Abstract
OBJECTIVE:
During the neonatal and infancy periods, some chronic liver diseases may lead to progressive hepatic fibrosis, which is a condition that can ultimately result in the loss of organ function and severe portal hypertension necessitating hepatic transplantation. In a previous report, pharmacological interventions were demonstrated to modulate hepatic fibrosis induced by bile duct ligation in young rats. The administration of pentoxifylline or prednisolone, or the combination of both, resulted in reduced fibrogenesis in portal spaces. The objectives of the present study were to evaluate the expression of transforming growth factor β and vascular endothelial growth factor after bile duct ligation in young rats and to assess the effect of those same drugs on cytokine expression.
METHODS:
In this experimental study, 80 young rats (21 or 22 days old) were submitted either to laparotomy and common bile duct ligation or to sham surgery. The animals were allocated into four groups according to surgical procedure, and the following treatments were administered: (1) common bile duct ligation + distilled water, (2) sham surgery + distilled water, (3) common bile duct ligation + pentoxifylline, or (4) common bile duct ligation + prednisolone. After 30 days, a hepatic fragment was collected from each animal for immunohistochemical analysis using monoclonal antibodies against transforming growth factor β and vascular endothelial growth factor. Digital morphometric and statistical analyses were performed.
RESULTS:
The administration of pentoxifylline reduced the transforming growth factor β-marked area and the amount of transforming growth factor β expressed in liver tissue. This effect was not observed after the administration of prednisolone. There was a significant reduction in vascular endothelial growth factor expression after the administration of either drug compared with the non-treatment group.
CONCLUSIONS:
The administration of pentoxifylline to cholestatic young rats resulted in the diminished expression of transforming growth factor β and vascular endothelial growth factor in liver tissue. The administration of steroids resulted in the diminished expression of vascular endothelial growth factor only. These pathways may be involved in hepatic fibrogenesis in young rats submitted to bile duct ligation and exposed to pentoxifylline or prednisolone.
Keywords: Liver Fibrogenesis, Experimental Cholestasis, Steroids, Pentoxifylline, TGFβ, VEGF
INTRODUCTION
Cholestatic disorders are responsible for chronic hepatic failure in a significant number of patients during infancy. In the neonatal period, the most frequent cholestatic disease requiring liver transplantation is biliary atresia, a condition for which the pathogenesis is not yet fully understood but that is absolutely fatal if left untreated (1).
Since the 1970s, some authors have suggested that the administration of corticosteroids may promote better late outcomes of biliary atresia in children who undergo Kasai's portoenterostomy (2-4). In recent years, some results of this new strategy have been published, but the available data still do not clearly demonstrate a difference in jaundice-free survival and a reduced need for early liver transplantation (5-7). Other potentially antifibrogenic drugs have also been experimentally tested in animals, including pentoxifylline (PTX), a phosphodiesterase inhibitor already used in clinical practice for the treatment of arterial insufficiency (8-10). Some studies have demonstrated the inhibition of inflammatory cytokines, such as tumor necrosis factor α, transforming growth factor β, and interleukins (ILs) 1, 6, and 8 (11,12). Other studies have failed to demonstrate this effect in animal models (9,13). Therefore, no consensus exists regarding the role of PTX in the inflammatory and fibrogenic cascades.
Our group has been investigating the effects of antifibrogenic drugs over the last few years, with a particular focus on their effects in growing animals. We reported our initial results in 2009 after developing a model to study portal fibrosis secondary to biliary obstruction in young rats (14-15). We concluded that hepatic fibrosis induced by bile duct ligation in young rats could be modulated by pharmacologic interventions. The administration of pentoxifylline or prednisolone (PRED), or the combination of both, resulted in diminished collagen-filled areas in the portal spaces. We have continued this work and now present an immunohistochemical analysis of the expression of transforming growth factor β (TGFβ) and vascular endothelial growth factor (VEGF) after bile duct ligation in young rats and the influence of these drugs on cytokine expression.
METHODS
This study was approved by the Ethical Committee for Research Project Analysis of our institution and was conducted according to international guidelines regarding the use of laboratory animals. All operative procedures were performed by the same surgeon.
Surgical procedures
Young (21- to 22-day-old) Wistar rats were submitted to common bile duct ligation (CBDL) as described in a previous report (15). Under ether anesthesia, a median laparotomy was performed, and the common bile duct was ligated and divided twice using 6.0 monofilament nylon ligation. The sham surgery (SHAM) consisted of the laparotomy and exposure of the hepatic hilum without duct ligation. The animals were randomly allocated into four groups (20 animals per group) according to the surgical procedure and were administered a solution as follows: 1) CBDL + distilled water; 2) SHAM + distilled water; 3) CBDL + PTX 10 mg/kg per day; or 4) CBDL + PRED 3 mg/kg per day.
The solutions were administered once a day via nasogastric tubing under sedation. The drug doses per kilogram were similar to those used in humans in the clinical setting.
After 30 days, cardiorespiratory arrest was induced by the inhalation of anesthesia (ether gas chamber at a high concentration). The animals were rapidly weighed and submitted to midline incision for the harvesting of a 1-cm3 hepatic fragment from the left lobe.
Histological analysis
The hepatic fragments were processed according to standard techniques (3-μm-thick sections of paraffin-embedded material) to obtain two slides for each animal. The slides were included in 3-aminopropyl-trietoxi-silano, and immunohistochemical reactions followed the biotin-streptavidin-peroxidase protocol. The primary antibodies were as follows: monoclonal anti-mouse TGFβ IgG (sc-52893, Santa Cruz Biotechnology, USA) and monoclonal anti-mouse VEGF IgG (sc-7269, Santa Cruz Biotechnology, Santa Cruz, CA, USA). The secondary antibody was the Vectastain anti-mouse ABCkit™ (Vector Laboratories, Burlingame, CA, USA) for both reactions. The protein block was accomplished with methanol/azide for the TGFβ slides and with the Novocastra™ system (Leica Microsystems, Milton Keynes, England, UK) for the VEGF reactions. The slides were analyzed under a Leica DMR microscope and digitized with Panoramic Scan Midi™ (3DHistech, Hungary Software, Budapest, Hungary).
Morphometric analysis
Aleatory areas were delineated on each digitized slide and processed with the aid of the software Image Pro-Plus 4.5.0.29 Windows XP version (Media Cybernetics Inc.). After image acquisition, the software was calibrated to automate the cytokine (TGFβ or VEGF) identification. Several measurements were performed: total area, cytokine-marked area (area +), integrated optical density (IOD; antibody color intensity measure, which reflects the amount of cytokine present in the tissue), antibody distribution or proportion (area +/total area), and mean density (IOD/area +).
Statistical analysis
Comparisons of the data between groups were performed using the software SPSS version 18.0 for Windows (SPSS Inc., Chicago, IL). As the data had a normal distribution, the analysis of variance and Tukey's post hoc test were applied to test for differences between groups. The hypothesis of sample equality was rejected for p<0.05.
RESULTS
Descriptive analysis
TGFβ expression
Sham: no hepatocyte presentation, expression in endothelial and hepatic stellate cells;
CBDL: mild expression in periportal hepatocytes, no expression in the biliary epithelium;
PRED: irregular hepatocyte expression in different areas of the hepatic lobule, clear presentation in endothelial and hepatic stellate cells;
PTX: less intense expression in hepatocytes and endothelial cells than in the PRED group.
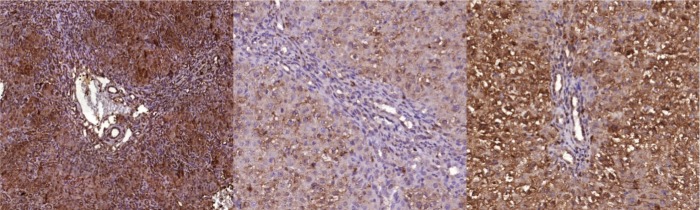
VEGF expression: All groups exhibited VEGF expression in hepatocytes, endothelial and hepatic stellate cells and the biliary epithelium. Only fibrous areas were not stained (Figure 1).
Figure 1.
Digitized slide microphotography showing Anti-VEGF immunohistochemical staining. From left to right: CBDL (100x magnification), PTX (200x magnification), and PRED (200x magnification).
Morphometric analysis
TGFβ expression
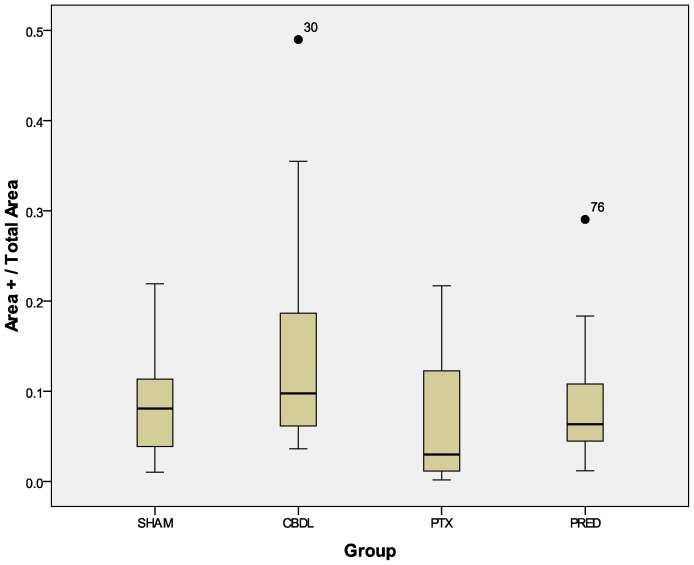
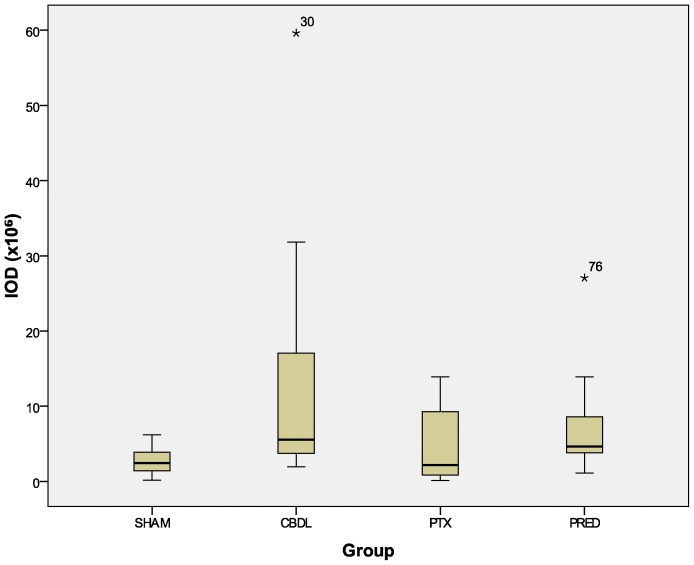
Validating the findings of the descriptive analysis, the administration of pentoxifylline (PTX) to growing rats reduced the size of the cytokine-marked area (Figure 2) and the amount of TGFβ expressed in liver tissue (Figure 3), as reflected by the integrated optical density (IOD). These effects were not observed after the administration of prednisolone (Figures 2 and 3).
Figure 2.
Ratio between the anti-TGFβ-stained area (µm2) and total area (µm2): PTX x CBDL, p = 0.032; PRED x CBDL, p = 0.089. The numbers in the graphic are outliers.
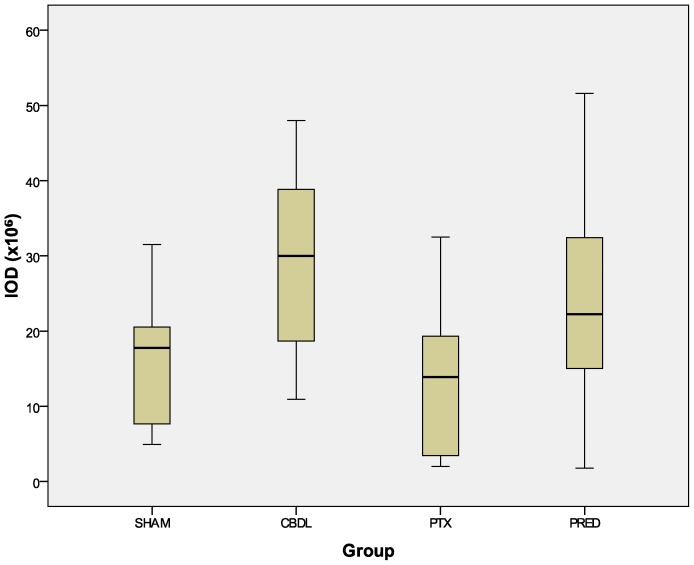
Figure 3.
Integrated optical density (IOD). Anti-TGFβ stained slides: PTX x CBDL, p = 0.032; PRED x CBDL, p = 0.231. The numbers in the graphic are outliers.
VEGF expression
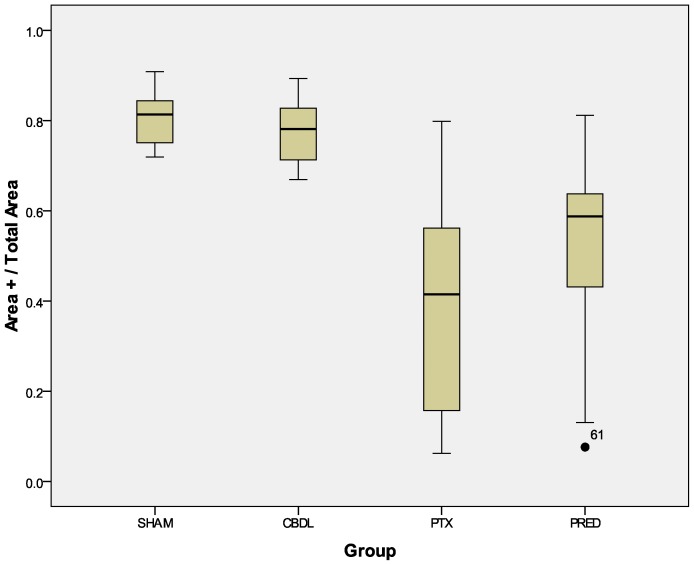
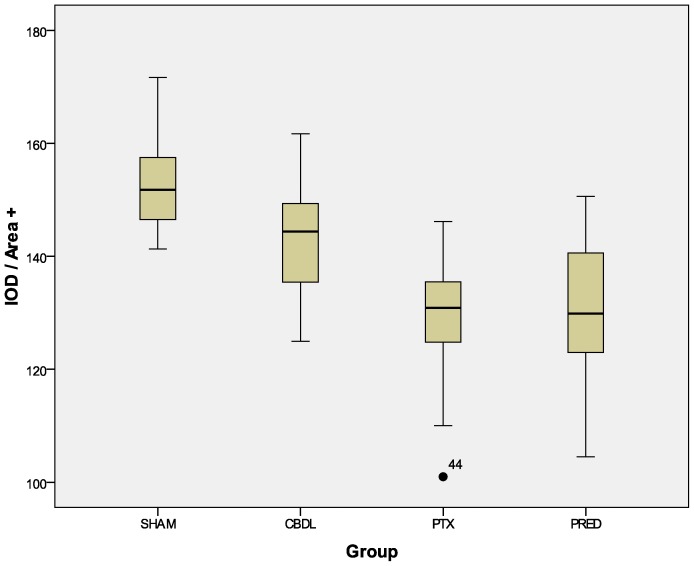
The reduction in the expression of VEGF after the administration of both drugs was quite remarkable. The distribution of this cytokine (Figure 4) was reduced in the PTX and PRED groups relative to the CBDL group (p<0.001). Although the decrease in the absolute amount of cytokine in the PRED group, as revealed by the IOD index, did not reach statistical significance (p = 0.195, Figure 5 compared with the CBDL group, when transformed into the mean density (ratio between the IOD and positive area), a significant decrease was revealed (p = 0.001, Figure 6.
Figure 4.
Ratio between the anti-VEGF-stained area (µm2) and total area (µm2): PTX x CBDL, p<0.001; PRED x CBDL, p<0.001. The number in the graphic is an outlier.
Figure 5.
Integrated optical density (IOD). Anti-VEGF-stained slides: PTX x CBDL, p<0.001; PRED x CBDL, p = 0.195.
Figure 6.
Mean density (ratio between the IOD and cytokine-marked area). Anti-VEGF-stained slides: PTX x CBDL, p<0.001; PRED x CBDL, p = 0.001. The number in the graphic is an outlier.
DISCUSSION
Regarding the treatment of biliary atresia in children, clinicians have been empirically prescribing steroids immediately post-Kasai surgery since the 1980s (16-18). In the wake of progressive liver disease following bile duct obstruction, steroids are hypothesized to suppress inflammation and promote bile flow (19), but recent clinical reports have been controversial (20,21) and have failed to clearly demonstrate an actual long-term benefit (i.e., transplant-free survival) of the adjuvant use of steroids after portoenterostomy.
Another medication included in this study was pentoxifylline (PTX), a potentially antifibrogenic drug (9-13,12. Classified as a nonspecific phosphodiesterase inhibitor, this drug has already been incorporated into the clinical setting for the treatment of chronic occlusive arterial disease because of its oxygen transport-enhancing effect. As phosphodiesterases regulate the intracellular levels of cyclic nucleotides (cyclic adenosine monophosphate and cyclic guanosine monophosphate), their inhibition can affect several processes, such as apoptosis, muscle contraction, cellular differentiation, migration, and proliferation (23-25). Specifically regarding hepatic fibrogenesis, since 1993, there have been some publications demonstrating that PTX can decrease the activation and proliferation of stellate cells and downregulate the production of IL-1, IL-6, and tumor necrosis factor α (all proinflammatory cytokines) (26).
In a previous report (15), we revealed that the administration of a steroid and pentoxifylline to recently weaned rats submitted to biliary obstruction could promote a reduction in the collagen-filled areas in liver tissue, indicating a potential mechanism for the pharmacological modulation of cholestasis-induced portal fibrosis. We believe that the use of growing animals, rather than adults, as experimental models may more accurately represent the histological abnormalities found in newborns or children affected by cholestasis (14).
The exact mechanisms involved in portal biliary fibrosis in young animals are not completely understood. The present investigation was conducted to contribute to the elucidation of the following specific question: how is hepatic collagen deposition secondary to biliary obstruction affected by the administration of steroids and PTX?
Among other cytokines, TGFβ and VEGF appear to actively participate in the processes of inflammatory cell recruitment and hepatic fibrogenesis and the mediation of the adaptive proliferative response of cholangiocytes to cholestasis (27-29). Are these cytokines affected by the administration of steroids and PTX?
In this model, we were able to demonstrate, through immunohistochemical analyses, that VEGF expression was substantially reduced following exposure to steroids and PTX. However, only PTX appeared to affect the liver tissue expression of TGFβ. We failed to demonstrate a significant difference in this expression after the administration of prednisolone. The impairment in the pathways of these two cytokines after the administration of PTX to these growing animals may explain the slightly more accentuated reduction in collagen deposition observed in our previous report compared with the PRED group.
Obviously, other cells, cytokines, signaling pathways, and mechanisms are involved in a process as complex as hepatic fibrogenesis. Therefore, subsequent experimental and clinical studies should be conducted to confirm the benefits of exposing jaundiced children to these medications, with the aim of reducing hepatic fibrosis and the need for liver transplantation.
ACKNOWLEDGMENTS
We thank FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) for providing financial support (grant #2009/07225-2).
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Sokol RJ, Mack C. Etiopathogenesis of biliary atresia. Semin Liver Dis. 2001;21(4):517–24. doi: 10.1055/s-2001-19032. [DOI] [PubMed] [Google Scholar]
- 2.Carceller A, Blanchard H, Alvarez F, St-Vil D, Bensoussan AL, Di Lorenzo M. Past and future of biliary atresia. J Pediatr Surg. 2000;35(5):717–20. doi: 10.1053/jpsu.2000.6034. [DOI] [PubMed] [Google Scholar]
- 3.Karrer FM, Lilly JR. Corticosteroid therapy in biliary atresia. J Pediatr Surg. 1985;20:693–5. doi: 10.1016/s0022-3468(85)80026-9. [DOI] [PubMed] [Google Scholar]
- 4.Muraji T, Higashimoto Y. The improved outlook for biliary atresia with corticosteroid therapy. J Pediatr Surg. 1997;32(7):1103–7. doi: 10.1016/s0022-3468(97)90408-5. [DOI] [PubMed] [Google Scholar]
- 5.Kobayashi H, Yamataka A, Koga H, Okazaki T, Tamura T, Urao M, et al. Optimum prednisolone usage in patients with biliary atresia postportoenterostomy. J Pediatr Surg. 2005;40(2):327–30. doi: 10.1016/j.jpedsurg.2004.10.017. [DOI] [PubMed] [Google Scholar]
- 6.Escobar MA, Jay CL, Brooks RM, West KW, Rescorla FJ, Molleston JP, et al. Effect of corticosteroid therapy on outcomes in biliary atresia after Kasai portoenterostomy. Journal of pediatric surgery. 2006;41(1):99-103; discussion 99- doi: 10.1016/j.jpedsurg.2005.10.072. [DOI] [PubMed] [Google Scholar]
- 7.Stringer MD, Davison SM, Rajwal SR, McClean P. Kasai portoenterostomy: 12-year experience with a novel adjuvant therapy regimen. Journal of pediatric surgery. 2007;42(8):1324–8. doi: 10.1016/j.jpedsurg.2007.03.026. [DOI] [PubMed] [Google Scholar]
- 8.Oberti F, Pilette C, Rifflet H, Maiga MY, Moreau A, Gallois Y, et al. Effects of simvastatin, pentoxifylline and spironolactone on hepatic fibrosis and portal hypertension in rats with bile duct ligation. Journal of hepatology. 1997;26(6):1363–71. doi: 10.1016/s0168-8278(97)80473-4. [DOI] [PubMed] [Google Scholar]
- 9.Peterson TC, Neumeister M. Effect of pentoxifylline in rat and swine models of hepatic fibrosis: role of fibroproliferation in its mechanism. Immunopharmacology. 1996;31(2-3):183–93. doi: 10.1016/0162-3109(95)00048-8. [DOI] [PubMed] [Google Scholar]
- 10.Ward A, Clissold SP. Pentoxifylline. A review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic efficacy. Drugs. 1987;34(1):50–97. doi: 10.2165/00003495-198734010-00003. [DOI] [PubMed] [Google Scholar]
- 11.Raetsch C, Jia JD, Boigk G, Bauer M, Hahn EG, Riecken EO, et al. Pentoxifylline downregulates profibrogenic cytokines and procollagen I expression in rat secondary biliary fibrosis. Gut. 2002;50(2):241–7. doi: 10.1136/gut.50.2.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schandené L, Vandenbussche P, Crusiaux A, Alègre ML, Abramowicz D, Dupont E, et al. Differential effects of pentoxifylline on the production of tumor necrosis factor-alpha (TNF&agr;) and interleukin-6 (IL-6) by monocytes and T cells. Immunology. 1992;76(1):30–4. [PMC free article] [PubMed] [Google Scholar]
- 13.Tarçin O, Avsar K, Demirtürk L, Gültepe M, Oktar BK, Ozdoğan OC, et al. In vivo inefficiency of pentoxifylline and interferon-alpha on hepatic fibrosis in biliary obstructed rats: assessment by tissue collagen content and prolidase activity. J Gastroenterol Hepatol. 2003;18(4):437–44. doi: 10.1046/j.1440-1746.2003.03004.x. [DOI] [PubMed] [Google Scholar]
- 14.Gibelli NE, Tannuri U, de Mello ES, Rodrigues CJ. Bile duct ligation in neonatal rats: Is it a valid experimental model for biliary atresia studies. Pediatr Transplant. 2009;13(1):81–7. doi: 10.1111/j.1399-3046.2008.00947.x. [DOI] [PubMed] [Google Scholar]
- 15.Andrade WdeC, Tannuri U, da Silva LF, Alves VA. Effects of the administration of pentoxifylline and prednisolone on the evolution of portal fibrogenesis secondary to biliary obstruction—an experimental study in growing animals. J Pediatr Surg. 2009;44(11):2071–7. doi: 10.1016/j.jpedsurg.2009.05.020. [DOI] [PubMed] [Google Scholar]
- 16.Meyers RL, Book LS, O'Gorman MA, Jackson WD, Black RE, Johnson DG, et al. High dose steroids, ursodeoxycholic acid, and chronic intravenous antibiotics improve bile flow after Kasai procedure in infants with biliary atresia. J Pediatr Surg. 2003;38(3):406–11. doi: 10.1053/jpsu.2003.50069. [DOI] [PubMed] [Google Scholar]
- 17.Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med. 2005;353(16):1711–23. doi: 10.1056/NEJMra050541. [DOI] [PubMed] [Google Scholar]
- 18.Muraji T, Nio M, Ohhama Y, Hashimoto T, Iwanaka T, Takamatsu H, et al. Postoperative corticosteroid therapy for bile drainage in biliary atresia: a nationwide survey. J Pediatr Surg. 2004;39(12):1803–5. doi: 10.1016/j.jpedsurg.2004.08.019. [DOI] [PubMed] [Google Scholar]
- 19.Miner PB, Gaito JM. Bile flow in response to pharmacologic agents. Biochem Pharmacol. 1979;28(7):1063–6. doi: 10.1016/0006-2952(79)90304-6. [DOI] [PubMed] [Google Scholar]
- 20.Suzuki T, Hashimoto T, Kondo S, Sato Y, Hussein MH. Evaluating patients' outcome post-Kasai operation: a 19-year experience with modification of the hepatic portoenterostomy and applying a novel steroid therapy regimen. Pediatr Surg Int. 2010;26(8):825–30. doi: 10.1007/s00383-010-2637-y. [DOI] [PubMed] [Google Scholar]
- 21.Sarkhy A, Schreiber RA, Milner RA, Barker CC. Does adjuvant steroid therapy post-Kasai portoenterostomy improve outcome of biliary atresia. Systematic review and meta-analysis. Can J Gastroenterol. 2011;25(8):440–4. doi: 10.1155/2011/125610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Windmeier C, Gressner AM. Pharmacological aspects of pentoxifylline with emphasis on its inhibitory actions on hepatic fibrosis. Gen Pharmacol. 1997;29:181–96. doi: 10.1016/s0306-3623(96)00314-x. [DOI] [PubMed] [Google Scholar]
- 23.Bender AT, Beavo JA. Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol Rev. 2006;58:488–520. doi: 10.1124/pr.58.3.5. [DOI] [PubMed] [Google Scholar]
- 24.Conti M, Beavo JA. Biochemistry and physiology of cyclic nucleotide phosphodiesterases: essential components in cyclic nucleotide signaling. Ann Rev Biochem. 2007;76:481–511. doi: 10.1146/annurev.biochem.76.060305.150444. [DOI] [PubMed] [Google Scholar]
- 25.Omori K, Kotera J. Overview of PDEs and their regulation. 2007;100(3):306–27. doi: 10.1161/01.RES.0000256354.95791.f1. Circ Res. [DOI] [PubMed] [Google Scholar]
- 26.Peterson T. Pentoxifylline prevents fibrosis in an animal model and inhibits platelet-derived growth factor driven proliferation of fibroblast. 1993;17(3):486–93. Hepatology. [PubMed] [Google Scholar]
- 27.Housset C. Biliary epithelial cell response to cholestasis. J Hepatol. 2000;32:14–15. [Google Scholar]
- 28.Kinnman N, Housset C. Peribiliary myofibroblasts in biliary type fibrosis. Front Biosci. 2002;7:d496–503. doi: 10.2741/A790. [DOI] [PubMed] [Google Scholar]
- 29.Gaudio E, Barbaro B, Alvaro D, Glaser S, Glaser S, Francis H, Ueno Y, et al. Vascular endothelial growth factor stimulates rat cholangiocyte proliferation via an autocrine mechanism. Gastroenterology. 2006;130(4):1270–82. doi: 10.1053/j.gastro.2005.12.034. [DOI] [PubMed] [Google Scholar]