Abstract
Purpose
Sarcoidosis is a non-caseating granulomatous disease for which a role for infectious antigens continues to strengthen. Recent studies have reported molecular evidence of mycobacteria or propionibacteria. We assessed for immune responses against mycobacterial and propionibacterial antigens in sarcoidosis bronchoalveolar lavage (BAL) using flow cytometry, and localized signals consistent with microbial antigens with sarcoidosis specimens, using matrix-assisted laser desorption ionization imaging mass spectrometry (MALDI-IMS).
Methods
BAL cells from 27 sarcoidosis, 14 PPD- controls, and 9 subjects with nontuberculosis mycobacterial (NTM) infections were analyzed for production of IFN-γ after stimulation with mycobacterial ESAT-6 and Propionibacterium acnes proteins. To complement the immunological data, MALDI-IMS was performed to localize ESAT-6 and Propionibacterium acnes signals within sarcoidosis and control specimens.
Results
CD4+ immunologic analysis for mycobacteria was positive in 17/27 sarcoidosis subjects, compared to 2/14 PPD-subjects, and 5/9 NTM subjects (p=00.008 and p=00.71 respectively, Fisher's exact test). There was no significant difference for recognition of P. acnes, which occurred only in sarcoidosis subjects that also recognized ESAT-6. Similar results were also observed for the CD8+ immunologic analysis. MALDI-IMS localized signals consistent with ESAT-6 only within sites of granulomatous inflammation, whereas P. acnes signals were distributed throughout the specimen.
Conclusions
MALDI-IMS localizes signals consistent with ESAT-6 to sarcoidosis granulomas, whereas no specific localization of P. acnes signals is detected. Immune responses against both mycobacterial and P. acnes are present within sarcoidosis BAL, but only mycobacterial signals are distinct from disease controls. These immunologic and molecular investigations support further investigation of the microbial community within sarcoidosis granulomas.
Keywords: Sarcoidosis, mycobacteria, propionibacteria, bronchoalveolar lavage, mass spectrometry, MALDI-IMS
Introduction
Sarcoidosis is a non-caseating granulomatous disease for which the role of infectious antigens continues to evolve. There are several lines of evidence suggesting an infectious origin, including clustering of cases, seasonal and geographic incidence, transmissibility, and the presence of oligoclonal TCR-αβ+CD4+ T cells at sites of granulomatous inflammation [1–7]. Multiple infectious agents have been suggested over the years as the etiologic agent; however, most recent reports favor either mycobacteria or propioni-bacteria as having a role in sarcoidosis pathogenesis.
Since the first description of sarcoidosis over a century ago, mycobacteria has been associated with the disease. Both the clinical presentation and histology demonstrate similarities to mycobacterial infection [8]. However, the many attempts to culture mycobacteria from sarcoid tissues have been unsuccessful. Advances in technology have renewed the association between mycobacteria and sarcoidosis. Mycobacterial proteins have been detected in sarcoidosis tissue samples using PCR, immunohistochemistry, and mass spectrometry [9–11]. Furthermore, independent laboratories have demonstrated mycobacterial-specific peripheral and pulmonary T cell immune responses [12–18].
There are also several studies in support of Propionibacterium acnes (P. acnes) having a role in sarcoidosis pathogenesis [19]. P. acnes is the only organism to have been cultured from sarcoidosis biopsy specimens [20]. In addition, a quantitative distinction in P. acnes DNA has been detected in sarcoid lymph nodes by both PCR and in situ hybridization, compared to control specimens [19, 21, 22]. Immune responses to P. acnes protein RP35 has been reported in ~18 % of sarcoidosis subjects, however these responses were not distinct from controls [23].
While a few studies have conducted molecular analysis of sarcoidosis specimens for both mycobacteria and propionibacteria, none to date have conducted both molecular and immunologic investigations for both organisms [19, 21]. In this study, we assessed for the presence of mycobacteria and propionibacteria in sarcoidosis bronchoalveolar lavage (BAL), using the complementary means of imaging mass spectrometry and flow cytometry. We analyzed for antigen-specific responses to mycobacterial early secreted antigenic target protein (ESAT-6) and P. acnes supernatant from BAL-derived T cells. In conjunction, we examined sarcoidosis tissue specimens for the presence of signals consistent with mycobacterial ESAT-6 and propionibacterial proteins using Matrix-Assisted Laser Desorption/Ionization (MALDI) Imaging Mass Spectrometry (IMS) [MALDI-IMS]. MALDI is a powerful tool for investigating the distribution of proteins and small molecules within biological systems through the in situ analysis of snap-frozen tissue sections [24].
Materials and Methods
Study Subjects
The study participants were recruited from The Cleveland Clinic (Cleveland, OH) and Vanderbilt University Medical Center (Nashville, TN). We prospectively enrolled patients who were undergoing bronchoscopy where sarcoidosis was a diagnostic consideration. All subjects provided written informed consent that was approved by the appropriate Institutional Review Boards. For the immunological response data, 50 subjects were enrolled in this study: 27 sarcoidosis subjects, 14 disease controls with a negative PPD test (PPD-), and 9 subjects with a nontuberculosis mycobacterial infection (NTM) such as Mycobacterium avium (Table I). Kruskal-Wallis analysis indicated there was a significant difference in age distributions across patient groups (p<0.001; Table I); a Pearson test performed across patient groups indicated no difference according to race or sex (Table I; bi-racial subjects were omitted from the race analysis). For the mass spectrometry analysis, 19 snap-frozen specimens (15 sarcoidosis and 4 controls) were analyzed (Table II). Demographic characteristics of the patient group and specimens are listed in Tables I and II, respectively. For each sarcoidosis subjects who underwent tuber-culin skin testing at the discretion of their physician, all were skin test negative. None of the sarcoidosis specimens were culture positive for infectious agents.
Table I.
Characteristics of the study population tested for immunologic response
| Characteristics | Sarcoidosis | PPD- | NTM | p value* |
|---|---|---|---|---|
| Number | 27 | 14 | 9 | |
| Sex, female/male | 17/10 | 11/3 | 5/4 | 0.46 |
| Age (yr), median (min, max) | 43 (25,64) | 49 (24,61) | 67 (51,75) | <0.001 |
| Race | 8AA:18 C:1AA/H | 3AA:10 C:1A/S | 1AA:8 C | 0.56 |
| Stage 0/I/II/III/IV | 1/10/15/1/0 | ND | ND | |
| IS at Branch | 10/17 | ND | ND | |
| Extrapulmonary disease, yes/no | 5/22 | ND | ND | |
| CD4+ response to ESAT-6 | 17 | 2 | 5 | 0.008 |
| CD4+ response to P. acnes | 6 | 6 | 3 | 0.38 |
| CD8+ response to ESAT-6 | 15 | 2 | 3 | 0.03 |
| CD8+ response to P.acnes | 6 | 5 | 2 | 0.68 |
| Percent CD4 of Total CD3+ cells median (min,max) | 65.2 (48.9,81.2) | 54.8 (34.6,68.7) | 70.9 (45.4, 90.2) | |
| Percent CD8 of Total CD3+ cells median (min,max) | 30.4 (17.1,50.8) | 42.7 (31.8,63.9) | 25.4 (12.7, 53.8) | |
| CD4/CD8 ratio, median (min,max) | 2.60 (0.56,70.54) | 1.30(0.19,36.57) | 2.95 (0.49,9.01) |
NTM non-Tuberculosis mycobacterial infection; yr years, min minimum, max maximum, ND not done, A Asian, AA African American, C Caucasian, H Hispanic, S Samoan, IS immunesuppressants.
The p values listed are for testing across all three patient groups. Statistical differences for age were assessed using the Wilcoxon rank sum test and for sex and race using the Pearson test. Statistical difference for the CD4+ and CD8+ response to ESAT-6 and P. acnes were calculated using Fisher's exact test. CD4+ or CD8+ response was defined as positive when the frequency of recognition was at least twice background fluorescence and greater than 0.5 %
Table II.
Demographic and pathologic information for sarcoidosis and control tissue specimens
| Diagnosis | Age/Race/Sex | Source of specimen | Pathologic diagnosis | Microbial signal |
|---|---|---|---|---|
| Sarcoid PP | 59AAF | Breast | Sarcoidosis | ESAT-6, P. acnes |
| Sarcoid RR | 39CF | Lymph Node | Sarcoidosis | ESAT-6 |
| Sarcoid ZZ | 68CM | Lymph Node | Sarcoidosis | P. acnes |
| Sarcoid 09 | 60CF | Lymph Node | Sarcoidosis | ESAT-6, P. acnes |
| Sarcoid 1 | 41AAF | Cutaneous | Sarcoidosis | |
| Sarcoid 2 | 55AF | Cutaneous | Sarcoidosis | P. acnes |
| Sarcoid 7 | 56AAF | Cutaneous | Sarcoidosis | |
| Sarcoid 10 | 57CF | Cutaneous | Sarcoidosis | ESAT-6 |
| Sarcoid 11 | 64CF | Cutaneous | Sarcoidosis | P. acnes |
| Sarcoid 14 | 65CF | Cutaneous | Sarcoidosis | |
| Sarcoid 15 | 49CM | Cutaneous | Sarcoidosis | ESAT-6 |
| Sarcoid 17 | 39AAF | Cutaneous | Sarcoidosis | P. acnes |
| Sarcoid 19 | 60AAF | Cutaneous | Sarcoidosis | |
| Sarcoid 20 | 53CF | Cutaneous | Sarcoidosis | P. acnes |
| Sarcoid 21 | 62CF | Cutaneous | Sarcoidosis | |
| Control AH | 74CF | Lung | Squamous cell carcinoma | |
| Control D | 15M | Lymph Node | Hodgkin's lymphoma | |
| Control N | 56CF | Lymph Node | Normal lymph node | P. acnes |
| Control T | 65CF | Lung | Squamous cell carcinoma |
Age age in years; F Female, M Male, A Asian, AA African American, C Caucasian
Synthesis of Mycobacterium Peptides
The ESAT-6 peptide, NNALQNLARTISEANTIGENQAMAS, was synthesized as described previously [25]. Each peptide was synthesized by solid-phase F-moc chemistry (Genemed Synthesis, San Diego, CA, USA), to a purity of>70%. Identity was confirmed by mass spectroscopy, and the purity was assessed by high performance liquid chromatography.
Synthesis of Propionibacterium Acnes Supernatant
One mg of Propionibacterium acnes (P. acnes) dried culture (ATCC # 6919) was transferred to a UV sterilized eppendorf tube. Six hundred μl of 1× PBS was added to the vial and reconstituted. The solution was boiled in water at 100°C for 10 min, then centrifuged at 14000 rpm for 10 min. The supernatant, at a concentration of 20 μg/ml, was used to stimulate sarcoidosis and control BAL cells.
BAL Cell Isolation and Culture
BAL fluid was obtained from diagnostic bronchoscopy and centrifuged at 1500 rpm for 15 min. The BAL cell pellet was washed with RPMI 1640 supplemented medium. BAL cells were either stored in liquid nitrogen in freezing media for future analysis or analyzed immediately. The culture medium used in all experiments was RPMI (Cellgro) and was prepared as previously described [26].
Flow Cytometry—Cytokine and Proliferation Assays
Intracellular staining was performed in order to identify IFN-γ secreting BAL-derived T cells in response to microbial stimulation. For the intracellular cytokine assay, 1–2×105 BAL cells were incubated in RPMI 1640 supplemented medium with or without antigen (ESAT-6 peptide or P. acnes supernatant; 20 μg/ml) or staphylococcal enterotoxin B (SEB; 10 μg/ml; Sigma) as positive control and the anti-CD28 and anti-CD49d Abs (1 μg/ml each; BD Biosciences) at 37°C under 5 % CO2 for 1 h before addition of BD GolgiStop (BD Biosciences). In addition, to assess for nonspecific recognition, neoantigen Keyhole Limpet Hemocyanin (KLH; 10 μg/ml; Calbiochem) was included as an additional negative control. Following a 6 h incubation at 37°C under 5 % CO2, cells were washed and stained with the surface antibodies anti-CD3, anti-CD4, and anti-CD8 (BD Biosciences) at 4°C for 30 min. After washing, fixation, and permeabilization, using a Fix & Perm Kit according to the manufacturer's instructions (BD Biosciences), anti-IFN-γ (BD Biosciences) was added at 4°C for 45 min. Cells were washed and analyzed via flow cytometry. The IFN-γ frequency was defined as the subject's percentage of stimulated CD3+CD4+ or CD3+CD8+ T cells minus their unstimulated background frequency. Based upon previous studies, a response was considered positive when the frequency of recognition was at least twice background fluorescence and greater than 0.5 % [27–29].
For the extracellular cytokine assay, 1–2×105 BAL cells were incubated in RPMI 1640 supplemented medium with or without ESAT-6 peptide or P. acnes supernatant (20 μg/ml), SEB, or KLH and the anti-CD28 and anti-CD49d (1 μg/ml each; BD Biosciences) at 37°C under 5 % CO2 for 24 h. Supernatants were then collected and analyzed for extracellular cytokine by using cytokine bead array according to the manufacturer's instructions (BD Biosciences).
All flow cytometry experiments were acquired with a FACSCalibur or LSR-II flow cytometer (BD Biosciences). Live cells were gated based on forward and side scatter properties and analysis was performed using FlowJo software (Tree Star). A minimum of 30,000 events were acquired per sample.
To determine proliferation and quantitate cell division, purified BAL cells were labeled with carboxyfluorescein succinimidyl ester (CFSE; Molecular Probes). Purified cells were first washed and resuspended in PBS. While vortexing the cells, CFSE was added at a final concentration of 5 μM. The mixture was vortexed for an additional 15 s and incubated at 37°C for 3 min. Labeling was quenched by the addition of 50 % FBS in PBS. Cells were washed once more with 50 % serum PBS, followed by two washes with RPMI 1640-supplemented medium. CFSE-labeled BAL cells were stimulated in RPMI 1640 supplemented medium with or without 10 μM peptide or SEB as positive control.
MALDI Analysis
MALDI-IMS was performed as previously described [30]. Basically, imaging mass spectrometry utilizes matrix-assisted laser desorption ionization time of flight mass spectrometry (MALDI-TOF-MS) to profile and map the distribution of proteins present in thin tissue sections. Nineteen snap frozen sarcoidosis specimens were analyzed using a histology directed mass spectral profiling approach [30]. In this technique, two sections of a tissue sample are collected; one that is stained for histological analysis and a serial unstained section on a MALDI target. Mass spectrometric analyses were performed in the linear, positive mode at +20 kV accelerating potential on a time-of-flight mass spectrometer (Bruker Autoflex II; Bruker Daltonik, Bremen, Germany), which was equipped with a SmartBeam™ laser (Nd:YAG, 355 nm) capable of operating at a repetition rate of 200 Hz. Image data were collected by coating the tissue section with sinapinic acid matrix in a regular array with a 300 μm center-to-center spacing using a Portrait 630 acoustic robotic spotter (Labcyte, Inc. Sunnyvale, CA). Spectra were acquired as described above and the protein ion images were viewed using FlexImaging (Bruker).
Statistical Analysis
Comparisons of the distribution of T cell frequencies were performed using the Wilcoxon rank sum test. Categorical comparisons, such as immune reactivity to mycobacterial antigens by individuals within a group, were analyzed using Fisher's exact test. Multiple comparisons were performed. All performed comparisons are reported, all p values are two-sided, and all analyses were performed using R (Version 2.1.1).
Results
Sarcoidosis CD4+ T Cells Exhibit Significant Distinctions in Microbial-Specific Recognition
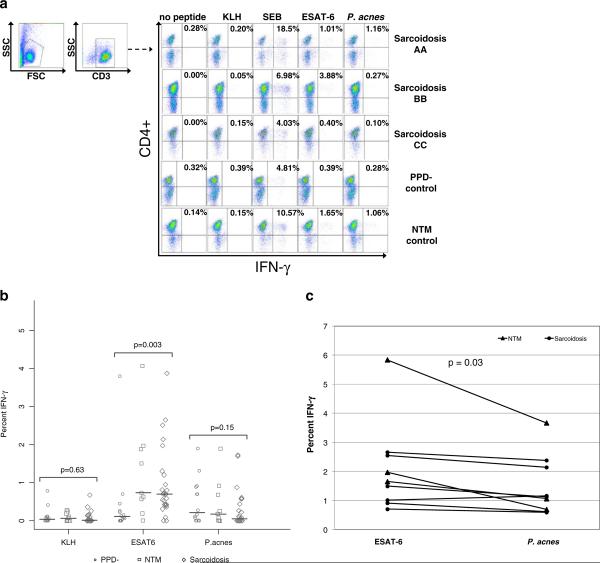
BAL cells from sarcoidosis subjects, disease controls with a negative PPD test (PPD-), or subjects with a nontuberculous mycobacteria (NTM) infection were stimulated with ESAT-6 peptide or P. acnes supernatant, then the CD4+ T cells were analyzed for intracellular cytokine production of IFN-γ by flow cytometry. Bronchoalveolar lavage (BAL) cells stimulated by staphylococcal enterotoxin B (SEB) served as the positive control, and BAL cells alone as the negative control. In order to assess for nonspecific recognition, neoantigen keyhole limpet hemocyanin (KLH), was included as an additional negative control. Seventeen of the 27 sarcoidosis BAL samples displayed a CD4+ IFN-γ response to ESAT-6 compared to two of the 14 PPD- controls and five of nine NTM controls (p=0.008, Fisher's exact test) (Table I). The distribution of the CD4+ T cell responses against ESAT-6 was significantly higher in the sarcoidosis subjects, compared to PPD- controls (p=0.003, Wilcoxon) (Fig. 1A, B). In contrast, the response to P. acnes was not significantly distinct between these two groups (p=0.15) (Fig. 1B). Six of 27 BAL samples displayed a CD4+ IFN-γ response to P. acnes compared to six of the 14 PPD- controls and three of the nine NTM controls (Table I)(p=0.38, Fisher's exact test). No responses to KLH in any of the populations were observed (Fig. 1B). Comparing CD4+ T cell responses to ESAT-6 and P. acnes antigen among the sarcoidosis subjects, there was a significant difference in the percentage of sarcoidosis subjects recognizing ESAT-6 antigen (17/27) versus P. acnes (6/27) (p=0.003, McNemar's test). It was interesting to note that the six sarcoidosis and three NTM subjects who displayed P. acnes recognition also recognized ESAT-6 (Fig. 1C). Among the sarcoidosis subjects who recognized both antigens, there were distinctions in the number of ESAT-6-specific CD4+ T cells, compared to P. acnes (p=0.03, Wilcoxon signed rank test) (Fig. 1C). No sarcoidosis or NTM subject recognized P. acnes solely.
Fig. 1.
Sarcoidosis BAL T cells demonstrated higher CD4+ T cell specific recognition of mycobacterial antigen compared to P. acnes. Intracellular cytokine staining for IFN-γ was performed on BAL cells from sarcoidosis, PPD-, and NTM subjects, after stimulation with neoantigen (KLH), ESAT-6 peptide, P. acnes supernatant, or SEB (positive control). a Shown are representative flow cytometry dot plots indicating percentage of CD3+CD4+ T cell responses to mycobacterial antigen from a sarcoidosis subject who demonstrated dual recognition of ESAT-6 and P. acnes (Sarcoidosis AA), a subject who recognized ESAT-6 solely (Sarcoidosis BB), a subject who displayed no recognition of either bacteria (Sarcoidosis CC), a NTM control, and a PPD-control. b Percent of CD3+CD4+ T cells that produced IFN-γ in response to KLH, ESAT-6, or P. acnes stimulation with no peptide control subtracted out for all 27 sarcoidosis subjects, 14 PPD- controls, and 9 NTM controls. Differences in the distributions of immune recognition of mycobacterial antigen by BAL CD3+CD4+ T cells were noted between sarcoidosis and disease controls, using the Wilcoxon rank sum test. Medians depicted by lines on figure. A response was considered positive when the frequency of recognition was at least twice background fluorescence and greater than 0.5 %. Stimulation of BAL cells with SEB resulted in a positive IFN-γ response for sarcoidosis subjects and disease controls. c The six sarcoidosis and three NTM subjects who displayed P. acnes recognition also recognized ESAT-6
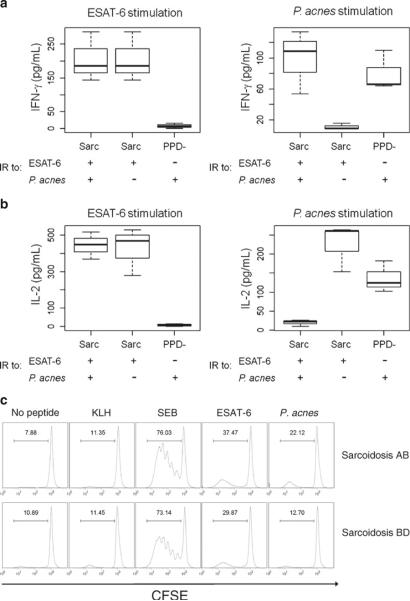
To extend these results, extracellular Th-1 cytokine expression following stimulation with ESAT-6 peptide or P. acnes supernatant for 24 h was obtained from BAL cells from three subjects who recognized P.acnes and mycobacterial-specific response and compared to three who displaced only immune responses against ESAT-6 peptide. The cells were chosen solely based upon cell availability. The ESAT-6 peptide elicited quantitatively higher levels of IFN-γ and IL-2 production as compared to the P. acnes supernatant (Fig. 2A, B). Furthermore, the cytokine levels induced by the P. acnes supernatant in the sarcoidosis subjects were similar to those of the PPD- controls who displayed a P. acnes specific response (Fig. 2A, B). In addition, BAL cells from the three subjects who displayed a P. acnes and mycobacterial-specific response demonstrated increased proliferation in response to ESAT-6 stimulation as compared to P. acnes stimulation (Fig. 2C). Taken together these results suggest that mycobacterial antigens elicit a stronger antigen-specific response as measured by cytokine production and proliferation, compared to P. acnes antigens.
Fig. 2.
Sarcoidosis BAL cells secrete Th1 cytokines and proliferate in response to mycobacterial antigen stimulation. Supernatants were collected from BAL cells 24 h after stimulation with neoantigen (KLH), ESAT-6 peptide, P. acnes supernatant, or SEB (positive control). Data for three sarcoidosis subjects who displayed immune responses (IR) to ESAT-6 and P. acnes, three sarcoidosis subjects who displayed responses to ESAT-6 but not P. acnes, and three PPD-controls who displayed responses to only P. acnes. a IFN-γ. b IL-2. There was a significant difference in extra-cellular Th1 cytokine production to mycobacterial antigens between sarcoidosis and control subjects. c Sarcoidosis BAL cells from subject who displayed responses to both ESAT-6 and P. acnes (Sarcoidosis AB) and subject who displayed responses only to ESAT-6 (Sarcoidosis BD) were CFSE-labeled and activated with neo-antigen (KLH), ESAT-6 peptide, P. acnes supernatant, or SEB. Day 4 post-activation, antigen-specific proliferation of CD4+ cells was assessed by gating on CD3+CD4+ T cells and analyzing the CFSE expression of this subset by flow cytometry. Percent proliferation is indicated by bracket above peaks. Similar results were found in the three other tested subjects
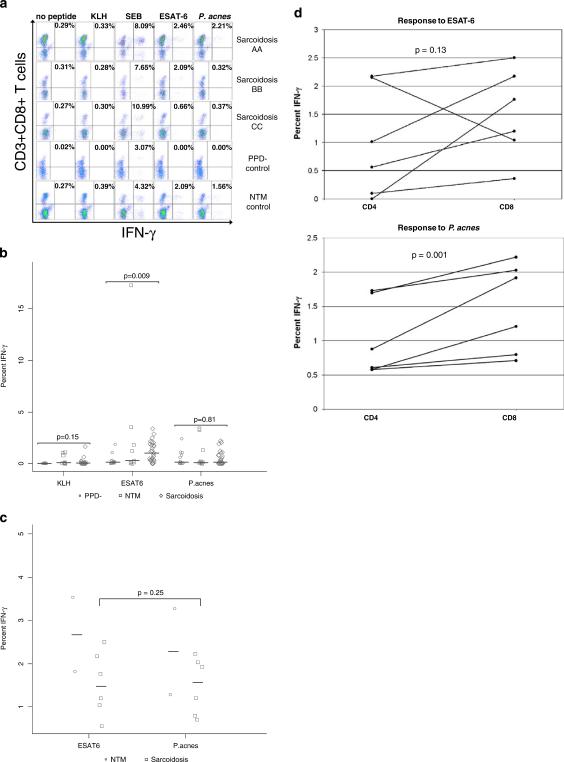
Sarcoidosis CD8+ T Cells Possess Stronger Responses to Mycobacterial Antigen than P. Acnes
Recently, mycobacterial-specific CD8+ T cell responses have been demonstrated in sarcoidosis subjects [12, 17]. To determine if mycobacterial and propionibacterial antigens elicit a CD8+ T cell immune response in sarcoidosis or control subjects, BAL cells were stimulated with ESAT-6 peptides or P. acnes supernatant, then the CD8+ T cells were analyzed for intracellular cytokine production of IFN-γ by flow cytometry. There was lack of significant immune responses to the neoantigen KLH; however strong CD8+ T cell responses were observed to ESAT-6 (Fig. 3A). Indeed, 15 of the 27 sarcoidosis subjects exhibited CD8+ IFN-γ responses compared to two of the 14 PPD- controls and three of the nine NTM controls (p=0.02 and 0.44 respectively, two-tailed Fisher's exact test) (Table I). The distribution of the CD8+ T cell responses against ESAT-6 was significantly higher in the sarcoidosis subjects, compared to PPD- controls (p=0.009, Wilcoxon) (Fig. 3A, B). In contrast, the response to P. acnes was not significantly distinct between these two groups (p=0.81) (Fig. 3A, B). Six of 27 BAL samples displayed a CD8+ IFN-γ response to P. acnes compared to five of the 14 PPD- controls and two of the nine NTM controls (p=0.46 and 1.0 respectively, two-tailed Fisher's exact test) (Table I). There was a significant difference in the percentage of sarcoidosis subjects with CD8+ T cell responses to ESAT-6 (15/27) compared to P. acnes (6/27) (p=0.008, McNemar's test) (Table I). Congruent with the CD4+ T cell responses, the same six sarcoidosis and two NTM subjects who displayed CD8+ antigen-specific responses against P. acnes also recognized ESAT-6 (Fig. 3C) and no sarcoidosis or NTM subjects recognized P. acnes solely. There was not a statistical difference in the magnitude of responses to ESAT-6 as compared to P. acnes in the sarcoidosis subjects who recognized both (p=0.25, Wilcoxon signed rank test) (Fig. 3C). Interestingly, for the dual responders there was a trend that the magnitude of the CD8+ T cell response was greater as compared to the CD4+ T cell response for both ESAT-6 and P. acnes stimulation (p=0.13 and p=0.001, respectively, Wilcoxon signed rank test) (Fig. 3D).
Fig. 3.
Sarcoidosis BAL CD8+ T cells display antigen-specific Th1 immune responses to mycobacterial antigen. Intracellular cytokine staining for IFN-γ was performed on BAL cells from sarcoidosis subjects and disease controls, after stimulation with neoantigen (KLH), ESAT-6 peptide, P. acnes supernatant, or SEB. a Shown are representative flow cytometry dot plots indicating percentage of CD3+CD4+ T cell responses to mycobacterial antigen from a sarcoidosis subject who demonstrated dual recognition of ESAT-6 and P. acnes (Sarcoidosis AA), a subject who recognized ESAT-6 solely (Sarcoidosis BB), a subject who displayed no recognition of either bacteria (Sarcoidosis CC), a NTM control, and a PPD- control. b Percent of CD3+CD8+ T cells that produced IFN-γ in response to KLH, ESAT-6, P. acnes supernatant stimulation with no peptide control subtracted out for sarcoidosis subjects and disease controls. Differences in the distributions of immune recognition of mycobacterial antigen by BAL CD3+CD8+ T cells were noted between the sarcoidosis and control subjects using the Wilcoxon rank sum test. Medians depicted by lines on figure. Stimulation of BAL cells with SEB resulted in a positive IFN-γ response for sarcoidosis subjects and disease controls. c The six sarcoidosis and two NTM subjects who displayed P. acnes recognition also recognized ESAT-6. d In the six sarcoidosis subjects who demonstrated dual recognition of the antigens, the trend was the CD8+ T cell response was stronger than the CD4+ T cell response. Differences in the distributions of immune recognition of ESAT-6 and P. acnes by BAL CD3+CD4+ and CD3+CD8+ T cells were measured using the Wilcoxon signed rank test
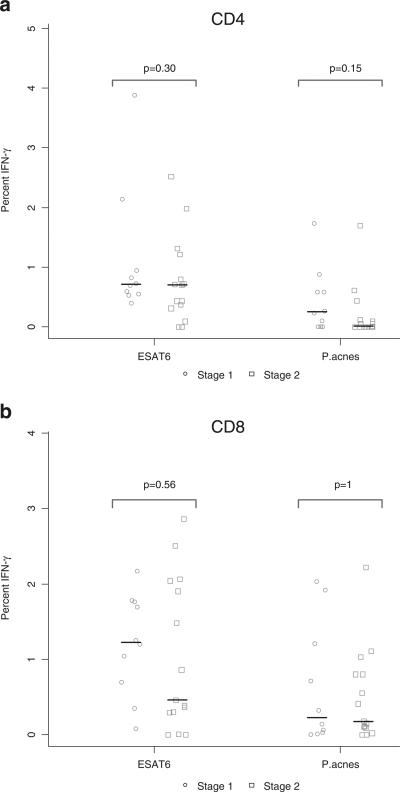
Presence of Bacteria Does Not Correlate with Radiographic Stages of Sarcoidosis
To rule out the possibility that the presence of mycobacteria and propionibacteria correlates with a specific radiographic stage, we analyzed the CD4+ and CD8+ T cell immune response data across stages I and II (Fig. 4A and B). We omitted stage 0 and stage III from the analysis because each stage only had one subject, and there were no subjects with stage IV. We previously showed no correlation with mycobacterial recognition and radiographic stage [17]. These results were confirmed in this study as well for both mycobacterial and propionibacterial recognition (Fig. 4A, B).
Fig. 4.
Presence of bacteria does not correlate with radiographic stages of sarcoidosis. Tested immune responses to ESAT-6 and P. acnes across radiographic stage. Medians depicted by lines on figure. Differences in the distribution of immune recognition in sarcoidosis patients across radiographic stage I and II were assessed using the Kruskal-Wallis rank sum test. There was no significant difference in the strength of response between stages. a CD4+ T cell immune response. b CD8+ T cell immune response
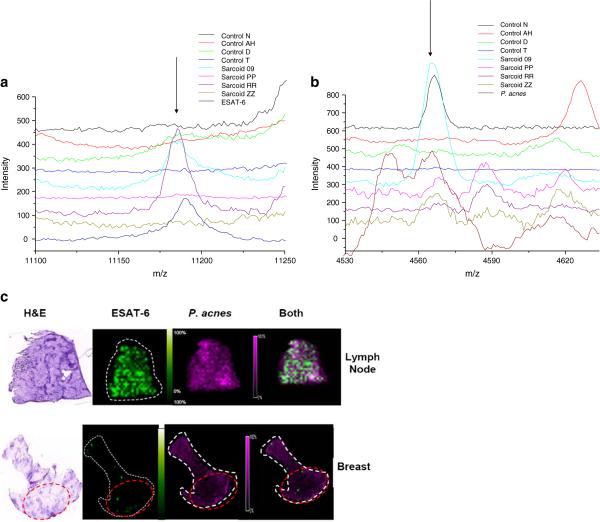
Signals Consistent with ESAT-6 and P. Acnes Are Present within Sarcoidosis Granulomas
Prior reports noted the presence of mycobacterial and propionibacterial proteins within sarcoidosis specimens, however precise localization within sarcoidosis and control specimens has not been performed to date. MALDI-IMS is a powerful tool for investigating the distribution of proteins and small molecules within biological systems. We utilized MALDI-IMS technology to localize ESAT-6 and propioni-bacterial proteins in tissue sections. We analyzed 15 snap-frozen sarcoidosis specimens [lymph node (3), skin (11), and breast (1)] and four snap-frozen control specimens [lung (2), lymph nodes (2)] (Table II), in conjunction with analysis of ESAT-6 protein and P. acnes protein supernatant. A protein signal possessing a mass-to-charge ratio (m/z) of 11,190 was detected in some sarcoidosis specimens and consistent with a signal from ESAT-6 whole protein, which exists as dimer. Signals consistent with the P. acnes supernatant (m/z 4561) were detected also (Table II). We detected signals consistent with ESAT-6 and/or P. acnes within 10 of 15 sarcoidosis samples. Representative analyses are shown in Fig. 5A, B. MALDI-IMS localized signals consistent with ESAT-6 to sites of granulomatous inflammation within the sarcoidosis specimens (Fig. 5C). For example, in a lymph node specimen that possessed granulomas throughout the specimens, signals consistent with ESAT-6 and P. acnes were seen throughout; however, in the breast biopsy which possessed discrete sites of granulomatous involvement, MALDI-IMS of this sample revealed that no ESAT-6 signal was detected outside of sites of granulomatous inflammation (Fig. 5C), whereas P. acnes signals (m/z 4561) were. It was also notable that variation in signal intensity was observed between samples, as well as within a given sample, suggesting variation in protein expression as well. Signals consistent with the P. acnes supernatant (m/z 4561) were detected throughout the sarcoidosis lymph node and breast biopsy specimens (Fig. 5B and C). Localization of the signals occurred irrespective of granulomatous involvement. Again there was variation in the signal intensity, insinuating variation in protein expression between samples, as well as within a given specimen. Signals consistent with P. acnes was noted in a single control lymph node (Table II).
Fig. 5.
MALDI-IMS localizes signals consistent with mycobacterial ESAT-6 and P. acnes within sarcoidosis granulomatous inflammation of a lymph node and a breast biopsy. Distinct protein signals corresponding to ESAT-6 and P. acnes were present within sarcoidosis specimens. (A/B) Signals consistent with ESAT-6 (~11,190 m/z) and P. acnes (~4,560 m/z) were present within sarcoidosis specimens. A representation of the peaks visualized with MALDI analysis of 15 snap-frozen sarcoidosis and four snap-frozen control specimens is depicted; 10 of 15 contained either ESAT-6 and/or P. acnes, compared to 1of 4 control specimen containing signals consistent with P. acnes. No signals consistent with ESAT-6 were noted in the control specimens. c Histologic staining of lymph nodes and the breast biopsy revealed granulomas throughout the lymph node, but only distinct areas of involvement within the breast biopsy (red circle). Localization of signals consistent with ESAT-6 and P. acnes were detected throughout both specimens. ESAT-6 signals were noted within areas of granulomatous involvement; whereas, signals consistent with P. acnes were present irrespective of granuloma involvement in the sarcoidosis specimen
Discussion
This study is the first to perform dual molecular and immunologic analysis for mycobacteria and propionibacteria in sarcoidosis specimens. This study confirms that while CD4+ and CD8+ immune responses to mycobacteria and propionibacteria are present within sarcoidosis BAL, they are distinct for mycobacterial recognition but not P. acnes, when compared to disease controls. Using MALDI-IMS, localization of signals consistent with mycobacterial ESAT-6 are present within sarcoidosis granulomas, whereas nonspecific localization of signals consistent with propioni-bacterial proteins was noted.
A prior report found an association between sarcoidosis and propionibacteria in sarcoidosis specimens [31]. In this study, we found that the six sarcoidosis and three NTM subjects who demonstrated CD4+ T cell specific recognition of P. acnes also recognized the mycobacterial antigen, ESAT-6 (Fig. 1). This was also true for the CD8+ T cell recognition (Fig. 3). All the sarcoidosis specimens were culture negative for M. tuberculosis. The detection of immune responses against ESAT-6 among subjects with non-tuberculous mycobacterial infection is noteworthy. The genome of organisms such as M. kansasii, M. szulgai, M. marinum, and M. riyadhense contain Region of Difference-1(RD-1), which encodes ESAT-6 and CFP-10. Secretion of ESAT-6 by NTM has also been previously reported [32, 33]. We chose NTM as a positive control because of these observations. Prior work has suggested an association of mycobacteria and propionibacteria in other granulomatous diseases such as tuberculosis and leprosy [34]. In this study, a direct association with P. acnes and sarcoidosis in BAL was not found. However, six sarcoidosis subjects demonstrated concomitant recognition of mycobacteria and propionibacteria. It is possible that systemic immune responses against ESAT-6 and P. acnes may also be present within sarcoidosis systemic responses.
Interestingly, our immunologic data demonstrates that mycobacteria elicit a stronger IFN-γ response compared to P. acnes. There are several studies suggesting that the strength of the immune response correlates with disease outcome in bacterial infections including tuberculosis [35]. We did not find a significant difference in immune responses across radiographic stages, however our analysis was performed on Stage I and II sarcoidosis BAL due to limitations of diagnostic BAL available for other sarcoidosis stages and thus these findings cannot be generalized to Stages 0, III, and IV.
Furthermore, in a recent report the cell invasiveness of P. acnes isolates from sarcoid lesions was studied and no differences were found between sarcoidosis and disease controls [36]. It is also noteworthy that despite the presence of mycobacterial antigen-specific CD4+ T cell responses in ~60 % of sarcoidosis subjects, the sarcoidosis BAL specimens are culture negative. Possible explanations for this include the following: 1) Although viable organisms are absent from sarcoidosis specimens, persistent microbial proteins remain. These proteins drive antigen-specific immune responses. 2) Viable organisms are present within sarcoidosis BAL that cannot be isolated under current culture conditions. This latter hypothesis will require the new techniques to identify viable mycobacteria within sarcoidosis specimens. These observed responses do not reflect active or latent tuberculosis infection. Each BAL specimen was culture negative for mycobacteria confirming that this is not M. tuberculosis infection. One also does not see immune responses against ESAT-6 in the BAL of subjects with latent tuberculosis [29].
One argument in support of the role of P. acnes in sarcoidosis pathogenesis is that there are quantitative differences in P. acnes DNA in Japanese and European sarcoidosis subjects as compared to disease controls [21]. A counterargument to this is that the increased levels of P. acnes DNA may reflect the replication of the organism to provide co-factors to support another microbe, such as mycobacteria. To address this point, we performed MALDI-IMS for microbial proteins in order to assess in a complementary fashion for mycobacteria and propionibacteria. Direct analysis of tissues of biological and clinical interest using MALDI-IMS has been shown to be successful for the study of the mid- to low molecular weight proteome and has been successfully used to characterize the expression of proteins and other organic biological compounds in numerous normal and diseased tissues [37, 38]. MALDI has been demonstrated for ultrasensitive detection to peptide amounts as low as 50×10−17 (50 ag). Utilizing this technology, signals consistent with ESAT-6 or P. acnes was detected in 10 of 15 sarcoidosis granulomatous samples. MALDI-IMS localized these ESAT-6 signals to sites of granulomatous inflammation while P. acnes proteins were detected throughout the tissue specimens of sarcoidosis and control samples (Fig. 5). ESAT-6 has been demonstrated to be a nidus for granuloma formation within tuberculosis specimens [39]. The work presented demonstrates only signals consistent with ESAT-6 and not the actual protein. However, the detection of signals consistent with ESAT-6 in sarcoidosis granulomas and absent from control specimens supports a more indepth investigation for ESAT-6 proteins within sarcoidosis granulomas.
Another plausible scenario is that both mycobacteria and propionibacteria cause sarcoidosis independently of each other. Several Japanese studies have identified propionibacteria in sarcoidosis utilizing multiple techniques [19, 21, 22]. Differences among this study and previously published work may be due to the racial composition of the study population. Sarcoidosis in Japanese subjects is characterized by eye, skin, and cardiac involvement, while in the United States and Europe, mainly affects the lungs. In this study, all subjects were from the United States and were either of African American or Caucasian background. Future investigations including subjects from the United States and Japan need to be conducted to determine if the different disease manifestations are the result of different infectious agents.
The localization of signals consistent with microbial proteins within sarcoidosis granulomas, as well as demonstration that they are targets of the adaptive immune responses further strengthens the role of infectious agents within sarcoidosis pathogenesis. The presence of responses against mycobacteria and propionibacteria, as well as evidence of signals consistent with both proteins within sarcoidosis granulomas suggest that these agents make important contributions. The relationship of mycobacteria and propionibacteria in sarcoidosis pathogenesis requires further investigation. Dissecting this relationship may lead to better therapeutic interventions, as well as identify mechanisms which determine disease outcome.
Acknowledgements
We thank Joan Isom, L.P.N. for her assistance with data collection and coordination and Jamie Allen for sample preparation for MALDI analysis.
Funding This work was supported by National Institutes of Health grants (RO1-HL83839; R01-AI65744; MO1 RR-00095, The Eliassen Foundation and Vanderbilt CTSA grant 1 UL1 RR024975 to W.P.D., T32 HL069765 to K.R., 5RO1-GM058008-11 to R.M.C.), funding from NFCR Center for Proteomics and Drug Action to R.M.C., and Vanderbilt Ingram Cancer Center Core Support Grant 5P30-CA068485-13 to E.H.S.
Footnotes
Conflict of interest statement The authors declare that they have no conflict of interest.
References
- 1.M'Koma AE, Blum DL, Norris JL, Koyama T, Billheimer D, Motley S, et al. Detection of pre-neoplastic and neoplastic prostate disease by MALDI profiling of urine. Biochem Biophys Res Commun. 2007;353(3):829–34. doi: 10.1016/j.bbrc.2006.12.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yanagisawa K, Shyr Y, Xu BJ, Massion PP, Larsen PH, White BC, et al. Proteomic patterns of tumour subsets in non-small-cell lung cancer. Lancet. 2003;362(9382):433–9. doi: 10.1016/S0140-6736(03)14068-8. [DOI] [PubMed] [Google Scholar]
- 3.Demirkok SS, Basaranoglu M, Coker E, Karayel T. Seasonality of the onset of symptoms, tuberculin test anergy and Kveim positive reaction in a large cohort of patients with sarcoidosis. Respirology. 2007;12(4):591–3. doi: 10.1111/j.1440-1843.2007.01062.x. [DOI] [PubMed] [Google Scholar]
- 4.Grunewald J, Wahlstrom J, Berlin M, Wigzell H, Eklund A, Olerup O. Lung restricted T cell receptor AV2S3+ CD4+ T cell expansions in sarcoidosis patients with a shared HLA-DRbeta chain conformation. Thorax. 2002;57(4):348–52. doi: 10.1136/thorax.57.4.348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kajdasz DK, Judson MA, Mohr LC, Jr, Lackland DT. Geographic variation in sarcoidosis in South Carolina: its relation to socioeconomic status and health care indicators. Am J Epidemiol. 1999;150(3):271–8. doi: 10.1093/oxfordjournals.aje.a009998. [DOI] [PubMed] [Google Scholar]
- 6.Morimoto T, Azuma A, Abe S, Usuki J, Kudoh S, Sugisaki K, et al. Epidemiology of sarcoidosis in Japan. Eur Respir J. 2007 Oct 24; doi: 10.1183/09031936.00075307. [DOI] [PubMed] [Google Scholar]
- 7.Wilsher ML. Seasonal clustering of sarcoidosis presenting with erythema nodosum. Eur Respir J. 1998;12(5):1197–9. doi: 10.1183/09031936.98.12051197. [DOI] [PubMed] [Google Scholar]
- 8.Sharma OP. Murray Kornfeld, American College Of Chest Physician, and sarcoidosis: a historical footnote: 2004 Murray Kornfeld Memorial Founders Lecture. Chest. 2005;128(3):1830–5. doi: 10.1378/chest.128.3.1830. [DOI] [PubMed] [Google Scholar]
- 9.Drake WP, Pei Z, Pride DT, Collins RD, Cover TL, Blaser MJ. Molecular analysis of sarcoidosis tissues for mycobacterium species DNA. Emerg Infect Dis. 2002;8(11):1334–41. doi: 10.3201/eid0811.020318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dubaniewicz A, Dubaniewicz-Wybieralska M, Sternau A, Zwolska Z, Izycka-Swieszewska E, Augustynowicz-Kopec E, et al. Mycobacterium tuberculosis complex and mycobacterial heat shock proteins in lymph node tissue from patients with pulmonary sarcoidosis. J Clin Microbiol. 2006;44(9):3448–51. doi: 10.1128/JCM.01433-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Song Z, Marzilli L, Greenlee BM, Chen ES, Silver RF, Askin FB, et al. Mycobacterial catalase-peroxidase is a tissue antigen and target of the adaptive immune response in systemic sarcoidosis. J Exp Med. 2005;201(5):755–67. doi: 10.1084/jem.20040429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen ES, Wahlstrom J, Song Z, Willett MH, Wiken M, Yung RC, et al. T cell responses to mycobacterial catalase-peroxidase profile a pathogenic antigen in systemic sarcoidosis. J Immunol. 2008;181(12):8784–96. doi: 10.4049/jimmunol.181.12.8784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Allen SS, Evans W, Carlisle J, Hajizadeh R, Nadaf M, Shepherd BE, et al. Superoxide dismutase A antigens derived from molecular analysis of sarcoidosis granulomas elicit systemic Th-1 immune responses. Respir Res. 2008;9:36. doi: 10.1186/1465-9921-9-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carlisle J, Evans W, Hajizadeh R, Nadaf M, Shepherd B, Ott RD, et al. Multiple Mycobacterium antigens induce interferon-gamma production from sarcoidosis peripheral blood mononuclear cells. Clin Exp Immunol. 2007;150(3):460–8. doi: 10.1111/j.1365-2249.2007.03510.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Drake WP, Dhason MS, Nadaf M, Shepherd BE, Vadivelu S, Hajizadeh R, et al. Cellular recognition of Mycobacterium tuberculosis ESAT-6 and KatG peptides in systemic sarcoidosis. Infect Immun. 2007;75(1):527–30. doi: 10.1128/IAI.00732-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hajizadeh R, Sato H, Carlisle J, Nadaf MT, Evans W, Shepherd BE, et al. Mycobacterium tuberculosis Antigen 85A induces Th-1 immune responses in systemic sarcoidosis. J Clin Immunol. 2007;27(4):445–54. doi: 10.1007/s10875-007-9080-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Oswald-Richter KA, Culver DA, Hawkins C, Hajizadeh R, Abraham S, Shepherd BE, et al. Cellular responses to mycobacterial antigens are present in sarcoidosis diagnostic bronchoalveolar lavage. Infect Immun. 2009;77(9):3740–8. doi: 10.1128/IAI.00142-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dubaniewicz A, Trzonkowski P, Dubaniewicz-Wybieralska M, Dubaniewicz A, Singh M, Mysliwski A. Mycobacterial heat shock protein-induced blood T lymphocytes subsets and cytokine pattern: comparison of sarcoidosis with tuberculosis and healthy controls. Respirology. 2007;12(3):346–54. doi: 10.1111/j.1440-1843.2007.01076.x. [DOI] [PubMed] [Google Scholar]
- 19.Ishige I, Usui Y, Takemura T, Eishi Y. Quantitative PCR of mycobacterial and propionibacterial DNA in lymph nodes of Japanese patients with sarcoidosis. Lancet. 1999;354(9173):120–3. doi: 10.1016/S0140-6736(98)12310-3. [DOI] [PubMed] [Google Scholar]
- 20.Abe C, Iwai K, Mikami R, Hosoda Y. Frequent isolation of Propionibacterium acnes from sarcoidosis lymph nodes. Zentralbl Bakteriol Mikrobiol Hyg A. 1984;256(4):541–7. doi: 10.1016/s0174-3031(84)80032-3. [DOI] [PubMed] [Google Scholar]
- 21.Eishi Y, Suga M, Ishige I, Kobayashi D, Yamada T, Takemura T, et al. Quantitative analysis of mycobacterial and propionibacterial DNA in lymph nodes of Japanese and European patients with sarcoidosis. J Clin Microbiol. 2002;40(1):198–204. doi: 10.1128/JCM.40.1.198-204.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yamada T, Eishi Y, Ikeda S, Ishige I, Suzuki T, Takemura T, et al. In situ localization of Propionibacterium acnes DNA in lymph nodes from sarcoidosis patients by signal amplification with catalysed reporter deposition. J Pathol. 2002;198(4):541–7. doi: 10.1002/path.1243. [DOI] [PubMed] [Google Scholar]
- 23.Ebe Y, Ikushima S, Yamaguchi T, Kohno K, Azuma A, Sato K, et al. Proliferative response of peripheral blood mononuclear cells and levels of antibody to recombinant protein from Propionibacterium acnes DNA expression library in Japanese patients with sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17(3):256–65. [PubMed] [Google Scholar]
- 24.Seeley EH, Caprioli RM. Molecular imaging of proteins in tissues by mass spectrometry. Proc Natl Acad Sci U S A. 2008;105(47):18126–31. doi: 10.1073/pnas.0801374105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pathan AA, Wilkinson KA, Klenerman P, McShane H, Davidson RN, Pasvol G, et al. Direct ex vivo analysis of antigen-specific IFN-gamma-secreting CD4 T cells in Mycobacterium tuberculosis-infected individuals: associations with clinical disease state and effect of treatment. J Immunol. 2001;167(9):5217–25. doi: 10.4049/jimmunol.167.9.5217. [DOI] [PubMed] [Google Scholar]
- 26.Oswald-Richter KA, Beachboard DC, Zhan X, Gaskill CF, Abraham S, Jenkins C, et al. Multiple mycobacterial antigens are targets of the adaptive immune response in pulmonary sarcoidosis. Respir Res. 2010;11:161. doi: 10.1186/1465-9921-11-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Breen RA, Hardy GA, Perrin FM, Lear S, Kinloch S, Smith CJ, et al. Rapid diagnosis of smear-negative tuberculosis using immunology and microbiology with induced sputum in HIV-infected and uninfected individuals. PLoS One. 2007;2(12):e1335. doi: 10.1371/journal.pone.0001335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fuhrmann S, Streitz M, Kern F. How flow cytometry is changing the study of TB immunology and clinical diagnosis. Cytometry A. 2008;73(11):1100–6. doi: 10.1002/cyto.a.20614. [DOI] [PubMed] [Google Scholar]
- 29.Jafari C, Ernst M, Kalsdorf B, Greinert U, Diel R, Kirsten D, et al. Rapid diagnosis of smear-negative tuberculosis by bronchoalveolar lavage enzyme-linked immunospot. Am J Respir Crit Care Med. 2006;174(9):1048–54. doi: 10.1164/rccm.200604-465OC. [DOI] [PubMed] [Google Scholar]
- 30.Cornett DS, Mobley JA, Dias EC, Andersson M, Arteaga CL, Sanders ME, et al. A novel histology-directed strategy for MALDIMS tissue profiling that improves throughput and cellular specificity in human breast cancer. Mol Cell Proteomics. 2006;5(10):1975–83. doi: 10.1074/mcp.M600119-MCP200. [DOI] [PubMed] [Google Scholar]
- 31.Hiramatsu J, Kataoka M, Nakata Y, Okazaki K, Tada S, Tanimoto M, et al. Propionibacterium acnes DNA detected in bronchoalveolar lavage cells from patients with sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2003;20(3):197–203. [PubMed] [Google Scholar]
- 32.Smith J, Manoranjan J, Pan M, Bohsali A, Xu J, Liu J, et al. Evidence for pore formation in host cell membranes by ESX-1-secreted ESAT-6 and its role in Mycobacterium marinum escape from the vacuole. Infect Immun. 2008;76(12):5478–87. doi: 10.1128/IAI.00614-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.van Ingen J, De Zwaan R, Dekhuijzen R, Boeree M, van Soolingen D. Region of difference 1 in nontuberculous Mycobacterium species adds a phylogenetic and taxonomical character. J Bacteriol. 2009;191(18):5865–7. doi: 10.1128/JB.00683-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Barksdale L, Kim KS. Propionibacterium, Corynebacterium, Mycobacterium and Lepra bacilli. Acta Leprol. 1984;2(2–4):153–74. [PubMed] [Google Scholar]
- 35.Sahiratmadja E, Alisjahbana B, de Boer T, Adnan I, Maya A, Danusantoso H, et al. Dynamic changes in pro- and anti-inflammatory cytokine profiles and gamma interferon receptor signaling integrity correlate with tuberculosis disease activity and response to curative treatment. Infect Immun. 2007;75(2):820–9. doi: 10.1128/IAI.00602-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Furukawa A, Uchida K, Ishige Y, Ishige I, Kobayashi I, Takemura T, et al. Characterization of Propionibacterium acnes isolates from sarcoid and non-sarcoid tissues with special reference to cell invasiveness, serotype, and trigger factor gene polymorphism. Microb Pathog. 2009;46(2):80–7. doi: 10.1016/j.micpath.2008.10.013. [DOI] [PubMed] [Google Scholar]
- 37.Atarashi R, Moore RA, Sim VL, Hughson AG, Dorward DW, Onwubiko HA, et al. Ultrasensitive detection of scrapie prion protein using seeded conversion of recombinant prion protein. Nat Methods. 2007;4(8):645–50. doi: 10.1038/nmeth1066. [DOI] [PubMed] [Google Scholar]
- 38.Chaurand P, Schwartz SA, Reyzer ML, Caprioli RM. Imaging mass spectrometry: principles and potentials. Toxicol Pathol. 2005;33(1):92–101. doi: 10.1080/01926230590881862. [DOI] [PubMed] [Google Scholar]
- 39.Volkman HE, Pozos TC, Zheng J, Davis JM, Rawls JF, Ramakrishnan L. Tuberculous granuloma induction via interaction of a bacterial secreted protein with host epithelium. Science. 2010;327(5964):466–9. doi: 10.1126/science.1179663. [DOI] [PMC free article] [PubMed] [Google Scholar]