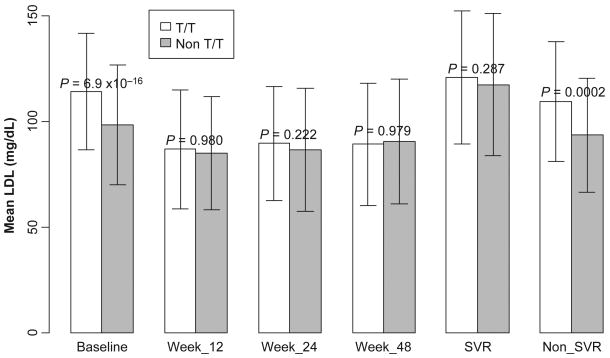
Fig. 1.
Mean serum low-density lipoprotein (LDL) (mM) at baseline, during treatment and at follow-up by host IL28B genotype [rs12980275 A/A (white) vs non-A/A (grey)] in Caucasians within the IDEAL pharmacogenomics cohort.
For Caucasians, there was a significant IL28B genotype-based difference in mean serum LDL-C at 24 weeks follow-up after treatment in non-SVR patients (P = 0.0002), but in patients who achieved SVR, IL28B was not associated with LDL-C (P = 0.287). This was further tested in a multivariable model of follow-up LDL-C, where formal interaction testing confirmed a significant interaction between treatment outcome and IL28B (P = 0.02 for interaction term) with no significant relationship in SVR patients (Tables S6 and S7).
Defining the role of low-density lipoprotein cholesterol and IL28B in predicting sustained viral response
Given the recognized genetic association between IL28B polymorphisms and treatment response [12–14] and also LDL-C levels and treatment response [8,9], we undertook multivariable modelling to more definitively assess the association of pretreatment LDL-C on SVR in this large genotype-informed cohort. We assessed models before and after the inclusion of host IL28B genotype as a covariate and compared each model's performance [r2 and area under the receiver operating curve (ROC)].
A baseline logistic regression model of SVR adjusted for the covariates of LDL-C, HCV RNA (log10 IU/mL), race, age, gender, significant fibrosis (METAVIR >F2), presence of any steatosis (>0%), fasting blood glucose level, alanine aminotransferase (ALT), low dose pegIFN and statin therapy but did not consider IL28B genotype. Higher pretreatment LDL-C was significantly associated with better treatment response [OR 1.16 per 10 mg/dL LDL (95%CI 1.11–1.20); P < 0.0001] with reasonable model performance (r2 19.2% ROC 0.721), confirming the previous association with SVR [9]. Including IL28B into the model improved the model's predictive performance (r2 30.6% ROC 78.3%), with significant interaction between LDL-C and IL28B (P-value for interaction = 0.029), reducing the effect and significance of LDL-C in SVR prediction, which was subsequently explored further.
Low-density lipoprotein cholesterol helps identify heterozygous IL28B genotype patients with high or low chance of sustained viral response
The significance of the association of LDL-C with SVR is dependent upon IL28B genotype. For heterozygous patients, LDL-C level was a significant predictor of SVR (per 10 mg/dL increase in LDL-C, OR 1.18 (95% CI 1.11–1.25), P < 0.001). In contrast, for patients with homozygous IL28B genotypes LDL-C did not aid in predicting response (IL28B good/poor response genotypes P = 0.097 and P = 0.308, respectively; Tables S10–S12).
Other recognized clinical and demographic factors continued to be significant in the prediction of SVR after inclusion of IL28B and LDL-C. Significant covariates associated with a better chance of SVR included Caucasian and Hispanic race relative to African American race (OR 2.11, P < 0.001 and OR 2.61, P = 0.008, respectively), fibrosis stage of ≤F2 (OR 2.48, P < 0.001); normal fasting plasma glucose (OR 1.33, P = 0.001) and lower HCV RNA (per 1 log10 IU/mL decrease, OR 2.15, P < 0001).
Because LDL-C remained a significant predictor of SVR for heterozygous IL28B genotype patients, we sought to better define the clinical utility of LDL-C in predicting SVR for this specific patient group. Dichotomizing the cohort into high LDL-C (>130 mg/dL) and low LDL-C (≤130 mg/dL), we considered a number of treatment endpoints. While there was no difference in rapid viral response, significantly higher rates of EVR, end of treatment responses (EOTR), and as expected SVR were seen in patients with LDL >130 mg/dL, while relapse rates were significantly lower (Table 3). Baseline LDL-C either as quartiles or at the 130 mg/dL cut-off point differentiated HCV viraemia across HCV RNA measurement timepoints (treatment weeks 2, 4, 12, Figures S1 & S2). Importantly, however, the LDL-C cut-off did not help to discriminate eventual SVR responses in slow responder patients with partial EVR (>2 log IU/mL reduction in HCV RNA but remaining HCV RNA positive after 12 weeks of therapy) and thus is unlikely to be useful to identify a treatment futility rule.
Table 3. Univariate analysis of on-treatment, end of treatment and sustained viral responses and relapse rates compared by baseline low-density lipoprotein cholesterol (LDL-C) thresholds in IL28B heterozygous patients.
| Week 4 response (RVR) | Week 12 response (cEVR) | Week 48 response (EOTR) | Week 24 follow-up response (SVR) | Relapse | |
|---|---|---|---|---|---|
| LDL-C > 130 mg/dL | 7.14% (7/98) | 55.4% (51/92) | 74.1% (60/81) | 47.1% (49/104) | 18.3% (11/49) |
| LDL-C ≤ 130 mg/dL | 5.12% (32/625) | 33.4% (204/611) | 47.6% (267/581) | 28.7% (185/644) | 32.6% (87/267) |
| Chi-square test P-value | 0.5594 | <0.0001* | <0.0001 | 0.0002 | 0.0432 |
RVR, rapid viral response; EOTR, end of treatment responses; SVR, sustained viral response; EVR, early viral response; pEVR, partial EVR; cEVR, HCV RNA negative after 12 weeks of therapy.
The test considers all three levels of EVR (cEVR, pEVR, and no EVR).
We further assessed SVR rates observed in the trial compared across a number of important clinical predictors of SVR (Table 4). LDL-C and high/low HCV RNA burden (≤600 000 IU/mL) were key factors. SVR decreased from 71% when comparing patients with high LDL-C and low HCV RNA, to 24% in patients with neither of these factors. In univariate analysis of the trial data, the high/low LDL-C threshold helped to differentiate SVR rates when considered across a number of other clinical factors such as significant fibrosis (>F2 METAVIR stage), African American ethnicity and abnormal fasting blood glucose.
Table 4. Univariate analysis of sustained viral response (SVR) rates observed from the IDEAL pharmacogenomics cohort for IL28B heterozygous patients with high or low low-density lipoprotein cholesterol (LDL-C) (≤/>130 mg/dL) compared with other prognostic factors associated with SVR.
| Clinical factor | LDL-C > 130 mg/dL SVR % (n/N) | LDL-C ≤ 130 mg/dL SVR % (n/N) | P-value* |
|---|---|---|---|
| HCV RNA ≤600 000 IU/mL | 71% (12/17) | 53% (59/112) | 0.00014 |
| HCV RNA >600 000 IU/mL | 43% 37/87 | 24% (126/532) | |
| F0–F2 METAVIR fibrosis | 47.9% (45/94) | 30.6% (175/571) | 0.00033 |
| F3–F4 METAVIR fibrosis | 40.0% (4/10) | 13.7% (10/73) | |
| Non-African American | 53.8% (42/78) | 30.6% (165/533) | 0.00008 |
| African American | 26.9% (7/26) | 18.0% (10/73) | |
| Normal fasting blood glucose | 50.0% (43/86) | 31.5% (165/524) | 0.00030 |
| Abnormal fasting blood glucose | 33.3% (6/18) | 16.7% (20/120) |
Cochran–Mantel–Haenszel test compares SVR rates for high vs low LDL-C after adjusting for hepatitis C virus (HCV) RNA level, METAVIR fibrosis, race, abnormal fasting glucose.
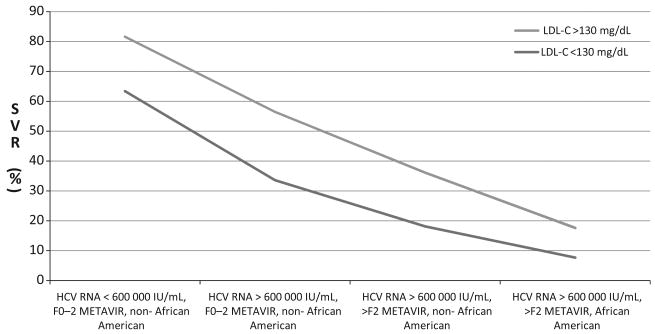
Finally, we sought to consider these variables together in a multivariable model using binary clinical thresholds as might be carried out in clinic. For model parsimony, we assumed no hepatic steatosis and normal glucose. This model confirmed that for IL28B heterozygous patients, the LDL-C threshold helped to differentiate the predicted treatment responses across the continuum of clinical and racial factors associated with poor response (R2 = 0.261, ROC = 76.0%) (Fig. 2). Patients with high LDL-C, low viral load, without advanced fibrosis (≤F2) and non-African American ethnicity had predicted SVR rates of 81%, while for patients without any of these positive treatment prognostic factors anticipated SVR is below 10%.
Fig. 2.
Low-density lipoprotein cholesterol (LDL-C) level above 130 mg/dL (light grey line) or below 130 mg/dL (dark grey line) differentiates sustained viral response (SVR) predication for IL28B heterozygous patients across the spectrum of adverse clinical predicators including high viral load [hepatitis C virus (HCV) RNA >600 000 IU/mL], advanced fibrosis (>METAVIR F2) and African American race in a logistic regression model of SVR.
Discussion
Altered lipid levels in patients infected with HCV have been thought primarily due to disruption of host lipid synthetic pathways mediated by viral factors such as the HCV core [25] and nonstructural (e.g. NS5A) [26] proteins. Through a genome-wide association approach, we have demonstrated host genetics are also critical. IL28B polymorphisms are the only common genetic variants significantly associated with host serum LDL-C levels in genotype-1 HCV infection. We thus validate the previously reported candidate gene association of IL28B polymorphism with LDL-C levels [15] and are able to extend this, demonstrating for the first time that there are no other common variants associated with LDL-C in genotype-1 HCV infection.
In this retrospective analysis, we have demonstrated that LDL-C's association with SVR is conditional upon IL28B genotype. LDL-C has predictive utility for patients with heterozygous IL28B genotype only and is not associated with SVR for patients homozygous for either the good or poor response IL28B alleles. In a multivariable model of SVR in IL28B heterozygous patients, established cut-offs for LDL (>130 mg/dL) and HCV RNA (≤600 000 IU/mL) provided a clear distinction of patients with high or low predicted SVR rate, independently of fibrosis and race.
Heterozygous IL28B genotype is the most common IL28B genotype in Caucasians. The heterozygous IL28B genotype is less clear in its predictive discrimination of SVR compared with the homozygous genotypes. Our data suggest patients with high LDL-C, low viral load and heterozygous IL28B genotype may anticipate much higher rates of SVR with existing pegIFN/RBV therapy. This distinction may be important for planning regimens for patients at more risk of toxicity or in treatment settings where direct acting antivirals (DAAs) may not be available. For the majority of heterozygous patients with low LDL-C and/or high HCV RNA, expected treatment responses to pegIFN/RBV are below the anticipated SVR rates seen with regimens including DAAs. Further prospective testing is necessary to validate the association of SVR with LDL-C and HCV RNA level for pegIFN/RBV regimens and to explore its use in DAA-inclusive regimens. IL28B may continue to play a significant, though attenuated role in DAA-inclusive peg-IFN/RBV regimens [27,28]. To define the clinical utility of LDL-C, our SVR modelling concentrated on IL28B heterozygous genotype patients based on data from a treatment naive cohort; however, more extensive modelling of SVR in larger cohorts including patients with more advanced fibrosis or prior nonresponse is needed, ideally prospectively.
In the GWAS analysis of other lipid measurements, no novel variants met genome-wide significance criteria; however, two polymorphisms implicated in previous genetic studies satisfied candidate gene criteria for significance. HDL-C was significantly associated with CETP variants and genetic variation near the APOA5 gene with hypertriglyceridaemia [22,23]. This demonstrates that common genetic variants important in non-HCV-infected populations may also have important effects on HDL and TG for patients with HCV.
Our data suggest that IL28B polymorphisms influence the biological associations between HCV infection and serum LDL-C, and the clinical utility of LDL-C for predicting treatment response to pegIFN/RBV therapy. Exogenous IFN therapy has been previously observed to decrease serum lipid levels, which may occur via reduced activities of lipoprotein lipase (LPL) and hepatic triglyceride lipase (HTGL) [24]. This may account for the lower LDL-C levels seen during therapy irrespective of host genotype or on-treatment responses. In contrast, LDL-C levels differ in HCV-infected vs cured patients, suggesting a direct role for the virus. Furthermore, in HCV-infected patients (whether pretreatment or non-SVR post-treatment) the effect of the virus on LDL-C level differs significantly by host IL28B genotype. The underlying mechanism of how IL28B genetic variation is associated with treatment-induced and spontaneous HCV clearance remains unclear, although it is presumed to be immune related. The viral dependency of the association between host genetics and LDL-C and the inverse correlation between HCV RNA levels and LDL-C (Figures S1 & S2), suggests LDL-C is a marker of HCV interference in lipid metabolism, mediated by host genetics.
Increased expression of interferon-stimulated genes (ISGs) provides a possible functional explanation and weak correlation has previously been found between ISG expression and LDL-C levels. Higher levels of ISG expression pretreatment have been associated with nonresponse to treatment [29,30] and have also been correlated with poor response IL28B genotype [31,32], although a recent study has suggested these factors may work independently [33]. Unfortunately, ISG expression data were not available in this clinical trial cohort. The functional mechanisms underlying these genetic associations remain undetermined. Further molecular and clinical research is required to understand how host genotype underlies the complex interaction between the HCV, the host immune system and lipid metabolism.
Conclusions
IL28B polymorphisms are the only common variants associated with LDL-C in G1-HCV. This association is HCV-dependent and was no longer significant after SVR. The clinical utility of serum LDL-C for predicting SVR is apparent only for patients with heterozygous IL28B genotype. In multivariable modelling of SVR, LDL-C level was significant for heterozygous IL28B patients and may potentially allow better prognostication of treatment response. Established thresholds for LDL-C and HCV RNA levels help to better identify heterozygous IL28B genotype patients with high or low anticipated SVR rates to pegIFN/RBV therapy and may help to better individualize care.
Supplementary Material
Supporting Information: Additional Supporting Information may be found in the online version of this article:
Fig. S1 Quartiles of LDL_C by HCV RNA level during treatment at week 2, week 4 and week 12 timepoints.
Fig. S2 LDL_C (</>130 mg/dL) by HCV RNA level during treatment at week 2, week 4 and week 12 time-points.
Table S1: Measures of linkage disequilibrium between genome wide significant SNPs and rs12980275 by race.
Table S2: Multiple regression model for baseline LDL in Caucasians.
Table S3: Top GWAS association polymorphisms in association with total cholesterol in Caucasians.
Table S4: Top GWAS association polymorphisms in association with serum HDL-cholesterol in Caucasians.
Table S5: Top GWAS association polymorphisms in association with serum triglycerides in Caucasisans.
Table S6: Multiple regression model for LDL at 24 weeks post treatment for non-SVR patients.
Table S7: Multiple regression model for LDL at 24 weeks post treatment for patients who attained SVR.
Table S8: Comparison between treatment response at the timepoint LDL was measured.
Table S9: Comparison between genotypes within treatment response levels at the timepoint LDL was measured.
Table S10: Multiple logistic regression model for SVR.
Table S11: Multiple logistic regression model results testing interaction between LDL and IL28B genotype (rs12980275) for SVR.
Table S12: Univariate comparison of SVR rates by each IL28B genotype (rs12980275).
Acknowledgments
The authors would like to thank the patients in the IDEAL trial as well as the principal investigators, study coordinators and nurses involved. The authors thank Megan Koehler, DCRI for her statistical review.
Funding: The study was funded by Schering-Plough Research Institute, Kenilworth, NJ (now Merck and Co.). Drs Clark and Thompson received funding support from the Duke Clinical Research Institute, the Richard Boebel Family Fund, the National Health and Medical Research Council of Australia (PJC: APP1017139) and the Gastroenterological Society of Australia. Dr Clark received funding from the National Centre in HIV Epidemiology and Clinical Research (now The Kirby Institute for Infection and Immunity in Society), University of New South Wales, Australia, and the AASLD/LIFER Clinical and Translational Research Fellowship in Liver Diseases Award. Dr Thompson received funding from the Royal Australian College of Physicians.
Abbreviations
- ALT
alanine aminotransferase
- CETP
cholesterylester transfer protein
- CHC
chronic hepatitis C
- DAA
direct acting antiviral
- EOTR
end of treatment responses
- EVR
early viral response
- G1
genotype 1
- GWAS
genome-wide association studies
- HCV
hepatitis C virus
- HDL-C
high-density lipoprotein cholesterol
- HTGL
hepatic triglyceride lipase
- IL28B
interleukin 28B
- ISG
interferon-stimulated gene
- LDL-C
low-density lipoprotein cholesterol
- LPL
lipoprotein lipase
- pegIFN/RBV
peginterferon/ribavirin
- ROC
receiver operating curve
- SD
standard deviation
- SNP
single nucleotide polymorphisms
- SVR
sustained viral response
- TC
total cholesterol
- TG
triglycerides
Appendix 1
Members of the IDEAL investigators: Ira Jacobson, Weill Cornell Medical College, New York, NY, USA; Fred Poor-dad, Cedars-Sinai Medical Center, Los Angeles, CA, USA; Eric Lawitz, Alamo Medical Research, San Antonio, TX, USA; Jonathan McCone, Mt. Vernon Endoscopy Center, Alexandria, VA, USA; Mitchell L. Shiffman, Virginia Commonwealth University, Richmond, VA, USA; Greg W. Galler, Kelsey Research Foundation, Houston, TX, USA; William M. Lee, University of Texas Southwestern Medical Center, Dallas, TX, USA, Robert Reindollar, Piedmont Healthcare, Statesville, NC, USA; John King, Louisiana State University, Shreveport, LA, USA; Reem Ghalib, The Liver Institute at Methodist Dallas Medical Center, Dallas, TX, USA; Bradley Freilich, Kansas City Gastroenterology and Hepatology, Kansas City, MO, USA; Lisa M. Nyberg, Kaiser Permanente, San Diego, CA, USA; Zachary Goodman, Armed Forces Institute of Pathology, Washington, DC, USA; Navdeep Boparai, Kenneth Koury, Clifford A. Brass, Schering-Plough Corporation, now Merck & Co., Inc., Whitehouse Station, NJ, USA.
Footnotes
Conflict of Interest: The authors disclose the following, Drs Thompson, Sulkowski, Afdhal, McHutchison and Muir report having received research and grant support from Schering-Plough (now Merck); Drs Afdhal, Harrison, McHutchison, Goldstein and Muir have received consulting fees or acted in an advisory capacity for Schering-Plough or Merck. Drs Albrecht and Pedicone are employees of Schering-Plough (now Merck & Co.) and are stockholders in this entity. Dr McHutchison is now employed by Gilead Sciences. Dr Noviello is a former employee of Schering-Plough and is now a consultant to Merck & Co. Drs Thompson, Ge, Urban, McHutchison and Goldstein are co-inventors of a patent application based on the IL28B finding.
Please note: Wiley-Blackwell are not responsible for the content or functionality of any supporting materials supplied by the authors. Any queries (other than missing material) should bedirected to the corresponding author for the article.
References
- 1.Agnello V, Ábel G, Elfahal M, Knight GB, Zhang QX. Hepatitis C virus and other Flaviviridae viruses enter cells via low density lipoprotein receptor. Proc Natl Acad Sci U S A. 1999;96(22):12766–12771. doi: 10.1073/pnas.96.22.12766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boulant S, Montserret R, Hope RG, et al. Structural determinants that target the hepatitis c virus core protein to lipid droplets. J Biol Chem. 2006;281(31):22236–22247. doi: 10.1074/jbc.M601031200. [DOI] [PubMed] [Google Scholar]
- 3.Miyanari Y, Atsuzawa K, Usuda N, et al. The lipid droplet is an important organelle for hepatitis C virus production. Nat Cell Biol. 2007;9(9):1089–1097. doi: 10.1038/ncb1631. [DOI] [PubMed] [Google Scholar]
- 4.Gastaminza P, Cheng G, Wieland S, Zhong J, Liao W, Chisari FV. Cellular determinants of hepatitis C virus assembly, maturation, degradation, and secretion. J Virol. 2008;82(5):2120–2129. doi: 10.1128/JVI.02053-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barth H, Liang TJ, Baumert TF. Hepatitis C virus entry: molecular biology and clinical implications. Hepatology. 2006;44(3):527–535. doi: 10.1002/hep.21321. [DOI] [PubMed] [Google Scholar]
- 6.Syed GH, Amako Y, Siddiqui A. Hepatitis C virus hijacks host lipid metabolism. Trends Endocrinol Metab. 2010;21(1):33–40. doi: 10.1016/j.tem.2009.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Poynard T, Ratziu V, McHutchison J, et al. Effect of treatment with peginterferon or interferon alfa-2b and ribavirin on steatosis in patients infected with hepatitis C. Hepatology. 2003;38(1):75–85. doi: 10.1053/jhep.2003.50267. [DOI] [PubMed] [Google Scholar]
- 8.Ramcharran D, Wahed AS, Conjeevaram HS, et al. Associations between serum lipids and hepatitis C antiviral treatment efficacy. Hepatology. 2010;52(3):854–863. doi: 10.1002/hep.23796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Harrison SA, Rossaro L, Hu KQ, et al. Serum cholesterol and statin use predict virological response to peginterferon and ribavirin therapy. Hepatology. 2010;52(3):864–874. doi: 10.1002/hep.23787. [DOI] [PubMed] [Google Scholar]
- 10.Serfaty L, Andreani T, Giral P, Carbonell N, Chazouilleres O, Poupon R. Hepatitis C virus induced hypobeta-lipoproteinemia: a possible mechanism for steatosis in chronic hepatitis C. J Hepatol. 2001;34(3):428–434. doi: 10.1016/s0168-8278(00)00036-2. [DOI] [PubMed] [Google Scholar]
- 11.Corey KE, Kane E, Munroe C, Barlow LL, Zheng H, Chung RT. Hepatitis C virus infection and its clearance alter circulating lipids: implications for long-term follow-up. Hepatology. 2009;50(4):1030–1037. doi: 10.1002/hep.23219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ge D, Fellay J, Thompson AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461(7262):399–401. doi: 10.1038/nature08309. [DOI] [PubMed] [Google Scholar]
- 13.Tanaka Y, Nishida N, Sugiyama M, et al. Genome-wide association of IL28B with response to pegylated interferon-alpha and ribavirin therapy for chronic hepatitis C. Nat Genet. 2009;41(10):1105–1109. doi: 10.1038/ng.449. [DOI] [PubMed] [Google Scholar]
- 14.Suppiah V, Moldovan M, Ahlenstiel G, et al. IL28B is associated with response to chronic hepatitis C interferon-alpha and ribavirin therapy. Nat Genet. 2009;41(10):1100–1104. doi: 10.1038/ng.447. [DOI] [PubMed] [Google Scholar]
- 15.Li JH, Lao XQ, Tillmann HL, et al. Interferon-lambda genotype and low serum low-density lipoprotein cholesterol levels in patients with chronic hepatitis C infection. Hepatology. 2010;51(6):1904–1911. doi: 10.1002/hep.23592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cai T, Dufour JF, Muellhaupt B, et al. Viral genotype-specific role of PNPLA3, PPARG, MTTP and IL28B in hepatitis C virus-associated steatosis. J Hepatol. 2011;55(3):529–535. doi: 10.1016/j.jhep.2010.12.020. [DOI] [PubMed] [Google Scholar]
- 17.Tillmann HL, Patel K, Muir AJ, et al. Beneficial IL28B genotype associated with lower frequency of hepatic steatosis in patients with chronic hepatitis C. J Hepatol. 2011;55(6):1195–1200. doi: 10.1016/j.jhep.2011.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McHutchison JG, Lawitz EJ, Shiffman ML, et al. Peginterferon alfa-2b or alfa-2a with ribavirin for treatment of hepatitis C infection. N Engl J Med. 2009;361(6):580–593. doi: 10.1056/NEJMoa0808010. [DOI] [PubMed] [Google Scholar]
- 19.Fellay J, Thompson AJ, Ge D, et al. ITPA gene variants protect against anaemia in patients treated for chronic hepatitis C. Nature. 2010;464(7287):405–408. doi: 10.1038/nature08825. [DOI] [PubMed] [Google Scholar]
- 20.Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38(8):904–909. doi: 10.1038/ng1847. [DOI] [PubMed] [Google Scholar]
- 21.Purcell S, Neale B, Todd-Brown K, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81(3):559–575. doi: 10.1086/519795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brown ML, Inazu A, Hesler CB, et al. Molecular basis of lipid transfer protein deficiency in a family with increased high-density lipoproteins. Nature. 1989;342(6248):448–451. doi: 10.1038/342448a0. [DOI] [PubMed] [Google Scholar]
- 23.Pennacchio LA, Olivier M, Hubacek JA, et al. An apolipoprotein influencing triglycerides in humans and mice revealed by comparative sequencing. Science. 2001;294(5540):169–173. doi: 10.1126/science.1064852. [DOI] [PubMed] [Google Scholar]
- 24.Shinohara E, Yamashita S, Kihara S, et al. Interferon alpha induces disorder of lipid metabolism by lowering postheparin lipases and cholesteryl ester transfer protein activities in patients with chronic hepatitis C. Hepatology. 1997;25(6):1502–1506. doi: 10.1002/hep.510250632. [DOI] [PubMed] [Google Scholar]
- 25.Moriishi K, Mochizuki R, Moriya K, et al. Critical role of PA28γ in hepatitis C virus-associated steatogenesis and hepatocarcinogenesis. Proc Natl Acad Sci U S A. 2007;104(5):1661–1666. doi: 10.1073/pnas.0607312104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang AG, Lee DS, Moon HB, et al. Non-structural 5A protein of hepatitis C virus induces a range of liver pathology in transgenic mice. J Pathol. 2009;219(2):253–262. doi: 10.1002/path.2592. [DOI] [PubMed] [Google Scholar]
- 27.Poordad F, Bronowicki JP, Gordon SC, et al. IL28B polymorphism predicts virologic response in patients with hepatitis C genoytpe 1 treated with Boceprevir (BOC) combination therapy. J Hepatol. 2011;54(Suppl. 1):S6. [Google Scholar]
- 28.Pol S, Aerssens J, Zeuzem S, et al. Similar SVR rates in IL28B CC, CT or TT prior relapser, partial- or null responder patients treated with telaprevir/peginterferon/ribavirin: retrospective analysis of the REALIZE study. J Hepatol. 2011;54(Suppl. 1):S6. [Google Scholar]
- 29.Sarasin-Filipowicz M, Oakeley EJ, Duong FHT, et al. Interferon signaling and treatment outcome in chronic hepatitis C. Proc Natl Acad Sci U S A. 2008;105(19):7034–7039. doi: 10.1073/pnas.0707882105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen L, Borozan I, Sun J, et al. Cell-type specific gene expression signature in liver underlies response to interferon therapy in chronic hepatitis C infection. Gastroenterology. 2010;138(3):1123–1133. doi: 10.1053/j.gastro.2009.10.046. [DOI] [PubMed] [Google Scholar]
- 31.Honda M, Sakai A, Yamashita T, et al. Hepatic ISG expression is associated with genetic variation in interleukin 28B and the outcome of IFN therapy for chronic hepatitis C. Gastroenterology. 2010;139(2):499–509. doi: 10.1053/j.gastro.2010.04.049. [DOI] [PubMed] [Google Scholar]
- 32.Urban TJ, Thompson AJ, Bradrick SS, et al. IL28B genotype is associated with differential expression of intrahepatic interferon-stimulated genes in patients with chronic hepatitis C. Hepatology. 2010;52(6):1888–1896. doi: 10.1002/hep.23912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dill MT, Duong FHT, Vogt JE, et al. Interferon-induced gene expression is a stronger predictor of treatment response than IL28B genotype in patients with hepatitis C. Gastroenterology. 2011;140(3):1021–1031. doi: 10.1053/j.gastro.2010.11.039. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supporting Information: Additional Supporting Information may be found in the online version of this article:
Fig. S1 Quartiles of LDL_C by HCV RNA level during treatment at week 2, week 4 and week 12 timepoints.
Fig. S2 LDL_C (</>130 mg/dL) by HCV RNA level during treatment at week 2, week 4 and week 12 time-points.
Table S1: Measures of linkage disequilibrium between genome wide significant SNPs and rs12980275 by race.
Table S2: Multiple regression model for baseline LDL in Caucasians.
Table S3: Top GWAS association polymorphisms in association with total cholesterol in Caucasians.
Table S4: Top GWAS association polymorphisms in association with serum HDL-cholesterol in Caucasians.
Table S5: Top GWAS association polymorphisms in association with serum triglycerides in Caucasisans.
Table S6: Multiple regression model for LDL at 24 weeks post treatment for non-SVR patients.
Table S7: Multiple regression model for LDL at 24 weeks post treatment for patients who attained SVR.
Table S8: Comparison between treatment response at the timepoint LDL was measured.
Table S9: Comparison between genotypes within treatment response levels at the timepoint LDL was measured.
Table S10: Multiple logistic regression model for SVR.
Table S11: Multiple logistic regression model results testing interaction between LDL and IL28B genotype (rs12980275) for SVR.
Table S12: Univariate comparison of SVR rates by each IL28B genotype (rs12980275).