Abstract
Background
Osteosarcoma cell lines and tumors have been shown to express EGFR and harbor amplifications at the EGFR locus. In this study, we further analyze the genomic features of EGFR in osteosarcoma tumors and investigate whether they correlate with PTEN expression and copy number status.
Methods
EGFR and PTEN expression were assessed by immunohistochemistry (n=28), and copy number alterations at the EGFR and PTEN loci were surveyed using Affymetrix® 50K single nucleotide polymorphism (SNP) arrays (n=31) and fluorescence in situ hybridization (FISH) (n=27). The EGFR tyrosine kinase domain was sequenced to survey for activating mutations (n=34). In addition, we assessed EGFRvIII expression using RT-PCR (n=24). Results were correlated with available clinical information on 59 patients, with a median age of 14.1 years (range, 5–23 years) and median follow up of 4.4 years.
Results
EGFR expression was detected in the majority of osteosarcoma tumors surveyed (23/28; 82%). SNP arrays revealed evidence of frequent copy number gains at 7p11.2 and losses at 10q23.21. A sizeable subset (16/27 cases; 59%) showed gains at the EGFR locus using FISH – amplification in 4/27 (15%) and balanced chromosome 7 polysomy in 12/27 (44%), and 12 cases showed deletions at the PTEN locus – biallelic deletions in 4/27 (15%) and monoallelic deletion in 9/27 (33%). No activating mutations in the EGFR tyrosine kinase domain, EGFRvIII expression, or association with clinical findings were detected.
Conclusion
EGFR expression and genomic gains at the EGFR locus are prevalent in osteosarcoma tumors, which also commonly harbor deletions at the PTEN locus.
Keywords: childhood cancer, osteosarcoma, epidermal growth factor receptor, tyrosine kinase, EGFRvIII, PTEN, single nucleotide polymorphism array
INTRODUCTION
The epidermal growth factor receptor (EGFR; ErbB1) is a cell membrane receptor with intrinsic protein tyrosine kinase activity that has been the subject of rigorous investigation in view of its involvement in several human cancers and its potential as a target of therapy.1 Several classes of anti-EGFR agents are currently approved for clinical use or in advanced clinical trials1 in adult patients with cancer. Phase I clinical trials of EGFR tyrosine kinase inhibitors, such as gefitinib, have included children with refractory solid tumors, including patients with osteosarcoma.2, 3 Similar phase I trials of gefinitib in adult patients did not include osteosarcoma patients.4, 5 Activation of EGFR in normal cells is induced by ligand binding, which leads to tightly regulated stimulation of proliferative and survival cell signaling pathways, most notably the phosphatidylinositol 3-kinase (PI3K)–AKT pathway.6, 7
Various types of EGFR alterations have been detected in human cancers, with varying clinical implications. Such alterations include aberrant EGFR expression, with or without gene amplification8–12, expression of a mutant form with in-frame deletion of exons 2 to 7 (EGFRvIII)13, and point mutations in the tyrosine kinase domain. The EGFRvIII mutant protein lacks the ligand-binding domain but is capable of constitutive phosphorylation of tyrosine residues and subsequent activation of downstream signaling cascades14, and its expression appears to confer sensitivity to tyrosine kinase inhibitors.15 Response to EGFR inhibitors appears to correlate with expression of EGFRvIII and concurrent loss of PTEN in some neoplasms.16, 17 In addition, somatic mutations in the tyrosine kinase domain of EGFR have been found to correlate with response to tyrosine kinase inhibitors in a subset of patients with non-small cell lung carcinoma.18–20 Furthermore, increased EGFR gene copy number has been found to be associated with higher response rates to gefitinib and lower progression rates in patients with lung21 and colon22 carcinoma.
Osteosarcoma is the most common primary malignant bone tumor in children and adolescents.23, 24 Advances in osteosarcoma therapy over the past several decades have enhanced patient outcomes, with most effective regimens currently including neoadjuvant and adjuvant chemotherapy coupled with local control that usually consists of limb-sparing surgery.25 However, outcome remains poor for most patients with metastatic or recurrent osteosarcoma and even for some patients with localized disease at presentation, despite intensification of initial chemotherapy and/or the use of second-line chemotherapeutic agents.26–32
Among childhood malignancies, expression of EGFR has been reported in Wilms tumor33, and it was recently found to correlate with the embryonal subtype of rhabdomyosarcoma34. In osteosarcoma, in vitro data from early passage osteosarcoma cells demonstrate constitutive EGFR phosphorylation whose abrogation leads to growth inhibition and apoptosis.35, 36 Furthermore, recent studies have demonstrated EGFR expression in osteosarcoma tumors.37, 38 Interestingly, EGFR expression appears to be associated with favorable clinical outcome in patients with osteosarcoma.37
In this study, we assess EGFR expression, copy number status, and tyrosine kinase domain mutations in a large group of osteosarcoma tumors. Furthermore, because loss of PTEN has been identified as an important molecular determinant of response to EGFR tyrosine kinase inhibitors in some cancers17, 39, we investigate whether these findings correlate with PTEN expression and copy number status, as well as with clinical features and disease outcome.
MATERIALS AND METHODS
Tumor samples and patient study group
This study was approved by the St Jude Children’s Research Hospital (SJCRH) Institutional Review Board. A total of 80 osteosarcoma cases were utilized, as summarized in Table S1. Due to limitations in available tumor material, only subsets of these 80 osteosarcoma tumor samples were amenable for use in the different experiments performed in this study. All tumors fulfilled the diagnostic criteria for osteosarcoma as defined in the World Health Organization classification.23 Formalin-fixed, paraffin-embedded (FFPE) tumors (n=28) were incorporated into tissue microarrays (TMA) using a manual arrayer (Beecher Instruments, Silver Spring, Maryland, USA). A minimum of three cores representing viable, non-decalcified osteosarcoma tumor tissue were collected from donor blocks. Cores from paraffin-embedded normal tissues were distributed in each of the TMA blocks as controls. Adequacy of tumor viability (<20% necrosis) and histologic subtype were assessed on all frozen tumor samples by routine microscopic examination of a representative section.
Clinical information was available on 59 patients. Only limited demographic and clinical data were available on the 21 remaining cases, which included patients who were referred for clinical and/or pathological consultation but were not admitted to or managed at SJCRH. Notably, the patients whose tumor material was used in this study were managed disparately on a variety of therapeutic protocols and nonprotocol regimens. The demographic and clinical features of the patient group are summarized in Table 1.
Table 1.
Summary of demographic and clinical features
| Total (n=59) | |
|---|---|
|
| |
| Age (years) | |
| Median (range) | 14.1 (5.01–23.5) |
| ≤14 | 28 |
| >14 | 31 |
|
| |
| Gender | |
| Female | 26 |
| Male | 33 |
|
| |
| Race | |
| Non-White | 22 |
| White | 37 |
|
| |
| Survival status | |
| Alive | 41 |
| Expired | 18 |
|
| |
| Site of primary tumor | |
| Extremity | 55 |
| Axial | 4 |
|
| |
| Disease at presentation (n=58) | |
| Local, resectable disease | 48 |
| Local, unresectable disease | 1 |
| Metastatic disease | 10 |
|
| |
| Rosen grade* (n=53) | |
| 1 or 2 | 22 |
| 3 or 4 | 31 |
|
| |
| Follow up (years) | |
| Median (range) | 4.4 (0.5–14) |
|
| |
| Outcome (%) | |
| 5-Year overall survival | 63.6 ± 10.3 |
| 5-Year event-free survival | 57.0 ± 10.0 |
Immunohistochemistry for assessment of EGFR and PTEN expression
Expression of EGFR was assessed using a monoclonal anti-EGFR antibody (31G7, 1:10; Zymed, San Francisco, California, USA) on the automated Ventana Benchmark XT immunostainer (Ventana Medical Systems, Tucson, Arizona, USA) using the manufacturer’s deparaffinization, antigen retrieval, and detection reagents as described previously.40 Expression of PTEN was assessed using a monoclonal anti-PTEN antibody (6H2.1, 1:400; Dako, Carpinteria, California, USA)41 on a semi-automoated Dako Automated Stainer (Dako). For PTEN immunostaining, antigen retrieval was carried out manually by placing tissue sections in plastic Coplin jars containing preheated TRIS/EDTA pH 9 solution in a household vegetable steamer (Sunbeam-Oster, Model Sunbeam 4713/5710, 900 W) for 30 minutes. Subsequent steps were performed using the Dako Automated Stainer (Dako) at room tempreature. We used 3,3′ diaminobenzidine/H2O2 (Dako) or 3-amino-9-ethyl carbazol as chromogens and hematoxylin as the counterstain. All immunostains were performed in duplicate. Negative control immunostains were performed as indicated above, less the primary antibody. Tonsil tissue (full sections and scattered cores in TMA blocks) served as positive control for both the EGFR and PTEN immunostains, with the former staining the squamous epithelium and the latter staining a subset of lymphocytes.
Immunostaining results were assessed as described previously.34 Immunostaining intensity was estimated for each tissue core as follows: 1, weak; 2, moderate (equivalent to immunostaining by positive control in TMA); or 3, strong. The percentage of tumor cells positive for EGFR and PTEN was estimated by counting at least 500 tumor cells in each core. Results were scored by multiplying the percentage of positive cells by the estimated intensity of immunostaining (score=percentage × intensity; range, 0 to 300). The core with the highest score determined the final value for each case. For the purposes of statistical analysis, a score of >20 was used to define positivity based on a data distribution plot (not shown), and tumors were considered to have strong EGFR or PTEN expression when the immunostaining score was ≥120.
Single nucleotide polymorphism (SNP) array analysis for assessment of EGFR and PTEN copy number status
SNP analysis was performed using the Affymetrix Mapping 50K Xba-1 240 array (Affymetrix, Santa Clara, California), which interrogates 59,015 SNPs across the whole human genome.42 DNA quality was confirmed by UV spectrophotometry and agarose gel electrophoresis. DNA samples were processed by the core laboratory in the Hartwell Center for Bioinformatics and Biotechnology at SJCRH using the recommended Affymetrix protocol. Briefly, 250 ng of genomic DNA from 31 frozen osteosarcoma tumors from 30 patients was digested to completion with Xba-I restriction endonuclease then ligated to an adapter containing a generic primer sequence. Three aliquots (25 ng each) of ligated DNA were amplified using conditions to enrich for 250 to 2000 bp DNA fragments and then pooled and purified. DNA was fragmented using DNase I and end-labeled with biotinylated nucleotide using terminal deoxynucleotidyl transferase. The labeled targets were added to a hybridization cocktail containing probe array controls and blocking agents (human Cot-1 and herring sperm DNA) and incubated overnight at 48°C on a GeneChip array. After hybridization, arrays were stringently washed and stained with Streptavidin Phycoerythrin (SAPE, Invitrogen - Molecular Probes, Carlsbad, California, USA) using the Affymetrix Fluidics Station 400. Arrays were scanned using the Affymetrix GeneChip Scanner 3000 and genotype calls determined using the Affymetrix Gene DNA Analysis Software (GDAS) version 3.0.
Fluorescence in situ hybridization for confirmation of EGFR and PTEN copy number status
Dual-color FISH was performed on 4 μm paraffin-embedded TMA sections. Probes were derived from BAC clones (Invitrogen; CHORI, Oakland, California, USA) and labeled with either FITC or Rhodamine fluorochromes. Target probes were paired with control probes from the opposite chromosomal arm. For EGFR copy number status assessment, a FITC-conjugated probe that is specific for the EGFR locus on chromosome 7p11.2 (RPC11-148P17; Invitrogen) was paired with a rhodamine-conjugated control probe specific for the CFTR locus on chromosome 7q31.2 (RP11-460J21 & CTB-133K23; Invitrogen). For PTEN copy number status assessment, a rhodamine-conjugated probe that is specific for the PTEN locus on chromosome 10q23.21 (RP11-380G5; Invitrogen) was paired with a FITC-conjugated control probe specific for 10p11.21 (RP11-254A5 & RP11-32212; Invitrogen). Nuclei were counterstained with DAPI (1000ng/ml) (Insitus) for viewing on a Nikon Eclipse E800 fluorescence microscope. Images were captured and processed with an exposure time ranging from 0.5–2 seconds for each fluorochrome using Cytovision v3.6 software (Applied Imaging, San Jose, California, USA).
Cores in which >90% of nuclei showed hybridization signals were considered informative, and those were scored for the number of fluorescent signals by counting 100 non-overlapping intact nuclei. FISH patterns were defined as follows: balanced disomy refers to the identification of dual signals from both the test and control probes; balanced polysomy refers to similar gains in copy numbers for both the test and control probes; gene amplification refers to unbalanced gains in copy number for the test probe.
Mutation analysis of the EGFR tyrosine kinase domain
Genomic DNA was extracted from 34 frozen osteosarcoma tumors and used to amplify EGFR exons 18, 19, 20, and 21 using primer sequences and PCR conditions that were kindly provided by Dr. Matthew Meyerson (Dana-Farber Cancer Institute, Boston, Massachussetts, USA) and available upon request. Following amplification, all PCR products were purified using the Qiaex II gel extraction kit (Qiagen, Valencia, CA, USA) or Microcon-100 purification columns (Millipore, Billerica, MA, USA). Purified DNA was then sequenced in both the sense and antisense directions on all cases by Big Dye Terminator (v.3) Chemistry using PCR primers on Applied Biosystem 3700 DNA Analyzers.
Assessment of EGFRvIII expression
RNA of adequate quality was extracted from 24 frozen osteosarcoma tumors and used to detect EGFRvIII mutant expression as described previously by Ji et al15. Briefly, RNA was retrotranscribed into first-strand cDNA using the iScript cDNA synthesis kit (Bio-Rad Laboratories, Hercules, California, USA). PCR was then used to amplify the region spanning EGFR exons 1 and 8. The RT-PCR products – 128 bp (EGFRvIII) and/or 929 bp (WT EGFR) – were visualized using 2% agarose gel electrophoresis.
Statistical analysis
Exact Pearson chi-square tests were used to test for associations among categorical variables. The Kaplan-Meier method was used to estimate event-free survival (EFS), defined as the time interval from diagnosis to first event (relapse, progression, second malignancy, or death from any cause) or to last follow up for patients without events.
Copy number and LOH analyses of the 50K SNP array data were performed using dChipSNP as described previously.43, 44 For copy number analysis, data were first normalized to a baseline array having median signal intensity at the probe intensity level with the invariant set normalization method. After normalization, the signal values for each SNP in each array were obtained with a model-based (perfect-match/mismatch difference model) method. Signal intensities for each SNP in each tumor were then compared with those from a reference set of 61 normal samples and copy numbers inferred using a hidden Markov Model (HMM).45, 46 LOH regions were identified using a HMM-based method and the 61 unpaired normal samples.
RESULTS
EGFR expression is common in osteosarcoma tumors
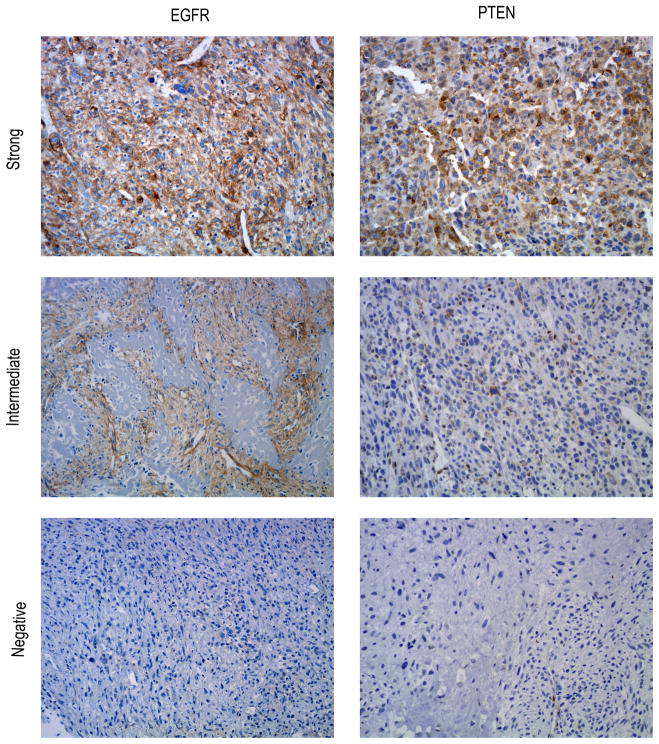
Immunostaining results are illustrated in Figure 1. EGFR expression was identified using immunohistochemistry in 23 of 28 (82%) osteosarcoma tumors, of which 19 of 23 (83%) exhibited strong expression. The average EGFR immunostaining score was 149 (median: 135; range among EGFR-positive cases: 50–300). The immunostaining pattern was membranous or membranous and cytoplasmic in all tumors.
Figure 1.
Panel illustrating EGFR and PTEN expression in osteosarcoma using immunohistochemistry. For EGFR, assigned scores for these illustrated cases were 300, 100, and 0. For PTEN, assigned scores for these illustrated cases were 300, 100, and 0.
PTEN immunostaining was identified in 19 of 28 (68%) osteosarcoma tumors. The average PTEN immunostaining score was 122 (median: 110; range among PTEN-positive cases: 30–300). Of the 23 EGFR-positive osteosarcoma tumors, 9 (39%) were PTEN-negative; however, there was no correlation between EGFR expression PTEN expression (p=0.64). Interestingly, two of the 28 osteosarcoma tumors exhibited chondroblastic morphology, and both were EGFR-positive and PTEN-negative.
Inferred genomic gains at 7p11.2 and losses at 10q23.21 are frequent in osteosarcoma tumors
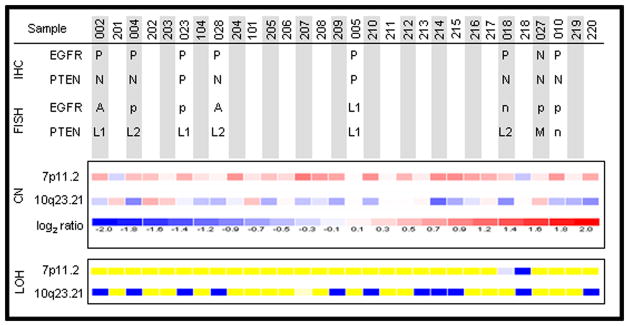
Using SNP arrays, we inferred the presence of frequent copy number gains in the region of the EGFR locus and frequent losses in the region of the PTEN locus in osteosarcoma tumors, as compared to the reference set (Figure 2). Of 31 tumors analyzed, at least 16 osteosarcoma tumors exhibited evidence of copy number gains in the chromosome 7p11.2 region whereas at least 12 exhibited evidence of copy number losses in the 10q23.21 region. Notably, 13 tumors (42%) harbored concurrent evidence of copy number gains at 7p11.2 and copy number losses at 10q23.21. In addition, 11 of 31 (35%) of osteosarcoma tumors analyzed show LOH at the PTEN locus, whereas only 2 cases show LOH at the EGFR locus.
Figure 2.
Results of 50K SNP array analysis in 31 osteosarcoma tumors demonstrate frequent genomic gains at 7p11.2, which includes the EGFR locus, and frequent genomic losses at 10q23.21, which includes the PTEN locus, using log2 ratio of tumor signal/mean signal of all 61 references for each SNP in each tumor case. In addition, loss of heterozygosity appears more common at 10q23.21 than at 7p11.2 (yellow: retention; dark blue: loss of heterozygosity; light blue: non-informative). These findings are matched with corresponding data (top panel) obtained using fluorescence in situ hybridization and immunohistochemistry where available. [A: amplification; CN: copy number; FISH: fluorescence in situ hybridization; IHC: immunohistochemistry; M: mixed pattern of monoallelic loss and polysomy; n: suboptimal hybridization; N: negative; p: balanced polysomy; P: positive; L1: monoallelic loss; L2: biallelic loss; LOH: loss of heterozygosity]
EGFR amplification is detected using FISH in a subset of osteosarcoma tumors
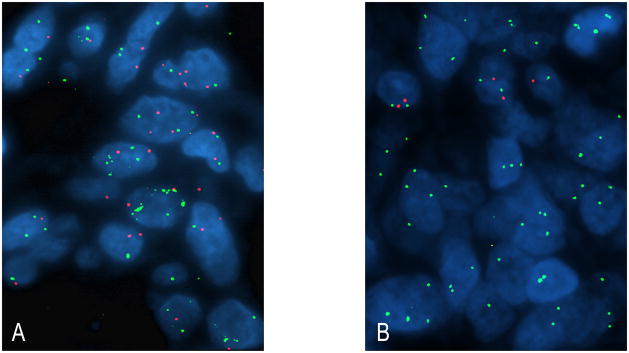
To begin understanding the molecular mechanisms underlying EGFR expression in osteosarcoma tumors and validate the data obtained from SNP arrays, we performed FISH on 28 osteosarcoma tumors to estimate the copy number status of the EGFR locus on chromosome 7p11.2. Of 27 (27/28, 96%) osteosarcoma tumors with interpretable hybridization signals, 4 (15%) showed evidence of EGFR gene amplification (Figure 3A). In addition, gain of material at 7p11.2 resulting from balanced chromosome 7 polysomy was identified in 12 (44%) tumors, monoallelic deletion was identified in 1 (4%) tumor, and 10 (37%) tumors had balanced disomy.
Figure 3.
(A) Fluorescence in situ hybridization demonstrating osteosarcoma tumor with EGFR gene amplification (green signal: EGFR test probe at 7p11.2; red signal: control probe at 7q31.2). (B) Another osteosarcoma tumor harboring biallelic loss of PTEN (red signal: PTEN test probe at 10q23.21; green signal: control probe at 10p11.21); note the presence of chromosome 10 polysomy.
All osteosarcoma tumors with EGFR amplification had high-level EGFR expression. However, of the 12 tumors with evidence of balanced polysomy, 9 had EGFR expression (6 strong, 3 weak) and 3 lacked EGFR expression. There was no correlation between EGFR copy number status, as assessed by FISH, and protein expression, as assessed by immunohistochemistry.
We also performed FISH on 28 osteosarcoma tumors to estimate the copy number status of the PTEN gene at 10q23.21. Of 27 (27/28, 96%) osteosarcoma tumors with interpretable hybridization signals, 4 (15%) tumors had biallelic deletion at the PTEN locus, 9 (33%) tumors had monoallelic deletion at the PTEN locus, 3 (11%) tumors showed balanced polysomy, 5 (19%) had a mixed pattern whereby subsets of tumor cells harbored monoallelic deletion of PTEN whereas others harbored balanced polysomy, and 6 (22%) had balanced disomy (Figure 3B). Because it was uncertain whether cases that demonstrated a mixed pattern should be assigned to the “loss” or “gain” categories, these cases were not included in subsequent statistical analyses.
When we examined the relation between PTEN copy number status and expression, we found a significant association between biallelic deletions of PTEN and negative or weak PTEN expression (p=0.041). In addition, osteosarcoma tumors with either monoallelic or biallelic deletion of PTEN tended to have weak or negative PTEN expression (p=0.057). Of note, we identified a correlation between EGFR amplification and monoallelic deletion of PTEN (p=0.05) in our study group. Interestingly, however, there was no correlation between EGFR amplification and biallelic deletion of PTEN.
EGFRvIII is not expressed in osteosarcoma tumors
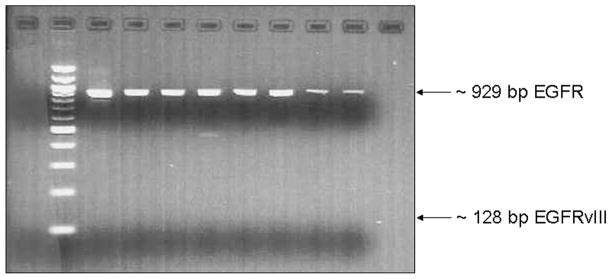
EGFRvIII, a mutant form of EGFR with in-frame deletion of exons 2–7 (801 bp), constitutively activates the PI3K signaling pathway and confers sensitivity to EGFR tyrosine kinase inhibitors16. Using RT-PCR and primers specific for EGFR exons 1 and 8, we found no evidence of deletion of exons 2–7 in any of the 24 osteosarcoma tumors surveyed. Gel electrophoresis showed variable intensity ~929 bp amplification bands, as expected for the wild type EGFR transcript (Figure 4).
Figure 4.
Agarose gel electrophoresis of RT-PCR products using EGFR exon 1 and exon 8 primers. The wild type EGFR mRNA is expected to yield a 929bp amplicon whereas the EGFRvIII mutant mRNA, in which exons 2 to 7 are deleted, is expected to yield a 128bp amplicon. None of the cases analyzed in our group showed expression of EGFRvIII.
Mutation analysis of the EGFR tyrosine kinase domain reveals no evidence of activating mutations
Certain mutations in the EGFR tyrosine kinase domain lead to constitutive activation and confer susceptibility to certain tyrosine kinase inhibitors.1 To determine whether such mutations are present in osteosarcoma tumors, we analyzed exons 18–21, which encode for the EGFR tyrosine kinase domain. Interestingly, known EGFR polymorphisms without resulting amino acid changes were noted in most cases, as summarized in Table S2. However, we did not identify known mutations that have been associated with constitutive EGFR activation in the 34 osteosarcoma tumors analyzed.
Correlation of EGFR and PTEN expression and copy number status with clinical features
We found no correlation between EGFR or PTEN expression and copy number status with presenting clinical features and outcome data, which included disease stage at presentation, primary tumor’s histologic response to chemotherapy (Rosen grade I and II vs III and IV), and survival.
DISCUSSION
In cancer cells, EGFR aberrations impact a variety of cell signaling pathways, notably the PI3K-AKT and JAK/STAT pathways.1 The PI3K-AKT pathway plays an important role in modulating the mammalian target of rapamycin (mTOR) and is effectively inhibited by PTEN.47 In vitro data indicate that EGFR and AKT signaling play a role in the pathogenesis of osteosarcoma35, 36, 48. Our data additionally demonstrate that in osteosarcoma tumors there is an overall genomic gain in EGFR and loss in PTEN. Such an imbalance implies that growth-promoting pathways influenced by EGFR, notably PI3K-AKT/mTOR, might be constitutively upregulated in osteosarcoma. In fact, AKT activation or functional loss of PTEN, the major negative regulator of PI3K, has been found to comprise an important cause of resistance to the anti-EGFR agent, gefitinib.49 Although further studies are needed to explore the downstream influence of EGFR aberrations in osteosarcoma tumors, evidence in support of such a contention includes data showing that inhibition of mTOR50 and PI3-AKT51 have adverse effects on a murine model of osteosarcoma and osteosarcoma cell lines, respectively. Interestingly, in a survey of 32 osteosarcoma tumors, we did not identify evidence of STAT3 activation (J. Khoury, unpublished data), arguing against a prominent role for the JAK/STAT pathway. Taken together, these data appear to indicate that osteosarcoma tumors could be biologically dependent on EGFR-modulated cell signaling pathways, especially PI3K-AKT/mTOR, a fact that might be exploitable for therapeutic purposes.
In addition to demonstrating that most osteosarcoma tumors harbor genomic gains at the EGFR locus using SNP arrays and FISH, our data also show that loss of PTEN is a common occurrence in osteosarcoma tumors. We were intrigued to identify a correlation between EGFR amplification and monoallelic loss of PTEN. Indeed, there is mounting evidence indicating that loss of PTEN in tumors with EGFR expression is associated with resistance to anti-EGFR tyrosine kinase inhibitors.39 In vitro evidence suggests that such resistance can be overcome by coupling anti-EGFR agents with mTOR inhibitors.52 Novel therapeutic approaches of this kind appear warranted if anti-EGFR therapy is to be investigated in osteosarcoma patients. Furthermore, since it appears that in a sizeable subset (44%) of osteosarcoma tumors the mechanism of copy number gain at the EGFR locus involves balanced chromosome 7 polysomy or gains in large segments of chromosome 7, further studies will be necessary to determine whether response to anti-EGFR therapy in osteosarcoma may be influenced by the mechanism of copy number gain.
Our data are in line with two recent studies that also demonstrate EGFR expression in a sizeable subset of osteosarcoma tumors, of which a subset also harbors gains at the EGFR locus.37, 38 Interestingly, one of those studies identified a correlation between EGFR expression and favorable clinical outcome.37 In our study group, we did not identify a correlation between expression and copy number status of EGFR, or PTEN, and clinical features. We speculate that the most likely causes include the non-uniformity of therapeutic regimens used to treat patients in our group, a limitation of this retrospective study, and the disparity in the number of pre-therapy versus post-therapy samples employed in the two studies.
In conclusion, data from this study using osteosarcoma tumor samples support in vitro and recent clinical data suggesting a role for the EGFR/PI3K-AKT pathway in osteosarcoma. Clinical investigative trials using approved and/or novel therapeutic agents that specifically target this pathway appear warranted.
Acknowledgments
This work was supported by NIH grants P01CA023099 (JDK) and R21CA98543 (JSD) and by the American Lebanese Syrian Associated Charities (ALSAC). The authors acknowledge the services of James Dalton, Charlene Henry, Catherine Billups, and the St. Jude Children’s Research Hospital Tissue Resources Facility.
Footnotes
Disclaimers:
None of the authors have any significant potential conflicts of interest to disclose.
- This study was presented, in part, at the 2007 Annual Meeting of the United States and Canadian Academy of Pathology.
References
- 1.Hynes NE, Lane HA. ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer. 2005 May;5(5):341–354. doi: 10.1038/nrc1609. [DOI] [PubMed] [Google Scholar]
- 2.Daw NC, Furman WL, Stewart CF, et al. Phase I and pharmacokinetic study of gefitinib in children with refractory solid tumors: a Children’s Oncology Group Study. J Clin Oncol. 2005 Sep 1;23(25):6172–6180. doi: 10.1200/JCO.2005.11.429. [DOI] [PubMed] [Google Scholar]
- 3.Freeman BB, 3rd, Daw NC, Geyer JR, Furman WL, Stewart CF. Evaluation of gefitinib for treatment of refractory solid tumors and central nervous system malignancies in pediatric patients. Cancer Invest. 2006 Apr-May;24(3):310–317. doi: 10.1080/07357900600632058. [DOI] [PubMed] [Google Scholar]
- 4.Baselga J, Rischin D, Ranson M, et al. Phase I safety, pharmacokinetic, and pharmacodynamic trial of ZD1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. J Clin Oncol. 2002 Nov 1;20(21):4292–4302. doi: 10.1200/JCO.2002.03.100. [DOI] [PubMed] [Google Scholar]
- 5.Ranson M, Hammond LA, Ferry D, et al. ZD1839, a selective oral epidermal growth factor receptor-tyrosine kinase inhibitor, is well tolerated and active in patients with solid, malignant tumors: results of a phase I trial. J Clin Oncol. 2002 May 1;20(9):2240–2250. doi: 10.1200/JCO.2002.10.112. [DOI] [PubMed] [Google Scholar]
- 6.Schlessinger J. Common and distinct elements in cellular signaling via EGF and FGF receptors. Science. 2004 Nov 26;306(5701):1506–1507. doi: 10.1126/science.1105396. [DOI] [PubMed] [Google Scholar]
- 7.Yarden Y, Sliwkowski MX. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol. 2001 Feb;2(2):127–137. doi: 10.1038/35052073. [DOI] [PubMed] [Google Scholar]
- 8.Bhargava R, Gerald WL, Li AR, et al. EGFR gene amplification in breast cancer: correlation with epidermal growth factor receptor mRNA and protein expression and HER-2 status and absence of EGFR-activating mutations. Mod Pathol. 2005 Aug;18(8):1027–1033. doi: 10.1038/modpathol.3800438. [DOI] [PubMed] [Google Scholar]
- 9.Hirsch FR, Varella-Garcia M, Bunn PA, Jr, et al. Epidermal growth factor receptor in non-small-cell lung carcinomas: correlation between gene copy number and protein expression and impact on prognosis. J Clin Oncol. 2003 Oct 15;21(20):3798–3807. doi: 10.1200/JCO.2003.11.069. [DOI] [PubMed] [Google Scholar]
- 10.Ohgaki H, Dessen P, Jourde B, et al. Genetic pathways to glioblastoma: a population-based study. Cancer Res. 2004 Oct 1;64(19):6892–6899. doi: 10.1158/0008-5472.CAN-04-1337. [DOI] [PubMed] [Google Scholar]
- 11.Sunpaweravong P, Sunpaweravong S, Puttawibul P, et al. Epidermal growth factor receptor and cyclin D1 are independently amplified and overexpressed in esophageal squamous cell carcinoma. J Cancer Res Clin Oncol. 2005 Feb;131(2):111–119. doi: 10.1007/s00432-004-0610-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kersemaekers AM, Fleuren GJ, Kenter GG, et al. Oncogene alterations in carcinomas of the uterine cervix: overexpression of the epidermal growth factor receptor is associated with poor prognosis. Clin Cancer Res. 1999 Mar;5(3):577–586. [PubMed] [Google Scholar]
- 13.Moscatello DK, Holgado-Madruga M, Godwin AK, et al. Frequent expression of a mutant epidermal growth factor receptor in multiple human tumors. Cancer Res. 1995 Dec 1;55(23):5536–5539. [PubMed] [Google Scholar]
- 14.Pedersen MW, Meltorn M, Damstrup L, Poulsen HS. The type III epidermal growth factor receptor mutation. Biological significance and potential target for anti-cancer therapy. Ann Oncol. 2001 Jun;12(6):745–760. doi: 10.1023/a:1011177318162. [DOI] [PubMed] [Google Scholar]
- 15.Ji H, Zhao X, Yuza Y, et al. Epidermal growth factor receptor variant III mutations in lung tumorigenesis and sensitivity to tyrosine kinase inhibitors. Proc Natl Acad Sci U S A. 2006 May 16;103(20):7817–7822. doi: 10.1073/pnas.0510284103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kuan CT, Wikstrand CJ, Bigner DD. EGF mutant receptor vIII as a molecular target in cancer therapy. Endocr Relat Cancer. 2001 Jun;8(2):83–96. doi: 10.1677/erc.0.0080083. [DOI] [PubMed] [Google Scholar]
- 17.Mellinghoff IK, Wang MY, Vivanco I, et al. Molecular determinants of the response of glioblastomas to EGFR kinase inhibitors. N Engl J Med. 2005 Nov 10;353(19):2012–2024. doi: 10.1056/NEJMoa051918. [DOI] [PubMed] [Google Scholar]
- 18.Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004 May 20;350(21):2129–2139. doi: 10.1056/NEJMoa040938. [DOI] [PubMed] [Google Scholar]
- 19.Paez JG, Janne PA, Lee JC, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004 Jun 4;304(5676):1497–1500. doi: 10.1126/science.1099314. [DOI] [PubMed] [Google Scholar]
- 20.Pao W, Miller V, Zakowski M, et al. EGF receptor gene mutations are common in lung cancers from “never smokers” and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc Natl Acad Sci U S A. 2004 Sep 7;101(36):13306–13311. doi: 10.1073/pnas.0405220101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cappuzzo F, Hirsch FR, Rossi E, et al. Epidermal growth factor receptor gene and protein and gefitinib sensitivity in non-small-cell lung cancer. J Natl Cancer Inst. 2005 May 4;97(9):643–655. doi: 10.1093/jnci/dji112. [DOI] [PubMed] [Google Scholar]
- 22.Moroni M, Veronese S, Benvenuti S, et al. Gene copy number for epidermal growth factor receptor (EGFR) and clinical response to antiEGFR treatment in colorectal cancer: a cohort study. Lancet Oncol. 2005 May;6(5):279–286. doi: 10.1016/S1470-2045(05)70102-9. [DOI] [PubMed] [Google Scholar]
- 23.Raymond AK, Ayala AG, Knuutila S. Conventional osteosarcoma. In: Fletcher CDM, Unni KK, Mertens F, editors. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of Soft Tissue and Bone. Lyon: IARC Press; 2002. pp. 264–270. [Google Scholar]
- 24.Ries LAG, Eisner MP, Kosary CL, et al. SEER Cancer Statistics Review, 1975–2002. Bethesda, MD: National Cancer Institute; 2005. Based on November 2004 SEER data submission, posted to the SEER web site. [Google Scholar]
- 25.Marina N, Gebhardt M, Teot L, Gorlick R. Biology and therapeutic advances for pediatric osteosarcoma. Oncologist. 2004;9(4):422–441. doi: 10.1634/theoncologist.9-4-422. [DOI] [PubMed] [Google Scholar]
- 26.Bacci G, Longhi A, Versari M, Mercuri M, Briccoli A, Picci P. Prognostic factors for osteosarcoma of the extremity treated with neoadjuvant chemotherapy: 15-year experience in 789 patients treated at a single institution. Cancer. 2006 Mar 1;106(5):1154–1161. doi: 10.1002/cncr.21724. [DOI] [PubMed] [Google Scholar]
- 27.Meyers PA, Heller G, Healey J, et al. Chemotherapy for nonmetastatic osteogenic sarcoma: the Memorial Sloan-Kettering experience. J Clin Oncol. 1992 Jan;10(1):5–15. doi: 10.1200/JCO.1992.10.1.5. [DOI] [PubMed] [Google Scholar]
- 28.Meyers PA, Heller G, Healey JH, et al. Osteogenic sarcoma with clinically detectable metastasis at initial presentation. J Clin Oncol. 1993 Mar;11(3):449–453. doi: 10.1200/JCO.1993.11.3.449. [DOI] [PubMed] [Google Scholar]
- 29.Mialou V, Philip T, Kalifa C, et al. Metastatic osteosarcoma at diagnosis: prognostic factors and long-term outcome--the French pediatric experience. Cancer. 2005 Sep 1;104(5):1100–1109. doi: 10.1002/cncr.21263. [DOI] [PubMed] [Google Scholar]
- 30.Rodriguez-Galindo C, Shah N, McCarville MB, et al. Outcome after local recurrence of osteosarcoma: the St. Jude Children’s Research Hospital experience (1970–2000) Cancer. 2004 May 1;100(9):1928–1935. doi: 10.1002/cncr.20214. [DOI] [PubMed] [Google Scholar]
- 31.Tabone MD, Kalifa C, Rodary C, Raquin M, Valteau-Couanet D, Lemerle J. Osteosarcoma recurrences in pediatric patients previously treated with intensive chemotherapy. J Clin Oncol. 1994 Dec;12(12):2614–2620. doi: 10.1200/JCO.1994.12.12.2614. [DOI] [PubMed] [Google Scholar]
- 32.Bielack SS, Kempf-Bielack B, Delling G, et al. Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1,702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol. 2002 Feb 1;20(3):776–790. doi: 10.1200/JCO.2002.20.3.776. [DOI] [PubMed] [Google Scholar]
- 33.Ghanem MA, Van Der Kwast TH, Den Hollander JC, et al. Expression and prognostic value of epidermal growth factor receptor, transforming growth factor-alpha, and c-erb B-2 in nephroblastoma. Cancer. 2001 Dec 15;92(12):3120–3129. doi: 10.1002/1097-0142(20011215)92:12<3120::aid-cncr10173>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 34.Ganti R, Skapek SX, Zhang J, et al. Expression and genomic status of EGFR and ErbB-2 in alveolar and embryonal rhabdomyosarcoma. Mod Pathol. 2006 Sep;19(9):1213–1220. doi: 10.1038/modpathol.3800636. [DOI] [PubMed] [Google Scholar]
- 35.Hughes DP, Thomas DG, Giordano TJ, Baker LH, McDonagh KT. Cell surface expression of epidermal growth factor receptor and Her-2 with nuclear expression of Her-4 in primary osteosarcoma. Cancer Res. 2004 Mar 15;64(6):2047–2053. doi: 10.1158/0008-5472.can-03-3096. [DOI] [PubMed] [Google Scholar]
- 36.Hughes DP, Thomas DG, Giordano TJ, McDonagh KT, Baker LH. Essential erbB family phosphorylation in osteosarcoma as a target for CI-1033 inhibition. Pediatr Blood Cancer. 2006 May 1;46(5):614–623. doi: 10.1002/pbc.20454. [DOI] [PubMed] [Google Scholar]
- 37.Kersting C, Gebert C, Agelopoulos K, et al. Epidermal growth factor receptor expression in high-grade osteosarcomas is associated with a good clinical outcome. Clin Cancer Res. 2007 May 15;13(10):2998–3005. doi: 10.1158/1078-0432.CCR-06-2432. [DOI] [PubMed] [Google Scholar]
- 38.Wen YH, Koeppen H, Garcia R, et al. Epidermal growth factor receptor in osteosarcoma: expression and mutational analysis. Hum Pathol. 2007 Aug;38(8):1184–1191. doi: 10.1016/j.humpath.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 39.Bianco R, Shin I, Ritter CA, et al. Loss of PTEN/MMAC1/TEP in EGF receptor-expressing tumor cells counteracts the antitumor action of EGFR tyrosine kinase inhibitors. Oncogene. 2003 May 8;22(18):2812–2822. doi: 10.1038/sj.onc.1206388. [DOI] [PubMed] [Google Scholar]
- 40.Ganti R, Skapek SX, Zhang J, et al. Expression and genomic status of EGFR and ErbB-2 in alveolar and embryonal rhabdomyosarcoma. Mod Pathol. 2006 May 26; doi: 10.1038/modpathol.3800636. [DOI] [PubMed] [Google Scholar]
- 41.Pallares J, Bussaglia E, Martinez-Guitarte JL, et al. Immunohistochemical analysis of PTEN in endometrial carcinoma: a tissue microarray study with a comparison of four commercial antibodies in correlation with molecular abnormalities. Mod Pathol. 2005 May;18(5):719–727. doi: 10.1038/modpathol.3800347. [DOI] [PubMed] [Google Scholar]
- 42.Matsuzaki H, Dong S, Loi H, et al. Genotyping over 100,000 SNPs on a pair of oligonucleotide arrays. Nat Methods. 2004 Nov;1(2):109–111. doi: 10.1038/nmeth718. [DOI] [PubMed] [Google Scholar]
- 43.Beroukhim R, Lin M, Park Y, et al. Inferring loss-of-heterozygosity from unpaired tumors using high-density oligonucleotide SNP arrays. PLoS Comput Biol. 2006 May;2(5):e41. doi: 10.1371/journal.pcbi.0020041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhao X, Li C, Paez JG, et al. An integrated view of copy number and allelic alterations in the cancer genome using single nucleotide polymorphism arrays. Cancer Res. 2004 May 1;64(9):3060–3071. doi: 10.1158/0008-5472.can-03-3308. [DOI] [PubMed] [Google Scholar]
- 45.Li C, Wong WH. Model-based analysis of oligonucleotide arrays: expression index computation and outlier detection. Proc Natl Acad Sci U S A. 2001 Jan 2;98(1):31–36. doi: 10.1073/pnas.011404098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Parmigiani G, Garrett ES, Irizarry R, Zeger SL. The analysis of gene expression data: methods and software. New York: Springer; 2003. DNA-Chip Analyzer (dChip) pp. 120–141. [Google Scholar]
- 47.Bjornsti MA, Houghton PJ. The TOR pathway: a target for cancer therapy. Nat Rev Cancer. 2004 May;4(5):335–348. doi: 10.1038/nrc1362. [DOI] [PubMed] [Google Scholar]
- 48.Fukaya Y, Ishiguro N, Senga T, et al. A role for PI3K-Akt signaling in pulmonary metastatic nodule formation of the osteosarcoma cell line, LM8. Oncol Rep. 2005 Oct;14(4):847–852. [PubMed] [Google Scholar]
- 49.She QB, Solit D, Basso A, Moasser MM. Resistance to gefitinib in PTEN-null HER-overexpressing tumor cells can be overcome through restoration of PTEN function or pharmacologic modulation of constitutive phosphatidylinositol 3′-kinase/Akt pathway signaling. Clin Cancer Res. 2003 Oct 1;9(12):4340–4346. [PubMed] [Google Scholar]
- 50.Wan X, Mendoza A, Khanna C, Helman LJ. Rapamycin inhibits ezrin-mediated metastatic behavior in a murine model of osteosarcoma. Cancer Res. 2005 Mar 15;65(6):2406–2411. doi: 10.1158/0008-5472.CAN-04-3135. [DOI] [PubMed] [Google Scholar]
- 51.Inoue R, Matsuki NA, Jing G, Kanematsu T, Abe K, Hirata M. The inhibitory effect of alendronate, a nitrogen-containing bisphosphonate on the PI3K-Akt-NFkappaB pathway in osteosarcoma cells. Br J Pharmacol. 2005 Nov;146(5):633–641. doi: 10.1038/sj.bjp.0706373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang MY, Lu KV, Zhu S, et al. Mammalian Target of Rapamycin Inhibition Promotes Response to Epidermal Growth Factor Receptor Kinase Inhibitors in PTEN-Deficient and PTEN-Intact Glioblastoma Cells. Cancer Res. 2006 Aug 15;66(16):7864–7869. doi: 10.1158/0008-5472.CAN-04-4392. [DOI] [PubMed] [Google Scholar]
- 53.Huvos AG, Rosen G, Marcove RC. Primary osteogenic sarcoma: pathologic aspects in 20 patients after treatment with chemotherapy en bloc resection, and prosthetic bone replacement. Arch Pathol Lab Med. 1977 Jan;101(1):14–18. [PubMed] [Google Scholar]
- 54.Rosen G, Marcove RC, Caparros B, Nirenberg A, Kosloff C, Huvos AG. Primary osteogenic sarcoma: the rationale for preoperative chemotherapy and delayed surgery. Cancer. 1979 Jun;43(6):2163–2177. doi: 10.1002/1097-0142(197906)43:6<2163::aid-cncr2820430602>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 55.Rosen G, Murphy ML, Huvos AG, Gutierrez M, Marcove RC. Chemotherapy, en bloc resection, and prosthetic bone replacement in the treatment of osteogenic sarcoma. Cancer. 1976 Jan;37(1):1–11. doi: 10.1002/1097-0142(197601)37:1<1::aid-cncr2820370102>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]