Abstract
Cell cycle regulators, such as cyclins, are often upregulated in many proliferative disorders, and Cyclin A2 is generally considered as a marker of aggressive cancers. Our recent work, which revealed decreased expression of Cyclin A2 upon metastasis of colorectal cancer, suggests a more complicated situation. Consistent with this, we identified a role for Cyclin A2, via RhoA, in regulation of the actin cytoskeleton and the control of cell invasion. Cyclin A2 also regulates spindle orientation which, when misoriented, could disrupt cell polarity and favor cancer cell detachment from the tumor as part of a transforming process, such as epithelial to mesenchymal transition (EMT). During EMT, cells undergo morphological and molecular changes toward a mesenchymal phenotype. Upregulation, or increased activity of some Rho GTPases, such as Cdc42, Rac1 or RhoC, increases the invasive potential of these cells. This correlates with the inverse relationship between RhoA and RhoC activities we observed in an epithelial cell type. Altogether, these observations raise the possibility that Cyclin A2 is instrumental in preventing EMT and therefore cancers of epithelial tissues.
Keywords: RhoA, RhoC, cell polarity, centrosome, epithelium, golgi, mitotic spindle
Recent data pointing to a novel function of Cyclin A2 add another component to the complex regulatory network that involves cell cycle regulators and cytoskeletal structures participating in the control of cell movement.1 As already known, Cyclin A2 is a key regulator of cell division, since it controls both S phase and G2/M transition in association with CDK2 and CDK1, respectively.2 During S phase, Cyclin A2 regulates the initiation and progression of DNA synthesis, and at the G2/M transition, it plays a critical role in triggering Cyclin B1-CDK1 activation.3-5 In mice, it is essential in embryonic cells and in the hematopoietic lineage, yet dispensable in fibroblasts.6,7
Surprisingly, depletion of Cyclin A2 is sufficient to increase cell motility of fibroblasts in 2D assays and cooperates with oncogenic transformation to increase their invasiveness in 3D collagen matrixes.1 Cyclin A2-deficient cells contain a perturbed cytoskeleton, where Actin filaments are cortical and the distribution of focal adhesions is altered. Interestingly, these defects are corrected by a Cyclin A2 mutant unable to activate its cognate kinases, CDK1 and CDK2. This is associated with a downregulation of the RhoA-ROCK pathway and decreased phosphorylation of Cofilin, which is involved in the reorganization of Actin filaments, consecutively leading to an increased cell migration and invasion. Importantly, pharmacological inhibition of ROCK in control fibroblasts leads to an increase in migration velocity similar to that of Cyclin A2-depleted cells.
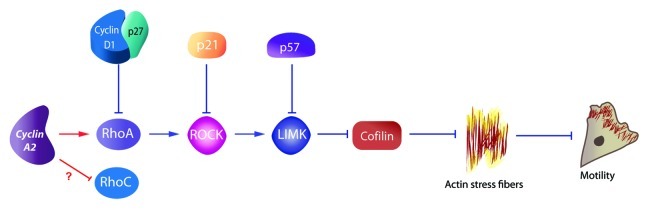
Cyclin D1 and the CDK inhibitors p21, p27 and p57 had also previously been shown to impinge upon the RhoA/ROCK pathway. Cyclin D1 binds directly to p27 and thereby blocks RhoA activation by inhibiting interaction with its GEF.8-11 Similarly, cytoplasmic p21 has been shown to bind and inhibit ROCK1, which promotes neurite extension by neuroblastoma cells and hippocampal neurons,12 while p57 was shown to sequester LIMK (Fig. 1).13 Within this scheme, Cyclin A2 binds directly to RhoA and facilitate its GTP loading by GEFs.1 This is consistent with the involvement of this GTPase in early mitosis, since its increased activity at that time leads to cortical retraction and cell rounding via its downstream effector ROCK.14 Moreover, formation of the contractile ring during cytokinesis has also been shown to depend upon RhoA activation in a precise zone at the cell equator.15
Figure 1. Cell cycle regulators and the Rho/ROCK pathway. Whereas Cyclin D1, p27, p21, and p57 collectively define the inhibitory arm of a regulatory loop linking the cell cycle to the RhoA/ROCK pathway, Cyclin A2 appears to be the first component in its activator arm.
Knockdown of Cyclin A2 and inhibition of CDK2 prevent cells from forming stable attachments of their mitotic spindle to the cell cortex.16 This resulted in the spindles failing to locate to the central position in the cells and undergo dramatic rotation. Moreover, Cyclin A2-CDK2 specifically associated with APC in late G2 phase and phosphorylated it on Ser1360. Mutation of this serine to alanine results in identical off-centered mitotic spindles. Thus, this Cyclin A2-CDK2-dependent phosphorylation within the mutation cluster region of APC affects astral microtubule attachment to the cortical surface in mitosis.
Another potential player in this complex ballet appears to be the Golgi apparatus. Recent studies suggest the existence of functional interactions between this organelle and the centrosome, the structure responsible for the nucleation of the mitotic spindle. The Golgi and the centrosome are in a close proximity at interphase and this intimacy is transiently lost during mitosis, when the Golgi is fragmented, and the two newly duplicated centrosomes migrate. Golgi-centrosomes interactions may play a central role in cell polarization, since both structures undergo re-orientation toward the leading edge of a migrating cell (for a review see ref. 17). Proteins of the Golgi apparatus are likely to be instrumental in centrosome organization and positioning, and microtubules nucleated at the centrosome seem to play the same role for the Golgi. CyclinA2 has also been shown to localize to the centrosome in a CDK-independent manner, and through binding of MCM5 and Orc1, prevents the formation of supernumerary centrosomes.18 Since Cdc42 plays a central role in cell polarization, this raises the question of its role in the potential coupling of this process to centrosome duplication and spindle orientation. A partial answer to this question was provided recently. Bray et al. showed that Cdc42 controls spindle orientation to position the apical surface during epithelial morphogenesis, at least in vitro.19
These data highlight thus another aspect of Cyclin A2 function in the control of cytoskeleton dynamics, which places this cyclin at the crossroads of intracellular processes, such as mitosis, and extracellular cues emanating from neighboring cells or from the extracellular matrix (Fig. 2).
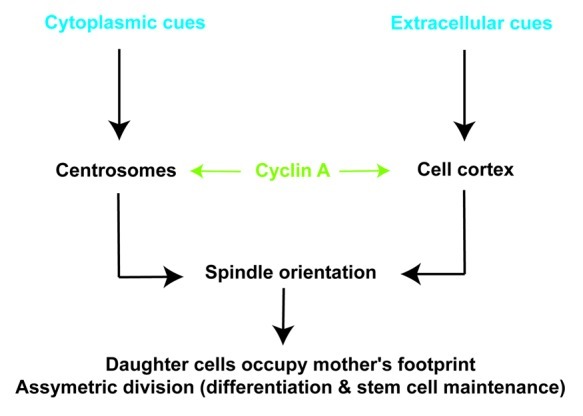
Figure 2. Cyclin A2 is at the interface between cytoplasmic and extracellular cues. When Cyclin A2 level is high, the two daughter cells orientate their bipolar mitotic spindles according to both their mother footprint and cues emanating from the lamina. If Cyclin A2 is abnormally downregulated, cell division is non-localized, cell spreading is isotropic and cells show increased motility.
If we place these observations within the context of the epithelium, the orientation of the mitotic spindle with respect to the lamina is a major issue, since this has been proposed to be instrumental in the determination of the fate of the two daughter cells. Moreover, in tumors, basal extrusion of a daughter cell could initiate metastasis in some situations.20 Therefore, mutations that couple spindle misorentiation with oncogene activation could facilitate tumor progression and metastasis spreading.
Until recently, Cyclin A2 was solely considered as a proliferation marker since, consistent with its known functions during the cell cycle, it is frequently overexpressed in highly proliferative cancers.3 However, our observations indicate that Cyclin A2 protein levels are significantly lower in an invasive colon carcinoma cell line derived from lymph node metastasis relative to a less-invasive counterpart issued from the primary site. More importantly, the same is observed upon metastasis of colon adenocarcinoma.1 Studies of renal, colorectal carcinoma and prostate cancer, found that proliferative tumors with low levels of Cyclin A2 were more aggressive than those with high Cyclin A2 expression.21-23 Moreover, Wang et al. established that Cyclin A2 levels were inversely correlated with invasiveness of oral squamous cell carcinoma (OSCC) both in vitro and in vivo.24
Altogether, these data indicate that Cyclin A2 downregulation could be an important step in the acquisition of an invasive property by epithelial cells, and thereby call for more studies on the involvement of this cyclin in the morphological changes that occur during metastasis. Along these lines, the epithelial to mesenchymal transition (EMT) appears to be instrumental in pathological situations such as fibrosis, tumor development and metastasis spreading.25-27 In the context of epithelial cancer, EMT provides one mechanism for tumor cells to invade the local tissue and blood vessels, setting the stage for metastatic spread. Therefore, EMT is hypothesized to contribute to tumor progression, and clinical evidence suggests that upregulation of EMT regulators in cancer cells correlates with poor patient outcome and tumor aggressiveness.28-30 EMT is likely to be triggered by complex networks of signals emanating from the tumor stroma, along with a variety of cytokines and growth factors, such as transforming growth factor β (TGFβ),31 epidermal growth factor (EGF),32 fibroblast growth factor (FGF)33 andhepatocyte growth factor (HGF).34 Consistent with this, Ras and TGFβ have been shown to cooperate in promoting EMT.35 During this process, epithelial cells undergo morphological remodeling toward a more elongated and fibroblastic morphology. Among these changes, cells lose cell-to-cell junctions, undergo Actin cytoskeleton rearrangement and acquire enhanced invasive properties. At the molecular level, EMT is characterized by a loss of E-cadherin, and the acquisition of mesenchymal markers, such as Vimentin, Fibronectin or N-cadherin. Transcription factors, such as Snail, Slug, Twist or Zeb-1 and 2, have been implicated in this process.36 For some cell types, the acquisition of a more motile phenotype during EMT has been attributed to the increased expression and activation of Rho GTPases, such as Rac1, Cdc42 and RhoC.37-39
Accordingly, overexpression of RhoC enhances the ability of melanoma cells to exit the circulatory system and colonize the lungs. Consistent with this, loss of RhoC does not affect tumor development while it leads to a drastic inhibition of metastasis. Nevertheless, RhoC-deficient mice are viable, indicating that this GTPase is dispensable for embryonic and post-natal development.40 RhoC and RhoA share more than 95% sequence similarity, thus it is not surprising that Cyclin A2 interacts as well with both GTPases.1,41 Indeed, when Cyclin A2 is knocked down in epithelial cells, such as normal mouse mammary epithelial cells (NMuMG), they exibit a strong downregulation of RhoA activity and an increase in RhoC activity (our unpublished observation).
Several reports support a possible role for RhoC in the EMT-related invasion and in metastasis spreading. As mentioned earlier, the breeding of RhoC-deficient mice to the tumor and metastasis-prone MMTV-PyMT strain established the requirement for RhoC in metastasis to the lungs.40 Consistent with this, increased RhoC expression in adenocarcinoma of pancreas,42 along with hepatocellular,43 breast,44 ovarian,45 bladder and esophageal cancers, has been observed and correlated with progression and poor prognosis.46 Moreover, invasion by several cell types, such as breast,47 colon carcinoma,48 or prostate cancer cell lines,49 was shown to be dependent on RhoC expression and activation. With regards to EMT, recent studies have established that increased expression and/or RhoC activation promotes invasion, whereas Rac1 and Cdc42 were generally considered as major players in this process.38,48 In these studies, increased RhoC activity is correlated with an increased transcription of the gene. This transcription is induced by upregulation of Twist and miR-10b, or is dependent on the transcription factor Ets-1 in breast cancer and colon carcinoma cell lines, respectively.38,48
In conclusion, these data highlight the intricate relationship between Cyclin A2 expression and Rho GTPases activity within the context of cell adhesion and motility. While Cdc42 had previously been shown to be instrumental for anchorage-independent expression of the Cylin A2 gene in primary mouse embryonic fibroblasts,50 this cyclin appears now to feed back on the activity of other GTPases, such as RhoA and RhoC. Altogether, these observations indicate that CyclinA2 is a potential novel player in the complex regulation of EMT, moreover, they reinforce the idea that cell cycle regulators are much more than just cell cycle regulators.
Acknowledgments
We thank Robert Hipskind for his kind review of our manuscript. This work and N. Arsic were supported by grants from Agence Nationale de la Recherche (BLANC06–3_142605), the Marie Curie reintegration program (MIRG-CT-2006–044922) and Association pour la Recherche contre le Cancer. N. Bendris was supported by a fellowship from the French Ministry of Education and Research and Fondation pour la Recherche Médicale.
Footnotes
Previously published online: www.landesbioscience.com/journals/smallgtpases/article/20791
References
- 1.Arsic N, Bendris N, Peter M, Begon-Pescia C, Rebouissou C, Gadéa G, et al. A novel function for Cyclin A2: control of cell invasion via RhoA signaling. J Cell Biol. 2012;196:147–62. doi: 10.1083/jcb.201102085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pagano M, Pepperkok R, Verde F, Ansorge W, Draetta G. Cyclin A is required at two points in the human cell cycle. EMBO J. 1992;11:961–71. doi: 10.1002/j.1460-2075.1992.tb05135.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yam CH, Fung TK, Poon RY. Cyclin A in cell cycle control and cancer. Cell Mol Life Sci. 2002;59:1317–26. doi: 10.1007/s00018-002-8510-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fung TK, Ma HT, Poon RY. Specialized roles of the two mitotic cyclins in somatic cells: cyclin A as an activator of M phase-promoting factor. Mol Biol Cell. 2007;18:1861–73. doi: 10.1091/mbc.E06-12-1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.De Boer L, Oakes V, Beamish H, Giles N, Stevens F, Somodevilla-Torres M, et al. Cyclin A/cdk2 coordinates centrosomal and nuclear mitotic events. Oncogene. 2008;27:4261–8. doi: 10.1038/onc.2008.74. [DOI] [PubMed] [Google Scholar]
- 6.Kalaszczynska I, Geng Y, Iino T, Mizuno S, Choi Y, Kondratiuk I, et al. Cyclin A is redundant in fibroblasts but essential in hematopoietic and embryonic stem cells. Cell. 2009;138:352–65. doi: 10.1016/j.cell.2009.04.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Murphy M, Stinnakre MG, Senamaud-Beaufort C, Winston NJ, Sweeney C, Kubelka M, et al. Delayed early embryonic lethality following disruption of the murine cyclin A2 gene. Nat Genet. 1997;15:83–6. doi: 10.1038/ng0197-83. [DOI] [PubMed] [Google Scholar]
- 8.Li Z, Jiao X, Wang C, Ju X, Lu Y, Yuan L, et al. Cyclin D1 induction of cellular migration requires p27(KIP1) Cancer Res. 2006;66:9986–94. doi: 10.1158/0008-5472.CAN-06-1596. [DOI] [PubMed] [Google Scholar]
- 9.Li Z, Wang C, Prendergast GC, Pestell RG. Cyclin D1 functions in cell migration. Cell Cycle. 2006;5:2440–2. doi: 10.4161/cc.5.21.3428. [DOI] [PubMed] [Google Scholar]
- 10.Li Z, Wang C, Jiao X, Lu Y, Fu M, Quong AA, et al. Cyclin D1 regulates cellular migration through the inhibition of thrombospondin 1 and ROCK signaling. Mol Cell Biol. 2006;26:4240–56. doi: 10.1128/MCB.02124-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Besson A, Gurian-West M, Schmidt A, Hall A, Roberts JM. p27Kip1 modulates cell migration through the regulation of RhoA activation. Genes Dev. 2004;18:862–76. doi: 10.1101/gad.1185504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tanaka H, Yamashita T, Asada M, Mizutani S, Yoshikawa H, Tohyama M. Cytoplasmic p21(Cip1/WAF1) regulates neurite remodeling by inhibiting Rho-kinase activity. J Cell Biol. 2002;158:321–9. doi: 10.1083/jcb.200202071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yokoo T, Toyoshima H, Miura M, Wang Y, Iida KT, Suzuki H, et al. p57Kip2 regulates actin dynamics by binding and translocating LIM-kinase 1 to the nucleus. J Biol Chem. 2003;278:52919–23. doi: 10.1074/jbc.M309334200. [DOI] [PubMed] [Google Scholar]
- 14.Maddox AS, Burridge K. RhoA is required for cortical retraction and rigidity during mitotic cell rounding. J Cell Biol. 2003;160:255–65. doi: 10.1083/jcb.200207130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bement WM, Benink HA, von Dassow G. A microtubule-dependent zone of active RhoA during cleavage plane specification. J Cell Biol. 2005;170:91–101. doi: 10.1083/jcb.200501131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Beamish H, de Boer L, Giles N, Stevens F, Oakes V, Gabrielli B. Cyclin A/cdk2 regulates adenomatous polyposis coli-dependent mitotic spindle anchoring. J Biol Chem. 2009;284:29015–23. doi: 10.1074/jbc.M109.042820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sütterlin C, Colanzi A. The Golgi and the centrosome: building a functional partnership. J Cell Biol. 2010;188:621–8. doi: 10.1083/jcb.200910001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ferguson RL, Pascreau G, Maller JL. The cyclin A centrosomal localization sequence recruits MCM5 and Orc1 to regulate centrosome reduplication. J Cell Sci. 2010;123:2743–9. doi: 10.1242/jcs.073098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bray K, Brakebusch C, Vargo-Gogola T. The Rho GTPase Cdc42 is required for primary mammary epithelial cell morphogenesis in vitro. Small Gtpases. 2011;2:247–58. doi: 10.4161/sgtp.2.5.18163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Slattum G, McGee KM, Rosenblatt J. P115 RhoGEF and microtubules decide the direction apoptotic cells extrude from an epithelium. J Cell Biol. 2009;186:693–702. doi: 10.1083/jcb.200903079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Migita T, Oda Y, Naito S, Tsuneyoshi M. Low expression of p27(Kip1) is associated with tumor size and poor prognosis in patients with renal cell carcinoma. Cancer. 2002;94:973–9. doi: 10.1002/cncr.10338. [DOI] [PubMed] [Google Scholar]
- 22.Li JQ, Miki H, Wu F, Saoo K, Nishioka M, Ohmori M, et al. Cyclin A correlates with carcinogenesis and metastasis, and p27(kip1) correlates with lymphatic invasion, in colorectal neoplasms. Hum Pathol. 2002;33:1006–15. doi: 10.1053/hupa.2002.125774. [DOI] [PubMed] [Google Scholar]
- 23.Mashal RD, Lester S, Corless C, Richie JP, Chandra R, Propert KJ, et al. Expression of cell cycle-regulated proteins in prostate cancer. Cancer Res. 1996;56:4159–63. [PubMed] [Google Scholar]
- 24.Wang YF, Chen JY, Chang SY, Chiu JH, Li WY, Chu PY, et al. Nm23-H1 expression of metastatic tumors in the lymph nodes is a prognostic indicator of oral squamous cell carcinoma. Int J Cancer. 2008;122:377–86. doi: 10.1002/ijc.23096. [DOI] [PubMed] [Google Scholar]
- 25.Kriz W, Kaissling B, Le Hir M. Epithelial-mesenchymal transition (EMT) in kidney fibrosis: fact or fantasy? J Clin Invest. 2011;121:468–74. doi: 10.1172/JCI44595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chapman HA. Epithelial-mesenchymal interactions in pulmonary fibrosis. Annu Rev Physiol. 2011;73:413–35. doi: 10.1146/annurev-physiol-012110-142225. [DOI] [PubMed] [Google Scholar]
- 27.Cheung C, Luo H, Yanagawa B, Leong HS, Samarasekera D, Lai JC, et al. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in coxsackievirus-induced myocarditis. Cardiovasc Pathol. 2006;15:63–74. doi: 10.1016/j.carpath.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 28.Zhang S, Wang X, Osunkoya AO, Iqbal S, Wang Y, Chen Z, et al. EPLIN downregulation promotes epithelial-mesenchymal transition in prostate cancer cells and correlates with clinical lymph node metastasis. Oncogene. [DOI] [PMC free article] [PubMed]
- 29.Ru GQ, Wang HJ, Xu WJ, Zhao ZS. Upregulation of Twist in gastric carcinoma associated with tumor invasion and poor prognosis. Pathol Oncol Res. 2011;17:341–7. doi: 10.1007/s12253-010-9332-0. [DOI] [PubMed] [Google Scholar]
- 30.Yang MH, Hsu DS, Wang HW, Wang HJ, Lan HY, Yang WH, et al. Bmi1 is essential in Twist1-induced epithelial-mesenchymal transition. Nat Cell Biol. 2010;12:982–92. doi: 10.1038/ncb2099. [DOI] [PubMed] [Google Scholar]
- 31.Zavadil J, Böttinger EP. TGF-beta and epithelial-to-mesenchymal transitions. Oncogene. 2005;24:5764–74. doi: 10.1038/sj.onc.1208927. [DOI] [PubMed] [Google Scholar]
- 32.Lo HW, Hsu SC, Xia W, Cao X, Shih JY, Wei Y, et al. Epidermal growth factor receptor cooperates with signal transducer and activator of transcription 3 to induce epithelial-mesenchymal transition in cancer cells via up-regulation of TWIST gene expression. Cancer Res. 2007;67:9066–76. doi: 10.1158/0008-5472.CAN-07-0575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Acevedo VD, Gangula RD, Freeman KW, Li R, Zhang Y, Wang F, et al. Inducible FGFR-1 activation leads to irreversible prostate adenocarcinoma and an epithelial-to-mesenchymal transition. Cancer Cell. 2007;12:559–71. doi: 10.1016/j.ccr.2007.11.004. [DOI] [PubMed] [Google Scholar]
- 34.Savagner P, Yamada KM, Thiery JP. The zinc-finger protein slug causes desmosome dissociation, an initial and necessary step for growth factor-induced epithelial-mesenchymal transition. J Cell Biol. 1997;137:1403–19. doi: 10.1083/jcb.137.6.1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Larue L, Bellacosa A. Epithelial-mesenchymal transition in development and cancer: role of phosphatidylinositol 3′ kinase/AKT pathways. Oncogene. 2005;24:7443–54. doi: 10.1038/sj.onc.1209091. [DOI] [PubMed] [Google Scholar]
- 36.Thiery JP, Sleeman JP. Complex networks orchestrate epithelial-mesenchymal transitions. Nat Rev Mol Cell Biol. 2006;7:131–42. doi: 10.1038/nrm1835. [DOI] [PubMed] [Google Scholar]
- 37.Bosco EE, Mulloy JC, Zheng Y. Rac1 GTPase: a “Rac” of all trades. Cell Mol Life Sci. 2009;66:370–4. doi: 10.1007/s00018-008-8552-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ma L, Teruya-Feldstein J, Weinberg RA. Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature. 2007;449:682–8. doi: 10.1038/nature06174. [DOI] [PubMed] [Google Scholar]
- 39.Wilkinson S, Paterson HF, Marshall CJ. Cdc42-MRCK and Rho-ROCK signalling cooperate in myosin phosphorylation and cell invasion. Nat Cell Biol. 2005;7:255–61. doi: 10.1038/ncb1230. [DOI] [PubMed] [Google Scholar]
- 40.Hakem A, Sanchez-Sweatman O, You-Ten A, Duncan G, Wakeham A, Khokha R, et al. RhoC is dispensable for embryogenesis and tumor initiation but essential for metastasis. Genes Dev. 2005;19:1974–9. doi: 10.1101/gad.1310805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wheeler AP, Ridley AJ. Why three Rho proteins? RhoA, RhoB, RhoC, and cell motility. Exp Cell Res. 2004;301:43–9. doi: 10.1016/j.yexcr.2004.08.012. [DOI] [PubMed] [Google Scholar]
- 42.Suwa H, Ohshio G, Imamura T, Watanabe G, Arii S, Imamura M, et al. Overexpression of the rhoC gene correlates with progression of ductal adenocarcinoma of the pancreas. Br J Cancer. 1998;77:147–52. doi: 10.1038/bjc.1998.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang W, Yang LY, Huang GW, Lu WQ, Yang ZL, Yang JQ, et al. Genomic analysis reveals RhoC as a potential marker in hepatocellular carcinoma with poor prognosis. Br J Cancer. 2004;90:2349–55. doi: 10.1038/sj.bjc.6601749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fritz G, Brachetti C, Bahlmann F, Schmidt M, Kaina B. Rho GTPases in human breast tumours: expression and mutation analyses and correlation with clinical parameters. Br J Cancer. 2002;87:635–44. doi: 10.1038/sj.bjc.6600510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Horiuchi A, Imai T, Wang C, Ohira S, Feng Y, Nikaido T, et al. Up-regulation of small GTPases, RhoA and RhoC, is associated with tumor progression in ovarian carcinoma. Lab Invest. 2003;83:861–70. doi: 10.1097/01.lab.0000073128.16098.31. [DOI] [PubMed] [Google Scholar]
- 46.Faried A, Faried LS, Usman N, Kato H, Kuwano H. Clinical and prognostic significance of RhoA and RhoC gene expression in esophageal squamous cell carcinoma. Ann Surg Oncol. 2007;14:3593–601. doi: 10.1245/s10434-007-9562-x. [DOI] [PubMed] [Google Scholar]
- 47.Simpson KJ, Dugan AS, Mercurio AM. Functional analysis of the contribution of RhoA and RhoC GTPases to invasive breast carcinoma. Cancer Res. 2004;64:8694–701. doi: 10.1158/0008-5472.CAN-04-2247. [DOI] [PubMed] [Google Scholar]
- 48.Bellovin DI, Simpson KJ, Danilov T, Maynard E, Rimm DL, Oettgen P, et al. Reciprocal regulation of RhoA and RhoC characterizes the EMT and identifies RhoC as a prognostic marker of colon carcinoma. Oncogene. 2006;25:6959–67. doi: 10.1038/sj.onc.1209682. [DOI] [PubMed] [Google Scholar]
- 49.Iiizumi M, Bandyopadhyay S, Pai SK, Watabe M, Hirota S, Hosobe S, et al. RhoC promotes metastasis via activation of the Pyk2 pathway in prostate cancer. Cancer Res. 2008;68:7613–20. doi: 10.1158/0008-5472.CAN-07-6700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Philips A, Roux P, Coulon V, Bellanger JM, Vié A, Vignais ML, et al. Differential effect of Rac and Cdc42 on p38 kinase activity and cell cycle progression of nonadherent primary mouse fibroblasts. J Biol Chem. 2000;275:5911–7. doi: 10.1074/jbc.275.8.5911. [DOI] [PubMed] [Google Scholar]