Abstract
Extracts of four plant portions (roots, stems, leaves and flowers) of Urtica dioica, (the stinging nettle) were prepared using accelerated solvent extraction (ASE) involving water, hexanes, methanol and dichloromethane. The extracts were evaluated for their anti-inflammatory and cytotoxic activity in an NF-κB luciferase and MTT assay using macrophage immune (RAW264.7) cells. A standardized commercial ethanol extract of nettle leaves were also evaluated. The methanolic extract of the flowering portions displayed significant anti-inflammatory activity on par with the standard anti-inflammatory agent celastrol (1) but was moderately cytotoxic. Alternatively, the polar extracts (water, methanol, ethanol) of the roots, stems and leaves plant portions displayed moderate to weak anti-inflammatory activity, while the methanol and especially the water soluble extracts exhibited noticeable cytotoxicity. In contrast, the lipophilic dichloromethane extracts of the roots, stems and leaves exhibited potent anti-inflammatory effects ≥ 1 with minimal cytotoxicity to RAW264.7 cells. Collectively these results suggest that using lipophilic extracts of the roots, stems or leaves of stinging nettle may be more effective then traditional tinctures (water, methanol, ethanol) to undergo clinical evaluations for the treatment of inflammatory disorders including arthritis. A chemical investigation into the lipophillic extracts of stinging nettle to identify the bioactive compound(s) responsible for their observed anti-inflammatory activity is further warranted.
Keywords: Urtica dioica (Stinging Nettle), Anti-inflammatory, Rheumatoid arthritis, Osteoarthritis, Nuclear factor-kB
Introduction
Herbal medicines have the potential to provide efficacious treatments for inflammatory disorders, particularly rheumatoid (RA) or osteoarthritis (OA) (Khanna et al., 2007, Soeken et al., 2003, Long et al., 2001). Historically, dozens of plants species have been repeatedly and independently selected by multiple cultures around the world for their analgesic/anti-inflammatory effects (Venkatesha et al., 2011a). Selected examples include Tripterygium wilfordii (thunder God vine), Camellia sinensis (green tea), Uncaria tomentosa (cat’s claw), and Zingiber offcinale (ginger). Extracts of these plants and or their characterized bioactive compound(s) demonstrate potent anti-inflammatory activity in vitro by inhibiting targets in the nuclear factor kappa B (NF-κB) pathway (Ahmed et al., 2005, Khanna et al., 2007, and Venkatesha et al., 2011a). Furthermore evaluations of each of these herbal medicines have shown positive anti-arthritic effects in vivo as well as clinically against RA or OA (Ahmed et al., 2005).
Another lesser known cosmopolitan ethnobotanical medicine is Urtica dioica, (the stinging nettle) used throughout North America and Europe for treating inflammatory disorders such as OA (Setty and Sigal 2005). Several preclinical studies demonstrating the anti-inflammatory therapeutic potential and proposed mechanism of action (MOA) of nettle have been reported (Chrubasik et al., 2007a), most notably its inhibition of myeloid dendritic cells (Broer and Behnke, 2002) as well as targets in the NF-kB pathway (Riehemann et al., 1999), however it’s precise MOA is not clear. Ten clinical trials evaluating its use for the treatment of OA have been conducted and generated mixed results in terms of their efficacy (Rayburn et al., 2009, Jacquet et al., 2009, Chrubasik et al., 2007a.) The root extracts of nettle have also been studied clinically for treatment of benign prostatic hyperplasia (BPH) (Chrubasik et al., 2007b). Also noteworthy is that the bioactive compound(s) responsible for these reported activities remains poorly understood.
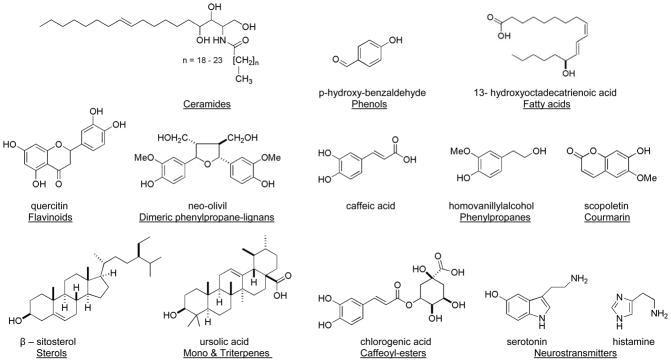
A number of structural classes have been reported from extracts of nettle and selected examples are shown in Fig. 1 (Cummings and Olsen, 2011; Chrubasik et al., 2007a, Ji et al., 2007). Surprisingly, definitive reports using bioassay guided fractionation to link any potent anti-inflammatory compound(s) derived from nettle for treating OA are lacking (Schulze-Tanzil et al., 2002). In addition several different extract preparations (e.g. water, ethanol, propanol,) have exhibited success clinically, making it difficult to pinpoint the overall active chemistry and further compare the efficacy of clinical studies side-by-side (Cameron et al., 2009; Randall et al., 2008; Chrubasik et al., 2007a). Traditionally water and ethanol soluble extracts of nettle have been used to investigate its anti-inflammatory effects. Unfortunately, standardized extracts of the more polar constituents have failed to agree with clinical efficacy (Chrubasik et al., 2007a). Interestingly, few investigators have looked closely into the lipophilic extracts of U. dioica despite previous reports of anti-inflammatory activity with potentially undefined chemistry (Chrubasik and Eisenberg, 1999).
Figure 1.
Selected examples of compounds and structural classes (underlined) reported from extracts of stinging nettle.
In a previous pilot clinical trial, we demonstrated that the topical use of a proprietary ethanol based extract of nettle for treatment of OA was both well tolerated and likely effective in reducing pain and improving function (Rayburn et al., 2009). Before initiating further OA evaluations, we realized that the MOA (Setty and Sigal 2005) and most effective formulation(s) of nettle (Chrubasik et al., 2007a) were not clearly defined for treating OA and that we could possibly benefit from conducting our own pre-clinical investigations to address these concerns. The purpose of the present study was to start by identifying the most potent extracts and ultimately the bioactive compound(s) derived from stinging nettle to faciliate evaluation of their anti-inflammatory and anti-arthritic mechanisms in order to lead to optimal formulations that would be the most effective for OA clinical evaluations. Based on the anti-inflammatory results reported for T. wilfordi, C. sinensis, U. tomentosa, and Z. officinale using models involving NF-κB (Ahmed et al., 2005), the fact that the NF-κB pathway has emerged as a potential anti-arthritic therapeutic target (Venkatesha et al., 2011a, Khanna et al., 2007), and the positive results observed for these species in treating RA and OA clinically (Ahmed et al., 2005), it seemed logical to begin our investigation of nettlein two parts. First by collecting fresh specimens of U. dioica and separating them into four major portions (roots, stems, leaves and flowers) for high-throughput accelerated solvent extraction (ASE) and second by evaluating these extracts for their anti-inflammatory and cytotoxic activities using a previously reported NF-κB luciferase and MTT assay (Johnson et al., 2012).
Materials and methods
Motivated by the positive results from our pilot clinical trial we began by obtaining wild stinging nettle specimens (~1 kg) of U. dioica from the Monterey Bay region of California (where the Mutsun Native American tribe has used the plant medicinally for centuries) and conducted an in vitro anti-inflammatory analysis of extracts from the roots (12.0 g), stems (11.2 g), leaves (15.6 g) and flowers (7.9 g). We employed a previously reported nuclear factor kappa B (NF-κB) luciferase assay (Wu et al., 2010) to measure the reduction of inflammation of transfected macrophage immune (RAW 264.7) cells that were stimulated by the endotoxin lipopolysaccharide (LPS). Cytotoxicity of these samples was also measured against macrophage (RAW 264.7) immune cells using a previously reported MTT assay (Wu et al., 2010). Extracts were prepared using our high-throughput extraction protocol involving accelerated solvent extraction (ASE) (Johnson et al., 2010) and included water (H2O), methanol (MeOH), dichloromethane (DCM) and hexanes. We further compared the nettle extracts with a commercial ethanol leaf extract, obtained from Gaia Herbs Inc., versus the standard anti-inflammatory agent celastrol (1) and the cytotoxin doxorubicin.
NF-kB luciferase assay
Extracts were tested in an NF-κB luciferase reporter assay in mouse macrophage (RAW264.7) immune cells to determine NF-κB activity. Stably transfected RAW264.7 cells with the NF-κB reporter gene were plated and evaluated. Following a 24 h recovery period, the cells were treated with the extract(s) for an additional 18 h in the presence of LPS (100 ng/ml). To check NF-κB -luciferase activity, the Luciferase Reporter Assay System purchased from Promega (Madison, WI) were used. Cell lysates (15 μl) from treated RAW264.7 cells were placed in opaque 96 well plates. Luciferase Assay Reagent (50 μl) was injected and read by a fluorometer (LMAX 2, Molecular devices). All extracts were evaluated in triplicate (n = 3) for their effects on RAW264.7 cells. The standard anti-inflammatory agent celastrol was obtained from Cayman Chemical Company, Ann Arbor, MI, USA
MTT Cytotoxicity assay
Extracts were tested at 10 μg/ml respectively using the MTT assay in murine macrophage (RAW264.7) immune cells. Cells in 96-well plates in the required growth medium were treated with extracts dissolved in DMSO for 20 h (RAW264.7). After incubation, MTT solution was added to wells and incubated for another 2 h. Media were removed and DMSO was added to dissolve purple precipitates. Then plates were read at 570 nm using a plate reader. All extracts were evaluated in triplicate (n = 3) for their effects on RAW264.7 cells. The standard cytotoxic agent doxorubicin was obtained from Cayman Chemical Company, Ann Arbor, MI, USA.
Results
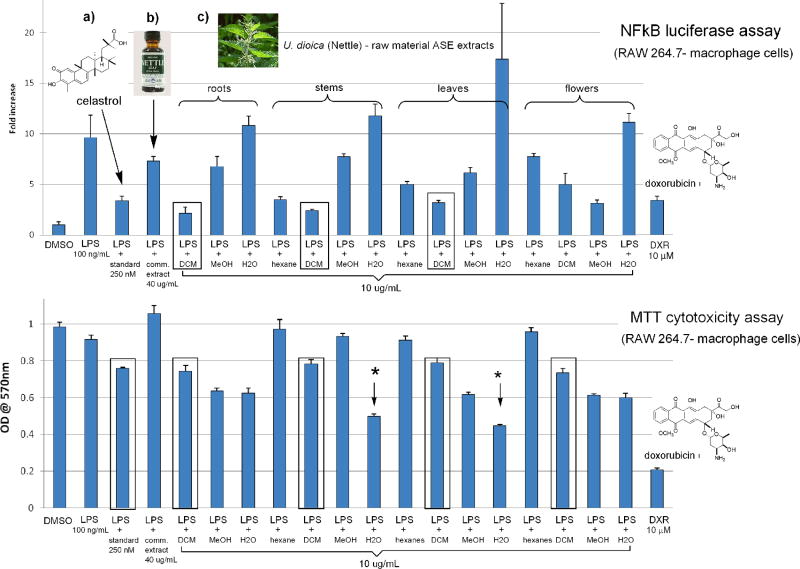
The anti-inflammatory and cytotoxic activity of four extracts from four major plant portions of U. dioica evaluated in the NF-κB luciferase assay (top) and MTT cytotoxicity assay (bottom) are shown in Fig. 2. These samples were screened alongside the standard compounds celastrol (1, 250 nM) and the known cytotoxin doxorubicin (10 μM). The yields of the water (H2O) extracts (in parenthesis) evaluated from the roots (192.7 mg), stems (248.1 mg), leaves (320.1 mg) and flowers (234.3 mg) displayed no anti-inflammatory effects up to 10 μg/ml in the NF-kB luciferase activity. In contrast, the stems and leaves H2O extracts were noticeably cytotoxic to macrophage (RAW264.7) immune cells in the MTT assay (asterisks), but were less toxic then doxorubicin. The methanol (MeOH) extracts of the roots (256.1 mg), stems (1,042.2 mg) and leaves (674.4 mg) were considerably less active than 1 in terms of their ability to reduce LPS induced activation of NF-κB and were on par with a standardized commercial ethanol leaf extract (40 μg/ml), and most were slightly cytotoxic. The MeOH extract (333.1 mg) of the flowering portion of U. dioica, effectively reduced LPS induced inflammation in the NF-κB assay but was also moderately cytotoxic in the MTT assay. Interestingly, the dichloromethane (DCM) extracts (boxed) of the roots (12.7 mg), stems (41.5 mg) and leaves (190.3 mg) were ≥ then 1 at reducing LPS induced inflammation in the NF-κB assay at 10 μg/ml. While yields of the hexanes extracts of the roots (1.0 mg), stems (2.9 mg), leaves (13.3 mg) and flowers (20.7 mg) also displayed anti-inflammatory activity but they were not as potent as the DCM extracts. Most importantly all of the lipophilic extracts exhibited minimal cytototoxicity to RAW264.7 cells in the MTT assay and were comparable to 1.
Figure 2.
Comparative bioassay data of: (a) standard NF-κB inhibitor (celastrol, 1); (b) a commercial ethanol leaf extract of U. dioica (stinging nettle) and (c) ASE extracts of U. dioica evaluated in the NF-κB luciferase and MTT cytotoxicity assays. The upper row depicts anti-inflammatory activity measured by the NF-κB assay. Samples with more potent anti-inflammatory activity are indicated by shorter graph bars. The bottom row gives the results of the MTT cytotoxicity assay. In this representation, shorter bars indicate greater cytotoxicity.
Discussion
An important aspect of this study involved using the NF-κB luciferase assay alongside the MTT cytotoxicity assay to identify selective non cytotoxic anti-inflammatory extracts. Many extracts or compounds can demonstrate potent anti-inflammatory effects by inhibiting LPS stimulated NF-κB activation but do so through a cytotoxic mechanism (e.g. doxorubicin), which serves to diminish their therapeutic potential. To the best of our knowledge, this is the first report to screen nettle extracts for both their anti-inflammatory and cytotoxic activity in macrophage (RAW 264.7) immune cells side by side. This heightens the impact of our discovery as the above results indicate the potential for in vivo and or clinical anti-inflammatory activity, without significant toxicity using lipophilic extracts, thereby improving the therapeutic index when administering nettle.
A recent clinical study showed that an unspecified dried extract of nettle combined with fish oil, vitamin E and zinc, in a proprietary product (Phytalgic®), decreased disease scores in patients with OA and reduces the use of analgesics or non steroidal anti-inflammatory drugs (Jacquet et al., 2009). The results of this study showed great promise, although some concerns were raised regarding detection bias and the way in which the overall study was conducted (Christensen et al., 2010). Regardless, if larger independent studies can repeat the incredible observed effect size of ~ 0.5 reported by Jacquet et al., 2009, OA therapy may be in for a dramatic change. Of considerable interest is that the nettle extract and fish oil used in this product were the only substances in which the chemistry was undefined in terms of its ascribed anti-arthritic activity. This leaves the reader to ponder whether the unreported compounds in nettle play a most crucial role in treating OA.
Some of the most encouraging results obtained overall from herbal medicines for the treatment of arthritis are from using T. wilfordii (Tao et al., 2002) which has spawned widespread investigation into the therapeutic potential of the extracts and or compounds prepared from this species. For example, a U.S. NIH-sponsored Phase 2b clinical trial using extracts of T. wilfordii for the treatment of RA showed significant improvement measured in the group receiving the plant extract (Goldbach-Mansky et al., 2009). Therapeutic lead compounds from T. wilfordi with proven anti-inflammatory activity that inhibit the NF-κB pathway include the lipophilic triterpene celastrol (1) and diterpenoid epoxide triptolide (2) (Fig. 3) (Lee et al., 2006; Ma et al., 2007). Compound 1 is currently being pursued as an important anti-arthritic therapeutic lead based on impressive results of both 1 and its parent extract demonstrating potent in vivo activity in the rat adjuvant-induced arthritis model of human RA (Venkatesha et al., 2011b). Compound 2 or its derivatives are in Phase I clinical trials for treating RA (Zhou et al., 2012). The fact that lipophilic extracts of nettle are on par with the anti-inflammatory standard 1 in our NF-κB and MTT assays in terms of their selective potency is promising and suggests that further exploration into the bioactive compound(s) within these extracts could uncover new therapeutic leads for treating OA.
Fig. 3.
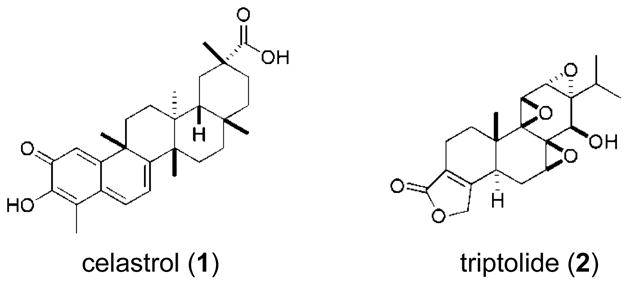
Chemical structures of celastrol (1) and triptolide (2)
Conclusion
Several noteworthy conclusions can be drawn from our investigation of the extracts of U. dioica (the stinging nettle). First, the polar water (H2O) extracts evaluated demonstrated no ability to reduce inflammation stimulated by LPS in our NFkB luciferase assay. In fact H2O extracts of the stems and leaves demonstrated noticeable cytotoxicity in the MTT assay. Second, most of the polar MeOH extracts of our specimens (including the roots, stems and leaves) were far less effective than the standard anti-inflammatory agent celastrol (1) and were comparable to a commercial ethanol extract of stinging nettle in terms of their activity. Third, in accordance with reports by others (Chrubasik et al., 2007), the MeOH extract of the flowering portion of nettle displayed significant anti-inflammatory activity however moderate cytotoxicity was also observed. Lastly and of particular note, the lipophilic DCM extracts of the roots, stems and leaves were equivalent to or more potent than the standard 1 at reducing LPS induced inflammation in the NFkB luciferase assay and were the least cytotoxic.
Further preclinical work is needed to elucidate the structure(s) of the active component(s) in the bioactive extract portions of nettle, and to determine optimal dosage and route of delivery. Subsequently, clinical trials can define a role for stinging nettle in the treatment of patients with OA and other inflammatory disorders. In summary, these findings suggest that using lipophilic extracts of the roots, stems or leaves of stinging nettle may serve as complimentary and or more effective formulations than traditional tinctures to undergo in vivo and or clinical evaluations for treating diseases involving inflammation, including OA. Ultimately ongoing investigations into the chemistry responsible for the anti-inflammatory activity observed in lipophillic nettle extracts merits further consideration.
Acknowledgments
This work was supported by grants from the NIH Fogarty International Center, International Cooperative Biodiversity Groups, award number 1U01TW008160-01 and Agricultural Food Research Initiative of the National Institute of Food and Agriculture, USDA, Grant #35621-04750. We are grateful to Professor Philip Crews (UC Santa Cruz) for allowing us to use his laboratory to conduct this research.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ahmed S, Anuntiyo J, Malemud CJ, Haqqi TM. Biological basis for the use of botanicals in osteoarthritis and rheumatoid arthritis. eCAM. 2005;2:301–308. doi: 10.1093/ecam/neh117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broer J, Behnke B. Immunosuppressant effect of IDS 30, a stinging nettle leaf extract, on myeloid dendritic cells in vitro. J Rheumatol. 2002;29:659–666. [PubMed] [Google Scholar]
- Cameron M, Gagnier JJ, Little CV, Parsons TJ, Blümle A, Chrubasik SA. Evidence of effectiveness of herbal medicinal products in the treatment of arthritis. Part 1. Phytother Res. 2009;23:1497–1515. doi: 10.1002/ptr.3007. [DOI] [PubMed] [Google Scholar]
- Christensen R, Bliddal H. Is Phytalgic® a goldmine for osteoarthritis patients or is there something fishy about this nutraceutical? A summary of findings and risk-of-bias assessment. Arthritis Res Ther. 2010;12:R105. doi: 10.1186/ar2909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chrubasik SA, Eisenberg E. Treatment of rheumatic pain with kampo medicine in Europe. Part II. Urtica dioica. Pain Clin. 1999;11:179–185. [Google Scholar]
- Chrubasik JE, Roufogalis BD, Wagner H, Chrubasik SA. A comprehensive review on nettle effect and efficacy profiles, Part I: Herba urticae. Phytomed. 2007a;14:423–435. doi: 10.1016/j.phymed.2007.03.004. [DOI] [PubMed] [Google Scholar]
- Chrubasik JE, Roufogalis BD, Wagner H, Chrubasik SA. A comprehensive review on the stinging nettle effect and efficacy profiles. Part II: Urticae radix. Phytomed. 2007b;14:568–579. doi: 10.1016/j.phymed.2007.03.014. [DOI] [PubMed] [Google Scholar]
- Cummings AJ, Olsen M. Mechanism of action of stinging nettles. Wilderness Environ Med. 2011;22:136–139. doi: 10.1016/j.wem.2011.01.001. [DOI] [PubMed] [Google Scholar]
- Goldman-Mansky R, Wilson M, Fleischmann R, Olsen N, Silverfield J, Kempf P, Kivitz A, Sherrer Y, Pucino F, Csako G, Costello R, Pham TH, Synder C, Heijde D, Tao X, Wesley R, Lipsky PE. Comparison of Tripterygium wilfordii Hook F versus sulfasalazine in the treatment of rheumatoid arthritis: A randomized trial. Ann Intern Med. 2009;51:229–240. doi: 10.7326/0003-4819-151-4-200908180-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacquet A, Girodet P, Pariente A, Forest K, Mallet L, Moore N. Phytalgic®, a food supplement, vs placebo in patients with osteoarthritis of the knee or hip: A randomized double-blind placebo-controlled clinical trial. Arthritis Res Ther. 2009;11:R192. doi: 10.1186/ar2891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji T, Liu C, Wang A, Yang J, Su Y, Yuan L, Feng X. Chemical constituents of Urtica dioicaL grown in Tibet autonomous region Zhong yao cai = J Chinese Medicinal Materials. 2007;30:662–664. [PubMed] [Google Scholar]
- Johnson TA, Morgan MVC, Aratow NA, Estee SA, Sashidhara KV, Loveridge ST, Segraves NL, Crews P. Assessing pressurized liquid extraction for the high-throughput extraction of marine-sponge-derived natural products. J Nat Prod. 2010;3:359–364. doi: 10.1021/np900565a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson TA, Sohn J, Vaske YM, White KN, Cohen TL, Vervoort HC, Tenney K, Valeriote FA, Bjeldanes LF, Crews P. Myxobacteria versus sponge-derived alkaloids: The bengamide family identified as potent immune modulating agents by scrutiny of LC–MS/ELSD libraries. Bioorg Med Chem. 2012;20:4348–4355. doi: 10.1016/j.bmc.2012.05.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khanna D, Sethi G, Ahn KS, Pandey MK, Kunnamakkara AB, Sung B, Aggarwal A, Aggarwal BB. Natural Products as a gold mine for arthritis treatment. Curr Opin Pharmacol. 2007;7:344–351. doi: 10.1016/j.coph.2007.03.002. [DOI] [PubMed] [Google Scholar]
- Lee J, Koo TH, Yoon H, Jung HS, Jin HZ, Lee K, Hong Y, Lee JJ. Inhibition of NF-κB activation through targeting IκB kinase by celastrol, a quinone methide triterpenoid. Biochem Pharmacol. 2006;72:1311–1321. doi: 10.1016/j.bcp.2006.08.014. [DOI] [PubMed] [Google Scholar]
- Long L, Soeken K, Ernst E. Herbal medicines for the treatment of osteoarthritis: a systematic review. Rheumatology. 2001;40:779–793. doi: 10.1093/rheumatology/40.7.779. [DOI] [PubMed] [Google Scholar]
- Ma J, Dey M, Yang H, Poulev A, Pouleva R, Dorn R, Lipsky RE, Kennelly EJ, Raskin I. Anti-inflammatory and immunosuppressive compounds from Tripterygium wilfordii. Phytochemistry. 2007;68:1172–1178. doi: 10.1016/j.phytochem.2007.02.021. [DOI] [PubMed] [Google Scholar]
- Randall C, Dickens A, White A, Sanders H, Fox M, Cambell J. Nettle sting for chronic knee pain: a randomized controlled pilot study. Complement Ther Medicine. 2008;16:66–72. doi: 10.1016/j.ctim.2007.01.012. [DOI] [PubMed] [Google Scholar]
- Rayburn K, Fleischbein E, Song J, Allen B, Kundert M, Leiter C, Bush T. Stinging nettle cream for osteoarthritis. Altern Ther Health Med. 2009;15(4):60–61. [PubMed] [Google Scholar]
- Riehemann K, Behnke B, Schulze-Osthoff K. Plant extracts from stinging nettle (Urtica dioica), an anti-rheumatic remedy, inhibit the proinflammatory transcription factor NF-κB. FEBS lett. 1999;442:89–94. doi: 10.1016/s0014-5793(98)01622-6. [DOI] [PubMed] [Google Scholar]
- Schulze-Tanzil G, De Souza P, Behnke B, Klingelhoefer S, Scheid A, Shakibaei M. Effects of the antirheumatic remedy Hox alpha - a new stinging nettle leaf extract - on matrix metalloproteinases in human chondrocytes in vitro. Histol Histopathol. 2002;17:477–485. doi: 10.14670/HH-17.477. [DOI] [PubMed] [Google Scholar]
- Setty AR, Sigal LH. Herbal medications: commonly used in the practice of rheumatology: mechanisms of action, efficacy, and side effects. Semin Arthritis Rheu. 2005;34:773–784. doi: 10.1016/j.semarthrit.2005.01.011. [DOI] [PubMed] [Google Scholar]
- Soeken KL, Miller SA, Ernst E. Herbal medicines for the treatment of rheumatoid arthritis: a systematic review. Rheumatology. 2003;42:652–659. doi: 10.1093/rheumatology/keg183. [DOI] [PubMed] [Google Scholar]
- Tao X, Younger J, Fan FZ, Wang B, Lipsky PE. Benefit of an extract of Tripterygium wilfordii hook F in patients with rheumatoid arthritis. Arthritis Rheum. 2002;7:1735–1743. doi: 10.1002/art.10411. [DOI] [PubMed] [Google Scholar]
- Venkatesha SH, Berman BM, Moudgil KD. Herbal medicinal products target defined biochemical and molecular mediators of inflammatory autoimmune arthritis. Bioorg Med Chem. 2011a;19:21–29. doi: 10.1016/j.bmc.2010.10.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatesha SH, Yu H, Rajaiah R, Tong L, Moudgil KD. Celastrus-derived celastrol suppresses autoimmune arthritis by modulating antigen-induced cellular and humoral effector responses. J Biol Chem. 2011b;17:15138–15146. doi: 10.1074/jbc.M111.226365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z, Yang Y, Ding J, Li Y, Miao Z. Triptolide: structural modifications, structure-activity relationships, bioactivities, clinical development and mechanisms. Nat Prod Rep. 2012;29:457–475. doi: 10.1039/c2np00088a. [DOI] [PubMed] [Google Scholar]