Background: Cis-acting sequences regulate region-specific DNA demethylation.
Results: DNA demethylation of a fragment from the mouse H19/Igf2 imprinted control region requires two Sox-Oct motifs, one of which is methylation-sensitive.
Conclusion: The Sox-Oct motif functions to induce DNA demethylation in P19 cells.
Significance: The Sox-Oct motif might be a key element to induce region-specific DNA demethylation as well as activate transcription in undifferentiated cells.
Keywords: DNA Methylation, Epigenetics, Gene Regulation, Transcription Factors, Transfection, DNA Demethylation, Sox-Oct Motif, Cis-acting Element, Imprinted Gene
Abstract
DNA demethylation is used to establish and maintain an unmethylated state. The molecular mechanisms to induce DNA demethylation at a particular genomic locus remain unclear. The mouse H19/insulin-like growth factor 2 (Igf2) imprinted control region (ICR) is a methylation state-sensitive insulator that regulates transcriptional activation of both genes. The unmethylated state of the ICR established in female germ cells is maintained during development, resisting the wave of genome-wide de novo methylation. We previously demonstrated that a DNA fragment (fragment b) derived from this ICR-induced DNA demethylation when it was transfected into undifferentiated mouse embryonal carcinoma cell lines. Moreover, two octamer motifs within fragment b were necessary to induce this DNA demethylation. Here, we demonstrated that both octamer motifs and their flanking sequences constitute Sox-Oct motifs (SO1 and SO2) and that the SO1 region, which requires at least four additional elements, including the SO2 region, contributes significantly to the induction of high-frequency DNA demethylation as a Sox-Oct motif. Moreover, RNAi-mediated inhibition of Oct3/4 expression in P19 cells resulted in a reduced DNA demethylation frequency of fragment b but not of the adenine phosphoribosyltransferase gene CpG island. The Sox motif of SO1 could function as a sensor for a hypermethylated state of the ICR to repress demethylation activity. These results indicate that Sox-Oct motifs in the ICR determine the cell type, DNA region, and allele specificity of DNA demethylation. We propose a link between the mechanisms for maintenance of the unmethylated state of the H19/Igf2 ICR and the undifferentiated cell-specific induction of DNA demethylation.
Introduction
Allele-specific transcription of two imprinted genes, maternally expressed H19 and paternally expressed insulin-like growth factor 2 (Igf2),2 are associated with the methylation status of the H19 promoter and an imprinted control region (ICR) 2–4 kb upstream of the H19 gene. The unmethylated state of this ICR and the H19 promoter is established in germ cells and is maintained during development, circumventing the genome-wide de novo methylation that occurs after the implantation stage (1–3).
DNA demethylation is a part of the process that regulates methylation status and is used to establish (4–7) and maintain a hypomethylated state (8–12). To understand the mechanisms of maintaining the unmethylated state of the ICR, we previously screened for cis-acting sequences required to induce DNA demethylation in undifferentiated cells, and identified an ∼160-bp DNA fragment (fragment b) in the ICR that could induce DNA demethylation. Two octamer motifs with 12 bp of intervening sequence (dyad Oct-binding sequence) in fragment b were required for the efficient induction of DNA demethylation and maintenance of the unmethylated state within the ICR on a transgene. The dyad Oct-binding sequence-like structure is conserved with three copies in the human H19 gene (hD0–hD2) and a single copy in the rat H19 gene (10). A recent report showed that a single nucleotide mutation in one of the octamer motifs in hD1 is associated with a hypermethylated H19/IGF2 ICR in familial Beckwith-Wiedemann syndrome patients (13) and therefore indicated that the dyad Oct-binding sequence-containing motif possibly functions to maintain an unmethylated state by induction of DNA demethylation in the human H19/IGF2 ICR. The two octamer motifs are surrounded by sequences that are well conserved among mouse, rat, and human; however, the function of these sequences in the induction of demethylation remains unclear.
The Sox-Oct motif is known to regulate the transcription of several pluripotency-related genes expressed in ES cells and the early embryo (14–22). Although DNA demethylation occurs in some of these genes (Pou5f1, Nanog, Fbx15, Utf1, and Fgf4) during induced pluripotent stem cell induction (23, 24), it is unknown whether the Sox-Oct motif is involved in gene-specific DNA demethylation.
In this study, we have examined the DNA sequence in fragment b necessary to induce regional DNA demethylation using a transient transfection-based assay in the embryonal carcinoma cell line P19. We show here that each of the two octamer motifs in fragment b (SO1 and SO2) constitutes a Sox-Oct motif, defined by sequence similarity and their protein-binding properties, and that at least one (SO1) functions to induce DNA demethylation as a Sox-Oct motif. Furthermore, the Oct3/4 protein is required for DNA demethylation of fragment b. This is the first demonstration that a Sox-Oct motif contributes to the induction of DNA demethylation. We also found that SO1 is a methylation-sensitive element. The requirement for both a hypermethylation-sensitive Sox-Oct motif and the Oct3/4 protein for regional demethylation provides a novel insight into the maintenance mechanisms of the unmethylated state of the mouse H19/Igf2 ICR.
EXPERIMENTAL PROCEDURES
Plasmid Construction
To examine the DNA demethylation-inducing capabilities of the test fragments, each fragment was inserted into the HindIII site of the plasmid pEdBSl. To avoid an effect of the CpG-rich region of the bacterial plasmid on the methylation status of the test fragments, the non-CpG island (CGI) region of H19 was cloned into pBluescript II SK(−) and was used for the insertion site of the test fragments (pEdBSl) (10). A series of mutants of fragment b (bmA, bmB, bmC, bmD, and b-dCpG) was generated by ligation of a mutated HhaI-HpaII fragment made by annealing of synthetic oligonucleotides to the flanking fragments. The DNA fragments I+II, II+III, and II were HinfI-HpaII, HhaI-HinfI, and HhaI-HpaII fragments of fragment b, respectively. First, these fragments were cloned in the same orientation into the SmaI site of pBluescriptII SK(−). Then, the cloned fragments were excised with HindIII and BamHI for further plasmid construction. The DNA fragments SO1, SO1mBm1, and IIIa were synthesized and directly cloned with appropriate flanking fragments into pEdBSl. The construction of plasmids containing the other test fragments has been described elsewhere (10).
To knock down Oct3/4 and Sox2, we used an shRNA expression vector containing a puromycin-resistance gene: pBAsi-mU6 pur (TaKaRa, Shiga, Japan). Synthetic oligonucleotides to express the shRNA were annealed and cloned into the HindIII-BamHI site of pBAsi-mU6 pur. The sequence of the oligonucleotides used for green fluorescent protein (GFP) shRNA and Oct3/4 shRNA expression were described in Chew et al. (15). The sequences of the oligonucleotides used for Sox2 shRNA expression, 5′-GATCCCCGAAGGAGCACCCGGATTATATCAAGAGTATAATCCGGGTGCTCCTTCTTTTTA-3′ and 5′-AGCTTAAAAAGAAGGAGCACCCGGATTATACTCTTGATATAATCCGGGTGCTCCTTCGGG-3′, were also based on sequences described in this work except for the underlined nucleotides. The test fragment to examine DNA demethylation frequency was inserted into the blunt-ended NdeI sites of pBAsi-mU6 pur. The test fragment for fragment b was excised with NcoI and SpeI from pEdBSl-b. An EcoRV-XhoI DNA fragment of the adenine phosphoribosyltransferase (Aprt) CGI region was isolated from a mouse cosmid genomic library. The Oct3/4 and Sox2 cDNAs were cloned from P19 cells and F9 cells, respectively, and transferred to an expression plasmid (pCAGGSneo), which expresses its cDNA insert under the control of a chicken β-actin and rabbit β-globin hybrid promoter, with a human CMV immediate early enhancer.
Cell Culture and Transfection
The mouse embryonal carcinoma cell line P19 was maintained in α-modification of Eagle's medium supplemented with 5% calf serum, 5% FBS, and penicillin-streptomycin sulfate. HeLa cells were maintained in DMEM supplemented with 10% FBS and penicillin-streptomycin sulfate.
For the transient transfection assay of DNA demethylation frequency, P19 cells were plated (0.5 × 105 cells in a 3.5-cm dish) 24 h before transfection, transfected with 1.5 μg of in vitro-methylated plasmid DNA using TransIT® LT1 transfection reagent (Mirus Bio, Madison, WI), transferred to 10-cm dishes 24 h after transfection and used for total DNA preparation 72 h after transfection. To prepare the OCT3/4- or SOX2-overexpressing nuclear extracts, HeLa cells were transfected with 0.4 pmol of each expression plasmid or an empty plasmid and used for protein extraction 48 h after transfection. To assess the role of Oct3/4 and Sox2 in DNA demethylation, P19 cells were transfected with 1.7 μg of in vitro-methylated shRNA expression plasmid carrying the test fragment, selected for 22 h with 1 μg/ml puromycin from 24 h after transfection, and used for preparation of DNA or protein.
DNA Demethylation Assay
Constructs used in the experiments were methylated in vitro with bacterial HpaII methyltransferase (M.HpaII; New England Biolabs), HhaI methyltransferase (M.HhaI; New England Biolabs), and CpG methyltransferase (M.SssI; New England Biolabs). Prior to transfection, methylation was confirmed by digestion with 8–10 units of HpaII or HhaI restriction endonucleases per 1 μg of methylated plasmid. Plasmids that were completely methylated at the appropriate sites were used for transfection experiments. In vitro-methylated plasmids were transfected into P19 cells. Total DNA was harvested 46 h (for the knockdown assay) or 72 h after transfection. The methylation status of the transfected and untransfected plasmids was determined by Southern hybridization. To determine the frequency of in vitro methylation with M.HpaII or M.HhaI, 0.2 ng of untransfected plasmid was mixed with 5 μg of P19 DNA and used for Southern hybridization.
Southern Hybridization
Five or 0.85 μg (knockdown experiments) of total DNA harvested from each transfectant was digested with BamHI (TaKaRa) or HindIII (TaKaRa) followed by digestion with the methylation-sensitive restriction enzymes HpaII (Fermentas), HhaI (TaKaRa), or SmaI (TaKaRa). Digested DNAs were electrophoresed on 1.3% agarose gels, transferred to nylon membranes (Hybond-XLTM; GE Healthcare) and used for hybridization. Hybridization was performed as described elsewhere (10). Hybridized filters were subjected to autoradiography and quantified using a BAS2500 (FUJIFILM, Tokyo, Japan). The DNA demethylation frequency of a restriction site was calculated as follows: the radioactive count of a band generated by digestion at the restriction site was divided by the total radioactive count of all the bands. The count of fragment 3 by HpaII digestion or fragment 2 by HhaI digestion (see Fig. 1a) was used to evaluate the demethylation frequency. Results are shown as mean and S.D. for three independent experiments.
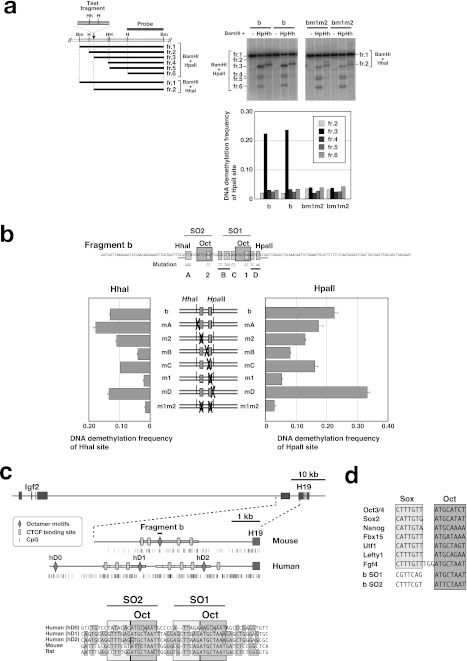
FIGURE 1.
Conserved sequences surrounding the two octamer motifs in fragment b are required to induce DNA demethylation. a, left: schematic representation of the restriction fragments observed in Southern hybridization analyses. Right upper panel: Southern hybridization analysis of fragment b and bm1m2. Bm, BamHI; H, HpaII; Hh, HhaI; fr., fragment. Right lower panel: DNA demethylation frequency of each HpaII site and HhaI site. The demethylation frequencies at the HhaI and HpaII sites were calculated as described under “Experimental Procedures.” b, DNA demethylation frequency at the HhaI or HpaII site within fragment b with or without mutation(s). The positions of the mutations are shown as light gray filled boxes on the nucleotide sequence of fragment b. Mutations B and D are substitutions of five and four nucleotides, respectively. The octamer motifs (Oct) and restriction endonuclease recognition sites (HhaI and HpaII) are indicated by dark gray filled boxes and underlines, respectively. Plasmids containing a test fragment were in vitro-methylated with M.HhaI and M.HpaII and transfected into P19 cells for 72 h. c, alignment of the octamer motifs and flanking sequences of the human, mouse, and rat H19/Igf2 ICR. The octamer motifs (dark gray filled boxes) are surrounded by well conserved sequences. Sox motif-like sequences are indicated by light gray filled boxes. d, the two octamer motifs and flanking sequences in fragment b are similar to Sox-Oct motifs located in the regulatory region of several pluripotency-related genes.
EMSA
Nuclear extracts were prepared as described by Schreiber et al. (25) from HeLa cells transfected with a mouse Oct3/4 expression vector, a mouse Sox2 expression vector, or empty vector and from P19 cells. Equal amounts of two nuclear extracts from the transfected HeLa cells were mixed and used as shown in Fig. 2. The binding reactions between the probe and 2 μg of nuclear extract were performed in 20.5 mm HEPES-KOH (pH 7.9), 2 mm Tris-HCl (pH 8.0), 40 mm KCl, 1.2 mm EDTA, 2 mm MgCl2, 1 mm dithiothreitol, 17% glycerol, 1 μg of poly(dG-dC), and 0.1 μl of protease inhibitor mixture (P8340; Sigma-Aldrich) at 4 °C for 60 min. Appropriate competitors at 300× molar excess or 2 μg of antibody were optionally added. Antibody was incubated with nuclear extract prior to the binding reaction at 4 °C for 60 min. Anti-Oct1 (sc-232x), anti-Oct4 (sc-8628x), and anti-Sox2 (sc-17320x) antibodies were purchased from Santa Cruz Biotechnology. The reaction mixtures were separated by 4% PAGE in 1× Tris-glycine-EDTA. The probe DNA fragments SO1, SO1m1, SO1mB, SO1m1mB, SO2, SO2mA, SO2mAm2, and Sox were generated by annealing the synthetic oligonucleotides SO1U (5′-ctTTGGGGCGTTCAGATGCTAATGATCTCCGGCTC-3′) and SO1L (5′-ctGAGCCGGAGATCATTAGCATCTGAACGCCCCAA-3′); SO1m1U (5′-ctTTGGGGCGTTCAGATGCTCCTGATCTCCGGCTC-3′) and SO1m1L (5′-ctGAGCCGGAGATCAGGAGCATCTGAACGCCCCAA-3′); SO1mBU (5′-ctTTGGTTCTGGCAGATGCTAATGATCTCCGGCTC-3′) and SO1mBL (5′-ctGAGCCGGAGATCATTAGCATCTGCCAGAACCAA-3′); SO1m1mBL (5′-ctTTGGTTCTGGCAGATGCTCCTGATCTCCGGCTC-3′) and SO1m1L (5′-ctGAGCCGGAGATCAGGAGCATCTGCCAGAACCAA-3′); SO2U (5′-TGATTTGCGCTTTCGTATTCTAATTGGGGC-3′) and SO2L (5′-CGCCCCAATTAGAATACGAAAGCGCAAATC-3′); SO2mAU (5′-TGATTTGCGCGGGCGTATTCTAATTGGGGC-3′) and SO2mAL (5′-CGCCCCAATTAGAATACGCCCGCGCAAATC-3′); SO2m2U (5′-TGATTTGCGCGGGCGTATTCTCCTTGGGGC-3′) and SO2m2L (5′-CGCCCCAAGGAGAATACGCCCGCGCAAATC-3′); SoxU (5′-AAAGATATCTTTGTTTCTTTC-3′) and SoxL (5′-TGAAAGAAACAAAGATATCTT-3′); SO1MeU (5′-ctTTGGGGMGTTCAGATGCTAATGATCTCCGGCTC-3′) and SO1MeL (5′-ctGAGCCGGAGATCATTAGCATCTGAAMGCCCCAA-3′); SO2MeU (5′-TGATTTGCGCTTTMGTATTCTAATTGGGGC-3′) and SO2MetL (5′-CGCCCCAATTAGAATAMGAAAGCGCAAATC-3′); SO1dCGU (5′-ctTTGGGGCTTTCAGATGCTAATGATCTCCGGCTC-3′) and SO1dCGL (5′-ctGAGCCGGAGATCATTAGCATCTGAAAGCCCCAA-3′); and SO2dCGU (5′-TGATTTGCGCTTTCATATTCTAATTGGGGC-3′) and SO2dCGL (5′-CGCCCCAATTAGAATATGAAAGCGCAAATC-3′), respectively. In the sequences (SO1MeU, SO1MeL, SO2MeU, and SO1MeL), M indicates 5-methylcytosine. The lowercase letters signify additional bases non-complementary to the genome.
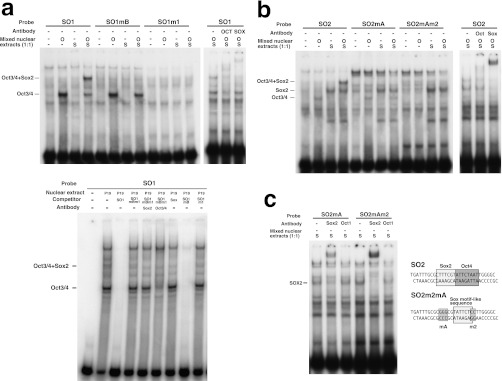
FIGURE 2.
Mutations that suppress DNA demethylation inhibit the binding of Sox2 and Oct3/4. a, upper panel: EMSA of the SO1 probe with or without a mutation. Nuclear extracts from HeLa cells transfected with an empty vector, an Oct3/4 expression plasmid (O), or a Sox2 expression plasmid (S) were mixed at a 1:1 ratio and used for binding reactions with the indicated probe. An anti-Oct3/4 (Oct) antibody or an anti-Sox2 (Sox) antibody was used for the supershift assay. Lower panel: EMSA of the SO1 probe with nuclear extract from P19 cells. The binding of Oct1 has been previously reported (10). b, binding of Oct3/4 and Sox2 to the SO2 probe with or without a mutation. c, Sox2 can bind to SO2 with mutations mA and m2. Anti-Sox2 (Sox2) or anti-Oct1 (Oct1) antibodies were used for the supershift assay.
Western Blotting
Whole-cell extracts were prepared from P19 cells with 0.1% SDS-radioimmune precipitation assay buffer. Twenty micrograms of total protein samples were electrophoresed on a 10% SDS-polyacrylamide gel and transferred onto a PVDF membrane (Immobilon; Millipore). The membrane was probed with goat anti-Oct4 antibody (1:5000; sc-8628X; Santa Cruz Biotechnology), goat anti-Sox2 antibody (1:5000; sc-17320X; Santa Cruz Biotechnology), or mouse anti-α-tubulin antibody (1:5000; T9026; Sigma) followed by an appropriate secondary antibody: horseradish peroxidase-conjugated donkey anti-goat (V8051; Promega), anti-rabbit (W401B; Promega), or anti-mouse (W402B; Promega). The signal was detected using ECL detection reagents (GE Healthcare). The intensity of the bands was calculated using ImageGauge (Fujifilm).
RESULTS
Octamer Motif-dependent DNA Demethylation Occurs on Fragment b in a Transiently Transfected Plasmid
In our previous study, to identify the cis-acting sequence that induces DNA demethylation, we transfected in vitro-methylated plasmids with various DNA fragments into P19 cells and examined the DNA methylation status of the stably integrated transgene. Because this stable transfection-based assay failed to reveal subtle differences in the demethylation-inducing activity of various DNA fragments, in this study, we examined the DNA methylation status of transiently transfected plasmids 72 h after transfection. This transient transfection assay system was successful in assessing the demethylation-inducing activity of the test fragments. Unlike demethylation of stably transfected transgenes, sequence-specific high-frequency demethylation of transiently transfected plasmids occurred only at the HpaII and HhaI sites proximal to the octamer motif (Fig. 1a). Therefore, we used the methylation status of these restriction sites to evaluate the DNA demethylation-inducing activity of the test fragments.
Induction of High-frequency DNA Demethylation Requires Sequences Flanking the Octamer Motif in Fragment b
To clarify the role of the conserved sequences within fragment b in DNA demethylation, we examined the DNA demethylation-inducing activity of a series comprising fragment b containing various mutations in the conserved sequence (mA to mD) or in either octamer motif (m1, m2).
Mutation at site B (mB) significantly repressed the demethylation frequency at both the HpaII and HhaI sites to almost the same levels as that conferred by mutation m1 (Fig. 1b and supplemental Fig. S1). This suggested that site B and site 1 function as a unit to induce high-frequency DNA demethylation. Because all the mutations except mutation mD reduced the demethylation frequency to various degrees, we concluded that conserved sequences at sites A, B, and C, as well as the two octamer motifs (sites 1 and 2) are required to induce maximum demethylation.
Mutations That Suppress DNA Demethylation Inhibit the Interaction of Sox2 and Oct3/4 with the Sox-Oct Motifs
An octamer motif and a Sox family protein-binding site (Sox motif) adjacent to the octamer motif, collectively called the Sox-Oct motif, are known to cooperatively regulate the transcription of several pluripotency-related genes. We identified sequence similarity between the Sox motif and the sequences flanking the octamer motifs in fragment b (Fig. 1, c and d) and hypothesized that this region could function as a Sox-Oct motif. To test this, we examined whether the two DNA fragments, identified by the SO1 and SO2 probes, respectively, which each contain an octamer motif and its flanking sequences, could bind to Sox2. We also tested whether the mutations that inhibit demethylation affected the binding of Sox2 (Fig. 2 and supplemental Fig. S2).
EMSAs on nuclear extracts from HeLa cells that overexpressed Oct3/4 or Sox2 revealed that Sox2 bound to both the SO1 probe and the SO2 probe and formed a ternary complex with Oct3/4 and each DNA fragment. The formation of the ternary complex was confirmed by a supershift assay with anti-Oct3/4 antibody or anti-Sox2 antibody.
As expected, mutations m1, mB, m2, and mA effectively suppressed complex formation between the predicted binding sites constituting the Sox-Oct motifs and the binding proteins. Mutation m1 in the SO1 octamer motif markedly suppressed the binding of Oct3/4, as reported previously (10). Mutation mB, which affects a putative Sox motif and a flanking two-nucleotide sequence, inhibited formation of the ternary complex but not Oct3/4 binding (Fig. 2a). Mutation mA in the putative SO2 Sox motif also inhibited the formation of the ternary complex but not Oct3/4 binding, indicating that mA suppressed the binding of Sox2 to the Sox motif. Oct3/4 binding to SO2 was suppressed by the combination of mA and m2, but not by mA alone (Fig. 2b).
Sox2 could not bind the SO1 probe by itself and required Oct3/4 for binding (Fig. 2a; SO1 probe). This was consistent with the observation that mutation of the SO1 octamer motif disrupted the binding of both Oct3/4 and Sox2 (Fig. 2a; SO1m1 probe). In contrast, mutation mB inhibited the binding of Sox2 but not Oct3/4 to the SO1 fragment. These binding properties of Oct3/4 and Sox2 were confirmed using nuclear extracts from P19 cells (Fig. 2a, lower panel). Oct3/4 and Sox2 expressed in P19 cells formed a ternary complex with the SO1 probe. When the SO1mB DNA fragment (SO1 without the Sox motif) was used as a competitor, all of the bands disappeared. In contrast, an SO1m1 competitor (SO1 without the octamer motif) failed to disrupt formation of the ternary complex. Thus, the sequence similarity and the binding of OCT3/4 and Sox2 to the SO1 probe indicate that the SO1 region can function as a Sox-Oct motif to induce DNA demethylation.
The patterns of protein binding to the SO2 region were complicated (Fig. 2b). Sox2 could bind alone to the SO2 probe independently of OCT3/4. Although Oct3/4, Sox2, and the SO2 probe formed a ternary complex, a significant amount of the binary complex of Sox2 and the SO2 probe remained (Fig. 2b, SO2 probe). Mutation mA suppressed the binding of SOX2 to the Sox-Oct motif; however, Sox2 still bound to the SO2 probe even in the presence of m2 and mA (SO2mAm2). This suggested that there is another Sox motif independent of the Sox-Oct motif in the SO2 region, and consistently, we found a well conserved Sox motif-like sequence within the Sox-Oct motif (Fig. 2c). The position of the second Sox motif is consistent with our observation that Oct3/4, Sox2, and the SO2 probe form either a ternary complex or a binary complex of Sox2 and the SO2 probe in a mutually exclusive manner. We concluded that the SO2 region can function as a Sox-Oct motif or a single Sox family protein-binding site.
High-frequency DNA Demethylation Requires the Entire Fragment b Region
To identify the DNA region sufficient to induce high-frequency DNA demethylation, we tested the DNA demethylation frequency at the HpaII site on various DNA fragments derived from fragment b (Fig. 3 and supplemental Fig. S3).
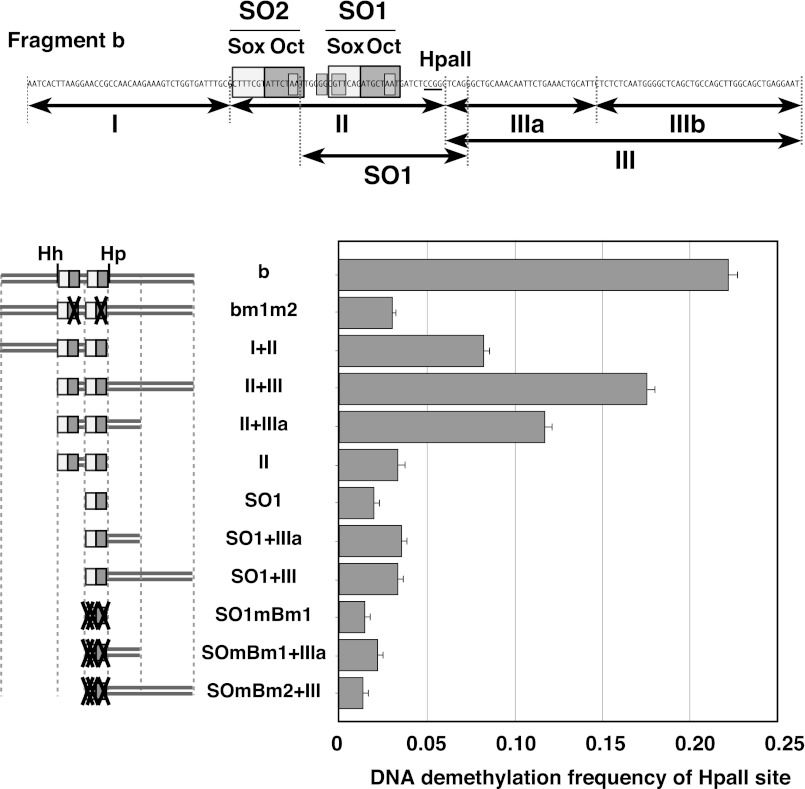
FIGURE 3.
Multiple elements are required to induce DNA demethylation. Upper panel: the positions of the fragments and mutations used are shown on the nucleotide sequence of fragment b. Lower panel: DNA demethylation frequency of the deletion mutants. The test DNAs were in vitro-methylated by M.HpaII, and transfected into P19 cells for 72 h. The demethylation frequency at the HpaII sites was determined as described under “Experimental Procedures.”
The region II DNA fragment, which contains both Sox-Oct motifs, induced very low-frequency demethylation. To induce high-frequency demethylation, region II required both region I and region III (Fig. 3). Deletion of subregion IIIb from fragment II+III resulted in modest decreases in the demethylation frequency compared with that of fragment II+III. Furthermore, neither region I nor III alone was sufficient to induce high-frequency demethylation because bm1m2 or SO1+III resulted in very low-frequency demethylation. Thus, the cis-acting elements required to induce high-frequency demethylation are located in multiple (at least four) portions of fragment b and additively enhance the demethylation frequency.
Oct3/4 Is Required for Sox-Oct Motif-dependent Induction of DNA Demethylation
We have shown that the Sox-Oct motif, especially the SO1 region, is involved in the induction of high-frequency regional DNA demethylation in P19 cells. Chromatin immunoprecipitation analysis of ES cells revealed that the Oct3/4-binding consensus sequence is a Sox-Oct motif identical to the Sox2-binding consensus sequence and that 74.3% of OCT3/4- and 60.3% of Sox2-binding sequences are clustered in the mouse genome (26). P19 is an undifferentiated pluripotent cell line that expresses Oct3/4 and Sox2. To assess the possibility that Sox2 and Oct3/4 are involved in the induction of DNA demethylation through interacting with the Sox-Oct motifs in fragment b, we suppressed the expression of these proteins in P19 cells by RNAi with shRNAs and evaluated the DNA demethylation frequency on fragment b or a fragment from the mouse Aprt gene CGI. The Aprt gene CGI is reported to undergo Sp1 binding site-dependent DNA demethylation in embryonal cells (9). Because the Sox-Oct motif is not reported to play a role in the transcriptional activation of Aprt and DNA demethylation of its CGI, we used the Aprt CGI as a control to determine whether Oct3/4 and Sox2 are required to induce region-specific DNA demethylation. Each DNA fragment was cloned into an shRNA expression plasmid. The plasmids were in vitro-methylated with M.HpaII and transfected into P19 cells. We confirmed that the methylated shRNA expression plasmids could suppress expression of the target proteins in puromycin-resistant cells (Fig. 4a).
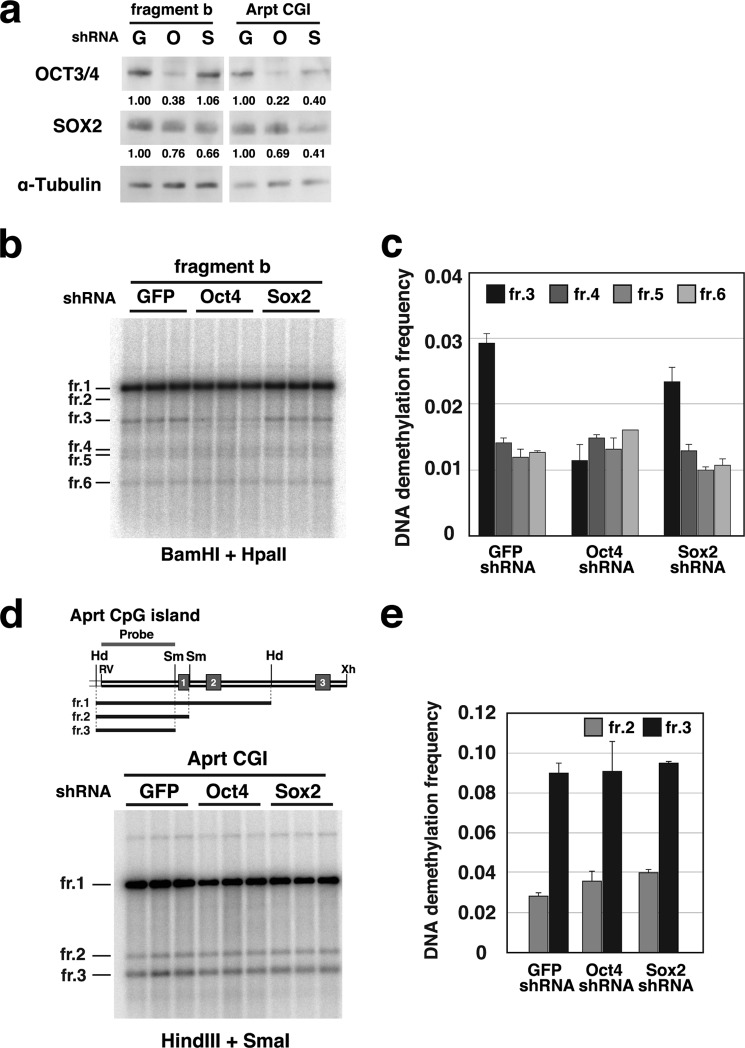
FIGURE 4.
Oct3/4 is involved in DNA demethylation of fragment b but not of the Aprt CGI. a, Western blot analysis of Oct3/4, Sox2, and α-tubulin expression. shRNA expression plasmids were in vitro-methylated with M.HpaII and transfected for 46 h. Proteins were extracted from P19 cells transfected with GFP shRNA (G), Oct3/4 shRNA (O), or Sox2 shRNA (S) expression plasmids with a test fragment (fr.; fragment b or the Aprt CGI). The indicated values for Oct3/4 and Sox2 were normalized to the value for α-tubulin and are shown relative to the values in cells transfected with the GFP shRNA expression plasmid. b, Southern hybridization analysis to estimate the DNA demethylation frequency of the transfected plasmids containing fragment b. DNA was digested with BamHI and HpaII and probed with a HpaII-BamHI fragment as indicated in Fig. 1a. c, DNA demethylation frequency at the HpaII sites on the plasmids containing fragment b. The demethylation frequency was determined as described in under “Experimental Procedures.” d, Southern hybridization analysis to evaluate the DNA demethylation frequency in the transfected Aprt CGI. DNA was digested with HindIII and SmaI and probed with a HindIII-SmaI fragment. e, DNA demethylation frequency at the HpaII sites in the mouse Aprt CGI fragment.
Expression of the Oct3/4 shRNA for 72 h resulted in a significantly reduced amount of harvested genomic DNA (on average, 28.7% of GFP shRNA) because of a decrease in cell number. Therefore, in these experiments, we examined the demethylation frequency 46 h after transfection. At this time point, there was no difference in the amount of DNA from puromycin-resistant cells transfected with GFP shRNA, Oct3/4 shRNA, or Sox2 shRNA (1.00:1.02:1.00, respectively). Sox2 suppression by shRNA resulted in no significant difference in DNA demethylation frequency of either fragment b or the Aprt CGI (Fig. 4). In contrast, Oct3/4 shRNA expression significantly reduced the DNA demethylation frequency of only the SO1-proximal HpaII site in fragment b but not of the other HpaII sites examined or the two SmaI sites in the Aprt CGI fragment. In this assay, the SO1-proximal HpaII site is the only HpaII site at which Sox-Oct motif-dependent demethylation occurred. Our data indicated that Oct3/4 is indispensable in inducing Sox-Oct motif-dependent DNA demethylation on fragment b.
A CpG Sequence in the Sox Motif of SO1 Functions as a Sensor of the Hypermethylated State
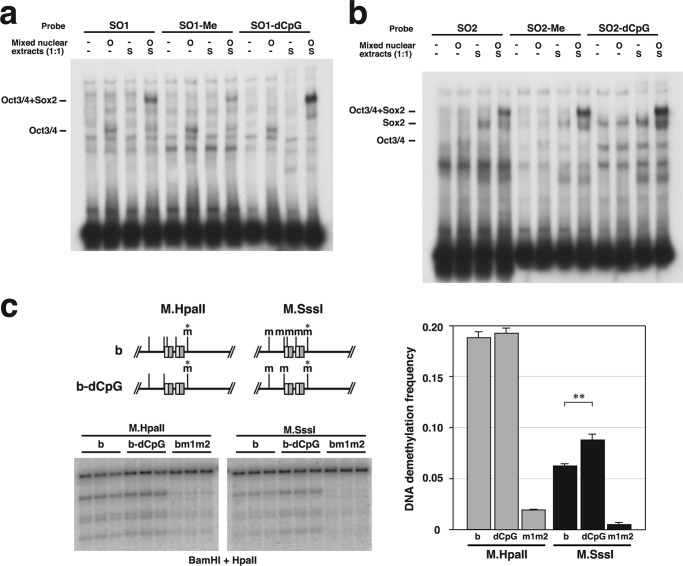
In previous work, we demonstrated by transfection assays that the DNA demethylation-inducing activity of fragment b was required to maintain an unmethylated state within the ICR in P19 cells. In contrast, when DNA fragments methylated with SssI methylase, which methylates all CpG sequences, were introduced into the genome of P19 cells, a highly methylated state was maintained (10). This result indicated that the octamer motif-dependent DNA demethylation-inducing activity of fragment b is hypermethylation sensitive and can maintain the unmethylated state of the ICR without affecting the hypermethylated state of the paternal allele. (The fully methylated endogenous ICR inherited from sperm is maintained during mouse development.) However, we could not determine the molecular mechanisms by which methylation of all of the CpGs suppressed the octamer motif-dependent induction of DNA demethylation. Because we revealed that each Sox-Oct motif in fragment b contains a CpG sequence (Fig. 1d), we tested the possibility that the CpG sequence in the Sox motifs of fragment b functions as a sensor of the hypermethylated state by regulating ternary complex formation and controls DNA demethylation-inducing activity.
EMSA with synthetic probes methylated at the CpG in the Sox motifs of SO1 or SO2 (supplemental Fig. S2) revealed that methylation of the CpG in SO1 but not in SO2, significantly suppressed formation of the ternary complex (Fig. 5, a and b). To determine the effect of methylation in the Sox motif on DNA demethylation-inducing activity, we generated fragment b with methylation-insensitive Sox-Oct motifs (b-dCpG), in which the CpG was substituted by CpT/ApG at SO1 and CpA/TpG at SO2. This sequence substitution of EMSA probes resulted in no reduced formation of the ternary complexes (Fig. 5, a and b). Fragment b and fragment b-dCpG in plasmids methylated with HpaII methylase equally induced DNA demethylation within the fragments (M.HpaII-treated b and b-dCpG); however, methylation at CpG in the Sox motifs significantly reduced the DNA demethylation frequency of the HpaII site within the fragments (Fig. 5c, M.SssI-treated b and b-dCpG). These results suggested that the CpG sequence in the Sox motif of fragment b functions as a sensor of the methylated state to control the frequency of DNA demethylation induction.
FIGURE 5.
The CpG sequence in the Sox motif of SO1 functions as a sensor of the methylated state to control the frequency of DNA demethylation induction. a, binding of Oct3/4 and Sox2 to the SO2 probe, SO1 with methylation (SO1-Me), and SO1 without a CpG sequence (SO1-dCpG). Nuclear extracts from HeLa cells transfected with an empty vector, an Oct3/4 expression plasmid (O), or a Sox2 expression plasmid (S) were mixed at a 1:1 ratio and used for binding reactions with the indicated probe. b, EMSA of the SO2 probe, SO2 with methylation (SO2-Me), and SO2 without a CpG sequence (SO2-dCpG). c, DNA demethylation frequency at the HpaII site (asterisk in the left upper panel) in fragment b (b), fragment b without the CpGs of the two Sox motifs (b-dCpG), or bm1m2 (bm1m2) on the plasmids methylated with HpaII methyltransferase (M.HpaII) or CpG methyltransferase (M.SssI). **, p < 0.01.
DISCUSSION
We have demonstrated that both octamer motifs in fragment b are flanked by a Sox2-binding site. As shown in Fig. 1, the demethylation frequency of fragment b with mutation mB (bmB) reduced demethylation to almost the same level as that of fragment b with mutation m1 (bm1). Either mutation mB or m1 disrupted ternary complex formation between Oct3/4, Sox2, and the SO1 probe DNA (Fig. 2). These results suggest that the Sox-Oct motif in the SO1 region is required to induce high-frequency demethylation. This is the first direct evidence that the Sox-Oct motif can be involved in induction of regional DNA demethylation.
The other Sox-Oct motif, SO2, was also indispensable for the induction of high-frequency DNA demethylation (Fig. 3; fragments II+III and SO1+III). However, it is unclear whether the Sox-Oct motif in the SO2 region functions as a Sox-Oct motif to induce DNA demethylation. Mutation mA resulted in an increased DNA demethylation frequency at the HhaI site only, probably because the interaction of a protein with site A would mask the methylated CpG at the HhaI site; conversely, this implies that when the HhaI site is demethylated, protein binding to site A must be excluded. This result indicates that binding of a Sox family protein to the Sox-Oct motif of the SO2 region is not necessary for demethylation at the HhaI site. The contribution of the second Sox2-binding site in the SO2 region to the demethylation at the HhaI site remains to be determined. In contrast, because mutations mA and m2 decreased the DNA demethylation frequency at the HpaII site, the SO2 region must require the Sox-Oct motif to induce maximal DNA demethylation. We conclude that the SO2 region functions as a Sox-Oct motif or as either a single Sox family protein-binding region or a single Oct3/4 protein-binding region in a mutually exclusive manner.
Mutation of the octamer motif that is homologous to the SO2 portion of fragment b has been found in Beckwith-Wiedemann syndrome patients (13). As shown in Fig. 1, the contribution of SO2 to the induction of DNA demethylation is smaller than that of SO1. The human SO2 homologous sequences, however, can be expected to show different demethylation-induction activities because the sequence of the octamer motif of the human SO2 homologous sequence is more similar to the octamer motif consensus sequence than is the mouse SO2. Interestingly, because the flanking sequences of the human octamer motifs are homologous to the Sox motif, the octamer motifs in the human genome may function as Sox-Oct motifs to induce DNA demethylation of the ICR.
Cis-acting sequences known to be involved in DNA demethylation and maintenance of the unmethylated state of the H19/Igf2 ICR are the Sox-Oct motif (in this work) and the CCCTC-binding factor binding site (11), but both of these elements are insufficient to understand the molecular mechanisms by which the mutations and deletions found in Beckwith-Wiedemann syndrome patients result in a hypermethylated ICR. To understand this, systematic screening of cis-acting elements is required. In this report, we demonstrated that our transient transfection-based assay system could be used to determine the core sequence sufficient to induce DNA demethylation. This simple and sensitive assay system should be widely applicable to screening for cis-acting sequences that induce DNA demethylation.
Repression of Sox2 expression for 46 h had weak and no effect on the DNA demethylation frequency of fragment b and the Aprt CGI, respectively, although the Sox2-binding site in the Sox-Oct motifs of SO1 was required to induce high-frequency demethylation. In ES cells, Sox-Oct motifs are reported to activate transcription of several pluripotency-related genes through interaction with Sox2, Sox4, Sox11, Sox15, and Oct3/4 (27). These Sox family proteins are thought to complement the effects of Sox2 depletion on transcriptional activation. Because several papers have reported that Sox2, Sox3, and Sox15 are expressed in P19 cells (28–30), it is possible that the effect of Sox2 depletion on DNA demethylation was also rescued by these other Sox family proteins through interaction with the Sox-Oct motifs.
In contrast, suppression of Oct3/4 expression by shRNA resulted in a significant reduction in demethylation on fragment b. We propose a model in which Oct3/4 and Sox family proteins recruit a DNA demethylation-related protein complex to fragment b through interaction with the Sox-Oct motifs (supplemental Fig. S4a). However, because the proteins involved in DNA demethylation of fragment b and the Aprt CGI remain unclear, we cannot exclude an alternative model in which Oct3/4 up-regulates proteins that function in DNA demethylation of fragment b but not of the Aprt CGI (supplemental Fig. S4b). These two models indicate that undifferentiated cell-specific DNA demethylation might function to maintain the unmethylated state of the mouse H19/Igf2 ICR in embryos.
We have shown that the Sox-Oct motif functions as a cis-acting sequence to induce DNA demethylation in the undifferentiated pluripotent embryonal carcinoma cell line P19 and that Sox-Oct motif-dependent DNA demethylation requires the Oct3/4 protein. Interestingly, our findings indicated that the presence of both Sox-Oct motifs is not essential to induce high-frequency DNA demethylation, because mutation mA, which disrupts only one Sox-Oct motif, resulted in a small decrease in DNA demethylation (76.9%) at the HpaII site (Figs. 1 and 2). A single Sox-Oct motif is involved in the transcriptional activation of several pluripotency-related genes in undifferentiated pluripotent cells (14–22). Although the relationship between transcriptional activation and the induction of DNA demethylation is not clear, our findings raise the possibility that, in undifferentiated pluripotent cells, the Sox-Oct motif associated with pluripotency-related genes is used to maintain the unmethylated state of functional DNA elements by DNA demethylation. It would be of interest to determine whether the Sox-Oct motif-containing DNA fragments associated with pluripotency-related genes can induce DNA demethylation.
Each of the two Sox-Oct motifs in the mouse H19/Igf2 ICR contains a CpG sequence. We demonstrated that one of the two Sox-Oct motifs, SO1, functions as a methylation-sensitive binding motif and that methylation of the CpG sequence repressed the DNA demethylation-inducing activity of fragment b. Suppression of DNA demethylation-inducing activity by the hypermethylated state is an important feature of cis-acting elements to maintain an unmethylated state in the maternal ICR without affecting the hypermethylated state in the paternal ICR. Our results indicated that the SO1 in the ICR could function as a methylation state-sensing element to discriminate the chromosomal parental origin (supplemental Fig. S5). Therefore, the SO1 might be a target for mutations that cause either aberrant hypermethylation of the maternal ICR or hypomethylation of the paternal ICR.
Acknowledgment
We thank Tomoko Nakanishi for helpful discussions.

This article contains supplemental Figs. S1–S5.
- IGF2
- insulin-like growth factor 2
- ICR
- imprinted control region
- CGI
- CpG island
- APRT
- adenine phosphoribosyl transferase
- M.HpaII
- HpaII methyltransferase
- M.HhaI
- HhaI methyltransferase
- M.SssI
- CpG methyltransferase.
REFERENCES
- 1. Tremblay K. D., Saam J. R., Ingram R. S., Tilghman S. M., Bartolomei M. S. (1995) A paternal-specific methylation imprint marks the alleles of the mouse H19 gene. Nat. Genet. 9, 407–413 [DOI] [PubMed] [Google Scholar]
- 2. Tremblay K. D., Duran K. L., Bartolomei M. S. (1997) A 5′ 2-kilobase-pair region of the imprinted mouse H19 gene exhibits exclusive paternal methylation throughout development. Mol. Cell Biol. 17, 4322–4329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ueda T., Abe K., Miura A., Yuzuriha M., Zubair M., Noguchi M., Niwa K., Kawase Y., Kono T., Matsuda Y., Fujimoto H., Shibata H., Hayashizaki Y., Sasaki H. (2000) The paternal methylation imprint of the mouse H19 locus is acquired in the gonocyte stage during foetal testis development. Genes Cells 5, 649–659 [DOI] [PubMed] [Google Scholar]
- 4. Kirillov A., Kistler B., Mostoslavsky R., Cedar H., Wirth T., Bergman Y. (1996) A role for nuclear NF-κB in B-cell-specific demethylation of the Igκ locus. Nat. Genet. 13, 435–441 [DOI] [PubMed] [Google Scholar]
- 5. Bruniquel D., Schwartz R. H. (2003) Selective, stable demethylation of the interleukin-2 gene enhances transcription by an active process. Nat. Immunol. 4, 235–240 [DOI] [PubMed] [Google Scholar]
- 6. Métivier R., Gallais R., Tiffoche C., Le Péron C., Jurkowska R. Z., Carmouche R. P., Ibberson D., Barath P., Demay F., Reid G., Benes V., Jeltsch A., Gannon F., Salbert G. (2008) Cyclical DNA methylation of a transcriptionally active promoter. Nature 452, 45–50 [DOI] [PubMed] [Google Scholar]
- 7. Bhutani N., Brady J. J., Damian M., Sacco A., Corbel S. Y., Blau H. M. (2010) Reprogramming towards pluripotency requires AID-dependent DNA demethylation. Nature 463, 1042–1047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Brandeis M., Frank D., Keshet I., Siegfried Z., Mendelsohn M., Nemes A., Temper V., Razin A., Cedar H. (1994) Sp1 elements protect a CpG island from de novo methylation. Nature 371, 435–438 [DOI] [PubMed] [Google Scholar]
- 9. Macleod D., Charlton J., Mullins J., Bird A. P. (1994) Sp1 sites in the mouse aprt gene promoter are required to prevent methylation of the CpG island. Genes Dev. 8, 2282–2292 [DOI] [PubMed] [Google Scholar]
- 10. Hori N., Nakano H., Takeuchi T., Kato H., Hamaguchi S., Oshimura M., Sato K. (2002) A dyad oct-binding sequence functions as a maintenance sequence for the unmethylated state within the H19/Igf2-imprinted control region. J. Biol. Chem. 277, 27960–27967 [DOI] [PubMed] [Google Scholar]
- 11. Rand E., Ben-Porath I., Keshet I., Cedar H. (2004) CTCF elements direct allele-specific undermethylation at the imprinted H19 locus. Curr. Biol. 14, 1007–1012 [DOI] [PubMed] [Google Scholar]
- 12. Schmitz K. M., Schmitt N., Hoffmann-Rohrer U., Schäfer A., Grummt I., Mayer C. (2009) TAF12 recruits Gadd45a and the nucleotide excision repair complex to the promoter of rRNA genes leading to active DNA demethylation. Mol. Cell 33, 344–353 [DOI] [PubMed] [Google Scholar]
- 13. Demars J., Shmela M. E., Rossignol S., Okabe J., Netchine I., Azzi S., Cabrol S., Le Caignec C., David A., Le Bouc Y., El-Osta A., Gicquel C. (2010) Analysis of the IGF2/H19 imprinting control region uncovers new genetic defects, including mutations of OCT-binding sequences, in patients with 11p15 fetal growth disorders. Hum. Mol. Genet. 19, 803–814 [DOI] [PubMed] [Google Scholar]
- 14. Okumura-Nakanishi S., Saito M., Niwa H., Ishikawa F. (2005) Oct-3/4 and Sox2 regulate Oct-3/4 gene in embryonic stem cells. J. Biol. Chem. 280, 5307–5317 [DOI] [PubMed] [Google Scholar]
- 15. Chew J. L., Loh Y. H., Zhang W., Chen X., Tam W. L., Yeap L. S., Li P., Ang Y. S., Lim B., Robson P., Ng H. H. (2005) Reciprocal transcriptional regulation of Pou5f1 and Sox2 via the Oct4/Sox2 complex in embryonic stem cells. Mol. Cell Biol. 25, 6031–6046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Tomioka M., Nishimoto M., Miyagi S., Katayanagi T., Fukui N., Niwa H., Muramatsu M., Okuda A. (2002) Identification of Sox-2 regulatory region which is under the control of Oct-3/4-Sox-2 complex. Nucleic Acids Res. 30, 3202–3213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kuroda T., Tada M., Kubota H., Kimura H., Hatano S. Y., Suemori H., Nakatsuji N., Tada T. (2005) Octamer and Sox elements are required for transcriptional cis regulation of Nanog gene expression. Mol. Cell Biol. 25, 2475–2485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Rodda D. J., Chew J. L., Lim L. H., Loh Y. H., Wang B., Ng H. H., Robson P. (2005) Transcriptional regulation of nanog by OCT4 and SOX2. J. Biol. Chem. 280, 24731–24737 [DOI] [PubMed] [Google Scholar]
- 19. Tokuzawa Y., Kaiho E., Maruyama M., Takahashi K., Mitsui K., Maeda M., Niwa H., Yamanaka S. (2003) Fbx15 is a novel target of Oct3/4 but is dispensable for embryonic stem cell self-renewal and mouse development. Mol. Cell Biol. 23, 2699–2708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Nishimoto M., Fukushima A., Okuda A., Muramatsu M. (1999) The gene for the embryonic stem cell coactivator UTF1 carries a regulatory element which selectively interacts with a complex composed of Oct-3/4 and Sox-2. Mol. Cell Biol. 19, 5453–5465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Nakatake Y., Fukui N., Iwamatsu Y., Masui S., Takahashi K., Yagi R., Yagi K., Miyazaki J., Matoba R., Ko M. S., Niwa H. (2006) Klf4 cooperates with Oct3/4 and Sox2 to activate the Lefty1 core promoter in embryonic stem cells. Mol. Cell Biol. 26, 7772–7782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Botquin V., Hess H., Fuhrmann G., Anastassiadis C., Gross M. K., Vriend G., Schöler H. R. (1998) New POU dimer configuration mediates antagonistic control of an osteopontin preimplantation enhancer by Oct-4 and Sox-2. Genes Dev. 12, 2073–2090 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Takahashi K., Yamanaka S. (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 [DOI] [PubMed] [Google Scholar]
- 24. Mikkelsen T. S., Hanna J., Zhang X., Ku M., Wernig M., Schorderet P., Bernstein B. E., Jaenisch R., Lander E. S., Meissner A. (2008) Dissecting direct reprogramming through integrative genomic analysis. Nature 454, 49–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Schreiber E., Matthias P., Müller M. M., Schaffner W. (1989) Rapid detection of octamer binding proteins with “mini-extracts,” prepared from a small number of cells. Nucleic Acids Res. 17, 6419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chen X., Xu H., Yuan P., Fang F., Huss M., Vega V. B., Wong E., Orlov Y. L., Zhang W., Jiang J., Loh Y. H., Yeo H. C., Yeo Z. X., Narang V., Govindarajan K. R., Leong B., Shahab A., Ruan Y., Bourque G., Sung W. K., Clarke N. D., Wei C. L., Ng H. H. (2008) Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell 133, 1106–1117 [DOI] [PubMed] [Google Scholar]
- 27. Masui S., Nakatake Y., Toyooka Y., Shimosato D., Yagi R., Takahashi K., Okochi H., Okuda A., Matoba R., Sharov A. A., Ko M. S., Niwa H. (2007) Pluripotency governed by Sox2 via regulation of Oct3/4 expression in mouse embryonic stem cells. Nat. Cell Biol. 9, 625–635 [DOI] [PubMed] [Google Scholar]
- 28. Xia C., Wang C., Zhang K., Qian C., Jing N. (2007) Induction of a high population of neural stem cells with anterior neuroectoderm characters from epiblast-like P19 embryonic carcinoma cells. Differentiation 75, 912–927 [DOI] [PubMed] [Google Scholar]
- 29. Jin Z., Liu L., Bian W., Chen Y., Xu G., Cheng L., Jing N. (2009) Different transcription factors regulate nestin gene expression during P19 cell neural differentiation and central nervous system development. J. Biol. Chem. 284, 8160–8173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Savage J., Conley A. J., Blais A., Skerjanc I. S. (2009) SOX15 and SOX7 differentially regulate the myogenic program in P19 cells. Stem Cells 27, 1231–1243 [DOI] [PubMed] [Google Scholar]