Abstract
Objectives:
The anticancer activity of shatavarins (containing shatavarin IV) isolated from the roots of Asparagus racemosus (Wild) was evaluated using in vitro and in vivo experimental models.
Material and Methods:
The shatavarin IV was isolated from ethyl acetate insoluble fraction (AR-2B) of chloroform:methanol (2:1) (AR-2) extract of A. racemosus roots. The cytotoxicity (in vitro) of shatavarin IV and other shatavarins rich fraction was carried out using of MTT assay using MCF-7 (human breast cancer), HT-29 (human colon adenocarcinoma), and A-498 (human kidney carcinoma) cell lines. The in vivo anticancer activity of shatavarins (containing shatavarin IV) was evaluated against Ehrlich ascites carcinoma (EAC) tumor bearing mice.
Results:
The isolated shatavarin IV (84.69 %) along with shatavarins rich fraction, coded AR-2B containing 5.05% shatavarin IV showed potent cytotoxicity. Oral administration of AR-2B to tumor bearing mice at doses of 250 and 500 mg/kg body weight for 10 days, showed significant reduction in percent increase in body weight, tumor volume, packed cell volume, viable tumor cell count, and increased non-viable cell count when compared to the untreated mice of the EAC control group. The restoration of hematological parameters towards normalcy was also observed.
Conclusion:
The result suggests that the shatavarins (containing shatavarin IV) rich fraction (AR-2B) exhibits significant anticancer activity in both in vitro and in vivo experimental models.
KEY WORDS: Anticancer, Asparagus racemosus, cytotoxicity, ehrlich ascites carcinoma, shatavarin IV
Introduction
Natural products have long been a fertile source of drugs against cancer, which is projected to become the major cause of death in this century. There are at least 250000 species of plants of which, more than 1000 plants have documented anticancer properties.[1] Out of 121 prescription anticancer drugs in use today, 90 are derived from plant species.[2] Many herbs have been evaluated in clinical studies and are currently being investigated phytochemically to understand their tumoricidal actions against various cancers.[3] Major classes of anticancer compounds include alkaloids, terpenoids, flavonoids, and lignans.[4] We have chosen steroids (terpenoids) from Asparagus racemosus roots, because terpenoids, like taxol are currently being widely used in cancer chemotherapy.[4]
The plant Asaparagus racemosus is reported to contain steroidal saponins as a major class along with minor steroids and their glucosides, alkaloids, flavonoids, and triterpenoids.[5] The major steroidal glucosides (saponins) such as shatavarin I to X (shatavarins) were reported from the roots.[6,7] The total extract, polar and non-polar extracts, and their formulations exhibit immuno-pharmacological activity in cancer chemotherapy.[8] The extracts of this plant also reported to produce inhibitory action on 7, 12-dimethylbenzanthracene (DMBA)-induced mammary carcinogenesis in rats.[9] Hence, in the present study, we investigated the anticancer properties of shatavarins (containing shatavain IV) from the roots of Asparagus racemosus (Wild).
Materials and Methods
Asparagus racemosus roots (AR) were collected from Tumkur district, Karnataka, India and was authenticated by Dr. Kannan, R&D Center, The Himalaya Drug Company, Bangalore, India.
Extraction and Fractionation
The air-dried powdered material (7.5 kg) was subjected to repeated extraction by maceration at room temperature with various solvents of different polarity such as chloroform (20 L), chloroform:methanol (2:1) (20 L) and methanol (20 L) successively for 7 days with each solvent. Each extract was filtered, concentrated and dried at reduced pressure, and controlled temperature (40-60°C), which yielded 23 g of chloroform extract (AR-1), 437 g chloroform:methanol (2:1) extract (AR-2) and 845 g of methanol extract (AR-3). The dried extracts of AR-1 (20 g), AR-2 (400 g), and AR-3 (800 g) were again subjected to fractionation by maceration at room temperature. AR-1 was fractionated with hexane (1 L) to separate hexane soluble (AR-1A) and insoluble (AR-1B) fractions, same way AR-2 with ethyl acetate (8 L) to separate ethyl acetate soluble (AR-2A) and insoluble (AR-2B) and AR-3 with n-butanol (15 L) to separate n-butanol soluble (AR-3A) and insoluble (AR-3B) fractions. Each fraction was filtered, concentrated and dried, which yielded 19.6 g of AR-1A, negligible residue of AR-1B, 10 g of AR-2A, 375 g of AR-2B, 24 g of AR-3A, and 700 g of AR-3B.
Identification and Estimation of Shatavarin IV/shatavarins
High performance thin layer chromatography (HPTLC)
All the AR extracts and its fractions were subjected to identification and estimation of shatavarin IV by HPTLC [Figure 1].[10] One gram sample was dissolved with methanol made up to 100 ml with same solvent. Shatavarin IV (B.No T9G034 (99%) from Natural Remedies) at 1 mg/ml concentration in methanol was used as standard. Ten microliter of sample and standard were spotted on CAMAG Linomat V as 12 mm band width on a precoated Silica gel 60 F254 plate of thickness (0.2 mm). The plate was developed in the solvent system chloroform:methanol:water 65:35:10 (lower layer). Identification of the bands of shatavarins (containing shatavarin IV) was done by dipping the plate with vanillin sulfuric acid reagent and heating at 110°C for 5 to 10 min and the concentration of shatavarin IV was estimated by scanning the plate at 640 nm.
Figure 1.

Identification of shatavarins (Steroidal saponins) in different extracts (AR-1, AR-2, AR-3 and fractions (AR-2B, AR2B-F7B, AR-3 AR-3A AR-3B, AR2B-Shat IV, Shatavarin IV)
Isolation and characterization of shatavarin IV
As per HPTLC analysis, shatavarins rich fraction AR-2B (130 g; containing shatavrain IV-5.05 %) was further subjected to column chromatography to separate shatavarin IV on Silica gel (60-120) by elution with chloroform (0.5 L), chloroform:ethyl acetate mixture (2 L), ethyl acetate (1 L), ethyl acetate:methanol mixture (5.5 L) with increasing polarity up to methanol (1.5 L) led to separation of nine fractions designated as AR2B-F1 to F9. Major fraction (AR-2BF7B) was further subjected for the identification and estimation of shatavarin IV by HPTLC [Figures 1 and 2].[10]
Figure 2.
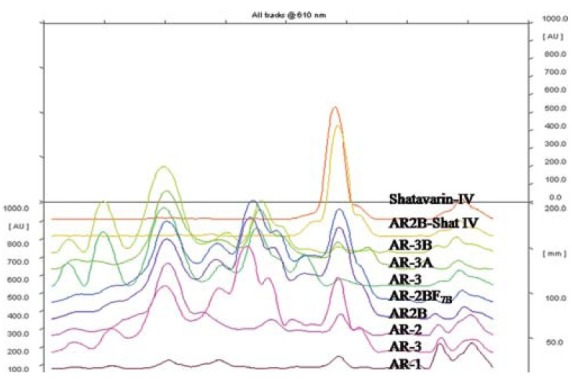
Estimation of shatavarin IV (shatavarins) in different extracts (AR-1, AR-2, AR-3) and fractions (AR-2B, AR2B-F7B, AR-3 AR-3A AR-3B, AR2B-Shat IV)
The crystalline powder AR2B-F7B(66 g) having shatavarin IV (5.17%) was subjected to repeated fractionation with ethyl acetate. The ethyl acetate fraction was evaporated and the residue was recrystallized with hot methanol to get white crystalline powder (3 g) which was coded as AR2B-Shat IV (shatavarin IV). The HPTLC [Figure 1] and mass spectra of isolated compound were matched with standard.
Cell Culture
Human breast cancer (MCF-7), human colon adeno carcinoma (HT-29), human kidney carcinoma (A-498), and Ehrlich ascites carcinoma (EAC) tumor cells were obtained from National Centre for Cell Sciences (Pune, India).
In Vitro Cytotoxic Activity
Preparation of sample
Stock solution: Since the extract AR-2, AR-2B, and fraction AR2B-F7B were rich in shatavarin IV, they were also analyzed for cytotoxicity along with isolated compound AR2B-Shat IV (shatavarin IV). Samples were separately dissolved in DMSO and volume was made up with DMEM supplemented with 2% inactivated NBCS to obtain a stock solution of 5 mg/ml, sterilized by filtration and stored at -20°C. Serial working solutions were prepared from stock at concentrations of 200, 100, 50, 25, and 12.5 mg/ml with DMEM.
Stock cells of MCF-7, HT-29, and A-498 cell lines were cultured in RPMI-1640 and DMEM supplemented with 10% sheep serum, penicillin (100 IU/ml), and streptomycin (100 μg/ml) in a humidified atmosphere of 5% CO2 at 37°C until confluent. The cells were dissociated with 0.2 % trypsin, 0.02 % EDTA in PBS. The cytotoxicity assay was carried out by adding 0.1 ml of cell suspension containing 10 000 cells to each well of a 96 well microtitre plate and fresh medium containing different concentrations of the samples were added at 24 h after seeding. Control cells were incubated without the test item and with DMSO (maximum 0.2 %). The microtitre plates were incubated at 37°C in a humidified incubator with 5% CO2 for a period of 3 days. The assay was done in triplicates. Cells were observed at different time intervals during incubation in the presence of the test item. Cellular viability was determined by the standard MTT assay method.[11]
In Vivo Anticancer Activity
The ascitic antitumor studies induced by EAC tumor cells were evaluated using healthy adult Swiss albino mice weighing 20-25 g. They were obtained from the animal house facility of The Himalaya Drug Company, Bangalore. All the animals were kept under standard laboratory conditions. Animals were provided with commercial pellet diet and purified water ad libitum. The study protocol was approved by the Institutional Animal Ethics Committee.
Fifty-four mice were divided in to five groups. Group I containing six animals, served as normal control, for which tumor cell inoculation was not done. The remaining animals were inoculated with EAC tumor cells (1 × 106 cells/mouse) intraperitoneally and divided into four groups containing 12 mice in each group. Group II, served as the tumor control. Groups I and II received an oral dose of 10 ml/kg b.wt. sodium CMC suspension (0.3%). Group III, served as a reference drug, which was treated orally with 20 mg/kg b.wt of 5-flurouracil (5-FU). Groups IV and V were treated with AR-2B fraction at 250 and 500 mg/kg b.wt., respectively. All the treatments were given orally at 24 h after tumor inoculation and continued once daily for 10 days. On the 11th day, six animals from each group were anesthetized and blood was collected by retro-orbital puncture for evaluation of hematological parameters, which included hemoglobin (Hb), red blood cell count (RBC), white blood cell count (WBC), and absolute count. The remaining animals in each of the groups were kept to check the mean survival time (MST) and percent increase in life span (% ILS) of the tumor bearing hosts.[12]
Tumor growth response: Antitumor effect of AR-2B was assessed by observation of change in body weight, ascitic tumor volume, packed cell volume, and viable tumor cell count, MST and % ILS.
Tumor Cell Count and Packed Cell Volume
The mice were dissected for collecting ascitic fluid from peritoneal cavity. The transplantable murine tumor was carefully collected with the help of sterile syringe to measure the tumor volume and the ascitic fluid was transferred to a graduated glass centrifuge tube and packed cell volume was determined by centrifuging at 100 g for 5 min. Viable and non-viable cell counting of ascitic cells were performed by trypan blue stain dye exclusion test (0.4% in normal saline). The cell counts were determined in a Neubauer counting chamber. The experiment was done in triplicates and mean count was taken.
MST and % ILS: The effect of AR-2B on tumor growth was observed by MST and %ILS. MST of each group containing six mice were monitored by recording the mortality daily for 6 weeks and % ILS was calculated by using the following equation.[13–16]
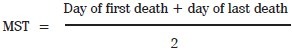
Statistical Analysis
The results are expressed as mean ± S.E.M. Data were analyzed using ANOVA followed by Dunnett's multiple comparison post hoc test. The CTC50 concentrations were calculated using linear regression analysis. The statistical analysis was performed using GraphPad Prism software (version 4.03).
Results
Out of all extracts and fractions, only AR-2 (4.18%) and AR-2B (5.05 %) contained high concentrations of shatavarin IV [Figure 1 and Table 1] and remaining samples had negligible content of shatavarin IV. According to the HPTLC [Figure 1 and Table 1], the AR2B-F7B fraction was rich in shatavarin IV. It was subjected to fractionation and crystallization until a single spot was obtained on HPTLC. The purified compound yielded 3 g of AR2B-Shat IV (shatavarin IV (84.89%)). The HPTLC [Figures 1 and 2] and mass spectra of isolated AR2B-Shat IV was an exact match to the standard shatavarin IV [Figure 3].
Table 1.
Comparative analysis of Shatavarin IV in different extract and fractions
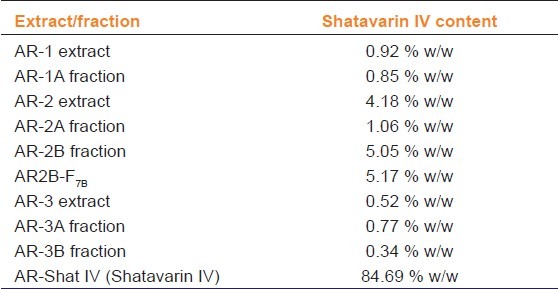
Figure 3.
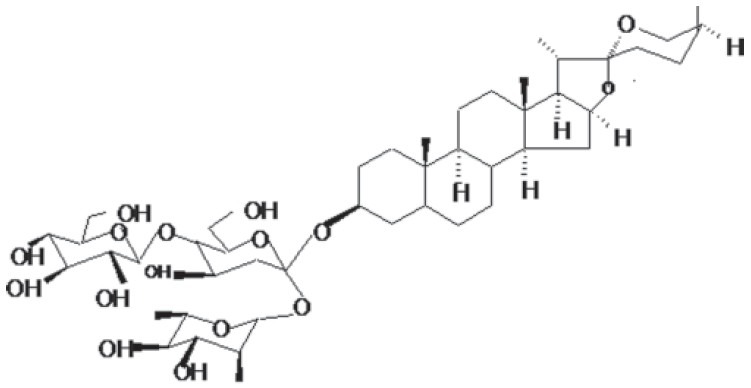
Structure of Shatavarin IV
In Vitro Cytotoxic Activity
AR-2 and AR-2B fractions were subjected to cytotoxicity along with separated fraction AR2B-F7B and isolated compound AR2B-Shat-IV (shatavarin IV). The results of CTC50 and % cytotoxicity of AR-2, AR-2B, AR2B-F7B, and AR2B-Shat-IV are shown in Figures 4a, b. The AR-2B, AR2B-F7B, and AR2B-Shat IV exhibited potent cytotoxicity on the MCF-7, HT-29, and A-498 cell lines.
Figure 4.
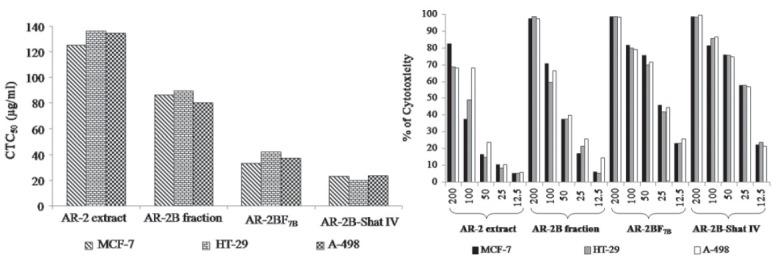
(a and b) In vitro cytotoxicity of AR-2, AR-2B, AR2B-F7B and AR2B-Shat IV
In Vivo Anticancer Activity
Effect on tumor growth
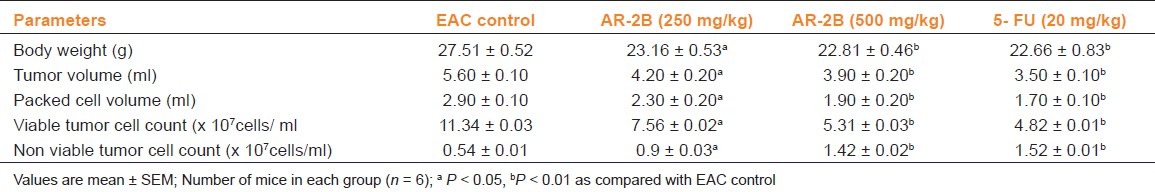
In the EAC tumor control group, the average life span of animals was found to be 21.45±1.58 days. AR-2B at doses of 250 and 500 mg/kg b.wt. showed a significant change in the average life span as compared to EAC control. The average life span of 5-FU treatment was found to be 27.48±1.40 days [Figure 5]. A dose dependent antitumor action of AR-2B was observed as a significant reduction in body weight, reduction in tumor volume, packed cell volume and viable tumor cell count, and increase in non-viable tumor cell count in both doses as compared to the EAC tumor control [Table 2].
Figure 5.
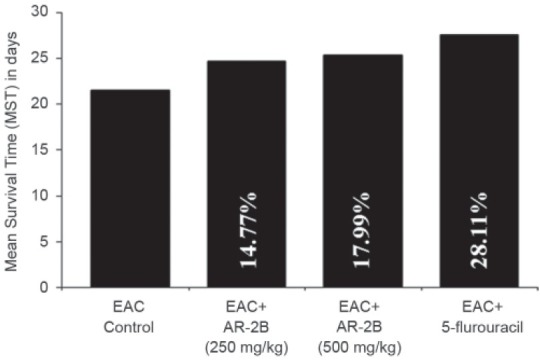
Effect of AR-2B extracts of Asparagus racemosus on survival time of EAC baring mice. Values are Mean ± SEM, No. of mice in each group: 6; aP < 0.05, bP < 0.01 as compared with EAC control
Table 2.
Effect of AR-2B fraction of Asparagus racemosus on tumor volume, packed cell volume, viable, and non-viable tumor cell count on EAC bearing mice
Effect of hematological parameters
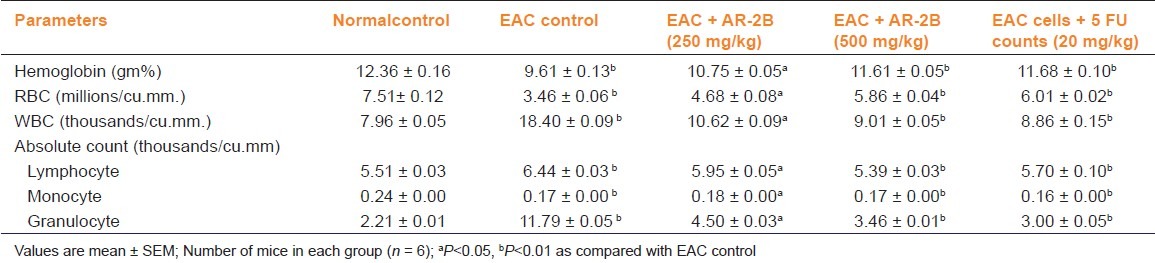
EAC tumor bearing mice showed a significant decrease in RBC count, hemoglobin, and a significant increase in WBC count as compared to normal control. There was also a significant increase in the lymphocyte count with an increase in neutrophil (granulocyte) counts in EAC tumor bearing mice as compared to normal control. Treatment with AR-2B significantly reversed EAC tumor-induced changes in hematological profiles [Table 3]. Similar observations were observed with 5-FU (Reference Drug).
Table 3.
Effect of AR-2B fraction Asparagus racemosus on hematological parameters of EAC bearing mice
Discussion
Natural products have been regarded as an important source that could contribute to potential chemotherapeutic agents. In particular, plant derived compounds have gained importance in anticancer therapy and some of the new chemotherapeutic agents currently available for use includes paclitaxel, vincristine, podophyllotoxin, and camptothecin.[12] The present investigation was carried out to evaluate the anticancer activity of major shatavarins (shatavarin IV) from AR-2B fraction of Asparagus racemosus roots. In vitro cytotoxicity study using MCF-7, HT-29, and A-498 cell lines showed potent activity with AR-2B (5.05% shatavarin IV) as well as its isolated compound AR2B-shat IV (shatavarin IV). These observations provided a basis for conducting the in vivo study of AR-2B using EAC tumor in mice.
The reliable criteria for judging the potency of an anticancer drug is prolongation of the life span of tumor bearing animals. There is also a significant increase in body weight in EAC tumor bearing mice due to the rapid increase in the ascitic tumor volume.[17] The mice bearing EAC tumor when orally administered with AR-2B showed a significant increase in the life span and a significant decrease in body weight. Treatment with AR-2B also showed a significant decrease in the tumor volume, packed cell volume, and viable tumor cell count, thereby indicating the anticancer nature of AR-2B. These results indicate either a direct cytotoxic effect on the tumor cells or local cytotoxic effect of AR-2B.
The major drawback of cancer chemotherapy encountered are myelosuppression and anemia.[18,19] The anemia encountered in the tumor bearing mice is due to the reduction in the RBC count or hemoglobin percentage and this may occur due to iron deficiency or due to hemolytic or myelopathic conditions. Treatment with AR-2B restored the hematological profiles in our study.
Excessive production of free radical causes oxidative stress, which leads to damage of macromolecules such as lipids and can induce lipid peroxidation in vivo. Increased peroxidation would cause degeneration of tissues. Lipid peroxide formed in the primary site would be transferred through the circulation and cause damage by propagating the process of lipid peroxidation. Malondialdehyde (MDA), the end product of lipid peroxidation was reported to be higher in cancer tissues than in non-diseased organs. Since Asparagus racemosus root induced elevation of antioxidant activity could be responsible for anticancer activity.[20–22] From the above findings it could be concluded that shatavarins (containing shatavarin IV) rich fraction AR-2B (containing 5.05% shatavarin IV) from Asparagus racemosus roots exhibited potent anticancer activity as demonstrated by in vitro cytotoxicity using MCF-7, HT-29, and A-498 cell lines as well as EAC induced tumor in mice.
Footnotes
Source(s) of Support: Nil.
Conflicting Interest: No.
References
- 1.Mukherjee AK, Basu S, Sarkar N, Ghosh AC. Advances in cancer therapy with plant based natural products. Curr Med Chem. 2001;8:1467–86. doi: 10.2174/0929867013372094. [DOI] [PubMed] [Google Scholar]
- 2.Shishodia S, Aggarwal BB. Guggulsterone inhibits NF-kB and IkBa kinase activation, suppresses expression of anti-apoptotic gene products, and enhances apoptosis. J Biol Chem. 2004;279:47148–58. doi: 10.1074/jbc.M408093200. [DOI] [PubMed] [Google Scholar]
- 3.Spiridon KE. Terrestrial plant-derived anticancer agents and plants used in anticancer research. Crit Rev Plant Sci. 2006;25:79–113. [Google Scholar]
- 4.Kim J, Park EJ. Cytotoxic anticancer candidates from natural resources. Curr Med Chem Anticancer Agents. 2002;2:485–537. doi: 10.2174/1568011023353949. [DOI] [PubMed] [Google Scholar]
- 5.Quality standards of Indian medicinal plants. Vol. 1. New Delhi: 2003. Indian Council of Medical Research; pp. 27–33. [Google Scholar]
- 6.Rastogi RM, Mehrotra BN. Compendium of Indian Medicinal Plants, Vol.IV: 1985-1989. CDRI, New Delhi: Lucknow & Publication & Information Directorate; 1995. p. 81. [Google Scholar]
- 7.Hayes PY, Jahidin AH, Lehmann R, Penman K, Kitching W, De Voss JJ. Steroidal saponins from the roots of Asparagus racemosus. Phytochemistry. 2008;69:796–804. doi: 10.1016/j.phytochem.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 8.Diwanay S, Chitre D, Patwardhan B. Immunoprotection by botanical drugs in cancer chemotherapy. J Ethnopharmacol. 2004;90:49–55. doi: 10.1016/j.jep.2003.09.023. [DOI] [PubMed] [Google Scholar]
- 9.Rao AR. Inhibitory action of Asparagus racemosus on DMBA-induced mammary carcinogenesis in rats. Int J Cancer. 1981;28:607–10. doi: 10.1002/ijc.2910280512. [DOI] [PubMed] [Google Scholar]
- 10.Ravikumar R, Chetty GL, Pandey RC, Sukhdev Chemistry of Ayurvedic Crude Drugs: Part VI-(Shatavari-1):Structure of Shatavarin-IV. Indian J Chem. 1987;26B:1012–7. [Google Scholar]
- 11.Vijayan P, Vinod Kumar S, Dhanaraj SA, Mukherjee PK, Suresh B. In vitro cytotoxicity and antitumor properties of Hypericum mysorense and Hyperium patulum. Phytother Res. 2003;1:952–6. doi: 10.1002/ptr.1271. [DOI] [PubMed] [Google Scholar]
- 12.Jagetia GC, Rao SK. Evaluation of antineoplastic activity Guduchi (Tinospora cordifolia) in Ehrlich Ascites Carcinoma bearing mice. Bio Pharm Bull. 2006;29:460–6. doi: 10.1248/bpb.29.460. [DOI] [PubMed] [Google Scholar]
- 13.Natesan S, Badani S, Dongre SH, Godavarthi A. Antitumor activity and antioxidant status of the methanol extract of Careya arborea bark against Dalton's lymphoma ascites-induced ascitic and solid tumor in mice. J Pharmacol Sci. 2007;103:12–23. doi: 10.1254/jphs.fp0060907. [DOI] [PubMed] [Google Scholar]
- 14.Gothoskar SV, Ranadive KJ. Anticancer screening of SAN-AB: An extract of marking nut, Semecarpus anacardium. Indian J Expt Biol. 1971;9:372–5. [PubMed] [Google Scholar]
- 15.Joharapurkar AA, Zambad SP, Wanjari MM, Umathe SN. In vivo evaluation of antioxidant activity of alcoholic extract of Rubia cordifolia Linn.and its influence on ethanol induced Immunosuppression. Indian J Pharmacol. 2003;35:232–6. [Google Scholar]
- 16.Vijayabaskaran M, Badkhal AK, Babu G, Sivakumar P, Perumal P, Sivakumar T, et al. Antitumor activity and antioxidant status of Symplocos racemosa Roxb. against Ehrlich ascites carcinoma in Swiss albino mice. Res J Pharm, Biol and Chemi Sci. 2010;1:306. [Google Scholar]
- 17.Price VE, Greenfield RE. In: Anemia in Cancer. In: Advances in Cancer Research. Greenstein JP, Haddow A, editors. New York: Academic Press; 1958. pp. 199–200. [DOI] [PubMed] [Google Scholar]
- 18.Maseki M, Nishigaki I, Hagishara M, Tomada Y, Yagi K. Lipid peroxide levels and lipid content of serum lipoprotein fractions of pregnant subjects with or without pre-eclampsia. Clin Chim Acta. 1981;115:155–61. doi: 10.1016/0009-8981(81)90071-1. [DOI] [PubMed] [Google Scholar]
- 19.Badami S, Reddy SM, Kumar EP, Vijayan P, Suresh B. Antitumor activity of total alkaloids fraction of Solanum pseudocapsicum leaves. Phytother Res. 2003;17:1001–4. doi: 10.1002/ptr.1229. [DOI] [PubMed] [Google Scholar]
- 20.Wiboonpun N, Phuwapraisirisan P, Tip-pyang S. Identification of antioxidant compound from Asparagus racemosus. Phytother Res. 2004;18:771–3. doi: 10.1002/ptr.1526. [DOI] [PubMed] [Google Scholar]
- 21.Bhatnagar M, Sisodia SS, Bhatnagar R. Antiulcer and antioxidant activity of Asparagus racemosus Willd and Withania somnifera Dunal in rats. Ann NY Acad Sci. 2005;1056:261–78. doi: 10.1196/annals.1352.027. [DOI] [PubMed] [Google Scholar]
- 22.Kamat JP, Boloor KK, Devasagayam TP, Venkatachalam SR. Antioxidant properties of Asparagus racemosus against damage induced by gamma-radiation in rat liver mitochondria. J Ethnopharmacol. 2000;71:425–35. doi: 10.1016/s0378-8741(00)00176-8. [DOI] [PubMed] [Google Scholar]


