Abstract
We designed a recyclable Hg2+ probe based on Rhodamine B isothiocyanate (RBITC) - poly (ethylene glycol) (PEG)-co-modified gold nanoparticles (AuNPs) with excellent robustness, selectivity and sensitivity. Based on a rational design, only Hg2+ can displace RBITC from the AuNP surfaces, resulting in a remarkable enhancement of RBITC fluorescence initially quenched by AuNPs. To maintain stability and monodispersity of AuNPs in real samples, thiol-terminated PEG was employed to bind with the remaining active sites of AuNPs. Besides, this displacement assay can be regenerated by resupplying free RBITC into the AuNPs solutions that were already used for detecting Hg2+. Importantly, the detection limit of this assay for Hg2+ (2.3 nM) was lower than the maximum limits guided by the United States Environmental Protection Agency as well as that permitted by the World Health Organization. The efficiency of this probe was demonstrated in monitoring Hg2+ in complex samples such as river water and living cells.
Keywords: gold nanoparticles, sensitivity, selectivity, recyclable detection, Rhodamine B isothiocyanate
This study provides a highly robust and recyclable gold nanoparticle (AuNP)-based displacement assay for mercuric ions (Hg2+) in aqueous solutions and living cells with ultra-high selectivity and sensitivity. Mercury contamination is an ongoing public concern because inorganic Hg2+ in contaminated water and soil can be transformed into methylmercury and accumulate in the human body through the food chain, posing severe threats to both human health and natural ecosystems.1 Long-term exposure to high Hg2+ levels can lead to serious and permanent damage to the central nervous system and other organs such as heart, kidneys, lungs, etc.2,3 Standard methods for monitoring trace Hg2+ include atomic absorption spectroscopy (AAS),4 inductively coupled plasma-mass spectrometry (ICPMS),5 and mass spectroscopy (MS).6 Although these methods are sensitive and powerful, they require sophisticated instruments and specialized personnel to carry out the operational procedures. Moreover, time-consuming sample preparation and tedious preconcentration procedures are also involved. During the past decade, researchers have devoted considerable effort to the development of simple Hg2+ probes, mainly based on fluorescent chemosensors using small organic molecules,7 polymeric materials,8 oligonucleotides,9 and proteins.10 Most of these systems, however, have limitations with respect to lack of water-solubility, cross-reactivity with other metal ions, and short emission wavelengths. Therefore, it is extremely important to develop simple, inexpensive, highly sensitive and selective Hg2+ probes that can provide real-time measurement of Hg2+ levels in environmental and biological samples.
In the past two decades, tremendous progress in the design of high-sensitivity sensors has been made as a result of the development of nanotechnology. Particularly, AuNP-based fluorescence assays have drawn considerable research interest, because AuNPs are a unique quencher for fluorescent dyes through energy-transfer and/or electron-transfer processes.11,12 The extremely high quenching efficiencies of AuNPs (up to 99.8%13,14) make them very useful in numerous fluorescence-based assays for analytes.15–20 For example, when Rhodamine B (RB) fluorescent dye molecules adsorb onto AuNP surfaces, their fluorescence is efficiently quenched by AuNPs. Upon addition of Hg2+, RBs are displaced from the AuNP surfaces to recover their fluorescence.15,16 The sensitivity of these assays is higher than those reported by using conventional colorimetric assays. However, the selectivity of these assays is so poor that it makes them unlikely to detect Hg2+ by the RB-AuNP system alone. In order to improve selectivity, pretreatment with thiol ligands and addition of masking agent (2,6-pyridinedicarboxylic acid, PDCA) were required.15,16 The pretreatment of AuNPs and involvement of masking agent make these assays laborious and time-consuming, and thus unsuitable for real-time detection of Hg2+. Additionally, most AuNP-based assays fail to precisely determine targeted analytes in their native environments such as environmental samples and biological fluids; because, AuNPs tend to aggregate in real samples, and thus are limited to pure aqueous solutions where Hg2+ is added.21 Therefore, it is vital to develop more stable, reliable and accurate detection methods with sufficient sensitivity and selectivity for Hg2+ in real samples.
We herein design an improved, recyclable displacement assay for Hg2+ detection for use in not only aqueous solutions but also living cells by using Rhodamine B isothiocyanate (RBITC) - poly (ethylene glycol) (PEG)-co-modified AuNPs (RBITC-PEG-AuNPs). The excellent selectivity of this assay for Hg2+ over competing species can be expected by comparing the stability constant (logKf) of metallic ions with isothiocyanate (ITC, -N=C=S), a high-affinity anchoring groups commonly used to attach organic molecules to colloidal AuNPs. We note that the logKf of Hg(ITC)n is ca. 21.8, whereas those of Co2+, Zn2+, Cd2+, Ni2+, Pb2+, Mn2+, Fe2+, Fe3+, Cr3+, Cu2+ and Au+ are ca. 1.72, 2.0, 2.8, 1.76, 1.48, 1.23, 1.31, 4.64, 3.08, 10.4 and 16.98 respectively.22 The logKf of Hg(ITC)n is the only one that is larger than that of Au(ITC)n, indicating that Hg2+ has the highest affinity toward ITC. As a result, only Hg2+ is capable of removing RBITC from the AuNP surface. The detachment of RBITC from AuNP surfaces induces the recovery of RBITC fluorescence that was initially quenched by AuNPs (Figure 1). After fluorescence detection, RBITC can be replenished on the AuNP surface, and the RBITC-PEG-AuNP platform can be used for additional rounds of Hg2+ detection. To address instability of nanomaterials in native samples, we elegantly achieve monodispersity of RBITC-PEG-AuNPs in real detection conditions by functionalizing AuNP surfaces with thiol terminated PEG. We previously reported that Hg2+ is unable to remove thiol ligands from Au surfaces, except in extremely high acidic solutions (pH 1.0).23 Therefore, after Hg2+ is added to the detection assay, only RBITC can be displaced from the AuNP surfaces leaving PEG stably adsorbed on AuNPs for continued detection.
Figure 1.
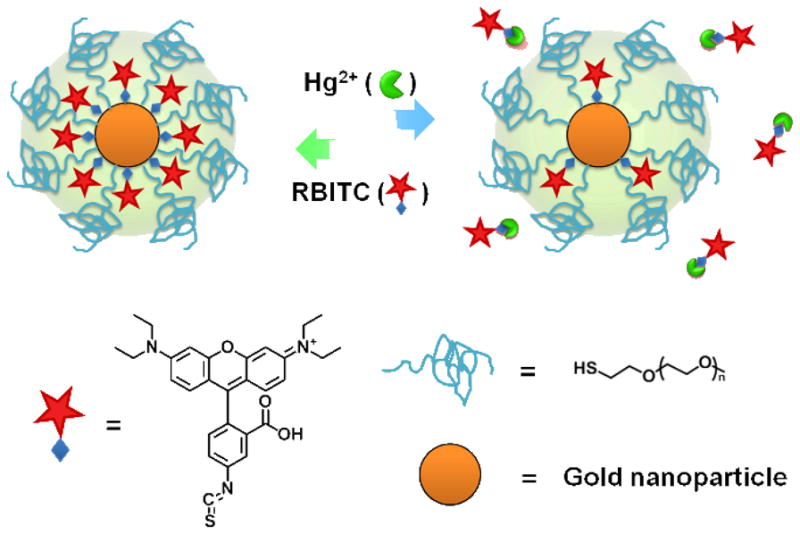
Schematic illustration of the design for detecting Hg2+ ions in a recyclable way.
RESULT AND DISCUSSION
RBITC was chosen as the model fluorophore in this study because it is water-soluble, photostable, and strongly fluorescent. More importantly, AuNPs can be ideal energy acceptors quenching RBITC fluorescence via the nanoparticle surface energy transfer (NSET) effect.11 RBITC molecules can tightly adsorb and anchor around AuNP surfaces by means of the strong affinity between the ITC group and Au.24 In general, the most commonly used 13 nm AuNPs are prepared using a citrate reduction method and are stabilized by the negatively-charged citrate. Upon addition of RBITC, the citrate, weakly adsorbed on Au surfaces, is replaced by RBITC molecules through Au–ITC bonds. It is worth noting that the metal-sulfur bond is found to be the strongest bond compared to other general functional chemical groups such as amines, carboxylic acids, phosphors, and alcohols. Therefore, the sulfur exchange method is the most common way to replace the original capping molecules and functionalize ligands onto Au surfaces.25–27
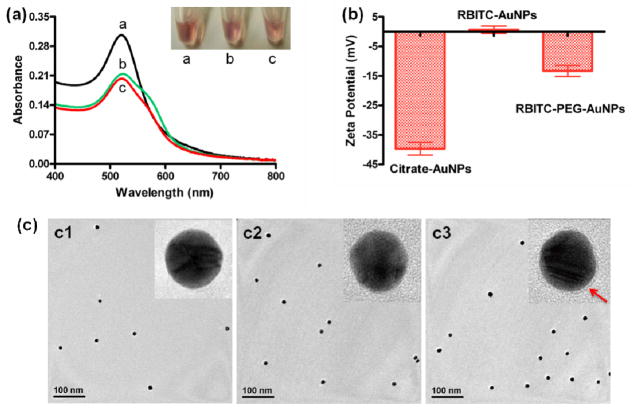
In order to utilize this assay in the real world, thiol terminated PEG was subsequently added and adsorbed onto the remaining active sites of AuNPs via Au–S bond.26 To confirm the quality of AuNPs after RBITC and PEG attachment, the AuNPs were characterized using UV-vis spectroscopy. As shown in Figure 2a, the as-prepared citrate-AuNPs solutions were red in color and showed a typical absorption band at 520 nm, which was attributed to the surface plasmon resonance of AuNPs whose size is about 13 nm.23 The UV-vis spectra of RBITC-AuNPs and RBITC-PEG-AuNPs are similar to that of the citrate-AuNPs. The dominant absorption peak at 520 nm demonstrated proper dispersion of the 13-nm AuNPs, while the appearance of a new peak at roughly 555 nm was attributed to the RBITC absorbance of the RBITC-AuNP conjugates, which is similar to the other reported dye-AuNP systems. 28–30 We employed zeta potential measurements to further investigate the surface charge variations of AuNPs before and after functionalization (Figure 2b). As reported elsewhere, the charge of citrate-AuNPs is negative (−39.7 ± 3.8 mV) because of the citric acid.23,26,31 Once modified with RBITC, the zeta potential of AuNPs increased to be 0.7 ± 2.2 mV, most likely due to the co-presence of the positively-charged quaternary ammonium and the negatively-charged carboxyl group in RBITC. After PEG adsorption on the remaining active sites of RBITC-AuNPs, the charge decreased to a negative value (−13.3 ± 3.2 mV), owing to the huge amount of ethylene oxide units in PEG chains.32–34 The surface functionalization was also supported by dynamic light scattering (DLS) data (Figure S1). The average hydrodynamic diameter of RBITC-AuNPs was 21 nm (similar to the size of citrate-AuNPs), while that of the RBITC-PEG-AuNPs increased to 33 nm. In addition, the morphology of the functionalized AuNPs was determinated by transmission electron microscopy (TEM) (Figure 2c). By using high-resolution TEM, we can observe a thin organic layer that surrounds the AuNPs with an approximately 2–3 nm thickness (Figure 2c3), while the organic layers on citrate-AuNP and RBITC-AuNP surfaces were too thin to be observed.
Figure 2.
(a) UV-vis spectra of citrate-AuNPs (a1), RBITC-AuNPs (a2) and RBITC-PEG-AuNPs (a3) and their corresponding solution colors. (b) Zeta potentials and (c) TEM images of citrate-AuNPs (c1), RBITC-AuNPs (c2), and RBITC-PEG-AuNPs (c3). High-resolution TEM was performed to analyze the organic layers surronding AuNPs. The red arrow indicates the PEG layer on the AuNP surface.
If some citric acid is still adsorbed on RBITC-AuNPs, it will compete for binding with Hg2+ and decrease the amount of Hg2+ available to displace RBITC from the AuNP surfaces. As a consequence, the sensitivity of this displacement assay will decrease. Therefore, it is necessary to add excess RBITC into citrate-AuNPs solutions during the preparation of the AuNP-based probe to replace all of the citric acid on the AuNP. The amount of RBITC on AuNP surfaces can be quantitatively measured by comparing the absorbance changes of free RBITC before and after mixing with citrate-AuNPs and subsequently adding PEG. RBITC (10 μM) in pure water exhibits a typical fluorescence emission band at around 580 nm; with the addition of citrate-AuNPs, the intensity of the fluorescence peak decreased significantly by about 50 % (Figure S2). The phenomenon demonstrates the presence of the NSET effect between AuNPs and RBITC. To quantify the amount of RBITC that attached onto AuNP surfaces, we measured the changes of UV-vis absorbance for excess, non-conjugated RBITC before and after mixing with citrate-AuNPs. With the assistance of a calibration curve of UV-vis absorbance versus RBITC concentrations (Figure S3), the amount of attached RBITC per AuNP was calculated to be 1200. It is worth noting that the thiol-terminated PEG subsequently added may interact with Au, resulting in the subtraction of RBITC from Au surfaces. So we explored the appropriate reaction time and concentration of PEG. Figure S4 shows the change of fluorescence intensities of RBITC solutions (10 μM) that were subsequently added with citrate-AuNPs (2.5 nM) and various concentrations of PEG (0–5 μM). The PEG functionality was recorded in 60 min. We observed that the fluorescence intensity for each solution increased significantly in 10 min and then displayed a very slow rise from 10 to 60 min. The absence of PEG was set as the control, whose fluorescence intensity had negligible change over time. We conclude that 10 min is sufficient for the PEG functionality of this system.
Next, we investigated the appropriate concentration of PEG by re-dissolving the RBITC-PEG-AuNPs pellets (functionalized by different concentrations of PEG) in phosphate-buffered saline (PBS). As shown in Figure S5, when functionalizing RBITC-AuNPs with low concentrations (0, 1, 2 μM) of PEG, new absorption peaks appeared between 600 and 700 nm in the UV-vis spectra, indicating the formation of Au aggregates.23,26 When we increased the PEG concentration to 3 μM, the RBITC-PEG-AuNPs remained well-dispersed in PBS. Therefore, we chose an optimized concentration of PEG (3 μM) for the modification, where AuNPs can be well-dispersed in high-salt solutions and simultaneously only a small number of RBITC (120 RBITC per AuNP) can be removed from the AuNP surfaces by PEG. In the process of preparing the RBITC-PEG-AuNPs, the absorbance changes of free RBITC in each step was monitored by UV-vis spectra (Figure S6). The results show that the addition of such low concentrations of PEG (3 μM) caused just a few RBITC to be released from AuNP surfaces.
Most nanomaterial-based probes tend to aggregate in complex samples, thus reducing their efficiency and accuracy in monitoring targeted analytes. We herein evaluated the stability of this probe in various complex solutions and those with different pH values. The as-prepared RBITC-PEG-AuNPs were purified by centrifugation and the pellets were re-dissolved in various complex solutions including tap water, river water, PBS, and 10-fold concentrated PBS. Pure water was set as the control. We found that the color of the RBITC-PEG-AuNP solutions remained red even after standing for several hours, and correspondingly, the UV-vis spectra showed the typical absorption band for mono-dispersed AuNPs (Figure S7). In contrast, the RBITC-AuNPs can only be re-dispersed in pure water. Upon mixing RBITC-AuNPs pellets with complex samples (including tap water, river water and PBS), the solution color changed from red to blue, indicating the formation of AuNP aggregates, which was confirmed by the appearance of a broad absorption peak at about 600 nm in the UV-visible spectrum (Figure S8). The results reveal that RBITC-PEG-AuNPs can be stably mono-dispersed in various complex samples, enabling the use of this probe for real-world detection.
In addition, we evaluated if pH values could influence the monodispersity of RBITC-PEG-AuNPs. We utilized the plots of A520/A650 (the ratio of absorbance at 520 nm and 650 nm) versus various pH values to investigate the aggregation states of RBITC-PEG-AuNPs. Higher A520/A650 values indicate lower degrees of aggregation of AuNPs. Figure S9 revealed that RBITC-PEG-AuNPs were well-dispersed in solutions ranging from pH 2.0 to 12.0, while RBITC-AuNPs only remained dispersed in pH 7.0–11.0 solutions. Based on this characterization, we can conclude that this nanosensor has the ability to detect Hg2+ not only in various complex samples but also in solutions with a broad range of pH values.
We noted that the fluorescence intensity of RBITC (1 μM) decreased significantly after reaction with varying concentrations of citrate-AuNPs ranging from 0 to 1000 pM (Figure 3). More citrate-AuNPs resulted in higher-efficiency fluorescence quenching. Based on resonance energy transfer from the RBITC molecules to the localized surface plasmon of AuNPs, a linear Stern–Volmer relationship describing the dynamic quenching effect between AuNP concentrations and RBITC fluorescence intensity should be obtained by Equation (1), where F0 and F are emission intensity of RBITC in the absence and presence of quencher, respectively; KSV is the Stern–Volmer quenching constant; [Q] is the concentration of the AuNP quencher.35
Figure 3.
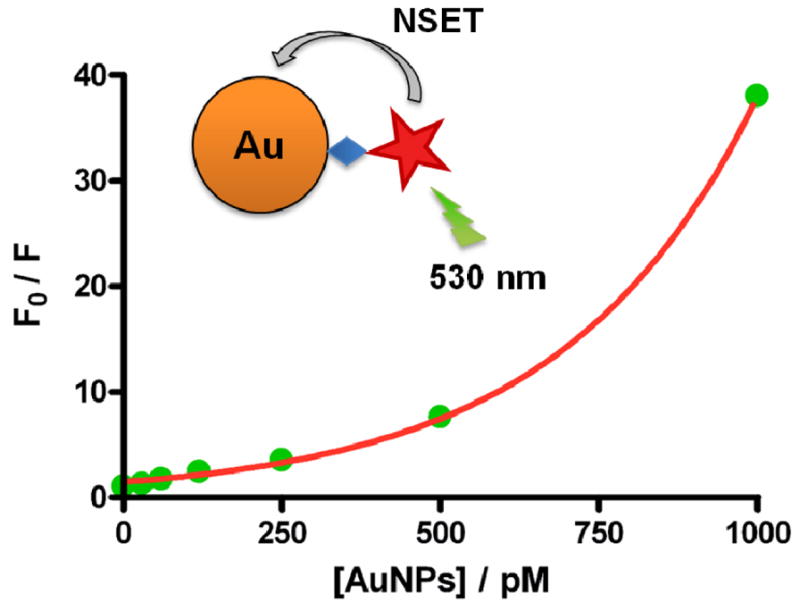
Stern–Volmer plots of RBITC quenching by varying concentrations (0, 30, 60, 120, 250, 500, and 1000 pM) of AuNPs (13 nm).
| (1) |
From the results plotted in Figure 3, however, we observed that the relationship between concentrations of AuNP quencher and F/F0 values was not linear but rather showed an upward deviation from linearity. This effect can be characterized by the combined effects of static quenching, based on the direct binding and association of dyes and AuNPs, and dynamic quenching described above. When taking both quenching effects into account, the quencher concentration factor is no longer in a linear relationship with the fluorescence intensities but rather a squared factor, leading to the extended Stern–Volmer relation:36
| (2) |
| (3) |
where kq is the bimolecular quenching rate constant; τ0 is the excited state lifetime in the absence of quencher; ka is the association constant of RBITC with AuNPs. We reason that the upward curvature of F0/F versus [Q] plots is most likely associated with the superquenching efficiency of AuNP, which arises from the efficient energy transfer from the RBITC donor to the AuNP acceptor. The AuNP acceptor has a large surface-to-volume ratio and an isotropic distribution of dipole vectors to accept the energy from RBITC. In the case of resonant excitation of surface plasmons on AuNPs, a small dipole in the excited RBITC can lead to a large dipole in the AuNP acceptor, resulting in the superquenching efficiencies.
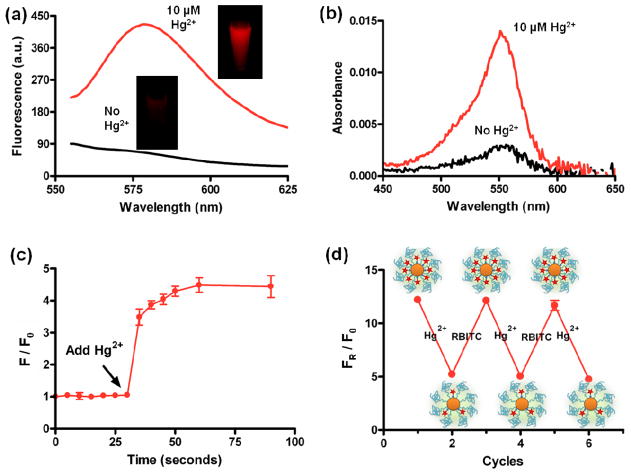
Next, we measured the fluorescence of RBITC-PEG-AuNPs before and after adding Hg2+. In the presence of Hg2+ (10 μM), the attached RBITC molecules were released from AuNP surfaces, and thus the quenched fluorescence of the attached RBITC on AuNPs recovered significantly (Figure 4a). The fluorescence enhancement was observed in a few seconds after the addition of Hg2+ (10 μM). The released RBITC was separated by centrifugation and measured using UV-vis spectroscopy. After separation, we observed the remarkable emergence of an absorption peak at around 555 nm, indicating that Hg2+ had the capacity of removing RBITC from AuNP surfaces. On the other hand, without addition of Hg2+, there was very weak absorbance at the same absorption band (Figure 4b).
Figure 4.
(a) Fluorescence emission spectra and photograph (Inset) of the RBITC-PEG-AuNPs solutions (2.5 nM) before and after addition of 10 μM of Hg2+ ions, and (b) their corresponding UV-vis spectra of released RBITC after centrifugation. (c) Plot of F/F0 values versus time upon the addition of 10 μM of Hg2+ ions into RBITC-PEG-AuNPs solution. (F and F0 are fluorescence intensity at 580 nm in the presence and absence of Hg2+). The arrow indicates the starting time of Hg2+ addition. (d) The FR/F0 values of RBITC-PEG-AuNPs solutions after incubation with Hg2+ and free RBITC alternatively. FR represents the fluorescence intensity of the remaining RBITC left on the RBITC-PEG-AuNPs (released by 100 mM of DTT) while F0 represents the fluorescence intensity of RBITC-PEG-AuNPs.
In order to better understand the response rate of the fluorescence recovery upon addition of Hg2+, we measured the changes of fluorescence intensities at different time intervals by testing the F/F0 values (the ratio of fluorescence intensity at 580 nm in the presence and absence of Hg2+). We found that, with the addition of Hg2+ (the arrow indicates the starting time of Hg2+ addition), F/F0 values increased significantly in 1 min and then remained constant with time (Figure 4c). The results revealed that the displacement reaction can be completed quickly, providing a rapid means to determine Hg2+.
Interestingly, RBITC-PEG-AuNPs can be regenerated by addition of free RBITC into the AuNPs solutions that were already used for detecting Hg2+ and where RBITC was partially released by Hg2+. We reasoned that the freshly added RBITC can bind with the remaining active sites of gold left by the released RBITC. To this study, we firstly treated RBITC-PEG-AuNPs with Hg2+, which we have shown displaces RBITC from the AuNP conjugates. After removing the released RBITC by centrifugation, we incubated the solutions with 100 mM of Dithiothreitol (DTT), a common agent used for completely replacing ligands from Au surfaces.37 At this point, any dye removed by DTT indicates the remaining RBITC left on the RBITC-PEG-AuNPs. We compared the FR/F0 values of the remaining RBITC after adding Hg2+ and free RBITC alternatively along with DTT incubation. FR represents the fluorescence intensity of the remaining RBITC left on the RBITC-PEG-AuNPs while F0 represents the fluorescence intensity of RBITC-PEG-AuNPs. With the addition of Hg2+, the remaining RBITC exhibited reduced FR/F0 values; while addition of free RBITC caused an increase of the FR/F0 values (Figure 4d). In other words, low amounts of RBITC are left on the AuNP after Hg2+ treatment, but this RBITC amount can be replaced onto the RBITC-PEG-AuNP conjugates by simply resupplying the AuNPs with RBITC molecules. It is worth noting that neither the addition of Hg2+ nor the subsequent resupplying of RBITC can induce the aggregation of RBITC-PEG-AuNPs, which was confirmed by the TEM images (Figure S10). The results indicate that this probe is robust and recyclable, making it particularly useful in remote areas.
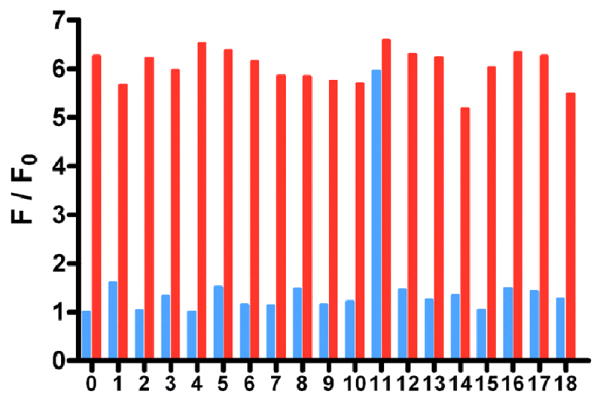
We then studied the selectivity of this nanosensor for Hg2+ by testing the fluorescence responses to other environmentally relevant metallic ions, including Ag+, Al3+, Ba2+, Ca2+, Cd2+, Co2+, Cr2+, Cu2+, Fe2+, Fe3+, K+, Mg2+, Mn2+, Na+, Ni2+, Pb2+ and Zn2+ (Figure 5), each with a concentration of 100 μM. Only Hg2+ (10 μM) induced a noticeable fluorescence recovery of RBITC. As we expected, none of the competing metallic ions interfered with the detection even at increased concentrations up to mM levels. In the control experiments, all metallic ions did not influence the fluorescence of free RBITC, indicating that the enhanced fluorescence of the solution is only due to the Hg2+-induced departing of RBITC from AuNP surfaces and thus fluorescence recovery. When the competing metallic ions were mixed with Hg2+, the solutions had similar behavior to that containing pure Hg2+, i.e., inducing remarkable fluorescence enhancement. This result means that the coexistence of various competing metallic ions would not influence the selectivity of this assay. Unlike many fluorescent assays for Hg2+, which depend on the addition of masking agents and/or require pretreatment to improve selectivity, our designed probe, does not require any extra agents.
Figure 5.
Selectivity of this assay for Hg2+ ions in aqueous solutions. Blue bars indicate the fluorescence recovery of RBITC-PEG-AuNPs solutions (2.5 nM) with addition of 100 μM metal ions of interest: (0) RBITC-PEG-AuNPs solution, (1) Ag+, (2) Al3+, (3) Ba2+, (4) Ca2+, (5) Cd2+, (6) Co2+, (7) Cr2+, (8) Cu2+, (9) Fe2+, (10) Fe3+, (11)Hg2+, (12) K+, (13) Mg2+, (14) Mn2+, (15) Na+, (16) Ni2+, (17) Pb2+ and (18) Zn2+. Red bars represent the eventual fluorescence recovery after addition of 10 μM Hg2+ ions to each solution containing other metal ions.
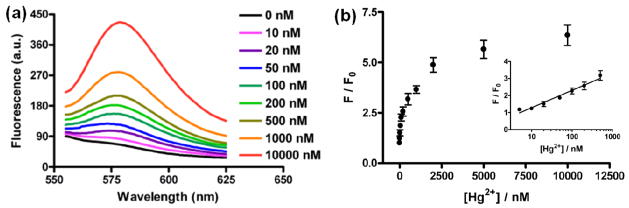
Next, we investigated the sensitivity of this assay for Hg2+ in aqueous solutions. We firstly prepared the solutions containing various concentrations of Hg2+ ranging from 0 to 10 μM. Later, aliquots of the as-prepared RBITC-PEG-AuNP pellets were added into the Hg2+ solutions. The eventual concentration of RBITC-PEG-AuNPs in each solution was determined to be 2.5 nM by UV-vis spectroscopy. We allowed the resulting solutions to incubate at room temperature for 1 min and then recorded the fluorescence of RBITC for each solution. We noted that the levels of fluorescence recovery highly depended on the concentration of Hg2+, where higher concentrations of Hg2+ led to stronger fluorescence recovery. As shown in Figure 6, with the increase of Hg2+ concentration, the fluorescence intensities at 580 nm increased gradually. The changes in fluorescence spectra were quantified by measuring the plots of F/F0 values versus the concentrations of Hg2+. We found the F/F0 values were linear with the Hg2+ concentrations within a range from 10 nM to 500 nM. The limit of detection (LOD) of this assay is 2.3 nM at a signal-to-noise ratio of 3. This value is particularly attractive because it is lower than the maximum level (10 nM) of Hg2+ in drinking water guided by the United States Environmental Protection Agency (EPA)38 as well as that (30 nM) permitted by the World Health Organization (WHO).39
Figure 6.
The sensitivity of this assay for Hg2+ ions by fluorescence recovery. (a) Fluorescence emission spectra of the RBITC-PEG-AuNPs solutions (2.5 nM) after addition of varying Hg2+ ions from 0 to 10−5M. (b) Plot of the fluorescence intensity at 580 nm versus various concentrations of Hg2+ ions in the RBITC-PEG-AuNPs solutions.
Although many nanosensors show great potential in bioanalysis with ultrahigh sensitivity, they only work well under specific conditions such as in pure water and are thus unavailable in complex samples. Encouraged by the excellent stability of RBITC-PEG-AuNPs in various complex solutions as well as the extraordinary selectivity and sensitivity of this probe in pure water, we next evaluated if this probe can be used to monitor Hg2+ in real environmental samples. Firstly, a water sample from the Potomac River near Washington, D.C. was collected and filtered through a 22 μm membrane to remove any insoluble substrates. The river water samples were spiked with varying concentrations of Hg2+ to evaluate the interference of unknown substances in the river water on the detection. The final concentrations of Hg2+ in the river water samples were set to be 0, 10, 20, 40, 80, 150, and 300 nM (1 mL of each concentration). The Hg2+-spiked solutions were mixed with the as-prepared RBITC-PEG-AuNP pellets, of which the eventual concentration of AuNPs was 2.5 nM. We observed that the pellets were easily dispersed, and all the solutions remained red, demonstrating proper dispersion of RBITC-PEG-AuNPs in the Hg2+-spiked solutions. Next, we measured the fluorescence recovery of each solution. As shown in Figure S11, the fluorescence intensity increased with the increase of the added Hg2+ concentration. The LOD at a signal-to-noise ratio of 3 for the added Hg2+ in the river water sample is determined to be 3.8 nM, which is close to that in pure water. The results indicated that the unknown matrices in river water have a negligible effect on the stability and detection of this probe. We suggest that this assay has the capability in monitoring environmentally relevant concentrations of Hg2+.
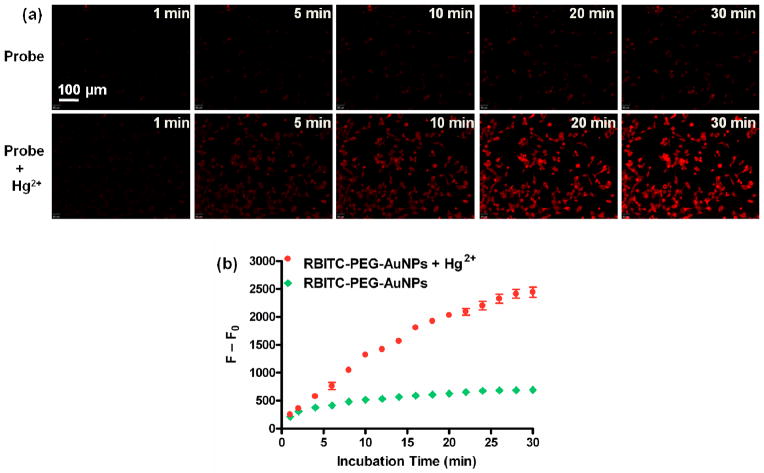
To further investigate the possibility of using this displacement assay for monitoring Hg2+ levels in organisms, we used this assay to detect the intracellular Hg2+ in living cells. MDA-MB-435 cells were firstly incubated with 10 μM of Hg2+ for 0.5 h, and then RBITC-PEG-AuNPs (20 nM) in the medium were added for another 0.5 h incubation. We observed that the fluorescence inside the living cells enhanced gradually, while those without the pretreatment of Hg2+ showed very weak intracellular fluorescence (Figure 7a). The enhancement of fluorescence intensity was quantified by the F-F0 values, where F is the mean fluorescence intensity of RBITC inside cells, and F0 is the background fluorescence (Figure 7b). The results revealed that our assay can be used to monitor intracellular Hg2+ in living cells. Additionally, bright-field measurements showed that the cells after treatment with Hg2+ (10 μM) and RBITC-PEG-AuNPs (20 nM) were still viable during the imaging (Figure S12). We performed MTT experiments to explore the cytotoxicity of RBITC-PEG-AuNPs. The results showed that the cell viability was between 95–100 % after incubation of various concentrations of RBITC-PEG-AuNPs ranging from 0 to 80 nM with cells for 24 h (Figure S13). This indicates that the probe does not affect the cell viability, making this probe particularly suitable for Hg2+ detection in biological samples.
Figure 7.
(a) Intracellular fluorescence at different incubation times. MDA-MB-435 cells were cultured in 8-well plates in the absence (top) and presence (bottom) of 10 μM Hg2+, and RBITC-PEG-AuNPs (20 nM) were added in the medium. The intracellular fluorescence increased over time, which was quantified by the F–F0 values (b). F is the mean fluorescence intensity of RBITC inside cells, and F0 is the background fluorescence.
CONCLUSION
In conclusion, we present a very robust, recyclable, inexpensive, ultrasensitive and selective gold nanoparticle-based NSET probe for monitoring Hg2+ levels in aqueous solutions and living cells. Based on our rational design, this assay was endowed with extraordinary selectivity for Hg2+ over competing analytes. Unlike many nanomaterial-based probes, the RBITC-PEG-AuNPs, described in this manuscript, remained well-dispersed in complex samples such as river water, tap water, and biological fluids, providing a novel means to screen Hg2+ levels in various real samples. Importantly, the sensitivity of this assay (2.3 nM) for Hg2+ in aqueous media is higher than both the EPA and the WHO standard limits. Furthermore, this probe can be recyclable and the detection can be completed in 1 min, enabling this assay for Hg2+ monitoring not only in developed areas but also in remote areas with limited resources. We believe this AuNP-based displacement assay is a promising point-of-care device to be used in many settings, especially in combination with other formats such as lab-on-chip and micro-fluidic devices.40–43
EXPERIMENTAL SECTION
Materials and Instrumentation
Chemicals such as Rhodamine B isothiocyanate (RBITC), Poly(ethylene glycol) (PEG) 2000, HAuCl4·3H2O, trisodium citrate, and metallic salts (AgNO3, Al(NO3)3·9H2O, Ba(NO3)2, CaCl2, Cd(SO4)·8H2O, CoCl2·6H2O, Cr(NO3)3·9H2O, Cu(NO3)2·3H2O, Fe(NO3)2·6H2O, Fe(NO3)3·9H2O, Hg(ClO4)2·3H2O, KNO3, MgSO4, MnSO4·2H2O, NaNO3, Ni(NO3)2·6H2O, Pb(NO3)2, Zn(NO3)2·6H2O) were purchased from major suppliers such as Sigma-Aldrich and Alfa Aesar and used as received. Distilled water was used throughout the work. The UV-vis spectra of gold colloidal solutions and Rhodamine B isothiocyanate (RBITC) solutions were recorded with a Genesys 10s UV-vis spectrophotometer. The fluorescence spectra were collected using an F-7000 fluorescence spectrophotometer (Hitachi, Tokyo, Japan) operating at an excitation wavelength at 530 nm, excitation and emission slit widths were 5 nm and 5 nm, respectively. The fluorescence intensities were collected at 580 nm. We used the emission fluorescence value over the initial emission fluorescence value (F/F0) to measure the variations of the fluorescence intensity. Dynamic light scattering (DLS) and zeta potential (ζ) were performed on a Zeta Sizer Nano ZS (Malvern Zetasizer 3000HS and He/Ne laser at 632.8 nm and scattering angles of 90 at 25 °C). TEM images were obtained by using a JEOL1400 model at an accelerating voltage of 100 kV.
Preparation of RBITC-AuNPs
The synthesis of citrate-AuNPs (13 nm) has been reported elsewhere.15,16,21 In brief, a stirred aqueous solution of HAuCl4 (41 mg, 1.0 mM) in 100 mL water was heated to reflux, and a trisodium citrate solution (114 mg, 38.8 mM) dissolved in hot water (10 mL) was added. The solution was heated under reflux with vigorous stirring for another 15 min, its color changed from pale yellow to deep red. The solution was cooled to room temperature with a slow and continuous stirring. The resulting solution was filtered with a PES membrane (filter unit is 22 μm) to remove some large clusters and insoluble compounds. The sizes of the nanoparticles were about 13 nm by TEM analysis, the corresponding absorption band is at ~520 nm.
In order to prepare RBITC-PEG-AuNPs, the pH value of the as-prepared citrate-AuNPs solution (2.5 nM, 1 mL) was adjusted by K2CO3 (100 mM) to be 8. A stock solution of RBITC (10 mM, 1 μL) was added into the citrate-AuNPs solution (2.5 nM, 1 mL) with vigorous shaking to allow adsorption of RBITC to the AuNP surface. The resulting solution was shaken (600 r/min) in the dark at room temperature for 1 hour for sufficient equilibration. Later, PEG (1 mM, 3 μL) was added into the solution of RBITC-AuNPs and agitated for 10 min at room temperature. The excess RBITC and PEG were removed by three runs of centrifugation (14000 r/min, 15 min) and the AuNP pellets were obtained and stored at 4 °C for further use. The fluorescence spectra of the RBITC-PEG-AuNPs solutions were measured with excitation at 530 nm. The decrease of the fluorescence of RBITC-PEG-AuNPs solutions indicated that RBITC molecules had adsorbed onto the surfaces of AuNPs and the fluorescence of RBITC was dramatically quenched by AuNPs.
Experimental Procedures for Detection of Hg2+ in Aqueous Solutions
A typical detection procedure for Hg2+ was performed as follows. A stock solution of Hg2+ was serially diluted with distilled water for various folds. The volume for each Hg2+ solution was 1 mL. Aliquots of the as-prepared RBITC-PEG-AuNP pellets were mixed with the different concentrations of Hg2+ solutions. The resulting mixtures were kept in the dark for 1 min, and then the fluorescence for each solution was measured at room temperature. All the measurements were repeated 3 times for each concentration.
To investigate the selectivity of this assay, 0.1 mmol of each kind of metal ions (Ag+, Al3+, Ba2+, Ca2+, Cd2+, Co2+, Cr2+, Cu2+, Fe2+, Fe3+, Hg2+, K+, Mg2+, Mn2+, Na+, Ni2+, Pb2+ and Zn2+) was dissolved in distilled water (1 mL) to afford 100 mM aqueous solution. The stock solutions were diluted to desired concentrations with distilled water when needed. Typically, 1 mL of each metallic salt (100 μM) was incubated with aliquots of the as-prepared RBITC-PEG-AuNP pellets, after 1 min incubation, the fluorescence intensities of the resulting solutions were measured and analyzed.
To evaluate the potential of this assay in practical applications, we collected river water from the Potomac River near Washington, D.C and used as real samples. The river water was filtered by using a PES membrane (filter unit is 0.22 μm) to remove the insoluble materials, and then spiked with different volumes of Hg2+ stock solutions to result in final concentrations of Hg2+ to be: 0, 10, 20, 40, 80, 150, and 300 nM (1 mL of each concentration). The spiked river water containing varying concentrations of Hg2+ was mixed with the RBITC-PEG-AuNP pellets, and the fluorescence for each solution was recorded after 1 min of incubation in the dark.
Cell culture
Human breast carcinoma malignant cell line (MDA-MB-435) was cultured in L-15 medium (GIBCO) supplemented with 10 % fetal bovine serum (GIBCO) and 1 % penicillin/streptomycin sulfate (antibiotic, GIBCO), and maintained at 37 °C in a humidified 5 % CO2 atmosphere. Before seeding, cells were washed with PBS, dissociated from culture plates with trypsin/EDTA (GIBCO), and resuspended in L-15 medium containing 10 % FBS. After centrifugation at 800 rpm for 3 minutes, the cell pellet was resuspended in L-15 medium containing 10 % of FBS (cell density is about 1×104/well).
Cell imaging methods
MDA-MB-435 cells were seeded in 8-well plates and cultured overnight. After cell attachment, the wells were washed with fresh medium three times and then 10 μM of Hg2+ in PBS were added and incubated for 30 min at room temperature. After another round of washing with medium three times, the Hg2+-treated cells were incubated with the RBITC-PEG-AuNPs solutions (20 nM) in fresh medium, then the fluorescence imaging of intracelluar Hg2+ was observed by RFP filter under an Olympus IX81 microscope. Time-lapse images were acquired at 1 min intervals for 0.5 h with 10× objective lens. The MDA-MB-435 cells without the pretreatment of Hg2+ were set as a control. For all fluorescence images, the microscope settings such as brightness, contrast, and exposure time were held constant to compare the relative intensity of intracellular Hg2+ fluorescence.
Cytotoxicity
The cytotoxicity of RBITC-PEG-AuNPs was investigated using the standard MTT assay protocol. In brief, MDA-MB-435 cells were incubated with various concentrations of RBITC-PEG-AuNPs (0, 1.25, 2.5, 5, 10, 20, 40, 80 nM) in medium for 24 h. The medium was replaced with 200 μL fresh media containing 20 μL of MTT solution (5 mg/mL), and the incubation proceeded for 4 h. The media was then removed, and 150 μL of dimethyl sulfoxide (DMSO) was added into each well to dissolve the internalized purple formazan crystals. An aliquot of 100 μL of solution in each well was transferred into a new 96-well plate. The absorbance at 570 nm was recorded using a microplate reader. The absorbance from the control cells was set as 100 % cell viability.
Supplementary Material
Acknowledgments
This work was supported in part, by the National Basic Research Program of China (973 program) (973 Program No. 2013CB733802, 2013CB733800), the National Science Foundation of China (NSFC) (81101101, 51273165, 51173117, 50830107, 81171399, and 81101077), the National Significant New Drugs Creation Program (2012ZX09505-001-001), the Jiangsu Province Social Development Program (BE2012622 and BL2012031), the Outstanding Professional Fund of Health Ministry in Jiangsu Province (RC2011095), the Chinese Academy of Sciences professorship for Senior International Scientists (2011T2J06), and the Intramural Research Program (IRP) of the National Institute of Biomedical Imaging and Bioengineering (NIBIB), National Institutes of Health (NIH).
Footnotes
Supporting Information Details of DLS measurements, UV-Vis and fluorescence emission spectra, and TEM Images of various AuNP formulas. Fluorescence microscope images of MDA-MB-435 cells treated with Hg2+ and RBITC-PEG-AuNPs. This material is available free of charge via the Internet at http://pubs.acs.org.
Contributor Information
Min Yang, Email: yangmin@jsinm.org.
Xingyu Jiang, Email: xingyujiang@nanoctr.cn.
Xiaoyuan Chen, Email: shawn.chen@nih.gov.
References
- 1.Bolger PM, Schwetz BA. Mercury and Health. N Engl J Med. 2002;347:1735–1736. doi: 10.1056/NEJMp020139. [DOI] [PubMed] [Google Scholar]
- 2.Clarkson TW, Magos L, Myers GJ. The Toxicology of Mercury-Current Exposures and Clinical Manifestations. N Engl J Med. 2003;349:1731–1737. doi: 10.1056/NEJMra022471. [DOI] [PubMed] [Google Scholar]
- 3.Onyido I, Norris AR, Buncel E. Biomolecule-Mercury Interactions: Modalities of DNA Base-Mercury Binding Mechanisms. Remediation Strategies. Chem Rev. 2004;104:5911–5929. doi: 10.1021/cr030443w. [DOI] [PubMed] [Google Scholar]
- 4.Erxleben H, Ruzicka J. Atomic Absorption Spectroscopy for Mercury, Automated by Sequential Injection and Miniaturized in Lab-on-Valve System. Anal Chem. 2005;77:5124–5128. doi: 10.1021/ac058007s. [DOI] [PubMed] [Google Scholar]
- 5.Jia XY, Gong DR, Han Y, Wei C, Duan TC, Chen HT. Fast Speciation of Mercury in Seawater by Short-Column High-Performance Liquid Chromatography Hyphenated to Inductively Coupled Plasma Spectrometry after On-line Cation Exchange Column Preconcentration. Talanta. 2012;88:724–729. doi: 10.1016/j.talanta.2011.10.026. [DOI] [PubMed] [Google Scholar]
- 6.Leermakers M, Baeyens W, Quevauviller P, Horvat M. Mercury in Environmental Samples: Speciation Artifacts and Validation. Trends Anal Chem. 2005;24:383–393. [Google Scholar]
- 7.Huang JH, Xu YF, Qian XH. A Rhodamine-Based Hg2+ Sensor with High Selectivity and Sensitivity in Aqueous Solution: A NS2-Containing Receptor. J Org Chem. 2009;74:2167–2170. doi: 10.1021/jo802297x. [DOI] [PubMed] [Google Scholar]
- 8.Kim IB, Bunz UHF. Modulating the Sensory Response of a Conjugated Polymer by Proteins: An Agglutination Assay for Mercury Ions in Water. J Am Chem Soc. 2006;128:2818–2819. doi: 10.1021/ja058431a. [DOI] [PubMed] [Google Scholar]
- 9.Ono A, Togashi H. Highly Selective Oligonucleotide-Based Sensor for Mercury(II) in Aqueous Solutions. Angew Chem, Int Ed. 2004;43:4300–4302. doi: 10.1002/anie.200454172. [DOI] [PubMed] [Google Scholar]
- 10.Chen P, He C. A General Strategy to Convert the MerR Family Proteins into Highly Sensitive and Selective Fluorescent Biosensors for Metal Ions. J Am Chem Soc. 2004;126:728–729. doi: 10.1021/ja0383975. [DOI] [PubMed] [Google Scholar]
- 11.Yun CS, Javier A, Jennings T, Fisher M, Hira S, Peterson S, Hopkins B, Reich NO, Strouse GF. Nanometal Surface Energy Transfer in Optical Rulers, Breaking the FRET Barrier. J Am Chem Soc. 2005;127:3115–3119. doi: 10.1021/ja043940i. [DOI] [PubMed] [Google Scholar]
- 12.Bunz UHF, Rotello VM. Gold Nanoparticle-Fluorophore Complexes: Sensitive and Discerning “Noses” for Biosystems Sensing. Angew Chem Int Ed. 2010;49:3268–3279. doi: 10.1002/anie.200906928. [DOI] [PubMed] [Google Scholar]
- 13.Dulkeith E, Ringler M, Klar TA, Feldmann J, Muñoz Javier A, Parak WJ. Gold Nanoparticles Quench Fluorescence by Phase Induced Radiative Rate Suppression. Nano Lett. 2005;5:585–589. doi: 10.1021/nl0480969. [DOI] [PubMed] [Google Scholar]
- 14.Swierczewska M, Lee S, Chen X. The Design and Application of Fluorophore–Gold Nanoparticle Activatable Probes. Phys Chem Chem Phys. 2011;13:9929–9941. doi: 10.1039/c0cp02967j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Darbha GK, Ray A, Ray PC. Gold Nanoparticle-Based Miniaturized Nanomaterial Surface Energy Transfer Probe for Rapid and Ultrasensitive Detection of Mercury in Soil, Water, and Fish. ACS Nano. 2007;1:208–214. doi: 10.1021/nn7001954. [DOI] [PubMed] [Google Scholar]
- 16.Huang CC, Chang HT. Selective Gold-Nanoparticle-Based “Turn-On” Fluorescent Sensors for Detection of Mercury(II) in Aqueous Solution. Anal Chem. 2006;78:8332–8338. doi: 10.1021/ac061487i. [DOI] [PubMed] [Google Scholar]
- 17.Liu DB, Chen WW, Wei JH, Li XB, Wang Z, Jiang XY. A Highly Sensitive, Dual-Readout Assay Based on Gold Nanoparticles for Organophosphorus and Carbamate Pesticides. Anal Chem. 2012;84:4185–4191. doi: 10.1021/ac300545p. [DOI] [PubMed] [Google Scholar]
- 18.Liu DB, Chen WW, Tian Y, He S, Zheng WF, Sun JS, Wang Z, Jiang XY. A Highly Sensitive Gold-Nanoparticle-Based Assay for Acetylcholinesterase in Cerebrospinal Fluid of Transgenic Mice with Alzheimer’s Disease. Adv Healthcare Mater. 2012;1:90–95. doi: 10.1002/adhm.201100002. [DOI] [PubMed] [Google Scholar]
- 19.Chen JL, Zheng AF, Chen AH, Gao YC, He CY, Kai XM, Wu GH, Chen YC. A Functionalized Gold Nanoparticles and Rhodamine 6G Based Fluorescent Sensor for High Sensitive and Selective Detection of Mercury(II) in Environmental Water Samples. Anal Chim Acta. 2007;599:134–142. doi: 10.1016/j.aca.2007.07.074. [DOI] [PubMed] [Google Scholar]
- 20.De M, Rana S, Akpinar H, Miranda OR, Arvizo RR, Bunz UHF, Rotello VM. Sensing of Proteins in Human Serum Using Conjugates of Nanoparticles and Green Fluorescent Protein. Nat Chem. 2009;1:461–465. doi: 10.1038/nchem.334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu DB, Wang Z, Jiang XY. Gold Nanoparticles for the Colorimetric and Fluorescent Detection of Ions and Small Organic Molecules. Nanoscale. 2011;3:1421–1433. doi: 10.1039/c0nr00887g. [DOI] [PubMed] [Google Scholar]
- 22.Smith RM, Martell AE. Critical Stability Constants. Plenum Press; New York: 1976. p. 4. Inorganic complexes. [Google Scholar]
- 23.Liu DB, Qu WS, Chen WW, Zhang W, Wang Z, Jiang XY. Highly Sensitive, Colorimetric Detection of Mercury(II) in Aqueous Media by Quaternary Ammonium Group-Capped Gold Nanoparticles at Room Temperature. Anal Chem. 2010;82:9606–9610. doi: 10.1021/ac1021503. [DOI] [PubMed] [Google Scholar]
- 24.Qian XM, Emory SR, Nie SM. Anchoring Molecular Chromophores to Colloidal Gold Nanocrystals: Surface-Enhanced Raman Evidence for Strong Electronic Coupling and Irreversible Structural Locking. J Am Chem Soc. 2012;134:2000–2003. doi: 10.1021/ja210992b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hammock ML, Sokolov AN, Stoltenberg RM, Naab BD, Bao Z. Organic Transistors with Ordered Nanoparticle Arrays as a Tailorable Platform for Selective, In Situ Detection. ACS Nano. 2012;6:3100–3108. doi: 10.1021/nn204830b. [DOI] [PubMed] [Google Scholar]
- 26.Liu DB, Chen WW, Sun K, Deng K, Zhang W, Wang Z, Jiang XY. Resettable, Multi-Readout Logic Gates Based on Controllably Reversible Aggregation of Gold Nanoparticles. Angew Chem, Int Ed. 2011;50:4103–4107. doi: 10.1002/anie.201008198. [DOI] [PubMed] [Google Scholar]
- 27.Kumar AS, Ye T, Takami T, Yu BC, Flatt AK, Tour JM, Weiss PS. Reversible Photo-Switching of Single Azobenzene Molecules in Controlled Nanoscale Environments. Nano Lett. 2008;8:1644–1648. doi: 10.1021/nl080323+. [DOI] [PubMed] [Google Scholar]
- 28.Joshi P, Chakraborti S, Ramirez-Vick JE, Ansari ZA, Shanker V, Chakrabarti P, Singh SP. The Anticancer Activity of Chloroquine-Gold Nanoparticles against MCF-7 Breast Cancer Cells. Colloids Surf B. 2012;95:195–200. doi: 10.1016/j.colsurfb.2012.02.039. [DOI] [PubMed] [Google Scholar]
- 29.Ding Y, Chen Z, Xie J, Guo R. Comparative Studies on Adsorption Behavior of Thionine on Gold Nanoparticles with Different Sizes. J Colloid Interface Sci. 2008;327:243–250. doi: 10.1016/j.jcis.2008.07.057. [DOI] [PubMed] [Google Scholar]
- 30.Narband N, Uppal M, Dunnill CW, Hyett G, Wilsonb M, Parkin IP. The Interaction between Gold Nanoparticles and Cationic and Anionic Dyes: Enhanced UV-visible Absorption. Phys Chem Chem Phys. 2009;11:10513–10518. doi: 10.1039/b909714g. [DOI] [PubMed] [Google Scholar]
- 31.Ojea-Jiménez I, López X, Arbiol J, Puntes V. Citrate-Coated Gold Nanoparticles As Smart Scavengers for Mercury(II) Removal from Polluted Waters. ACS Nano. 2012;6:2253–2260. doi: 10.1021/nn204313a. [DOI] [PubMed] [Google Scholar]
- 32.Rostro-Kohanloo BC, Bickford LR, Payne CM, Day ES, Anderson LJE, Zhong M, Lee S, Mayer KM, Zal T, Adam L, et al. The Stabilization and Targeting of Surfactant-Synthesized Gold Nanorods. Nanotechnology. 2009;20:434005. doi: 10.1088/0957-4484/20/43/434005. [DOI] [PubMed] [Google Scholar]
- 33.Lipka J, Semmler-Behnke M, Sperling RA, Wenk A, Takenaka S, Schleh C, Kissel T, Parak WJ, Kreyling WG. Biodistribution of PEG-Modified Gold Nanoparticles Following Intratracheal Instillation and Intravenous Injection. Biomaterials. 2010;31:6574–6581. doi: 10.1016/j.biomaterials.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 34.Liopo A, Conjusteau A, Tsyboulski D, Ermolinsky B, Kazansky A, Oraevsky A. Biocompatible Gold Nanorod Conjugates for Preclinical Biomedical Research. J Nanomed Nanotechnol. 2012;S2:001–010. doi: 10.4172/2157-7439.S2-001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fan CH, Wang S, Hong JW, Bazan GC, Plaxco KW, Heeger AJ. Beyond Superquenching: Hyper-Efficient Energy Transfer from Conjugated Polymers to Gold Nanoparticles. Proc Natl Acad Sci US A. 2003;100:6297–6301. doi: 10.1073/pnas.1132025100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thipperudrappa J, Biradar DS, Hanagodimath SM. Simultaneous Presence of Static and Dynamic Component in the Fluorescence Quenching of Bis-MSB by CCl4 and Aniline. J Lumin. 2007;1244:45–50. [Google Scholar]
- 37.Stoeva SI, Lee J, Smith JE, Rosen ST, Mirkin CA. Multiplexed Detection of Protein Cancer Markers with Biobarcoded Nanoparticle Probes. J Am Chem Soc. 2006;128:8378–8379. doi: 10.1021/ja0613106. [DOI] [PubMed] [Google Scholar]
- 38.http://water.epa.gov/drink/contaminants/basicinformation/mercury.cfm#four
- 39.http://www.who.int/water_sanitation_health/dwq/fulltext.pdf
- 40.Zhang WH, Lin SC, Wang CM, Hu J, Li C, Zhuang ZX, Zhou YL, Mathies RA, Yang CJ. PMMA/PDMS Valves and Pumps for Disposable Microfluidics. Lab Chip. 2009;9:3088–3094. doi: 10.1039/b907254c. [DOI] [PubMed] [Google Scholar]
- 41.Hu M, Yan J, He Y, Lu HT, Weng LX, Song SP, Fan CH, Wang LH. Ultrasensitive, Multiplexed Detection of Cancer Biomarkers Directly in Serum by Using a Quantum Dot-Based Microfluidic Protein Chip. ACS Nano. 2010;4:488–494. doi: 10.1021/nn901404h. [DOI] [PubMed] [Google Scholar]
- 42.Du Y, Chen CG, Zhou M, Dong SJ, Wang EK. Microfluidic Electrochemical Aptameric Assay Integrated On-Chip: A Potentially Convenient Sensing Platform for the Amplified and Multiplex Analysis of Small Molecules. Anal Chem. 2011;83:1523–1529. doi: 10.1021/ac101988n. [DOI] [PubMed] [Google Scholar]
- 43.Xiang Y, Lu Y. Portable and Quantitative Detection of Protein Biomarkers and Small Molecular Toxins Using Antibodies and Ubiquitous Personal Glucose Meters. Anal Chem. 2012;84:4174–4178. doi: 10.1021/ac300517n. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.