Abstract
Background
Experimental autoimmune encephalomyelitis (EAE) depends on the initial activation of CD4+ T cells responsive to myelin autoantigens. The key antigen presenting cell (APC) population that drives the activation of naïve T cells most efficiently is the dendritic cell (DC). As such, we should be able to trigger EAE by transfer of DC that can present the relevant autoantigen(s). Despite some sporadic reports, however, models of DC-driven EAE have not been widely adopted. We sought to test the feasibility of this approach and whether activation of the DC by toll-like receptor (TLR)-4 ligation was a sufficient stimulus to drive EAE.
Findings
Host mice were seeded with myelin basic protein (MBP)-reactive CD4+ T cells and then were injected with DC that could present the relevant MBP peptide which had been exposed to lipopolysaccharide as a TLR-4 agonist. We found that this approach induced robust clinical signs of EAE.
Conclusions
DC are sufficient as APC to effectively drive the differentiation of naïve myelin-responsive T cells into autoaggressive effector T cells. TLR-4-stimulation can activate the DC sufficiently to deliver the signals required to drive the pathogenic function of the T cell. These models will allow the dissection of the molecular requirements of the initial DC-T cell interaction in the lymphoid organs that ultimately leads to autoimmune pathology in the central nervous system.
Keywords: Multiple sclerosis, Experimental autoimmune encephalomyelitis, Dendritic cells, Myelin basic protein
Findings
The clinical manifestation of experimental autoimmune encephalomyelitis (EAE) depends on the initial activation of CD4+ T cells responsive to myelin autoantigens and their differentiation into autoaggressive effector T cells which, when re-isolated from the inflamed central nervous system (CNS) can produce a range of pro-inflammatory cytokines including IFN-γ, IL-17, TNF-α and granulocyte-macrophage colony-stimulating factor (GM-CSF) [1]. The immunological consensus is that the key antigen presenting cell (APC) population, capable of driving the activation of naïve T cells most efficiently, is the dendritic cell (DC) [2]. As such, many studies have shown that inoculation of mice with antigen-loaded DC can substitute for active immunization with the antigen in adjuvant to provoke a T cell response. Achieving this as a robust experimental option for the induction of EAE would allow us to probe the molecular requirements for the initial DC-T cell interaction in the lymphoid organs that ultimately leads to autoimmune pathology in the CNS. It is notable, however, that despite some sporadic reports [3-5], this has not as yet been achieved. Classically, the induction of EAE requires the use of complete Freund’s adjuvant (CFA), which contains a range of mycobacterial-derived molecules to trigger DC activation through their pattern-recognition receptors (PRRs). The most common means of activating DC prior to their use for cellular immunization is to use the toll-like receptor (TLR)-4 agonist lipopolysaccharide (LPS). Thus, one possible reason for the lack of a robust DC-driven model of EAE may be that LPS-activated DC cannot provide all of the signals to the naïve myelin-response T cells to engender strong autoaggressive function.
To formally test this possibility, we sought to induce EAE using bone marrow-derived DC (BMDC) that had received only the TLR4-signal in vitro, by activation with LPS. To maximize our chances of success we first sought to obviate two possible confounding issues – the size of the naïve myelin-responsive repertoire and the ability of the DC to present the autoantigen. We therefore started by developing a transgenic two-cell transfer model. We have previously reported that C57BL/6xB10.PL mice are resistant to EAE induction using the Ac1-9 peptide of myelin basic protein (MBP), unless they are first seeded with a cohort of naïve MBP-responsive T cells derived from the Tg4 T cell receptor (TCR) transgenic mouse [6]. This approach therefore allowed us to control the size of the Ac1-9-responsive T cell repertoire. To remove the possibility that BMDC may for some reason fail to present the Ac1-9 peptide efficiently, we made use of a second transgenic line, the AMK35 mouse, in which major histocompatibility molecule (MHC) class II-expressing cells constitutively express the Ac1-9 peptide, covalently bound to the AU molecule [7].
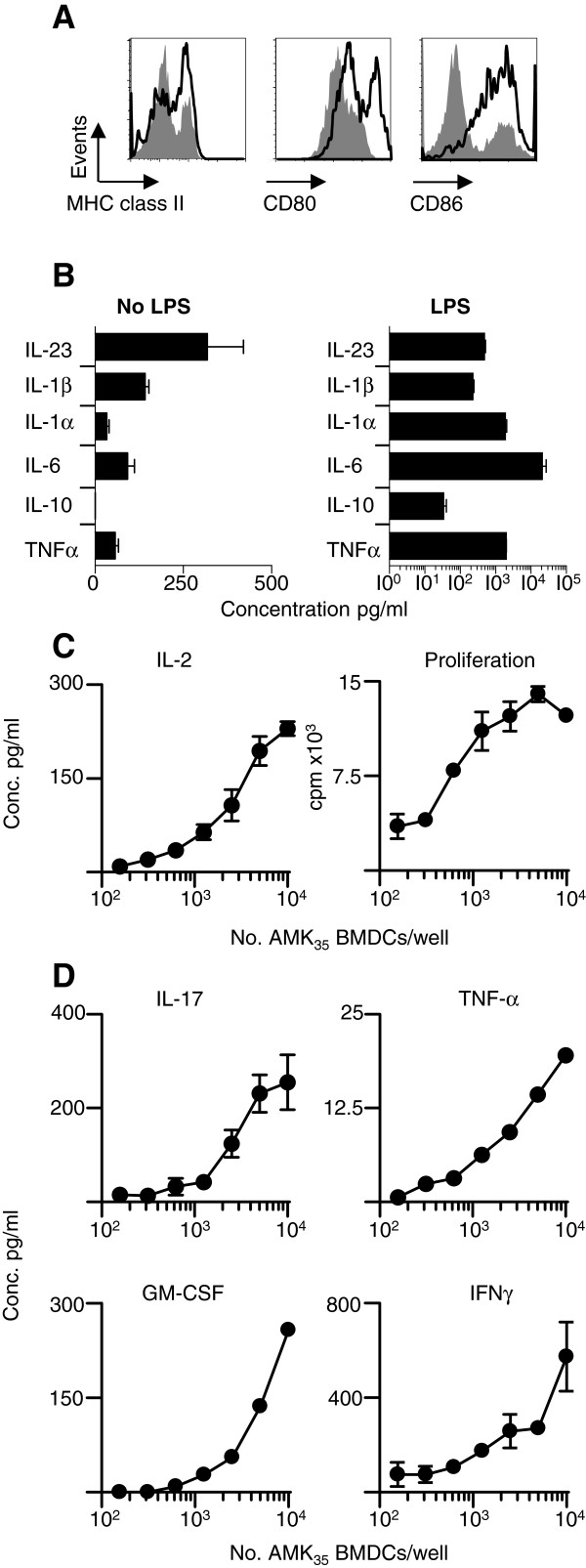
BMDC generated from AMK35 mice showed elevated expression of Au, CD80 and CD86 after overnight exposure to LPS (Figure 1A) and supernatants from these cultures contained elevated levels of pro-inflammatory cytokines (Figure 1B), notably IL-1β, IL-6 and IL-23, each of which has been described as being required for EAE development using gene-deficient mice [8-10]. These LPS-activated AMK35 DC also efficiently induced the in vitro proliferation of naïve Tg4 CD4+ T cells (Figure 1C), and their production of IFN-γ, IL-17, TNF-α and GM-CSF (Figure 1D), without any addition of exogenous antigen to the culture. Thus, LPS-activated DC appeared to have many of the key attributes associated with EAE pathology. To test this in vivo, we seeded C57BL/6xB10.PL mice with a cohort of naïve Tg4 CD4+ T cells and one-day later subcutaneously administered LPS-activated AMK35 DC. Ten out of 12 mice developed clinical signs of EAE (Figure 2A). However, the extent and, importantly, the timing of onset of clinical signs showed variation.
Figure 1.
MBP loaded BMDC can prime MBP responsive T cells. BMDC were generated in the presence of 20ng/ml of recombinant GM-CSF for nine days as previously described [11] and activated with 0.1μg/ml LPS and 5ng/ml of GM-CSF for an additional 18 hours. A) Cells were stained for MHC class II, CD80, CD86 expression with (open histogram) or without (grey histogram) LPS maturation and analyzed by FACS. B) Cytokine concentrations in BMDC supernatants were sampled after 18-hour culture with or without LPS. C and D) To study the primary activation of Tg4 T cells, varying numbers (as stated) of AMK35[7] BMDC were cultured with 2x104 CD4+ Tg4.CD45.1+ T cells [12] per well. Cell proliferation was assessed by thymidine incorporation. The results are expressed as mean counts per minute ± standard error of the mean. Tg4 T cell production of cytokines (IL-2, IL-17A, GM-CSF, TNF-α and IFN-γ) was assessed in culture supernatants by ELISA. IL-2 was measured in supernatants after 48 hours of culture and IFN-γ, TNF-α, GM-CSF and IL-17A were measured after 72 hours of culture. Data are representative of four independent experiments. BMDC, bone marrow-derived dendritic cells;FACS, fluorescence activated cell sorting; GM-CSF, granulocyte-macrophage colony-stimulating factor; LPS, lipopolysaccharide; MBP, myelin basic protein.
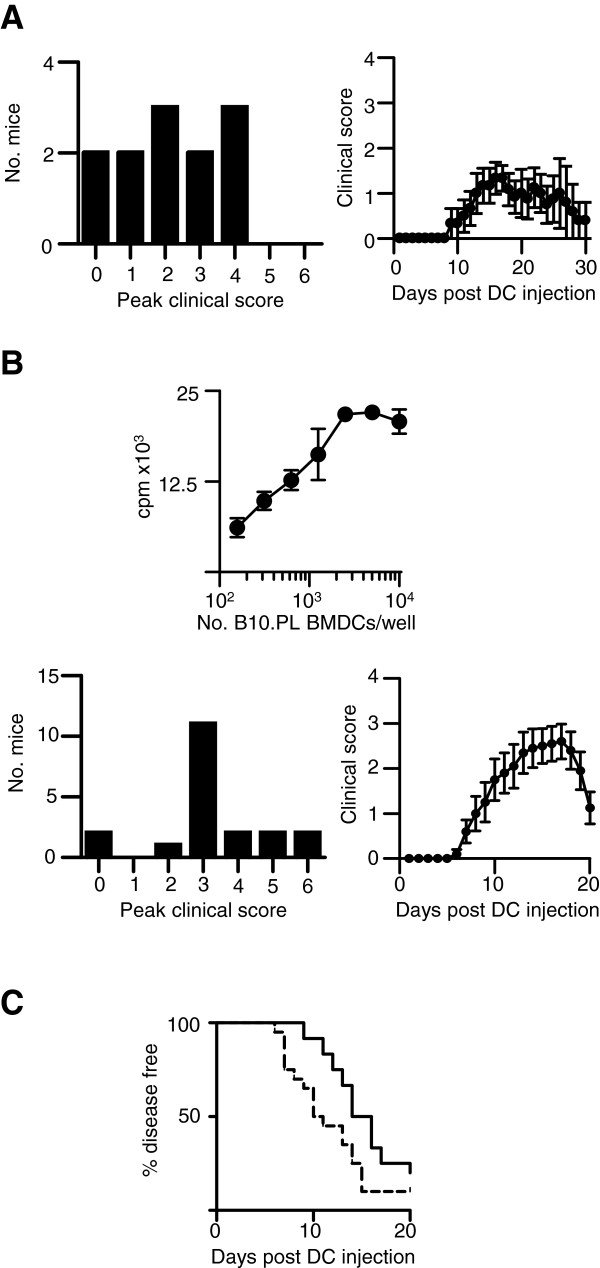
Figure 2.
MBP loaded BMDC can initiate EAE in mice seeded with MBP-reactive T cells. BMDC were generated from AMK35 or B10.PL mice and matured with LPS as described in Figure 1. A total of 2x106 Tg4.CD45.1 CD4+ T cells was transferred into B10.PLxC57BL/6 mice one day prior to s.c. injection of 2x106 LPS-conditioned AMK35 BMDC (A) or B10.PL BMDC pulsed with 0.1μM MBP Ac1-9(4Tyr) analog peptide (B). of Pertussis toxin was administered (200 ngi.p.) at the time of BMDC transfer. Clinical signs of EAE were assessed daily with the following scoring system: 0, no signs; 1, flaccid tail; 2, impaired righting reflex and/or gait; 3, partial hind limb paralysis; 4, total hind limb paralysis; 5, hind limb paralysis with partial front limb paralysis; 6, moribund or dead. A) Plots show maximal clinical scores of mice administered AMK35 BMDC and the clinical disease curve of three pooled, independent experiments. B) A total of 2x104 Tg4 CD4+ T cells was cultured with LPS-conditioned B10.PL BMDC pulsed with 0.1μM MBP Ac1-9(4Tyr) and assessed for proliferation (thymidine incorporation). Plots show maximal clinical scores of mice administered B10.PL BMDC pulsed with 0.1μM MBP Ac1-9(4Tyr) and the clinical disease curve of three pooled, independent experiments. C) Plot showing timing of disease onset in mice administered AMK35 DC (black line, n=12) and MBP Ac1-9(4Tyr) pulsed DC (dashed line, n=20). All mice were bred under specific pathogen-free conditions at the University of Edinburgh and all experiments had local ethical approval and were performed in accordance with UK legislation. BMDC, bone marrow-derived dendritic cells; DC, dendritic cells; EAE, experimental autoimmune encephalomyelitis; LPS, lipopolysaccharide; MBP, myelin basic protein.
The use of BMDC derived from mice in which antigen presentation is driven by a transgene, of course, has limitations. For example, it can be envisaged that it would be desirable to study DC expressing multiple autoantigenic peptides, either separately or together. To extend our study in this direction we therefore generated BMDC from AU+ non-transgenic BMDC, from B10.PL mice. These BMDC were activated with LPS and loaded with the Ac1-9(4Tyr) peptide in vitro. This variant peptide binds to the Au molecule with high affinity, allowing stable antigen presentation (unlike the wild type Ac1-9 peptide). These antigen-loaded non-transgenic DC could efficiently activate naïve Tg4 T cells in vitro (Figure 2B) and induced a robust disease course in the T cell/DC transfer model in C57BL/6xB10.PL mice (Figure 2B). Of note, there was a trend for the onset of disease to be earlier and more synchronous than achieved with AMK35 DC (Figure 2C).
The two novel models of DC-driven EAE we have described have a number of advantages over the few earlier reports of EAE following transfer of autoantigen-loaded DC. Previous studies using BMDC pulsed with the 35–55 peptide of myelin oligodendrocyte glycoprotein (MOG) in C57BL/6 mice have required repeated injections of BMDC to induce EAE [3] or have required concurrent administration of CFA to drive robust disease [5]. A previous report of EAE involving MBP-pulsed DC differed markedly from ours in that five times as many MBP-reactive T cells were transferred and the host mice were irradiated prior to cell transfer [4]. Of note, the wild type MBP peptide (lacking the insertion of a Tyr residue at position 4) has such low affinity for Au that APC cannot be pulsed with that peptide to allow efficient loading into the Au peptide binding groove [13]. It is therefore difficult to reconcile that observation with the reported ability of DC loaded with the wild type peptide to activate MBP-responsive T cells for the initiation of EAE [4]. Unlike that previous report, we used two systems in which the presentation of the autoantigenic peptide-MHC complex by the DC was highly stable. AMK35 DC, expressing transgenic peptide-MHC [7] induced variable degrees of EAE, particularly in terms of day of onset, but this difficulty was overcome using non-transgenic DC pulsed with the Ac1-9(4Tyr) peptide, which shows extraordinary affinity for the AU molecule [14]. DC that are loaded with this peptide in vivo can maintain expression of the peptide-MHC complex for at least seven days (SMA, unpublished). Given that the consensus is that BMDC probably persist for only a few days after administration, we can be confident that the DC we transferred were presenting the peptide to T cells for a sustained length of time. It is conceivable that the MOG(35–55)-Ab complex might be prone to more rapid degradation in DC transfer experiments. Nevertheless, it is evident from other studies that the use of MOG(35–55)-loaded C57BL/6 DC can be sufficient to provide TCR-signaling in vivo, as we ourselves have shown [15,16], but it was not sufficient to trigger robust EAE, which still required subsequent immunization with CFA in those models.
A key paradigm is that autoreactive T cells can develop into autoaggressive T cells if they recognize (self, or cross-reactive non-self) antigen presented during infection (that is, on activated APC). The use of CFA to induce EAE mimics infection due to the presence of heat-killed mycobacteria. However, CFA contains multiple moieties capable of stimulating APC through a range of PRRs. Although LPS is not one of these moieties, heat shock proteins 65 and 70 have been described as TLR-4 agonists [17], as has pertussis toxin [18], which is routinely used during EAE induction. Less complex adjuvants, including LPS, have been reported to be able to substitute for CFA [19-22], but results have been variable between studies. Indeed, exposure to LPS can protect against EAE if it occurs before immunization with autoantigen in CFA [23]. Those studies have not provided clear information on which APC need to be activated through a given PRR to drive pathology, or which of the multiple consequences of PRR-ligation are non-redundant for the induction of EAE. Furthermore, it is clear that expression of the same TLR on different immune cells can have markedly different effects on disease outcome [24,25].
From our experiments described here, we can conclude that a) DC are sufficient as APC to effectively drive the differentiation of naïve myelin-responsive T cells into autoaggressive effector T cells and b) exposure to a TLR-4 agonist can activate the DC sufficiently to deliver the signals required to drive the pathogenic function of the T cell. These new model systems will therefore allow comparison of a range of PRR stimuli upon the DC and the use of gene-knockout/knockdown DC to help to probe the key molecular events in the initial DC-T cell dialogue that lead ultimately to autoimmune CNS inflammation in EAE.
Abbreviations
APC: Antigen presenting cell; BMDC: Bone marrow derived dendritic cell; CFA: Complete Freund’s adjuvant; CNS: Central nervous system; DC: Dendritic cell; EAE: Experimental autoimmune encephalomyelitis; ELISA: Enzyme-linked immunosorbent assay; FACS: Fluorescence activated cell sorting; GM-CSF: Granulocyte-macrophage colony-stimulating factor; IFN: Interferon; IL: Interleukin; LPS: Lipopolysaccharide; MBP: Myelin basic protein; MHC: Major histocompatibility molecules; MOG: Myelin oligodendrocyte glycoprotein; PRRs: Pattern-recognition receptors; TCR: T cell receptor; TLR: Toll-like receptor; TNF: Tumor necrosis factor; Tyr: Yrosine.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
RJM, ASM and SMA designed the experiments. RJM, HC, DT, ROC and MDL performed the experiments. RJM and SMA drafted the manuscript. FCK and BA generated the AMK35 transgenic mouse. All authors have read and approved the final manuscript.
Contributor Information
Richard J Mellanby, Email: Richard.Mellanby@ed.ac.uk.
Helen Cambrook, Email: H.E.Cambrook@sms.ed.ac.uk.
Darryl G Turner, Email: Darryl.Turner@ed.ac.uk.
Richard A O’Connor, Email: R.OConnor@ed.ac.uk.
Melanie D Leech, Email: mel.leech@ed.ac.uk.
Florian C Kurschus, Email: kurschus@uni-mainz.de.
Andrew S MacDonald, Email: andrew.macdonald@ed.ac.uk.
Bernd Arnold, Email: b.arnold@dkfz-heidelberg.de.
Stephen M Anderton, Email: steve.anderton@ed.ac.uk.
Acknowledgements
This work was supported by grants for the Medical Research Council and the Wellcome Trust. RJM was funded by a Wellcome Trust Veterinary Postdoctoral Fellowship.
References
- Petermann F, Korn T. Cytokines and effector T cell subsets causing autoimmune CNS disease. FEBS Lett. 2011;585:3747–3757. doi: 10.1016/j.febslet.2011.03.064. [DOI] [PubMed] [Google Scholar]
- Steinman RM. Lasker Basic Medical Research Award. Dendritic cells: versatile controllers of the immune system. Nat Med. 2007;13:1155–1159. doi: 10.1038/nm1643. [DOI] [PubMed] [Google Scholar]
- Aghdami N, Gharibdoost F, Moazzeni SM. Experimental autoimmune encephalomyelitis (EAE) induced by antigen pulsed dendritic cells in the C57BL/6 mouse: influence of injection route. ExpAnim. 2008;57:45–55. doi: 10.1538/expanim.57.45. [DOI] [PubMed] [Google Scholar]
- Dittel BN, Visintin I, Merchant RM, Janeway CA Jr. Presentation of the self antigen myelin basic protein by dendritic cells leads to experimental autoimmune encephalomyelitis. J Immunol. 1999;163:32–39. [PubMed] [Google Scholar]
- Weir CR, Nicolson K, Backstrom BT. Experimental autoimmune encephalomyelitis induction in naive mice by dendritic cells presenting a self-peptide. Immunol Cell Biol. 2002;80:14–20. doi: 10.1046/j.1440-1711.2002.01056.x. [DOI] [PubMed] [Google Scholar]
- Ryan KR, McCue D, Anderton SM. Fas-mediated death and sensory adaptation limit the pathogenic potential of autoreactive T cells after strong antigenic stimulation. J LeukocBiol. 2005;78:43–50. doi: 10.1189/jlb.0205059. [DOI] [PubMed] [Google Scholar]
- Kurschus FC, Oelert T, Liliensiek B, Buchmann P, Wraith DC, Hammerling GJ, Arnold B. Experimental autoimmune encephalomyelitis in mice expressing the autoantigen MBP 1–10 covalently bound to the MHC class II molecule I-Au. IntImmunol. 2006;18:151–162. doi: 10.1093/intimm/dxh357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okuda Y, Sakoda S, Bernard CC, Fujimura H, Saeki Y, Kishimoto T, Yanagihara T. IL-6-deficient mice are resistant to the induction of experimental autoimmune encephalomyelitis provoked by myelin oligodendrocyte glycoprotein. IntImmunol. 1998;10:703–708. doi: 10.1093/intimm/10.5.703. [DOI] [PubMed] [Google Scholar]
- Sutton C, Brereton C, Keogh B, Mills KH, Lavelle EC. A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J Exp Med. 2006;203:1685–1691. doi: 10.1084/jem.20060285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, Lucian L, To W, Kwan S, Churakova T, Zurawski S, Wiekowski M, Lira SA, Gorman D, Kastelein RA, Sedgwick JD. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature. 2003;421:744–748. doi: 10.1038/nature01355. [DOI] [PubMed] [Google Scholar]
- MacDonald AS, Straw AD, Dalton NM, Pearce EJ. Cutting edge: Th2 response induction by dendritic cells: a role for CD40. J Immunol. 2002;168:537–540. doi: 10.4049/jimmunol.168.2.537. [DOI] [PubMed] [Google Scholar]
- Liu GY, Fairchild PJ, Smith RM, Prowle JR, Kioussis D, Wraith DC. Low avidity recognition of self-antigen by T cells permits escape from central tolerance. Immunity. 1995;3:407–415. doi: 10.1016/1074-7613(95)90170-1. [DOI] [PubMed] [Google Scholar]
- Fairchild PJ, Wildgoose R, Atherton E, Webb S, Wraith DC. An autoantigenic T cell epitope forms unstable complexes with class II MHC: a novel route for escape from tolerance induction. IntImmunol. 1993;5:1151–1158. doi: 10.1093/intimm/5.9.1151. [DOI] [PubMed] [Google Scholar]
- Anderton SM, Manickasingham SP, Burkhart C, Luckcuck TA, Holland SJ, Lamont AG, Wraith DC. Fine specificity of the myelin-reactive T cell repertoire: implications for TCR antagonism in autoimmunity. J Immunol. 1998;161:3357–3364. [PubMed] [Google Scholar]
- Hochweller K, Anderton SM. Systemic administration of antigen-loaded CD40-deficient dendritic cells mimics soluble antigen administration. Eur J Immunol. 2004;34:990–998. doi: 10.1002/eji.200324782. [DOI] [PubMed] [Google Scholar]
- Perona-Wright G, Jenkins SJ, O'Connor RA, Zienkiewicz D, McSorley HJ, Maizels RM, Anderton SM, MacDonald AS. A pivotal role for CD40-mediated IL-6 production by dendritic cells during IL-17 induction in vivo. J Immunol. 2009;182:2808–2815. doi: 10.4049/jimmunol.0803553. [DOI] [PubMed] [Google Scholar]
- Bulut Y, Michelsen KS, Hayrapetian L, Naiki Y, Spallek R, Singh M, Arditi M. Mycobacterium tuberculosis heat shock proteins use diverse Toll-like receptor pathways to activate pro-inflammatory signals. J BiolChem. 2005;280:20961–20967. doi: 10.1074/jbc.M411379200. [DOI] [PubMed] [Google Scholar]
- Kerfoot SM, Long EM, Hickey MJ, Andonegui G, Lapointe BM, Zanardo RC, Bonder C, James WG, Robbins SM, Kubes P. TLR4 contributes to disease-inducing mechanisms resulting in central nervous system autoimmune disease. J Immunol. 2004;173:7070–7077. doi: 10.4049/jimmunol.173.11.7070. [DOI] [PubMed] [Google Scholar]
- Segal BM, Chang JT, Shevach EM. CpG oligonucleotides are potent adjuvants for the activation of autoreactiveencephalitogenic T cells in vivo. J Immunol. 2000;164:5683–5688. doi: 10.4049/jimmunol.164.11.5683. [DOI] [PubMed] [Google Scholar]
- Wolf NA, Amouzegar TK, Swanborg RH. Synergistic interaction between Toll-like receptor agonists is required for induction of experimental autoimmune encephalomyelitis in Lewis rats. J Neuroimmunol. 2007;185:115–122. doi: 10.1016/j.jneuroim.2007.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Visser L, de Jan-Heer H, Boven LA, van Riel D, van Meurs M, Melief MJ, Zahringer U, van Strijp J, Lambrecht BN, Nieuwenhuis EE, Laman JD. Proinflammatory bacterial peptidoglycan as a cofactor for the development of central nervous system autoimmune disease. J Immunol. 2005;174:808–816. doi: 10.4049/jimmunol.174.2.808. [DOI] [PubMed] [Google Scholar]
- Tigno-Aranjuez JT, Jaini R, Tuohy VK, Lehmann PV, Tary-Lehmann M. Encephalitogenicity of complete Freund's adjuvant relative to CpG is linked to induction of Th17 cells. J Immunol. 2009;183:5654–5661. doi: 10.4049/jimmunol.0900645. [DOI] [PubMed] [Google Scholar]
- Buenafe AC, Bourdette DN. Lipopolysaccharide pretreatment modulates the disease course in experimental autoimmune encephalomyelitis. J Neuroimmunol. 2007;182:32–40. doi: 10.1016/j.jneuroim.2006.09.004. [DOI] [PubMed] [Google Scholar]
- Lampropoulou V, Hoehlig K, Roch T, Neves P, Calderon-Gomez E, Sweenie CH, Hao Y, Freitas AA, Steinhoff U, Anderton SM, Fillatreau S. TLR-activated B cells suppress T cell-mediated autoimmunity. J Immunol. 2008;180:4763–4773. doi: 10.4049/jimmunol.180.7.4763. [DOI] [PubMed] [Google Scholar]
- Lampropoulou V, Calderon-Gomez E, Roch T, Neves P, Shen P, Stervbo U, Boudinot P, Anderton SM, Fillatreau S. Suppressive functions of activated B cells in autoimmune diseases reveal the dual roles of Toll-like receptors in immunity. Immunol Rev. 2010;233:146–161. doi: 10.1111/j.0105-2896.2009.00855.x. [DOI] [PubMed] [Google Scholar]